Dissimilar Responses of Ancient Grapevines Recovered in Navarra (Spain) to Arbuscular Mycorrhizal Symbiosis in Terms of Berry Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Biological Material
2.2. Experimental Design
2.3. Plant Determinations
2.4. Berry Determinations
2.5. Statistical Analyses
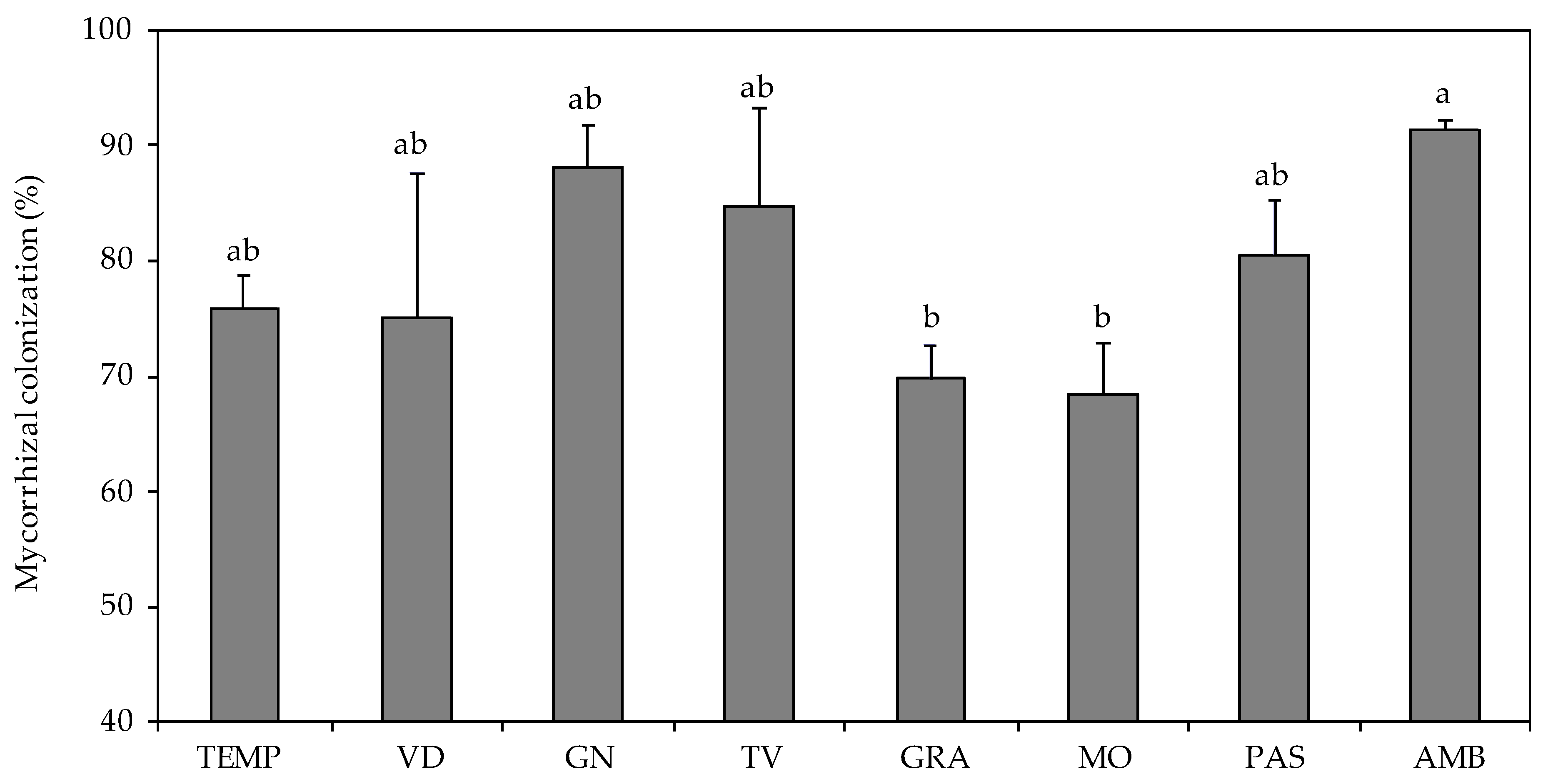
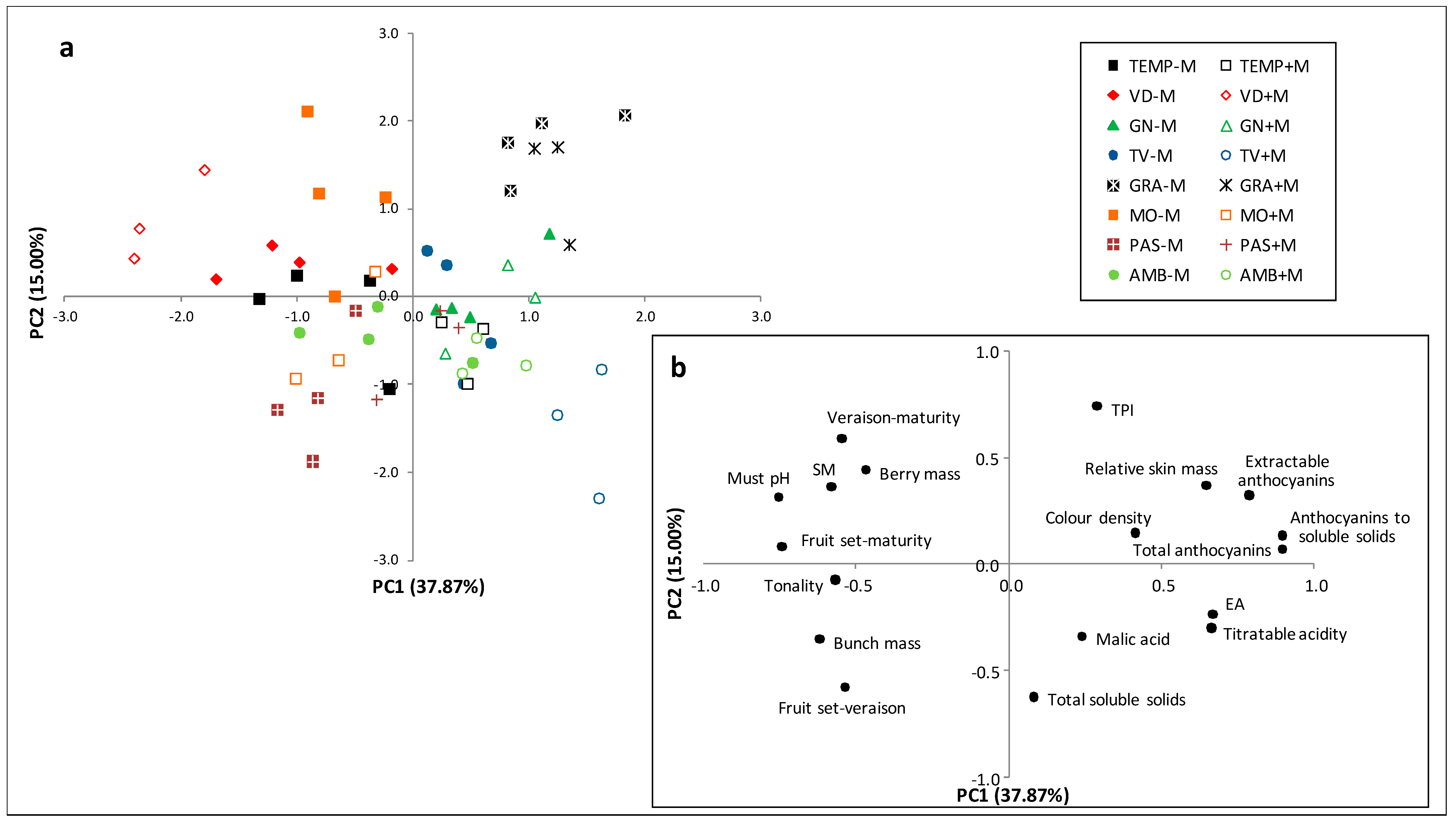
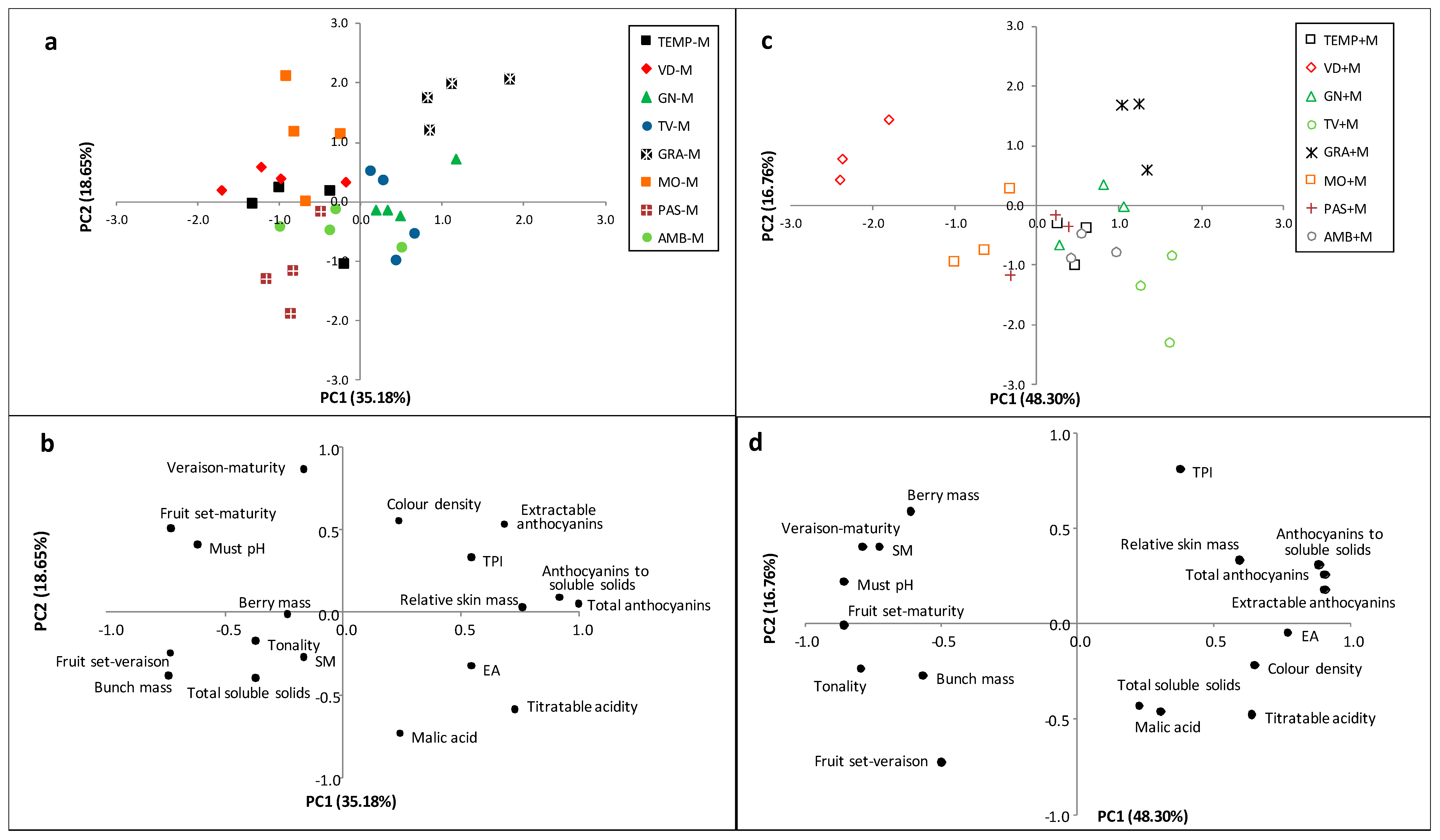
3. Results
3.1. Principal Component Analysis
3.2. Plant Characteristics
3.3. Berry Composition
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OIV Organisation Internationale de la Vigne et du Vin. State of the Vitiviniculture World Market in 2018. Available online: www.oiv.int (accessed on 7 September 2019).
- Sweetman, C.; Sadras, V.O.; Hancock, R.D.; Soole, K.L.; Ford, C.M. Metabolic effects of elevated temperature on organic acid degradation in ripening Vitis vinifera fruit. J. Exp. Bot. 2014, 65, 5975–5988. [Google Scholar] [CrossRef]
- Sadras, V.O.; Morán, M.A. Elevated temperature decouples anthocyanins and sugars in berries of Shiraz and Cabernet Franc. Aust. J. Grape Wine Res. 2012, 18, 115–122. [Google Scholar] [CrossRef]
- Morales-Castilla, I.; García de Cortázar-Atauri, I.; Cook, B.I.; Lacombe, T.; Parker, A.; van Leeuwen, C.; Nicholas, K.A.; Wolkovich, E.M. Diversity buffers winegrowing regions from climate change losses. Proc. Natl. Acad. Sci. USA 2020, 117, 2864–2869. [Google Scholar] [CrossRef]
- Eyduran, S.P.; Akin, M.; Ercisli, S.; Eyduran, E.; Maghradze, D. Sugars, organic acids, and phenolic compounds of ancient grape cultivars (Vitis vinifera L.) from Igdir province of Eastern Turkey. Biol. Res. 2015, 48, 2. [Google Scholar] [CrossRef]
- Loureiro, M.D.; Moreno-Sanz, P.; Suárez, B. Agronomical characterization of minority grapevine cultivars from Asturias (Spain). Ciéncia Téc. Vitiv. 2017, 32, 102–114. [Google Scholar] [CrossRef]
- Labagnara, T.; Bergamini, C.; Caputo, A.R.; Cirigliano, P. Vitis vinifera L. germplasm diversity: a genetic and ampelometric study in ancient vineyards in the South of Basilicata region (Italy). Vitis 2018, 57, 1–8. [Google Scholar]
- Balda, P.; Ibáñez, J.; Sancha, J.C.; Martínez de Toda, F. Characterization and identification of minority red grape varieties recovered in Rioja, Spain. Am. J. Enol. Vitic. 2014, 65, 148–152. [Google Scholar] [CrossRef]
- Urrestarazu, J.; Miranda, C.; Santesteban, L.G.; Royo, J.B. Recovery and identification of grapevine varieties cultivated in old vineyards from Navarre (Northeastern Spain). Sci. Hortic. 2015, 191, 65–73. [Google Scholar] [CrossRef]
- Jiménez, C.; Peiró, R.; Yuste, A.; García, J.; Martínez-Gil, F.; Gisbert, C. Looking for old grapevine varieties. Vitis 2019, 58, 59–60. [Google Scholar]
- Ocete, C.A.; Arroyo, R.; Lovicu, G.; Rodríguez-Miranda, A.; Valle, J.M.; Cantos, M.; García, J.L.; Lara, M.; González de Canales, F.; Llompart, J.; et al. An inventory of the relic Eurasian wild grapevine populational nuclei in Huelva province (Andalusia, Spain). Vitis 2019, 58, 53–57. [Google Scholar]
- Ibáñez, J.; Carreño, J.; Yuste, J.; Martínez-Zapater, J. Grapevine breeding and clonal selection programs in Spain. In Grapevine Breeding Programs for the Wine Industry; Elsevier: Sawston, UK, 2015; pp. 183–209. [Google Scholar]
- Martínez de Toda, F.; García, J.; Balda, P. Preliminary results on forcing vine regrowth to delay ripening to a cooler period. Vitis 2019, 58, 17–22. [Google Scholar]
- Balestrini, R.; Magurno, F.; Walker, C.; Lumini, E.; Bianciotto, V. Cohorts of arbuscular mycorrhizal fungi (AMF) in Vitis vinifera, a typical Mediterranean fruit crop. Environ. Microbiol. Rep. 2010, 3, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Ocete, R.; Armendáriz, I.; Cantos, M.; Álvarez, D.; Azcón, R. Ecological characterization of wild grapevine habitats focused on arbuscular mycorrhizal symbiosis. Vitis 2015, 54, 207–211. [Google Scholar]
- Schreiner, R.P. Mycorrhizal colonization of grapevine rootstocks under field conditions. Am. J. Enol. Vitic. 2003, 54, 143–149. [Google Scholar]
- Popescu, G.C. Arbuscular mycorrhizal fungi-an essential tool to sustainable vineyard development: a review. Curr. Trends Nat. Sci. 2016, 5, 107–116. [Google Scholar]
- Torres, N.; Goicoechea, N.; Zamarreño, A.; Antolín, M.C. Mycorrhizal symbiosis affects ABA metabolism during berry ripening in Vitis vinifera L. cv. Tempranillo grown under climate change scenarios. Plant. Sci. 2018, 274, 383–393. [Google Scholar] [CrossRef]
- Avio, L.; Turrini, A.; Giovannetti, M.; Sbrana, C. Designing the ideotype mycorrhizal symbionts for the production of healthy food. Front. Plant. Sci. 2018, 9, 1089. [Google Scholar] [CrossRef]
- Torres, N.; Goicoechea, N.; Morales, F.; Antolín, M.C. Berry quality and antioxidant properties in Vitis vinifera cv. Tempranillo as affected by clonal variability, mycorrhizal inoculation and temperature. Crop. Past. Sci. 2016, 67, 961–977. [Google Scholar] [CrossRef]
- Gabriele, M.; Gerardi, C.; Longo, V.; Lucejko, J.; Degano, I.; Pucci, L.; Domenici, V. The impact of mycorrhizal fungi on Sangiovese red wine production: phenolic compounds and antioxidant properties. LWT-Food Sci. Technol. 2016, 72, 310–316. [Google Scholar] [CrossRef]
- Ponce, M.A.; Scervino, J.M.; Erra-Balsells, R.; Ocampo, J.A.; Godeas, A.M. Flavonoids from shoots and roots of Trifolium repens (white clover) grown in presence or absence of the arbuscular mycorrhizal fungus Glomus intraradices. Phytochemistry 2004, 65, 1925–1930. [Google Scholar] [CrossRef]
- Bruisson, S.; Maillot, P.; Schellenbaum, P.; Walter, B.; Gindro, K.; Deglène-Benbrahim, L. Arbuscular mycorrhizal symbiosis stimulates key genes of the phenylpropanoid biosynthesis and stilbenoid production in grapevine leaves in response to downy mildew and grey mould infection. Phytochemistry 2016, 131, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Balestrini, R.; Salvioli, A.; DalMolin, A.; Novero, M.; Gabelli, G.; Paparelli, E.; Marroni, F.; Bonfante, P. Impact of an arbuscular mycorrhizal fungus versus a mixed microbial inoculum on the transcriptome reprogramming of grapevine roots. Mycorrhiza 2017, 27, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Torres, N.; Hilbert, G.; Antolín, M.C.; Goicoechea, N. Aminoacids and flavonoids profiling in Tempranillo berries can be modulated by the arbuscular mycorrhizal fungi. Plants 2019, 8, 400. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008; p. 800. ISBN 9780123705266. [Google Scholar]
- Staddon, P.L.; Fitter, A.H.; Graves, J.D. Effect of elevated atmospheric CO2 on mycorrhizal hyphal production and phosphorus inflow in Plantago lanceolata and Trifolium repens in association with the arbuscular mycorrhizal fungus Glomus mosseae. Glob. Change Biol. 1999, 5, 347–358. [Google Scholar] [CrossRef]
- MAPA (Ministerio de Agricultura, Pesca y Alimentación). Available online: https://www.mapa.gob.es/es/ (accessed on 7 September 2019).
- Mullins, M.G. Test-plants for investigations of the physiology of fruiting in Vitis vinifera L. Nature 1966, 209, 419–420. [Google Scholar] [CrossRef]
- Agnolucci, M.; Battini, F.; Cristani, C.; Giovannetti, M. Diverse bacterial communities are recruited on spores of different arbuscular mycorrhizal fungal isolates. Biol. Fertil. Soils 2015, 51, 379–389. [Google Scholar] [CrossRef]
- Trotel-Aziz, P.; Abou-Mansour, E.; Courteaux, B.; Rabenoelina, F.; Clément, C.; Fontaine, F.; Aziz, A. Bacillus subtilis PTA-271 counteracts botryosphaeria dieback in grapevine, triggering immune responses and detoxification of fungal phytotoxins. Front. Plant. Sci. 2019, 10, 25. [Google Scholar] [CrossRef]
- Coombe, B.G. Adoption of a system for identifying grapevine growth stages. Aust. J. Grape Wine Res. 1995, 1, 104–110. [Google Scholar] [CrossRef]
- Kliewer, W.M.; Dokoozlian, N.K. Leaf area/crop weight ratios of grapevines: influence on fruit composition and wine quality. Am. J. Enol. Vitic. 2005, 56, 170–181. [Google Scholar]
- Ollat, N.; Gény, L.; Soyer, J.P. Les boutures fructifères de vigne: validation d’un modèle d’étude de la physiologie de la vigne. I. Principales caractéristiques de l’appareil végétatif. J. Int. Sci. Vigne Vin 1998, 32, 1–9. [Google Scholar]
- Koske, R.E.; Gemma, J.N. A modified procedure for staining roots to detect VA mycorrhizas. Mycol. Res. 1989, 92, 486–488. [Google Scholar] [CrossRef]
- Giovannetti, M.; Mosse, B. An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytol. 1980, 87, 489–500. [Google Scholar] [CrossRef]
- OIV. Compendium of International Methods of Analysis of Wines and Musts, Vol. I; International Organisation of Vine and Wine: Paris, France, 2018; ISBN 979-10-91799-80-5. [Google Scholar]
- Saint-Cricq, N.; Vivas, N.; Glories, Y. Maturité phénolique: définition et contrôle. Rev. Fr. d’Oenol. 1998, 173, 22–25. [Google Scholar]
- Ribéreau-Gayon, J.; Stonestreet, E. Le dosage des anthocyanes dans le vin rouge. Bull. Soc. Chim. Fr. 1965, 9, 2649–2652. [Google Scholar]
- Nadal, M. Phenolic maturity in red grapes. In Methodologies and Results in Grapevine Research; Delrot, S., Medrano-Gil, H., Or, E., Bavaresco, L., Grando, S., Eds.; Springer Science+Business Media, B.V.: Dordrecht, The Netherlands, 2010; pp. 389–409. [Google Scholar]
- EEC (European Union Commission Regulation) N° 2676/90. Community Methods for the Analysis; Official Journal of the European Communities: Luxemburg, 1990; L 272:1-92. [Google Scholar]
- Glories, Y. La couleur des vins rouges. 2e partie: mesure, origine et interprétation. OENO One 1984, 18, 253–271. [Google Scholar] [CrossRef]
- Gisbert, C.; Peiró, R.; San Pedro, T.; Olmos, A.; Jiménez, C.; García, J. Recovering ancient grapevine varieties: from genetic variability to in vitro conservation, a case study. In Grapes and Wines. Advances in Production, Processing, Analysis and Valorization; Jordão, A.M., Cosme, F., Eds.; Intechopen: London, UK, 2015; pp. 3–21. [Google Scholar]
- Torres, N.; Antolín, M.C.; Goicoechea, N. Arbuscular mycorrhizal symbiosis as a promising resource for improving berry quality in grapevines under changing environments. Front. Plant. Sci. 2018, 9, 897. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Handbook of Enology: The Microbiology of Wine and Vinifications, 2nd ed.; Wiley & Sons: Chichester, UK, 2006; Vol.1. [Google Scholar]
- Poni, S.; Gatti, M.; Palliotti, A.; Dai, Z.; Duchêne, E.; Truong, T.T.; Ferrara, G.; Matarrese, M.S.; Gallotta, A.; Bellincontro, A.; et al. Grapevine quality: A multiple choice issue. Sci. Hort. 2018, 234, 445–462. [Google Scholar] [CrossRef]
- Karagiannidis, N.; Nikolaou, N.; Ipsilantis, I.; Zioziou, E. Effects of different N fertilizers on the activity of Glomus mosseae and on grapevine nutrition and berry composition. Mycorrhiza 2007, 18, 43–50. [Google Scholar] [CrossRef]
- Georgiev, V.; Ananga, A.; Tsolova, V. Recent advances and uses of grape flavonoids as nutraceuticals. Nutrients 2014, 6, 391–415. [Google Scholar] [CrossRef]
- Giovinazzo, G.; Grieco, F. Functional properties of grape and wine polyphenols. Plant. Foods Hum. Nutr. 2015, 70, 454–462. [Google Scholar] [CrossRef]
- He, F.; Mu, L.; Yan, G.-L.; Liang, N.N.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.-Q. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010, 15, 9057–9091. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, N.; Antolín, M.C. Increased nutritional value in food crops. Microb. Biotechnol. 2017, 10, 1004–1007. [Google Scholar] [CrossRef] [PubMed]
| Variety | Clone | Code | Reproductive Cycle | Bunch Mass (g bunch−1) | Berry Mass (g) |
|---|---|---|---|---|---|
| Tempranillo | T23 | TEMP | Medium | 329 | 2.58 |
| Vidadillo | T75 | VID | Medium | 326 | 2.29 |
| Grand Noir | T48 | GN | Medium | 173 | 1.27 |
| Tinto Velasco | T73 | TV | Medium | 158 | 2.13 |
| Graciano | T72 | GRA | Medium | 85 | 1.17 |
| Morate | T71 | MO | Long | 216 | 1.65 |
| Pasera | T85 | PAS | Long | 449 | 2.63 |
| Ambrosina | T46 | AMB | Long | 219 | 1.74 |
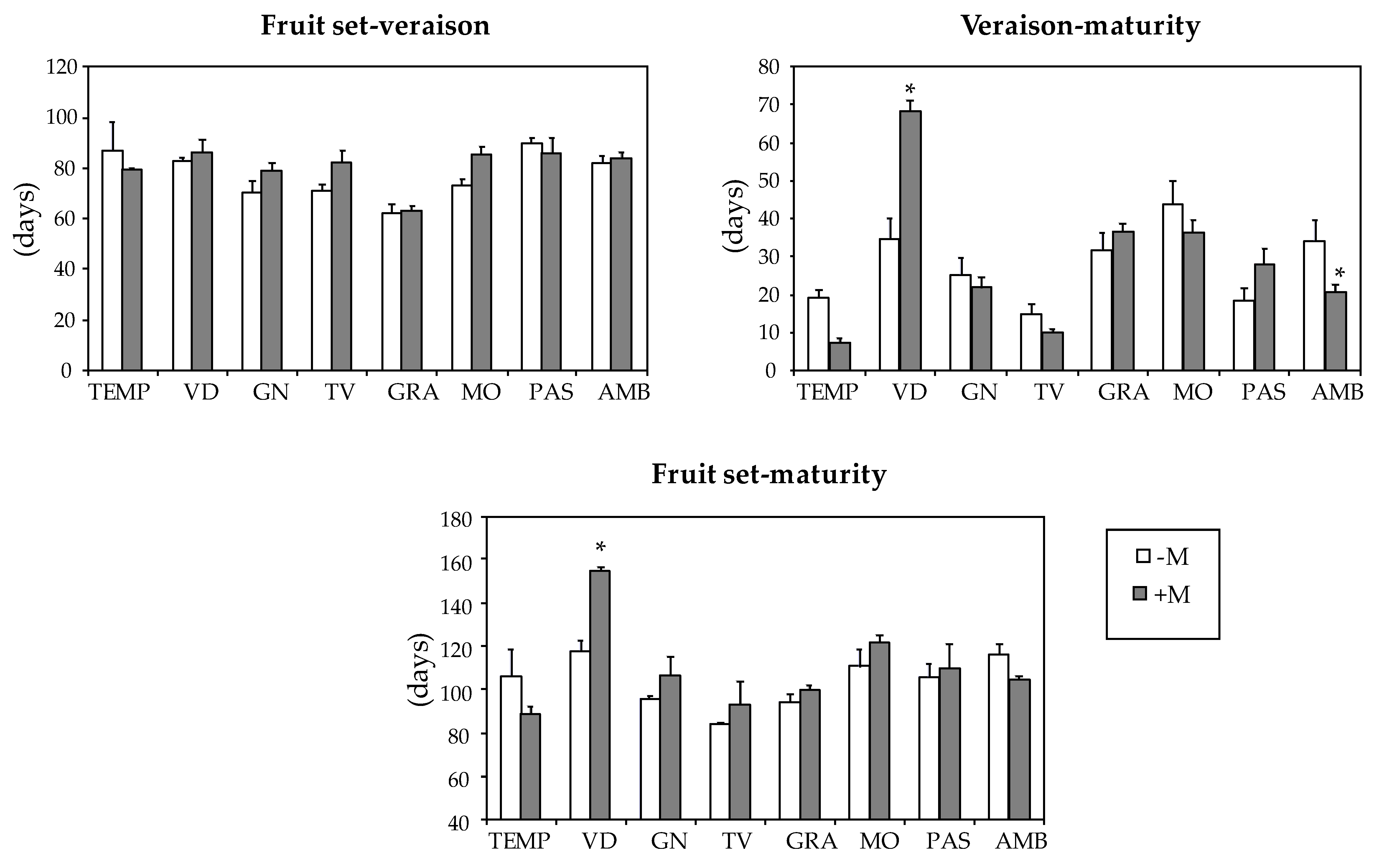
| Varieties | Fruit Set-Veraison (Days) | Veraison-Maturity (Days) | Fruit set-Maturity (Days) |
|---|---|---|---|
| Main effects | |||
| Variety (V) | |||
| TEMP | 83 ab | 14 d | 98 bcd |
| VID | 84 ab | 49 a | 133 a |
| GN | 74 b | 24 cd | 100 bcd |
| TV | 76 b | 13 d | 88 d |
| GRA | 62 c | 34 bc | 96 cd |
| MO | 78 ab | 41 ab | 115 b |
| PAS | 88 a | 22 cd | 107 bc |
| AMB | 82 ab | 28 c | 111 bc |
| Mycorrhization (M) | |||
| -M | 77 a | 28 a | 103 a |
| +M | 81 a | 29 a | 110 a1 |
| ANOVA2 | |||
| Variety (V) | *** | *** | *** |
| Mycorrhization (M) | ns | ns | ns |
| V × M | ns | *** | *** |
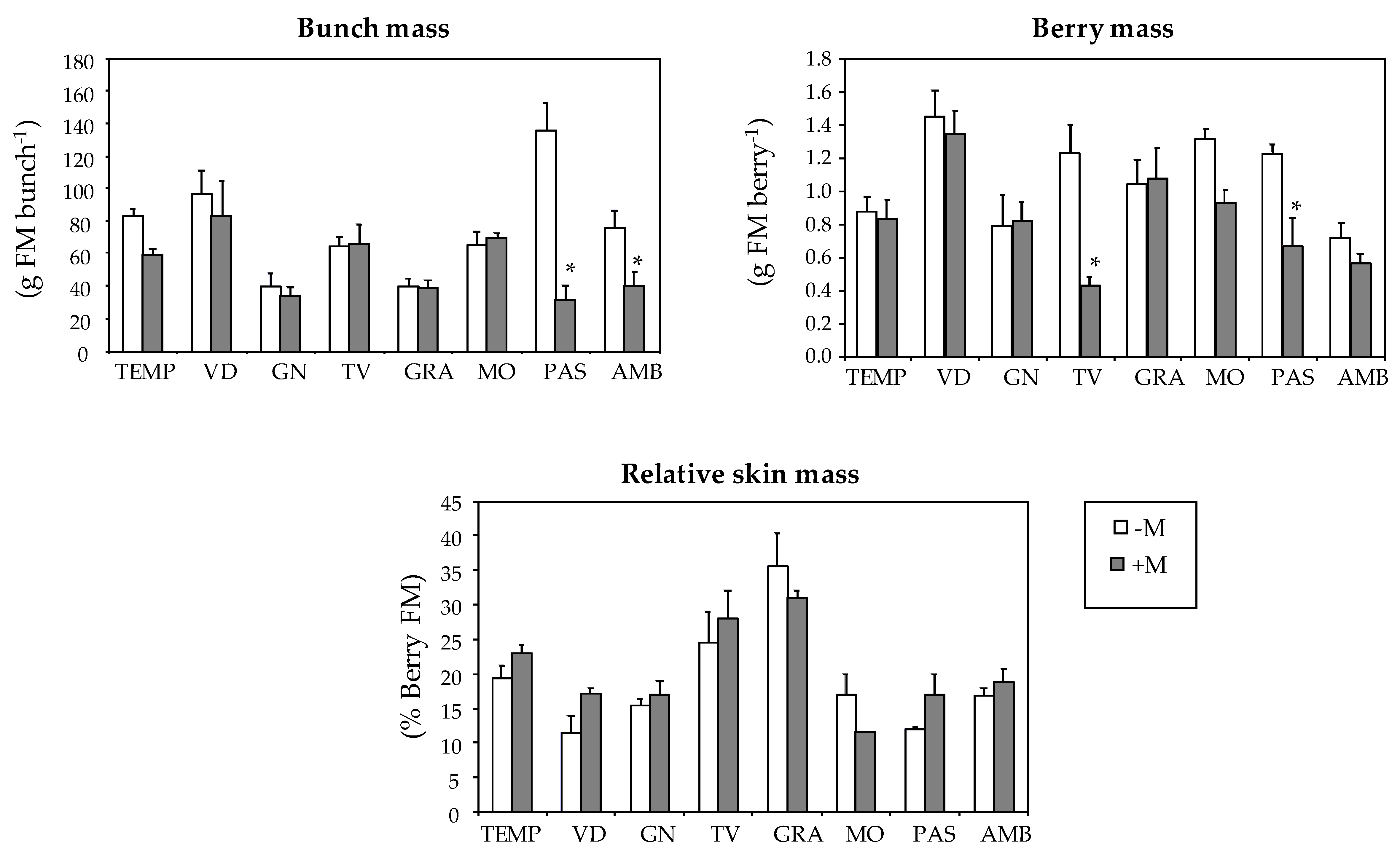
| Varieties | Bunch Mass (g FM3 bunch−1) | Berry Mass (g FM berry−1) | Relative Skin Mass (% berry FM) |
|---|---|---|---|
| Main effects | |||
| Variety (V) | |||
| TEMP | 72.8 a | 0.86 bc | 20.9 bc |
| VID | 91.1 a | 1.41 a | 13.9 d |
| GN | 37.5 b | 0.81 bc | 16.1 cd |
| TV | 65.1 ab | 0.89 bc | 26.0 b |
| GRA | 39.5 b | 1.06 b | 33.6 a |
| MO | 67.3 ab | 1.16 ab | 14.6 d |
| PAS | 91.2 a | 0.99 bc | 14.1 d |
| AMB | 60.5 ab | 0.66 c | 17.7 cd |
| Mycorrhization (M) | |||
| -M | 75.0 a | 1.08 a | 19.0 a |
| +M | 53.2 b | 0.84 b | 20.4 a1 |
| ANOVA2 | |||
| Variety (V) | *** | *** | *** |
| Mycorrhization (M) | *** | *** | ns |
| V × M | *** | * | ns |
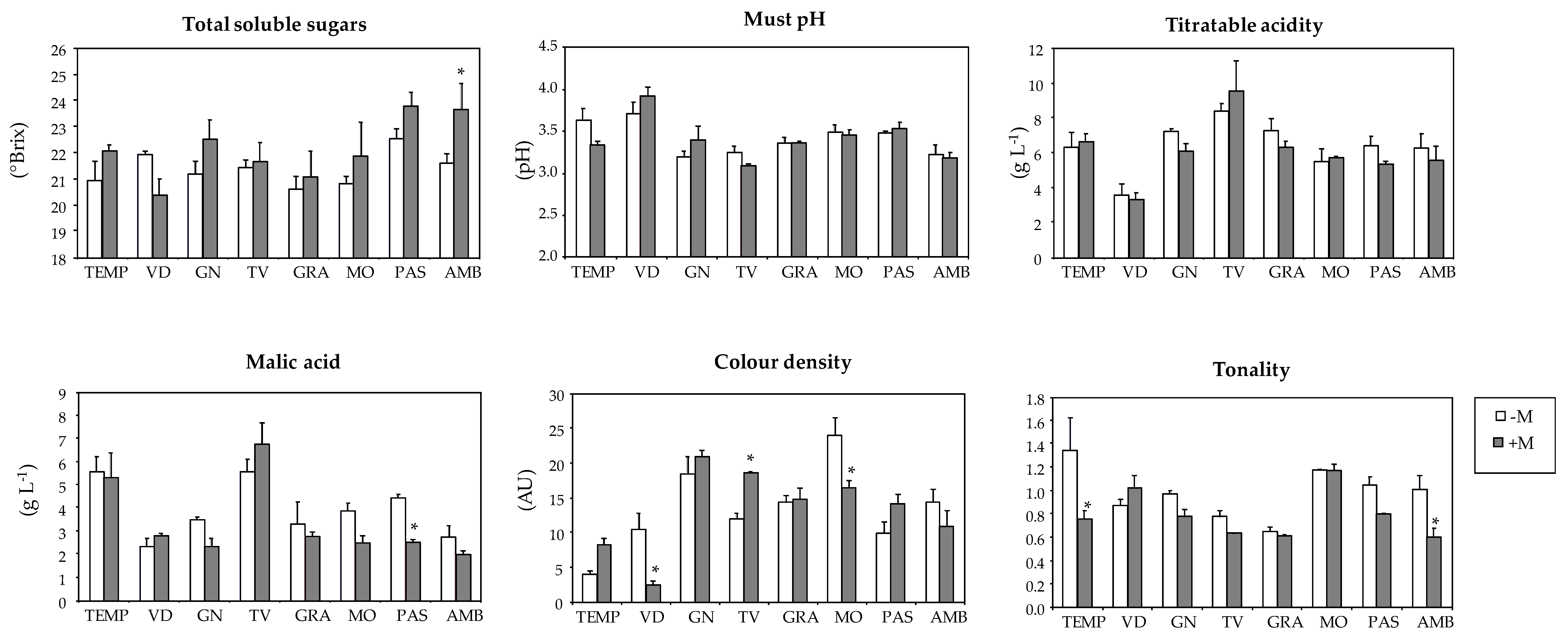
| Varieties | Total Soluble Solids (°Brix) | Must pH | Titratable Acidity (g L−1) | Malic Acid (g L−1) | Colour Density (AU) 3 | Tonality Index |
|---|---|---|---|---|---|---|
| Main effects | ||||||
| Variety (V) | ||||||
| TEMP | 21.4 a | 3.50 b | 6.45 b | 5.43 a | 5.8 c | 1.09 ab |
| VID | 21.3 a | 3.80 a | 3.49 c | 2.51 b | 7.0 c | 0.93 abc |
| GN | 21.8 a | 3.28 b | 6.75 b | 2.97 b | 19.5 a | 0.89 bcd |
| TV | 21.5 a | 3.18 d | 8.89 a | 6.04 a | 14.8 b | 0.72 cd |
| GRA | 20.8 a | 3.36 bcd | 6.84 b | 3.07 b | 14.6 b | 0.63 d |
| MO | 21.3 a | 3.47 bc | 5.59 b | 3.26 b | 20.8 a | 1.17 a |
| PAS | 23.1 a | 3.50 b | 5.95 b | 3.59 b | 11.7 b | 0.94 abc |
| AMB | 22.5 a | 3.20 d | 5.98 b | 2.41 b | 12.9 b | 0.83 bcd |
| Mycorrhization (M) | ||||||
| -M | 21.4 b | 3.42 a | 6.37 a | 3.89 a | 13.4 a | 0.98 a |
| +M | 22.1 a | 3.41 a | 6.07 a | 3.36 a | 13.3 a | 0.79 b 1 |
| ANOVA2 | ||||||
| Variety (V) | ns | *** | *** | *** | *** | *** |
| Mycorrhization (M) | ** | ns | ns | ns | ns | *** |
| V × M | ns | ns | ns | ns | *** | * |
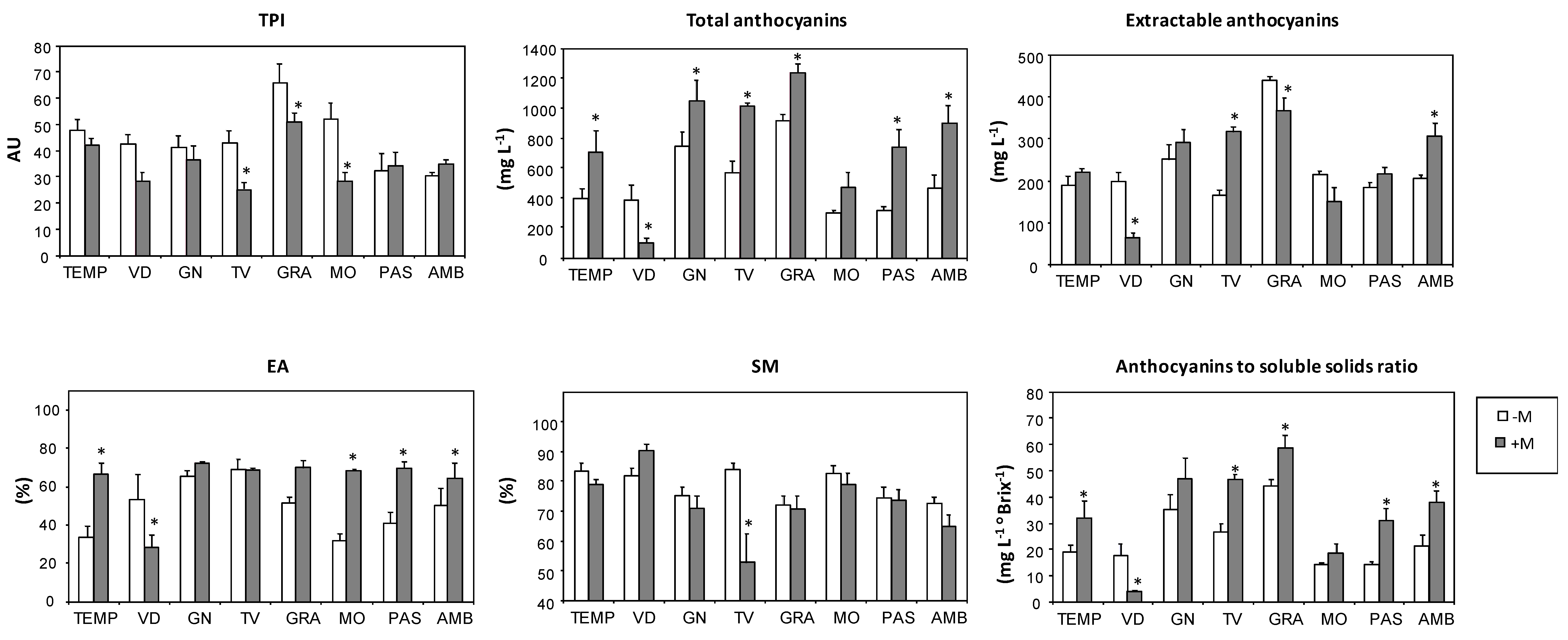
| Varieties | TPI3 (AU)4 | Total Anthocyanins (mg L−1) | Extractable Anthocyanins (mg L−1) | EA5 (%) | SM6 (%) | Anthocyanin/Soluble Solids (mg L−1 °Brix−1) |
|---|---|---|---|---|---|---|
| Main effects | ||||||
| Variety (V) | ||||||
| TEMP | 45.3 b | 527.4 cd | 202.0 bcd | 47.8 b | 81.6 ab | 24.4 cde |
| VID | 36.5 bc | 261.1 d | 139.5 d | 42.5 b | 85.6 a | 11.6 f |
| GN | 39.2 bc | 871.6 ab | 268.4 b | 68.3 a | 73.4 bcd | 40.3 ab |
| TV | 35.1 bc | 758.5 bc | 230.0 bc | 68.9 a | 70.8 cd | 35.2 bc |
| GRA | 59.5 a | 1048.5 a | 407.4 a | 59.4 ab | 71.5 bcd | 50.5 a |
| MO | 41.8 bc | 370.5 d | 186.4 cd | 47.2 b | 81.1 abc | 16.1 ef |
| PAS | 33.0 bc | 495.6 cd | 197.4 bcd | 53.2 ab | 74.2 bcd | 21.3 def |
| AMB | 32.2 c | 649.5 bc | 248.4 bc | 56.2 ab | 69.3 d | 28.5 cd |
| Mycorrhization (M) | ||||||
| -M | 44.3 a | 509.7 b | 230.3 a | 49.5 b | 78.3 a | 24.0 b |
| +M | 35.1 b | 773.7 a | 241.1 a | 63.4 a | 72.7 a | 34.5 a1 |
| ANOVA2 | ||||||
| Variety (V) | *** | *** | *** | *** | *** | *** |
| Mycorrhization (M) | *** | *** | ns | *** | ns | *** |
| V × M | * | ** | *** | *** | ns | ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antolín, M.C.; Izurdiaga, D.; Urmeneta, L.; Pascual, I.; Irigoyen, J.J.; Goicoechea, N. Dissimilar Responses of Ancient Grapevines Recovered in Navarra (Spain) to Arbuscular Mycorrhizal Symbiosis in Terms of Berry Quality. Agronomy 2020, 10, 473. https://doi.org/10.3390/agronomy10040473
Antolín MC, Izurdiaga D, Urmeneta L, Pascual I, Irigoyen JJ, Goicoechea N. Dissimilar Responses of Ancient Grapevines Recovered in Navarra (Spain) to Arbuscular Mycorrhizal Symbiosis in Terms of Berry Quality. Agronomy. 2020; 10(4):473. https://doi.org/10.3390/agronomy10040473
Chicago/Turabian StyleAntolín, M. Carmen, David Izurdiaga, Leyre Urmeneta, Inmaculada Pascual, Juan José Irigoyen, and Nieves Goicoechea. 2020. "Dissimilar Responses of Ancient Grapevines Recovered in Navarra (Spain) to Arbuscular Mycorrhizal Symbiosis in Terms of Berry Quality" Agronomy 10, no. 4: 473. https://doi.org/10.3390/agronomy10040473
APA StyleAntolín, M. C., Izurdiaga, D., Urmeneta, L., Pascual, I., Irigoyen, J. J., & Goicoechea, N. (2020). Dissimilar Responses of Ancient Grapevines Recovered in Navarra (Spain) to Arbuscular Mycorrhizal Symbiosis in Terms of Berry Quality. Agronomy, 10(4), 473. https://doi.org/10.3390/agronomy10040473






