Design of Graphene- and Polyaniline-Containing Functional Polymer Hydrogel as a New Adsorbent for Removal of Chromium (VI) Ions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
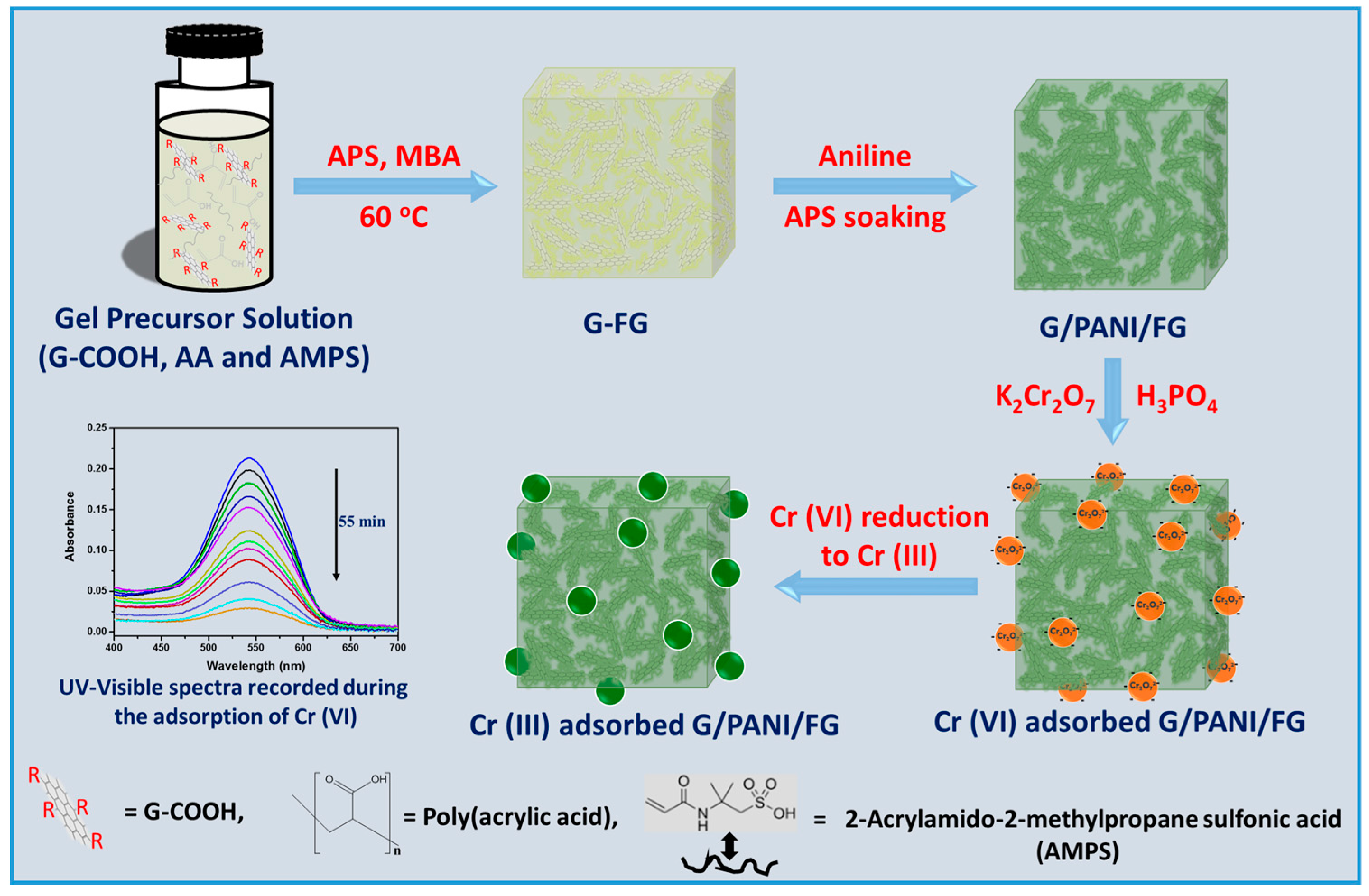
2.2. Preparation of G/PANI/FG
2.2.1. Preparation of G/P(AMPS-co-AA)
2.2.2. PANI Modification on G/P(AMPS-co-AA)/PANI/FG
2.3. Instrumentation
2.4. Experiments for Removal of Cr (VI)
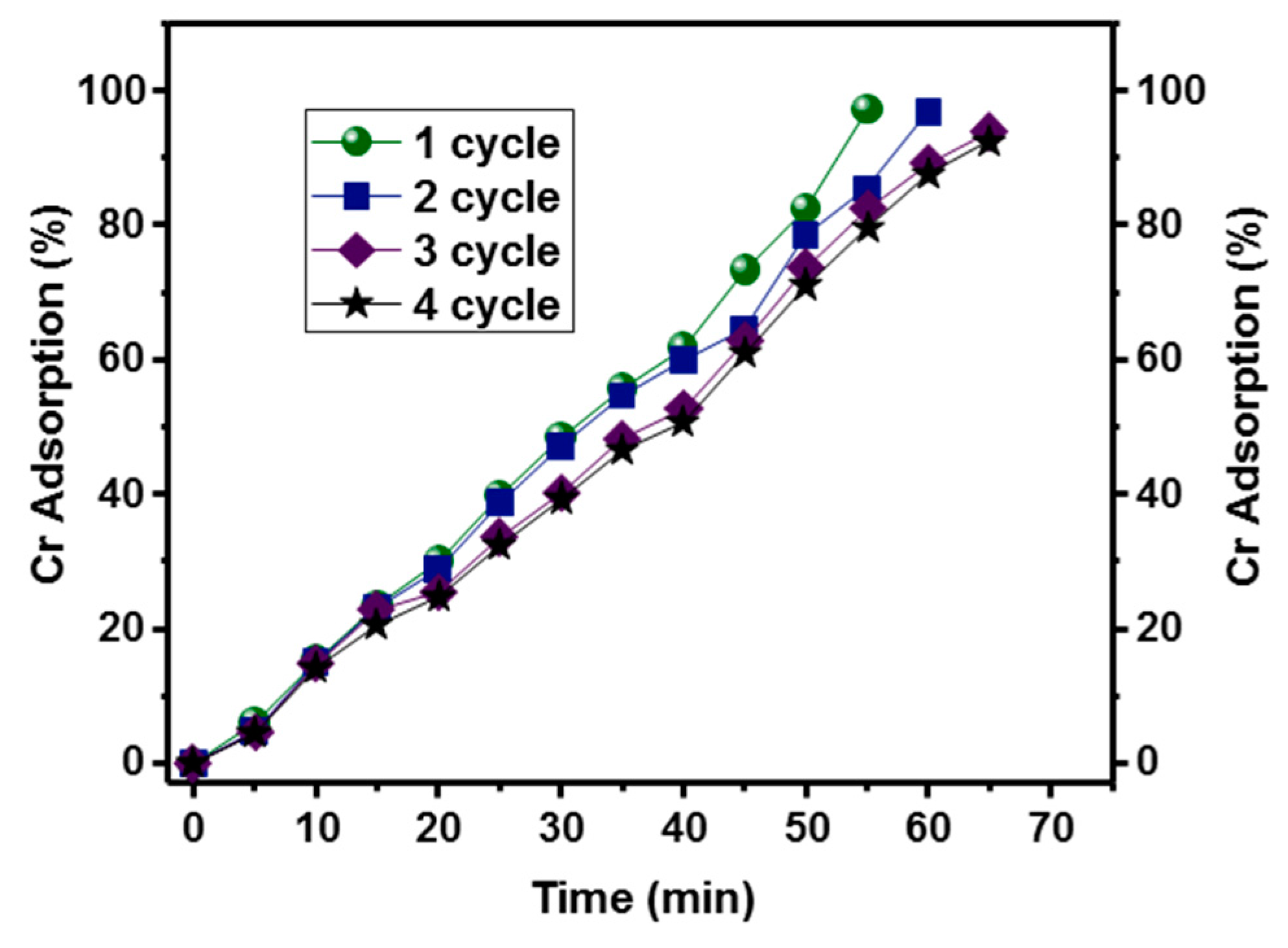
2.5. Reproducibility
3. Results and Discussion
3.1. Morphology
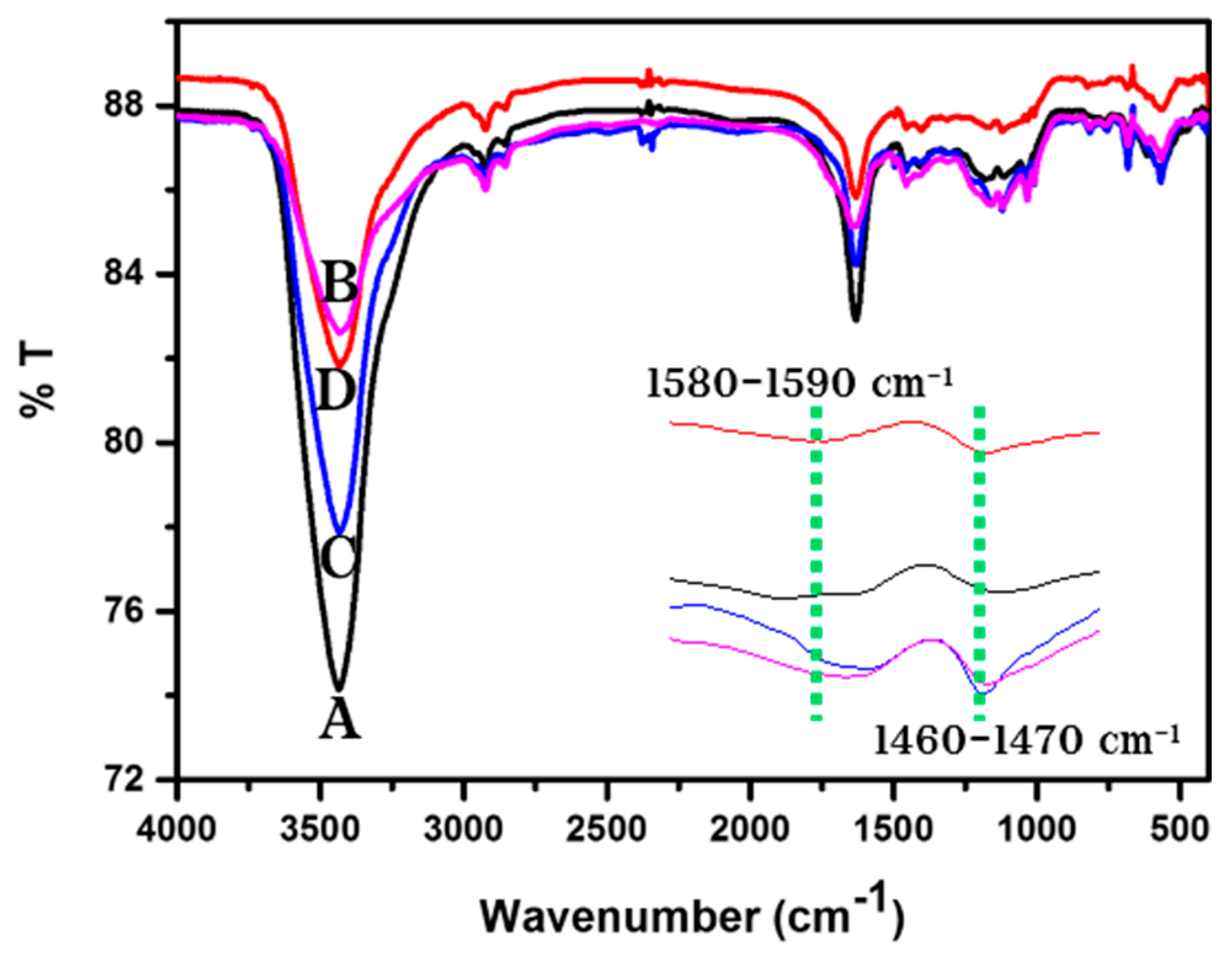
3.2. FTIR Studies
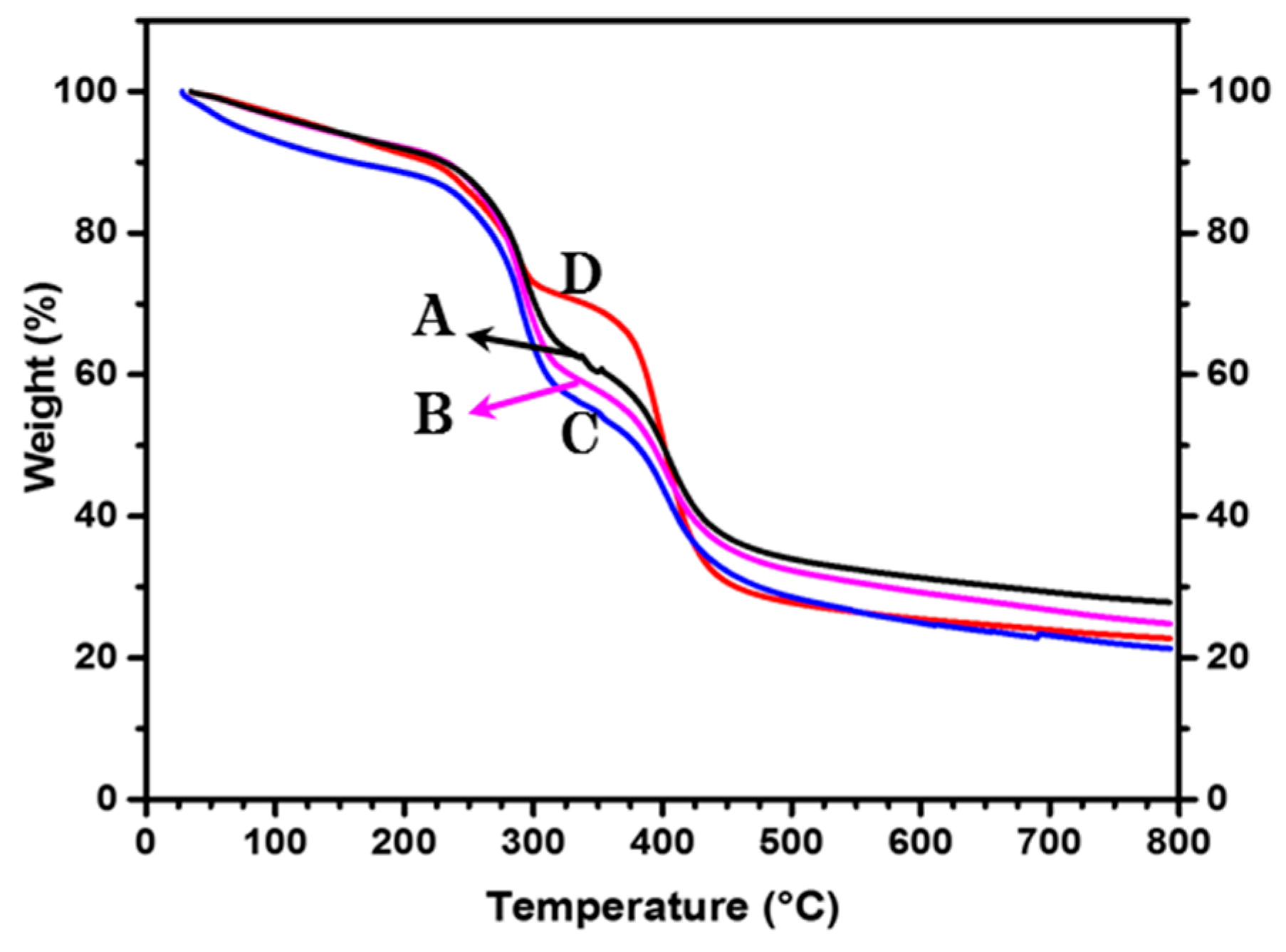
3.3. Thermogravimetric Analysis (TGA)
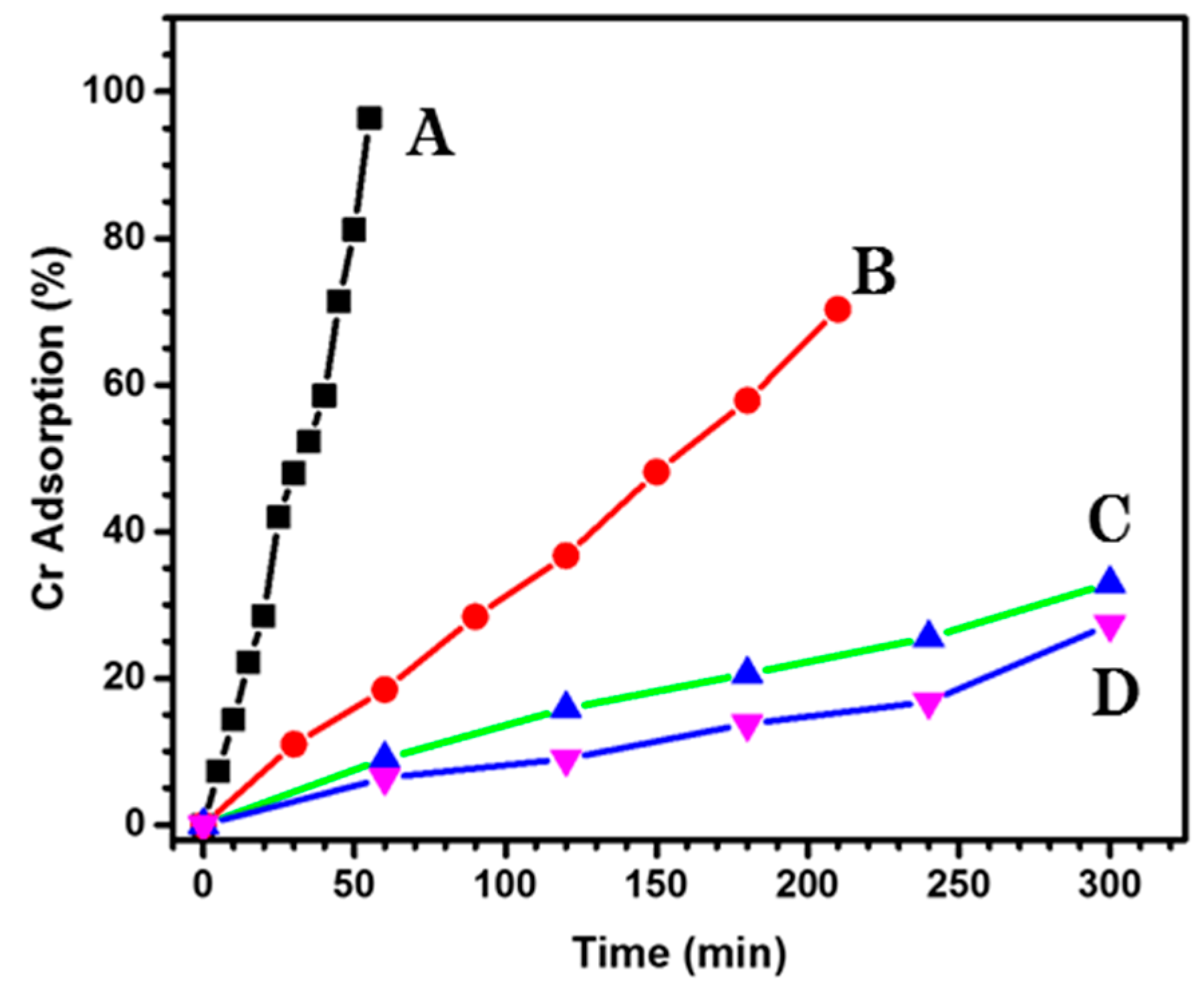
3.4. Adsorption Kinetics of Cr (VI)
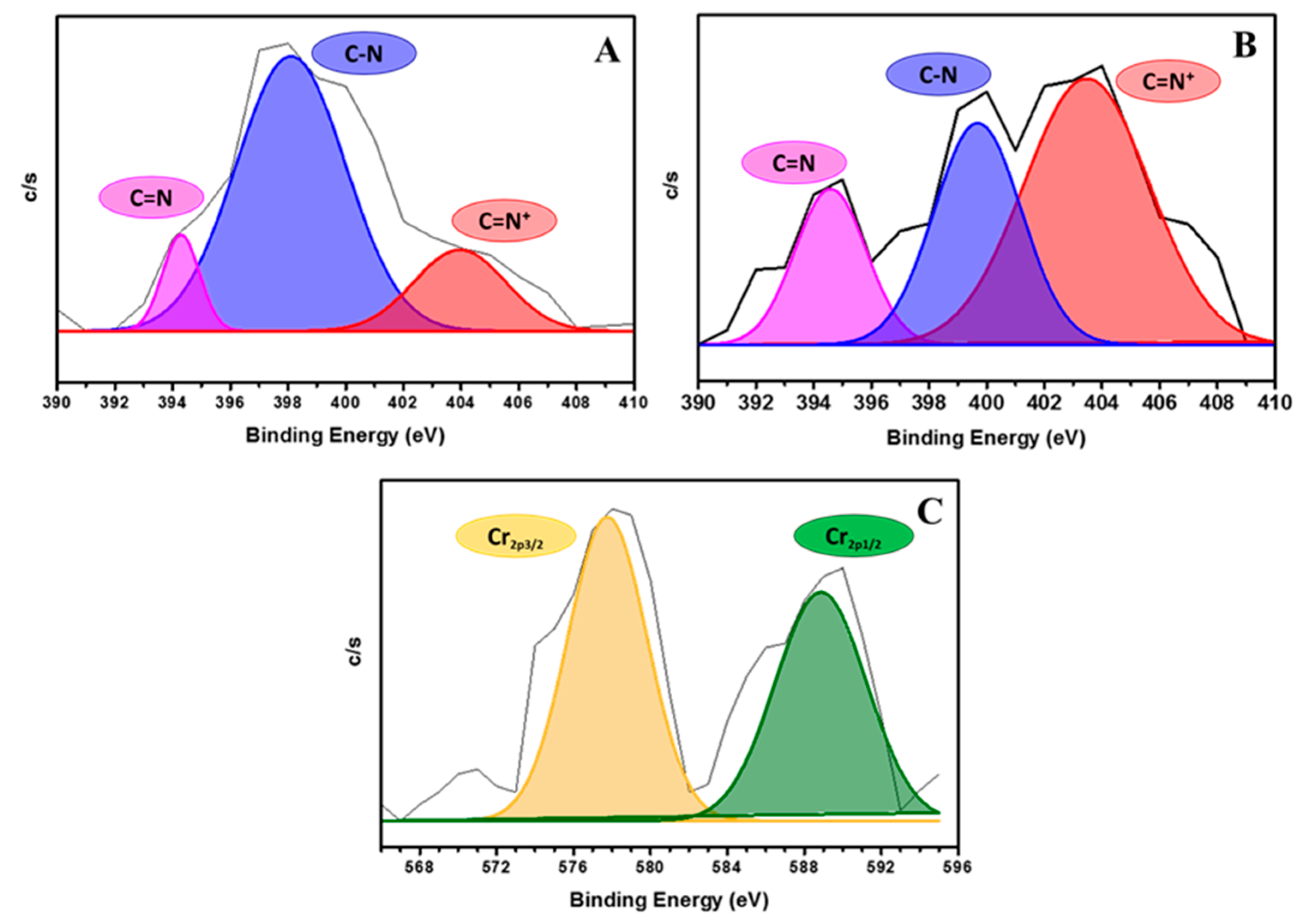
3.5. Mechanism of Cr (VI) Adsorption
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hsu, L.; Wang, S.; Lin, Y.; Wang, M.; Chiang, P.; Liu, J.; Kuan, W.; Chen, C.; Tzou, Y. Cr (VI) removal on fungal biomass of Neurospora crassa: The importance of dissolved organic carbons derived from the biomass to Cr (VI) reduction. Environ. Sci. Technol. 2010, 44, 6202–6208. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, J.; Persson, I.; Oromieh, A.; van Schaik, J.; Sjöstedt, C.; Kleja, D. Chromium(III) complexation to natural organic matter: Mechanisms and modeling. Environ. Sci. Technol. 2014, 48, 1753–1761. [Google Scholar] [CrossRef] [PubMed]
- Gheju, M. Hexavalent chromium reduction with zero-valent iron (ZVI) in aquatic systems. Water Air Soil Pollut. 2011, 222, 103–148. [Google Scholar] [CrossRef]
- Mukherjee, K.; Saha, R.; Ghosh, A.; Saha, B. Chromium removal technologies. Res. Chem. Intermed. 2013, 39, 2267–2286. [Google Scholar] [CrossRef]
- Sharma, S.; Petrusevski, B.; Amy, G. Chromium removal from water: A review. J. Water Supply Res. Technol. 2008, 57, 541–553. [Google Scholar] [CrossRef]
- Barrera-Díaz, C.; Lugo-Lugo, V.; Bilyeu, B. A review of chemical, electrochemical and biological methods for aqueous Cr (VI) reduction. J. Hazard. Mater. 2012, 223, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, K.; Zhang, Z.; Borthwick, A. Land-use suitability analysis for urban development in Beijing. J. Environ. Manag. 2014, 145, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tian, Y.; Guo, Y.; Gao, H.; Li, H.; Yan, S. Introduction of α-MnO2 nanosheets to NH2 graphene to remove Cr6+ from aqueous solutions. RSC Adv. 2015, 5, 44096–44106. [Google Scholar] [CrossRef]
- Lei, Y.; Chen, F.; Luo, Y.; Zhang, L. Three-dimensional magnetic graphene oxide foam/Fe3O4 nanocomposite as an efficient absorbent for Cr (VI) removal. J. Mater. Sci. 2014, 49, 4236–4245. [Google Scholar] [CrossRef]
- Han, X.; Wong, Y.; Wong, M.; Tam, N. Biosorption and bioreduction of Cr (VI) by a microalgal isolate, Chlorella miniata. J. Hazard. Mater. 2007, 146, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Sundaram, K.; Iyengar, G.; Lee, K. A novel chitosan functional gel included with multiwall carbon nanotube and substituted polyaniline as an adsorbent for efficient removal of chromium ion. Chem. Eng. J. 2015, 267, 51–64. [Google Scholar] [CrossRef]
- Li, X.; Huang, M.; Duan, W.; Yang, Y. Novel multifunctional polymers from aromatic diamines by oxidative polymerizations. Chem. Rev. 2002, 102, 2925–3030. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ma, H.; Peng, J.; Zhang, Y.; Chen, J.; Wang, L.; Xu, L.; Li, J.; Zhai, M. Facile synthesis of a novel polymeric ionic liquid gel and its excellent performance for hexavalent chromium removal. Dalton Trans. 2015, 44, 7618–7625. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Li, X.; Peng, J.; Xu, L.; Li, J.; Li, H.; Zhai, M. A new imidazolium-based polymeric ionic liquid gel with high adsorption capacity for perrhenate. RSC Adv. 2016, 6, 69052–69059. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, B.; Gao, T.; Wu, X.; Lou, S.; Zhou, S. Synthesis of orange-like Fe3O4/PPy composite microspheres and their excellent Cr (VI) ion removal properties. J. Mater. Chem. 2012, 22, 9034–9040. [Google Scholar] [CrossRef]
- Chiou, N.; Epstein, A. Polyaniline nanofibers prepared by dilute polymerization. Adv. Mater. 2005, 17, 1679–1683. [Google Scholar] [CrossRef]
- Guo, X.; Fei, G.; Su, H.; De Zhang, L. High-performance and reproducible polyaniline nanowire/tubes for removal of Cr (VI) in aqueous solution. J. Phys. Chem. C 2011, 115, 1608–1613. [Google Scholar] [CrossRef]
- Tian, Y.; Li, H.; Liu, Y.; Cui, G.; Sun, Z.; Yan, S. Morphology-dependent enhancement of template-guided tunable polyaniline nanostructures for the removal of Cr (VI). RSC Adv. 2016, 6, 10478–10486. [Google Scholar] [CrossRef]
- Olad, A.; Nabavi, R. Application of polyaniline for the reduction of toxic Cr (VI) in water. J. Hazard. Mater. 2007, 147, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ma, H.; Wang, B. Removal of chromium(VI) from aqueous solutions using polyaniline doped with sulfuric acid. Ind. Eng. Chem. Res. 2010, 49, 9998–10004. [Google Scholar] [CrossRef]
- Ansari, R. Application of polyaniline and its composites for adsorption/recovery of chromium(VI) from aqueous solutions. Acta Chim. Slov. 2006, 53, 88. [Google Scholar]
- Wang, J.; Pan, K.; Giannelis, E.; Cao, B. Polyacrylonitrile/polyaniline core/shell nanofiber mat for removal of hexavalent chromium from aqueous solution: mechanism and applications. RSC Adv. 2013, 3, 8978–8987. [Google Scholar] [CrossRef]
- Shen, Y.; Fang, Q.; Chen, B. Environmental applications of three-dimensional graphene-based macrostructures: Adsorption, transformation, and detection. Environ. Sci. Technol. 2014, 49, 67–84. [Google Scholar] [CrossRef] [PubMed]
- WooáLee, J.; BináKim, S. Enhanced Cr (VI) removal using iron nanoparticle decorated graphene. Nanoscale 2011, 3, 3583–3585. [Google Scholar]
- Zhang, K.; Kemp, K.; Chandra, V. Homogeneous anchoring of TiO2 nanoparticles on graphene sheets for waste water treatment. Mater. Lett. 2012, 81, 127–130. [Google Scholar] [CrossRef]
- Ju, M.; Jeon, I.; Lim, K.; Kim, J.; Choi, H.; Choi, I.; Eom, Y.; Kwon, Y.; Ko, J.; Lee, J.; et al. Edge-carboxylated graphene nanoplatelets as oxygen-rich metal-free cathodes for organic dye-sensitized solar cells. Energy Environ. Sci. 2014, 7, 1044–1052. [Google Scholar] [CrossRef]
- Huang, L.; Li, C.; Yuan, W.; Shi, G. Strong composite films with layered structures prepared by casting silk fibroin—Graphene oxide hydrogels. Nanoscale 2013, 5, 3780–3786. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Feng, J.; Wu, P. Highly elastic graphene oxide-epoxy composite aerogels via simple freeze-drying and subsequent routine curing. J. Mater. Chem. A 2013, 1, 3495–3502. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Nakao, S.; Kimura, S. Plasma-graft filling polymerization: Preparation of a new type of pervaporation membrane for organic liquid mixtures. Macromolecules 1991, 24, 5522–5527. [Google Scholar] [CrossRef]
- Adrus, N.; Ulbricht, M. Novel hydrogel pore-filled composite membranes with tunable and temperature-responsive size-selectivity. J. Mater. Chem. 2012, 22, 3088–3098. [Google Scholar] [CrossRef]
- Yu, P.; Li, Y.; Zhao, X.; Wu, L.; Zhang, Q. Graphene-wrapped polyaniline nanowire arrays on nitrogen-doped carbon fabric as novel flexible hybrid electrode materials for high-performance supercapacitor. Langmuir 2014, 30, 5306–5313. [Google Scholar] [CrossRef] [PubMed]
- Mitra, M.; Kulsi, C.; Chatterjee, K.; Kargupta, K.; Ganguly, S.; Banerjee, D.; Goswami, S. Reduced graphene oxide-polyaniline composites—Synthesis, characterization and optimization for thermoelectric applications. RSC Adv. 2015, 5, 31039–31048. [Google Scholar] [CrossRef]
- Kabiri, K.; Azizi, A.; Zohuriaan-Mehr, M.; Marandi, G.; Bouhendi, H. Poly(acrylic acid-sodium styrene sulfonate) organogels: Preparation, characterization, and alcohol superabsorbency. J. Appl. Polym. Sci. 2011, 119, 2759–2769. [Google Scholar] [CrossRef]
- Gomez, V.; Callao, M. Chromium determination and speciation since 2000. Trends Anal. Chem. 2006, 25, 1006–1015. [Google Scholar] [CrossRef]
- Laslau, C.; Zujovic, Z.; Zhang, L.; Bowmaker, G.; Travas-Sejdic, J. Morphological evolution of self-assembled polyaniline nanostuctures obtained by pH-stat chemical oxidation. Chem. Mater. 2009, 21, 954–962. [Google Scholar] [CrossRef]
- Xu, G.; Wang, J.; Li, C. Preparation of hierarchically nanofibrous membrane and its high adaptability in hexavalent chromium removal from water. Chem. Eng. J. 2012, 198, 310–317. [Google Scholar] [CrossRef]
- Lin, Y.; Cai, W.; Tian, X.; Liu, X.; Wang, G.; Liang, C. Polyacrylonitrile/ferrous chloride composite porous nanofibers and their strong Cr-removal performance. J. Mater. Chem. 2011, 21, 991–997. [Google Scholar] [CrossRef]
- Bhaumik, M.; Maity, A.; Srinivasu, V.; Onyango, M. Enhanced removal of Cr (VI) from aqueous solution using polypyrrole/Fe3O4 magnetic nanocomposite. J. Hazard. Mater. 2011, 190, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Biesinger, M.; Brown, C.; Mycroft, J.; Davidson, R.; McIntyre, N. X-ray photoelectron spectroscopy studies of chromium compounds. Surf. Interface Anal. 2004, 36, 1550–1563. [Google Scholar] [CrossRef]
- Manning, B.; Kiser, J.; Kwon, H.; Kanel, S. Spectroscopic investigation of Cr(III)-and Cr (VI)-treated nanoscale zerovalent iron. Environ. Sci. Technol. 2007, 41, 586–592. [Google Scholar] [CrossRef] [PubMed]


| Feed composition of the gel in stage I | Precursor gel | Further inclusion stage II | Final gel designation |
|---|---|---|---|
| G (10 mg), AA (1.125 M) and AMPS (0.125 M) | G/P(AMPS-co-AA) | PANI | G/PANI/FG1 |
| G (10 mg) and AA (1.125 M) | G/* PAA | PANI | G/PANI/FG2 |
| G (10 mg) and AMPS (0.125 M) | G/** PAMPS | PANI | G/PANI/FG3 |
| AA (1.125 M) and AMPS (0.125 M) | P(AMPS-co-AA) | PANI | PANI/*** GNG |
| Area (%) | ||
|---|---|---|
| Before Cr (VI) adsorption | After Cr (VI) adsorption | |
| C=N+ | 18.09 | 52.23 |
| C–N | 73.25 | 30.34 |
| C=N | 08.65 | 17.41 |
| * imine/amine ratio | 0.365 | 6.27 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chin, J.-S.; Gopalan, A.-I.; Muthuchamy, N.; Lee, K.-P. Design of Graphene- and Polyaniline-Containing Functional Polymer Hydrogel as a New Adsorbent for Removal of Chromium (VI) Ions. Polymers 2016, 8, 445. https://doi.org/10.3390/polym8120445
Chin J-S, Gopalan A-I, Muthuchamy N, Lee K-P. Design of Graphene- and Polyaniline-Containing Functional Polymer Hydrogel as a New Adsorbent for Removal of Chromium (VI) Ions. Polymers. 2016; 8(12):445. https://doi.org/10.3390/polym8120445
Chicago/Turabian StyleChin, Jae-Seo, Anantha-Iyengar Gopalan, Nallal Muthuchamy, and Kwang-Pill Lee. 2016. "Design of Graphene- and Polyaniline-Containing Functional Polymer Hydrogel as a New Adsorbent for Removal of Chromium (VI) Ions" Polymers 8, no. 12: 445. https://doi.org/10.3390/polym8120445
APA StyleChin, J.-S., Gopalan, A.-I., Muthuchamy, N., & Lee, K.-P. (2016). Design of Graphene- and Polyaniline-Containing Functional Polymer Hydrogel as a New Adsorbent for Removal of Chromium (VI) Ions. Polymers, 8(12), 445. https://doi.org/10.3390/polym8120445









