Controlled Morphing of Microbubbles to Beaded Nanofibers via Electrically Forced Thin Film Stretching
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
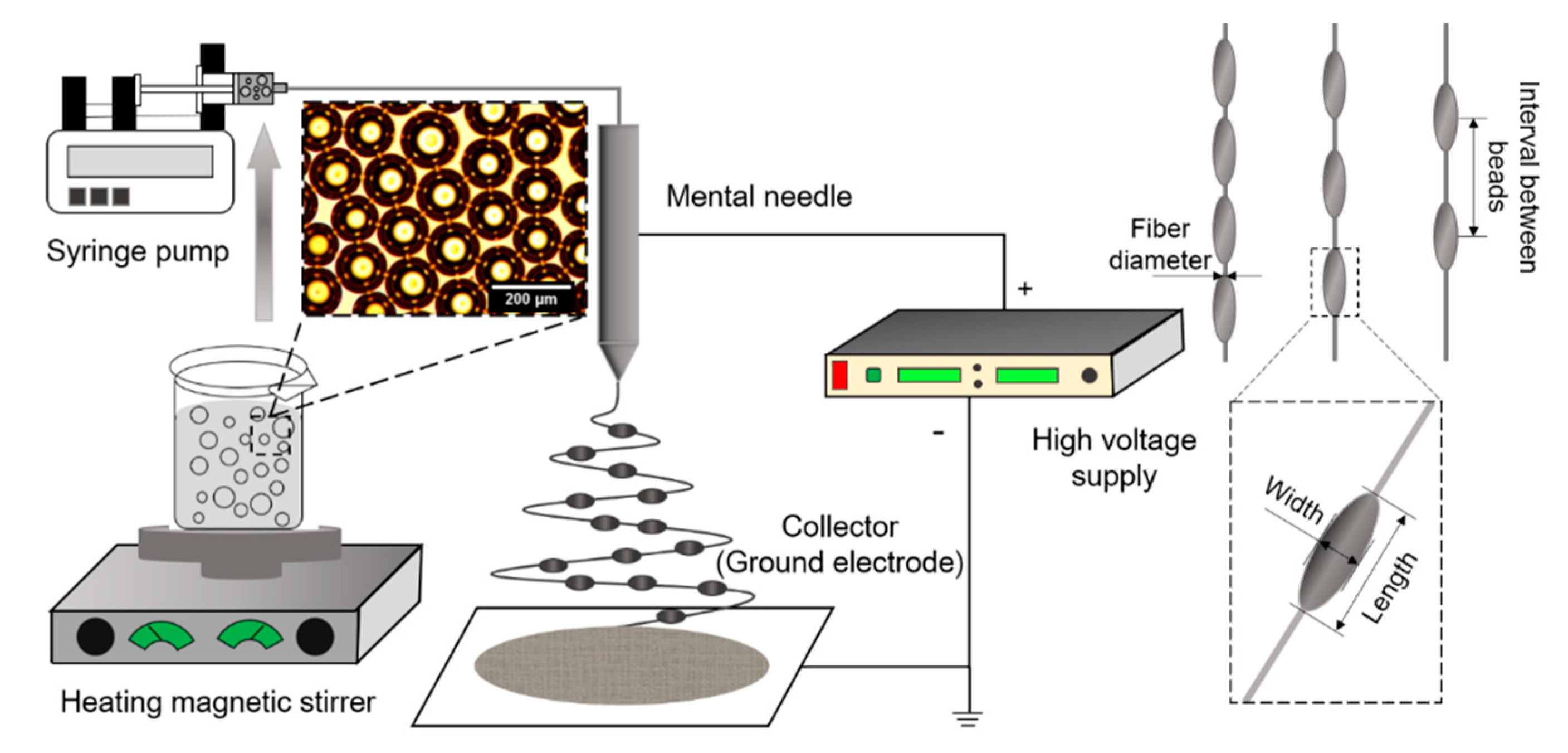
2.2.1. Beads-On Nanofiber Preparation
2.2.2. Beaded Nanofiber Characterization
2.2.3. In Vitro Drug Release Assessment
2.3. Statistical Analysis
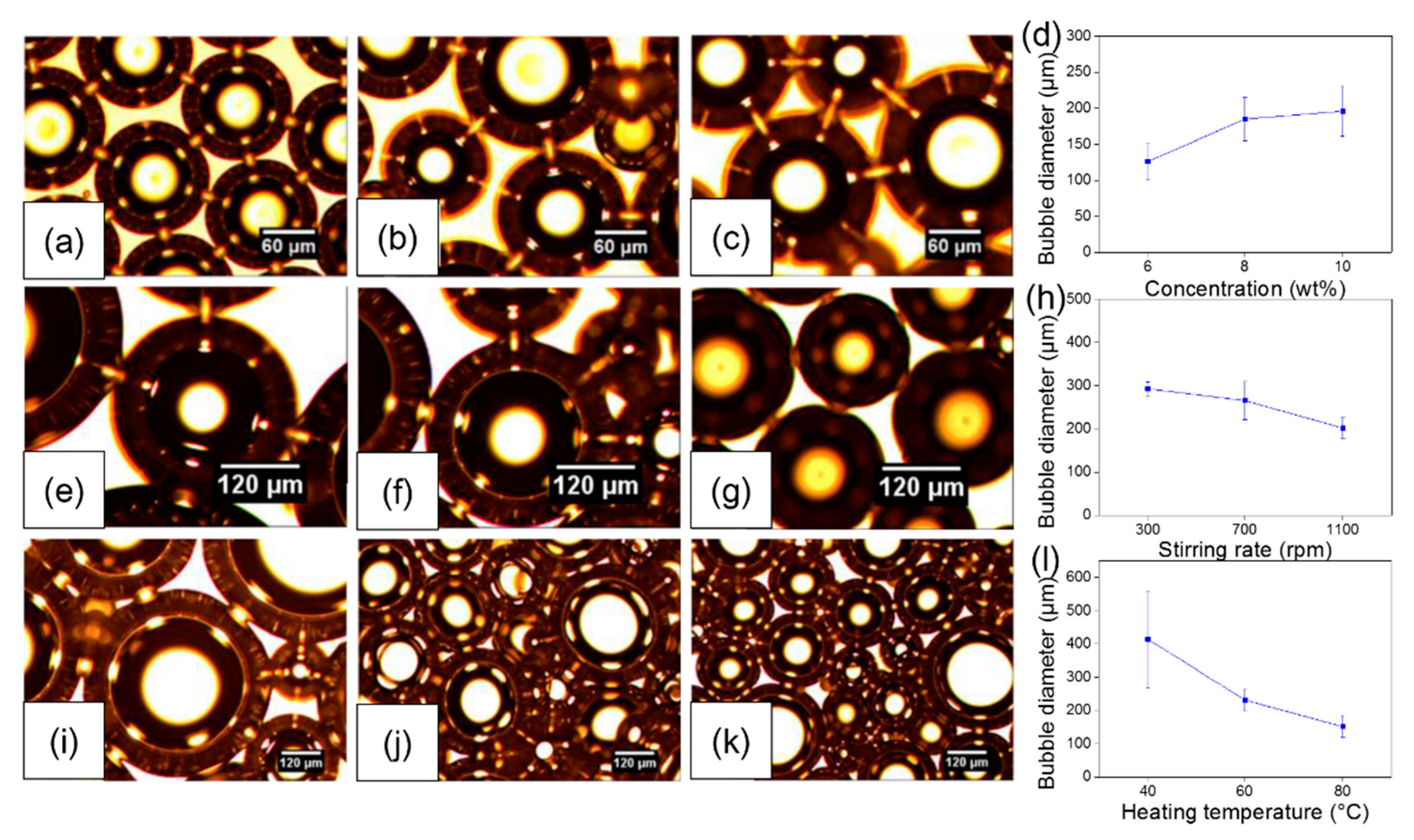
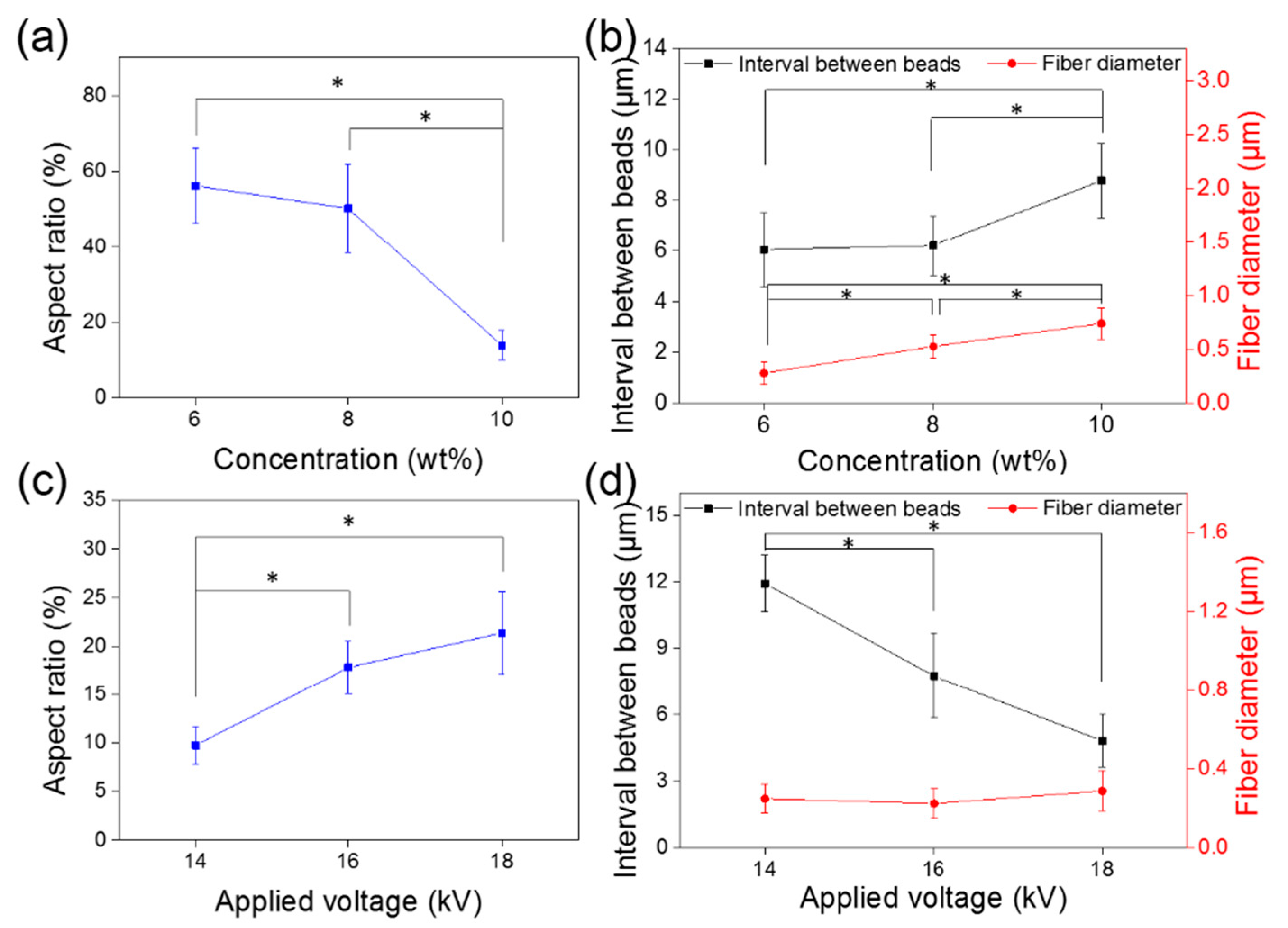
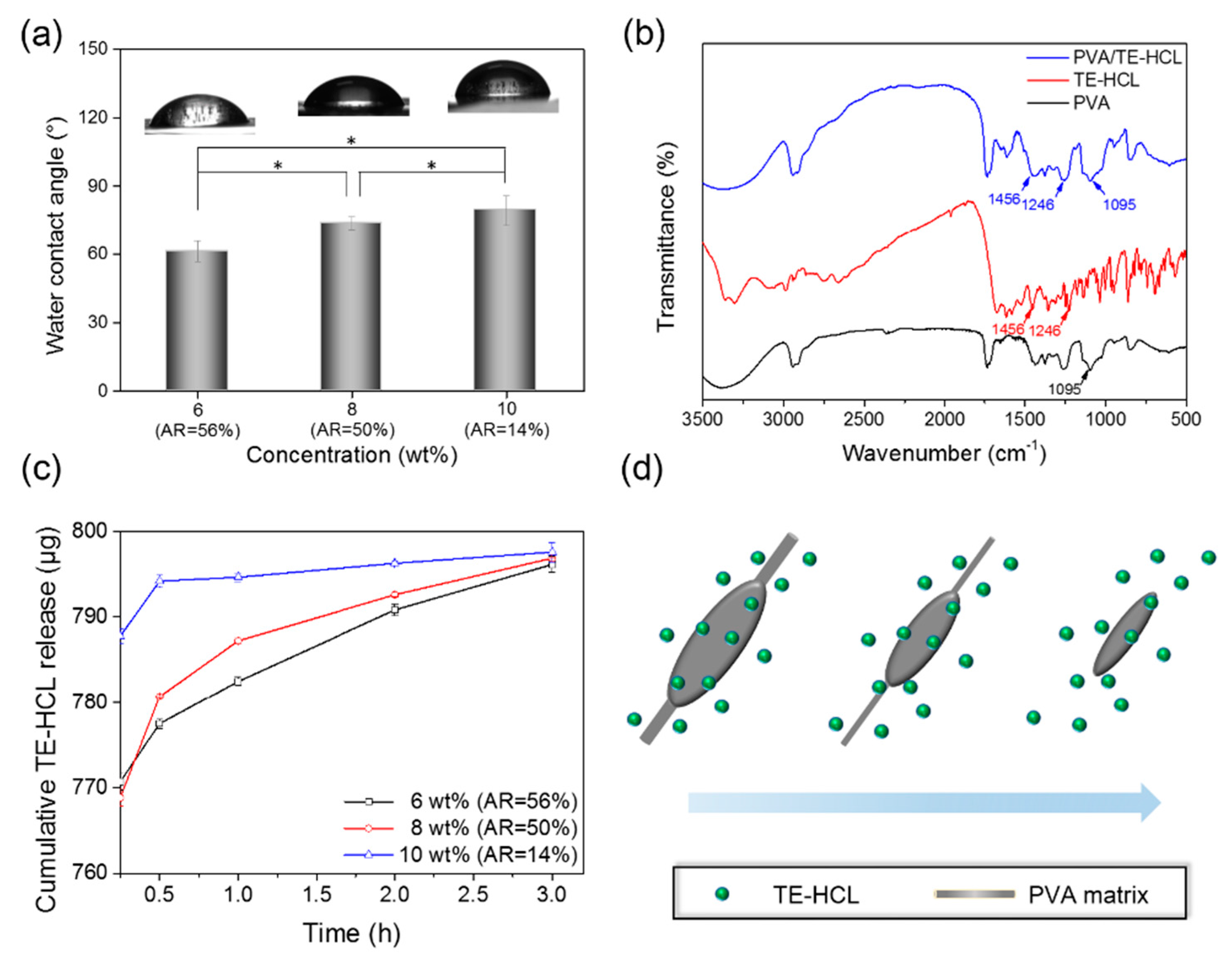
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Forward, K.M.; Flores, A.; Rutledge, G.C. Production of core/shell fibers by electrospinning from a free surface. Chem. Eng. Sci. 2013, 104, 250–259. [Google Scholar] [CrossRef]
- Kataria, K.; Gupta, A.; Rath, G.; Mathur, R.B.; Dhakate, S.R. In vivo wound healing performance of drug loaded electrospun composite nanofibers transdermal patch. Int. J. Pharm. 2014, 469, 102–110. [Google Scholar] [CrossRef]
- Yao, Z.C.; Chang, M.W.; Ahmad, Z.; Li, J.S. Encapsulation of rose hip seed oil into fibrous zein films for ambient and on demand food preservation via coaxial electrospinning. J. Food Eng. 2016, 191, 115–123. [Google Scholar] [CrossRef]
- Jiang, T.; Carbone, E.J.; Lo, W.H.; Laurencin, C.T. Electrospinning of polymer nanofibers for tissue regeneration. Prog. Polym. Sci. 2015, 46, 1–24. [Google Scholar] [CrossRef]
- Lu, W.; Sun, J.; Jiang, X. Recent advances in electrospinning technology and biomedical applications of electrospun fibers. J. Mater. Chem. B 2014, 2, 2369–2380. [Google Scholar] [CrossRef]
- Wang, B.; Zheng, H.; Chang, M.W.; Ahmad, Z.; Li, J.S. Hollow polycaprolactone composite fibers for controlled magnetic responsive antifungal drug release. Colloids Surf. B 2016, 145, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Chang, M.W.; Li, Y.; Qi, Y.; Wu, J.; Ahmad, Z.; Li, J.S. Janus particle synthesis via aligned non-concentric angular nozzles and electrohydrodynamic co-flow for tunable drug release. RSC Adv. 2016, 6, 77174–77178. [Google Scholar] [CrossRef]
- Zhang, C.; Gao, C.; Chang, M.W.; Ahmad, Z.; Li, J.S. Continuous micron-scaled rope engineering using a rotating multi-nozzle electrospinning emitter. Appl. Phys. Lett. 2016, 109, 151903. [Google Scholar] [CrossRef]
- Li, T.; Xin, D.; Tian, L.; Hu, J.; Yang, X.; Ramakrishna, S. The control of beads diameter of bead-on-string electrospun nanofibers and the corresponding release behaviors of embedded drugs. Mater. Sci. Eng. C 2017, 74, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Cipitria, A. Design, fabrication and characterization of PCL electrospun scaffolds—A review. J. Mater. Chem. 2011, 21, 9419–9453. [Google Scholar] [CrossRef]
- Avelina, F.; Sergio, T.G.; Josemaria, L. Novel route to stabilization of bioactive antioxidants by encapsulation in electrospun fibers of zein prolamine. Food Hydrocoll. 2009, 23, 1427–1432. [Google Scholar]
- Qi, H.; Hu, P.; Xu, J.; Wang, A. Encapsulation of Drug Reservoirs in Fibers by Emulsion Electrospinning: Morphology Characterization and Preliminary Release Assessment. Biomacromolecules 2006, 7, 2327–2330. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Greiner, A. On the way to clean and safe electrospinning—Green electrospinning: Emulsion and suspension electrospinning. Polym. Adv. Technol. 2011, 22, 372–378. [Google Scholar] [CrossRef]
- Fuh, Y.K.; Wu, Y.C.; He, Z.Y.; Huang, Z.M.; Hu, W.W. The control of cell orientation using biodegradable alginate fibers fabricated by near-field electrospinning. Mater. Sci. Eng. C 2016, 62, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Sirsi, S.; Borden, M. Microbubble Compositions, Properties and Biomedical Applications. Bubble Sci. Eng. Technol. 2009, 1, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.; Tabata, H.; Sanchez, M.; Cagna, A.; Koyama, D.; Krafft, M.P. Microbubbles with a Self-Assembled Poloxamer Shell and a Fluorocarbon Inner Gas. Langmuir 2016, 32, 12461–12467. [Google Scholar]
- Nguyen, P.; Nikolova, G.; Polavarapu, P.; Waton, G.; Phuoc, L.; Pourroy, G.; Krafft, M. Compressible multi-scale magnetic constructs: Decorating the outer surface of self-assembled microbubbles with iron oxide nanoparticles. RSC Adv. 2013, 3, 7743–7746. [Google Scholar] [CrossRef]
- Zhang, C.; Yuan, X.; Wu, L.; Han, Y.; Sheng, J. Study on morphology of electrospun poly (vinyl alcohol) mats. Eur. Polym. J. 2005, 41, 423–432. [Google Scholar] [CrossRef]
- Yao, Z.C.; Jin, L.J.; Ahmad, Z.; Huang, J.; Chang, M.W.; Li, J.S. Ganoderma lucidum polysaccharide loaded sodium alginate micro-particles prepared via electrospraying in controlled deposition environments. Int. J. Pharm. 2017, 524, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.C.; Chen, S.C.; Ahmad, Z.; Huang, J.; Chang, M.W.; Li, J.S. Essential oil bioactive fibrous membranes prepared via coaxial electrospinning. J. Food Sci. 2017, 82, 1412–1422. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhou, W.; Chang, M.W.; Ahmad, Z.; Li, J.S. Impact of substrate geometry on electrospun fiber deposition and alignment. J. Appl. Polym. Sci. 2017, 134, 44823. [Google Scholar] [CrossRef]
- Wang, J.C.; Chang, M.W.; Ahmad, Z.; Li, J.S. Fabrication of patterned polymer-antibiotic composite fibers via electrohydrodynamic (EHD) printing. J. Drug Deliv. Sci. Technol. 2016, 35, 114–123. [Google Scholar] [CrossRef]
- Gao, Y.; Chang, M.W.; Ahmad, Z.; Li, J.S. Magnetic-responsive microparticles with customized porosity for drug delivery. RSC Adv. 2016, 6, 88157–88167. [Google Scholar] [CrossRef]
- Palangetic, L.; Reddy, N.K.; Srinivasan, S.; Cohen, R.E.; Mckinley, G.H.; Clasen, C. Dispersity and spinnability: Why highly polydisperse polymer solutions are desirable for electrospinning. Polymer 2014, 55, 4920–4931. [Google Scholar] [CrossRef]
- Rieger, K.A.; Birch, N.P.; Schiffman, J.D. Electrospinning chitosan/poly(ethylene oxide) solutions with essential oils: Correlating solution rheology to nanofiber formation. Carbohydr. Polym. 2016, 139, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sano, M.; Zhang, T.A.; Wang, Q.; He, J.C. Intensification of Bubble Disintegration and Dispersion by Mechanical Stirring in Gas Injection Refining. ISIJ Int. 2009, 49, 17–23. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, D.; Chang, M.W.; Ahmad, Z.; Li, J.S. Optimising the shell thickness-to-radius ratio for the fabrication of oil-encapsulated polymeric microspheres. Chem. Eng. J. 2015, 284, 963–971. [Google Scholar] [CrossRef]
- Supponen, O.; Kobel, P.; Obreschkow, D.; Farhat, M. The inner world of a collapsing bubble. Phys. Fluids 2015, 27, 94–98. [Google Scholar] [CrossRef]
- Yang, R.R.; He, J.H.; Xu, L.; Yu, J.Y. Effect of solution concentration on diameter and morphology of PVA nanofibres in bubble electrospinning process. Mater. Sci. Technol. 2010, 26, 1313–1316. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Chahal, S.; Hussain, F.S.J.; Kumar, A.; Yusoff, M.M. Fabrication, characterization and in vitro biocompatibility of electrospun hydroxyethyl cellulose/poly (vinyl) alcohol nanofibrous composite biomaterial for bone tissue engineering. Chem. Eng. Sci. 2016, 144, 17–29. [Google Scholar] [CrossRef]
- Yao, Z.C.; Gao, Y.; Chang, M.W.; Ahmad, Z.; Li, J.S. Regulating poly-caprolactone fiber characteristics in-situ during one-step coaxial electrospinning via enveloping liquids. Mater. Lett. 2016, 183, 202–206. [Google Scholar] [CrossRef]
- Kim, C.H.; Khil, M.S.; Kim, H.Y.; Lee, H.U.; Jahng, K.Y. An improved hydrophilicity via electrospinning for enhanced cell attachment and proliferation. J. Biomed. Mater. Res. Part B 2006, 78, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Jiang, L.; Zhou, Z.; Wu, X.; Wang, Y. Preparation and properties of electrospun soy protein isolate/polyethylene oxide nanofiber membranes. ACS. Appl. Mater. Interfaces 2012, 4, 4331–4337. [Google Scholar] [CrossRef] [PubMed]
- Askari, P.; Zahedi, P.; Rezaeian, I. Three-layered electrospun PVA/PCL/PVA nanofibrous mats containing tetracycline hydrochloride and phenytoin sodium: A case study on sustained control release, antibacterial, and cell culture properties. J. Appl. Polym. Sci. 2015, 133, 43309. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, Z.-C.; Yuan, Q.; Ahmad, Z.; Huang, J.; Li, J.-S.; Chang, M.-W. Controlled Morphing of Microbubbles to Beaded Nanofibers via Electrically Forced Thin Film Stretching. Polymers 2017, 9, 265. https://doi.org/10.3390/polym9070265
Yao Z-C, Yuan Q, Ahmad Z, Huang J, Li J-S, Chang M-W. Controlled Morphing of Microbubbles to Beaded Nanofibers via Electrically Forced Thin Film Stretching. Polymers. 2017; 9(7):265. https://doi.org/10.3390/polym9070265
Chicago/Turabian StyleYao, Zhi-Cheng, Qiantailang Yuan, Zeeshan Ahmad, Jie Huang, Jing-Song Li, and Ming-Wei Chang. 2017. "Controlled Morphing of Microbubbles to Beaded Nanofibers via Electrically Forced Thin Film Stretching" Polymers 9, no. 7: 265. https://doi.org/10.3390/polym9070265
APA StyleYao, Z.-C., Yuan, Q., Ahmad, Z., Huang, J., Li, J.-S., & Chang, M.-W. (2017). Controlled Morphing of Microbubbles to Beaded Nanofibers via Electrically Forced Thin Film Stretching. Polymers, 9(7), 265. https://doi.org/10.3390/polym9070265








