Anther Extrusion and Its Association with Fusarium Head Blight in CIMMYT Wheat Germplasm
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Field Trials and Phenotyping
2.3. Statistical Analyses
2.4. Genotyping
2.5. Linkage and QTL Mapping
3. Results
3.1. Phenotypic Analysis
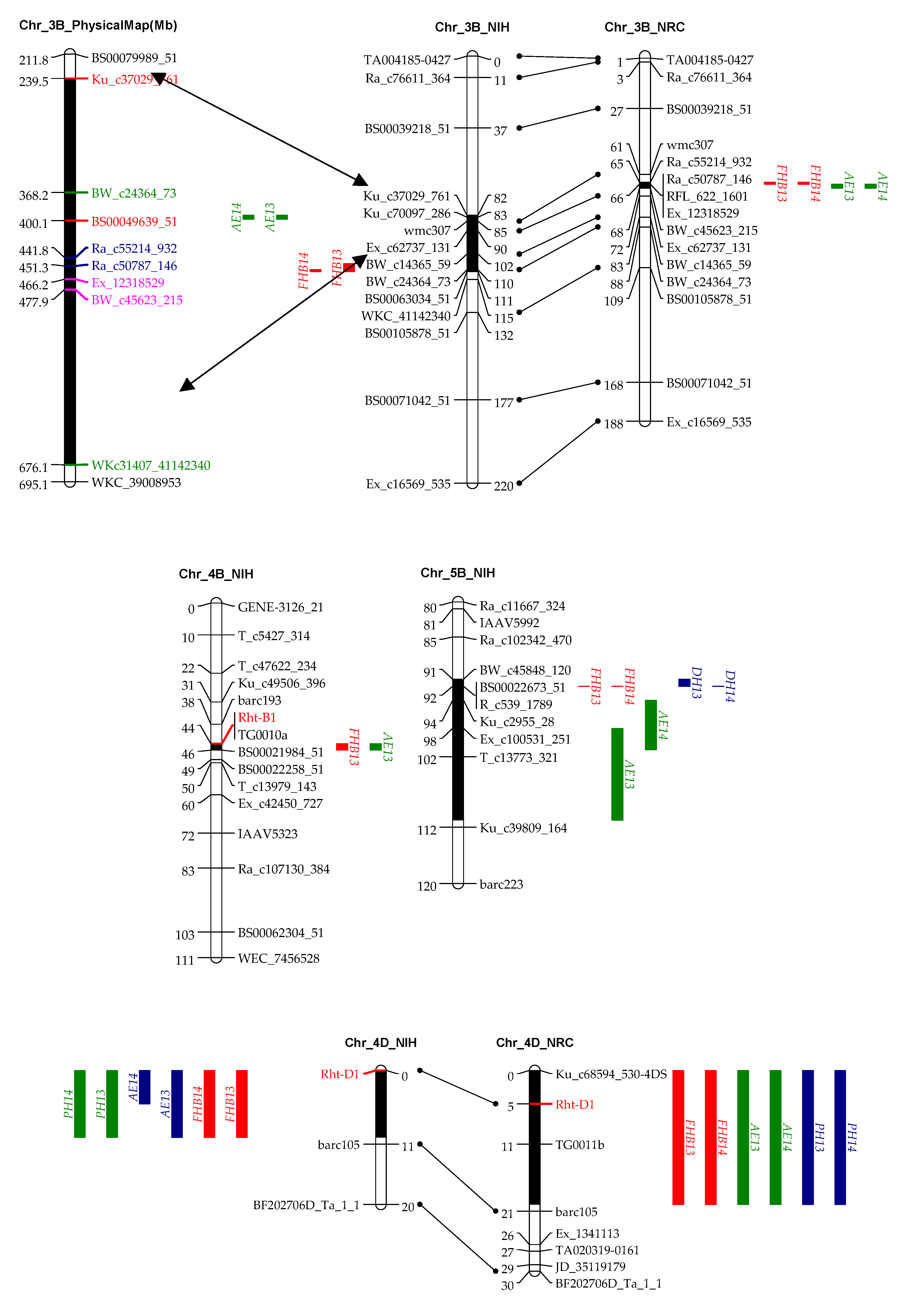
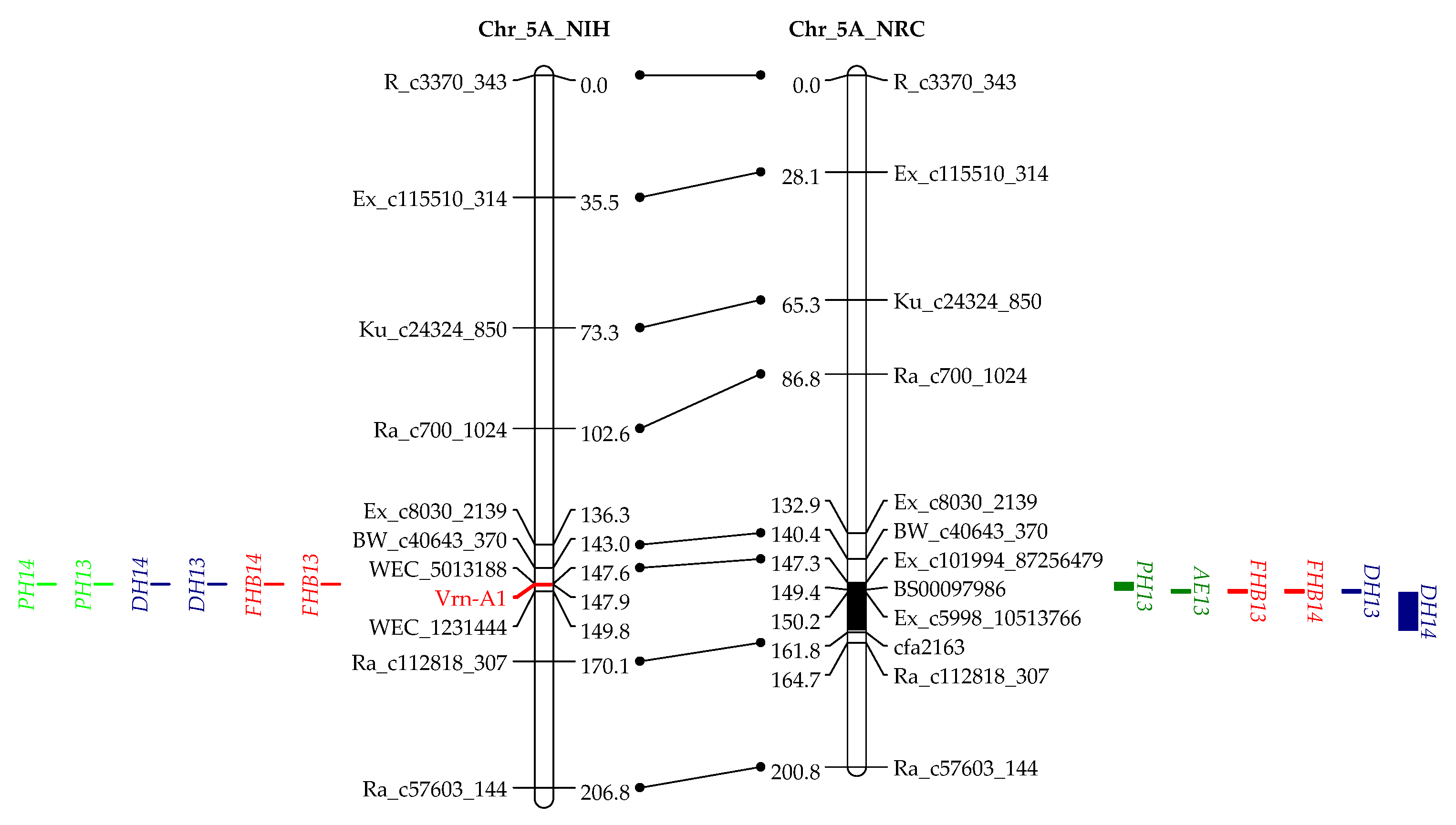
3.2. Genotyping and Linkage Analysis
3.3. QTL Mapping for AE
3.4. QTL Mapping for FHB Resistance
4. Discussion
4.1. AE as a Morphological Marker for FHB
4.2. Rht-B1 and Rht-D1 with Major Phenotypic Effects on Both AE and FHB
4.3. QTL Associated with AE, DH, and FHB
4.4. QTL on 3BL Was Also Important for AE and FHB Resistance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okada, T.; Jayasinghe, J.E.A.R.M.; Eckermann, P.; Watson-Haigh, N.S.; Warner, P.; Hendrikse, Y.; Baes, M.; Tucker, E.J.; Laga, H.; Kato, K.; et al. Effects of Rht-B1 and Ppd-D1 loci on pollinator traits in wheat. Theor. Appl. Genet. 2019, 132, 1965–1979. [Google Scholar] [CrossRef]
- McMullen, M.; Bergstrom, G.; De Wolf, E.; Dill-Macky, R.; Hershman, D.; Shaner, G.; Van Sanford, D. A unified effort to fight an enemy of wheat and barley: Fusarium Head Blight. Plant Dis. 2012, 96, 1712–1728. [Google Scholar] [CrossRef] [PubMed]
- Mesterhazy, A. Types and components of resistance to Fusarium head blight of wheat. Plant Breed. 1995, 114, 377–386. [Google Scholar] [CrossRef]
- Skinnes, H.; Semagn, K.; Tarkegne, Y.; Marøy, A.G.; Bjørnstad, Å. The inheritance of anther extrusion in hexaploid wheat and its relationship to Fusarium head blight resistance and deoxynivalenol content. Plant Breed. 2010, 129, 149–155. [Google Scholar] [CrossRef]
- Lu, Q.; Lillemo, M.; Skinnes, H.; He, X.; Shi, J.; Ji, F.; Dong, Y.; Bjornstad, A. Anther extrusion and plant height are associated with Type I resistance to Fusarium head blight in bread wheat line ‘Shanghai-3/Catbird’. Theor. Appl. Genet. 2013, 126, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Wei, Y.; Cao, W.; Lan, X.; Yu, M.; Chen, Z.; Chen, G.; Zheng, Y. Confirmation of the relationship between plant height and Fusarium head blight resistance in wheat (Triticum aestivum L.) by QTL meta-analysis. Euphytica 2010, 174, 343–356. [Google Scholar] [CrossRef]
- Muqaddasi, Q.H.; Brassac, J.; Börner, A.; Pillen, K.; Röder, M.S. Genetic Architecture of Anther Extrusion in Spring and Winter Wheat. Front. Plant Sci. 2017, 8, 754. [Google Scholar] [CrossRef] [PubMed]
- Muqaddasi, Q.H.; Jayakodi, M.; Börner, A.; Röder, M.S. Identification of consistent QTL with large effect on anther extrusion in doubled haploid populations developed from spring wheat accessions in German Federal ex situ Genebank. Theor. Appl. Genet. 2019, 132, 3035–3045. [Google Scholar] [CrossRef]
- He, X.; Singh, P.K.; Dreisigacker, S.; Singh, S.; Lillemo, M.; Duveiller, E. Dwarfing genes Rht-B1b and Rht-D1b are associated with both type I FHB susceptibility and low anther extrusion in two bread wheat populations. PLoS ONE 2016, 11, e0162499. [Google Scholar] [CrossRef]
- Strange, R.; Deramo, A.; Smith, H. Virulence enhancement of Fusarium graminearum by choline and betaine and of Botrytis cinerea by other constituents of wheat germ. Trans. Br. Mycol. Soc. 1978, 70, 201–207. [Google Scholar] [CrossRef]
- Kubo, K.; Fujita, M.; Kawada, N.; Nakajima, T.; Nakamura, K.; Maejima, H.; Ushiyama, T.; Hatta, K.; Matsunaka, H. Minor differences in anther extrusion affect resistance to Fusarium head blight in wheat. J. Phytopathol. 2013, 161, 308–314. [Google Scholar] [CrossRef]
- Strange, R.N.; Smith, H. A fungal growth stimulant in anthers which predisposes wheat to attack by Fusarium graminearum. Physiol. Plant Pathol. 1971, 1, 141–150. [Google Scholar] [CrossRef]
- Strange, R.N.; Smith, H.; Majer, J.R. Choline, one of two fungal growth stimulants in anthers responsible for the susceptibility of wheat to fusarium graminearum. Nature 1972, 238, 103–104. [Google Scholar] [CrossRef]
- Pearce, R.B.; Strange, R.N.; Smith, H. Glycinebetaine and choline in wheat: Distribution and relation to infection by Fusarium graminearum. Phytochemistry 1976, 15, 953–954. [Google Scholar] [CrossRef]
- Muqaddasi, Q.H.; Pillen, K.; Plieske, J.; Ganal, M.W.; Röder, M.S. Genetic and physical mapping of anther extrusion in elite European winter wheat. PLoS ONE 2017, 12, e0187744. [Google Scholar] [CrossRef]
- Buerstmayr, H.; Ban, T.; Anderson, J.A. QTL mapping and marker-assisted selection for Fusarium head blight resistance in wheat: A review. Plant Breed. 2009, 128, 1–26. [Google Scholar] [CrossRef]
- Muqaddasi, Q.H.; Reif, J.C.; Roder, M.S.; Basnet, B.R.; Dreisigacker, S. Genetic Mapping Reveals Large-Effect QTL for Anther Extrusion in CIMMYT Spring Wheat. Agronomy 2019, 9, 407. [Google Scholar] [CrossRef]
- De Vries, A.P. Flowering biology of wheat, particularly in view of hybrid seed production—A review. Euphytica 1971, 20, 152–170. [Google Scholar] [CrossRef]
- Couture, L. Receptivite de cultivars de cereales de printemps a la contamination des graines sur inflorescence par les Fusarium spp. Can. J. Plant Sci. 1982, 62, 29–34. [Google Scholar] [CrossRef]
- Zhu, H.; Gilchrist, L.; Hayes, P.; Kleinhofs, A.; Kudrna, D.; Liu, Z.; Prom, L.; Steffenson, B.; Toojinda, T.; Vivar, H. Does function follow form? Principal QTLs for Fusarium head blight (FHB) resistance are coincident with QTLs for inflorescence traits and plant height in a doubled-haploid population of barley. Theor. Appl. Genet. 1999, 99, 1221–1232. [Google Scholar] [CrossRef]
- He, X.; Skinnes, H.; Oliver, R.E.; Jackson, E.W.; Bjørnstad, Å. Linkage mapping and identification of QTL affecting deoxynivalenol (DON) content (Fusarium resistance) in oats (Avena sativa L.). Theor. Appl. Genet. 2013, 126, 2655–2670. [Google Scholar] [CrossRef]
- Emrich, K.; Wilde, F.; Miedaner, T.; Piepho, H.P. REML approach for adjusting the Fusarium head blight rating to a phenological date in inoculated selection experiments of wheat. Theor. Appl. Genet. 2008, 117, 65–73. [Google Scholar] [CrossRef]
- He, X.; Dreisigacker, S.; Singh, R.P.; Singh, P.K. Genetics for low correlation between Fusarium head blight disease and deoxynivalenol (DON) content in a bread wheat mapping population. Theor. Appl. Genet. 2019, 132, 2401–2411. [Google Scholar] [CrossRef]
- He, X.; Singh, P.K.; Duveiller, E.; Schlang, N.; Dreisigacker, S.; Singh, R.P. Identification and characterization of international Fusarium head blight screening nurseries of wheat at CIMMYT, Mexico. Eur. J. Plant Pathol. 2013, 136, 123–134. [Google Scholar] [CrossRef]
- Dreisigacker, S.; Sehgal, D.; Reyes-Jaimez, A.; Luna-Garrido, B.; Muñoz-Zavala, S.; Núñez-Ríos, C.; Mollins, J.; Mall, S. CIMMYT Wheat Molecular Genetics: Laboratory Protocols and Applications to Wheat Breeding; CIMMYT: Texcoco, Mexico, 2016. [Google Scholar]
- Dreisigacker, S.; Wang, X.; Cisneros, B.A.M.; Jing, R.L.; Singh, P.K. Adult-plant resistance to Septoria tritici blotch in hexaploid spring wheat. Theor. Appl. Genet. 2015, 128, 2317–2329. [Google Scholar] [CrossRef]
- Wang, S.; Wong, D.; Forrest, K.; Allen, A.; Chao, S.; Huang, B.E.; Maccaferri, M.; Salvi, S.; Milner, S.G.; Cattivelli, L. Characterization of polyploid wheat genomic diversity using a high-density 90 000 single nucleotide polymorphism array. Plant Biotechnol. J. 2014, 12, 787–796. [Google Scholar] [CrossRef]
- Ooijen, J.W.V. MapQTL® 6, Software for the Mapping of Quantitative Trait Loci in Experimental Populations of Diploid Species; Kyazma BV: Wageningen, The Netherlands, 2009. [Google Scholar]
- Muqaddasi, Q.H.; Reif, J.C.; Li, Z.; Basnet, B.R.; Dreisigacker, S.; Röder, M.S. Genome-wide association mapping and genome-wide prediction of anther extrusion in CIMMYT spring wheat. Euphytica 2017, 213, 73. [Google Scholar] [CrossRef]
- Graham, S.; Browne, R.A. Anther extrusion and Fusarium head blight resistance in European wheat. J. Phytopathol. 2009, 157, 580–582. [Google Scholar] [CrossRef]
- Pugh, G.W.; Johann, H.; Dickson, J. Factors affecting infection of wheat heads by Gibberella saubiuetii. J. Agric. Res. 1933, 46, 771–797. [Google Scholar]
- Buerstmayr, M.; Buerstmayr, H. Comparative mapping of quantitative trait loci for Fusarium head blight resistance and anther retention in the winter wheat population Capo x Arina. Theor. Appl. Genet. 2015, 128, 1519–1530. [Google Scholar] [CrossRef]
- He, X.; Lillemo, M.; Shi, J.; Wu, J.; Bjørnstad, Å.; Belova, T.; Dreisigacker, S.; Duveiller, E.; Singh, P. QTL characterization of Fusarium head blight resistance in CIMMYT bread wheat line Soru# 1. PLoS ONE 2016, 11, e0158052. [Google Scholar]
- Steiner, B.; Buerstmayr, M.; Wagner, C.; Danler, A.; Eshonkulov, B.; Ehn, M.; Buerstmayr, H. Fine-mapping of the Fusarium head blight resistance QTL Qfhs.ifa-5A identifies two resistance QTL associated with anther extrusion. Theor. Appl. Genet. 2019. [Google Scholar] [CrossRef]
- Zhang, Q. Preliminary study on identification method of wheat resistant to scab in breeding process. Seed 1982, 2, 19–22. [Google Scholar] [CrossRef]
- Reitan, L.; Skinnes, H. Breeding strategies for resistance to Fusarium in graminor cereal breeding. In Bioforsk FOKUS; Bioforsk: Aas, Norway, 2010; Volume 5, p. 33. [Google Scholar]
- Taylor, M. Incorporation of Fusarium head blight resistance into European winter wheat breeding programmes. In Proceedings of the 2nd International Symposium on Fusarium Head Blight: Incorporating the 8th European Fusarium Seminar, Orlando, FL, USA, 11–15 Decmber 2004; Canty, S.M., Boring, T., Wardwell, J., Ward, R.W., Eds.; Michigan State University: East Lansing, MI, USA, 2004; pp. 195–196. [Google Scholar]
- Gosman, N.; Steed, A.; Hollins, T.W.; Bayles, R.; Jennings, P.; Nicholson, P. Semi-dwarfing Rht-B1 and Rht-D1 loci of wheat differ significantly in their influence on resistance to Fusarium head blight. Theor. Appl. Genet. 2009, 118, 695–702. [Google Scholar] [CrossRef]
- Peng, J.; Richards, D.E.; Hartley, N.M.; Murphy, G.P.; Devos, K.M.; Flintham, J.E.; Beales, J.; Fish, L.J.; Worland, A.J.; Pelica, F. ‘Green revolution’genes encode mutant gibberellin response modulators. Nature 1999, 400, 256–261. [Google Scholar] [CrossRef]
- Pearce, S.; Saville, R.; Vaughan, S.P.; Chandler, P.M.; Wilhelm, E.P.; Sparks, C.A.; Al-Kaff, N.; Korolev, A.; Boulton, M.I.; Phillips, A.L. Molecular characterization of Rht-1 dwarfing genes in hexaploid wheat. Plant Physiol. 2011, 157, 1820–1831. [Google Scholar] [CrossRef]
- Mutasa-Göttgens, E.; Hedden, P.H. Gibberellin as a factor in floral regulatory networks. J. Exp. Bot. 2009, 60, 1979–1989. [Google Scholar] [CrossRef]
- Chhun, T.; Aya, K.; Asano, K.; Yamamoto, E.; Morinaka, Y.; Watanabe, M.; Kitano, H.; Ashikari, M.; Matsuoka, M.; Ueguchi-Tanaka, M. Gibberellin regulates pollen viability and pollen tube growth in rice. Plant Cell 2007, 19, 3876–3888. [Google Scholar] [CrossRef]
- Youssef, H.M.; Eggert, K.; Koppolu, R.; Alqudah, A.M.; Poursarebani, N.; Fazeli, A.; Sakuma, S.; Tagiri, A.; Rutten, T.; Govind, G. VRS2 regulates hormone-mediated inflorescence patterning in barley. Nat. Genet. 2017, 49, 157–161. [Google Scholar] [CrossRef]
- Herter, C.P.; Ebmeyer, E.; Kollers, S.; Korzun, V.; Leiser, W.L.; Wurschum, T.; Miedaner, T. Rht24 reduces height in the winter wheat population ‘Solitar x Bussard’ without adverse effects on Fusarium head blight infection. Theor. Appl. Genet. 2018, 131, 1263–1272. [Google Scholar] [CrossRef]
- Sutka, J.; Galiba, G.; Vagujfalvi, A.; Gill, B.S.; Snape, J.W. Physical mapping of the Vrn-A1 and Fr1 genes on chromosome 5A of wheat using deletion lines. Theor. Appl. Genet. 1999, 99, 199–202. [Google Scholar] [CrossRef]
- Yan, L.; Loukoianov, A.; Blechl, A.; Tranquilli, G.; Ramakrishna, W.; SanMiguel, P.; Bennetzen, J.L.; Echenique, V.; Dubcovsky, J. The wheat VRN2 gene is a flowering repressor down-regulated by vernalization. Science 2004, 303, 1640–1644. [Google Scholar] [CrossRef]
- Li, C.; Lin, H.; Chen, A.; Lau, M.; Jernstedt, J.; Dubcovsky, J.J.b. Wheat VRN1 and FUL2 play critical and redundant roles in spikelet meristem identity and spike determinacy. BioRxiv 2019, 146, dev175398. [Google Scholar]

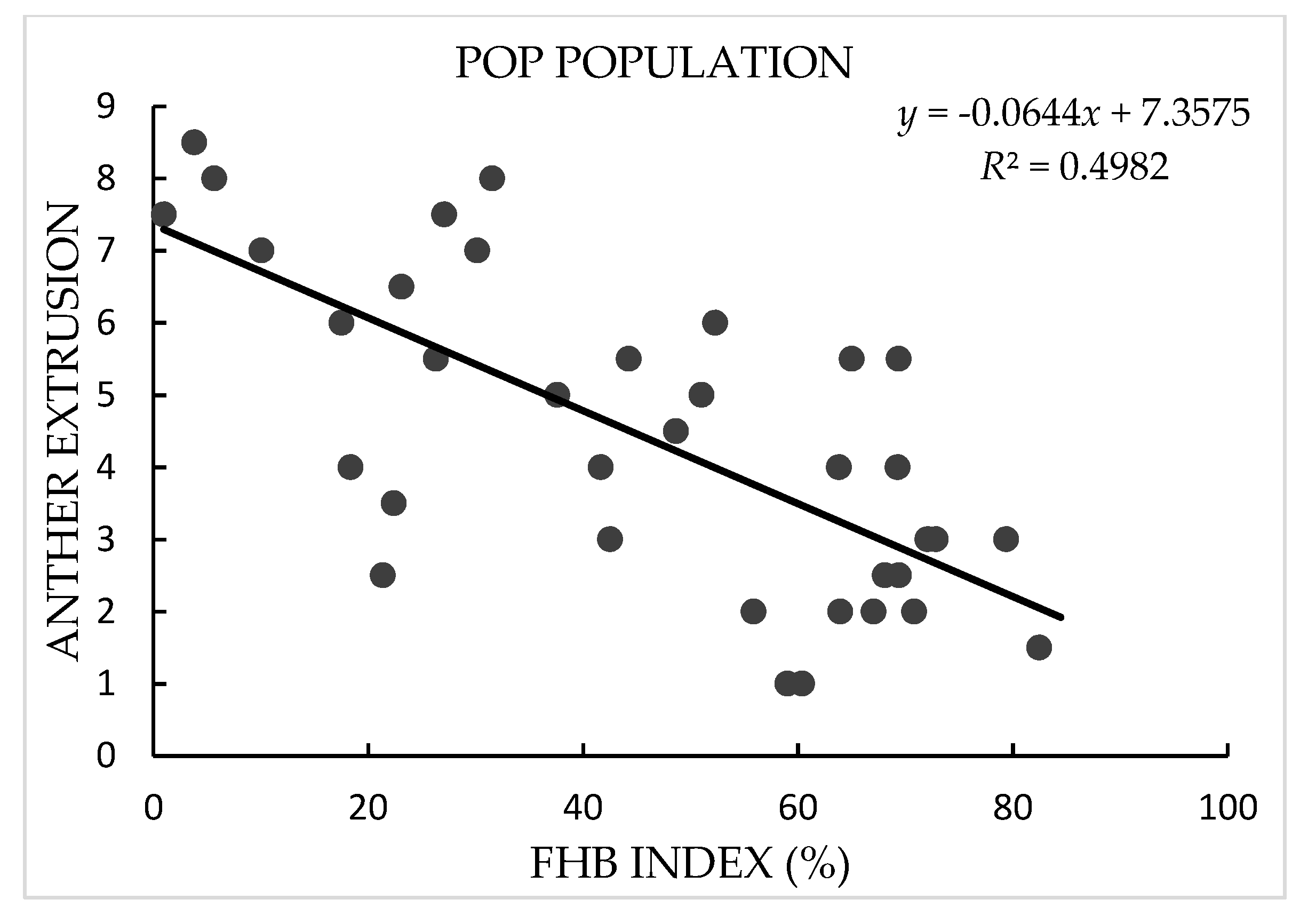
| Population | Source | DF | Mean Square | F-Value | p-Value | Heritability |
|---|---|---|---|---|---|---|
| NIH | Genotype | 196 | 14.97 | 9.66 | <0.0001 | 0.83 |
| Year | 1 | 324.92 | 209.67 | <0.0001 | ||
| Genotype × year | 196 | 2.52 | 1.63 | <0.0001 | ||
| Rep (year) | 2 | 23.27 | 15.02 | <0.0001 | ||
| Error | 392 | 1.55 | ||||
| NRC | Genotype | 184 | 14.07 | 6.82 | <0.0001 | 0.74 |
| Year | 1 | 231.62 | 112.28 | <0.0001 | ||
| Genotype × year | 184 | 3.61 | 1.75 | <0.0001 | ||
| Rep (year) | 2 | 26.43 | 12.81 | <0.0001 | ||
| Error | 368 | 2.06 | ||||
| FHBSN | Genotype | 35 | 4.67 | 5.67 | <0.0001 | 0.82 |
| Rep | 1 | 1.68 | 2.04 | 0.1620 | ||
| Error | 35 | 0.82 | ||||
| POP | Genotype | 35 | 9.44 | 7.04 | <0.0001 | 0.86 |
| Rep | 1 | 0.06 | 0.04 | 0.8399 | ||
| Error | 35 | 1.34 |
| Population | Source | DF | Mean Square | F-Value | p-Value | Heritability |
|---|---|---|---|---|---|---|
| NIH | Genotype | 196 | 1734.59 | 18.2 | <0.0001 | 0.92 |
| Year | 1 | 297.47 | 3.12 | 0.0781 | ||
| Genotype × year | 195 | 136.17 | 1.43 | 0.0016 | ||
| Rep (year) | 2 | 1173.78 | 12.32 | <0.0001 | ||
| Error | 390 | 95.308 | ||||
| NRC | Genotype | 184 | 1927.59 | 19.33 | <0.0001 | 0.74 |
| Year | 1 | 6655.90 | 66.73 | <0.0001 | ||
| Genotype × year | 184 | 279.66 | 2.80 | <0.0001 | ||
| Rep (year) | 2 | 699.51 | 7.01 | 0.001 | ||
| Error | 362 | 99.74 | ||||
| FHBSN | Genotype | 35 | 504.16 | 10.79 | <0.0001 | 0.91 |
| Rep | 1 | 27.90 | 0.60 | 0.4448 | ||
| Error | 35 | 46.72 | ||||
| POP | Genotype | 35 | 1135.29 | 13.52 | <0.0001 | 0.93 |
| Rep | 1 | 1454.45 | 17.32 | 0.0002 | ||
| Error | 35 | 83.99 |
| Population | Chr. | Position | Left Marker | Right Marker | 2013 | 2014 | Source b | Traits Associated c | ||
|---|---|---|---|---|---|---|---|---|---|---|
| LOD | PVE (%) a | LOD | PVE (%) | |||||||
| NIH | 1BS | 46.9–54.1 | wmc406 | barc8 | 2.2 | 3 | I | |||
| 3BL | 82.1–84.1 | Ku_c37029_761 | BS00049639_51 | 3.3 | 4.5 | 5.5 | 8.8 | I | FHB | |
| 4BS | 44.1–46.3 | Rht-B1 | BS00021984_51 | 4.0 | 6.1 | N | FHB, PH | |||
| 4DS | 0–10.7 | Rht-D1 | barc105 | 10.6 | 16.1 | 3.0 | 4.6 | I | FHB, PH | |
| 5BL | 98.4–111.8 | Ex_c100531_251 | Ku_c39809_164 | 4.4 | 6.2 | 4.3 | 6.8 | N | FHB, DH | |
| 6BS | 65.5–66.0 | WEC_3267186 | Ku_c9507_142 | 3.4 | 5.2 | I | ||||
| 7Ac | 118.7–121.7 | BS00006674_51 | BW_c66165_77 | 2.8 | 3.8 | 4 | 6.8 | N | ||
| NRC | 3BL | 66.0–67.9 | RFL_622_1601 | BW_c45623_215 | 5.0 | 6.9 | 2.1 | 3.1 | R | FHB |
| 4AL | 78.4–79.4 | Ex_c8658_335 | Ex_9657856 | 3.4 | 4.7 | N | ||||
| 4DS | 0.0–21.3 | Ku_c68594_530 | barc105 | 12.6 | 19.7 | 6.0 | 11.8 | R | FHB, PH | |
| 5AL | 149.4–150.2 | BS00097986 | Ex_c5998_10513766 | 4.1 | 5.6 | R | FHB, PH, DH | |||
| 6AS | 18.7–19.3 | IACX2250 | BS00037006_51 | 4.4 | 6.1 | 2.6 | 4.8 | N | ||
| 7BS | 46.2–53.3 | Ku_c101472_574 | GENE-4333_211 | 2.1 | 3.9 | R | DH | |||
| Population | Chr. | Position (cM) | Left Marker | Right Marker | 2013 | 2014 | R Source b | Traits Associated c | ||
|---|---|---|---|---|---|---|---|---|---|---|
| LOD | PVE (%) a | LOD | PVE (%) | |||||||
| 3BL | 109.6–115.5 | BW_c24364_73 | wKc31407_41142340 | 4.0 | 4.8 | 3.0 | 4.4 | I | AE | |
| 4BS | 44.1–46.3 | Rht-B1 | BS00021984_51 | 4.2 | 5.5 | N | AE, PH | |||
| NIH | 4DS | 0–10.7 | Rht-D1 | barc105 | 10.1 | 12.7 | 6.0 | 9.2 | I | AE, PH |
| 5AL | 147.6–147.9 | WEC_5013188 | Vrn-A1 | 15.8 | 21.2 | 13.2 | 21.7 | I | AE, DH, PH | |
| 5BL | 92.1–92.4 | BS00022673_51 | R_c539_1789 | 4.1 | 4.8 | 2.3 | 3.4 | N | AE, DH | |
| 2AL | 91.3–96.9 | BW_c4743_63 | Ra_c38018_278 | 3.1 | 4.2 | N | ||||
| 2DS | 37.3–55.6 | BS00022276_51 | Ex_c39215_100 | 4.6 | 5.8 | 2.3 | 3.8 | N | DH, PH | |
| NRC | 3BL | 64.9–65.6 | Ra_c55214_932 | Ra_c50787_146 | 3.1 | 3.8 | 2.6 | 3.6 | R | AE |
| 4AL | 129.3–136.8 | IAAV1383 | Ra_32274079 | 3.2 | 3.9 | 2.2 | 3 | N | ||
| 4DS | 0.0–21.3 | Ku_c68594_530 | barc105 | 7.7 | 10.2 | 9.4 | 14.4 | R | AE, PH | |
| 5AL | 149.4–164.7 | BS00097986 | Ra_c112818_307 | 13.4 | 18.7 | 6.4 | 9.2 | R | AE, DH, PH | |
| 7AS | 54.9–61.6 | Ex_c109881_701 | BS00023225_51 | 6.1 | 9.7 | 2.9 | 4.7 | R | DH, PH | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, K.; He, X.; Dreisigacker, S.; He, Z.; Singh, P.K. Anther Extrusion and Its Association with Fusarium Head Blight in CIMMYT Wheat Germplasm. Agronomy 2020, 10, 47. https://doi.org/10.3390/agronomy10010047
Xu K, He X, Dreisigacker S, He Z, Singh PK. Anther Extrusion and Its Association with Fusarium Head Blight in CIMMYT Wheat Germplasm. Agronomy. 2020; 10(1):47. https://doi.org/10.3390/agronomy10010047
Chicago/Turabian StyleXu, Kaijie, Xinyao He, Susanne Dreisigacker, Zhonghu He, and Pawan K. Singh. 2020. "Anther Extrusion and Its Association with Fusarium Head Blight in CIMMYT Wheat Germplasm" Agronomy 10, no. 1: 47. https://doi.org/10.3390/agronomy10010047
APA StyleXu, K., He, X., Dreisigacker, S., He, Z., & Singh, P. K. (2020). Anther Extrusion and Its Association with Fusarium Head Blight in CIMMYT Wheat Germplasm. Agronomy, 10(1), 47. https://doi.org/10.3390/agronomy10010047






