Do Soil Warming and Changes in Precipitation Patterns Affect Seed Yield and Seed Quality of Field-Grown Winter Oilseed Rape?
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Setup
2.3. Plant Cultivation, Crop Development Measurement, Biomass Harvests and Seed Quality Analyses
2.4. Statistical Analyses
3. Results
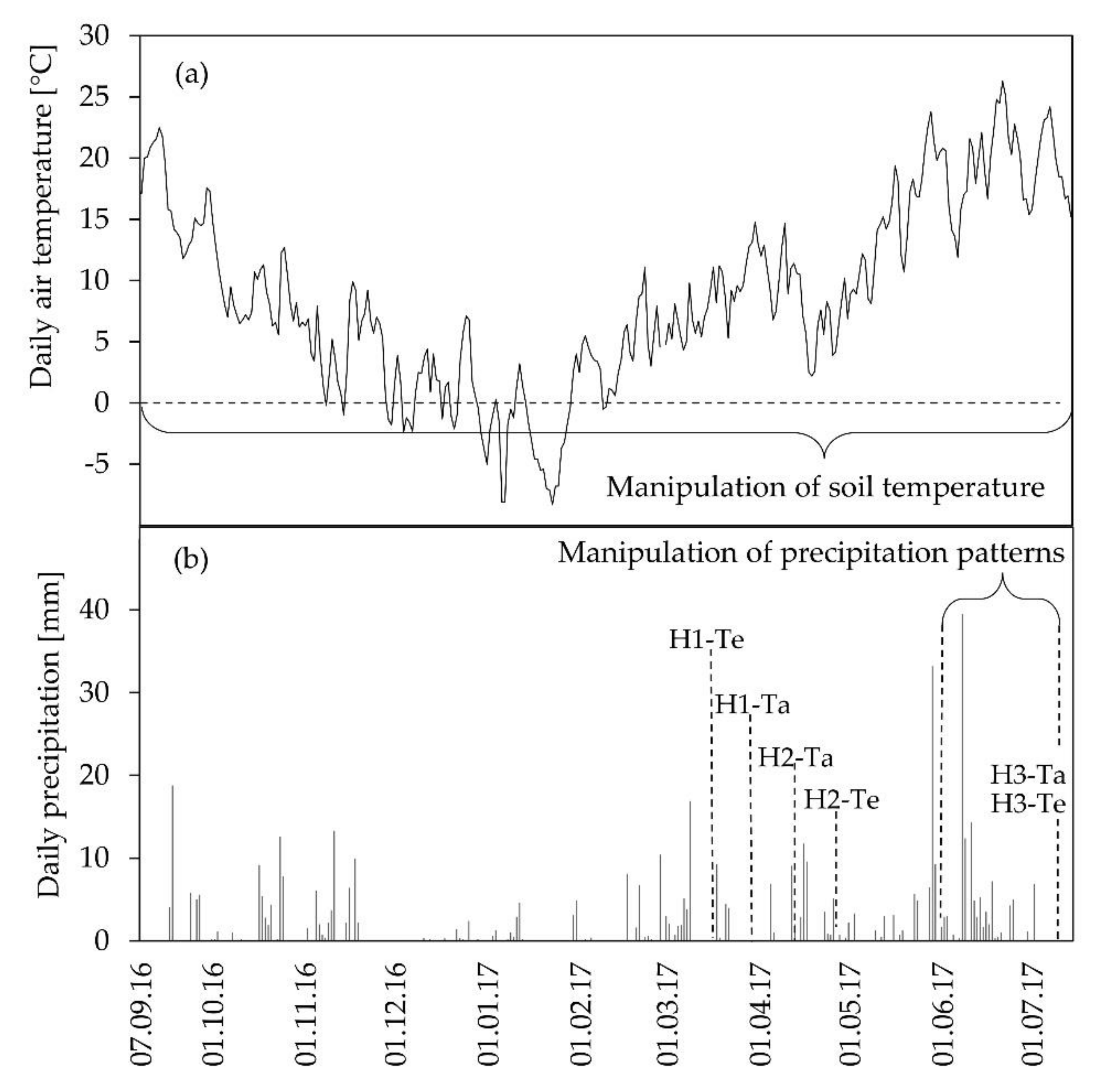
3.1. Environmental Conditions and Biomass Harvests
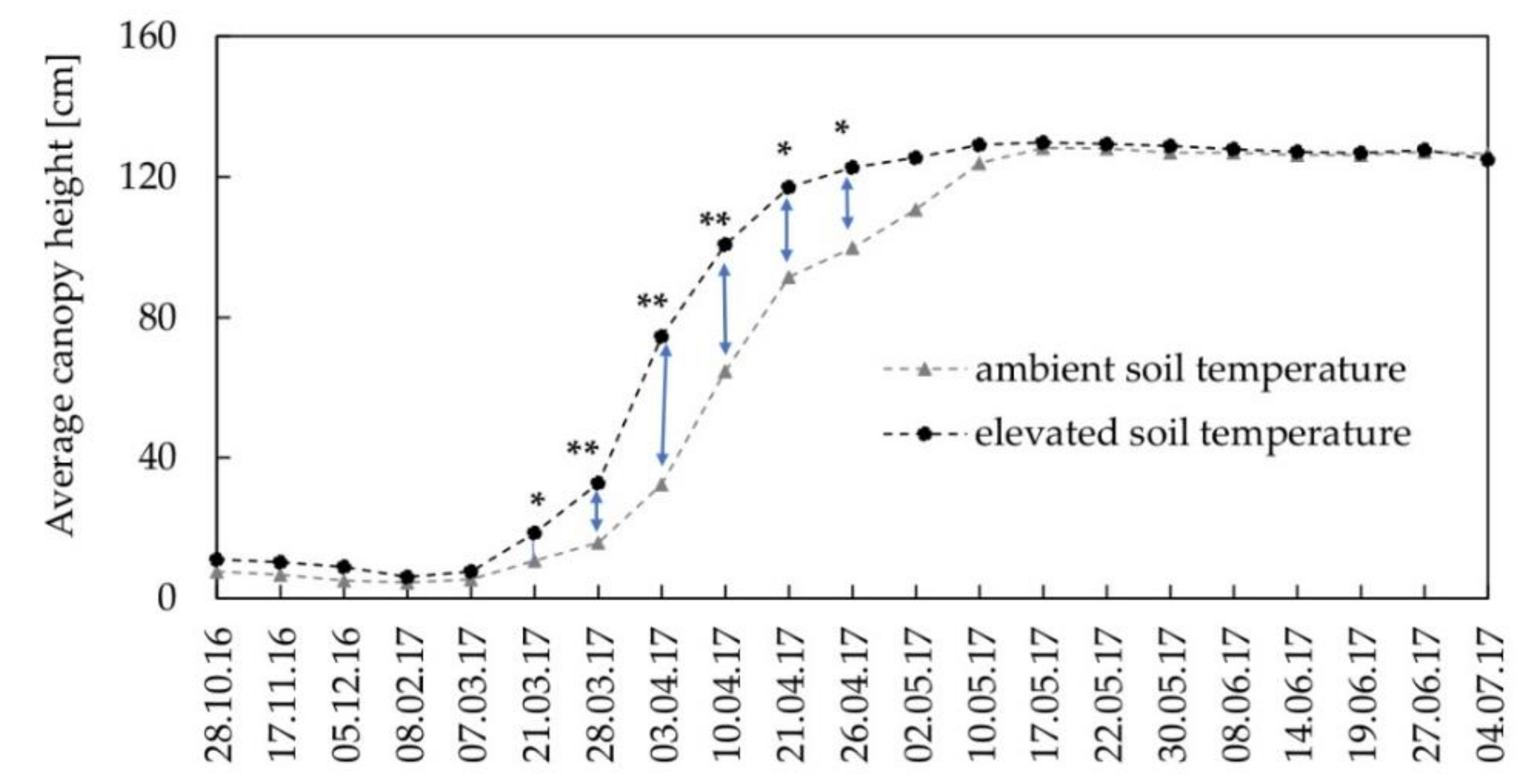
3.2. Crop Development
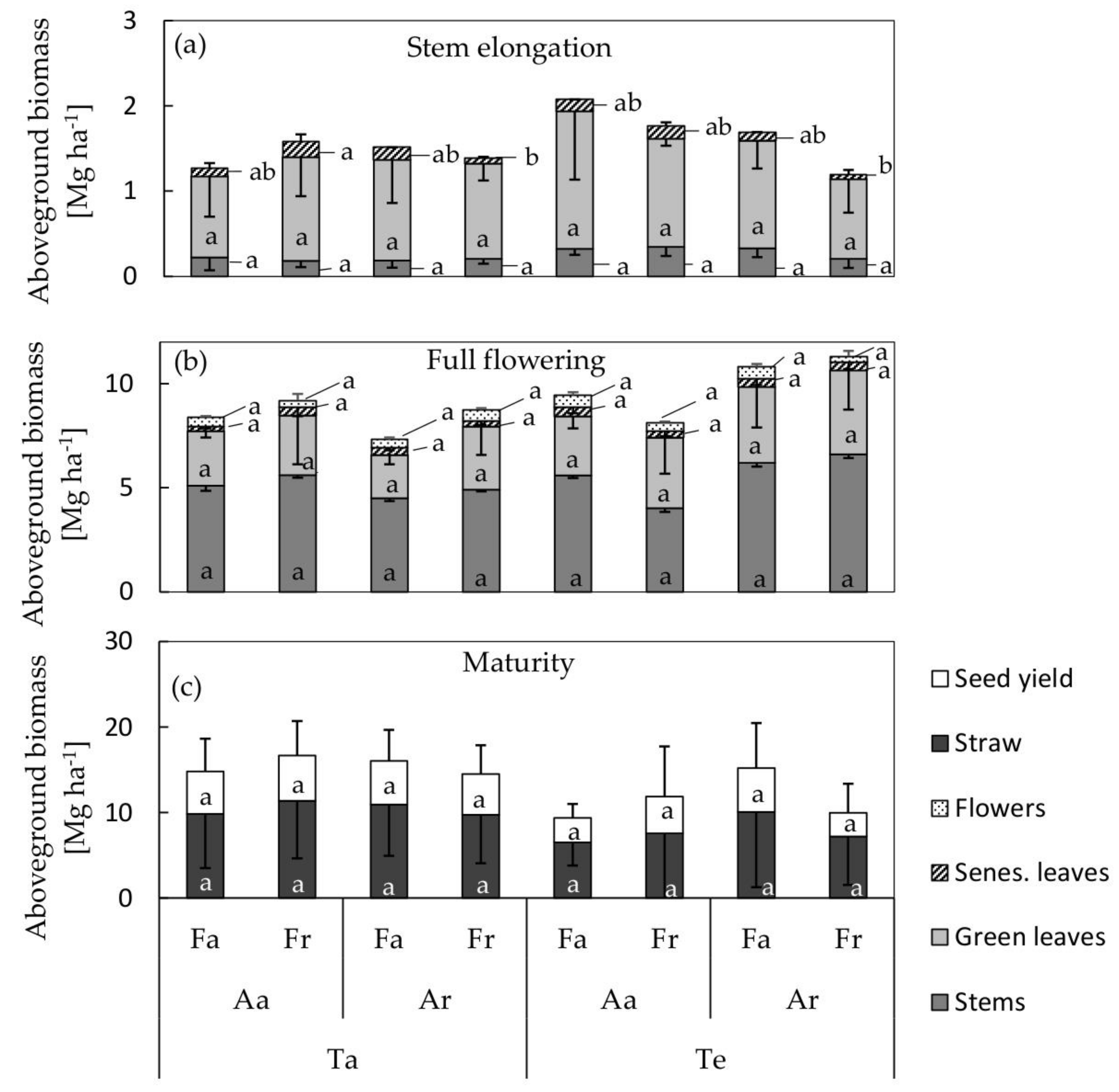
3.3. Biomass Allocation and Seed Yield
3.4. Seed Quality
4. Discussion
4.1. Crop Development and Crop Yield Parameters
4.2. Seed Quality
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DaMatta, F.M.; Grandis, A.; Arenque, B.C.; Buckeridge, M.S. Impacts of climate changes on crop physiology and food quality. Food Res. Int. 2010, 43, 1814–1823. [Google Scholar] [CrossRef]
- DWD. Nationaler Klimareport. Klima—Gestern, Heute und in der Zukunft, 3rd ed.; DWD: Offenbach am Main, Germany, 2017; pp. 1–46. [Google Scholar]
- Zheng, D.; Hunt, E.R., Jr.; Running, S.W. A daily soil temperature model based on air temperature and precipitation for continental applications. Clim. Res. 1993, 2, 183–191. [Google Scholar] [CrossRef]
- Patil, R.H.; Laegdsmand, M.; Olesen, J.E.; Porter, J.R. Growth and yield response of winter wheat to soil warming and rainfall patterns. J. Agric. Sci. 2010, 148, 553–566. [Google Scholar] [CrossRef]
- Bowen, G.D. Soil temperature, root growth and plant function. In Plant Roots: The Hidden Half, 1st ed.; Waisel, Y., Eshel, A., Kafkaki, U., Eds.; Plenum Press: New York, NY, USA, 1991; pp. 309–330. [Google Scholar]
- Gavito, M.E.; Curtis, P.S.; Mikkelsen, T.N.; Jakobsen, I. Interactive effects of soil temperature, atmospheric carbon dioxide and soil N on root development, biomass and nutrient uptake of winter wheat during vegetative growth. J. Exp. Bot. 2001, 52, 1913–1923. [Google Scholar] [CrossRef]
- Istanbulluoglu, A.; Arslan, B.; Gocmen, E.; Gezer, E.; Pasa, C. Effects of deficit irrigation regimes on the yield and growth of oilseed rape (Brassica napus L.). Biosyst. Eng. 2010, 105, 388–394. [Google Scholar] [CrossRef]
- Bodner, G.; Nakhforoosh, A.; Kaul, H.-P. Management of crop water under drought: A review. Agron. Sustain. Dev. 2015, 35, 401–442. [Google Scholar] [CrossRef]
- Barnabás, B.; Jäger, K.; Fehér, A. The effect of drought and heat stress on reproductive processes in cereals. Plant Cell Environ. 2008, 31, 11–38. [Google Scholar] [CrossRef]
- Walker, K.C.; Booth, E.J. Agricultural aspects of rape and other Brassica products. Eur. J. Lipid Sci. Technol. 2001, 103, 441–446. [Google Scholar] [CrossRef]
- Weymann, W.; Böttcher, U.; Sieling, K.; Kage, H. Effects of weather conditions during different growth phases on yield formation of winter oilseed rape. Field Crops Res. 2015, 173, 41–48. [Google Scholar] [CrossRef]
- Brown, J.K.M.; Beeby, R.; Penfield, S. Yield instability of winter oilseed rape modulated by early winter temperature. Sci. Rep. 2019, 9, 6953. [Google Scholar] [CrossRef]
- Champolivier, L.; Merrien, A. Effects of water stress applied at different growth stages to Brassica napus L. var. oleifera on yield, yield components and seed quality. Eur. J. Agron. 1996, 5, 153–160. [Google Scholar] [CrossRef]
- Bouchereau, A.; Clossais-Besnard, N.; Bensaoud, A.; Leport, L.; Renard, M. Water stress effects on rapeseed quality. Eur. J. Agron. 1996, 5, 19–30. [Google Scholar] [CrossRef]
- Piper, E.L.; Boote, K.I. Temperature and cultivar effects on soybean seed oil and protein concentrations. J. Am. Oil Chem. Soc. 1999, 76, 1233–1241. [Google Scholar] [CrossRef]
- Pipolo, A.E.; Sinclair, T.R.; Camara, G.M.S. Effects of temperature on oil and protein concentration in soybean seeds cultured in vitro. Ann. Appl. Biol. 2004, 144, 71–76. [Google Scholar] [CrossRef]
- Jensen, C.R.; Mogensen, V.O.; Mortensen, G.; Fieldsend, J.K.; Milford, G.F.J.; Andersen, M.N.; Thage, J.H. Seed glucosinolate, oil and protein contents of field-grown rape (Brassica napus L.) affected by soil drying and evaporative demand. Field Crops Res. 1996, 47, 93–105. [Google Scholar] [CrossRef]
- Alexander, J.; Auðunsson, G.A.; Benford, D.; Cockburn, A.; Cravedi, J.-P.; Dogliotti, E.; Di Domenico, A.; Férnandez-Cruz, M.L.; Fürst, P.; Fink-Gremmels, J.; et al. Glucosinolates as undesirable substances in animal feed.: Scientific Panel on Contaminants in the Food Chain. EFSA J. 2008, 1–76. [Google Scholar]
- Deutscher Wetterdienst. Vieljährige Mittelwerte: Tabelle B: Mittelwerte für den Bezugsstandort am Ende der Referenzperiode 1961–1990. Available online: https://www.dwd.de/DE/leistungen/klimadatendeutschland/vielj_mittelwerte.html (accessed on 3 February 2020).
- Agricultural Technology Centre (LTZ) Augustenberg. Station Hohenheim. Available online: http://www.wetter-bw.de/ (accessed on 3 February 2020).
- Poll, C.; Marhan, S.; Back, F.; Niklaus, P.A.; Kandeler, E. Field-scale manipulation of soil temperature and precipitation change soil CO2 flux in a temperate agricultural ecosystem. Agr. Ecosyst. Environ. 2013, 165, 88–97. [Google Scholar] [CrossRef]
- Meier, U. Growth Stages of Mono- and Dicotyledonous Plants. BBCH Monograph, 2nd ed.; Blackwell: Oxford, UK, 2001; pp. 29–34. [Google Scholar]
- Umweltbundesamt. Künftige Klimaänderungen in Deutschland—Regionale Projektionen für das 21. Jahrhundert; Umweltbundesamt: Dessau, Germany, 2006; pp. 1–7. [Google Scholar]
- Högy, P.; Poll, C.; Marhan, S.; Kandeler, E.; Fangmeier, A. Impacts of temperature increase and change in precipitation pattern on crop yield and yield quality of barley. Food Chem. 2013, 136, 1470–1477. [Google Scholar] [CrossRef]
- European Commission. Regulation (EC) No 152/2009 III C. Determination of the content of crude protein. Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Off. J. Eur. Union 2009, 152, 54/15–54/19.
- European Commission. Regulation (EC) No 152/2009 III H. Determination of crude oils and fats. Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Off. J. Eur. Union 2009, 152, 54/37–54/39.
- European Commission. Regulation (EC) No 152/2009 III F. Determination of amino acids (except tryptophan). Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Off. J. Eur. Union 2009, 152, 54/23–54/32.
- European Commission. Regulation (EC) No 152/2009 III G. Determination of Tryptophan. Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Off. J. Eur. Union 2009, 152, 54/32–54/37.
- DIN EN ISO 12966-1:2015-03. Animal and Vegetable Fats and Oils—Gas Chromatography of Fatty Acid Methyl Esters—Part 1: Guidlines on Modern Gas Chromatography of Fatty Acid Methyl Esters (ISO 12966-1:2014); Beuth: Berlin, Germany, 2015. [Google Scholar]
- Högy, P.; Franzaring, J.; Schwadorf, K.; Breuer, J.; Schütze, W.; Fangmeier, A. Effects of free-air CO2 enrichment on energy traits and seed quality of oilseed rape. Agr. Ecosyst. Environ. 2010, 139, 239–244. [Google Scholar] [CrossRef]
- DIN EN ISO 9167-1: 2013-12. Rapssamen—Bestimmung des Glucosinolatgehaltes—Teil 1: HPLC-Verfahren; Beuth: Berlin, Germany, 2013. [Google Scholar]
- Böttcher, C.; Krähmer, A.; Stürtz, M.; Widder, S.; Schulz, H. Comprehensive metabolite profiling of onion bulbs (Allium cepa) using liquid chromatography coupled with electrospray ionization quadrupole time-of-flight mass spectrometry. Metabolomics 2017, 13, 10893. [Google Scholar] [CrossRef]
- Kozak, M.; Piepho, H.-P. What’s normal anyway? Residual plots are more telling than significance tests when checking ANOVA assumptions. J. Agron. Crop Sci. 2018, 204, 86–98. [Google Scholar] [CrossRef]
- Bamminger, C.; Poll, C.; Sixt, C.; Högy, P.; Wüst, D.; Kandeler, E.; Marhan, S. Short-term response of soil microorganisms to biochar addition in a temperate agroecosystem under soil warming. Agr. Ecosyst. Environ. 2016, 233, 308–317. [Google Scholar] [CrossRef]
- Singh, V.K.; Shukla, A.K.; Singh, A.K. Impact of climate change on plant-microbe interactions under agroecosystems. In Climate Change and Agricultural Ecosystems: Current Challenges and Adaptation; Choudhary, K.K., Kumar, A., Singh, A.K., Eds.; Woodhead Publishing: Duxford, UK; Cambridge, UK; Kidlington, UK, 2019. [Google Scholar]
- Statistisches Landesamt Baden-Württemberg. Ernte der Hauptfeldfrüchte in Baden-Württemberg 2017: Artikel-Nr. 3354 17001, C II 1-j/17 (3). In Statistische Berichte Baden-Württemberg; Statistisches Landesamt Baden-Württemberg, Ed.; Statistisches Landesamt Baden-Württemberg: Stuttgart, Germany, 2017; pp. 1–7. [Google Scholar]
- Stone, P.J.; Sorensen, I.B.; Jamieson, P.D. Effect of soil temperature on phenology, canopy development, biomass and yield of maize in a cool-temperate climate. Field Crops Res. 1999, 63, 169–178. [Google Scholar] [CrossRef]
- Yadav, S.S.; Hegde, V.S.; Habibi, A.B.; Dia, M.; Verma, S. Climate change, agriculture and food security. In Food Security and Climate Change; Yadav, S.S., Redden, R.J., Hatfield, J.L., Ebert, A.W., Hunter, D., Eds.; John Wiley & Sons Ltd: Hoboken, NJ, USA, 2019. [Google Scholar]
- Hatfield, J.L. Combined impacts of carbon, temperature, and drought to sustain food production. In Food Security and Climate Change; Yadav, S.S., Redden, R.J., Hatfield, J.L., Ebert, A.W., Hunter, D., Eds.; John Wiley & Sons Ltd: Hoboken, NJ, USA, 2019. [Google Scholar]
- Aamir, M.; Rai, K.K.; Dubey, M.K.; Zehra, A.; Tripathi, Y.N.; Divyanshu, K.; Samal, S.; Upadhyay, R.S. Impact of climate change on soil carbon exchange, ecosystem dynamics, and plant-microbe interactions. In Climate Change and Agricultural Ecosystems: Current Challenges and Adaptation; Choudhary, K.K., Kumar, A., Singh, A.K., Eds.; Woodhead Publishing: Duxford, UK; Cambridge, UK; Kidlington, UK, 2019. [Google Scholar]
- Kannojia, P.; Sharma, P.K.; Sharma, K. Climate change and soil. In Climate Change and Agricultural Ecosystems: Current Challenges and Adaptation; Choudhary, K.K., Kumar, A., Singh, A.K., Eds.; Woodhead Publishing: Duxford, UK; Cambridge, UK; Kidlington, UK, 2019. [Google Scholar]
- Savin, R.; Stone, P.J.; Nicolas, M.E.; Wardlaw, I.F. Grain growth and malting quality of barley. 1. Effects of heat stress and moderately high temperature. Aust. J. Agric. Res. 1997, 48, 615–624. [Google Scholar] [CrossRef]
- Faraji, A.; Latifi, N.; Soltani, A.; Rad, A.H.S. Seed yield and water use efficiency of canola (Brassica napus L.) as affected by high temperature stress and supplemental irrigation. Agric. Water Manag. 2009, 96, 132–140. [Google Scholar] [CrossRef]
- Kutcher, H.R.; Warland, J.S.; Brandt, S.A. Temperature and precipitation effects on canola yields in Saskatchewan, Canada. Agric. For. Meteorol. 2010, 150, 161–165. [Google Scholar] [CrossRef]
- Alemayehu, F.R.; Frenck, G.; van der Linden, L.; Mikkelsen, T.N.; Jørgensen, R.B. Can barley (Hordeum vulgare L. s.l.) adapt to fast climate changes?: A controlled selection experiment. Genet. Resour. Crop Evol. 2014, 61, 151–161. [Google Scholar] [CrossRef]
- Ingvordsen, C.H.; Lyngkjær, M.F.; Peltonen-Sainio, P.; Mikkelsen, T.N.; Stockmarr, A.; Jørgensen, R.B. How a 10-day heatwave impacts barley grain yield when superimposed onto future levels of temperature and CO2 as single and combined factors. Agric. Ecosyst. Environ. 2018, 259, 45–52. [Google Scholar] [CrossRef]
- Singer, S.D.; Zou, J.; Weselake, R.J. Abiotic factors influence plant storage lipid accumulation and composition. Plant Sci. 2016, 243, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hlavinka, P.; Trnka, M.; Semerádová, D.; Dubrovský, M.; Žalud, Z.; Možný, M. Effect of drought on yield variability of key crops in Czech Republic. Agric. For. Meteorol. 2009, 149, 431–442. [Google Scholar] [CrossRef]
- Morison, J.I.L.; Lawlor, D.W. Interactions between increasing CO2 concentration and temperature on plant growth. Plant Cell Environ. 1999, 22, 659–682. [Google Scholar] [CrossRef]
- Sowerby, A.; Emmet, B.A.; Tietema, A.; Beier, C. Contrasting effects of repeated summer drought on soil carbon efflux in hydric and mesic heathland soils. Glob. Chang. Biol. 2008, 14, 2388–2404. [Google Scholar] [CrossRef]
- Beier, C.; Beierkuhnlein, C.; Wohlgemuth, T.; Penuelas, J.; Emmett, B.; Körner, C.; Boeck, H.d.; Hesselbjerg Christensen, J.; Leuzinger, S.; Janssens, I.A.; et al. Precipitation manipulation experiments-challenges and recommendations for the future. Ecol. Lett. 2012, 15, 899–911. [Google Scholar] [CrossRef]
- Mailer, R.J.; Pratley, J.E. Field studies of moisture availability effects on glucosinolate and oil concentration in the seed of rape, (Brassica napus L.) and turnip rape (Brassica rapa L. var. silvestris (Lam.) Briggs). Can. J. Plant Sci. 1990, 70, 399–407. [Google Scholar] [CrossRef]
- Good, A.G.; Zaplachinski, S.T. The effects of drought stress on free amino acid accumulation and protein synthesis in Brassica napus. Physiol Plant 1994, 90, 9–14. [Google Scholar] [CrossRef]
- Dyer, J.M.; Stymne, S.; Green, A.G.; Carlsson, A.S. High-value oils from plants. Plant J. 2008, 54, 640–655. [Google Scholar] [CrossRef]
- Del Carmen Martínez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The physiological importance of glucosinolates on plant response to abiotic stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef] [PubMed]
- Wittstock, U.; Burow, M. Glucosinolate breakdown in Arabidopsis: Mechanism, regulation and biological significance. Arabidopsis Book 2010, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- De March, G.; McGregor, D.I.; Séguin-Shwartz, G. Glucosinolate content of maturing pods and seeds of high and low glucosinolate summer rape. Can. J. Plant Sci. 1989, 69, 929–932. [Google Scholar] [CrossRef]
- Pritchard, F.M.; Eagles, H.A.; Norton, R.M.; Salisbury, P.A.; Nicolas, M. Environmental effects on seed composition of Victorian canola. Aust. J. Exp. Agric. 2000, 40, 679–685. [Google Scholar] [CrossRef]


| Treatment Number | Roof | Treatment Description | Treatment Short Form |
|---|---|---|---|
| 1 | Yes | AMB soil temp. × AMB prec. amount × AMB prec. frequency | TaAaFa |
| 2 | Yes | AMB soil temp. × AMB prec. amount × RED prec. frequency | TaAaFr |
| 3 | Yes | AMB soil temp. × RED prec. amount × AMB prec. frequency | TaArFa |
| 4 | Yes | AMB soil temp. × RED prec. amount × RED prec. frequency | TaArFr |
| 5 | Yes | ELE soil temp. × AMB prec. amount × AMB prec. frequency | TeAaFa |
| 6 | Yes | ELE soil temp. × AMB prec. amount × RED prec. frequency | TeAaFr |
| 7 | Yes | ELE soil temp. × RED prec. amount × AMB prec. frequency | TeArFa |
| 8 | Yes | ELE soil temp. × RED prec. amount × RED prec. frequency | TaArFr |
| 9 | No | AMB soil temp. × AMB prec. amount × AMB prec. frequency | TaAaFa |
| 10 | No | ELE soil temp. × AMB prec. amount × AMB prec. frequency | TeAaFa |
| Harvests | Development Stage | Harvest Date Ambient Soil Temperature | Harvest Date Elevated Soil Temperature |
|---|---|---|---|
| Harvest 1 | DC 31, stem elongation | 27 March 2017 | 15 March 2017 |
| Harvest 2 | DC 65, full flowering | 24 April 2017 | 10 April 2017 |
| Harvest 3 | DC 99, maturity | 11 July 2017 | 11 July 2017 |
| Response Variable | T | A | F | T × A | T × F | A × F | T × A × F |
|---|---|---|---|---|---|---|---|
| Amino acid concentration [% protein] | |||||||
| Essential [% protein] | |||||||
| Isoleucine | ns | ns | 0.040 | 0.048 | ns | ns | ns |
| Phenylalanine | ns | 0.041 | ns | 0.086 | ns | ns | ns |
| Lysine | ns | ns | ns | 0.015 | ns | ns | 0.015 |
| Non-essential [% protein] | |||||||
| Asparagine/aspartic acid | ns | ns | 0.077 | ns | ns | ns | ns |
| Glutamine/glutamic acid | 0.086 | ns | 0.070 | ns | ns | ns | ns |
| Proline | ns | 0.082 | ns | ns | ns | ns | ns |
| Alanine | ns | ns | ns | ns | 0.090 | ns | ns |
| Fatty acids concentration [% oil] | |||||||
| Saturated fatty acids [% oil] | |||||||
| Capric acid | ns | ns | ns | 0.023 | ns | ns | ns |
| Myristic acid | ns | ns | ns | ns | ns | ns | 0.047 |
| Lignoceric acid | ns | ns | ns | ns | ns | 0.048 | ns |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drebenstedt, I.; Hart, L.; Poll, C.; Marhan, S.; Kandeler, E.; Böttcher, C.; Meiners, T.; Hartung, J.; Högy, P. Do Soil Warming and Changes in Precipitation Patterns Affect Seed Yield and Seed Quality of Field-Grown Winter Oilseed Rape? Agronomy 2020, 10, 520. https://doi.org/10.3390/agronomy10040520
Drebenstedt I, Hart L, Poll C, Marhan S, Kandeler E, Böttcher C, Meiners T, Hartung J, Högy P. Do Soil Warming and Changes in Precipitation Patterns Affect Seed Yield and Seed Quality of Field-Grown Winter Oilseed Rape? Agronomy. 2020; 10(4):520. https://doi.org/10.3390/agronomy10040520
Chicago/Turabian StyleDrebenstedt, Ireen, Leonie Hart, Christian Poll, Sven Marhan, Ellen Kandeler, Christoph Böttcher, Torsten Meiners, Jens Hartung, and Petra Högy. 2020. "Do Soil Warming and Changes in Precipitation Patterns Affect Seed Yield and Seed Quality of Field-Grown Winter Oilseed Rape?" Agronomy 10, no. 4: 520. https://doi.org/10.3390/agronomy10040520
APA StyleDrebenstedt, I., Hart, L., Poll, C., Marhan, S., Kandeler, E., Böttcher, C., Meiners, T., Hartung, J., & Högy, P. (2020). Do Soil Warming and Changes in Precipitation Patterns Affect Seed Yield and Seed Quality of Field-Grown Winter Oilseed Rape? Agronomy, 10(4), 520. https://doi.org/10.3390/agronomy10040520





