Bio-Based Adhesives and Evaluation for Wood Composites Application
Abstract
:1. Introduction
2. Lignin
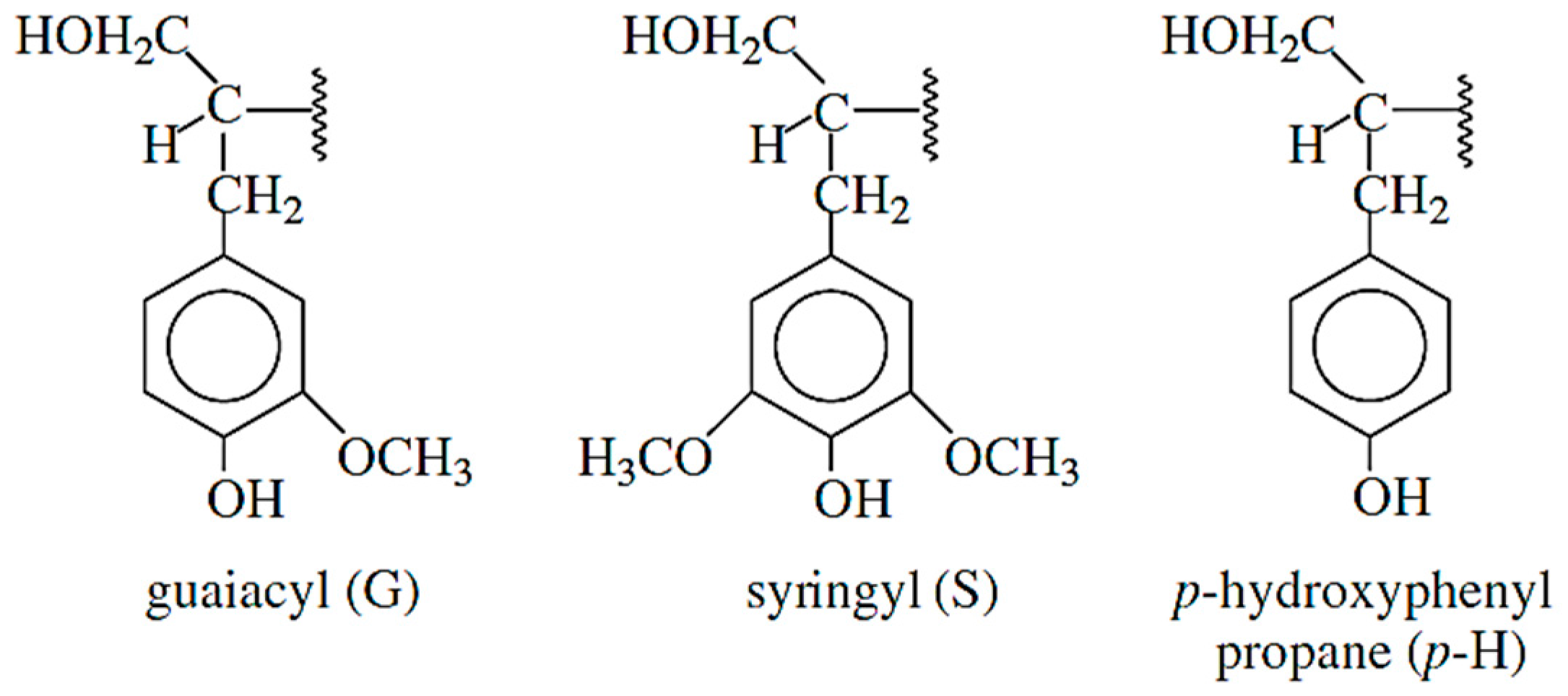
2.1. Chemistry of Lignin
2.2. Lignin-Based Phenol-Formaldehyde Adhesives
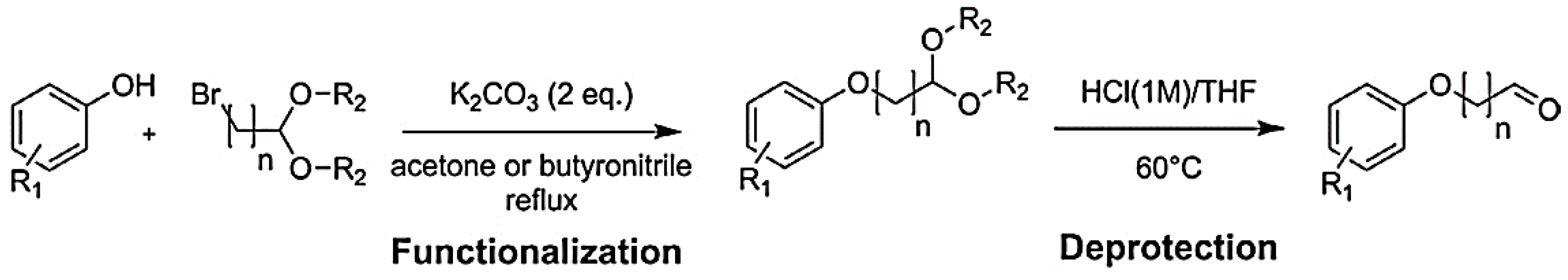
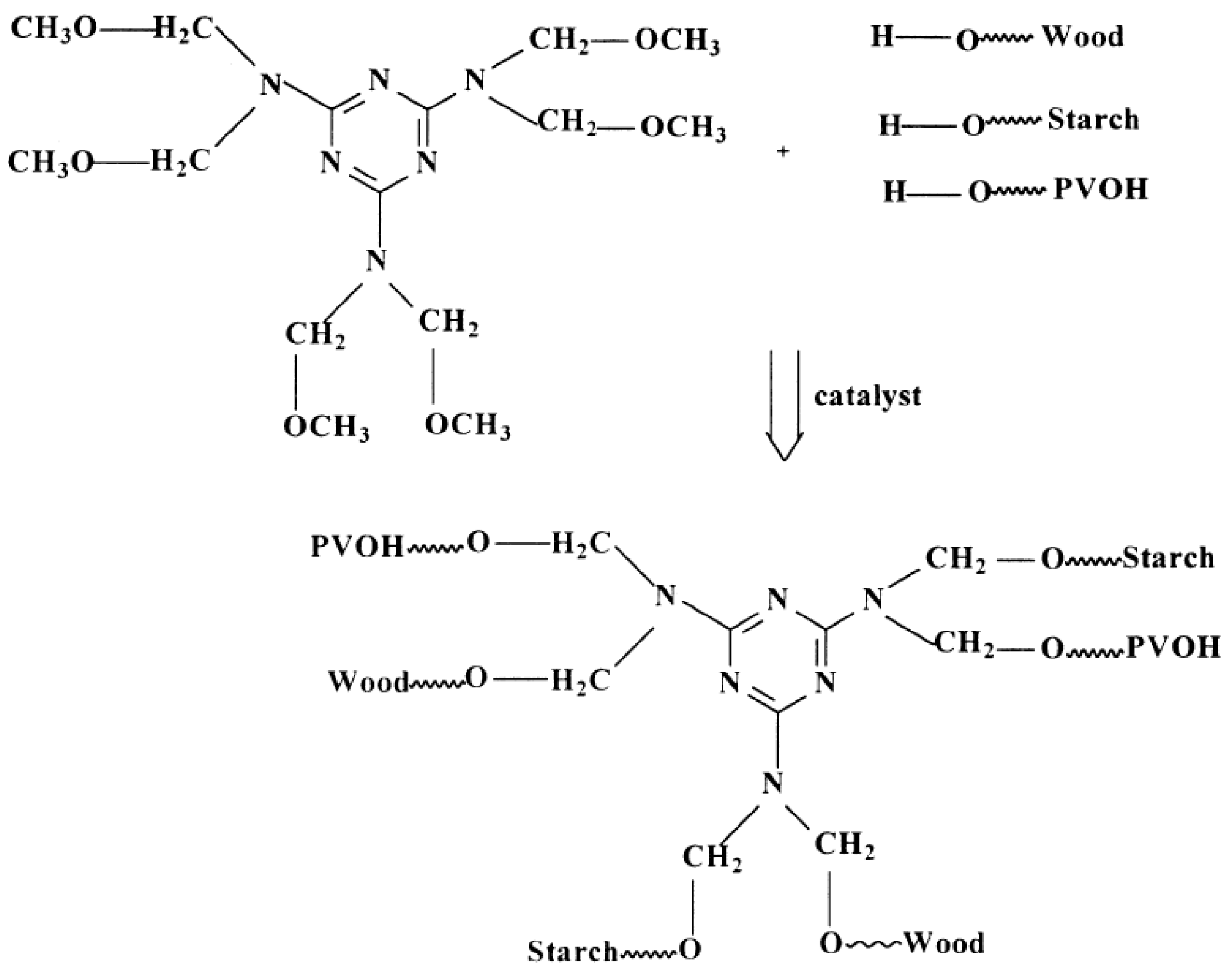
2.3. Formaldehyde-Free Lignin-Based Adhesives
3. Starch
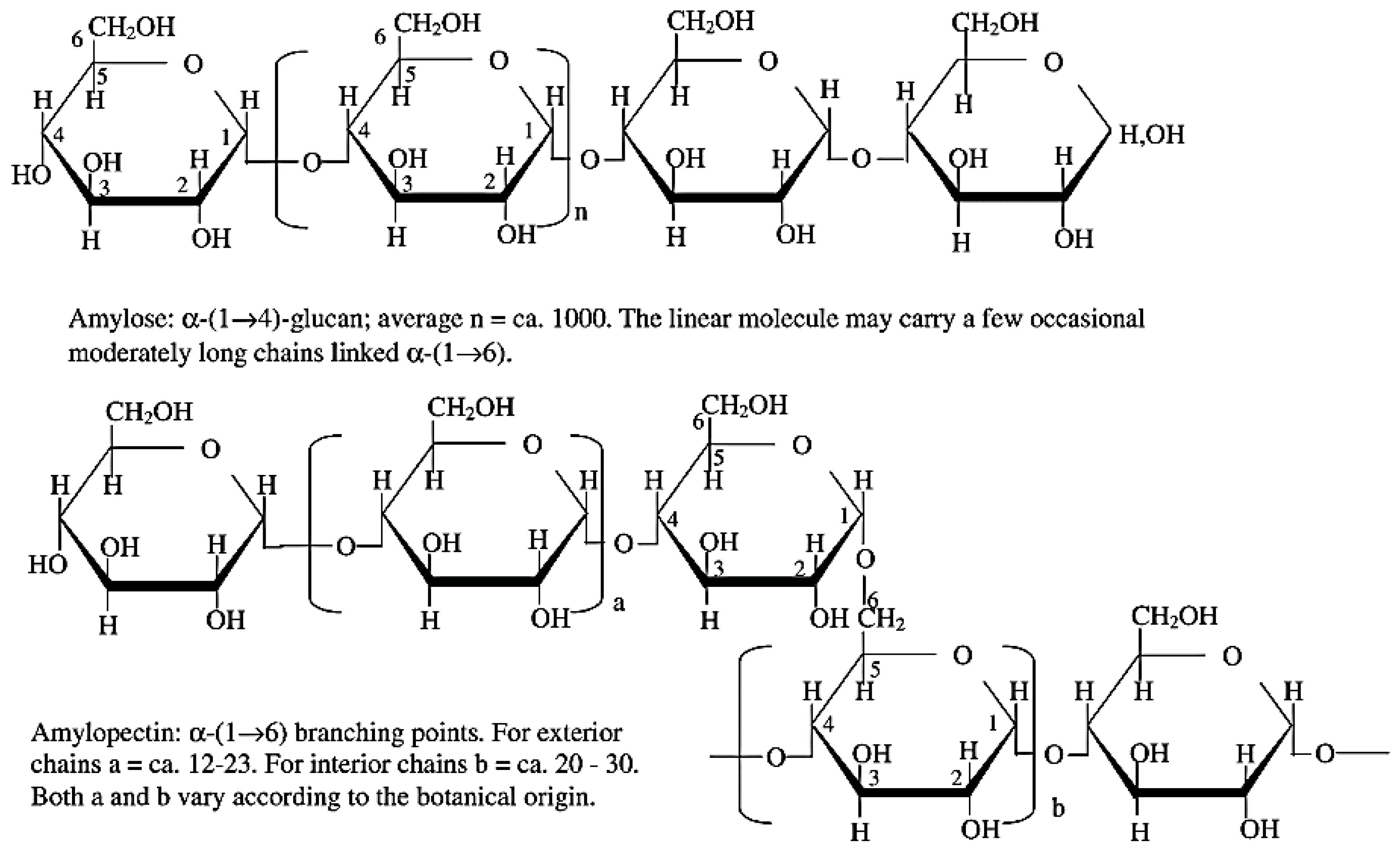
3.1. Chemistry of Starch
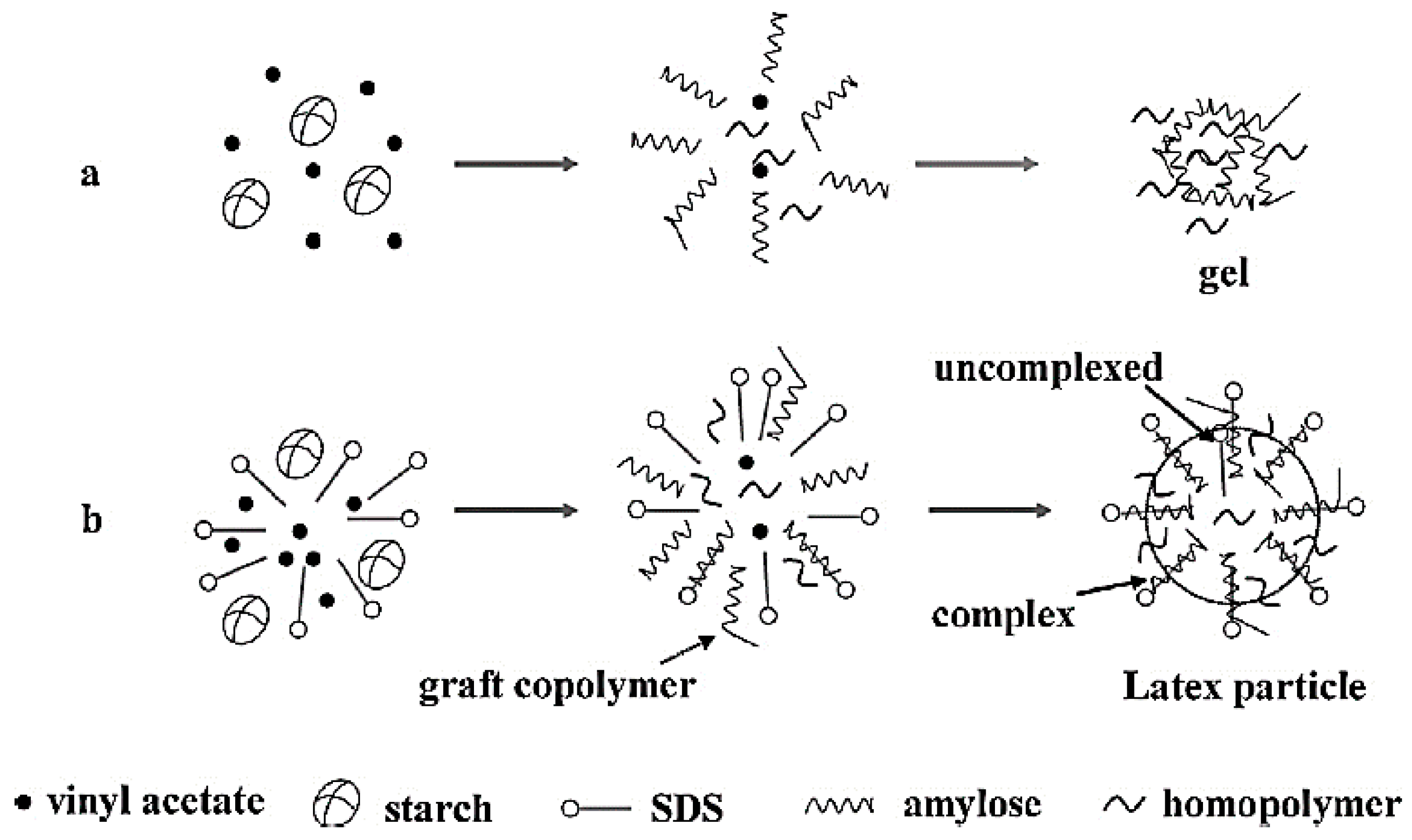
3.2. Starch-Based Wood Adhesives
4. Plant Protein
4.1. Soy Protein-Based Wood Adhesives
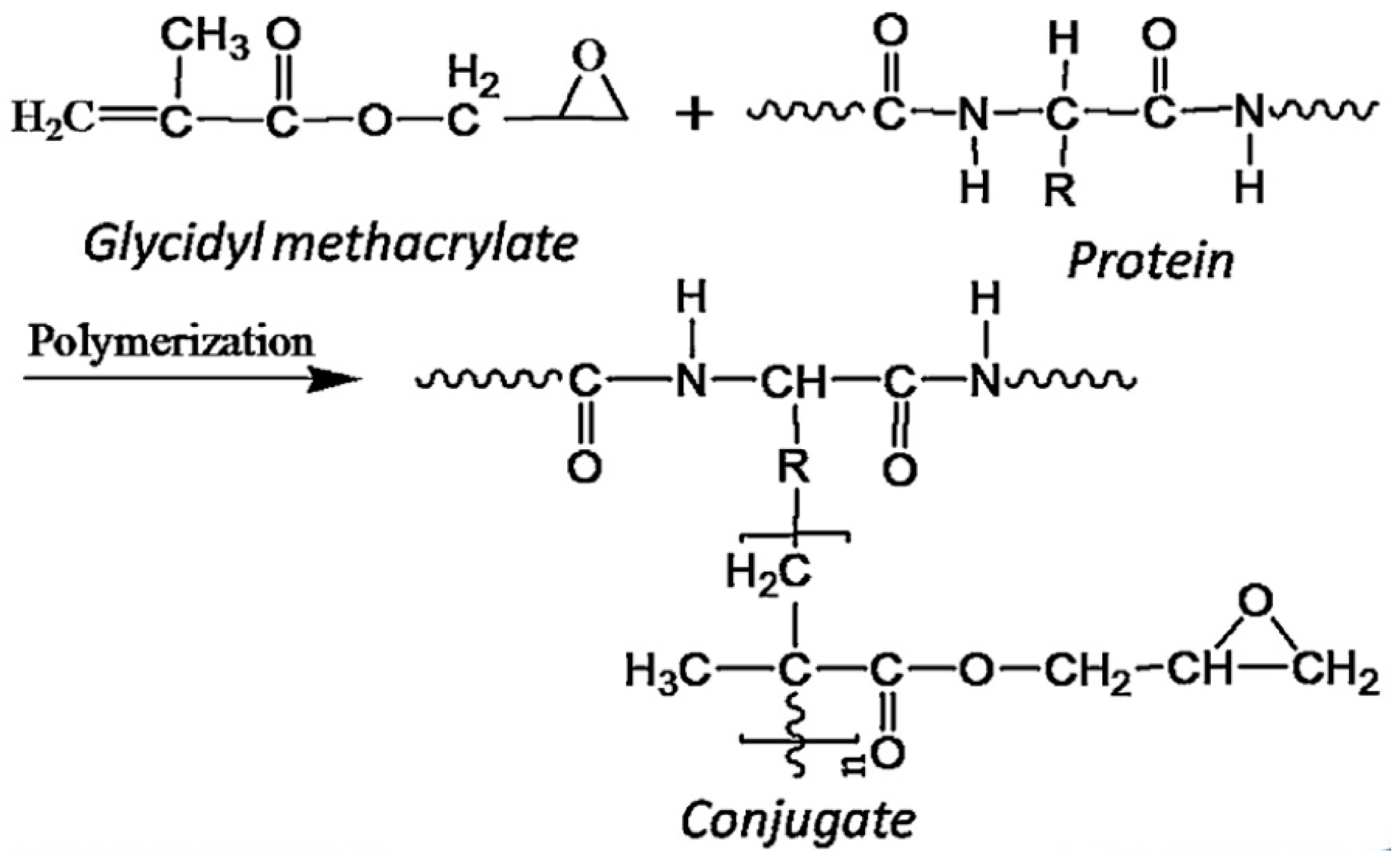
4.2. Canola Protein-Based Wood Adhesives
4.3. Cotton Protein-Based Wood Adhesives
4.4. Wheat Gluten-Based Wood Adhesives
5. Evaluation of Wood Adhesives
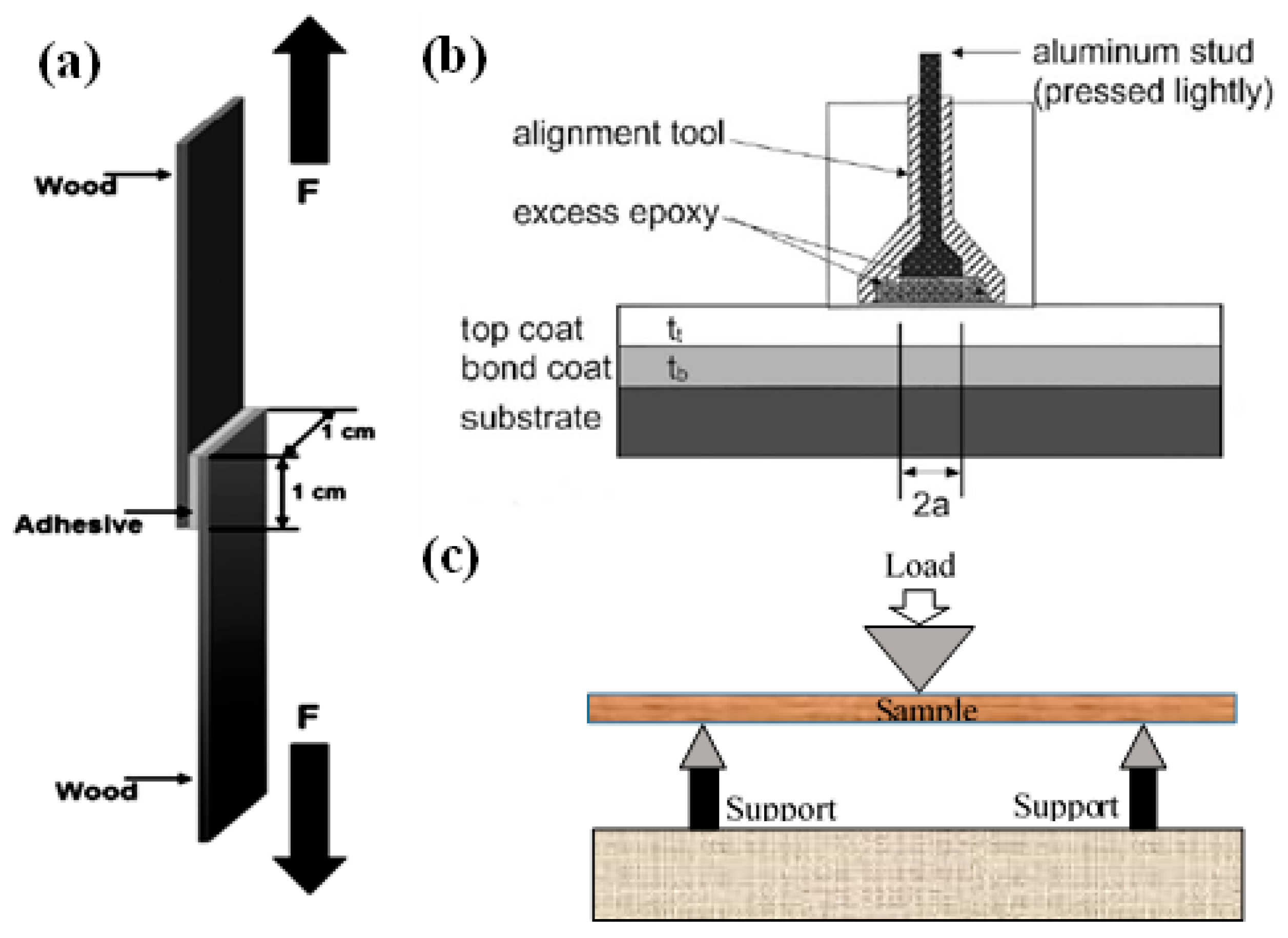
5.1. Wood Adhesive Bonding Strength Evaluation
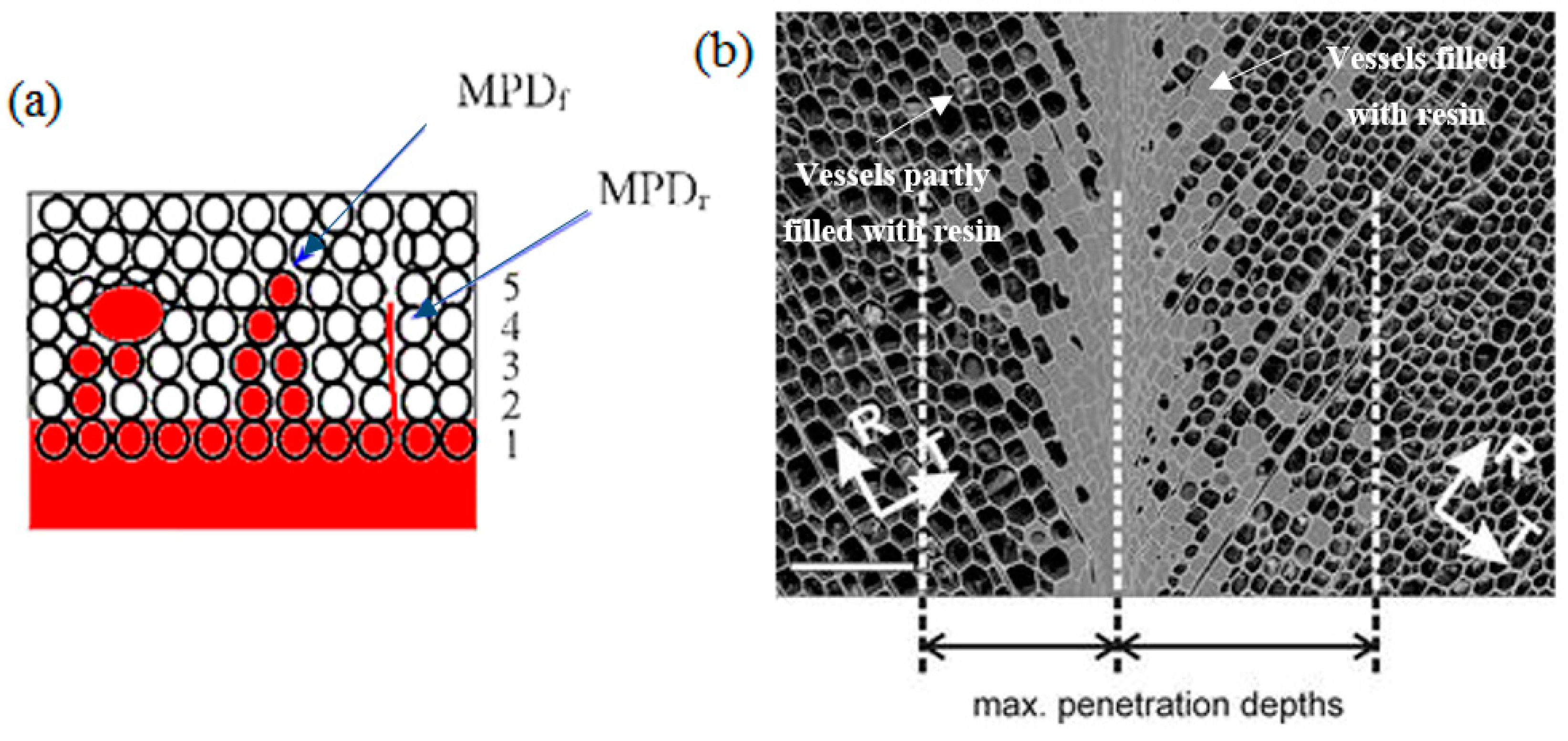
5.2. Wood Adhesive Penetration
5.2.1. Factors Affecting Wood Adhesive Penetration
5.2.2. Characterization Techniques of Wood Adhesive Penetration
6. Conclusions and Perspective
Acknowledgments
Conflicts of Interest
References
- Hüttermann, A.; Mai, C.; Kharazipour, A. Modification of lignin for the production of new compounded materials. Appl. Microbiol. Biotechnol. 2001, 55, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Youngquist, J.A. Wood-base Composites and Panel Products. In Wood Handbook: Wood as an Engineering Material; USDA Forest Service, Forest Products Laboratory: Madison, WI, USA, 1999; pp. 1–31. [Google Scholar]
- Moubarik, A.; Grimi, N.; Boussetta, N.; Pizzi, A. Isolation and characterization of lignin from Moroccan sugar cane bagasse: Production of lignin-phenol-formaldehyde wood adhesive. Ind. Crop. Prod. 2013, 45, 296–302. [Google Scholar] [CrossRef]
- Zhang, W.; Ma, Y.; Wang, C.; Li, S.; Zhang, M.; Chu, F. Preparation and properties of lignin-phenol-formaldehyde resins based on different biorefinery residues of agricultural biomass. Ind. Crop. Prod. 2013, 43, 326–333. [Google Scholar] [CrossRef]
- Jin, Y.; Cheng, X.; Zheng, Z. Preparation and characterization of phenol-formaldehyde adhesives modified with enzymatic hydrolysis lignin. Bioresour. Technol. 2010, 101, 2046–2048. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ma, Y.; Xu, Y.; Wang, C.; Chu, F. Lignocellulosic ethanol residue-based lignin-phenol-formaldehyde resin adhesive. Int. J. Adhes. Adhes. 2013, 40, 11–18. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, Y.; Yuan, T.-Q.; Sun, R.-C. Lignin-phenol-formaldehyde resin adhesives prepared with biorefinery technical lignins. J. Appl. Polym. Sci. 2015, 132, 1–8. [Google Scholar] [CrossRef]
- Roffael, E.; Dix, B. Lignin and ligninsulfonate in non-conventional bonding–An overview. Holz als Roh- und Werkst 1991, 49, 199–205. [Google Scholar] [CrossRef]
- He, G.; Yan, N. Effect of moisture content on curing kinetics of pMDI resin and wood mixtures. Int. J. Adhes. Adhes. 2005, 25, 450–455. [Google Scholar] [CrossRef]
- Kunaver, M.; Medved, S.; Čuk, N.; Jasiukaityte, E.; Poljanšek, I.; Strnad, T. Application of liquefied wood as a new particle board adhesive system. Bioresour. Technol. 2010, 101, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Klasnja, B.; Kopitovic, S. Lignin-phenol-formaldehyde resins as adhesive in the production of plywood. Holz als Roh- und Werkst 1992, 50, 282–285. [Google Scholar] [CrossRef]
- Pizzi, A. Recent developments in eco-efficient bio-based adhesives for wood bonding: Opportunities and issues. J. Adhes. Sci. Technol. 2006, 20, 829–846. [Google Scholar] [CrossRef]
- Wang, P.; Cheng, L.; Gu, Z.; Li, Z.; Hong, Y. Assessment of starch-based wood adhesive, quality by confocal Raman Microscopic detection of reaction homogeneity. Carbohydr. Polym. 2015, 131, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, J.; Li, C.; Gu, Z.; Cheng, L.; Hong, Y. Effects of montmorillonite addition on the performance of starch-based wood adhesive. Carbohydr. Polym. 2015, 115, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Nordqvist, P.; Thedjil, D.; Khosravi, S.; Lawther, M.; Malmstrom, E.; Khabbaz, F. Wheat Gluten Fractions as Wood Adhesives—Glutenins Versus Gliadins. J. Appl. Polym. Sci. 2012, 123, 1530–1538. [Google Scholar] [CrossRef]
- Santoni, I.; Pizzo, B. Evaluation of alternative vegetable proteins as wood adhesives. Ind. Crop. Prod. 2013, 45, 148–154. [Google Scholar] [CrossRef]
- Pan, Z.; Cathcart, A.; Wang, D. Properties of particleboard bond with rice bran and polymeric methylene diphenyl diisocyanate adhesives. Ind. Crop. Prod. 2006, 23, 40–45. [Google Scholar] [CrossRef]
- Trosa, A.; Pizzi, A. A no-aldehyde emission hardener for tannin-based wood adhesives for exterior panels. Holz als Roh- und Werkst 2001, 59, 266–271. [Google Scholar] [CrossRef]
- Feng, S.; Yuan, Z.; Leitch, M.; Xu, C.C. Adhesives formulated from bark bio-crude and phenol formaldehyde resole. Ind. Crop. Prod. 2015, 76, 258–268. [Google Scholar] [CrossRef]
- Feng, S.; Yuan, Z.; Leitch, M.; Shui, H.; Xu, C.C. Effects of bark extraction before liquefaction and liquid oil fractionation after liquefaction on bark-based phenol formaldehyde resoles. Ind. Crop. Prod. 2016, 84, 330–336. [Google Scholar] [CrossRef]
- Kong, X.; Liu, G.; Curtis, J.M. Characterization of canola oil based polyurethane wood adhesives. Int. J. Adhes. Adhes. 2011, 31, 559–564. [Google Scholar] [CrossRef]
- Kadla, J.F.; Kubo, S.; Venditti, R.A.; Gilbert, R.D.; Compere, A.L.; Griffith, W. Lignin-based carbon fibers for composite fiber applications. Carbon 2002, 40, 2913–2920. [Google Scholar] [CrossRef]
- Irvine, G.M. The significance of the glass transition of lignin in thermomechanical pulping. Wood Sci. Technol. 1985, 19, 139–149. [Google Scholar] [CrossRef]
- Vfizquez, G.; Gonzfilez, J.; Freire, S.; Antorrena, G. Effect of chemical modification of lignin on the gluebond performance of lignin-phenolic resins. Bioresour. Technol. 1997, 60, 191–198. [Google Scholar] [CrossRef]
- Tejado, A.; Peña, C.; Labidi, J.; Echeverria, J.M.; Mondragon, I. Physico-chemical characterization of lignins from different sources for use in phenol-formaldehyde resin synthesis. Bioresour. Technol. 2007, 98, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Leitch, M.; Xu, C.C. Synthesis of phenol–formaldehyde resol resins using organosolv pine lignins. Eur. Polym. J. 2009, 45, 3380–3388. [Google Scholar] [CrossRef]
- Benar, P.; Gonçalves, A.R.; Mandelli, D.; Schuchardt, U. Eucalyptus organosolv lignins: Study of the hydroxymethylation and use in resols. Bioresour. Technol. 1999, 68, 11–16. [Google Scholar] [CrossRef]
- Matsushita, Y.; Wada, S.; Fukushima, K.; Yasuda, S. Surface characteristics of phenol-formaldehyde-lignin resin determined by contact angle measurement and inverse gas chromatography. Ind. Crop. Prod. 2006, 23, 115–121. [Google Scholar] [CrossRef]
- Ahvazi, B.; Wojciechowicz, O.; Ton-That, T.-M.; Hawari, J. Preparation of lignopolyols from wheat straw soda lignin. J. Agric. Food Chem. 2011, 59, 10505–10516. [Google Scholar] [CrossRef] [PubMed]
- Ghaffar, S.H.; Fan, M. Lignin in straw and its applications as an adhesive. Int. J. Adhes. Adhes. 2014, 48, 92–101. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, Z.; Ye, L.; Ge, K.; Zhao, T. The production of lignin-phenol-formaldehyde resin derived carbon fibers stabilized by BN preceramic polymer. Mater. Lett. 2015, 142, 49–51. [Google Scholar] [CrossRef]
- Olivares, M.; Aceituno, H.; Neiman, G.; Rivera, E.; Sellers, T.J. lignin-modified phenolic adhesives for bonding radiata pine plywood. For. Prod. J. 1995, 45, 63–67. [Google Scholar]
- Pizzi, A. Wood products and green chemistry. Ann. For. Sci. 2016, 73, 185–203. [Google Scholar] [CrossRef]
- Newman, W.H.; Glasser, W.G. Engineering plastics from lignin XII. Synthesis and Performance of Lignin Adhesives with Isocyanate and Melamine. Holzforschung 1985, 39, 345–353. [Google Scholar] [CrossRef]
- Olivares, M.; Guzmán, J.A.; Natho, A.; Saavedra, A. Kraft lignin utilization in adhesives. Wood. Sci. Technol. 1988, 22, 157–165. [Google Scholar] [CrossRef]
- Gonçalves, A.R.; Benar, P. Hydroxymethylation and oxidation of Organosolv lignins and utilization of the products. Bioresour. Technol. 2001, 79, 103–111. [Google Scholar] [CrossRef]
- Çetin, N.S.; Özmen, N. Studies on lignin-based adhesives for particleboard panels. Turkish J. Agric. For. 2003, 27, 183–189. [Google Scholar]
- Qiao, W.; Li, S.; Guo, G.; Han, S.; Ren, S.; Ma, Y. Synthesis and characterization of phenol-formaldehyde resin using enzymatic hydrolysis lignin. J. Ind. Eng. Chem. 2014, 21, 1417–1422. [Google Scholar] [CrossRef]
- Ma, Y.; Zhao, X.; Chen, X.; Wang, Z. An approach to improve the application of acid-insoluble lignin from rice hull in phenol-formaldehyde resin. Colloids Surf. A Physicochem. Eng. Asp. 2011, 377, 284–289. [Google Scholar] [CrossRef]
- Zheng, Z.; Huang, Y.; Zou, J.; Zhang, H. Phenolation of using sulfuric acid as a catalyst and application to PF resin adhesives. J. Biotechnol. 2008, 136, S410–S411. [Google Scholar] [CrossRef]
- Tejado, A.; Kortaberria, G.; Labidi, J.; Echeverria, J.M.; Mondragon, I. Isoconversional kinetic analysis of novolac-type lignophenolic resins cure. Thermochim. Acta 2008, 471, 80–85. [Google Scholar] [CrossRef]
- Akhtar, T.; Lutfullah, G.; Ullah, Z. Ligonsulfonate-phenolformaldehyrde adhesive: A potenstial binder for wood panel industries. J. Chem. Soc. Pak. 2011, 33, 535–538. [Google Scholar]
- Domínguez, J.C.; Oliet, M.; Alonso, M.V.; Rojo, E.; Rodríguez, F. Structural, thermal and rheological behavior of a bio-based phenolic resin in relation to a commercial resol resin. Ind. Crop. Prod. 2013, 42, 308–314. [Google Scholar]
- El Mansouri, N.-E.; Pizzi, A.; Salvado, J. Lignin-based polycondensation resins for wood adhesives. J. Appl. Polym. Sci. 2007, 103, 1690–1699. [Google Scholar] [CrossRef]
- Peng, W.; Riedl, B. The chemorheology of phenol-formaldehyde thermoset resin and mixtures of the resin with lignin fillers. Polymer (Guildf) 1994, 35, 1280–1286. [Google Scholar] [CrossRef]
- Donmez Cavdar, A.; Kalaycioglu, H.; Hiziroglu, S. Some of the properties of oriented strandboard manufactured using kraft lignin phenolic resin. J. Mater. Process. Technol. 2008, 202, 559–563. [Google Scholar] [CrossRef]
- Siddiqui, H. Production of Lignin-based Phenolic Resin Using De-Polymerized Kraft Lignin and Process Optimization. Master’s Thesis, The University of Western Ontario, London, ON, Canada, 2013. [Google Scholar]
- Kouisni, L.; Fang, Y.; Paleologou, M.; Ahvazi, B.; Hawari, J.; Zhang, Y. Kraft lignin recovery and its use in the preparation of lignin-based phenol formaldehyde resins for plywood. Cellul. Chem. Technol. 2011, 45, 515–520. [Google Scholar]
- Park, Y.; Doherty, W.; Halley, P.J. Developing lignin-based resin coatings and composites. Ind. Crop. Prod. 2008, 27, 163–167. [Google Scholar] [CrossRef]
- Çetin, N.S.; Özmen, N. Use of organosolv lignin in phenol-formaldehyde resins for particleboard production: I. Organosolv lignin modified resins. Int. J. Adhes. Adhes. 2002, 22, 477–480. [Google Scholar] [CrossRef]
- Vázquez, G.; Rodríguez-Bona, C.; Freire, S.; González-Álvarez, J.; Antorrena, G. Acetosolv pine lignin as copolymer in resins for manufacture of exterior grade plywoods. Bioresour. Technol. 1999, 70, 209–214. [Google Scholar] [CrossRef]
- Dongre, P.; Driscoll, M.; Amidon, T.; Bujanovic, B. Lignin-furfural based adhesives. Energies 2015, 8, 7897–7914. [Google Scholar] [CrossRef]
- El Mansouri, N.E.; Pizzi, A.; Salvado, J. Lignin-based wood panel adhesives without formaldehyde. Holz als Roh- und Werkst 2007, 65, 65–70. [Google Scholar] [CrossRef]
- Lei, H.; Pizzi, A.; Du, G. Environmentally Friendly Mixed Tannin/Lignin Wood resins. J. Appl. Polym. Sci. 2008, 107, 203–209. [Google Scholar] [CrossRef]
- Navarrete, P.; Mansouri, H.R.; Pizzi, A.; Tapin-Lingua, S.; Benjelloun-Mlayah, B.; Pasch, H. Wood Panel Adhesives from Low Molecular Mass Lignin and Tannin without Synthetic Resins. J. Adhes. Sci. Technol. 2010, 24, 1597–1610. [Google Scholar] [CrossRef]
- Mansouri, H.R.; Navarrete, P.; Pizzi, A.; Tapin-Lingua, S.; Benjelloun-Mlayah, B.; Pasch, H. Synthetic-resin-free wood panel adhesives from mixed low molecular mass lignin and tannin. Eur. J. Wood Wood Prod. 2011, 69, 221–229. [Google Scholar] [CrossRef]
- Foyer, G.; Chanfi, B.H.; Boutevin, B.; Caillol, S.; David, G. New method for the synthesis of formaldehyde-free phenolic resins from lignin-based aldehyde precursors. Eur. Polym. J. 2016, 74, 296–309. [Google Scholar] [CrossRef]
- Foyer, G.; Chanfi, B.H.; Virieux, D.; David, G.; Caillol, S. Aromatic dialdehyde precursors from lignin derivatives for the synthesis of formaldehyde-free and high char yield phenolic resins. Eur. Polym. J. 2016, 77, 65–74. [Google Scholar] [CrossRef]
- Geng, X.; Li, K. Investigation of wood adhesives from kraft lignin and polyethylenimine. J. Adhes. Sci. Technol. 2006, 20, 847–858. [Google Scholar] [CrossRef]
- Liu, Y.; Li, K. Preparation and Characterization of Demethylated Lignin-Polyethylenimine Adhesives. J. Adhes. 2006, 82, 593–605. [Google Scholar] [CrossRef]
- Yuan, Y.; Guo, M.; Liu, F. Preparation and Evaluation of Green Composites Using Modified Ammonium Lignosulfonate and Polyethylenimine as a Binder. BioResources 2014, 9, 836–848. [Google Scholar] [CrossRef]
- Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N. Eco-friendly composite resins based on renewable biomass resources: Polyfurfuryl alcohol/lignin thermosets. Eur. Polym. J. 2010, 46, 1016–1023. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, F.; Lv, C.; Tang, L.; Wei, K.; Liu, X. Properties of formaldehyde-free environmentally friendly lignocellulosic composites made from poplar fibres and oxygen-plasma-treated enzymatic hydrolysis lignin. Compos. Part B Eng. 2013, 53, 369–375. [Google Scholar] [CrossRef]
- Xu, Q.; Wen, J.; Wang, Z. Preparation and Properties of Cassava Starch-based Wood Adhesives. BioResources 2016, 11, 6756–6767. [Google Scholar] [CrossRef]
- Qiao, Z.; Gu, J.; Lv, S.; Cao, J.; Tan, H.; Zhang, Y. Preparation and Properties of Normal Temperature Cured Starch-Based Wood Adhesive. BioResources 2016, 11, 4839–4849. [Google Scholar] [CrossRef]
- Tester, R.F.; Karkalas, J.; Qi, X. Starch—Composition, fine structure and architecture. J. Cereal Sci. 2004, 39, 151–165. [Google Scholar] [CrossRef]
- Buleon, A.; Colonna, P.; Planchot, V.; Ball, S. Starch granules: Structure and biosynthesis. Int. J. Biol. Macromol. 1998, 23, 85–112. [Google Scholar] [CrossRef]
- Parker, R.; Ring, S.G. Aspects of the Physical Chemistry of Starch. J. Cereal Sci. 2001, 34, 1–17. [Google Scholar] [CrossRef]
- Oates, C.G. Towards an understanding of Starch granule structure and hydrolysis. Trends Food Sci. Technol. 1997, 8, 375–382. [Google Scholar] [CrossRef]
- Huang, Z.; Lu, J.; Li, X.; Tong, Z. Effect of mechanical activation on physico-chemical properties and structure of cassava starch. Carbohydr. Polym. 2007, 68, 128–135. [Google Scholar] [CrossRef]
- Patel, K.F.; Mehta, H.U.; Srivastava, H.C. Kinetics and Mechanism of Oxidation of Starch with Sodium Hypochloride. J. Appl. Polym. Sci. 1974, 399, 389–399. [Google Scholar] [CrossRef]
- Gumul, D.; Gambuś, H.; Gibiński, M. Air Oxidation of Potato Starch Over Zinc (II) Catalyst. Carbohydr. Polym. 2005, 8, 45–50. [Google Scholar]
- Xie, F.; Yu, L.; Liu, H.; Chen, L. Starch modification using reactive extrusion. Starch 2006, 58, 131–139. [Google Scholar] [CrossRef]
- Siau, C.L.; Karim, A.A.; Norziah, M.H.; Wan Rosli, W.D. Effects of cationization on DSC thermal profiles, pasting and emulsifying properties of sago starch. J. Sci. Food Agric. 2004, 84, 1722–1730. [Google Scholar] [CrossRef]
- Emami, S.; Perera, A.; Meda, V.; Tyler, R.T. Effect of Microwave Treatment on Starch Digestibility and Physico-chemical Properties of Three Barley Types. Food Bioprocess Technol. 2012, 5, 2266–2274. [Google Scholar] [CrossRef]
- Lewandowicz, G.; Fornal, J.; Walkowski, A. Effect of microwave radiation on physico-chemical properties and structure of potato and tapioca starches. Carbohydr. Polym. 2000, 42, 193–199. [Google Scholar] [CrossRef]
- Luo, Z.; Fu, X.; He, X.; Luo, F.; Gao, Q.; Yu, S. Effect of ultrasonic treatment on the physicochemical properties of maize starches differing in amylose content. Starch 2008, 60, 646–653. [Google Scholar] [CrossRef]
- Czechowska-Biskup, R.; Rokita, B.; Lotfy, S.; Ulanski, P.; Rosiak, J.M. Degradation of chitosan and starch by 360-kHz ultrasound. Carbohydr. Polym. 2005, 60, 175–184. [Google Scholar] [CrossRef]
- Gunaratne, A.; Hoover, R. Effect of heat—Moisture treatment on the structure and physicochemical properties of tuber and root starches. Carbohydr. Polym. 2002, 49, 425–437. [Google Scholar] [CrossRef]
- Kaur, B.; Ariffin, F.; Bhat, R.; Karim, A.A. Progress in starch modification in the last decade. Food Hydrocoll. 2012, 26, 398–404. [Google Scholar] [CrossRef]
- Shariffa, Y.N.; Karim, A.A.; Fazilah, A.; Zaidul, I.S.M. Enzymatic hydrolysis of granular native and mildly heat-treated tapioca and sweet potato starches at sub-gelatinization temperature. Food Hydrocoll. 2009, 23, 434–440. [Google Scholar] [CrossRef]
- Chung, H.J.; Lim, H.S.; Lim, S.T. Effect of partial gelatinization and retrogradation on the enzymatic digestion of waxy rice starch. J. Cereal Sci. 2006, 43, 353–359. [Google Scholar] [CrossRef]
- Tan, H.; Zhang, Y.; Weng, X. Preparation of the plywood using starch-based adhesives modified with blocked isocyanates. Procedia Eng. 2011, 15, 1171–1175. [Google Scholar] [CrossRef]
- Onusseit, H. Starch in Industrial adhesives: New developments. Ind. Crop. Prod. 1993, 1, 141–146. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Z.; Gu, Z.; Hong, Y.; Cheng, L. Preparation, characterization and properties of starch-based wood adhesive. Carbohydr. Polym. 2012, 88, 699–706. [Google Scholar] [CrossRef]
- Zhou, L.; Han, C.; Zhang, S.; Gao, Q.; Li, H.; Li, J. Preparation and properties of environmental friendly wood adhesives based on wild acorn starch. Appl. Mech. Mater. 2011, 121–126, 2834–2838. [Google Scholar] [CrossRef]
- Yang, L.; Liu, J.; Du, C.; Qiang, Y. Preparation and Properties of Cornstarch Adhesive. Adv. J. Food Sci. Technol. 2013, 5, 1068–1072. [Google Scholar]
- Zhang, Y.; Ding, L.; Gu, J.; Tan, H.; Zhu, L. Preparation and properties of a starch-based wood adhesive with high bonding strength and water resistance. Carbohydr. Polym. 2015, 115, 32–57. [Google Scholar] [CrossRef] [PubMed]
- Amini, M.H.M.; Hashim, R.; Hiziroglu, S.; Sulaiman, N.S.; Sulaiman, O. Properties of particleboard made from rubberwood using modified starch as binder. Compos. Part B Eng. 2013, 50, 259–264. [Google Scholar] [CrossRef]
- Wang, S.; Shi, J.; Xu, W. Synthesis and Characterization of Starch-based Aqueous Polymer Isocyanate Wood Adhesive. BioResources 2015, 10, 7653–7666. [Google Scholar] [CrossRef]
- Imam, S.H.; Gordon, S.H.; Mao, L.; Chen, L. Environmentally friendly wood adhesive from a renewable plant polymer: Characteristics and optimization. Polym. Degrad. Stab. 2001, 73, 529–533. [Google Scholar] [CrossRef]
- Imam, S.H.; Mao, L.; Chen, L.; Greene, R.V. Wood Adhesive from Crosslinked Poly(Vinyl Alcohol) and Partially Gelatinized Starch: Preparation and Properties. Starch 1999, 51, 225–229. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L. Reaction mechanism of carboxymethyl starch-based wood adhesive. Comput. Model. New Technol. 2014, 18, 1150–1155. [Google Scholar]
- Kim, B.S.; Lim, S.-T. Removal of heavy metal ions from water by cross-linked carboxymethyl corn starch. Carbohydr. Polym. 1999, 39, 217–223. [Google Scholar] [CrossRef]
- Selamat, M.E.; Sulaiman, O.; Hashim, R.; Hiziroglu, S.; Nadhari, W.N.A.W.; Sulaiman, N.S. Measurement of some particleboard properties bonded with modified carboxymethyl starch of oil palm trunk. J. Int. Meas. Confed. 2014, 53, 251–259. [Google Scholar] [CrossRef]
- Moubarik, A.; Pizzi, A.; Allal, A.; Charrier, F.; Charrier, B. Cornstarch and tannin in phenol-formaldehyde resins for plywood production. Ind. Crop. Prod. 2009, 30, 188–193. [Google Scholar] [CrossRef]
- Moubarik, A.; Allal, A.; Pizzi, A.; Charrier, F.; Charrier, B. Characterization of a formaldehyde-free cornstarch-tannin wood adhesive for interior plywood. Eur. J. Wood Wood Prod. 2010, 68, 427–433. [Google Scholar] [CrossRef]
- Wang, Z.; Gu, Z.; Li, Z.; Hong, Y.; Cheng, L. Effects of urea on freeze—Thaw stability of starch-based wood adhesive. Carbohydr. Polym. 2013, 95, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Desai, S.D.; Patel, J.V.; Sinha, V.K. Polyurethane adhesive system from biomaterial-based polyol for bonding wood. Int. J. Adhes. Adhes. 2003, 23, 393–399. [Google Scholar] [CrossRef]
- Wang, Z.; Gu, Z.; Hong, Y.; Cheng, L.; Li, Z. Bonding strength and water resistance of starch-based wood adhesive improved by silica nanoparticles. Carbohydr. Polym. 2011, 86, 72–76. [Google Scholar] [CrossRef]
- Li, Z.; Wang, J.; Cheng, L.; Gu, Z.; Hong, Y.; Kowalczyk, A. Improving the performance of starch-based wood adhesive by using sodium dodecyl sulfate. Carbohydr. Polym. 2014, 99, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Qi, G.; Sun, X.S.; Stamm, M.J.; Wang, D. Physicochemical properties and adhesion performance of canola protein modified with sodium bisulfite. J. Am. Oil Chem. Soc. 2012, 89, 897–908. [Google Scholar] [CrossRef]
- Chen, M.; Chen, Y.; Zhou, X.; Lu, B.; He, M.; Sun, S. Improving Water Resistance of Soy-Protein Wood Adhesive by Using Hydrophilic Additives. Bioresources 2015, 10, 41–54. [Google Scholar] [CrossRef]
- Kalapathy, U.; Hettiarachchy, N.S.; Myers, D.; Hanna, M.A. Modification of Soy proteins and Their Adhesive Properties on Woods. JAOCS. J. Am. Oil Chem. Soc. 1995, 72, 507–510. [Google Scholar] [CrossRef]
- Li, X.; Li, Y.; Zhong, Z.; Wang, D.; Ratto, J.A.; Sheng, K. Mechanical and water soaking properties of medium density fiberboard with wood fiber and soybean protein adhesive. Bioresour. Technol. 2009, 100, 3556–3562. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, J.; Bernard, G.M. Preparation and characterization of canola protein isolate-poly(glycidyl methacrylate) conjugates: A bio-based adhesive. Ind. Crop. Prod. 2014, 57, 124–131. [Google Scholar] [CrossRef]
- He, Z.; Cheng, H.N.; Chapital, D.C.; Dowd, M.K. Sequential fractionation of cottonseed meal to improve its wood adhesive properties. J. Am. Oil Chem. Soc. 2014, 91, 151–158. [Google Scholar] [CrossRef]
- He, Z.; Chapital, D.C.; Cheng, H.N.; Thomas Klasson, K.; Olanya, O.M.; Uknalis, J. Application of tung oil to improve adhesion strength and water resistance of cottonseed meal and protein adhesives on maple veneer. Ind. Crop. Prod. 2014, 61, 398–402. [Google Scholar] [CrossRef]
- Nordqvist, P.; Nordgren, N.; Khabbaz, F.; Malmström, E. Plant proteins as wood adhesives: Bonding performance at the macro- and nanoscale. Ind. Crop. Prod. 2013, 44, 246–252. [Google Scholar] [CrossRef]
- D’Amico, S.; Müller, U.; Berghofer, E. Effect of hydrolysis and denaturation of wheat gluten on adhesive bond strength of wood joints. J. Appl. Polym. Sci. 2013, 129, 2429–2434. [Google Scholar] [CrossRef]
- Lei, H.; Du, G.; Wu, Z.; Xi, X.; Dong, Z. Cross-linked soy-based wood adhesives for plywood. Int. J. Adhes. Adhes. 2014, 50, 199–203. [Google Scholar] [CrossRef]
- Liu, Y.; Li, K. Development and characterization of adhesives from soy protein for bonding wood. Int. J. Adhes. Adhes. 2007, 27, 59–67. [Google Scholar] [CrossRef]
- Hettiarachchy, N.S.; Kalapathy, U.; Myers, D.J. Alkali-modified soy protein with improved adhesive and hydrophobic properties. J. Am. Oil Chem. Soc. 1995, 72, 1461–1464. [Google Scholar] [CrossRef]
- Rassam, G. Use of Soy/PF resin for Old Corrugated Container (OCC)-wood Composites. Mater. Lett. 2008, 62, 3236–3239. [Google Scholar] [CrossRef]
- Chen, N.; Lin, Q.; Rao, J.; Zeng, Q. Water resistances and bonding strengths of soy-based adhesives containing different carbohydrates. Ind. Crop. Prod. 2013, 50, 44–49. [Google Scholar] [CrossRef]
- Ciannamea, E.M.; Stefani, P.M.; Ruseckaite, R.A. Medium-density particleboards from modified rice husks and soybean protein concentrate-based adhesives. Bioresour. Technol. 2010, 101, 818–825. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, S.; Khabbaz, F.; Nordqvist, P.; Johansson, M. Protein-based adhesives for particleboards. Ind. Crop. Prod. 2010, 32, 275–283. [Google Scholar] [CrossRef]
- Li, N.; Qi, G.; Sun, X.S.; Wang, D. Effects of Sodium Bisulfite on the Physicochemical and Adhesion Properties of Canola Protein Fractions. J. Polym. Environ. 2012, 20, 905–915. [Google Scholar] [CrossRef]
- Cheng, H.N.; Ford, C.; Dowd, M.K.; He, Z. Use of additives to enhance the properties of cottonseed protein as wood adhesives. Int. J. Adhes. Adhes. 2016, 68, 156–160. [Google Scholar] [CrossRef]
- Cheng, H.N.; Ford, C.; Dowd, M.K.; He, Z. Soy and cottonseed protein blends as wood adhesives. Ind. Crop. Prod. 2016, 85, 324–330. [Google Scholar] [CrossRef]
- Khosravi, S.; Nordqvist, P.; Khabbaz, F.; Johansson, M. Protein-based adhesives for particleboards-Effect of application process. Ind. Crop. Prod. 2011, 34, 1509–1515. [Google Scholar] [CrossRef]
- Somord, K.; Tawichai, N.; Soykeabkaew, N. Bio-based adhesive from wheat gluten. In Proceedings of The 26th Annual Meeting of the Thai Society for Biotechnology and International Conference, Chiang Rai, Thailand, 26–29 November 2014.
- El-Wakil, N.A.; Abou-Zeid, R.E.; Fahmy, Y.; Mohamed, A.Y. Modified Wheat Gluten as a Binder in Particleboard Made from Reed. J. Appl. Phys. 2007, 106, 3592–3599. [Google Scholar] [CrossRef]
- Lagel, M.C.; Pizzi, A.; Redl, A.; Al-Marzouki, F.M. Phenol-wheat protein-formaldehyde thermoset wood adhesives. Eur. J. Wood Wood Prod. 2015, 73, 439–448. [Google Scholar] [CrossRef]
- Nordqvist, P.; Lawther, M.; Malmström, E.; Khabbaz, F. Adhesive properties of wheat gluten after enzymatic hydrolysis or heat treatment–A comparative study. Ind. Crop. Prod. 2012, 38, 139–145. [Google Scholar] [CrossRef]
- Akyildiz, M.H.; Kesik, H.İ. Effect of heat treatment on the adhesion strength of water based wood varnishes. Wood Res. 2015, 60, 987–994. [Google Scholar]
- Heon Kwon, J.; Lee, S.H.; Ayrilmis, N.; Hyung Han, T. Tensile shear strength of wood bonded with urea-formaldehyde with different amounts of microfibrillated cellulose. Int. J. Adhes. Adhes. 2015, 60, 88–91. [Google Scholar] [CrossRef]
- Ozdemir, T.; Hiziroglu, S.; Kocapnar, M. Adhesion strength of cellulosic varnish coated wood species as function of their surface roughness. Adv. Mater. Sci. Eng. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Cheng, L.; Guo, H.; Gu, Z.; Li, Z.; Hong, Y. Effects of compound emulsifiers on properties of wood adhesive with high starch content. Int. J. Adhes. Adhes. 2017, 72, 92–97. [Google Scholar] [CrossRef]
- Kohl, J.G.; Singer, I.L. Pull-off behavior of epoxy bonded to silicone duplex coatings. Prog. Org. Coat. 1999, 36, 15–20. [Google Scholar] [CrossRef]
- Ramos, N.M.M.; Simões, M.L.; Delgado, J.M.P.Q.; De Freitas, V.P. Reliability of the pull-off test for in situ evaluation of adhesion strength. Constr. Build. Mater. 2012, 31, 86–93. [Google Scholar] [CrossRef]
- Ozdemir, T.; Hiziroglu, S.; Kocapinar, M. Effect of Heartwood and Sapwood Ratio on Adhesion Strength of Finished Wood Species. Adv. Mater. Sci. Eng. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Gaff, M.; Gasparik, M. Influence of densification on bending strength of laminated beech wood. BioResources 2015, 10, 1506–1518. [Google Scholar] [CrossRef]
- Örs, Y.; Atar, M.; Keskin, H. Bonding strength of some adhesives in wood materials impregnated with Imersol-Aqua. Int. J. Adhes. Adhes. 2004, 24, 287–294. [Google Scholar] [CrossRef]
- Mckinley, P.E.; Ching, D.J.; Kamke, F.A.; Zauner, M.; Xiao, X. Micro X-Ray Computed Tomography of Adhesive Bonds in Wood. Wood Fiber Sci. 2016, 48, 2–16. [Google Scholar]
- Kläusler, O.; Clauß, S.; Lübke, L.; Trachsel, J.; Niemz, P. Influence of moisture on stress-strain behaviour of adhesives used for structural bonding of wood. Int. J. Adhes. Adhes. 2013, 44, 57–65. [Google Scholar] [CrossRef]
- Song, Y.H.; Seo, J.H.; Choi, Y.S.; Kim, D.H.; Choi, B.H.; Cha, H.J. Mussel adhesive protein as an environmentally-friendly harmless wood furniture adhesive. Int. J. Adhes. Adhes. 2016, 70, 260–264. [Google Scholar] [CrossRef]
- Adamopoulos, S.; Bastani, A.; Gascón-Garrido, P.; Militz, H.; Mai, C. Adhesive bonding of beech wood modified with a phenol formaldehyde compound. Eur. J. Wood Wood Prod. 2012, 70, 897–901. [Google Scholar] [CrossRef]
- Wei, S.; Shi, J.; Gu, J.; Wang, D.; Zhang, Y. Dynamic wettability of wood surface modified by acidic dyestuff and fixing agent. Appl. Surf. Sci. 2012, 258, 1995–1999. [Google Scholar] [CrossRef]
- Li, X.; Luo, J.; Gao, Q.; Li, J. A sepiolite-based united cross-linked network in a soybean meal-based wood adhesive and its performance. RSC Adv. 2016, 6, 45158–45165. [Google Scholar] [CrossRef]
- Davalos, J.F.; Qiao, P.Z.; Trimble, B.S. Fiber-reinforced composite and wood bonded interfaces: Part 1. Durability and shear strength. J. Compos. Technol. Res. 2000, 22, 224–231. [Google Scholar]
- Ebewele, R.; River, B.; Koutsky, J. Tapered Double Cantilever Beam Fracture Tests of Phenolic-Wood Adhesive Joints. Wood Fiber Sci. 1979, 11, 197–213. [Google Scholar]
- Custódio, J.; Broughton, J.; Cruz, H. A review of factors influencing the durability of structural bonded timber joints. Int. J. Adhes. Adhes. 2009, 29, 173–185. [Google Scholar] [CrossRef]
- Frangi, A.; Fontana, M.; Mischler, A. Shear behaviour of bond lines in glued laminated timber beams at high temperatures. Wood Sci. Technol. 2004, 38, 119–126. [Google Scholar] [CrossRef]
- Kamke, F.A.; Lee, J.N. Adhesive penetration in wood: A review. Wood Fiber Sci. 2007, 39, 5–20. [Google Scholar]
- Vallée, T.; Tannert, T.; Fecht, S. Adhesively Bonded Connections in the Context of Timber Engineering—A Review. J. Adhes. 2015, 93, 257–287. [Google Scholar] [CrossRef]
- Haag, A.P.; Maier, R.M.; Combie, J.; Geesey, G.G. Bacterially derived biopolymers as wood adhesives. Int. J. Adhes. Adhes. 2004, 24, 495–502. [Google Scholar] [CrossRef]
- Liu, D.; Chen, H.; Chang, P.R.; Wu, Q.; Li, K.; Guan, L. Biomimetic soy protein nanocomposites with calcium carbonate crystalline arrays for use as wood adhesive. Bioresour. Technol. 2010, 101, 6235–6241. [Google Scholar] [CrossRef] [PubMed]
- Hass, P.; Wittel, F.K.; Mendoza, M.; Herrmann, H.J.; Niemz, P. Adhesive penetration in beech wood: Experiments. Wood Sci. Technol. 2012, 46, 243–256. [Google Scholar] [CrossRef]
- Mendoza, M.; Hass, P.; Wittel, F.K.; Niemz, P.; Herrmann, H.J. Adhesive penetration in Beech wood Part II: Penetration Model. Wood Sci. Technol. 2010, 46, 529–549. [Google Scholar] [CrossRef]
- Follrich, J.; Vay, O.; Veigel, S.; Müller, U. Bond strength of end-grain joints and its dependence on surface roughness and adhesive spread. J Wood Sci. 2010, 56, 429–434. [Google Scholar] [CrossRef]
- Gardner, D.J. Chapter 19-Adhesion Mechanisms of Durable Wood Adhesive Bonds. Charact. Cellul. Cell Wall 2005, 254–266. [Google Scholar]
- Stelte, W.; Holm, J.K.; Sanadi, A.R.; Barsberg, S.; Ahrenfeldt, J.; Henriksen, U.B. A study of bonding and failure mechanisms in fuel pellets from different biomass resources. Biomass Bioenergy 2011, 35, 910–918. [Google Scholar] [CrossRef]
- Shi, S.Q.; Gardner, D.J. Dynamic Adhesive Wettability of Wood. Wood Fiber Sci. 2001, 33, 58–68. [Google Scholar]
- Gavrilovic-Grmusa, I.; Miljkovic, J.; Djiporovic-Momcilovic, M.; Radosevic, G. Penetration of urea formaldehyde adhesives in wood tissue, Part II: Radial penetration of UF adhesives into silver fir. Glas Umarskog Fak 2008, 98, 39–48. [Google Scholar] [CrossRef]
- Singh, A.P.; Anderson, C.R.; Warnes, J.M.; Matsumura, J. The effect of planing on the microscopic structure of Pinus radiata wood cells in relation to penetration of PVA glue. Holz als Roh- und Werkst 2002, 60, 333–341. [Google Scholar] [CrossRef]
- Edalat, H.; Faezipour, M.; Thole, V.; Kamke, F.A. A new quantitative method for evaluation of adhesive penetration pattern in particulate wood-based composites: Elemental counting method. Wood Sci. Technol. 2014, 48, 703–712. [Google Scholar] [CrossRef]
- Johnson, S.E.; Kamke, F.A. Quantitative analysis of gross adhesive penetration in wood using fluorescence microscopy. J. Adhes. 1992, 40, 47–61. [Google Scholar] [CrossRef]
- Paris, J.L.; Kamke, F.A. Quantitative wood–adhesive penetration with X-ray computed tomography. Int. J. Adhes. Adhes. 2015, 61, 71–80. [Google Scholar] [CrossRef]
- Konnerth, J.; Harper, D.; Lee, S.H.; Rials, T.G.; Gindl, W. Adhesive penetration of wood cell walls investigated by scanning thermal microscopy (SThM). Holzforschung 2008, 62, 91–98. [Google Scholar] [CrossRef]
- Wang, W.; Yan, N. Characterizing liquid resin penetration in wood using a mercury intrusion porosimeter. Wood Fiber Sci. 2005, 37, 505–513. [Google Scholar]
- Follrich, J.; Teischinger, A.; Gindl, W.; Müller, U. Effect of grain angle on shear strength of glued end grain to flat grain joints of defect-free softwood timber. Wood Sci. Technol. 2007, 41, 501–509. [Google Scholar] [CrossRef]
- Laborie, M.-P.P.G.; Salmén, L.; Frazier, C.E.; Salmen, L.; Frazier, C.E. A morphological study of the wood/phenol-formaldehyde adhesive interphase. J. Adhes. Sci. Technol. 2006, 20, 729–741. [Google Scholar] [CrossRef]
- Jakes, J.E.; Hunt, C.G.; Yelle, D.J.; Lorenz, L.; Hirth, K.; Gleber, S.C. Synchrotron-based X-ray fluorescence microscopy in conjunction with nanoindentation to study molecular-scale interactions of phenol-formaldehyde in wood cell walls. ACS Appl. Mater. Interfaces 2015, 7, 6584–6589. [Google Scholar] [CrossRef] [PubMed]
- Veigel, S.; Müller, U.; Keckes, J.; Obersriebnig, M.; Gindl-Altmutter, W. Cellulose nanofibrils as filler for adhesives: Effect on specific fracture energy of solid wood-adhesive bonds. Cellulose 2011, 18, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Cheng, E.Z.; Sun, X. Effects of wood-surface roughness, adhesive viscosity and processing pressure on adhesion strength of protein adhesive. J. Adhes. Sci. Technol. 2006, 20, 997–1017. [Google Scholar] [CrossRef]
- Kučerová, I. Methods to measure the penetration of consolidant solutions into “dry” wood. J. Cult. Herit 2012, 13, 191–195. [Google Scholar] [CrossRef]
- Modzel, G.; Kamke, F.A.; De Carlo, F. Comparative analysis of a wood: Adhesive bondline. Wood Sci. Technol. 2011, 45, 147–158. [Google Scholar] [CrossRef]
- Gavrilović-Grmuša, I.; Dunky, M.; Djiporović-Momčilović, M.; Popović, M.; Popović, J. Influence of pressure on the radial and tangential penetration of adhesive resin into poplar wood and on the shear strength of adhesive joints. BioResources 2016, 11, 2238–2255. [Google Scholar] [CrossRef]
- Cyr, P.-L.; Riedl, B.; Wang, X.-M. Investigation of Urea-Melamine-Formaldehyde (UMF) resin penetration in Medium-Density Fiberboard (MDF) by High Resolution Confocal Laser Scanning Microscopy. Eur J. Wood Wood Prod. 2008, 66, 129–134. [Google Scholar] [CrossRef]
- Ren, D.; Frazier, C.E. Wood/adhesive interactions and the phase morphology of moisture-cure polyurethane wood adhesives. Int. J. Adhes. Adhes. 2012, 34, 55–61. [Google Scholar] [CrossRef]
- Bastani, A.; Adamopoulos, S.; Koddenberg, T.; Militz, H. Study of adhesive bondlines in modified wood with fluorescence microscopy and X-ray micro-computed tomography. Int. J. Adhes. Adhes. 2016, 68, 351–358. [Google Scholar] [CrossRef]
- Pakdel, H.; Cyr, P.L.; Riedl, B.; Deng, J. Quantification of urea formaldehyde resin in wood fibers using X-ray photoelectron spectroscopy and confocal laser scanning microscopy. Wood Sci. Technol. 2008, 42, 133–148. [Google Scholar] [CrossRef]
- Bolton, A.J.; Dinwoodie, J.M.; Davies, D.A. The validity of the use of SEM/EDAX as a tool for the detection of UF resin penetration into wood cell walls in particleboard. Wood Sci. Technol. 1988, 22, 345–356. [Google Scholar] [CrossRef]
- Egerton, R. Limitations of Electron Energy-Loss Spectroscopy. Microsc. Microanal. 2006, 12, 102–103. [Google Scholar] [CrossRef]
- Kim, M.G. Examination of Selected Synthesis Parameters for Typical Wood Adhesive-Type Urea—Formaldehyde Resins by 13 C-NMR Spectroscopy. I. J. Polym. Sci. Part A Polym. Chem. 1999, 37, 995–1007. [Google Scholar] [CrossRef]
- Wang, Y.; Spencer, P. Quantifying adhesive penetration in adhesive/dentin interface using confocal Raman microspectroscopy. J. Biomed. Mater. Res. 2002, 59, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Konnerth, J.; Valla, A.; Gindl, W. Nanoindentation mapping of a wood-adhesive bond. Appl. Phys. A Mater. Sci. Process. 2007, 88, 371–375. [Google Scholar] [CrossRef]
- Paris, J.L.; Kamke, F.A.; Xiao, X. X-ray computed tomography of wood-adhesive bondlines: Attenuation and phase-contrast effects. Wood Sci. Technol. 2015, 49, 1185–1208. [Google Scholar] [CrossRef]
- Gierlinger, N.; Hansmann, C.; Röder, T.; Sixta, H.; Gindl, W.; Wimmer, R. Comparison of UV- and confocal Raman microscopy to measure the melamine-formaldehyde resin content within cell walls of impregnated spruce wood. Holzforschung 2005, 59, 210–213. [Google Scholar] [CrossRef]

| Type of Bio-Adhesive | Advantages | Disadvantages |
|---|---|---|
| Lignin-based wood adhesive | Utilization of by-products from paper pulping industries Improving the thermal properties Improving the modulus of elasticity Improving the water resistance Good bonding strength | Low level of substitution (max lignin content ≈ 50 wt %) Decreasing the curing rate Increasing the viscosity of adhesive Need chemical modification to improve its reactivity Solubility depends on the type of lignin |
| Starch-based wood adhesive | High level of substitution Good bonding strength Good film formation property | Poor water resistance Slow drying process Poor storage stability Need pre-treatment to improve the water resistance |
| Plant protein-based wood adhesive | High level of substitution Good adhesion strength Improving thermal stability | Need pre-treatment to improve the water resistance Poor water resistance |
| Technique | Application | Advantages | Disadvantages | Reference |
|---|---|---|---|---|
| Scanning probe microscopy (SPM)/nanoindentation | Cell-wall penetration | Adhesives penetration map | Modulus difference required, two techniques combination | [136,164,178] |
| Transmitted microscopy | Gross-penetration | Rapid, quantitative evaluation, color contrast | Specimen preparation difficult | |
| Fluorescent microscopy | Gross-penetration | High color contrast, color filtering, rapid, quantitative measurement | Fluorescer stain | [136,155,158,164,172] |
| Fluorescent infrared spectroscopy (FTIR) | Gross-penetration | Chemical bonding | No penetration depth and bond line information | [171] |
| Confocal laser scanning microscopy (CLSM) | Gross-penetration Cell-wall penetration | Adhesive distribution and penetration, 3D view, penetration to single fiber | Low scanning speed, low resolution in Z-direction, image damage | [170,173] |
| X-ray photoelectron spectroscopy (XPS) | Cell-wall penetration | Penetration to single fiber, quantification measurement | No penetration depth and bond line information, limitation in large scale | [148,169] |
| Scanning electron microscopy (SEM)/energy-dispersive X-ray analysis (EDAX) | Gross-penetration Cell-wall penetration | Adhesive distribution, penetration, bond line morphology | Gray image, poor contrast, quantitative measurement difficult, large excitation volume | [150,174] |
| Transmission electron microscopy (TEM) | Cell-wall penetration | Adhesive penetration, bond line morphology, morphology of diffusion in cell wall | Gray image, poor contrast, quantitative measurement difficult, slow | [174] |
| Scanning thermal microscopy (SThM) | Cell-wall penetration | Distribution at bond line area, high spatial resolution, simple specimen preparation, specimen preparation is simple | Rely on thermal conductivity difference, assisted by AFM, resolution depend on surface height variation, smaller image size | [149,160] |
| Electron energy loss spectroscopy (EELS) | Gross-penetration Cell-wall penetration | Monitoring penetration, high resolution, adhesive distribution | Combine with transmission electron microscopy, expansive, slow, radiation damage | [161,175] |
| 13C CP/MAS NMR | Gross-penetration Cell-wall penetration | Nanoscale observation, cell-wall penetration, relationship of molecular weight and penetration | Lack of morphology analysis, distribution and penetration depth | [152,171,176] |
| X-ray microtomography (XMT) | Gross-penetration Cell-wall penetration | 3D view, pattern of adhesive | Gray image, poor contrast | [168] |
| Micro X-ray computed tomography (XmCT) | Gross-penetration Cell-wall penetration | Adhesive distribution, penetration, high resolution, 3D view | Size limitation, pre-treatment of adhesives | [135,172,179] |
| Raman spectroscopy | Cell-wall penetration | Higher lateral resolution, interlayer composition measurement, adhesive diffusion detection | Not suitable for bond line morphology characterization, penetration depth and adhesive distribution | [177,180] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferdosian, F.; Pan, Z.; Gao, G.; Zhao, B. Bio-Based Adhesives and Evaluation for Wood Composites Application. Polymers 2017, 9, 70. https://doi.org/10.3390/polym9020070
Ferdosian F, Pan Z, Gao G, Zhao B. Bio-Based Adhesives and Evaluation for Wood Composites Application. Polymers. 2017; 9(2):70. https://doi.org/10.3390/polym9020070
Chicago/Turabian StyleFerdosian, Fatemeh, Zihe Pan, Guchuhan Gao, and Boxin Zhao. 2017. "Bio-Based Adhesives and Evaluation for Wood Composites Application" Polymers 9, no. 2: 70. https://doi.org/10.3390/polym9020070
APA StyleFerdosian, F., Pan, Z., Gao, G., & Zhao, B. (2017). Bio-Based Adhesives and Evaluation for Wood Composites Application. Polymers, 9(2), 70. https://doi.org/10.3390/polym9020070






