Poly(Lactic-co-Glycolic Acid): Applications and Future Prospects for Periodontal Tissue Regeneration
Abstract
:1. Introduction
2. Properties of PLGA Related to Periodontal Therapy
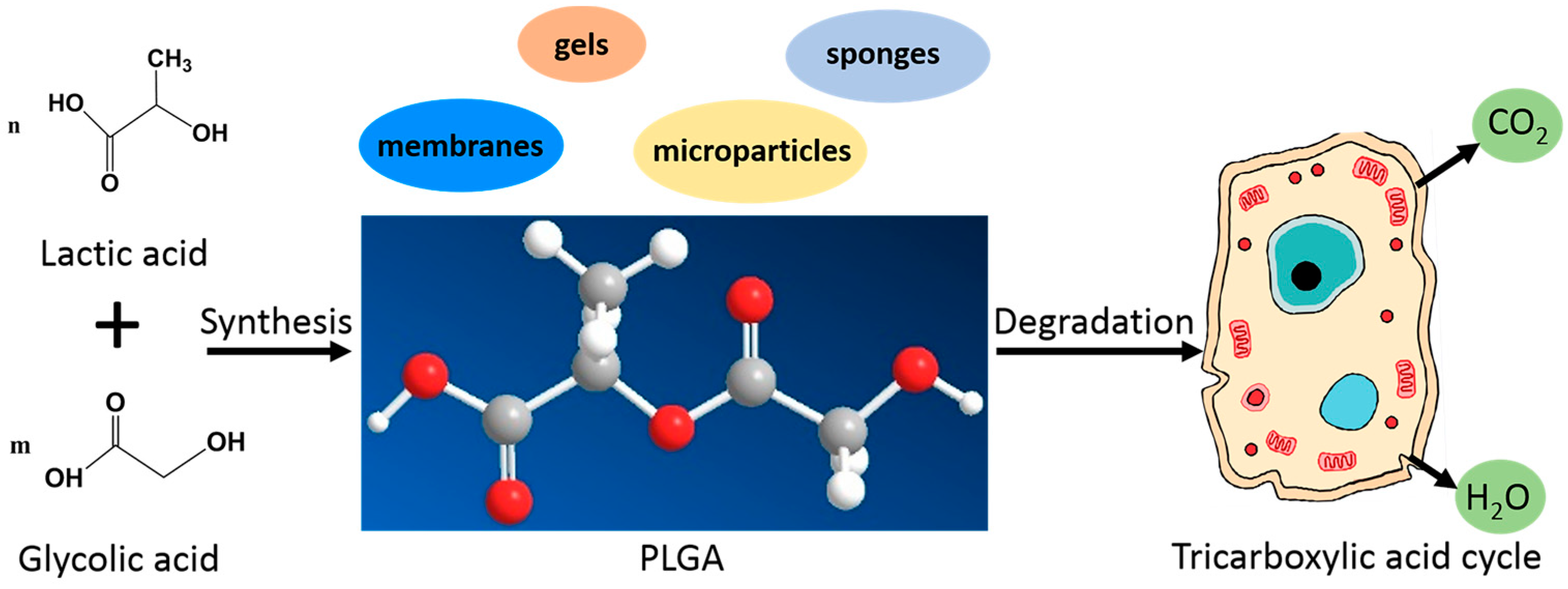
2.1. Physiochemical Properties
2.2. Biocompatibility
2.3. Biodegradability
3. Currently Commercially Available PLGA Products
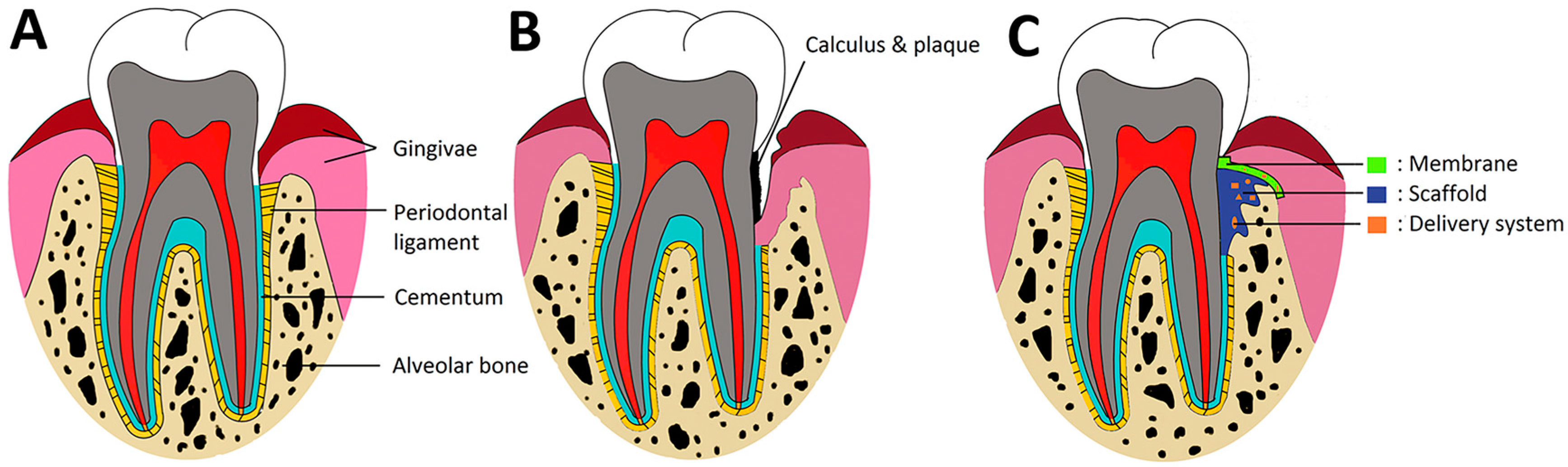
4. Application of PLGA for Periodontal Regeneration
4.1. PLGA Barrier Membranes
4.2. PLGA-Based Bone Scaffolds
4.3. PLGA for Periodontal Drug Delivery
4.3.1. Growth Factors Delivery
4.3.2. Antimicrobial Drug Delivery
5. Future Directions of PLGA for Periodontal Regeneration
5.1. Surface Modification of PLGA Membranes for GTR
5.2. Hydrophobicity Modification
5.3. Improve Bioactivity
5.4. PLGA Based Cell Engineering
6. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eke, P.I.; Dye, B.A.; Wei, L.; Slade, G.D.; Thornton-Evans, G.O.; Borgnakke, W.S.; Taylor, G.W.; Page, R.C.; Beck, J.D.; Genco, R.J. Update on Prevalence of Periodontitis in Adults in the United States: NHANES 2009 to 2012. J. Periodontol. 2015, 86, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Chen, F.M.; An, Y.; Zhang, R.; Zhang, M. New insights into and novel applications of release technology for periodontal reconstructive therapies. J. Control. Release 2011, 149, 92–110. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Zhang, J.; Zhang, M.; An, Y.; Chen, F.; Wu, Z.F. A review on endogenous regenerative technology in periodontal regenerative medicine. Biomaterials 2010, 31, 7892–7927. [Google Scholar] [CrossRef] [PubMed]
- Susin, C.; Wikesjo, U.M. Regenerative periodontal therapy: 30 Years of lessons learned and unlearned. Periodontology 2000 2013, 62, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Sam, G.; Pillai, B.R. Evolution of Barrier Membranes in Periodontal Regeneration—“Are the third Generation Membranes really here?”. J. Clin. Diagn. Res. 2014, 8, ZE14–ZE17. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—a materials perspective. Dent. Mater. 2012, 28, 703–721. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Lei, C.; Meng, L.Y.; Wang, C.N.; Song, Y.L. Chitosan as a barrier membrane material in periodontal tissue regeneration. J. Biomed. Mater. Res. B 2012, 100b, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Parrish, L.C.; Miyamoto, T.; Fong, N.; Mattson, J.S.; Cerutis, D.R. Non-bioabsorbable vs. bioabsorbable membrane: Assessment of their clinical efficacy in guided tissue regeneration technique. A systematic review. J. Oral Sci. 2009, 51, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Shimauchi, H.; Nemoto, E.; Ishihata, H.; Shimomura, M. Possible functional scaffolds for periodontal regeneration. Jpn. Dent. Sci. Rev. 2013, 49, 118–130. [Google Scholar] [CrossRef]
- Kim, J.H.; Park, C.H.; Perez, R.A.; Lee, H.Y.; Jang, J.H.; Lee, H.H.; Wall, I.B.; Shi, S.; Kim, H.W. Advanced biomatrix designs for regenerative therapy of periodontal tissues. J. Dent. Res. 2014, 93, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Patil, S.; Chaubey, K.K.; Thakur, R.K.; Goyel, P. Clinical evaluation of hydroxyapatite and beta-tricalcium phosphate composite graft in the treatment of intrabony periodontal defect: A clinico-radiographic study. J. Indian Soc. Periodontol. 2014, 18, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Sculean, A.; Nikolidakis, D.; Schwarz, F. Regeneration of periodontal tissues: Combinations of barrier membranes and grafting materials—Biological foundation and preclinical evidence: A systematic review. J. Clin. Periodontol. 2008, 35 (Suppl. 8), 106–116. [Google Scholar] [CrossRef] [PubMed]
- Greenstein, G.; Polson, A. The role of local drug delivery in the management of periodontal diseases: A comprehensive review. J. Periodontol. 1998, 69, 507–520. [Google Scholar] [CrossRef] [PubMed]
- King, G.N. The importance of drug delivery to optimize the effects of bone morphogenetic proteins during periodontal regeneration. Curr. Pharm. Biotechnol. 2001, 2, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Ulery, B.D.; Nair, L.S.; Laurencin, C.T. Biomedical applications of biodegradable polymers. J. Polym. Sci. Part B Polym. Phys. 2011, 49, 832–864. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.J.; Kau, Y.C.; Chou, C.; Chen, J.K.; Wu, R.H.; Yeh, W.L. Electrospun PLGA/collagen nanofibrous membrane as early-stage wound dressing. J. Membr. Sci. 2010, 355, 53–59. [Google Scholar] [CrossRef]
- Houchin, M.L.; Topp, E.M. Chemical degradation of peptides and proteins in PLGA: A review of reactions and mechanisms. J. Pharm. Sci. 2008, 97, 2395–2404. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.; Wolke, J.; Mikos, A.; Jansen, J. Injectable PLGA microsphere/calcium phosphate cements: Physical properties and degradation characteristics. J. Biomater. Sci. Polym. Ed. 2006, 17, 1057–1074. [Google Scholar] [CrossRef] [PubMed]
- Biondi, M.; Ungaro, F.; Quaglia, F.; Netti, P.A. Controlled drug delivery in tissue engineering. Adv. Drug Deliv. Rev. 2008, 60, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Parent, M.; Nouvel, C.; Koerber, M.; Sapin, A.; Maincent, P.; Boudier, A. PLGA in situ implants formed by phase inversion: Critical physicochemical parameters to modulate drug release. J. Control. Release 2013, 172, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.A. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials 2000, 21, 2475–2490. [Google Scholar] [CrossRef]
- Wu, X.S.; Wang, N. Synthesis, characterization, biodegradation, and drug delivery application of biodegradable lactic/glycolic acid polymers. Part II: Biodegradation. J. Biomater. Sci. Polym. Ed. 2001, 12, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Tang, X.; Zhang, J.; Lu, W.; Lin, X.; Zhang, Y.; Tian, B.; Yang, H.; He, H. PEG–PLGA copolymers: Their structure and structure-influenced drug delivery applications. J. Control. Release 2014, 183, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Sophocleous, A.M.; Desai, K.G.; Mazzara, J.M.; Tong, L.; Cheng, J.X.; Olsen, K.F.; Schwendeman, S.P. The nature of peptide interactions with acid end-group PLGAs and facile aqueous-based microencapsulation of therapeutic peptides. J. Control. Release 2013, 172, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Pignatello, R.; Sarpietro, M.G.; Castelli, F. Synthesis and biological evaluation of a new polymeric conjugate and nanocarrier with osteotropic properties. J. Funct. Biomater. 2012, 3, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Cenni, E.; Avnet, S.; Granchi, D.; Fotia, C.; Salerno, M.; Micieli, D.; Sarpietro, M.G.; Pignatello, R.; Castelli, F.; Baldini, N. The Effect of Poly(d,l-Lactide-co-Glycolide)-Alendronate Conjugate Nanoparticles on Human Osteoclast Precursors. J. Biomater. Sci. Polym. Ed. 2012, 23, 1285–1300. [Google Scholar] [CrossRef] [PubMed]
- Mittal, G.; Sahana, D.K.; Bhardwaj, V.; Kumar, M.N.V.R. Estradiol loaded PLGA nanoparticles for oral administration: Effect of polymer molecular weight and copolymer composition on release behavior in vitro and in vivo. J. Control. Release 2007, 119, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Leung, L.; Chan, C.; Baek, S.; Naguib, H. Comparison of morphology and mechanical properties of PLGA bioscaffolds. Biomed. Mater. 2008, 3, 025006. [Google Scholar] [CrossRef] [PubMed]
- Semete, B.; Booysen, L.; Lemmer, Y.; Kalombo, L.; Katata, L.; Verschoor, J.; Swai, H.S. In vivo evaluation of the biodistribution and safety of PLGA nanoparticles as drug delivery systems. Nanomedicine 2010, 6, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Rücker, M.; Laschke, M.W.; Junker, D.; Carvalho, C.; Schramm, A.; Mülhaupt, R.; Gellrich, N.C.; Menger, M.D. Angiogenic and inflammatory response to biodegradable scaffolds in dorsal skinfold chambers of mice. Biomaterials 2006, 27, 5027–5038. [Google Scholar] [CrossRef] [PubMed]
- Landes, C.A.; Ballon, A.; Roth, C. Maxillary and mandibular osteosyntheses with PLGA and P (L/DL) LA implants: A 5-year inpatient biocompatibility and degradation experience. Plast. Reconstr. Surg. 2006, 117, 2347–2360. [Google Scholar] [CrossRef] [PubMed]
- Inanc, B.; Elcin, A.E.; Elcin, Y.M. Osteogenic induction of human periodontal ligament fibroblasts under two-and three-dimensional culture conditions. Tissue Eng. 2006, 12, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Hakkarainen, M.; Albertsson, A.C.; Karlsson, S. Weight losses and molecular weight changes correlated with the evolution of hydroxyacids in simulated in vivo degradation of homo- and copolymers of PLA and PGA. Polym. Degrad. Stab. 1996, 52, 283–291. [Google Scholar] [CrossRef]
- Farahani, T.D.; Entezami, A.A.; Mobedi, H.; Abtahi, M. Degradation of poly(d,l-lactide-co-glycolide) 50:50 implant in aqueous medium. Iran. Polym. J. 2005, 14, 753–763. [Google Scholar]
- Zhang, H.; Zhou, L.; Zhang, W. Control of scaffold degradation in tissue engineering: A review. Tissue Eng. B Rev. 2014, 20, 492–502. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Owen, G.R.; Jackson, J.K.; Chehroudi, B.; Brunette, D.M.; Burt, H.M. An in vitro study of plasticized poly(lactic-co-glycolic acid) films as possible guided tissue regeneration membranes: Material properties and drug release kinetics. J. Biomed. Mater. Res. A 2010, 95, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Anderson, J.M.; Shive, M.S. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 2012, 64, 72–82. [Google Scholar] [CrossRef]
- Tracy, M.A.; Ward, K.L.; Firouzabadian, L.; Wang, Y.; Dong, N.; Qian, R.; Zhang, Y. Factors affecting the degradation rate of poly(lactide-co-glycolide) microspheres in vivo and in vitro. Biomaterials 1999, 20, 1057–1062. [Google Scholar] [CrossRef]
- Samadi, N.; Abbadessa, A.; Di Stefano, A.; van Nostrum, C.F.; Vermonden, T.; Rahimian, S.; Teunissen, E.A.; van Steenbergen, M.J.; Amidi, M.; Hennink, W.E. The effect of lauryl capping group on protein release and degradation of poly(d,l-lactic-co-glycolic acid) particles. J. Control. Release 2013, 172, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Zolnik, B.S.; Burgess, D.J. Effect of acidic pH on PLGA microsphere degradation and release. J. Control. Release 2007, 122, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Grayson, A.C.R.; Cima, M.J.; Langer, R. Size and temperature effects on poly(lactic-co-glycolic acid) degradation and microreservoir device performance. Biomaterials 2005, 26, 2137–2145. [Google Scholar] [CrossRef] [PubMed]
- Hurzeler, M.B.; Quinones, C.R.; Caffesse, R.G.; Schupbach, P.; Morrison, E.C. Guided periodontal tissue regeneration in class II furcation defects following treatment with a synthetic bioabsorbable barrier. J. Periodontol. 1997, 68, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Yamada, S. Evaluation of the regenerative effect of a 25% doxycycline-loaded biodegradable membrane for guided tissue regeneration. J. Periodontol. 2000, 71, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Kurtis, B.; Unsal, B.; Cetiner, D.; Gultekin, E.; Ozcan, G.; Celebi, N.; Ocak, O. Effect of polylactide/glycolide (PLGA) membranes loaded with metronidazole on periodontal regeneration following guided tissue regeneration in dogs. J. Periodontol. 2002, 73, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.Y.; Jung, U.W.; Kim, C.S.; Lee, Y.K.; Cho, K.S.; Chai, J.K.; Kim, C.K.; Choi, S.H. Effects of a tetracycline blended polylactic and polyglycolic acid membrane on the healing of one-wall intrabony defects in beagle dogs. Biomed. Mater. 2007, 2, S106–S110. [Google Scholar] [CrossRef] [PubMed]
- Carlo Reis, E.C.; Borges, A.P.; Araujo, M.V.; Mendes, V.C.; Guan, L.; Davies, J.E. Periodontal regeneration using a bilayered PLGA/calcium phosphate construct. Biomaterials 2011, 32, 9244–9253. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Cortellini, P.; Suvan, J.E.; Adriaens, P.; Baldi, C.; Dubravec, D.; Fonzar, A.; Fourmousis, I.; Magnani, C.; Muller-Campanile, V.; et al. Generalizability of the added benefits of guided tissue regeneration in the treatment of deep intrabony defects. Evaluation in a multi-center randomized controlled clinical trial. J. Periodontol. 1998, 69, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Aimetti, M.; Romano, F.; Pigella, E.; Pranzini, F.; Debernardi, C. Treatment of wide, shallow, and predominantly 1-wall intrabony defects with a bioabsorbable membrane: A randomized controlled clinical trial. J. Periodontol. 2005, 76, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Balusubramanya, K.V.; Ramya, R.; Govindaraj, S.J. Clinical and radiological evaluation of human osseous defects (mandibular grade ii furcation involvement) treated with bioresorbable membrane: Vicryl mesh. J. Contemp. Dent. Pract. 2012, 13, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.; Weber, F.E.; Grunder, U.; Andreoni, C.; Burkhardt, R.; Jung, R.E. A randomized controlled clinical multicenter trial comparing the clinical and histological performance of a new, modified polylactide-co-glycolide acid membrane to an expanded polytetrafluorethylene membrane in guided bone regeneration procedures. Clin. Oral Implant. Res. 2014, 25, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.; Becker, B.E.; Mellonig, J.; Caffesse, R.G.; Warrer, K.; Caton, J.G.; Reid, T. A prospective multi-center study evaluating periodontal regeneration for Class II furcation invasions and intrabony defects after treatment with a bioabsorbable barrier membrane: 1-year results. J. Periodontol. 1996, 67, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, P.; Giovannoli, J.L.; Mattout, C.; Davarpanah, M.; Etienne, D. Clinical evaluation of a bioabsorbable regenerative material in mandibular class II furcation therapy. J. Clin. Periodontol. 1997, 24, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Mattson, J.S.; Gallagher, S.J.; Jabro, M.H. The use of 2 bioabsorbable barrier membranes in the treatment of interproximal intrabony periodontal defects. J. Periodontol. 1999, 70, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Stavropoulos, A.; Sculean, A.; Karring, T. GTR treatment of intrabony defects with PLA/PGA copolymer or collagen bioresorbable membranes in combination with deproteinized bovine bone (Bio-Oss). Clin. Oral Investig. 2004, 8, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Pretzl, B.; Kim, T.S.; Holle, R.; Eickholz, P. Long-term results of guided tissue regeneration therapy with non-resorbable and bioabsorbable barriers. IV. A case series of infrabony defects after 10 years. J. Periodontol. 2008, 79, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Gupta, N.D. Comparative evaluation of decalcified freeze-dried bone allograft use alone and in combination with polylactic acid, polyglycolic acid membrane in the treatment of noncontained human periodontal infrabony defects. Quintessence Int. 2012, 43, 761–768. [Google Scholar] [PubMed]
- Vaquette, C.; Fan, W.; Xiao, Y.; Hamlet, S.; Hutmacher, D.W.; Ivanovski, S. A biphasic scaffold design combined with cell sheet technology for simultaneous regeneration of alveolar bone/periodontal ligament complex. Biomaterials 2012, 33, 5560–5573. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Zhao, Y.M.; Zhang, R.; Jin, T.; Sun, H.H.; Wu, Z.F.; Jin, Y. Periodontal regeneration using novel glycidyl methacrylated dextran (Dex-GMA)/gelatin scaffolds containing microspheres loaded with bone morphogenetic proteins. J. Control. Release 2007, 121, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Dali, G.; Velazquez-Cayon, R.; Serrera-Figallo, M.A.; Rodriguez-Gonzalez-Elipe, A.; Gutierrez-Perez, J.L.; Torres-Lagares, D. Importance of Poly(lactic-co-glycolic acid) in Scaffolds for Guided Bone Regeneration: A Focused Review. J. Oral Implantol. 2015, 41, e152–e157. [Google Scholar] [CrossRef] [PubMed]
- Chhabra, V.; Gill, A.S.; Sikri, P.; Bhaskar, N. Evaluation of the relative efficacy of copolymerized polylactic-polyglycolic acids alone and in conjunction with polyglactin 910 membrane in the treatment of human periodontal infrabony defects: A clinical and radiological study. Indian J. Dent. Res. 2011, 22, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Minenna, L.; Herrero, F.; Sanz, M.; Trombelli, L. Adjunctive effect of a polylactide/polyglycolide copolymer in the treatment of deep periodontal intra-osseous defects: A randomized clinical trial. J. Clin. Periodontol. 2005, 32, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Serino, G.; Rao, W.; Iezzi, G.; Piattelli, A. Polylactide and polyglycolide sponge used in human extraction sockets: Bone formation following 3 months after its application. Clin. Oral Implant. Res. 2008, 19, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Shelton, R.M.; Jin, Y.; Chapple, I.L. Localized delivery of growth factors for periodontal tissue regeneration: Role, strategies, and perspectives. Med. Res. Rev. 2009, 29, 472–513. [Google Scholar] [CrossRef] [PubMed]
- Elangovan, S.; Karimbux, N. Review Paper: DNA Delivery Strategies to Promote Periodontal Regeneration. J. Biomater. Appl. 2010, 25, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Sood, S.; Gupta, S.; Mahendra, A. Gene therapy with growth factors for periodontal tissue engineering—A review. Med. Oral Patol. Oral Cir. Bucal 2012, 17, e301–e310. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.M.; Cortellini, P. Periodontal tissue engineering and regeneration: Consensus report of the sixth European workshop on periodontology. J. Clin. Periodontol. 2008, 35 (Suppl. 8), 83–86. [Google Scholar] [CrossRef] [PubMed]
- Haidar, Z.S.; Hamdy, R.C.; Tabrizian, M. Delivery of recombinant bone morphogenetic proteins for bone regeneration and repair. Part A: Current challenges in BMP delivery. Biotechnol. Lett. 2009, 31, 1817–1824. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.H.; Qu, T.J.; Zhang, X.H.; Yu, Q.; Chen, F.M. Designing biomaterials for in situ periodontal tissue regeneration. Biotechnol. Prog. 2012, 28, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Min, C.; Wikesjö, U.M.E.; Park, J.; Chae, G.; Pippig, S.D.; Bastone, P.; Kim, C.; Kim, C. Wound healing/regeneration using recombinant human growth/differentiation factor-5 in an injectable poly-lactide-co-glycolide-acid composite carrier and a one-wall intra-bony defect model in dogs. J. Clin. Periodontol. 2011, 38, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Herberg, S.; Siedler, M.; Pippig, S.; Schuetz, A.; Dony, C.; Kim, C.K.; Wikesjo, U.M. Development of an injectable composite as a carrier for growth factor-enhanced periodontal regeneration. J. Clin. Periodontol. 2008, 35, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Wikesjö, U.M.E.; Koo, K.; Lee, J.; Kim, Y.; Pippig, S.D.; Bastone, P.; Kim, C.S.; Kim, C.K. Maturation of alveolar bone following implantation of an rhGDF-5/PLGA composite into 1-wall intra-bony defects in dogs: 24-week histometric observations. J. Clin. Periodontol. 2012, 39, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.T.; Wikesjo, U.M.; Jung, U.W.; Lee, J.S.; Kim, T.G.; Kim, C.K. Comparison between a beta-tricalcium phosphate and an absorbable collagen sponge carrier technology for rhGDF-5-stimulated periodontal wound healing/regeneration. J. Periodontol. 2013, 84, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.H.; Bennett, W.; Herberg, S.; Bastone, P.; Pippig, S.; Rodriguez, N.A.; Susin, C.; Wikesjo, U.M. Evaluation of an injectable rhGDF-5/PLGA construct for minimally invasive periodontal regenerative procedures: A histological study in the dog. J. Clin. Periodontol. 2010, 37, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.H.; Subbiah, R.; Kim, I.H.; Han, D.K.; Park, K. Dual growth factor delivery using biocompatible core-shell microcapsules for angiogenesis. Small 2013, 9, 3468–3476. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.; Dovban, A.S.; Lim, L.P.; Chong, L.Y.; Kuo, M.Y.; Wang, C. Dual delivery of PDGF and simvastatin to accelerate periodontal regeneration in vivo. Biomaterials 2013, 34, 9990–9997. [Google Scholar] [CrossRef] [PubMed]
- Slots, J.; Macdonald, E.S.; Nowzar, H. Infectious aspects of periodontal regeneration. Periodontology 2000 1999, 19, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.F.; Lee, Y.Y.; Chi, L.Y.; Chen, Y.T.; Hung, S.L.; Ling, L.J. Bacterial penetration through antibiotic-loaded guided tissue regeneration membranes. J. Periodontol. 2009, 80, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Frongia, M.E.; Cardellach, M.; Miller, C.A.; Stafford, G.P.; Leggett, G.J.; Hatton, P.V. Functionalised nanoscale coatings using layer-by-layer assembly for imparting antibacterial properties to polylactide-co-glycolide surfaces. Acta Biomater. 2015, 21, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar-Mohammadi, M.; Zamani, M.; Prabhakaran, M.P.; Bahrami, S.H.; Ramakrishna, S. Electrospinning of PLGA/gum tragacanth nanofibers containing tetracycline hydrochloride for periodontal regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.S.; Lee, C.C.; Wang, Y.P.; Chen, H.J.; Lai, C.H.; Hsieh, W.L.; Chen, Y.W. Controlled-release of tetracycline and lovastatin by poly(d,l-lactide-co-glycolide acid)-chitosan nanoparticles enhances periodontal regeneration in dogs. Int. J. Nanomed. 2016, 11, 285–297. [Google Scholar]
- Liu, D.Z.; Chen, W.P.; Lee, C.P.; Wu, S.L.; Wang, Y.C.; Chung, T.W. Effects of alginate coated on PLGA microspheres for delivery tetracycline hydrochloride to periodontal pockets. J. Microencapsul. 2004, 21, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xia, Y.; Lu, X.; Zhou, X.; Zhang, F.; Gu, N. Effects of surface functionalization of PLGA membranes for guided bone regeneration on proliferation and behavior of osteoblasts. J. Biomed. Mater. Res. A 2013, 101A, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Park, G.E.; Pattison, M.A.; Park, K.; Webster, T.J. Accelerated chondrocyte functions on NaOH-treated PLGA scaffolds. Biomaterials 2005, 26, 3075–3082. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.H.; Kang, S.G.; Lee, J.H. Degradation behavior of hydrophilized PLGA scaffolds prepared by melt-molding particulate-leaching method: Comparison with control hydrophobic one. J. Mater. Sci. Mater. Med. 2006, 17, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Gelmedin, V.; Delaney, A.; Jennelle, L.; Hawdon, J.M. Expression profile of heat shock response factors during hookworm larval activation and parasitic development. Mol. Biochem. Parasitol. 2015, 202, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fortunati, E.; Mattioli, S.; Visai, L.; Imbriani, M.; Fierro, J.L.; Kenny, J.M.; Armentano, I. Combined effects of Ag nanoparticles and oxygen plasma treatment on PLGA morphological, chemical, and antibacterial properties. Biomacromolecules 2013, 14, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Jamuna-Thevi, K.; Saarani, N.N.; Abdul Kadir, M.R.; Hermawan, H. Triple-layered PLGA/nanoapatite/lauric acid graded composite membrane for periodontal guided bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 43, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Filipowska, J.; Pawlik, J.; Cholewa-Kowalska, K.; Tylko, G.; Pamula, E.; Niedzwiedzki, L.; Szuta, M.; Laczka, M.; Osyczka, A.M. Incorporation of sol-gel bioactive glass into PLGA improves mechanical properties and bioactivity of composite scaffolds and results in their osteoinductive properties. Biomed. Mater. 2014, 9, 065001. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.P.; Cui, F.Z. Surface modification of polyester biomaterials for tissue engineering. Biomed. Mater. 2007, 2, R24–R37. [Google Scholar] [CrossRef] [PubMed]
- Wojak-Cwik, I.; Hintze, V.; Schnabelrauch, M.; Moeller, S.; Dobrzynski, P.; Pamula, E.; Scharnweber, D. Poly (l-lactide-co-glycolide) scaffolds coated with collagen and glycosaminoglycans: Impact on proliferation and osteogenic differentiation of human mesenchymal stem cells. J. Biomed. Mater. Res. A 2013, 101, 3109–3122. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.M.; Gritsch, K.; Salles, V.; Attik, G.N.; Grosgogeat, B. Surface Entrapment of Fibronectin on Electrospun PLGA Scaffolds for Periodontal Tissue Engineering. BioRes. Open Access 2014, 3, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.G.; Park, T.G. Biomimicking extracellular matrix: Cell adhesive RGD peptide modified electrospun poly (d,l-lactic-co-glycolic acid) nanofiber mesh. Tissue Eng. 2006, 12, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Kawazoe, N.; Lin, X.; Dong, J.; Chen, G. The influence of structural design of PLGA/collagen hybrid scaffolds in cartilage tissue engineering. Biomaterials 2010, 31, 2141–2152. [Google Scholar] [CrossRef] [PubMed]
- Wilberforce, S.I.; Finlayson, C.E.; Best, S.M.; Cameron, R.E. The influence of the compounding process and testing conditions on the compressive mechanical properties of poly(d,l-lactide-co-glycolide)/alpha-tricalcium phosphate nanocomposites. J. Mech. Behav. Biomed. Mater. 2011, 4, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Douglas, T.; Pamula, E.; Hauk, D.; Wiltfang, J.; Sivananthan, S.; Sherry, E.; Warnke, P.H. Porous polymer/hydroxyapatite scaffolds: Characterization and biocompatibility investigations. J. Mater. Sci. Mater. Med. 2009, 20, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Xing, J.; Zhang, X.; Dong, S.; Zhao, Y.; Wang, L.; Li, H.; Yang, F.; Xu, C.; Zhang, W. Exogenous hydrogen sulfide prevents cardiomyocyte apoptosis from cardiac hypertrophy induced by isoproterenol. Mol. Cell. Biochem. 2013, 381, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, W.; Liu, J.S.; Wang, J.; Yang, P.; Li, M.L.; Zhao, Z.H. Expression of osteoclastogenesis inducers in a tissue model of periodontal ligament under compression. J. Dent. Res. 2011, 90, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.W.; Yang, H.S.; Seo, S.W.; Han, D.K.; Kim, B.S. Apatite-coated poly(lactic-co-glycolic acid) microspheres as an injectable scaffold for bone tissue engineering. J. Biomed. Mater. Res. A 2008, 85, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Inanc, B.; Arslan, Y.E.; Seker, S.; Elcin, A.E.; Elcin, Y.M. Periodontal ligament cellular structures engineered with electrospun poly(dl-lactide-co-glycolide) nanofibrous membrane scaffolds. J. Biomed. Mater. Res. A 2009, 90, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Akita, D.; Morokuma, M.; Saito, Y.; Yamanaka, K.; Akiyama, Y.; Sato, M.; Mashimo, T.; Toriumi, T.; Arai, Y.; Kaneko, T.; et al. Periodontal tissue regeneration by transplantation of rat adipose-derived stromal cells in combination with PLGA-based solid scaffolds. Biomed. Res. 2014, 35, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Shang, S.; Yang, F.; Cheng, X.; Walboomers, X.F.; Jansen, J.A. The effect of electrospun fibre alignment on the behaviour of rat periodontal ligament cells. Eur. Cells Mater. 2010, 19, 180–192. [Google Scholar] [CrossRef]
- Jin, Q.M.; Zhao, M.; Webb, S.A.; Berry, J.E.; Somerman, M.J.; Giannobile, W.V. Cementum engineering with three-dimensional polymer scaffolds. J. Biomed. Mater. Res. A 2003, 67, 54–60. [Google Scholar] [CrossRef] [PubMed]
| Trade Name | Manufacturer | Composition (ratio) | Form | Degradation time | Biocompatibility and tissue response | Mechanical properties | Features |
|---|---|---|---|---|---|---|---|
| Vicryl-Netz | Ethicon | PLA/PGA 10/90 | Mesh | 4–12 weeks | Inert, lack of tissue integration, no reactions in the surrounding tissues | Semicrystalline, relatively soft | Well adaptable, easy handling, elimination of membrane removal |
| Resolut | W.L. Gore | PGA/PLGA | Mesh | 20–24 weeks | Good tissue integration, low inflammatory response in the surrounding tissues | Rigid, elastic | Retains its mechanical strength for 4 months to resist membrane collapse |
| Vicryl | Ethicon | PLA/PGA 8/92 | Mesh | 12 weeks | Good biocompatibility, limited inflammatory response | Semicrystalline, low elasticity, hold its tensile strength for 2–3 weeks in tissues | Easy to handle surgically |
| Suture | |||||||
| Polysorb | U.S. Surgical | PLA/PGA 80/20 | Mesh | 8–10 weeks | Minimal tissue reaction | High tensile strength, low elasticity | Easy handling |
| Suture | |||||||
| Dermagraft | ATS | PLA/PGA 10/90 | Mesh | 4 weeks | Good biocompatibility, limited immunological rejection, no inflammatory response | Great elasticity, porosity 95%, mechanical properties comparable to the native skin | Favorable for cell adherence |
| LactoSorb Screws and Plates | Walter Lorenz Surgical | PLA/PGA | Scaffold | 48 weeks | Well tolerated, induced bone formation without causing adverse tissue responses | High tensile strength and stiffness, retains 70% of original strength at 8 weeks | Plates can be heated and molded to shape multiple times without compromising their mechanical strength |
| Biologically Quiet | Instrument Makar | PLA/PGA 85/15 | Scaffold | 24 weeks | No abnormal tissue reactions | Rigid, high mechanical strength, even stronger than the metal screws | Easy handling, avoidance of reoperation to remove the implants |
| Fisiograft | Ghimas S.p.A | PLA/PGA | Sponge | 12–16 weeks | Biocompatible, totally absorbed in 3–4 months | Varies with different forms, mainly as bone filling materials | Osteoconductive, totally absorbable |
| Power | |||||||
| Gel | |||||||
| Lupron Depot | TAP | PLA/PGA 50/50 | Microparticle | 4 weeks | Minimal toxicity and minimal mechanical irritation to the surrounding tissues | Powders | Capable of delivering a sustained drug therapeutic level for 1 month |
| Zoladex | Astra-Zeneca | PLA/PLGA | Microparticle | 4 weeks | Good biocompatibility, nontoxicity in most tissues | Powders | Monthly subcutaneous injection, increase patient compliance |
| ReGel | Macro-Med | PLGA–PEG–PLGA | Hydrogel | 1–6 weeks | High biocompatibility | Some degree of flexibility | Compatible with tissues, ideally suited to deliver hydrophobic small molecules |
| References | Animal types | Defect types | Length months | Treatment groups | New cementum (mm) | New bone (mm) | Connective tissue adhesion (mm) | Junctional epithelium extension (mm) |
|---|---|---|---|---|---|---|---|---|
| Hurzeler et al., 1997 [46] | rhesus monkeys | Intrabony defects | 5 | A flap operation only | 0.20 ± 0.39 | 0.19 ± 0.37 | 0.20 ± 0.39 | N |
| PLGA membrane | 2.74 ± 0.69 * | 2.64 ± 0.74 * | 2.80 ± 0.75 * | N | ||||
| Hurzeler et al., 1997 [42] | rhesus monkeys | Class II furcation defects | 5 | A flap operation only | 0.83 ± 0.19 | 1.14 ± 0.35 | 0.92 ± 0.26 | N |
| PLGA membrane | 2.88 ± 0.63 * | 2.78 ± 0.53 * | 3.28 ± 0.55 * | N | ||||
| Chang et al., 2000 [47] | dogs | Intrabony defects | 3 | PLGA membrane | 4.03 ± 0.16 | 1.78 ± 0.22 | N | 0.92 ± 0.11 |
| PLGA membrane loaded with 25% doxycycline | 3.89 ± 0.22 | 2.67 ± 0.30 * | N | 1.04 ± 0.12 | ||||
| Kurtis et al., 2002 [48] | dogs | Intrabony defects | 2 | A flap operation only | 0.97 ± 0.04 | 0.62 ± 0.07 | 0.96 ± 0.02 | 2.28 ± 0.06 |
| PLGA membrane | 1.46 ± 0.09 * | 2.01 ± 0.08 * | 1.24 ± 0.02 * | 1.16 ± 0.10 * | ||||
| PLGA membrane Loaded with metronidazole | 1.53 ± 0.10 * | 2.05 ± 0.08 * | 1.20 ± 0.02 * | 1.13 ± 0.10 * | ||||
| Kim et al., 2007 [49] | dogs | Intrabony defects | 2 | A flap operation only | 2.00 ± 0.70 | 1.46 ± 0.68 | 0.85 ± 0.43 | N |
| PLGA membrane | 3.16 ± 0.37 * | 2.39 ± 0.52 * | 0.69 ± 0.17 | N | ||||
| PLGA membraneLoaded with tetracycline | 3.72 ± 0.53 * | 2.88 ± 0.66 * | 0.64 ± 0.10 | N | ||||
| Reis et al., 2011 [50] | dogs | Class II furcation defects | 4 | A flap operation only | N | trabeculaenumber = 1 | 3.80 ± 1.34 | N |
| PLGA membrane combined with CaP particles | N | trabeculaenumber ≈ 3 * | 1.80 ± 0.44 *,# | N |
| References | Patients number | Defect type (defect number) | Length (months) | Treatment groups | Pocket probing depth reduction (mm) | Clinical attachment level gain (mm) | Gingival margin recession change (mm) | Radiologic bone fill (mm) | Vertical/horizontal furcation depth reduction (mm) |
|---|---|---|---|---|---|---|---|---|---|
| Becker et al., 1996 [55] | 50 | class II furcation invasions/31 | 12 | PLGA membrane | 2.5 ± 1.4 | 2.1 ± 1.6 | −0.4 ± 1.0 | N | 1.8 ± 2.0 |
| Becker et al., 1996 [55] | 50 | Intrabony defects/30 | 12 | PLGA membrane | 4.0 ± 1.5 | 2.9 ± 2.0 | 1.2 ± 1.6 | N | N |
| Bouchard et al., 1997 [56] | 30 | class II furcation defects/30 | 12 | ePTFE membrane | 1.8 ± 0.3 | 1.2 ± 0.3 | N | N | 2.7 ± 0.3 |
| PLGA membrane | 2.1 ± 0.4 | 1.5 ± 0.5 | N | N | 2.5 ± 0.4 | ||||
| Tonetti et al., 1998 [51] | 154 | Infrabony defects/154 | 12 | flap operation only | N | 2.18 ± 1.46 | N | N | N |
| PLGA membrane | N | 3.04 ± 1.64 * | N | N | N | ||||
| Mattson et al., 1999 [57] | 19 | Infrabony defects/23 | 6 | collagen membrane | 1.66 ± 1.81 | 1.00 ± 1.82 | 0.66 ± 1.11 | 2.1 ± 2.18 | N |
| PLGA membrane | 2.61 ± 1.75 | 2.01 ± 1.87 | 0.60 ± 0.99 | 1.67 ± 2.10 | N | ||||
| PLGA membrane | 1.8 ± 1.3 | 1.4 ± 1.2 | N | N | N | ||||
| Stavropoulos et al., 2004 [58] | 28 | Infrabony defects/28 | 12 | PLGA membrane + Bio-Oss | 4.0 ± 1.2 | 2.9 ± 2.3 | 1.1 ± 1.6 | N | N |
| collagen membrane + Bio-Oss | 5.1 ± 1.7 | 3.9 ± 1.3 | 1.2 ± 0.8 | N | N | ||||
| Aimetti et al., 2005 [52] | 18 | Infrabony defects/36 | 12 | flap operation only | 2.39 ± 0.92 | 1.50 ± 0.99 | 0.89 ± 0.58 | 1.05 ± 0.94 | N |
| PLGA membrane | 3.44 ± 0.78 * | 2.89 ± 0.90 * | 0.56 ± 0.92 * | 2.13 ± 1.21 * | N | ||||
| Pretzl et al., 2009 [59] | 12 | Infrabony defects/24 | 120 | ePTFE membrane | 2.4 ± 1.6 | −1.7 ± 1.3 | N | 0.8 ± 0.6 | N |
| PLGA membrane | 4.2 ± 2.5 | 0.2 ± 2.0 | N | 2.76 ± 1.70 | N | ||||
| Agarwal et al., 2012 [60] | 12 | Infrabony defects/16 | 6 | bone allograft only | 2.00 ± 0.19 | 1.38 ± 0.1 | N | 0.63 ± 0.26 | N |
| bone allograft with PLGA membrane | 2.75 ± 0.37 | 1.50 ± 0.27 | N | 1.13 ± 0.23 | 1.37 ± 1.12 | ||||
| Balusubramanya et al., 2012 [53] | 7 | class II furcation defects/22 | 6 | flap operation only | N | 1.09 ± 0.94 | N | N | 1.54 ± 1.04 * |
| PLGA membrane | N | 2.18 ± 0.6 * | N | N |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, X.; Xu, C.; Wu, G.; Ye, Q.; Wang, C. Poly(Lactic-co-Glycolic Acid): Applications and Future Prospects for Periodontal Tissue Regeneration. Polymers 2017, 9, 189. https://doi.org/10.3390/polym9060189
Sun X, Xu C, Wu G, Ye Q, Wang C. Poly(Lactic-co-Glycolic Acid): Applications and Future Prospects for Periodontal Tissue Regeneration. Polymers. 2017; 9(6):189. https://doi.org/10.3390/polym9060189
Chicago/Turabian StyleSun, Xiaoyu, Chun Xu, Gang Wu, Qingsong Ye, and Changning Wang. 2017. "Poly(Lactic-co-Glycolic Acid): Applications and Future Prospects for Periodontal Tissue Regeneration" Polymers 9, no. 6: 189. https://doi.org/10.3390/polym9060189
APA StyleSun, X., Xu, C., Wu, G., Ye, Q., & Wang, C. (2017). Poly(Lactic-co-Glycolic Acid): Applications and Future Prospects for Periodontal Tissue Regeneration. Polymers, 9(6), 189. https://doi.org/10.3390/polym9060189






