Cropping System Redesign for Improved Weed Management: A Modeling Approach Illustrated with Giant Ragweed (Ambrosia trifida)
Abstract
1. Introduction
1.1. Overview
1.2. Giant Ragweed (Ambrosia trifida L.)
1.3. Options for Cropping System Redesign in the U.S. Corn Belt
2. Model Development and Evaluation
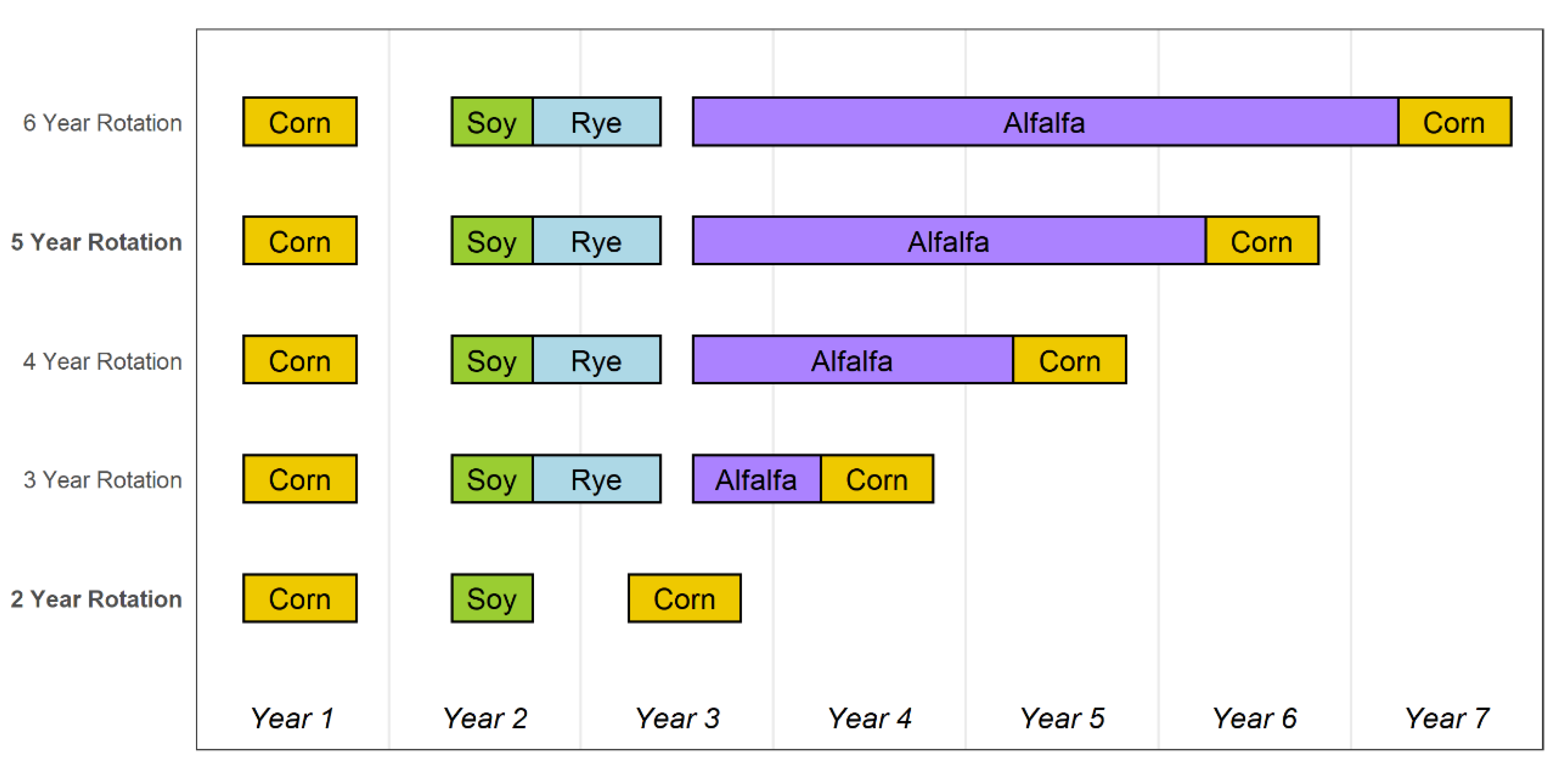
2.1. Cropping System Characteristics
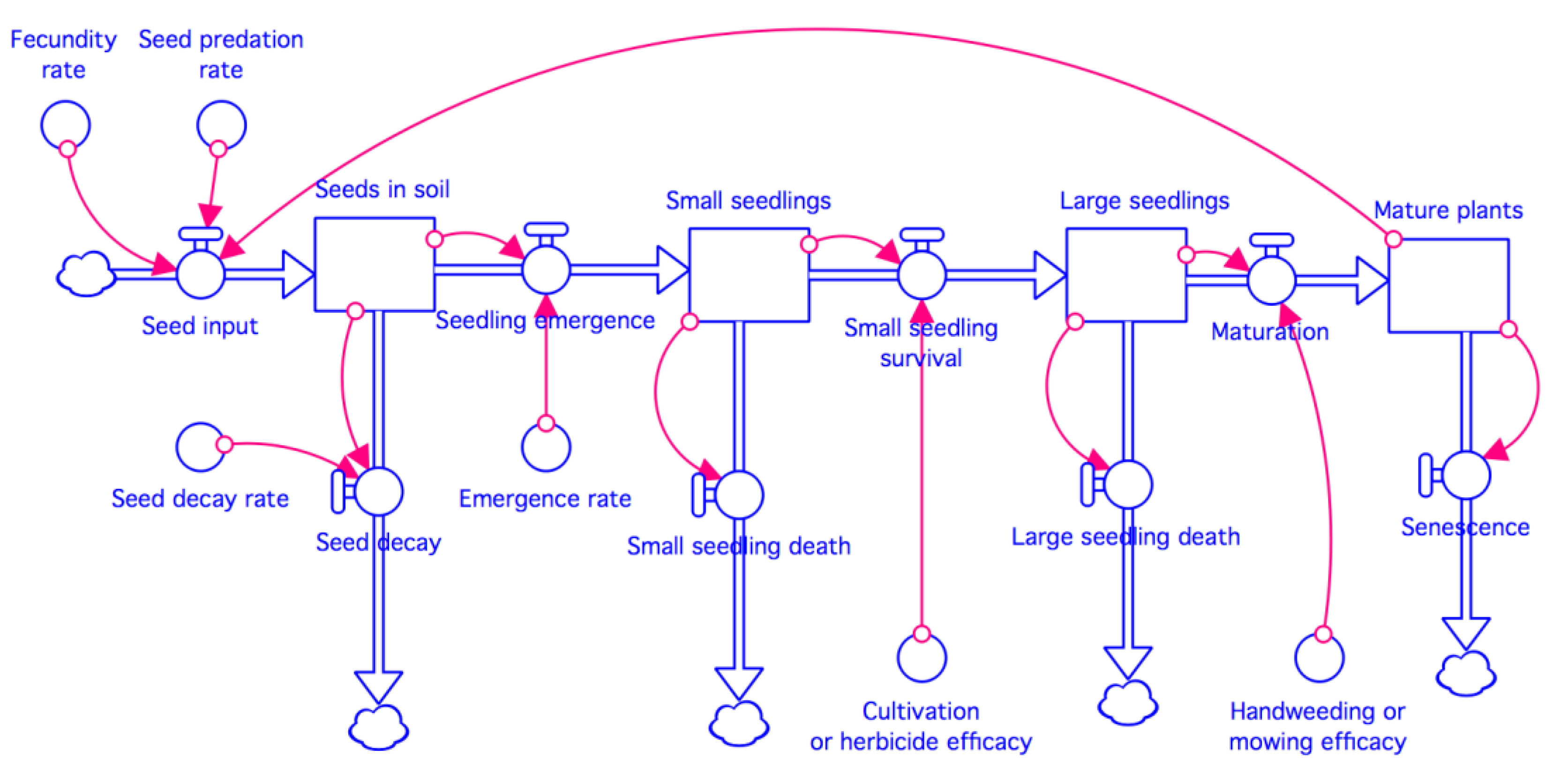
2.2. Model Concept and Structure
2.3. Giant Ragweed Demographic Parameters
2.4. Model Calculations and Scenario Evaluations
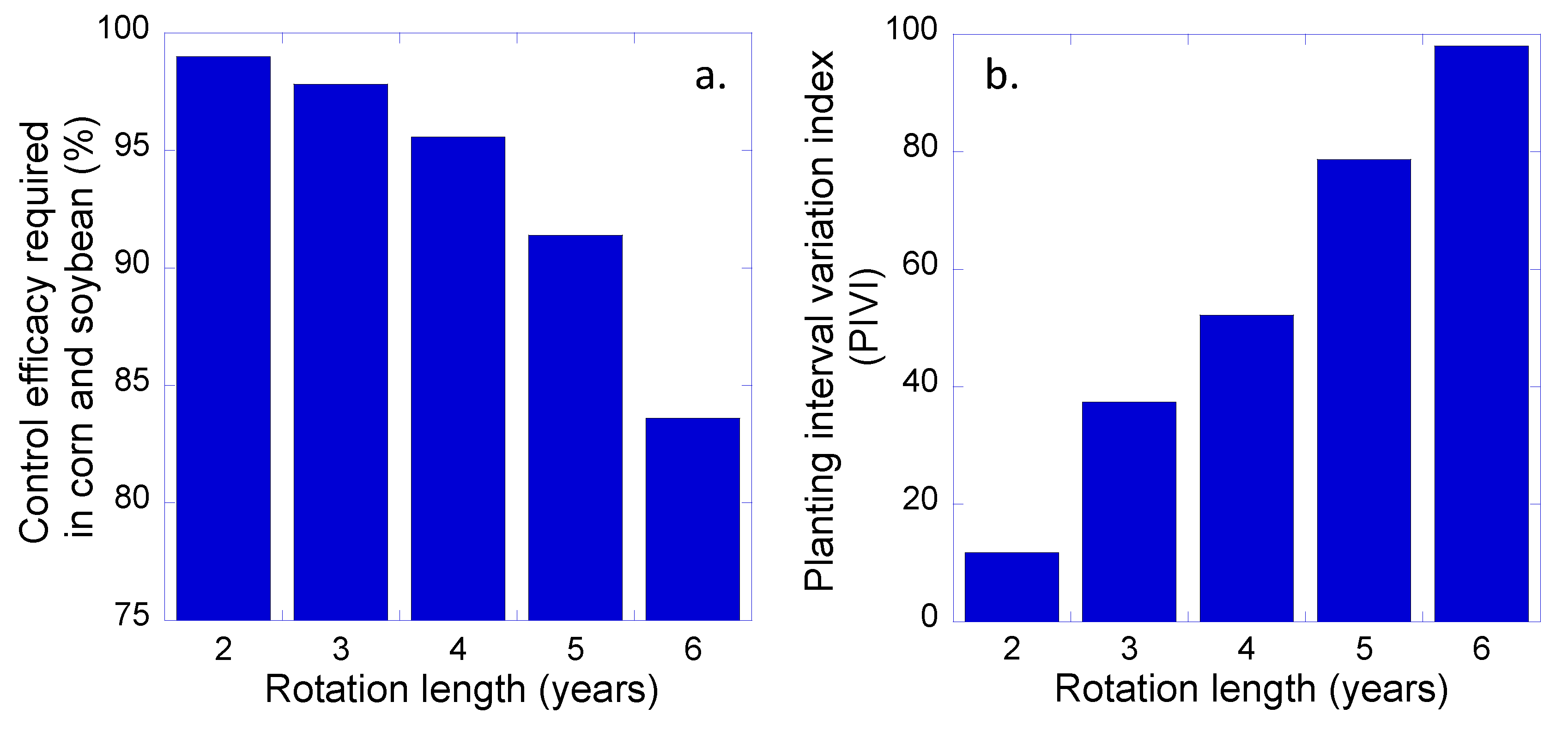
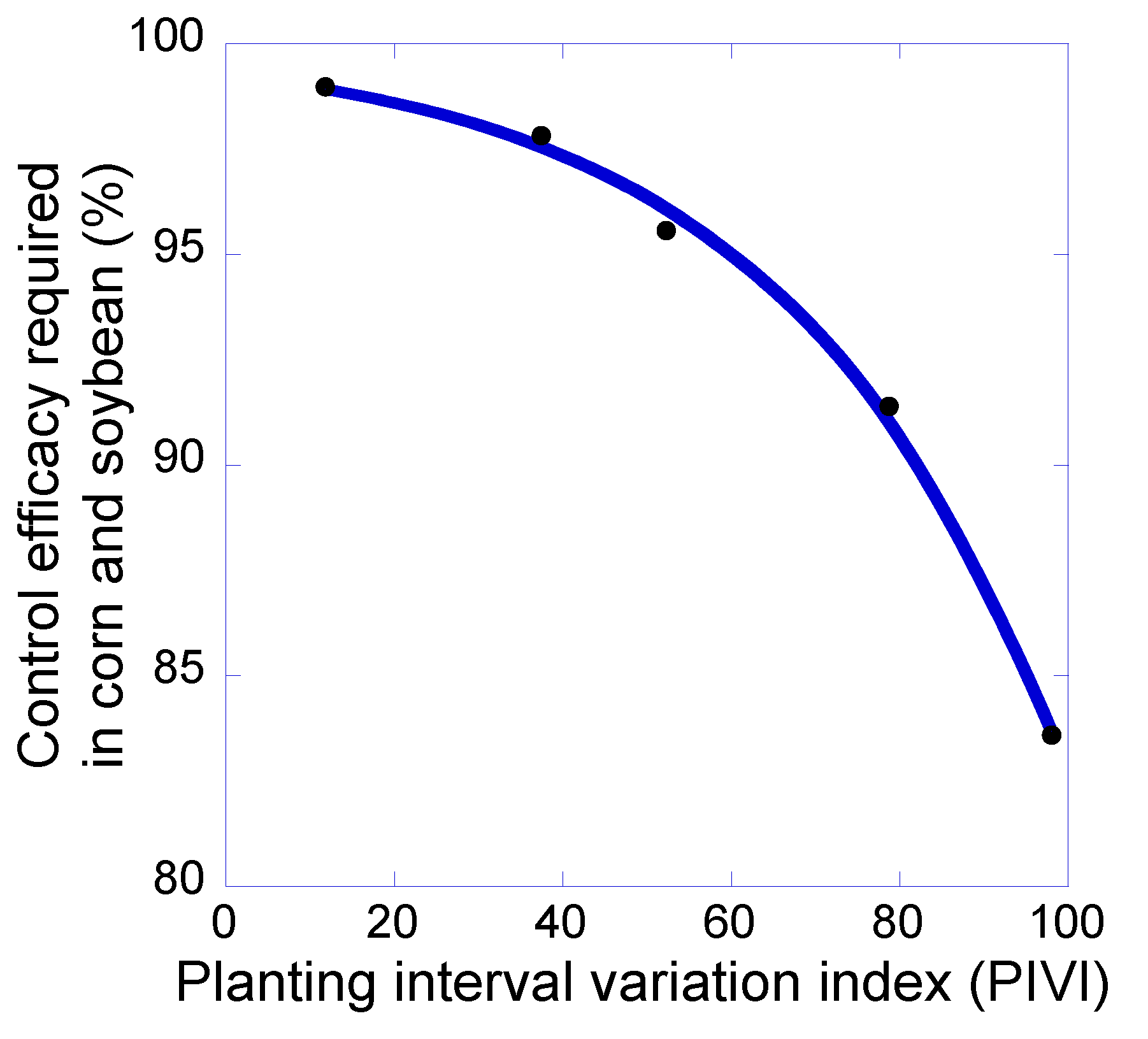
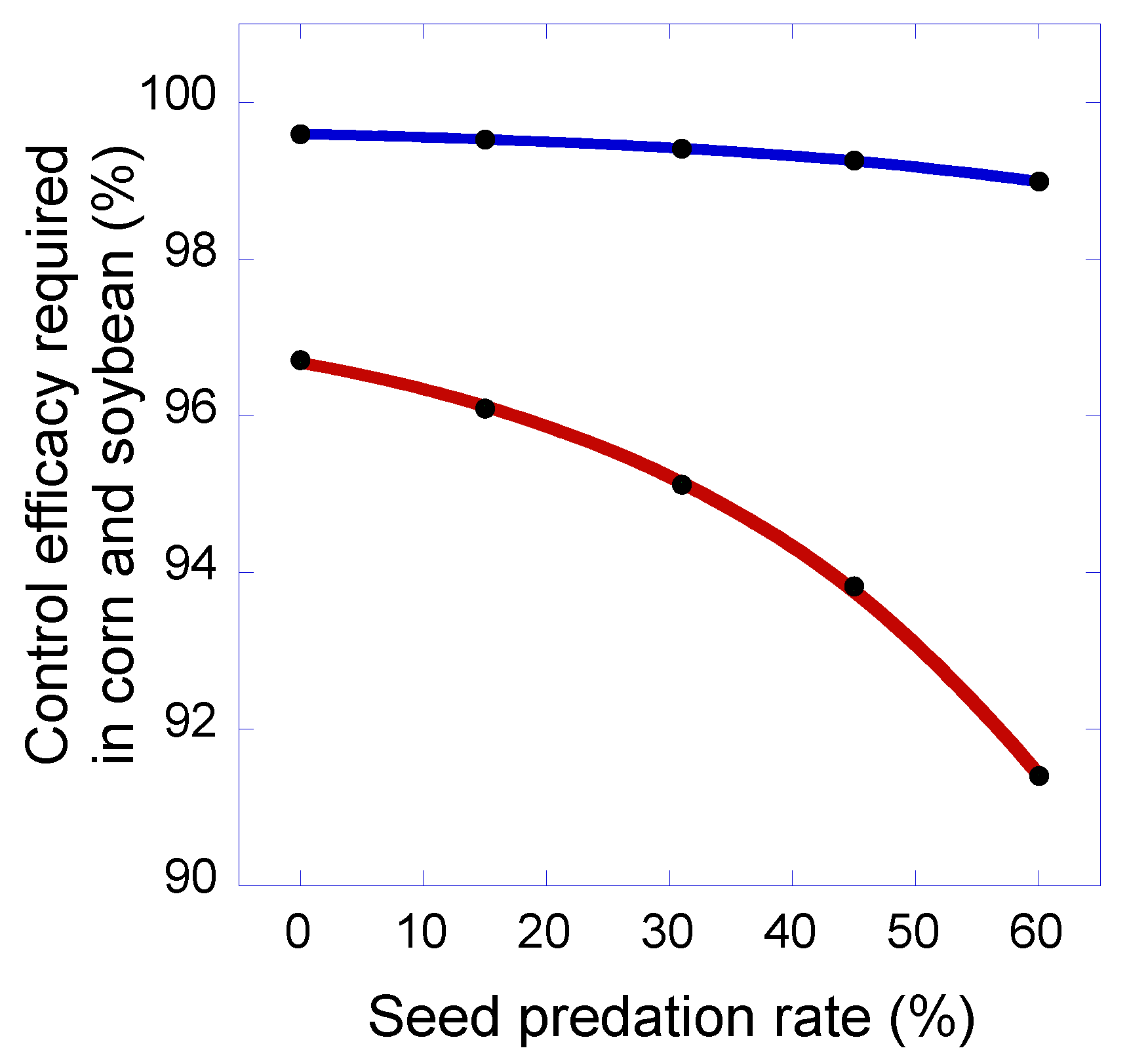
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 1–43. [Google Scholar] [CrossRef]
- Heap, I. Trends in the development of herbicide-resistant weeds. In Integrated Weed Management for Sustainable Agriculture; Zimdahl, R.L., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2018; pp. 169–192. [Google Scholar]
- Preston, C. Assessing and minimizing the environmental effects of herbicides. In Integrated Weed Management for Sustainable Agriculture; Zimdahl, R.L., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2018; pp. 133–167. [Google Scholar]
- Westwood, J.H.; Charudattan, R.; Duke, S.O.; Fennimore, S.A.; Marrone, P.; Slaughter, D.C.; Swanton, C.; Zollinger, R. Weed management in 2050: Perspectives on the future of weed science. Weed Sci. 2018, 66, 275–285. [Google Scholar] [CrossRef]
- Gonsolus, J.L.; Buhler, D.D. A risk management perspective on integrated weed management. J. Crop Prod. 1999, 2, 167–187. [Google Scholar] [CrossRef]
- Gallandt, E.R.; Brainerd, D.; Brown, B. Developments in physical weed control. In Integrated Weed Management for Sustainable Agriculture; Zimdahl, R.L., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2018; pp. 261–283. [Google Scholar]
- Liebman, M.; Davis, A.S. Managing weeds in organic farming systems: An ecological approach. In Organic Farming: The Ecological System; Francis, C., Ed.; American Society of Agronomy: Madison, WI, USA; Crop Science Society of America: Madison, WI, USA; Soil Science Society of America: Madison, WI, USA, 2009; pp. 173–195. [Google Scholar]
- Mortensen, D.A.; Egan, J.F.; Maxwell, B.D.; Ryan, M.R.; Smith, R.G. Navigating a critical juncture for sustainable weed management. Bioscience 2012, 62, 75–84. [Google Scholar] [CrossRef]
- Bagavathiannan, M.V.; Davis, A.S. An ecological perspective on managing weeds during the great selection for herbicide resistance. Pest Manag. Sci. 2018, 74, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.S. Weed ecology and population dynamics. In Integrated Weed Management for Sustainable Agriculture; Zimdahl, R.L., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2018; pp. 3–28. [Google Scholar]
- Beckie, H.J.; Harker, K.N. Our top 10 herbicide-resistant weed management practices. Pest Manag. Sci. 2017, 73, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, J.R. The use of rotations and cover crops to manage weeds. In Integrated Weed Management for Sustainable Agriculture; Zimdahl, R.L., Ed.; Burleigh Dodds Science Publishing: Cambridge, UK, 2018; pp. 227–260. [Google Scholar]
- Liebman, M.; Gallandt, E.R. Many little hammers: Ecological management of crop-weed interactions. In Ecology in Agriculture; Jackson, L.E., Ed.; Academic Press: Orlando, FL, USA, 1997; pp. 287–339. [Google Scholar]
- Weisberger, D.A.; Nichols, V.; Liebman, M. Does diversifying crop rotations suppress weeds? A meta-analysis. PLoS ONE 2019. [Google Scholar] [CrossRef]
- Gaba, S.; Fried, G.; Kazakou, E.; Chauvel, B.; Navas, M.-L. Agroecological weed control using a functional approach: A review of cropping systems diversity. Agron. Sustain. Dev. 2014, 34, 103–119. [Google Scholar] [CrossRef]
- Levins, R. The strategy of model building in population biology. Am. Sci. 1966, 54, 421–431. [Google Scholar]
- Regnier, E.E.; Harrison, S.K.; Loux, M.M.; Holloman, C.; Venkatesh, R.; Diekmann, F.; Taylor, R.; Ford, R.A.; Stoltenberg, D.E.; Hartzler, R.G.; et al. Certified Crop Advisors’ perceptions of giant ragweed (Ambrosia trifida) distribution, herbicide resistance, and management in the Corn Belt. Weed Sci. 2016, 64, 361–377. [Google Scholar] [CrossRef]
- Bazzaz, F.A. Demographic consequences of plant physiological traits: Some case studies. In Perspectives on Plant Population Ecology; Dirzo, R., Surukhán, J., Eds.; Sinauer Associates: Sunderland, MA, USA, 1984; pp. 324–346. [Google Scholar]
- Harrison, S.K.; Regnier, E.E.; Schmoll, J.T.; Webb, J.E. Competition and fecundity of giant ragweed in corn. Weed Sci. 2001, 49, 224–229. [Google Scholar] [CrossRef]
- Goplen, J.J.; Sheaffer, C.C.; Becker, R.L.; Coulter, J.A.; Breitenback, F.R.; Behnken, L.M.; Johnson, G.A.; Gunsolus, J.L. Giant ragweed (Ambrosia trifida) seed production and retention in soybean and field margins. Weed Technol. 2016, 30, 246–253. [Google Scholar] [CrossRef]
- Harrison, S.K.; Regnier, E.E.; Schmoll, J.T. Postdispersal predation of giant ragweed (Ambrosia trifida) seed in no-tillage corn. Weed Sci. 2003, 51, 955–964. [Google Scholar] [CrossRef]
- Harrison, S.K.; Regnier, E.E.; Schmoll, J.T.; Harrison, J.M. Seed size and burial effects on giant ragweed (Ambrosia trifida) emergence and seed demise. Weed Sci. 2007, 55, 16–22. [Google Scholar] [CrossRef]
- Goplen, J.J.; Sheaffer, C.C.; Becker, R.L.; Coulter, J.A.; Breitenback, F.R.; Behnken, L.M.; Johnson, G.A.; Gunsolus, J.L. Seedbank depletion and emergence patterns of giant ragweed (Ambrosia trifida) in Minnesota cropping systems. Weed Sci. 2017, 65, 52–60. [Google Scholar] [CrossRef]
- Karlen, D.L.; Hurley, E.G.; Andrews, S.S.; Cambardella, C.A.; Meek, D.W.; Duffy, M.D.; Mallarino, A.P. Crop rotation effects on soil quality at three northern Corn/Soybean Belt locations. Agron. J. 2006, 98, 484–495. [Google Scholar] [CrossRef]
- Broussard, W.P.; Turner, R.E.; Westra, J.V. Do federal farm policies influence surface water quality? Agric. Ecosyst. Environ. 2012, 158, 103–109. [Google Scholar] [CrossRef]
- Hatfield, J.L.; McMullen, L.D.; Jones, C.S. Nitrate-nitrogen patterns in the Raccoon River Basin related to agricultural practices. J. Soil Water Conserv. 2009, 64, 190–199. [Google Scholar] [CrossRef]
- US. Department of Agriculture-National Agricultural Statistics Service. 2017 Census of Agriculture; USDA-NASS: Washington, DC, USA, 2019; Available online: https://www.nass.usda.gov/Publications/AgCensus/2017/ (accessed on 11 February 2020).
- Alexander, R.B.; Smith, R.A.; Schwarz, G.E.; Boyer, E.W.; Nolan, J.V.; Brakebill, J.W. Differences in phosphorus and nitrogen delivery to the Gulf of Mexico from the Mississippi River Basin. Environ. Sci. Technol. 2008, 42, 822–830. [Google Scholar] [CrossRef]
- Heathcote, A.J.; Filstrup, C.T.; Downing, J.A. Watershed sediment losses to lakes accelerating despite agricultural soil conservation efforts. PLoS ONE 2013, e53554. [Google Scholar] [CrossRef]
- Sulc, R.M.; Tracy, B.F. Integrated crop–livestock systems in the U.S. Corn Belt. Agron. J. 2007, 99, 335–345. [Google Scholar] [CrossRef]
- Poffenbarger, H.; Artz, G.; Dahlke, G.; Edwards, W.; Hanna, M.; Russell, J.; Sellers, H.; Liebman, M. An economic analysis of integrated crop-livestock systems in Iowa, USA. Agric. Syst. 2017, 157, 51–69. [Google Scholar] [CrossRef]
- Goplen, J.J.; Coulter, J.A.; Sheaffer, C.C.; Becker, R.L.; Breitenbach, F.R.; Behnken, L.M.; Gonsulus, J.L. Economic performance of crop rotations in the presence of herbicide-resistant giant ragweed. Agron. J. 2018, 110, 260–268. [Google Scholar] [CrossRef]
- Hunt, N.; Hill, J.; Liebman, M. Cropping system diversity effects on nutrient discharge, soil erosion, and agronomic performance. Environ. Sci. Tech. 2019, 53, 1344–1352. [Google Scholar] [CrossRef]
- Hunt, N.; Hill, J.; Liebman, M. Reducing freshwater toxicity while maintaining weed control, profits, and productivity: Effects of increased crop rotation diversity and reduced herbicide usage. Environ. Sci. Technol. 2017, 51, 1707–1717. [Google Scholar] [CrossRef]
- Hunt, N.D.; Liebman, M.; Thakar, S.K.; Hill, J.D. Fossil energy use, climate change impacts, and air quality-related human health damages of conventional and diversified cropping systems in Iowa, USA. Environ. Sci. Technol. 2020, in press. [Google Scholar]
- Mallory, E.; Molloy, T. Winter Rye Variety Trial: 2017 Results; University of Maine Cooperative Extension: Orono, ME, USA, 2018; Available online: https://extension.umaine.edu/grains-oilseeds/wp-content/uploads/sites/16/2018/05/Winter-Rye-Variety-Trial-2017-Results.pdf (accessed on 11 February 2020).
- Wiersma, J.; Wells, S.; García y García, A. Winter Rye Field Crop Trials Results; Minnesota Agricultural Experiment Station: St. Paul, MN, USA, 2018; Available online: https://www.maes.umn.edu/sites/maes.umn.edu/files/2018_winter_rye_final.pdf (accessed on 11 February 2020).
- Wilde, P.; Schmiedchen, B.; Menzel, J.; Gordillo, A.; Fowler, D.B. Brasetto hybrid winter rye. Can. J. Plant Sci. 2018, 98, 195–198. [Google Scholar] [CrossRef]
- McGhee, M.L.; Stein, H.H. Apparent and standardized ileal digestibility of AA and starch in hybrid rye, barley, wheat, and corn fed to growing pigs. J. Anim. Sci. 2018, 96, 3319–3329. [Google Scholar] [CrossRef]
- Smit, M.N.; Zhou, X.; Landero, J.L.; Young, M.G.; Beltranena, E. Hybrid Rye Replacing Wheat Grain for Hogs. Available online: https://pdfs.semanticscholar.org/f7ae/d9b2e8750b6b1824de72f5bb7eaa24b08983.pdf (accessed on 11 February 2020).
- Barnes, D.K.; Goplen, B.P.; Baylor, J.E. Highlights in the USA and Canada. In Alfalfa and Alfalfa Improvement; Hanson, A.A., Barnes, D.K., Hill, R.R., Jr., Eds.; American Society of Agronomy-Crop Science Society of America-Soil Science Society of America: Madison, WI, USA, 1988; pp. 1–24. [Google Scholar]
- Mahoney, K.J.; McNaughton, K.E.; Sikkema, P.H. Control of glyphosate-resistant giant ragweed in winter wheat. Weed Technol. 2015, 29, 868–873. [Google Scholar] [CrossRef]
- Wortman, S.E.; Davis, A.S.; Schutte, B.J.; Lindquist, J.L.; Cardina, J.; Felix, J.; Sprague, C.L.; Dille, J.A.; Ramirez, A.H.M.; Reicks, G.; et al. Local conditions, not regional gradients, drive demographic variation of giant ragweed (Ambrosia trifida) and common sunflower (Helianthus annuus) across northern U.S. Maize Belt. Weed Sci. 2012, 60, 440–450. [Google Scholar] [CrossRef]
- Butler, R.A.; Brouder, S.M.; Johnson, W.G.; Gibson, K.D. Response of four summer annual weed species to mowing frequency and height. Weed Technol. 2013, 27, 798–802. [Google Scholar] [CrossRef]
- Glettner, C.E.; Stoltenberg, D.E. Noncompetitive growth and fecundity of Wisconsin giant ragweed resistance to glyphosate. Weed Sci. 2017, 63, 273–281. [Google Scholar] [CrossRef]
- Davis, A.S.; Taylor, E.C.; Haramoto, E.R.; Renner, K.A. Annual postdispersal weed seed predation in contrasting field environments. Weed Sci. 2013, 61, 296–302. [Google Scholar] [CrossRef]
- Davis, A.S.; Fu, X.; Schutte, B.J.; Berhow, M.A.; Dalling, J.W. Interspecific variation in persistence of buried weed seeds follows trade-offs among physiological, chemical, and physical seed defenses. Ecol. Evol. 2016, 6, 6836–6845. [Google Scholar] [CrossRef]
- Brown, B.; Gallandt, E.R. Evidence of synergy with ‘stacked’ intrarow cultivation tools. Weed Res. 2018, 58, 284–291. [Google Scholar] [CrossRef]
- Westerman, P.R.; Liebman, M.; Menalled, F.D.; Heggenstaller, A.H.; Hartzler, R.G.; Dixon, P.M. Are many little hammers effective? Velvetleaf (Abutilon theophrasti) population dynamics in two- and four-year crop rotation systems. Weed Sci. 2005, 53, 382–392. [Google Scholar] [CrossRef]
- Beckie, H.J.; Johnson, E.N.; Leeson, J.Y.; Shirriff, S.W.; Kapiniak, A. Selection and evolution of acetyl-CoA carboxylase (ACC)-inhibitor resistance in wild oat (Avena fatua L.) in a long-term alternative cropping systems study. Can. J. Plant Sci. 2014, 94, 727–731. [Google Scholar] [CrossRef]
- Westerman, P.R.; Liebman, M.; Heggenstaller, A.H.; Forcella, F. Integrating measurements of seed availability and removal to estimate weed seed losses due to predation. Weed Sci. 2006, 54, 566–574. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liebman, M.; Nichols, V.A. Cropping System Redesign for Improved Weed Management: A Modeling Approach Illustrated with Giant Ragweed (Ambrosia trifida). Agronomy 2020, 10, 262. https://doi.org/10.3390/agronomy10020262
Liebman M, Nichols VA. Cropping System Redesign for Improved Weed Management: A Modeling Approach Illustrated with Giant Ragweed (Ambrosia trifida). Agronomy. 2020; 10(2):262. https://doi.org/10.3390/agronomy10020262
Chicago/Turabian StyleLiebman, Matt, and Virginia A. Nichols. 2020. "Cropping System Redesign for Improved Weed Management: A Modeling Approach Illustrated with Giant Ragweed (Ambrosia trifida)" Agronomy 10, no. 2: 262. https://doi.org/10.3390/agronomy10020262
APA StyleLiebman, M., & Nichols, V. A. (2020). Cropping System Redesign for Improved Weed Management: A Modeling Approach Illustrated with Giant Ragweed (Ambrosia trifida). Agronomy, 10(2), 262. https://doi.org/10.3390/agronomy10020262




