Stereocomplexation, Thermal and Mechanical Properties of Conetworks Composed of Star-Shaped l-Lactide, d-Lactide and ε-Caprolactone Oligomers Utilizing Sugar Alcohols as Core Molecules
Abstract
:1. Introduction
2. Experimental Section
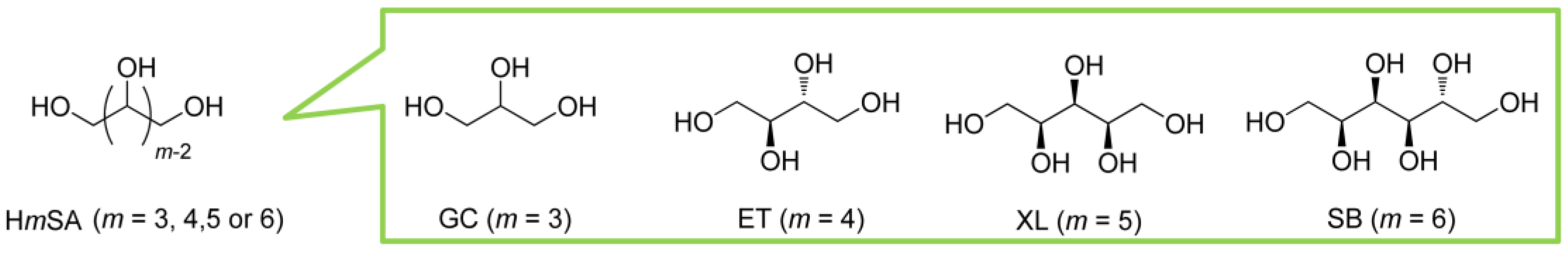
2.1. Materials and Reagents
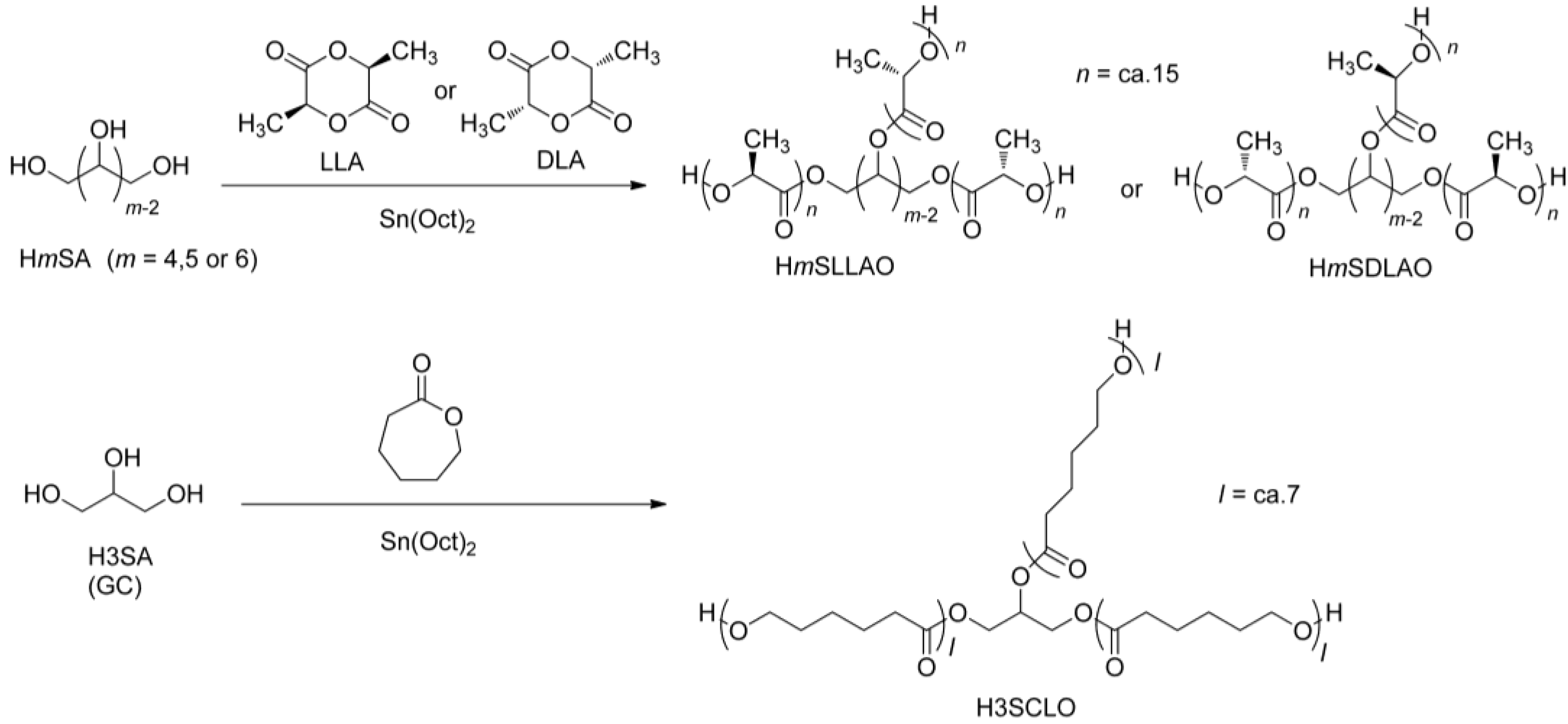
2.2. Syntheses of HmSLLAO and HmSDLAO (m = 4, 5 or 6)
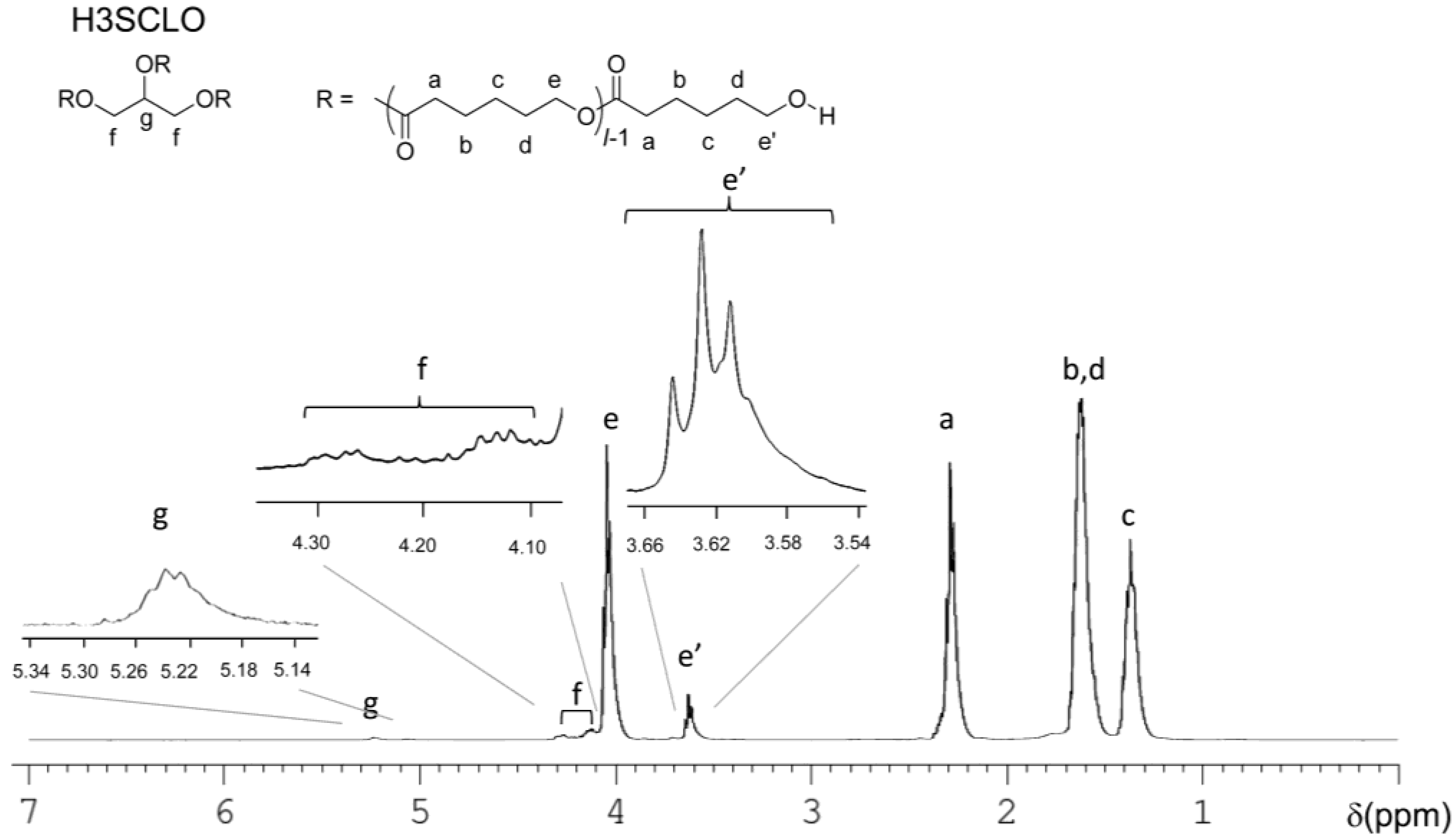
2.3. Synthesis of H3SCLO
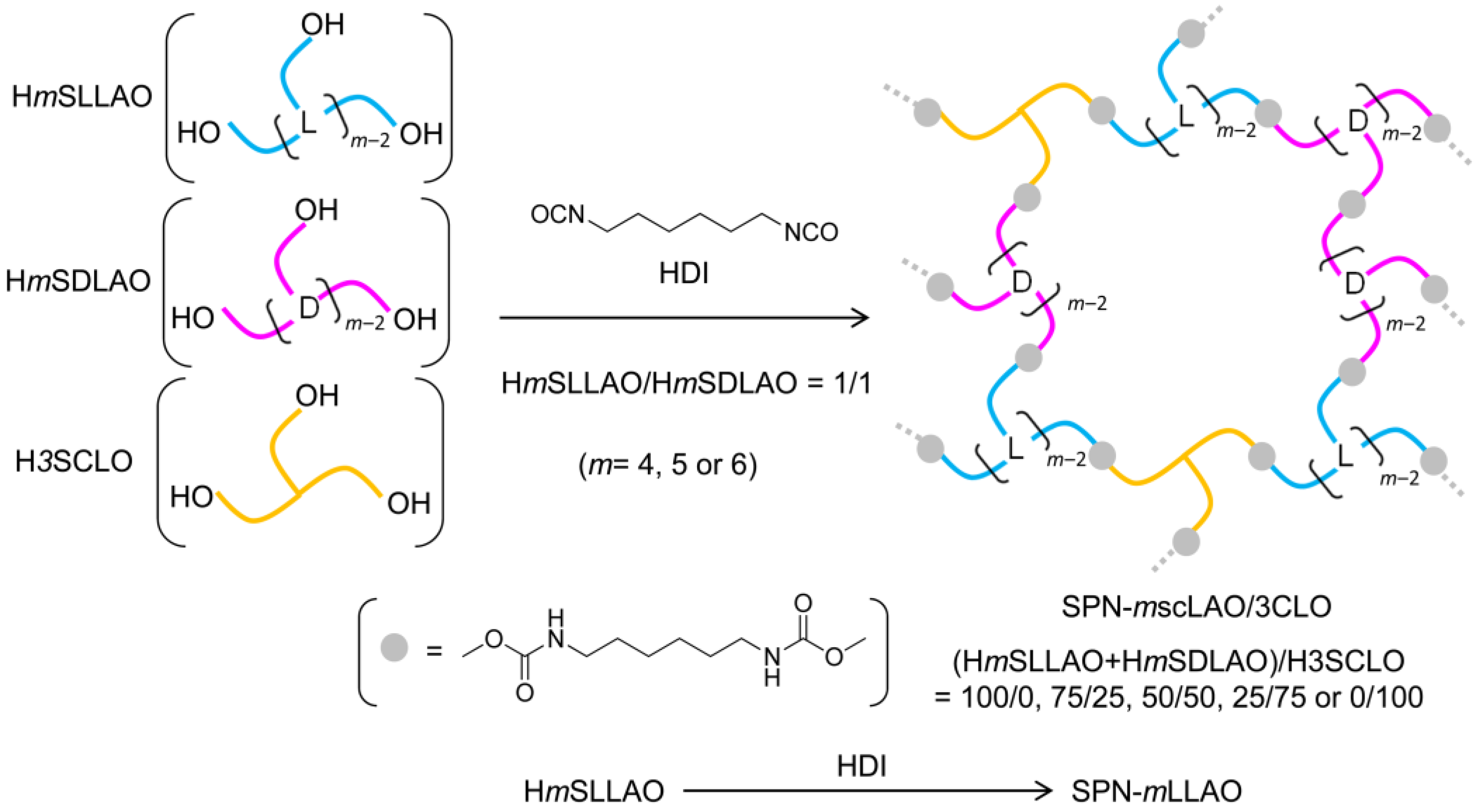
2.4. Syntheses of SPN-mscLAO/3CLO and SPN-mLLAO (m = 4, 5 or 6)
2.5. Characterization and Measurements
3. Results and Discussion
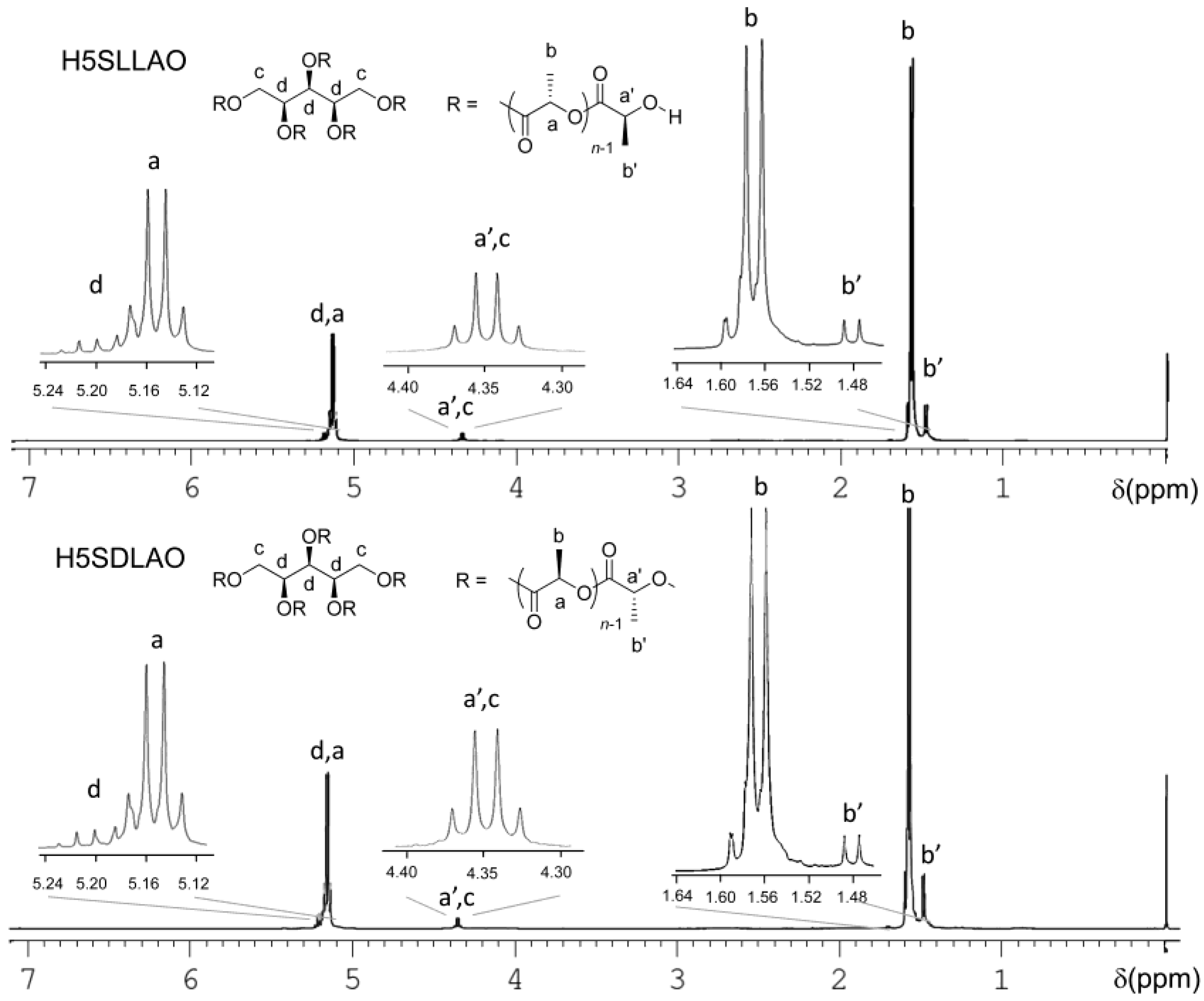
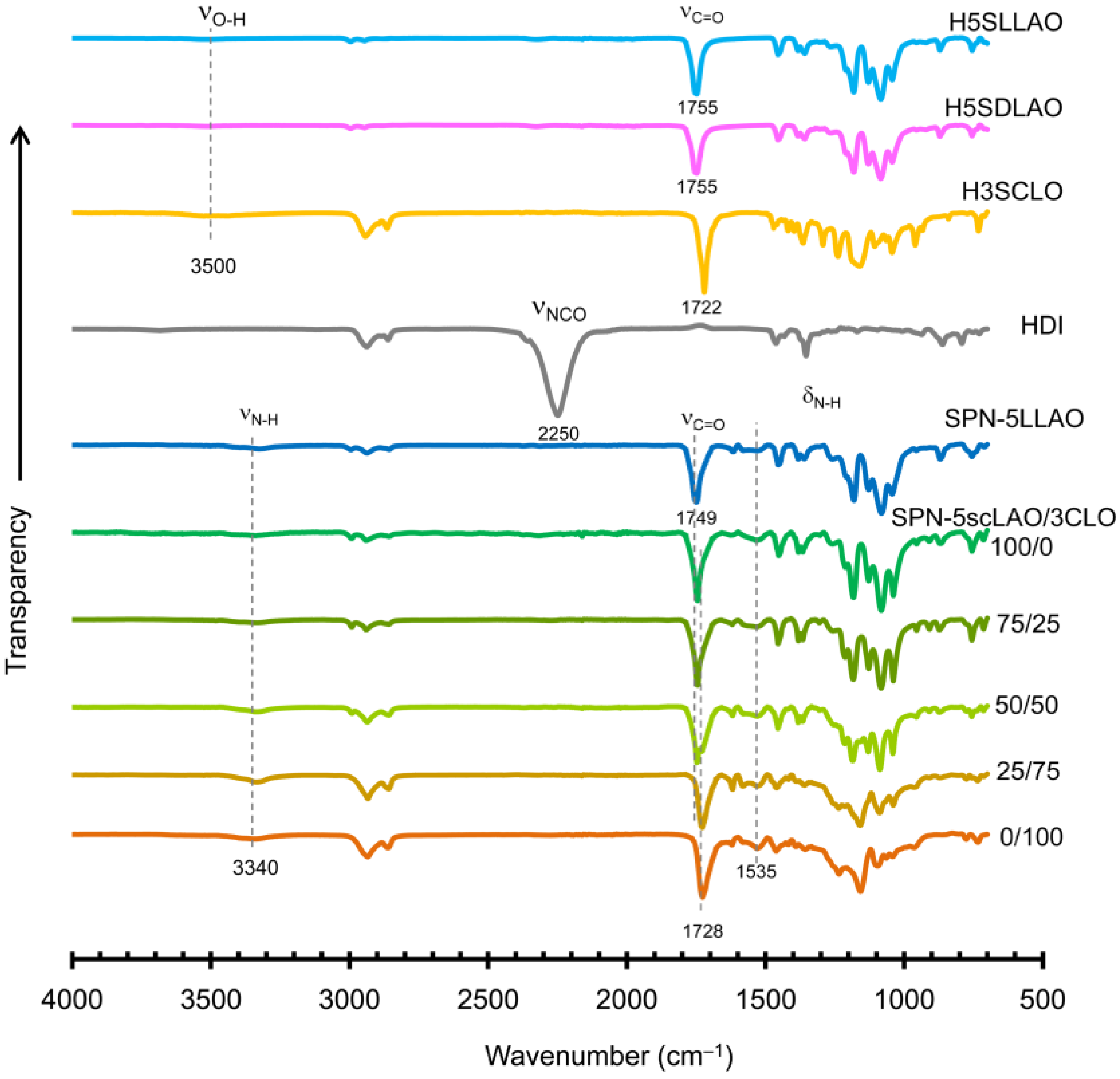
3.1. Characterization of HmSLLAOs, HmSDLAOs and H3SCLO
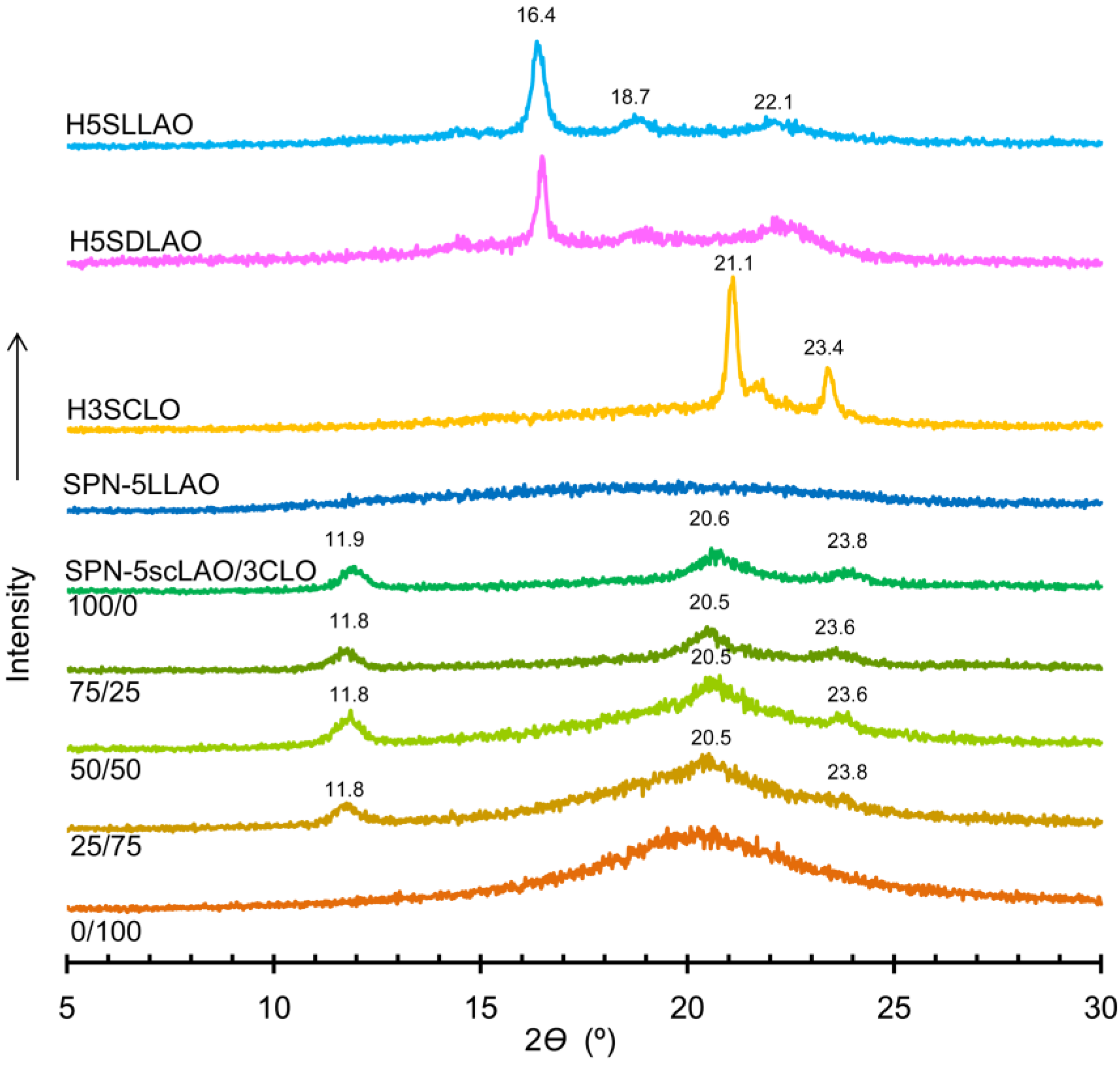
3.2. Characterization of SPN-mscLAO/3CLOs and SPN-mLLAOs
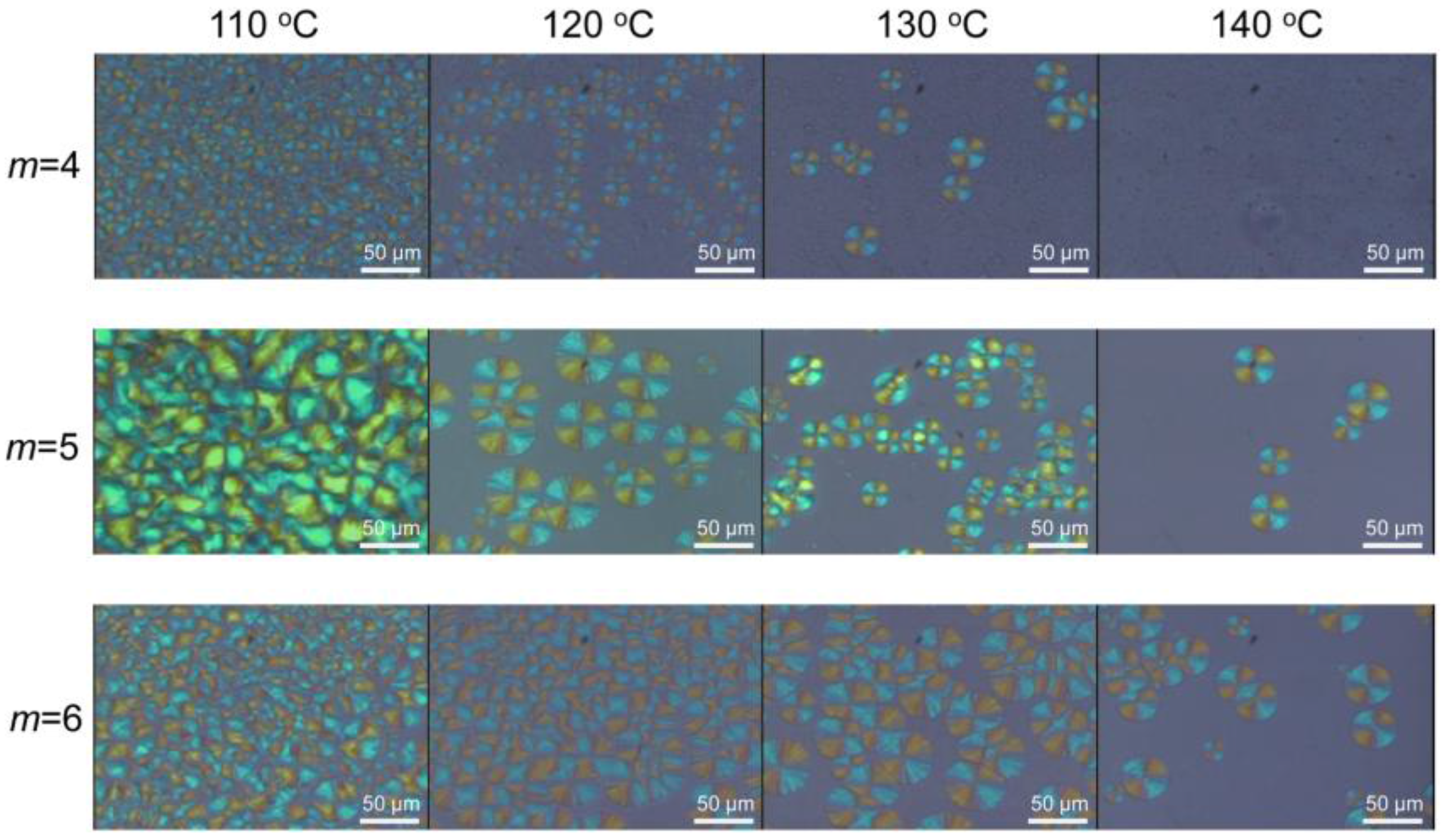
3.3. Stereocomplex Crystallization Behavior of SPN-mscLAO/3CLOs
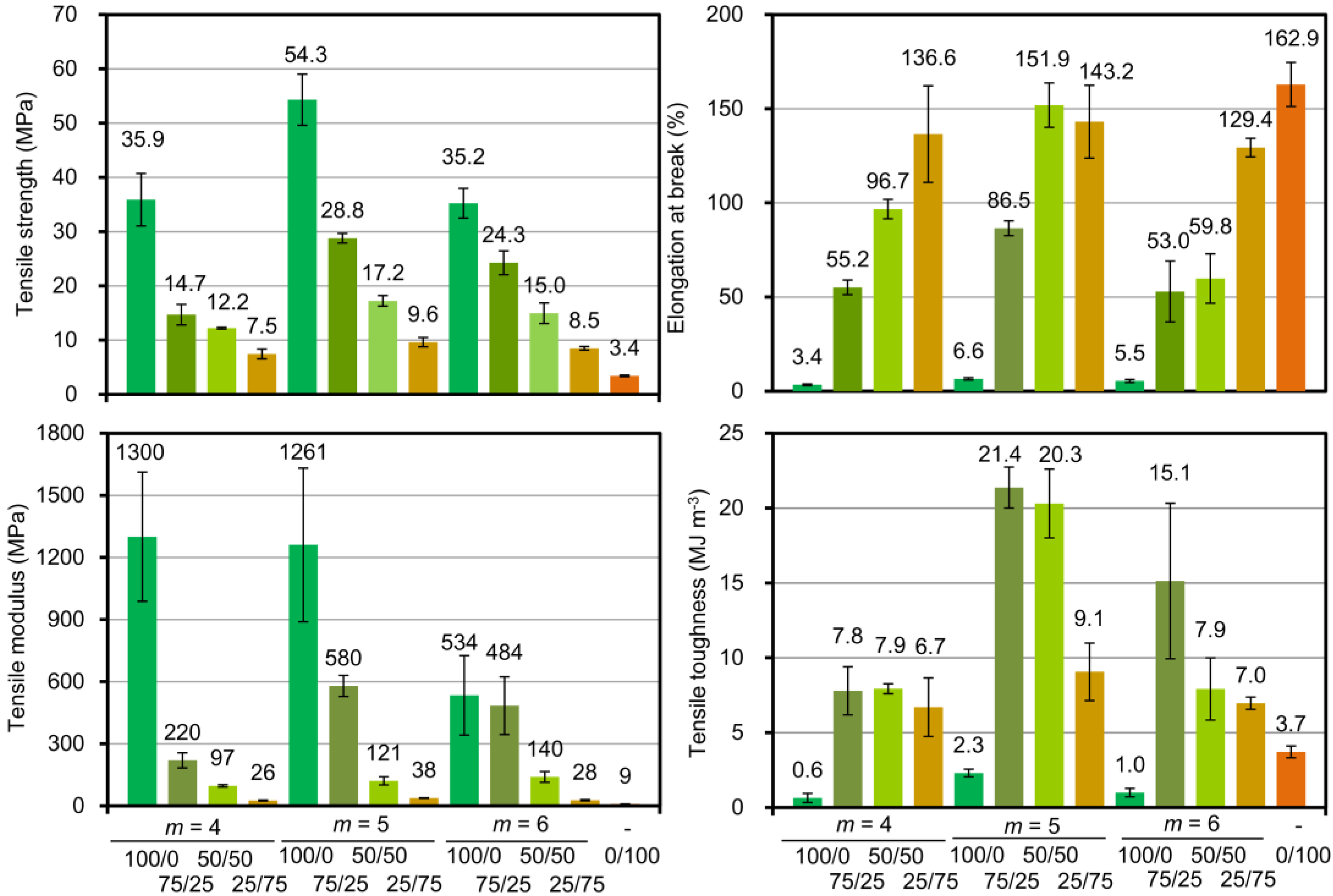
3.4. Thermal and Mechanical Properties of SPN-mscLAO/3CLOs
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cameron, D.J.A.; Shaver, M.P. Aliphatic polyester polymer stars: Synthesis, properties and applications in biomedicine and nanotechnology. Chem. Soc. Rev. 2011, 40, 1761–1776. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, W.; Li, J. Star polymers: Advances in biomedical applications. Prog. Polym. Sci. 2015, 46, 55–85. [Google Scholar] [CrossRef]
- Jahandideh, A.; Muthukumarappan, K. Star-shaped lactic acid based systems and their thermosetting resins; Synthesis, characterization, potential opportunities and drawbacks. Eur. Polym. J. 2017, 87, 360–379. [Google Scholar] [CrossRef]
- Lee, J.S.; Choo, D.-J.; Kim, S.H.; Kim, Y.H. Synthesis and degradation property of star-shaped polylactide. Polymer (Korea) 1998, 6, 880–889. [Google Scholar]
- Lee, S.-H.; Kim, S.H.; Han, Y.-K.; Kim, Y.H. Synthesis and degradation of end-group-functionalized polylactide. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 973–985. [Google Scholar] [CrossRef]
- Kim, E.S.; Kim, B.C.; Kim, S.H. Structural effect of linear and star-shaped poly(l-lactic acid) on physical properties. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 939–946. [Google Scholar] [CrossRef]
- Wang, L.; Dong, C.-M. Synthesis, crystallization kinetics, and spherulitic growth of linear and star-shaped poly(l-lactide)s with different numbers of arms. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 2226–2236. [Google Scholar] [CrossRef]
- Lang, M.; Wong, R.P.; Chu, C.C. Synthesis and structural analysis of functionalized poly(ɛ-caprolactone)-based three-arm star polymers. J. Polym. Sci. Part A Polym. Chem. 2002, 40, 1127–1141. [Google Scholar] [CrossRef]
- Shi, M.; Zhang, H.; Chen, J.; Wan, X.; Zhou, Q. Synthesis and characterization of a novel star shapes rod-coil block copolymer. Polym. Bull. 2004, 52, 401–408. [Google Scholar] [CrossRef]
- Wang, J.-L.; Wang, L.; Dong, C.-M. Synthesis, crystallization, and morphology of star-shaped poly ɛ-caprolactone). J. Polym. Sci. Part A Polym. Chem. 2005, 43, 5449–5457. [Google Scholar] [CrossRef]
- Choi, J.; Kim, I.-K.; Kwak, S.-Y. Synthesis and characterization of a series of star-branched poly(ɛ-caprolactone)s with the variation in arm numbers and lengths. Polymer 2005, 46, 9725–9735. [Google Scholar] [CrossRef]
- Wang, J.-L.; Dong, C.-M. Physical properties, crystallization kinetics, and spherulitic growth of well-defined poly(ɛ-caprolactone)s with different arms. Polymer 2006, 47, 3218–3228. [Google Scholar] [CrossRef]
- Cai, L.; Wang, S. Poly(ɛ-caprolactone) acrylates synthesized using a facile method for fabricating networks to achieve controllable physicochemical properties and tunable cell responses. Polymer 2010, 51, 164–177. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Lee, S.-R. Polylactones. 40. Nanopretzels by macrocyclic polymerization of lactones via a spirocyclic tin initiator derived from pentaerythritol. Macromolecules 1996, 29, 8689–8695. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Fechner, B. Polylactones. LVIII. Star-shaped polylactones with functional end groups via ring-expansion polymerization with a spiroinitiator. J. Polym. Sci. Part A Polym. Chem. 2002, 43, 1047–1057. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Rost, S. Polylactones, 67. Macromol. Chem. Phys. 2004, 205, 1031–1038. [Google Scholar] [CrossRef]
- Helou, M.; Miserque, O.; Brusson, J.-M.; Carpentier, J.-F.; Guillaume, S.M. Highly effective and green catalytic approach toward α,ω-dihydroxy-telechelic poly(trimethylenecarbonate). Macromol. Rapid Commun. 2009, 30, 2128–2135. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Li, F.; Li, Q.; Li, Y.; Jia, L.; Yang, J.; Fang, Q.; Cao, A. Preparation and crystallization kinetics of new structurally well-defined star-shaped biodegradable poly(l-lactide) initiated with diverse natural sugar alcohols. Biomacromolecules 2005, 6, 2236–2247. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.Y.; Jiang, N.; Gan, Z.H. Effects of multi-arm structure on crystallization and biodegradation of star-shaped poly(ɛ-caprolactone). Macromol. Biosci. 2008, 8, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Xy, Z.; Niu, Y.; Yang, L.; Xie, W.; Li, H.; Gan, Z.; Wang, Z. Morphology, rheology and crystallization behavior of polylactide composites prepared through addition of five-armed star polylactide grafted multiwalled carbon nanotubes. Polymer 2010, 51, 730–737. [Google Scholar]
- Teng, L.; Xu, X.; Nie, W.; Zhou, Y.; Song, L.; Chen, P. Synthesis and degradability of a star-shaped polylactide based on l-lactide and xylitol. J. Polym. Res. 2015, 22, 83. [Google Scholar] [CrossRef]
- Teng, L.; Nie, W.; Zhou, Y.; Song, L.; Chen, P. Synthesis and characterization of star-shaped PLLA with sorbitol as core and its microspheres application in controlled drug release. J. Appl. Polym. Sci. 2015. [Google Scholar] [CrossRef]
- Goddard, A.R.; Pérez-Nieto, S.; Passos, T.M.; Quilty, B.; Caemichael, K.; Irvine, D.J.; Howdle, S.M. Controlled polymerisation and purification of branched poly(lactic acid) surfactants in supercritical carbon dioxide. Green Chem. 2016, 18, 4772–4786. [Google Scholar] [CrossRef]
- Isikgor, F.H.; Remzi Becer, C. Lignocellulosic biomass: A sustainable platform for the production of bio-based chemicals and polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef]
- Hejna, A.; Kosmela, P.; Formela, K.; Piszczyk, L.; Haponiuk, J.T. Potential applications of crude glycerol in polymer technology–Current state and perspectives. Renew. Sustain. Energy Rev. 2016, 66, 449–475. [Google Scholar] [CrossRef]
- Yang, S.-W.; Park, J.-B.; Han, N.S.; Ryu, Y.-W.; Seo, J.-H. Production of erythritol from glucose by an osmophilic mutant of Candida magnolia. Biotechnol. Lett. 1999, 21, 887–890. [Google Scholar] [CrossRef]
- Rao, R.S.; Jyothi, C.P.; Prakasham, R.S.; Sarma, P.N.; Rao, L.V. Xylitol production from corn fiber and sugarcane bagasse hydrolysates by Candida tropicalis. Bioresour. Technol. 2006, 97, 1974–1978. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.; Nieto-Márquez, A.; Alonso, E. Bimetallic Ru:Ni/MCM-48 catalysts for the effective hydrogenation of d-glucose into sorbitol. Appl. Catal. A Gen. 2017, 59, 49–59. [Google Scholar] [CrossRef]
- Amsden, B.G. Curable, biodegradable elastomers: Emerging biomaterials for drug delivery and tissue engineering. Soft Matter 2007, 3, 1335–1348. [Google Scholar] [CrossRef]
- Storey, R.F.; Wiggins, J.S.; Puckett, A.D. Hydrolyzable poly(ester-urethane) networks from l-lysine diisocyante and d,l-lactide/ε-caprolactone homo- and copolyester triols. J. Polym. Sci. Part A Polym. Chem. 1994, 32, 2345–2363. [Google Scholar] [CrossRef]
- Amsden, B.G.; Misra, G.; Gu, F.; Younes, M. Synthesis and characterization of a photo-cross-linked biodegradable elastomer. Biomacromolecules 2004, 5, 2479–2486. [Google Scholar] [CrossRef] [PubMed]
- Karikari, A.; Edwards, W.F.; Mecham, J.B.; Long, T.E. Influence of peripheral hydrogen bonding on the mechanical properties of photo-cross-linked star-shaped poly(d,l-lactide) networks. Biomacromolecules 2005, 6, 2866–2874. [Google Scholar] [CrossRef] [PubMed]
- Nagata, M.; Sato, Y. Synthesis and properties of photocurable biodegradable multiblock copolymers based on pol(ε-caprolactone) and pol(l-lactide) segments. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 2426–2439. [Google Scholar] [CrossRef]
- Chang, S.K.; Zeng, C.; Li, J.; Ren, J. Synthesis of polylactide-based thermoset resin and its curing kinetics. Polym. Int. 2012, 61, 1492–1502. [Google Scholar] [CrossRef]
- Bakare, F.O.; Skrifvars, M.; Åkesson, D.; Wang, Y.; Afshar, S.J.; Esmaeili, N. Synthesis and characterization of bio-based thermosetting resins from lactic acid and glycerol. J. Appl. Polym. Sci. 2014. [Google Scholar] [CrossRef]
- Jahandideh, A.; Esmaeili, N.; Muthukumarappan, K. Effect of lactic acid chain length on thermomechanical properties of star-LA-Xylitol resins and jute reinforced biocompoistes. Polym. Int. 2017, 66, 1021–1030. [Google Scholar] [CrossRef]
- Shibata, M.; Katoh, M.; Takase, H.; Shibita, A. Stereocomplex formation in stereoblock copolymer networks composed of 4-armed star-shaped lactide oligomers and a 2-armed ɛ-caprolactone oligomer. Polym. Chem. 2015, 6, 4123–4132. [Google Scholar] [CrossRef]
- Shibita, A.; Kawasaki, S.; Shimasaki, T.; Teramoto, N.; Shibata, M. Stereocomplexation in copolymer networks incorporating enantiomeric glycerol-based 3-armed lactide oligomers and a 2-armed ɛ-caprolactone oligomer. Materials 2016, 9, 519. [Google Scholar] [CrossRef] [PubMed]
- Isono, T.; Kondo, Y.; Otsuka, I.; Nishiyama, Y.; Borsali, R.; Kakuchi, T.; Satoh, T. Synthesis and stereocomplex formation of star-shaped stereoblock polylactides consisting of poly(l-lactide) and poly(d-lactide) arm. Macromolecules 2013, 46, 8509–8518. [Google Scholar] [CrossRef]
- Patrício, T.; Bártolo, B. Thermal stability of PCL/PLA blends produced by physical blending process. Procedia Eng. 2013, 59, 292–297. [Google Scholar] [CrossRef]
- Woo, E.M.; Chang, L. Crystallization and morphology of stereocomplexes in nonequimolar mixtures of poly(l-lactic acid) with excess poly(d-lactic acid). Polymer 2011, 52, 6080–6089. [Google Scholar] [CrossRef]
- Pant, H.R.; Neupane, M.P.; Pant, B.; Panthia, G.; Oh, H.J.; Lee, M.H.; Kim, H.Y. Fabrication of highly porous poly (ε-caprolactone) fibers for novel tissue scaffold via water-bath electrospinning. Colloids Surf. B 2011, 88, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Shibita, A.; Takase, H.; Shibata, M. Semi-interpenetrating polymer networks composed of poly(l-lactide) and diisocyanate-bridged 4-arm star-shaped ɛ-caprolactone oligomers. Polymer 2014, 55, 5407–5416. [Google Scholar] [CrossRef]
- Shibita, A.; Shimasaki, T.; Teramoto, N.; Shibata, M. Tough conetworks composed of 4-armed star-shaped oligomers of l-lactide, d-lactide and ɛ-caprolactone. Polym. Bull. 2017. [Google Scholar] [CrossRef]
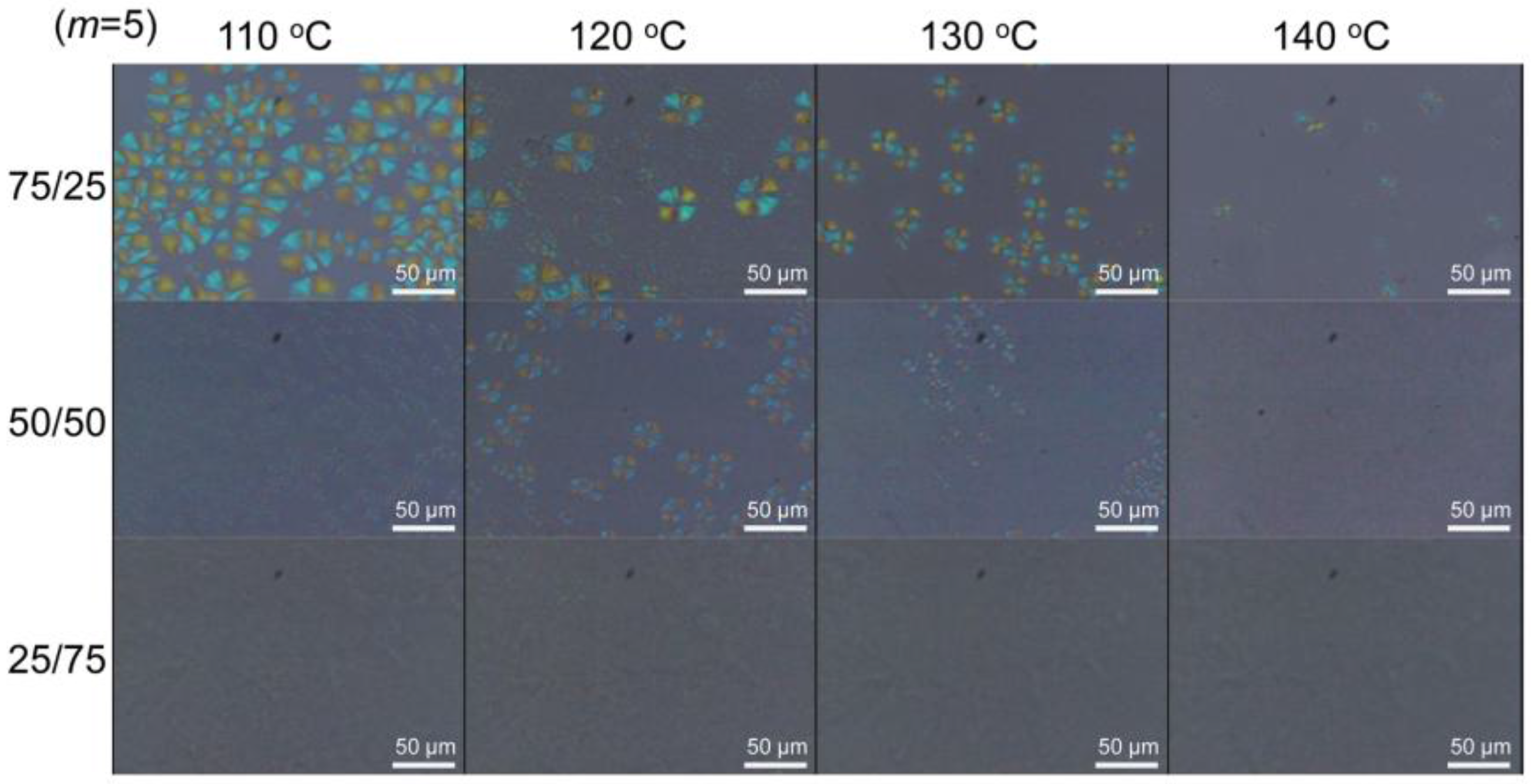
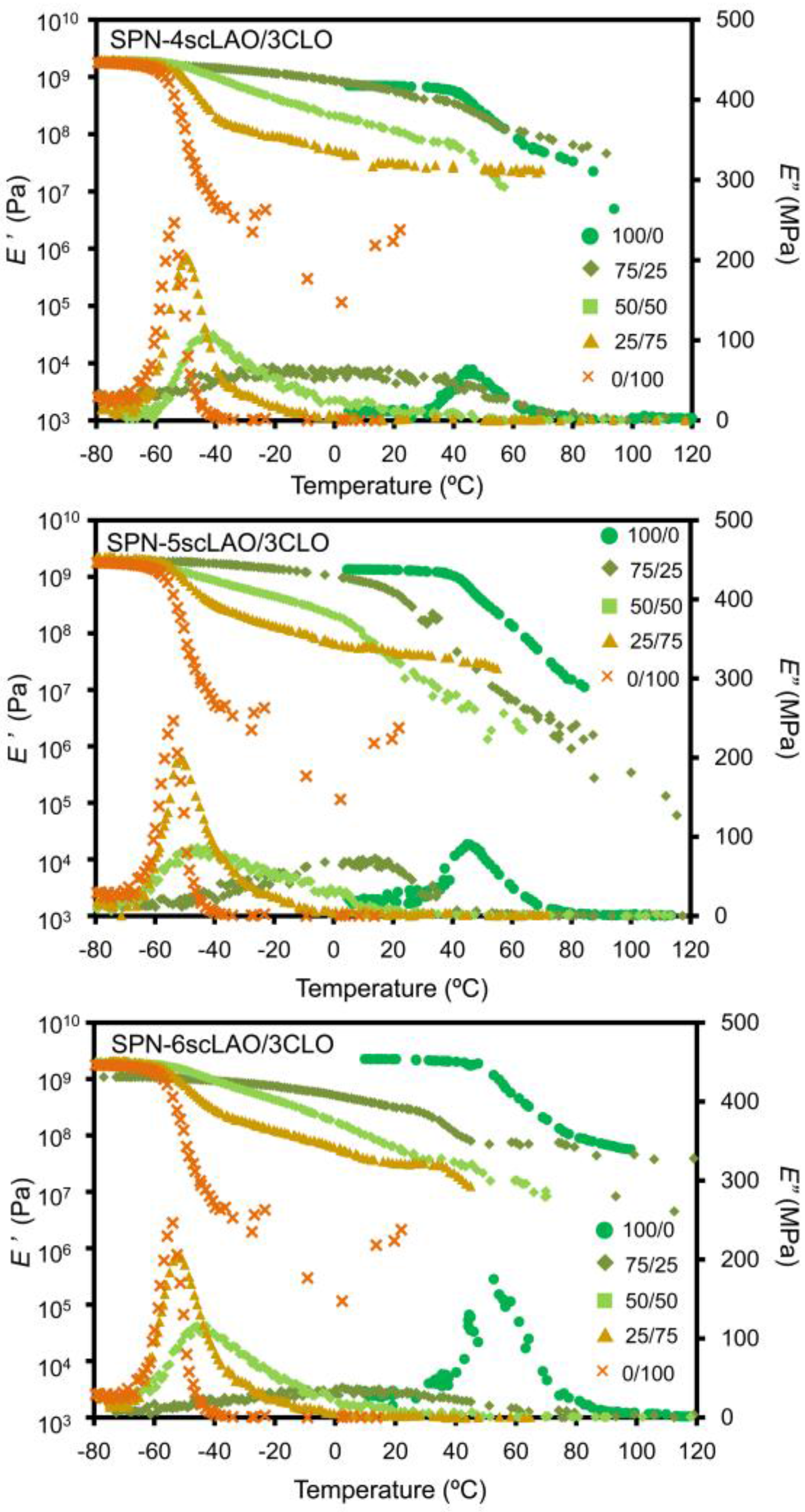
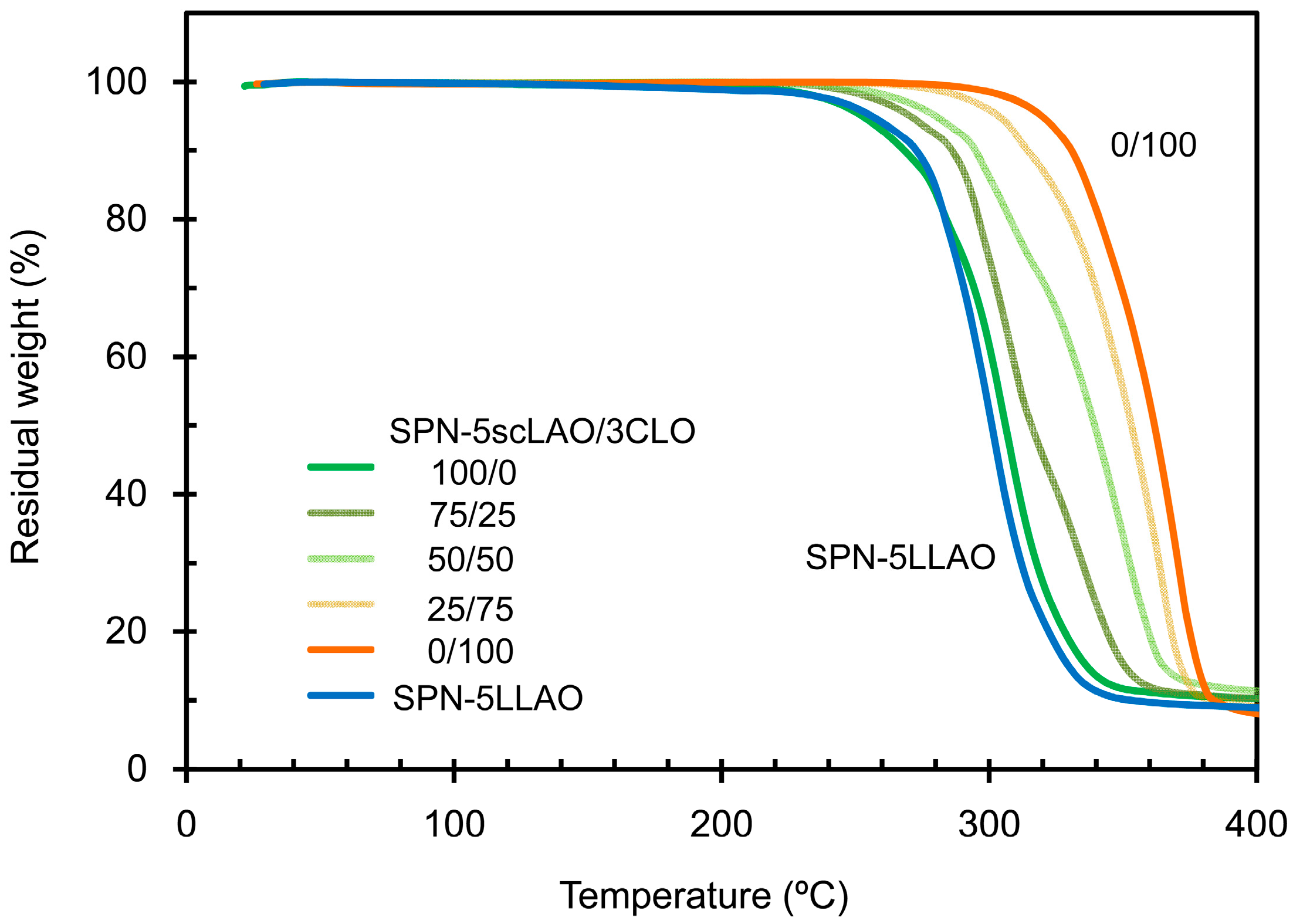
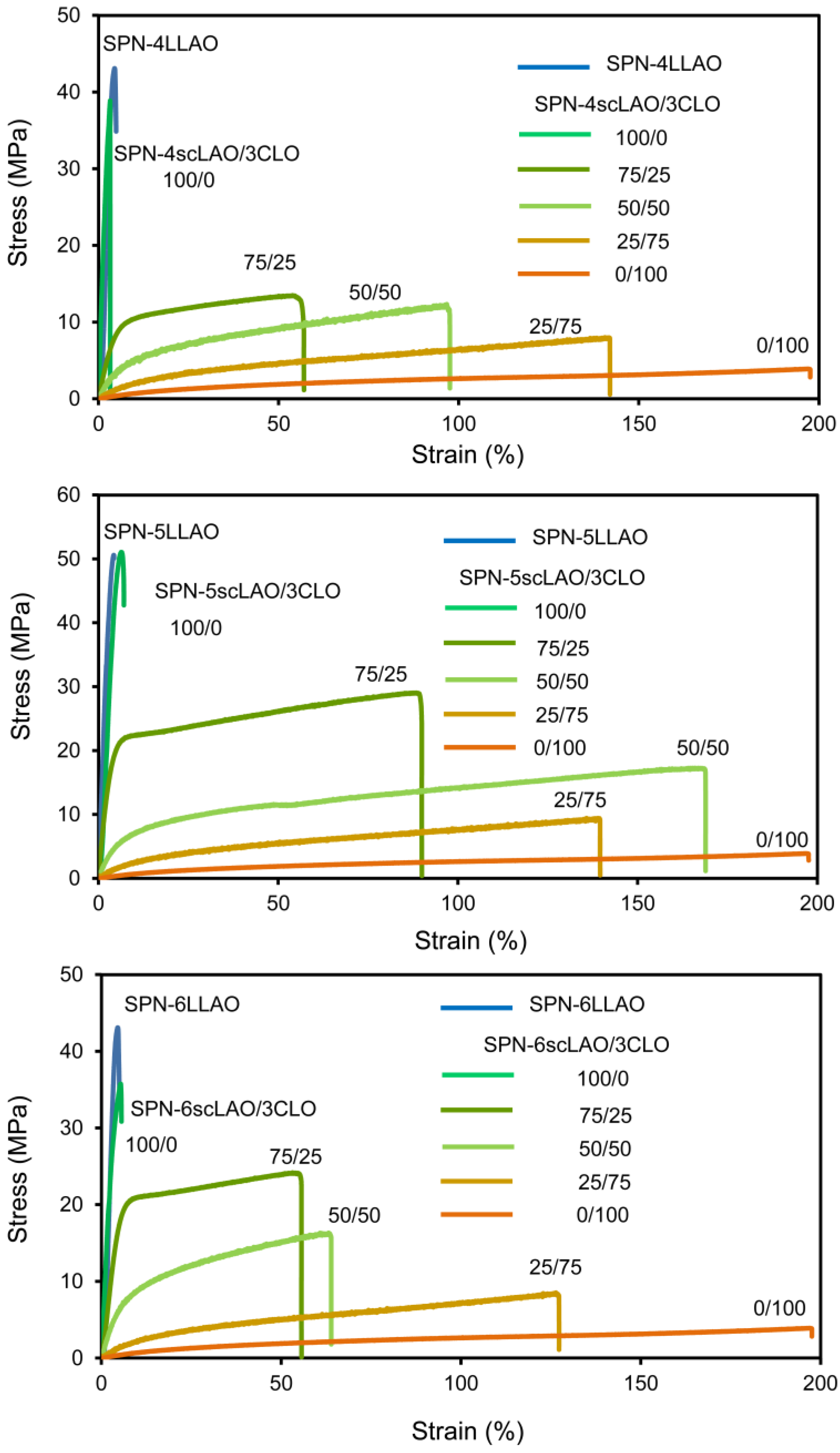
| Sample | Initiator | Feed molar ratio of LLA(DLA)/OH or CL/OH | Yield (%) | Theo. n | Obs. n *1 | Mn *2 | Mw *2 |
|---|---|---|---|---|---|---|---|
| H3SCLO | GC | 19.1 | 98 | 6.4 | 7.1 | 1560 | 2870 |
| H4SLLAO | ET | 28.0 | 94 | 14.0 | 15.4 | 2970 | 5100 |
| H4SDLAO | ET | 28.0 | 94 | 14.0 | 14.8 | 2900 | 4910 |
| H5SLLAO | XL | 35.8 | 99 | 14.3 | 15.7 | 3750 | 6430 |
| H5SDLAO | XL | 33.3 | 98 | 13.3 | 14.9 | 3240 | 6030 |
| H6SLLAO | SB | 36.3 | 97 | 12.1 | 14.6 | 4590 | 7210 |
| H6SDLAO | SB | 40.2 | 93 | 13.4 | 16.6 | 4720 | 7890 |
| Sample | First heating data | Second heating data | ||||||
|---|---|---|---|---|---|---|---|---|
| Tg | Tm,x *2 | ∆Hm,x *2 | χc,x *2 | Tg | Tm,x *2 | ∆Hm,x *2 | χc,x *2 | |
| (°C) | (°C) | (J·g−1) | (%) | (°C) | (°C) | (J·g−1) | (%) | |
| H3SCLO | - | 55.9 | 60.6 | 43.6 | - | 38.4, 47.8 | 21.8, 41.1 | 45.3 *3 |
| SPN-3CLO | −44.8 | 29.3 | 27.0 | 21.7 | −43.8 | 33.0 | 32.8 | 26.3 |
| H4SLLAO | 36.6 | 132.4 | 16.8 | 18.1 | 36.1 | - | 0 | 0 |
| H4SDLAO | 41.2 | 132.1 | 17.3 | 18.6 | 46.7 | - | 0 | 0 |
| H5SLLAO | 41.1 | 139.0 | 23.4 | 25.2 | 50.7 | - | 0 | 0 |
| H5SDLAO | 49.2 | 136.0 | 28.3 | 30.4 | 38.0 | - | 0 | 0 |
| H6SLLAO | - | 135.4 | 29.9 | 32.1 | 34.5 | - | 0 | 0 |
| H6SDLAO | - | 130.3 | 23.2 | 25.0 | 31.4 | - | 0 | 0 |
| SPN-4LLAO | 62.3 | - | 0 | 0 | 26.4 | - | 0 | 0 |
| SPN-5LLAO | 58.2 | - | 0 | 0 | 28.9 | - | 0 | 0 |
| SPN-6LLAO | 67.0 | - | 0 | 0 | 31.4 | - | 0 | 0 |
| SPN-4scLAO | 56.0 | 175.0 | 19.3 | 14.8 | 43.9 | - | 0 | 0 |
| SPN-5scLAO | 61.5 | 187.5 | 31.0 | 23.7 | 35.8 | 173.0 | 29.4 | 22.5 |
| SPN-6scLAO | 65.4 | 187.0 | 31.5 | 24.3 | 31.4 | 170.9 | 30.6 | 23.6 |
| Sample | Tg | Tm,CLO | ∆Hm,CLO | χc,CLO | Tc,LAO | ∆Hc,LAO | Tm,LAO | ∆Hm,LAO | χc,LAO | |
|---|---|---|---|---|---|---|---|---|---|---|
| (°C) | (°C) | (J·g−1) | (%) | (°C) | (J·g−1) | (°C) | (J·g−1) | (%) | ||
| First heating data | ||||||||||
| SPN-4scLAO/3CLO | 75/25 | - | - | - | 0 | - | 0 | 177.7 | 19.6 | 20.2 |
| 50/50 | - | - | - | 0 | - | 0 | 175.1 | 10.1 | 15.7 | |
| 25/75 | - | - | - | 0 | - | 0 | 176.1 | 6.23 | 19.6 | |
| SPN-5scLAO/3CLO | 75/25 | - | - | 0 | 0 | - | 0 | 187.8 | 25.7 | 26.4 |
| 50/50 | - | - | 0 | 0 | - | 0 | 187.8 | 15.8 | 24.5 | |
| 25/75 | −41.2 | 17.3 | 5.4 | 5.7 | - | 0 | 187.8 | 5.64 | 17.6 | |
| SPN-6scLAO/3CLO | 75/25 | 42.1 | - | 0 | 0 | - | 0 | 184.4 | 24.4 | 25.2 |
| 50/50 | −30.3 | - | 0 | 0 | - | 0 | 184.7 | 17.1 | 26.6 | |
| 25/75 | −44.1 | 11.1 | 2.4 | 2.5 | - | 0 | 185.5 | 4.54 | 14.2 | |
| Second heating data | ||||||||||
| SPN-4scLAO/3CLO | 75/25 | - | - | 0 | 0 | 73.7 | −16.1 | 168.4 | 11.5 | 11.8 |
| 50/50 | - | - | 0 | 0 | 59.3 | −8.18 | 170.1 | 11.6 | 18.1 | |
| 25/75 | - | - | 0 | 0 | 68.7 | −2.78 | 173.4 | 2.43 | 7.6 | |
| SPN-5scLAO/3CLO | 75/25 | - | - | 0 | 0 | 71.7 | −18.4 | 173.4 | 21.4 | 22.0 |
| 50/50 | - | - | 0 | 0 | 74.7 | −8.00 | 181.8 | 10.8 | 16.8 | |
| 25/75 | −39.0 | 32.0 | 5.5 | 5.8 | 92.9 | −1.82 | 185.1 | 2.26 | 7.1 | |
| SPN-6scLAO/3CLO | 75/25 | 27.7 | - | 0 | 0 | 66.9 | −15.0 | 169.9 | 19.5 | 20.1 |
| 50/50 | −31.1 | - | 0 | 0 | 49.0 | −3.88 | 171.6 | 3.1 | 4.9 | |
| 25/75 | −40.9 | 28.6 | 6.7 | 7.1 | 82.8 | −1.48 | 180.4 | 1.4 | 4.3 | |
| Sample | E′′ Peak temperature (°C) | Td5 (°C) | Char yield at 400 °C (%) | |
|---|---|---|---|---|
| SPN-4LLAO | - | 264 | 8.7 | |
| SPN-4scLAO/3CLO | 100/0 | 44.5 | 255 | 4.5 |
| 75/25 | 18.1 | 265 | 6.6 | |
| 50/50 | −43.6 | 276 | 7.2 | |
| 25/75 | −52.5 | 327 | 7.8 | |
| SPN-5LLAO | - | 261 | 2.6 | |
| SPN-5scLAO/3CLO | 100/0 | 45.9 | 254 | 10.2 |
| 75/25 | 14.7 | 272 | 10.1 | |
| 50/50 | −45.8 | 281 | 11.2 | |
| 25/75 | −52.5 | 304 | 8.9 | |
| SPN-6LLAO | - | 265 | 10.3 | |
| SPN-6scLAO/3CLO | 100/0 | 52.6 | 254 | 9.2 |
| 75/25 | 19.1 | 266 | 9.5 | |
| 50/50 | −50.3 | 278 | 9.0 | |
| 25/75 | −52.3 | 291 | 9.6 | |
| (SPN-3CLO) | 0/100 | −53.9 | 320 | 7.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugane, K.; Takahashi, H.; Shimasaki, T.; Teramoto, N.; Shibata, M. Stereocomplexation, Thermal and Mechanical Properties of Conetworks Composed of Star-Shaped l-Lactide, d-Lactide and ε-Caprolactone Oligomers Utilizing Sugar Alcohols as Core Molecules. Polymers 2017, 9, 582. https://doi.org/10.3390/polym9110582
Sugane K, Takahashi H, Shimasaki T, Teramoto N, Shibata M. Stereocomplexation, Thermal and Mechanical Properties of Conetworks Composed of Star-Shaped l-Lactide, d-Lactide and ε-Caprolactone Oligomers Utilizing Sugar Alcohols as Core Molecules. Polymers. 2017; 9(11):582. https://doi.org/10.3390/polym9110582
Chicago/Turabian StyleSugane, Kaito, Hayato Takahashi, Toshiaki Shimasaki, Naozumi Teramoto, and Mitsuhiro Shibata. 2017. "Stereocomplexation, Thermal and Mechanical Properties of Conetworks Composed of Star-Shaped l-Lactide, d-Lactide and ε-Caprolactone Oligomers Utilizing Sugar Alcohols as Core Molecules" Polymers 9, no. 11: 582. https://doi.org/10.3390/polym9110582
APA StyleSugane, K., Takahashi, H., Shimasaki, T., Teramoto, N., & Shibata, M. (2017). Stereocomplexation, Thermal and Mechanical Properties of Conetworks Composed of Star-Shaped l-Lactide, d-Lactide and ε-Caprolactone Oligomers Utilizing Sugar Alcohols as Core Molecules. Polymers, 9(11), 582. https://doi.org/10.3390/polym9110582






