Controlled Release of Lysozyme from Double-Walled Poly(Lactide-Co-Glycolide) (PLGA) Microspheres
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Microspheres
2.2.2. Characterization of Lysozyme Loaded Microspheres
Particle Size Analysis
Microspheres Surface Morphology
Determining Microspheres Polymer Orientation
Lysozyme Encapsulation Efficiency
Microspheres Yield
In Vitro Release Studies
Lysozyme Bioactivity Measurements
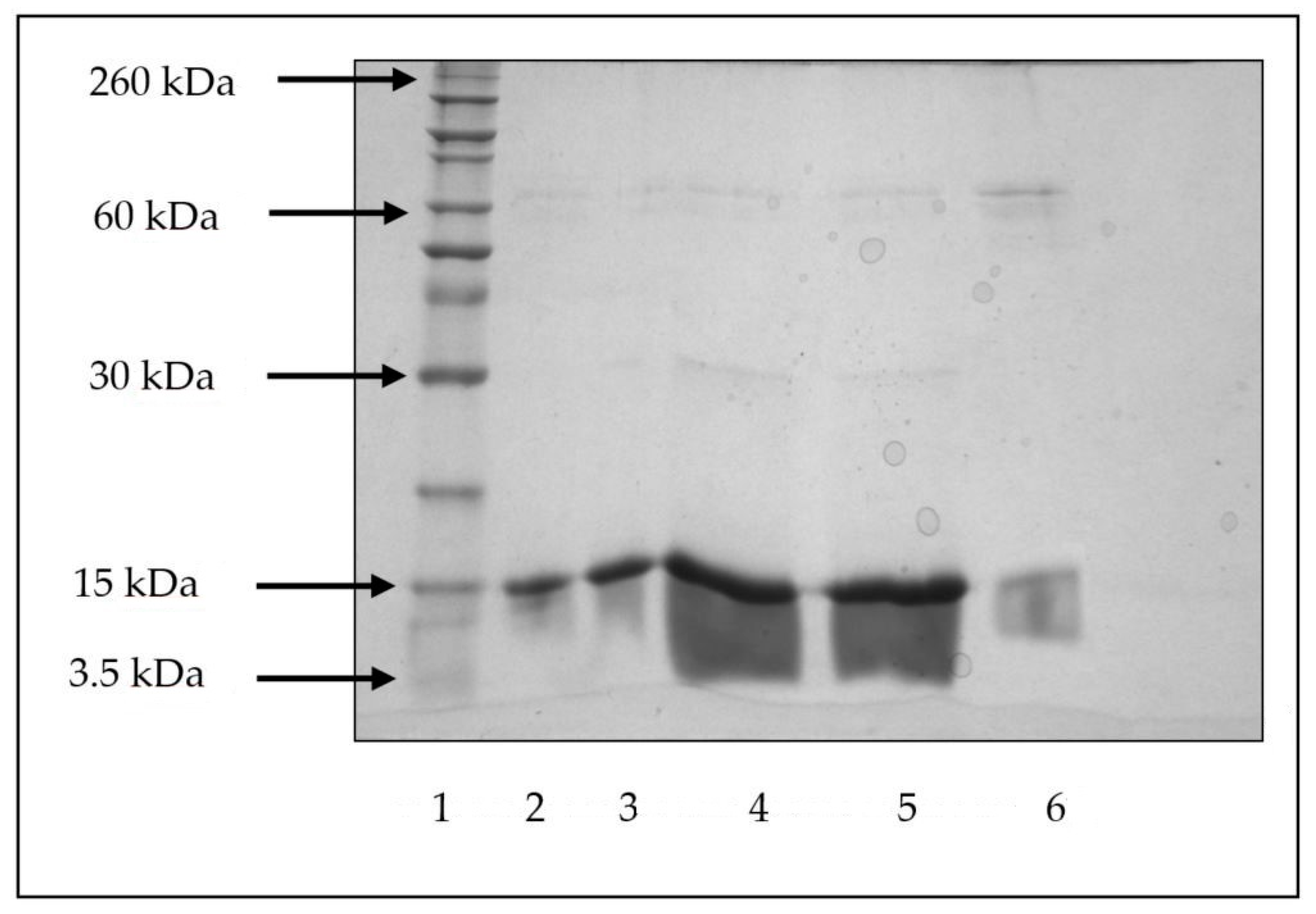
2.2.3. Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.3. Statistical Analysis
3. Results and Discussion
3.1. Particle Size and Size Distribution of Microspheres
3.2. Surface Morphology of Lysozyme Loaded Microspheres
3.3. Microspheres Polymer Orientation Studies
3.4. Lysozyme Encapsulation Efficiency
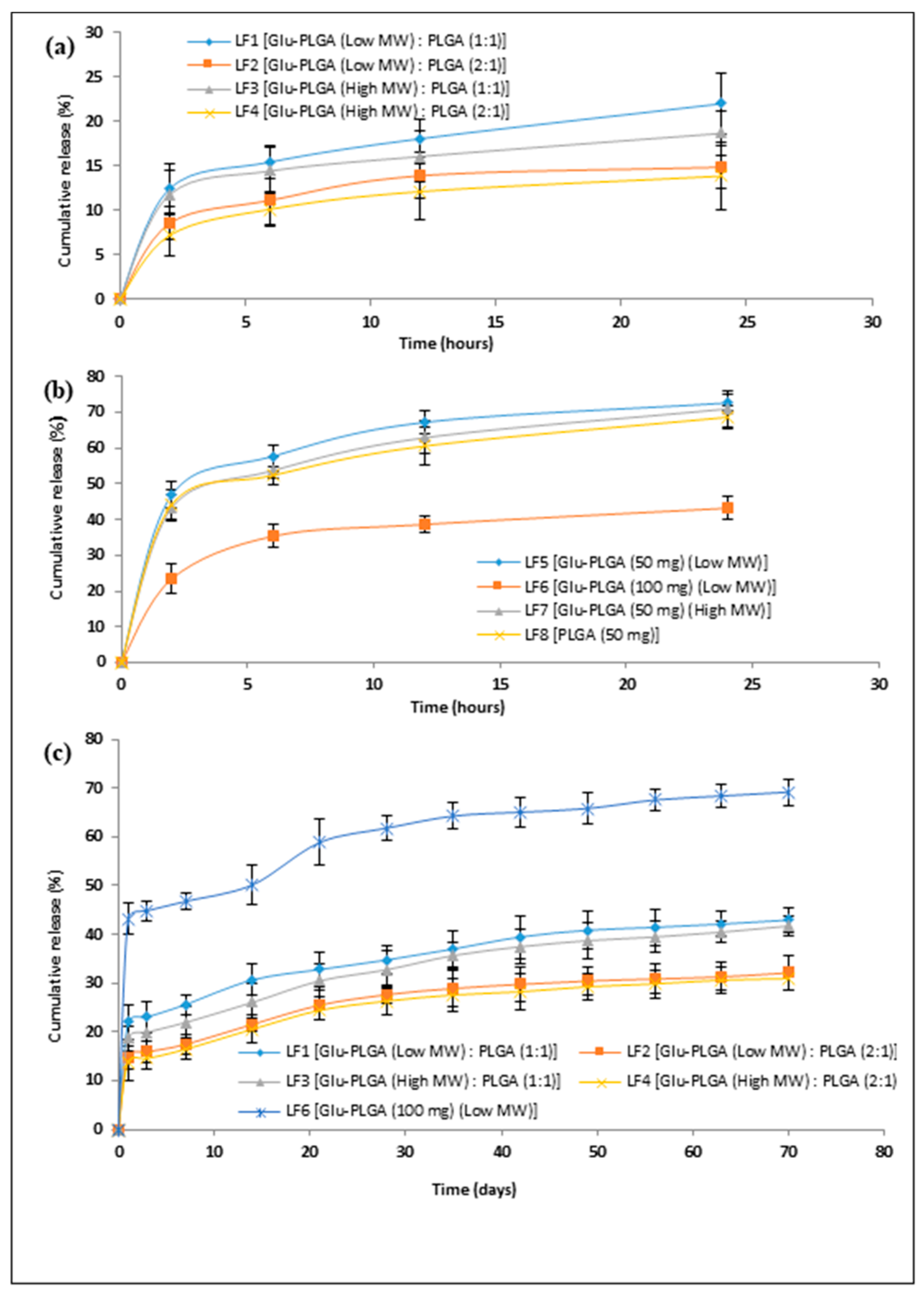
3.5. In Vitro Lysozyme Release
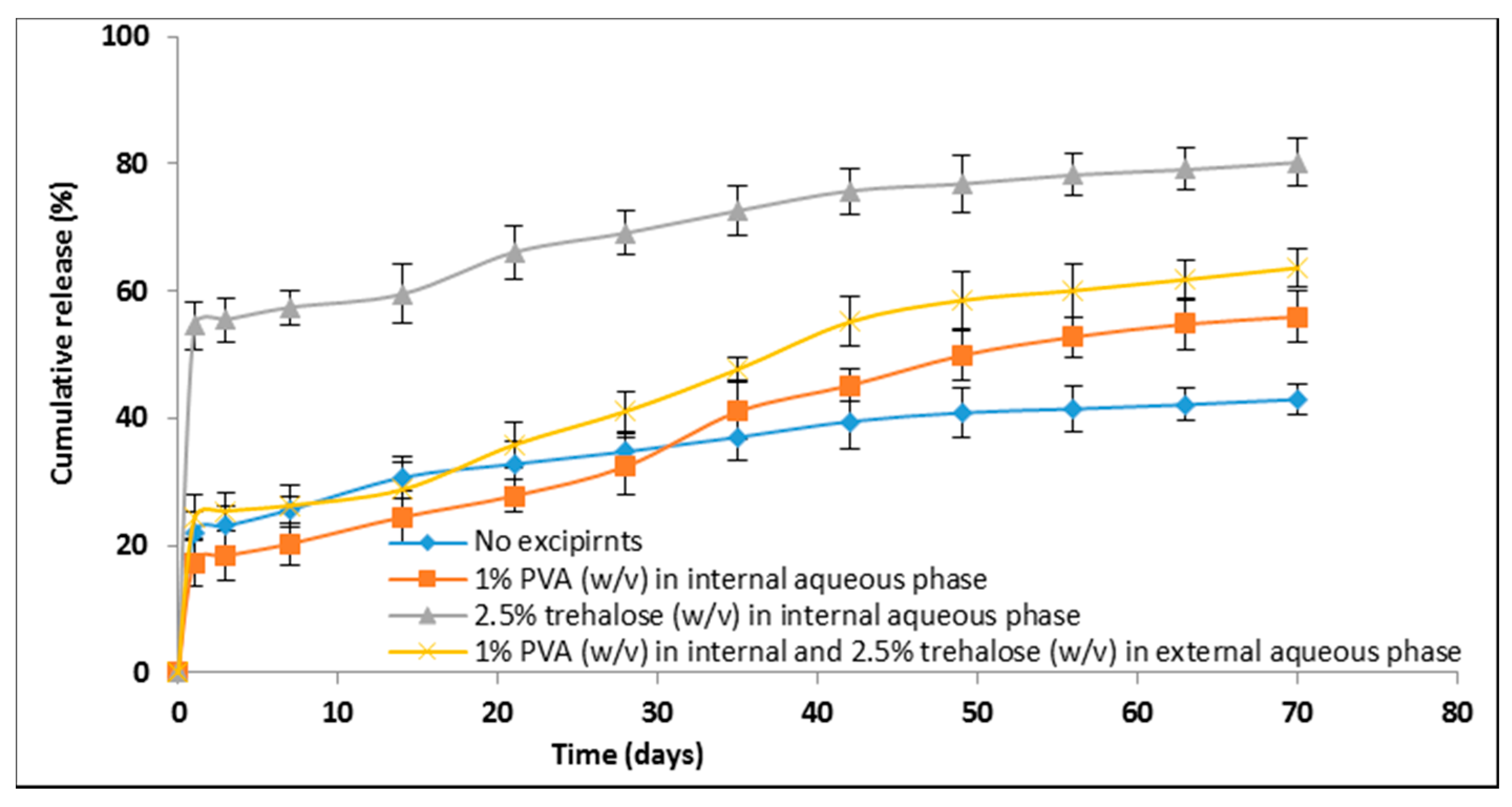
3.6. Stabilization of Lysozyme during Emulsification
3.6.1. Excipients Effect on Microspheres Size
3.6.2. Excipients Effect on Surface Morphology
3.6.3. Excipients Effect on Lysozyme Encapsulation Efficiency
3.6.4. Excipients Effect on In Vitro Lysozyme Release Profiles
3.7. Lysozyme Stability during Encapsulation and In Vitro Release
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zimmer, A.; Kreuter, J. Microspheres and nanoparticles used in ocular delivery systems. Adv. Drug Deliv. Rev. 1995, 16, 61–73. [Google Scholar] [CrossRef]
- Bala, I.; Hariharan, S.; Ravi Kumar, M.N.V. PLGA nanoparticles in drug delivery: The state of the art. Crit. Rev. Ther. Drug Carr. Syst. 2004, 21, 387–422. [Google Scholar] [CrossRef]
- Anderson, J.M.; Shive, M.S. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 2012, 64, 72–82. [Google Scholar] [CrossRef]
- Samadi, N.; Abbadessa, A.; Di Stefano, A.; van Nostrum, C.F.; Vermonden, T.; Rahimian, S.; Teunissen, E.A.; van Steenbergen, M.J.; Amidi, M.; Hennink, W.E. The effect of lauryl capping group on protein release and degradation of poly(d,l-lactic-co-glycolic acid) particles. J. Control. Release 2013, 172, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Renukuntla, J.; Vadlapudi, A.D.; Patel, A.; Boddu, S.H.; Mitra, A.K. Approaches for enhancing oral bioavailability of peptides and proteins. Int. J. Pharm. 2013, 477, 75–93. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; van der Walle, C.F.; Ravi Kumar, M.N. Antacid co-encapsulated polyester nanoparticles for peroral delivery of insulin: Development, pharmacokinetics, biodistribution and pharmacodynamics. Int. J. Pharm. 2013, 440, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Devrim, B.; Bozkir, A. Preparation and in vitro evaluation of surface-modified poly(lactide-co-glycolide) microparticles as biodegradable drug carriers for pulmonary peptide and protein delivery. J. Microencapsul. 2014, 31, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid /polyglycolic acid copolymers. Biomaterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- Göpferich, A. Mechanisms of polymer degradation and erosion. Biomaterials 1996, 17, 103–114. [Google Scholar] [CrossRef]
- Huang, X.; Brazel, C.S. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J. Control. Release 2001, 73, 121–136. [Google Scholar] [CrossRef]
- Kim, H.K.; Park, T.G. Microencapsulation of human growth hormone within biodegradable polyester microspheres: Protein aggregation stability and incomplete release mechanism. Biotechnol. Bioeng. 1999, 65, 659–667. [Google Scholar] [CrossRef]
- Jiang, G.; Woo, B.H.; Kang, F.; Singh, J.; Deluca, P.P. Assessment of protein release kinetics, stability and protein polymer interaction of lysozyme encapsulated poly(d,l-lactide-co-glycolide) microspheres. J. Control. Release 2002, 79, 137–145. [Google Scholar] [CrossRef]
- Lee, T.H.; Wang, J.; Wang, C. Double-walled microspheres for the sustained release of a highly water soluble drug: Characterization and irradiation studies. J. Control. Release. 2002, 83, 437–452. [Google Scholar] [CrossRef]
- Rahman, N.A.; Mathiowitz, E. Localization of bovine serum albumin in double-walled microspheres. J. Control. Release 2004, 94, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.L.; She, Y.C.; Widjaja, E.; Chong, H.C.; Tan, N.S.; Loo, S.C.J. Fabrication and drug release study of double-layered microparticles of various sizes. J. Pharm. Sci. 2012, 101, 2787–2797. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.C.; Lin, R.; Wang, C. Fabrication of double-walled microspheres for the sustained release of doxorubicin. J. Colloid Interface Sci. 2005, 291, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Navaei, A.; Rasoolian, M.; Momeni, A.; Emami, S.; Rafienia, M. Double-walled microspheres loaded with meglumineantimoniate: Preparation, characterization and in vitro release study. Drug Dev. Ind. Pharm. 2014, 40, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Göpferich, A.; Alonso, M.J.; Langer, R. Development and characterization of microencapsulated microspheres. Pharm. Res. 1994, 11, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Watano, S.; Nakamura, H.; Hamada, K.; Wakamatsu, Y.; Tanabe, Y.; Dave, R.N.; Pfeffer, R. Fine particle coating by a novel rotating fluidized bed coater. Powder Technol. 2004, 141, 172–176. [Google Scholar] [CrossRef]
- Lee, H.K.; Park, J.H.; Kwon, K.C. Double-walled microparticles for single shot vaccine. J. Control. Release 1997, 44, 283–293. [Google Scholar] [CrossRef]
- Berkland, C.; Pollauf, E.; Pack, D.W.; Kim, K. Uniform double-walled polymer microspheres of controllable shell thickness. J. Control. Release 2004, 96, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Ribeiro, P.F.; Pack, D.W. Controlled protein release from monodisperse biodegradable double-wall microspheres of controllable shell thickness. J. Control. Release 2013, 172, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W. A water-in-oil-in-oil-in-water (W/O/O/W) method for producing drug-releasing, double-walled microspheres. Int. J. Pharm. 2009, 374, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Devrim, B.; Bozkir, A. Preparation and evaluation of double-walled microparticles prepared with a modified water-in-oil-in-oil-in-water (w1/o/o/w3) method. J. Microencapsul. 2013, 30, 741–754. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.D.; Shen, X.C.; Tao, L. Modified emulsion solvent evaporation method for fabricating core—Shell microspheres. Int. J. Pharm. 2013, 452, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Ansary, R.H.; Rahman, M.M.; Awang, M.B.; Katas, H.; Hadi, H.; Mohamed, F.; Doolaanea, A.A.; Kamaruzzaman, Y.B. Preparation, characterization and in vitro release study of BSA-loaded double-walled glucose-poly(lactide-co-glycolide) microspheres. Arch. Pharm. Res. 2016, 39, 1242–1256. [Google Scholar] [CrossRef] [PubMed]
- Ansary, R.H.; Rahman, M.M.; Awang, M.B.; Katas, H.; Hadi, H.; Doolaanea, A.A. Preparation, characterization and in vitro release studies of insulin-loaded double-walled poly(lactide-co-glycolide) microspheres. Drug Deliv. Transl. Res. 2016, 6, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Sah, H. A new strategy to determine the actual protein content of poly(lactide-co-glycolide) microspheres. J. Pharm. Sci. 1997, 86, 1315–1318. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi, A.H.; van Steenbergen, M.J.; Talsma, H. Preparation and characterization of protein loaded microspheres based on a hydroxylated aliphatic polyester, poly(lactic-co-hydroxymethyl glycolic acid). J. Control. Release 2009, 138, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Kokai, L.E.; Tan, H.; Jhunjhunwala, S.; Little, S.R.; Frank, J.W.; Marra, K.G. Protein bioactivity and polymer orientation is affected by stabilizer incorporation for double-walled microspheres. J. Control. Release 2010, 141, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Diwan, M.; Park, T.G. Pegylation enhances protein stability during encapsulation in PLGA microspheres. J. Control. Release 2001, 73, 233–244. [Google Scholar] [CrossRef]
- Milacic, V.; Schwendeman, S.P. Lysozyme release and polymer erosion behavior of injectable implants prepared from PLGA-PEG block copolymers and PLGA/PLGA-PEG blends. Pharm. Res. 2014, 31, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Igartua, M.; Hernandez, R.M.; Esquisabel, A.; Gascon, A.R.; Calvo, M.B.; Pedraz, J.L. Influence of formulation variables on the in vitro release of albumin from biodegradable microparticulate systems. J. Microencapsul. 1997, 14, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Crotts, G.; Park, T.G. Protein delivery from poly(lactic-co-glycolic acid) biodegradable microspheres: Release kinetics and stability issues. J. Microencapsul. 1998, 15, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Sah, H. Protein instability toward organic solvent/water emulsification: Implications for protein microencapsulation into microspheres. PDA J. Pharm. Sci. Technol. 1999, 53, 3–10. [Google Scholar] [PubMed]
- van de Weert, M.; Hoechstetter, J.; Hennink, W.E.; Crommelin, D.J. The effect of a water/organic solvent interface on the structural stability of lysozyme. J. Control Release 2000, 68, 351–359. [Google Scholar] [CrossRef]
- Srinivasan, C.; Katare, Y.K.; Muthukumaran, T.; Panda, A.K. Effect of additives on encapsutation efficiency, stability and bioactivity of entrapped lysozyme from biodegradable polymer particles. J. Microencapsul. 2005, 22, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Paillard-Giteau, A.; Tran, V.T.; Thomas, O.; Garric, X.; Coudane, J.; Marchal, S.; Chourpa, I.; Benoit, J.P.; Montero-Menei, C.N.; Venier-Julienne, M.C. Effect of various additives and polymers on lysozyme release from PLGA microspheres prepared by an s/o/w emulsion technique. Eur. J. Pharm. Biopharm. 2010, 75, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Pistel, K.F.; Kissel, T. Effects of salt addition on the microencapsulation of proteins using W/O/W double emulsion technique. J. Microencapsul. 2000, 17, 467–483. [Google Scholar] [PubMed]
- Zhang, J.X.; Zhu, K.J. An improvement of double emulsion technique for preparing bovine serum albumin-loaded PLGA microspheres. J. Microencapsul. 2004, 21, 775–785. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Timasheff, S.N. The stabilization of proteins by sucrose. J. Biol. Chem. 1981, 256, 7193–7201. [Google Scholar] [PubMed]
- Fu, K.; Pack, D.W.; Klibanov, A.M.; Langer, R. Visual evidence of acidic environment within degrading Poly(lactic-co-glycolic acid) (PLGA) microspheres. Pharm. Res. 2000, 17, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Schwendeman, S.P. Comparison of the effects of Mg(OH)2 and sucrose on the stability of bovine serum albumin encapsulated in injectable poly (d,l-lactide-co-glycolide) implants. Biomaterials 2002, 23, 239–245. [Google Scholar] [CrossRef]
- Xia, Y.; Xu, Q.; Wang, C.; Pack, D.W. Protein encapsulation in and release from monodisperse double-wall polymer microspheres. J. Pharm. Sci. 2013, 102, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Dutta, C.; Tiwari, A. Disulfide-bond scrambling promotes amorphous aggregates in lysozyme and bovine serum albumin. J. Phys. Chem. B 2015, 119, 3969–3981. [Google Scholar] [CrossRef] [PubMed]



| Formulation Code | Method | Glu-PLGA * (Low MW) (mg) | Glu-PLGA ** (High MW) (mg) | PLGA (mg) | DCM (mL) | EA (mL) |
|---|---|---|---|---|---|---|
| LF1 | w1/o/o/w2 | 50 | - | 50 | 1 | 1 |
| LF2 | w1/o/o/w2 | 100 | - | 50 | 1 | 1 |
| LF3 | w1/o/o/w2 | - | 50 | 50 | 1 | 1 |
| LF4 | w1/o/o/w2 | - | 100 | 50 | 1 | 1 |
| LF5 | w1/o/w2 | 50 | - | - | 1 | - |
| LF6 | w1/o/w2 | 100 | - | - | 1 | - |
| LF7 | w1/o/w2 | - | 50 | - | 1 | - |
| LF8 | w1/o/w2 | - | - | 50 | 1 | - |
| Formulation Code | Mean Particle Size (µm ± SD) | Span ± SD | Actual Drug Loading (% ± SD) | Encapsulation Efficiency (% ± SD) | Microspheres Yield (%) |
|---|---|---|---|---|---|
| LF1 | 1.64 ± 0.58 | 1.01 ± 0.08 | 2.13 ± 0.10 | 71.04 ± 3.08 | 58.85 ± 4.15 |
| LF2 | 7.79 ± 1.03 | 1.34 ± 0.12 | 2.49 ± 0.12 | 82.84 ± 3.89 | 63.54 ± 3.28 |
| LF3 | 2.65 ± 0.45 | 1.14 ± 0.05 | 2.17 ± 0.11 | 72.15 ± 3.54 | 61.28 ± 3.72 |
| LF4 | 8.04 ± 1.21 | 1.31 ± 0.07 | 2.52 ± 0.14 | 83.92 ± 4.66 | 67.36 ± 3.86 |
| LF5 | 6.32 ± 1.07 | 1.27 ± 0.13 | 1.59 ± 0.11 | 51.67 ± 3.56 | 64.18 ± 4.32 |
| LF6 | 7.18 ± 1.38 | 1.38 ± 0.10 | 1.85 ± 0.13 | 62.03 ± 4.15 | 66.45 ± 3.55 |
| LF7 | 5.73 ± 1.21 | 1.22 ± 0.12 | 1.65 ± 0.09 | 53.88 ± 2.97 | 62.40 ± 4.14 |
| LF8 | 5.20 ± 1.10 | 1.16 ± 0.11 | 1.62 ± 0.12 | 52.23 ± 3.90 | 63.12 ± 3.37 |
| Formulation Code | Lysozyme Released from the Microspheres after 70 Days (% ± SD ) | Lysozyme Extracted from the Insoluble Residues (% ± SD) | Recovery (% ± SD) | Specific Bioactivity of the Released Lysozyme (% ± SD) |
|---|---|---|---|---|
| LF1 | 42.97 ± 2.48 | 18.54 ± 3.65 | 61.51 ± 3.73 | 100.78 ± 4.90 |
| LF2 | 32.13 ± 3.41 | 13.98 ± 4.38 | 46.11 ± 1.16 | 101.43 ± 4.60 |
| LF3 | 41.74 ± 2.02 | 19.75 ± 3.21 | 61.48 ± 4.37 | 99.54 ± 3.25 |
| LF4 | 30.97 ± 2.38 | 12.14 ± 3.53 | 43.10 ± 1.21 | 99.14 ± 5.56 |
| LF6 | 69.21 ± 2.69 | 21.02 ± 3.11 | 90.23 ± 4.92 | 101.15 ± 4.08 |
| Excipients Present in Internal Aqueous Phase (w/v)% | Excipients Present in External Aqueous Phase (w/v)% | Mean Particle Size (µm ± SD) | Span ± SD | Encapsulation Efficiency (± SD) |
|---|---|---|---|---|
| - | - | 1.64 ± 0.58 | 1.01 ± 0.08 | 71.04 ± 3.08 |
| 1% PVA | - | 1.73 ± 0.53 | 1.12 ± 0.05 | 75.65 ± 4.62 |
| 2.5% trehalose | - | 4.34 ± 1.20 | 1.20 ± 0.12 | 54.15 ± 3.80 |
| 1% PVA | 2.5% trehalose | 2.61 ± 0.50 | 1.09 ± 0.07 | 82.52 ± 3.28 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ansary, R.H.; Rahman, M.M.; Mohamad, N.; Arrif, T.M.; Latif, A.Z.A.; Katas, H.; Nik, W.S.B.W.; Awang, M.B. Controlled Release of Lysozyme from Double-Walled Poly(Lactide-Co-Glycolide) (PLGA) Microspheres. Polymers 2017, 9, 485. https://doi.org/10.3390/polym9100485
Ansary RH, Rahman MM, Mohamad N, Arrif TM, Latif AZA, Katas H, Nik WSBW, Awang MB. Controlled Release of Lysozyme from Double-Walled Poly(Lactide-Co-Glycolide) (PLGA) Microspheres. Polymers. 2017; 9(10):485. https://doi.org/10.3390/polym9100485
Chicago/Turabian StyleAnsary, Rezaul H., Mokhlesur M. Rahman, Nasir Mohamad, Tengku M. Arrif, Ahmad Zubaidi A. Latif, Haliza Katas, Wan Sani B. Wan Nik, and Mohamed B. Awang. 2017. "Controlled Release of Lysozyme from Double-Walled Poly(Lactide-Co-Glycolide) (PLGA) Microspheres" Polymers 9, no. 10: 485. https://doi.org/10.3390/polym9100485
APA StyleAnsary, R. H., Rahman, M. M., Mohamad, N., Arrif, T. M., Latif, A. Z. A., Katas, H., Nik, W. S. B. W., & Awang, M. B. (2017). Controlled Release of Lysozyme from Double-Walled Poly(Lactide-Co-Glycolide) (PLGA) Microspheres. Polymers, 9(10), 485. https://doi.org/10.3390/polym9100485




