Fully Biodegradable Biocomposites with High Chicken Feather Content
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Alkali Treatment of the CFs
2.3. Biocomposite Preparation
2.4. Characterization of the Biocomposites
2.4.1. Density of the Biocomposites
2.4.2. Water Absorption of Biocomposites
2.4.3. Thermogravimetric Analysis (TGA)
2.4.4. Dynamic Scanning Calorimetry (DSC)
2.4.5. Mechanical Testing
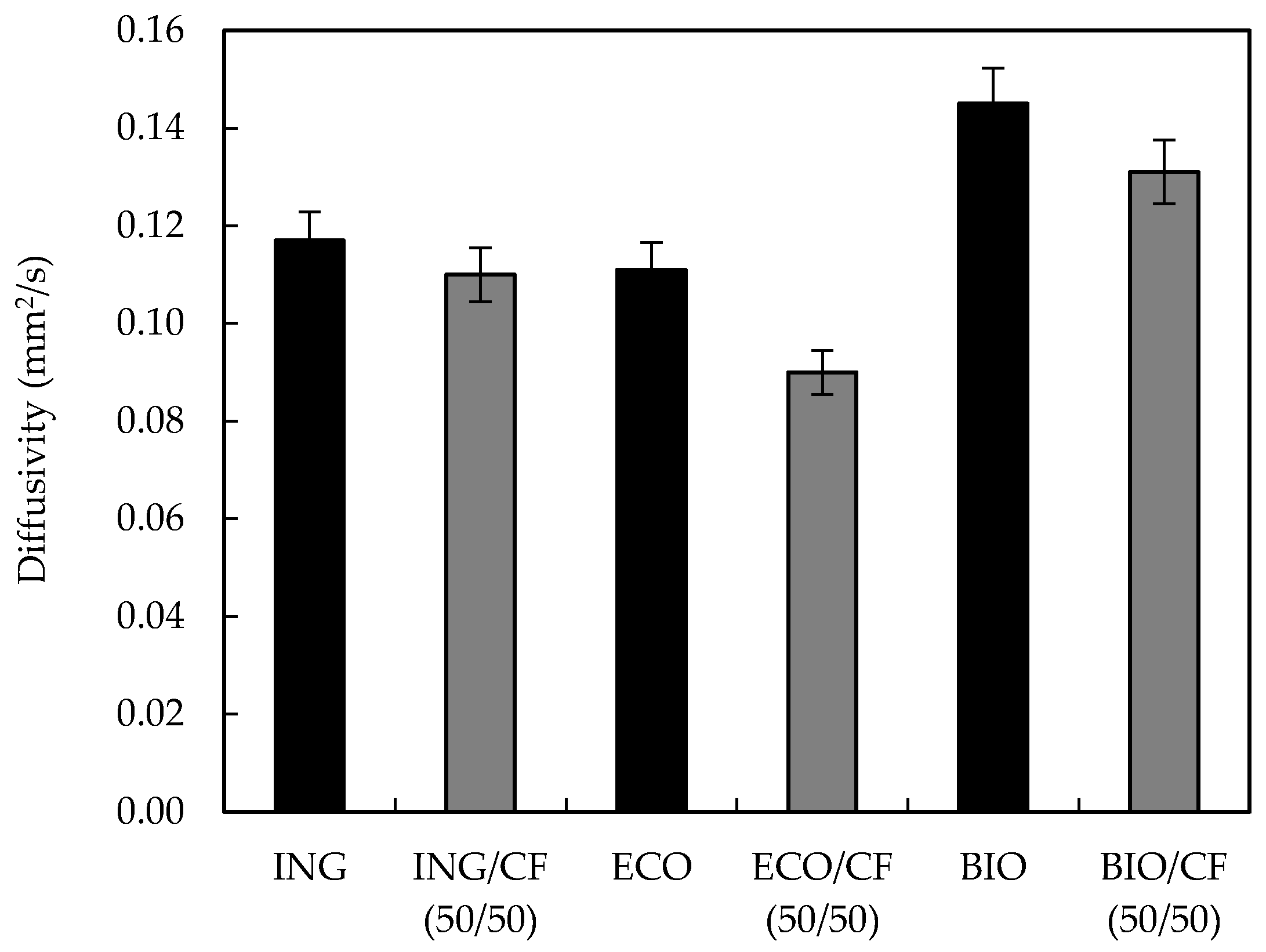
2.4.6. Thermal Diffusivity
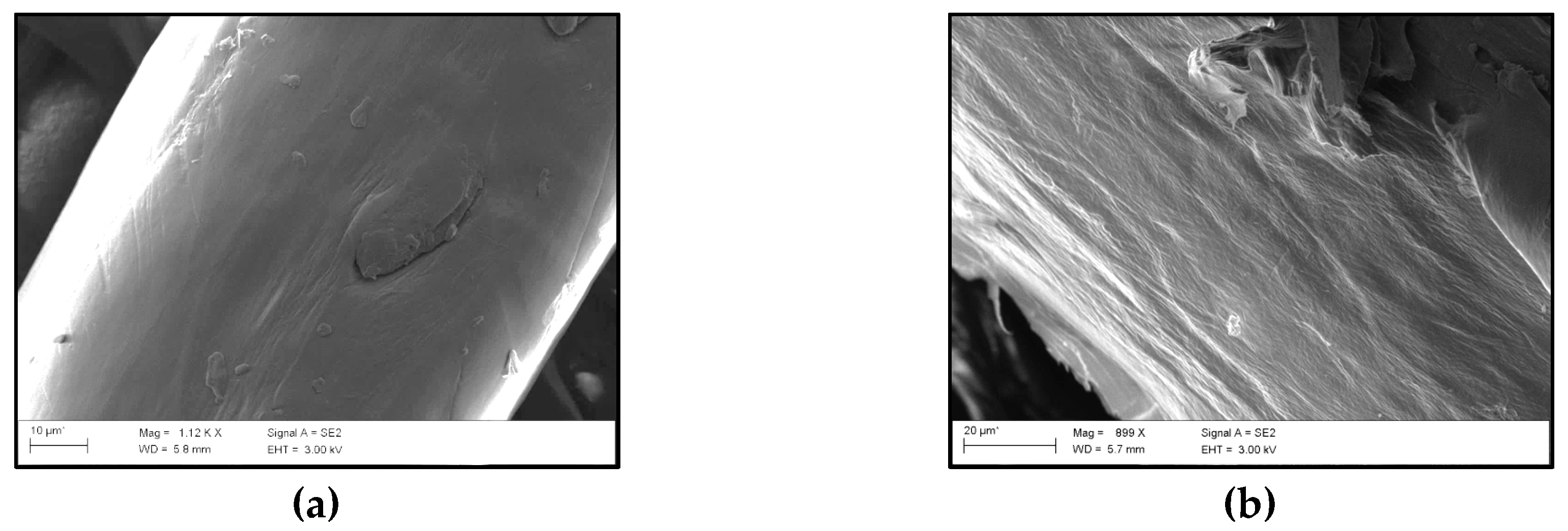
2.4.7. FE–SEM
3. Results and Discussions
3.1. Density
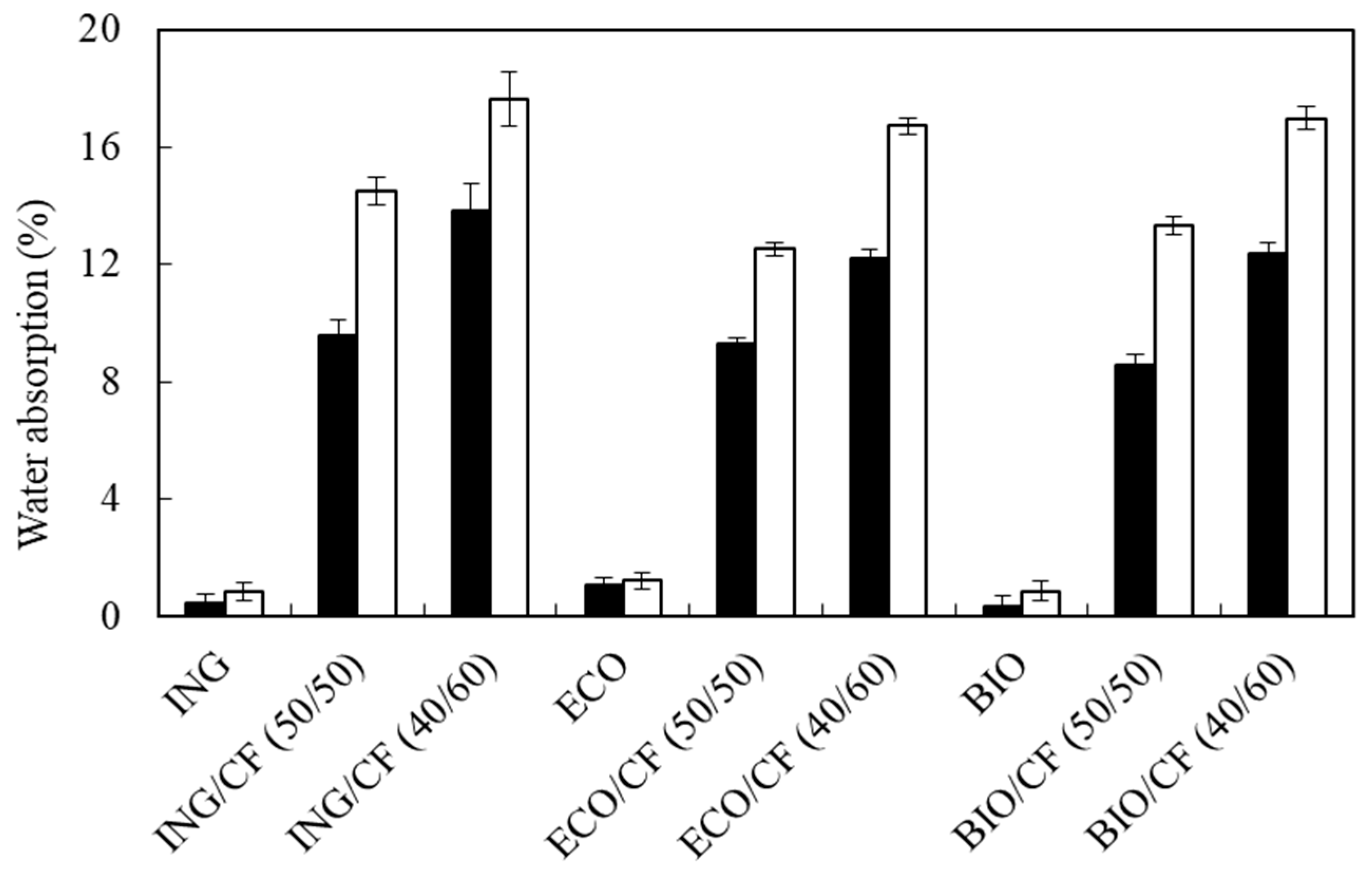
3.2. Water Absorption
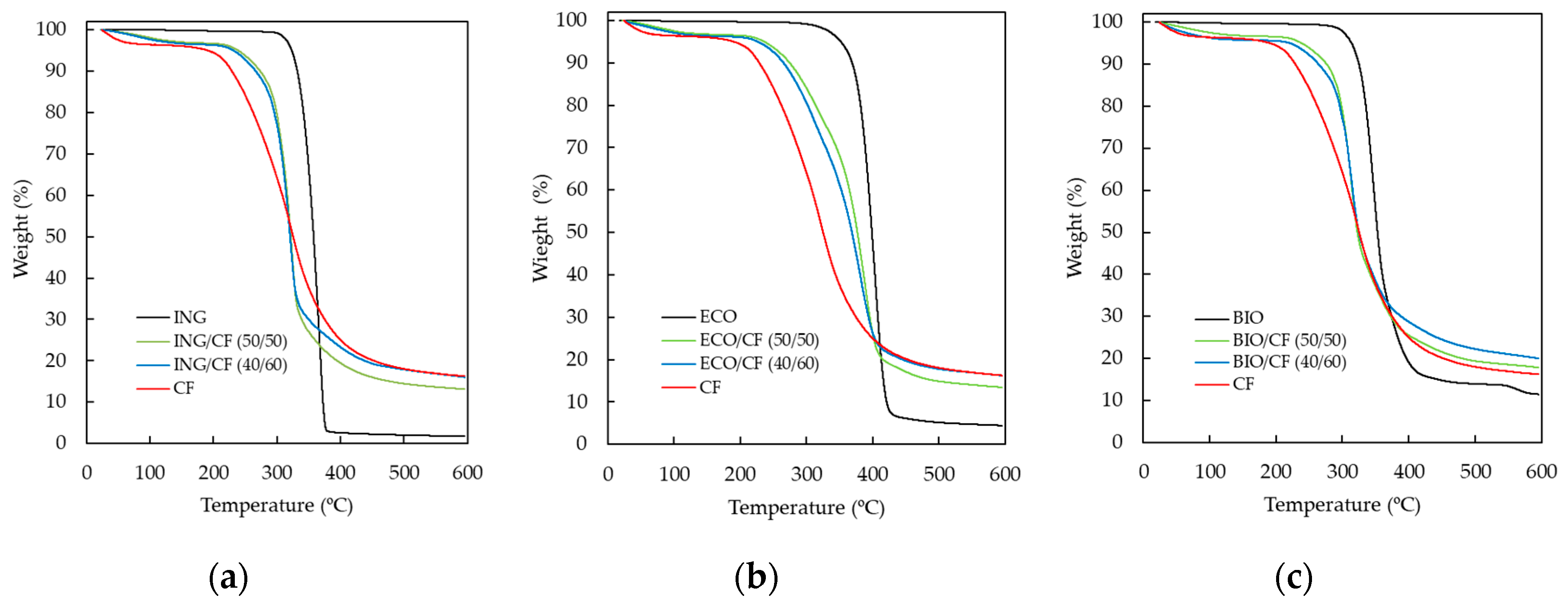
3.3. Thermogravimetric Analysis (TGA)
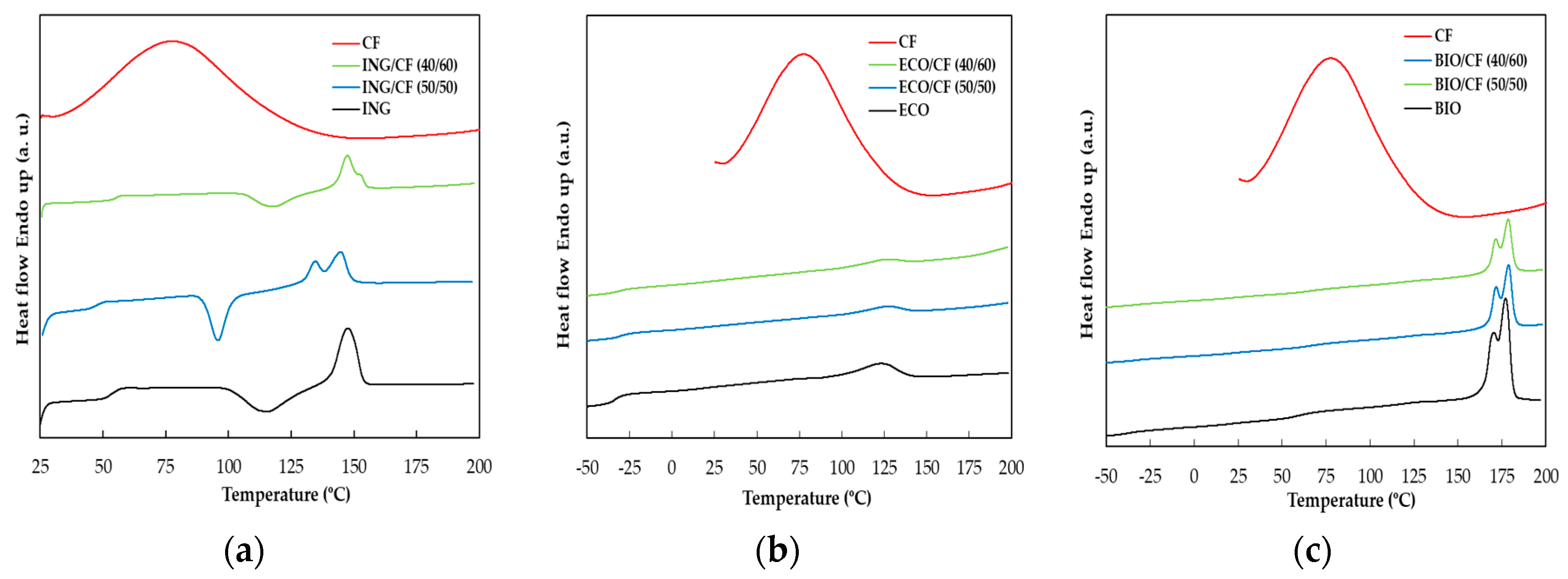
3.4. DSC Analysis
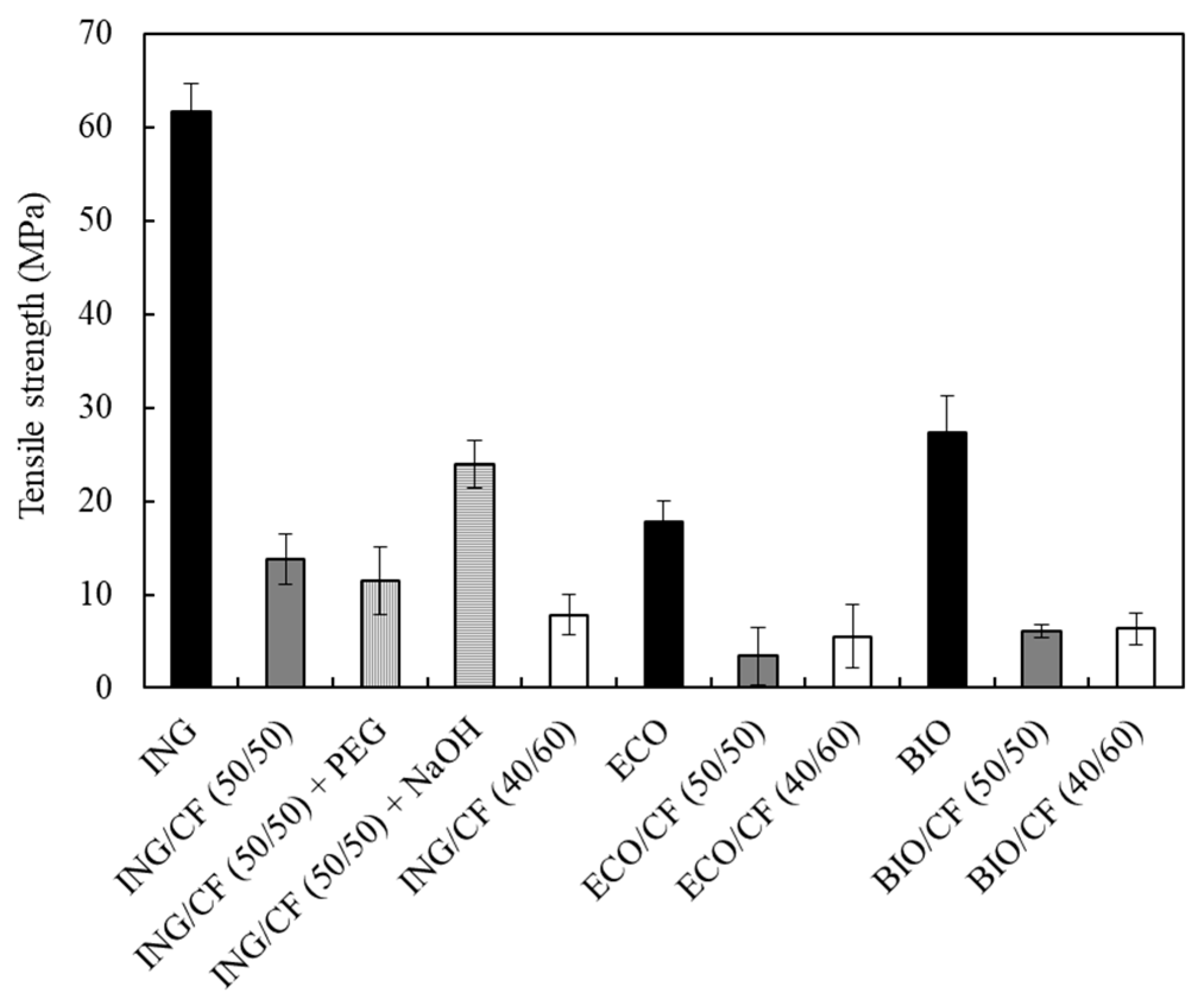
3.5. Mechanical Properties of the Biocomposites
3.6. Morphology of the Biocomposites
3.7. Thermal Diffusivity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mohanty, A.; Misra, M.; Drzal, L. Natural Fibers, Biopolymers and Biocomposites; CRC Press, Taylor & Francis: Boca Ratón, FL, USA, 2005; pp. 1–36. ISBN 9780849317415. [Google Scholar]
- Pickering, K.L.; Efendy, M.G.A.; Le, T.M. A review of recent developments in natural fibre composite and their mechanical performance. Compos. Part A Appl. Sci. Manuf. 2016, 83, 98–112. [Google Scholar] [CrossRef]
- Conzatti, L.; Giunco, F.; Stagnaro, P.; Patrucco, A.; Marano, C.; Rink, M.; Marsano, E. Composites based on polypropylene and short wool fibres. Compos. Part A Appl. Sci. Manuf. 2013, 47, 165–171. [Google Scholar] [CrossRef]
- Alam, A.K.M.M.; Shubhra, Q.T.H.; Al-Imran, G.; Barai, S.; Islam, M.R.; Rahman, M.M. Preparation and characterization of natural silk fiber-reinforced polypropylene and synthetic E-glass fiber-reinforced polypropylene composites: A comparative study. J. Compos. Mater. 2011, 45, 2301–2308. [Google Scholar] [CrossRef]
- Kumar, D.; Boopathy, S.R.; Sangeetha, D.; Bharathiraja, G. Investigation of Mechanical Properties of Horn Powder-Filled Epoxy Composite. J. Mech. Eng. 2017, 63, 138–147. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, R.; Zhang, Y.; Wu, L. Sustainable and practical utilization of feather keratin by a novel physicochemical pretreatment: High density steam flash-explosion. Green Chem. 2012, 14, 3352–3360. [Google Scholar] [CrossRef]
- European Commission. Available online: http://ec.europa.eu/agriculture/poultry_en (accessed on 9 November 2017).
- Korniłłowicz-Kowalska, T.; Bohacz, J. Biodegradation of keratin waste: Theory and practical aspects. Waste Manag. 2011, 31, 1689–1701. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N. Non-food industrial applications of poultry feathers. Waste Manag. 2015, 45, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Rastogi, S.; Terry, A.E.; Popescu, C. Self-organization of polypeptides obtained on dissolution of feather keratins in superheated water. Biomacromolecules 2007, 8, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.C.; Li, F.Y.; He, Y.F.; Wang, Y.; Wang, R.M. Study on effective extraction of chicken feather keratin and their films for controlling drug release. Biomater. Sci. 2013, 1, 528–536. [Google Scholar] [CrossRef]
- Schmidt, W.F.; Line, M.J. Physical and chemical structures of poultry feather fiber fractions in fiber process development. In Proceedings of the 1996 TAPPI Nonwovens Conference, Charlotte, NC, USA, 11–13 March 1996; pp. 135–141. [Google Scholar]
- Qin, X. Chicken Feather Fibre Mat/PLA Composites for Thermal Insulation. Master’s Thesis, University of Waikato, Hamilton, New Zealand, 2015. [Google Scholar]
- Acda, M.N. Waste chicken feather as reinforcement in cement-bonded composites. Philipp. J. Sci. 2010, 139, 161–166. [Google Scholar]
- Saucedo-Rivalcoba, V.; Martínez-Hernández, A.L.; Martínez-Barrera, G.; Velasco-Santos, C.; Castaño, V.M. (Chicken feathers keratin)/polyurethane membranes. Appl. Phys. A 2011, 104, 219–228. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Light-weight polypropylene composites reinforced with whole chicken feathers. J. Appl. Polym. Sci. 2010, 116, 3668–3675. [Google Scholar] [CrossRef]
- Huda, S.; Yang, Y. Composites from ground quill and polypropylene. Compos. Sci. Technol. 2008, 68, 790–798. [Google Scholar] [CrossRef]
- Jiménez-Cervantes, E.; Velasco-Santos, C.; Martínez-Hernández, A.L.; Rivera-Armenta, J.L.; Mendoza-Martínez, A.M.; Castaño, V.M. Composites from chicken feathers quill and recycled polypropylene. J. Compos. Mater. 2015, 49, 275–283. [Google Scholar]
- Yang, Y.; Reddy, N. Utilizing discarded plastic bags as matrix material for composites reinforced with chicken feathers. J. Appl. Polym. Sci. 2013, 130, 307–312. [Google Scholar] [CrossRef]
- Cañavate, J.; Aymerich, J.; Garrido, N.; Colom, X.; Macanás, J.; Molins, G.; Álvarez, M.D.; Carrillo, F. Properties and optimal manufacturing conditions of chicken feather/poly(lactic acid) biocomposites. J. Compos. Mater. 2016, 50, 1671–1683. [Google Scholar] [CrossRef]
- Cheng, S.; Lau, K.T.; Liu, T.; Zhao, Y.; Lam, P.M.; Yin, Y. Mechanical and thermal properties of chicken feather fiber/PLA Green composites. Compos. Part B Eng. 2009, 40, 650–654. [Google Scholar] [CrossRef]
- Baba, B.O.; Özmen, U. Preparation and mechanical characterization of chicken feather/PLA composites. Polym. Compos. 2015, 38, 837–845. [Google Scholar] [CrossRef]
- Reddy, N.; Jiang, J.; Yang, Y. Biodegradable composites containing chicken feathers as matrix and jute fibers as reinforcement. J. Polym. Environ. 2014, 22, 310–317. [Google Scholar] [CrossRef]
- Arai, K.M.; Takahashi, R.; Yokote, Y.; Akahane, K. Amino-acid sequence of feather keratin from fowl. Eur. J. Biochem. 1983, 132, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Huda, M.S.; Schmidt, W.F.; Misra, M.; Drzal, L.T. Effect of fiber surface treatment of poultry feather fibers on the properties of their polymer matrix composites. J. Appl. Polym. Sci. 2013, 128, 1117–1124. [Google Scholar] [CrossRef]
- Ghani, S.A.; Tan, S.J.; Yeng, T.S. Properties of chicken feather fiber-filled low density polyethylene composites: The effect of polyethylene grafted maleic anhydride. Polym. Plast. Technol. Eng. 2013, 52, 495–500. [Google Scholar] [CrossRef]
- Kuma, P.; Bajpai, P.K.; Singh, I.; Madaan, J. Development and characterization of PLA-based green composites: A review. J. Thermoplast. Compos. Mater. 2012, 10, 1–30. [Google Scholar]
- Masutani, K.; Kimura, Y. PLA Synthesis. From the Monomer to the Polymer. In Poly(lactic acid) Science and Technology: Processing, Properties, Additives and Applications; RSC: London, UK, 2014. [Google Scholar]
- Martin, O.; Avérous, L. Poly(lactic acid): Plasticization and properties of biodegradable multiphase system. Polymer 2001, 45, 6209–6219. [Google Scholar] [CrossRef]
- Bordes, P.; Pollet, E.; Avérous, L. Nano-biocomposites: Biodegradable polyester/nanoclay systems. Prog. Polym. Sci. 2009, 34, 125–155. [Google Scholar] [CrossRef]
- Briassoulis, D.; Babou, E.; Hiskakis, M. Degradation Behaviour and Field Performance of Experimental Biodegradable Drip Irrigation Systems. J. Polym. Environ. 2011, 19, 341–361. [Google Scholar] [CrossRef]
- Islam, M.S.; Pickering, K.L.; Foreman, N.J. Influence of Alkali Treatment on the Interfacial and Physico-Mechanical Properties of Industrial Hemp Fibre Reinforced Polylactic Acid Composites. Compos. Part A Appl. Sci. Manuf. 2010, 41, 596–603. [Google Scholar] [CrossRef]
- ISO 9427:2003. Wood-Based Panels. Determination of Density; International Organization for Standardization: Geneva, Switzerland, 2003.
- ASTM D570-98. Standard Test Method for Water Absorption of Plastics; ASTM International: West Conshohocken, PA, USA, 2010.
- ISO 527-1:2012. Plastics—Determination of Tensile Properties; International Organization for Standardization: Geneva, Switzerland, 2012.
- ASTM E1461-13. Standard Test Method for Thermal Diffusivity by the Flash Method; ASTM International: West Conshohocken, PA, USA, 2013.
- Barone, J.R.; Schmidt, W.F. Polyethylene reinforced with keratin fibers obtained from chicken feathers. Compos. Sci. Technol. 2005, 65, 173–181. [Google Scholar] [CrossRef]
- Carrillo, F.; Rahalli, A.; Cañavate, J.; Colom, X. Composites from keratin biofibers. Study of compatibility using polyolephinic matrices. In Proceedings of the 15th European Conference on Composite Materials (ECCM-15) Conference, Venice, Italy, 24–28 June 2012; pp. 1–15. [Google Scholar]
- Carrillo, F.; Rahhali, A.; Cañavate, J.; Colom, X. Biocomposites using waste whole chicken feathers and thermoplastic matrices. J. Reinf. Plast. Compos. 2013, 32, 1419–1429. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Structure and Properties of Chicken Feather Barbs as Natural Protein Fibers. J. Polym. Environ. 2007, 15, 81–87. [Google Scholar] [CrossRef]
- Barone, J.R.; Gregoire, N.T. Characterization of fibre-polymer interactions and transcrystallity in short keratin fiber-polypropylene composites. Plast. Rubber Compos. 2006, 35, 287–293. [Google Scholar] [CrossRef]
- Martínez-Hernández, A.L.; Velasco-Santos, C.; de Icaza, M.; Castaño, V.M. Microstructural characterization of keratin fibres from chicken feathers. Int. J. Environ. Pollut. 2005, 23, 162–178. [Google Scholar] [CrossRef]
- Takahashi, K.; Yakamoto, H.; Yokote, Y.; Hattori, M. Thermal Behavior of Fowl Feather Keratin. Biosci. Biotechnol. Biochem. 2004, 68, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Huda, M.S.; Drzal, L.T.; Mohanty, A.K.; Misra, M. Chopped glass and recycled newspaper as reinforcement fibers in injection molded poly(lactic acid) (PLA) composites: A comparative study. Compos. Sci. Technol. 2006, 66, 1813–1824. [Google Scholar] [CrossRef]
- Houshyar, S.; Shanks, R.A.; Hodzic, A. The effect of fiber concentration on mechanical and thermal properties of fiber-reinforced polypropylene composites. J. Appl. Polym. Sci. 2005, 96, 2260–2272. [Google Scholar] [CrossRef]
- Cheung, H.Y.; Lau, K.T.; Tao, X.M.; Hui, D. A potential material for tissue engineering: Silkworm silk/PLA biocomposite. Compos. Part B Eng. 2008, 39, 1026–1033. [Google Scholar] [CrossRef]
- Ghani, S.A.; Jahari, M.H.; Soo-Jin, T. Effects of sodium hydroxide treatment on the properties of low-density polyethylene composites filled with chicken feather fiber. J. Vinyl Addit. Technol. 2014, 20, 36–41. [Google Scholar] [CrossRef]
- Bismarck, A.; Mohanty, A.K.; Aranberri-Askargorta, I.; Czapla, S.; Misra, M.; Hinrichsen, G.; Springer, J. Surface characterization of natural fibers; surface properties and the water up-take behavior of modified sisal and coir fibers. Green Chem. 2001, 3, 100–107. [Google Scholar] [CrossRef]
- Winandy, J.E.; Muehl, J.H.; Micales, J.A.; Raina, A.; Schmidt, W. Potential of chicken feather fibre in wood MDF composites. In Proceedings of the EcoComp, London, UK, 1–2 September 2003; p. 20. [Google Scholar]
- Peter, S. Material Revolution: Sustainable and Multi-Purpose Materials for Design and Architecture; Birkhäuser: Basel, Switzerland, 2011; ISBN 13:978-3034606639. [Google Scholar]





| Polymer (Sample Code) | Type of Matrix | Melting Temperature (°C) | Density (g/cm3) | Supplier |
|---|---|---|---|---|
| Ingeo 2003D (ING) | PLA | 145–160 | 1.24 | NatureWorks (Minnetonka, MN, USA) |
| Ecoflex C1200 (ECO) | PBAT | 120 | 1.25–1.27 | BASF (Ludwigshafen, Germany) |
| Bio-Flex 6611 (BIO) | PLA/copolyester blend | 150–170 | 1.29 | FKuR (Willich, Germany) |
| Matrix | Polymer/CF Ratio | Density (g/cm3) | % of Density Reduction |
|---|---|---|---|
| Compared to Neat Polymer | |||
| ING | 100/0 | 1.16 | - |
| 50/50 | 1.10 | 5.17 | |
| 40/60 | 1.00 | 13.79 | |
| ECO | 100/0 | 1.22 | - |
| 50/50 | 1.06 | 13.11 | |
| 40/60 | 1.01 | 17.21 | |
| BIO | 100/0 | 1.28 | - |
| 50/50 | 1.08 | 15.63 | |
| 4960 | 1.02 | 20.31 |
| Samples | T (5%) (°C) | T (25%) (°C) | T (50%) (°C) |
|---|---|---|---|
| CF | 191 | 276 | 325 |
| ING | 323 | 345 | 358 |
| ING/CF (50/50) | 238 | 305 | 322 |
| ING/CF (40/60) | 231 | 302 | 321 |
| ECO | 352 | 386 | 398 |
| ECO/CF (50/50) | 236 | 330 | 377 |
| ECO/CF (40/60) | 227 | 316 | 369 |
| BIO | 314 | 339 | 353 |
| BIO/CF (50/50) | 239 | 304 | 323 |
| BIO/CF (40/60) | 221 | 303 | 325 |
| Sample | Tg1 (°C) | Tg2 (°C) | ΔHcc (J/g) | Tm (°C) | ΔHm (J/g) | χ (%) |
|---|---|---|---|---|---|---|
| ING | 53.6 | 19.5 | 147.6 | 22.7 | 3.48 | |
| ING/CF (50/50) | 47.4 | 15.2 | 144.6 | 17.9 | 5.78 | |
| ING/CF (40/60) | 55.6 | 9.00 | 147.4 | 11.8 | 7.50 | |
| ECO | −34.1 | 120.4 | 9.8 | 8.60 | ||
| ECO/CF (50/50) | −30.4 | 116.8 | 4.3 | 7.54 | ||
| ECO/CF (40/60) | −30.7 | 116.6 | 2.7 | 5.92 | ||
| BIO | −37.8 | 58.7 | 176.9 | 28.6 | 30.5 | |
| BIO/CF (50/50) | −33.1 | 65.6 | 178.7 | 14.4 | 30.7 | |
| BIO/CF (40/60) | −32.1 | 64.8 | 178.5 | 13.5 | 36.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aranberri, I.; Montes, S.; Azcune, I.; Rekondo, A.; Grande, H.-J. Fully Biodegradable Biocomposites with High Chicken Feather Content. Polymers 2017, 9, 593. https://doi.org/10.3390/polym9110593
Aranberri I, Montes S, Azcune I, Rekondo A, Grande H-J. Fully Biodegradable Biocomposites with High Chicken Feather Content. Polymers. 2017; 9(11):593. https://doi.org/10.3390/polym9110593
Chicago/Turabian StyleAranberri, Ibon, Sarah Montes, Itxaso Azcune, Alaitz Rekondo, and Hans-Jürgen Grande. 2017. "Fully Biodegradable Biocomposites with High Chicken Feather Content" Polymers 9, no. 11: 593. https://doi.org/10.3390/polym9110593
APA StyleAranberri, I., Montes, S., Azcune, I., Rekondo, A., & Grande, H.-J. (2017). Fully Biodegradable Biocomposites with High Chicken Feather Content. Polymers, 9(11), 593. https://doi.org/10.3390/polym9110593







