Characterization of Epoxy Composites Reinforced with Wax Encapsulated Microcrystalline Cellulose
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Coating Cellulose Particles
2.3. Differential Interference Contrast (DIC) Microscopy
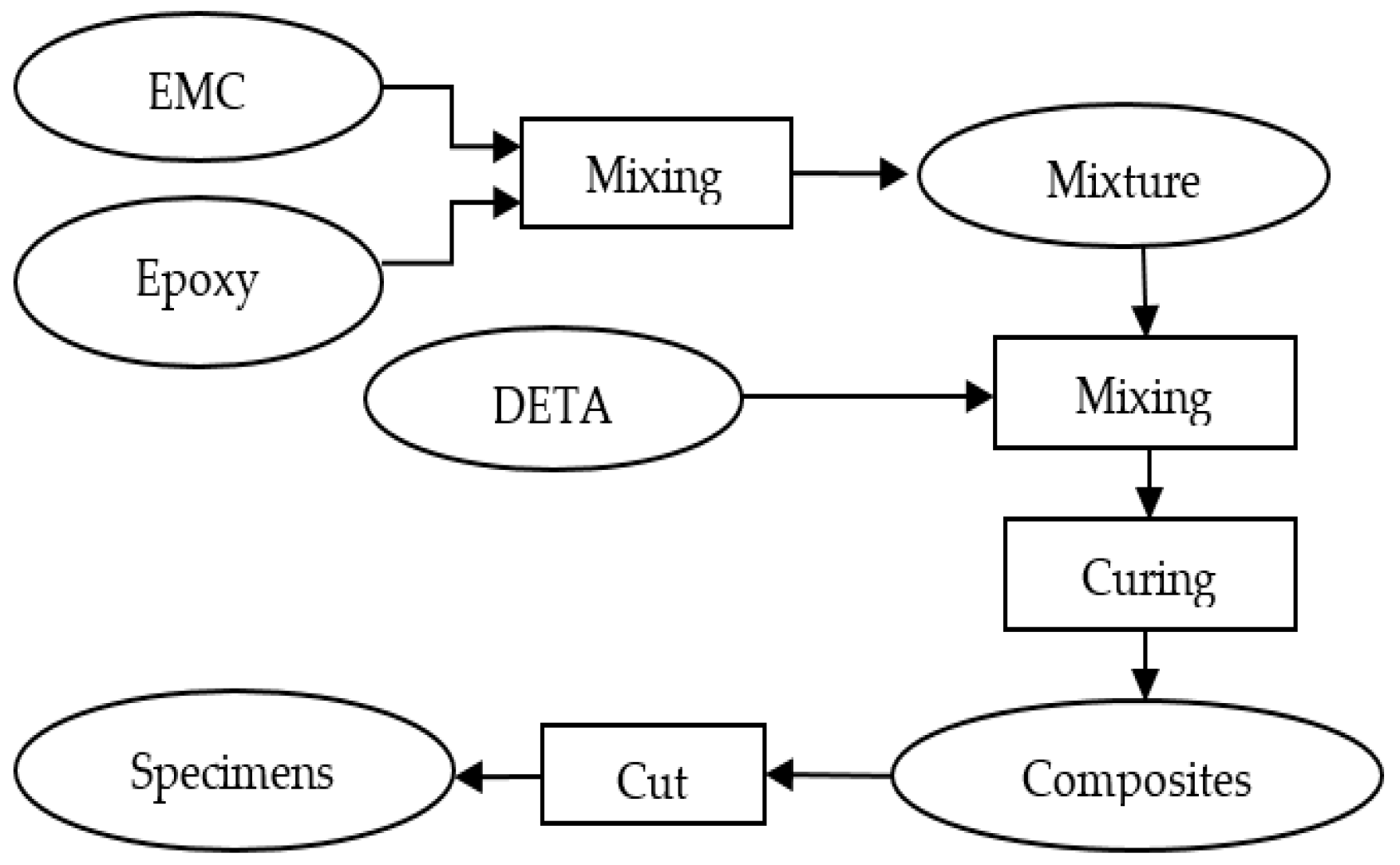
2.4. Preparation of EMC Reinforced Epoxy Composites
2.5. Water Absorption
2.6. Mechanical Properties
2.7. Scanning Electron Microscopy (SEM)
2.8. Fourier Transform Infrared Spectroscopy
2.9. Thermal Analysis
2.10. Data Statistical Analysis
3. Results and Discussion
3.1. MCC and EMC Morphology
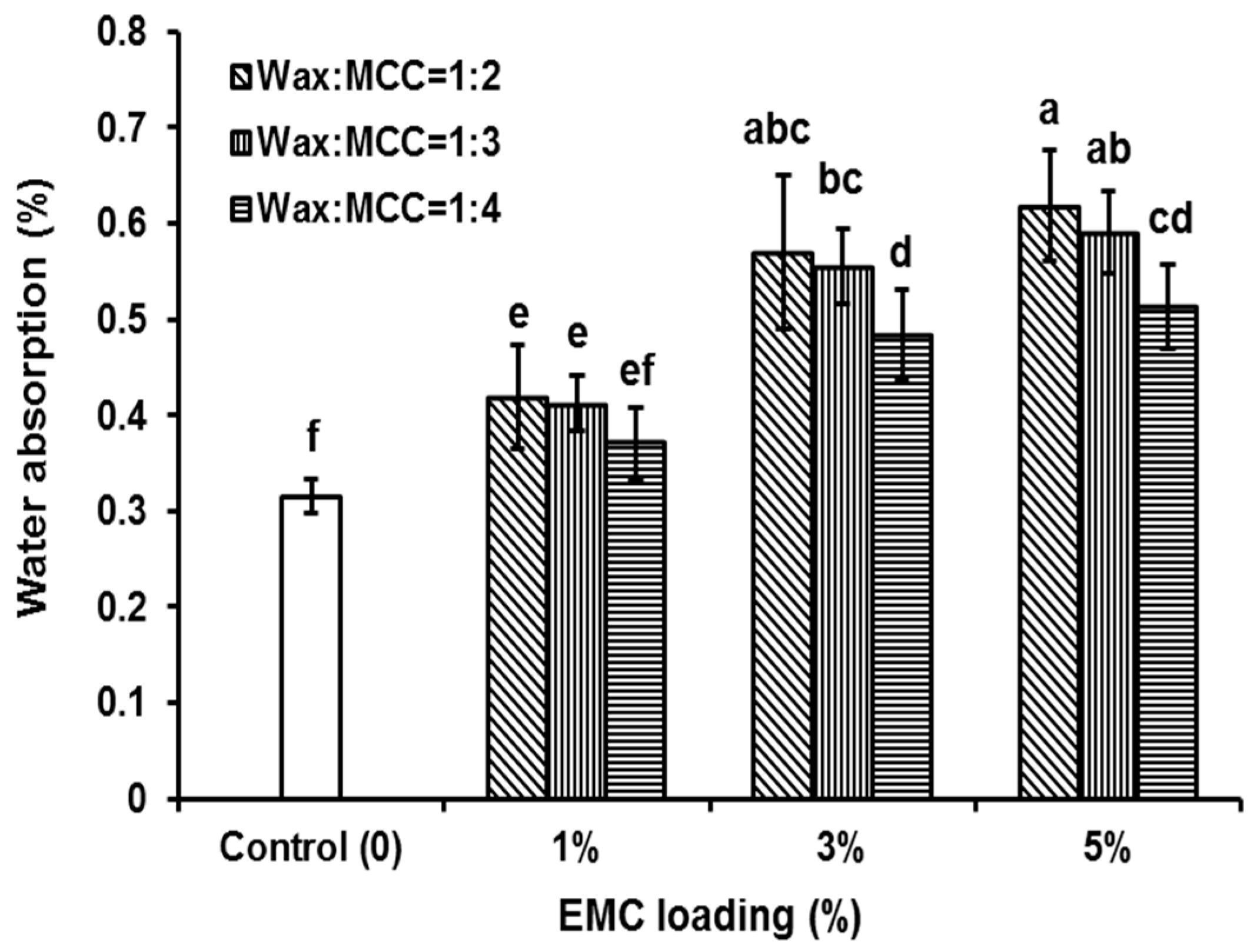
3.2. Water Absorption
3.3. Flexure Properties
3.4. SEM Observation of EMC/Epoxy Resin Composites
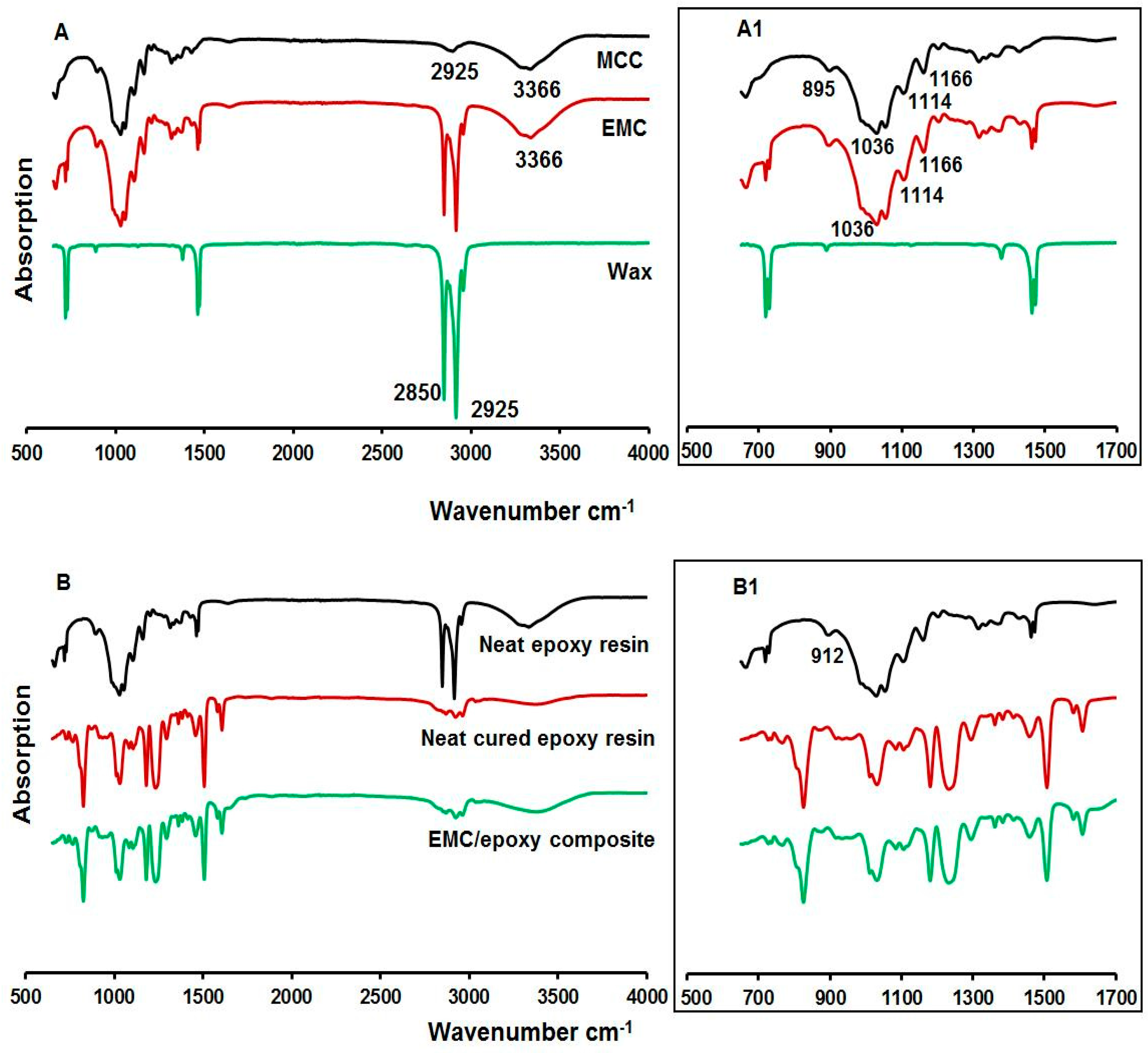
3.5. FT-IR Spectroscopy
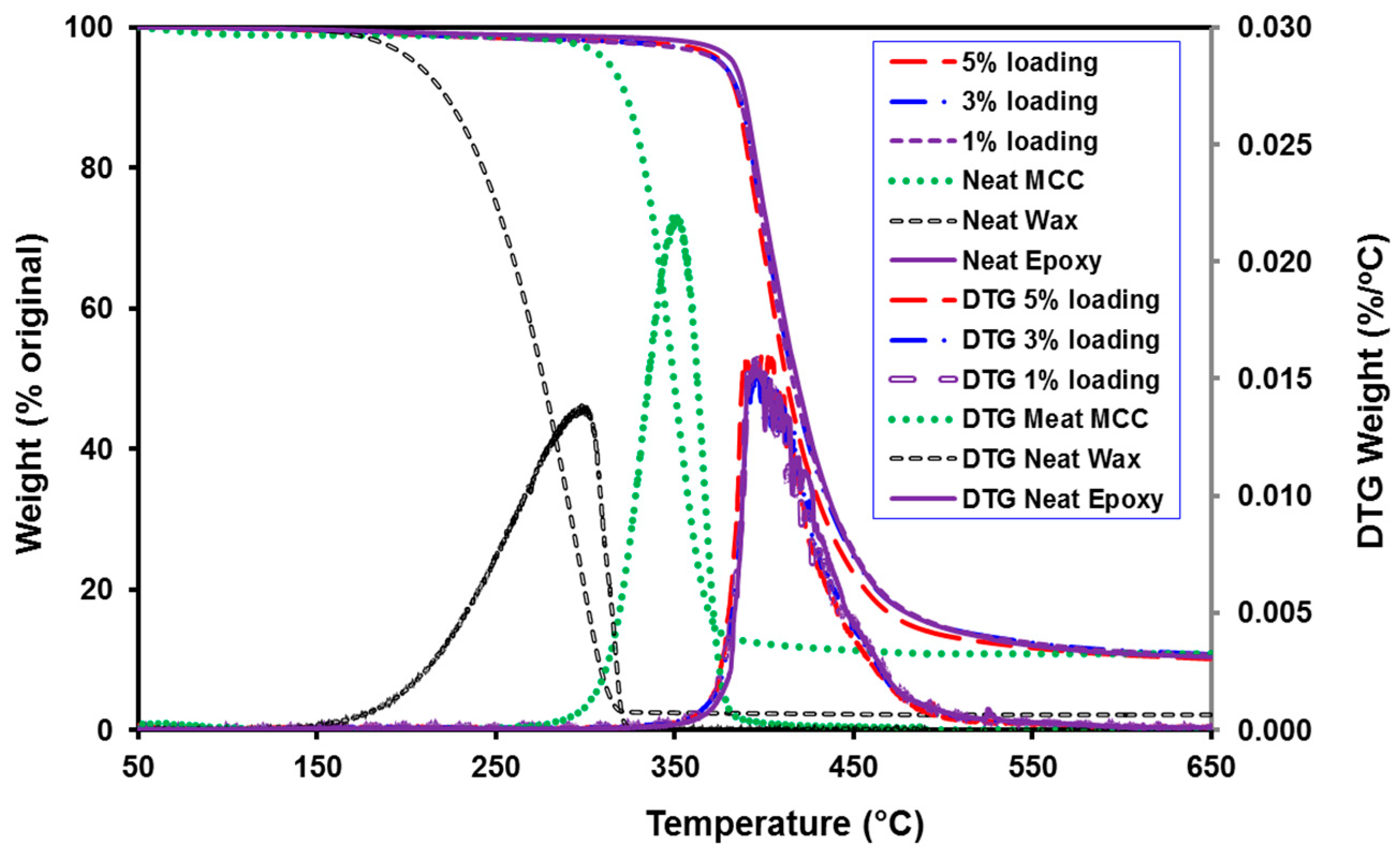
3.6. Thermal Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DIC | Differential Interference Contrast Microscopy |
| EMC | Encapsulated Microcrystalline Cellulose |
| FTIR | Fourier Transform Infrared spectroscopy |
| MCC | Microcrystalline Cellulose |
| TGA | Thermogravimetric Analyses |
References
- Alamri, H.; Low, I.M. Effect of water absorption on the mechanical properties of nano-filler reinforced epoxy nanocomposites. Mater. Des. 2012, 42, 214–222. [Google Scholar] [CrossRef]
- Baley, C.; Davies, P.; Grohens, Y.; Dolto, G. Application of interlaminar tests to marine composites. Relation between glass fibre/polymer interfaces and interlaminar properties of marine composites. Appl. Compos. Mater. 2004, 11, 99–126. [Google Scholar] [CrossRef]
- Gojny, F.H.; Wichmann, M.H.G.; Köpke, U.; Fiedler, B.; Schulte, K. Carbon nanotube-reinforced epoxy-composites: Enhanced stiffness and fracture toughness at low nanotube content. Compos. Sci. Technol. 2004, 64, 2363–2371. [Google Scholar] [CrossRef]
- Rotrekl, J.; Sikora, A.; Kaprálková, L.; Dybal, J.; Kelnar, I. Effect of an organoclay on the reaction-induced phase-separation in a dynamically asymmetric epoxy/PCL system. Express Polym. Lett. 2013, 7, 1012–1019. [Google Scholar] [CrossRef]
- Rout, J.; Misra, M.; Tripathy, S.S.; Nayak, S.K.; Mohanty, A.K. The influence of fibre treatment on the performance of coir-polyester composites. Compos. Sci. Technol. 2001, 61, 1303–1310. [Google Scholar] [CrossRef]
- Chen, W.; Gu, J.; Xu, S. Exploring nanocrystalline cellulose as a green alternative of carbon black in natural rubber/butadiene rubber/styrene-butadiene rubber blends. Express Polym. Lett. 2014, 8, 659–668. [Google Scholar] [CrossRef]
- Lee, K.Y.; Ho, K.K.C.; Schlufter, K.; Bismarck, A. Hierarchical composites reinforced with robust short sisal fibre preforms utilising bacterial cellulose as binder. Compos. Sci. Technol. 2012, 72, 1479–1486. [Google Scholar] [CrossRef]
- Kamel, S.; Ali, N.; Jahangir, K.; Shah, S.; El-Gendy, A. Pharmaceutical significance of cellulose: A review. Express Polym. Lett. 2008, 2, 758–778. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Hinrichsen, G. Biofibres, biodegradable polymer and composites: An overview. Mater. Eng. 2000, 276, 1–24. [Google Scholar]
- Gassan, J.; Bledzki, A.K. Possibilities to improve the properties of natural fiber reinforced plastics by fiber modification—Jute polypropylene composites. Appl. Compos. Mater. 2000, 7, 373–385. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Bhat, A.H.; Ireana Yusra, A.F. Green composites from sustainable cellulose nanofibrils: A review. Carbohydr. Polym. 2012, 87, 963–979. [Google Scholar] [CrossRef]
- Lavoine, N.; Desloges, I.; Dufresne, A.; Bras, J. Microfibrillated cellulose—Its barrier properties and applications in cellulosic materials: A review. Carbohydr. Polym. 2012, 90, 735–764. [Google Scholar] [CrossRef] [PubMed]
- La Mantia, F.P.; Morreale, M. Green composites: A brief review. Compos. Part A Appl. Sci. Manuf. 2011, 42, 579–588. [Google Scholar] [CrossRef]
- Kim, D.B.; Jo, S.M.; Lee, W.S.; Pak, J.J. Physical agglomeration behavior in preparation of cellulose-N-methyl morpholine N-oxide hydrate solutions by simple mixing. J. Appl. Polym. Sci. 2004, 93, 1687–1697. [Google Scholar] [CrossRef]
- Silviya, E.K.; Unnikrishnan, G.; Varghese, S.; Guthrie, J.T. Thermal and mechanical characterization of EVA/banana fiber-derived cellulose composites. J. Appl. Polym. Sci. 2012, 125, 786–792. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Reihmane, S.; Gassan, J. Properties and modification methods for vegetable fibers for natural fiber composites. J. Appl. Polym. Sci. 1996, 59, 1329–1336. [Google Scholar] [CrossRef]
- Johansson, L.S.; Tammelin, T.; Campbell, J.M.; Setälä, H.; Österberg, M. Experimental evidence on medium driven cellulose surface adaptation demonstrated using nanofibrillated cellulose. Soft Matter 2011, 7, 10917–10924. [Google Scholar] [CrossRef]
- Gu, W.; Li, H.; Zhan, H.; Ding, J.; Zhang, X. The interface interaction between water-soluble chitosan (DD = 50%) and peroxide bleached reed kraft pulp. J. Appl. Polym. Sci. 2011, 121, 2606–2613. [Google Scholar] [CrossRef]
- Goutianos, S.; Peijs, T.; Nystrom, B.; Skrifvars, M. Development of flax fibre based textile reinforcements for composite applications. Appl. Compos. Mater. 2006, 13, 199–215. [Google Scholar] [CrossRef]
- Dai, X.; Shen, X. Research on microcapsules of phase change materials. Rare Met. 2006, 25, 393–399. [Google Scholar] [CrossRef]
- Emås, M.; Nyqvist, H. Methods of studying aging and stabilization of spray-congealed solid dispersions with carnauba wax. 1. Microcalorimetric investigation. Int. J. Pharm. 2000, 197, 117–127. [Google Scholar] [CrossRef]
- Kamble, R.; Maheshwari, M.; Paradkar, A.; Kadam, S. Melt solidification technique: Incorporation of higher wax content in ibuprofen beads. AAPS PharmSciTech 2004, 5, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Rule, J.D.; Brown, E.N.; Sottos, N.R.; White, S.R.; Moore, J.S. Wax-protected catalyst microspheres for efficient self-healing materials. Adv. Mater. 2005, 17, 205–208. [Google Scholar] [CrossRef]
- Lesar, B.; Humar, M. Use of wax emulsions for improvement of wood durability and sorption properties. Eur. J. Wood Wood Prod. 2011, 69, 231–238. [Google Scholar] [CrossRef]
- Kurt, R.; Krause, A.; Militz, H.; Mai, C. Hydroxymethylated resorcinol (HMR) priming agent for improved bondability of wax-treated wood. Eur. J. Wood Wood Prod. 2008, 66, 333–338. [Google Scholar] [CrossRef]
- Evans, P.D.; Wingate-Hill, R.; Cunningham, R.B. Wax and oil emulsion additives: How effective are they at improving the performance of preservative-treated wood? For. Prod. J. 2009, 59, 66–71. [Google Scholar]
- Cataldia, A.; Corcioneb, C.; Frigioneb, M.; Pegorettia, A. Photocurable resin/microcrystalline cellulose composites for wood protection: Physical-mechanical characterization. Prog. Org. Coat. 2016, 99, 230–239. [Google Scholar] [CrossRef]
- Pan, Y.; Sun, S.; Cheng, Q.; Celikbag, Y.; Via, B.K.; Wang, X.; Sun, R. Technical note: Melt dispersion technique for preparing paraffin wax microspheres for cellulose encapsulation. Wood Fiber Sci. 2014, 46, 1–6. [Google Scholar]
- Pan, Y.; Liu, Y.; Cheng, Q.; Celikbag, Y.; Via, B.K.; Wang, X.; Sun, R. Surface coating of microcrystalline cellulose with surfactants and paraffin wax. Eur. J. Wood Prod. 2016, 74, 763–765. [Google Scholar] [CrossRef]
- Saba, N.; Jawaid, M.; Alothman, O.Y.; Paridah, M.T.; Hassan, A. Recent advances in epoxy resin, natural fiber-reinforced epoxy composites and their applications. J. Reinf. Plast. Compos. 2016, 35, 447–470. [Google Scholar] [CrossRef]
- Chen, M.C.; Hourston, D.J.; Sun, W.B. Miscibility and fracture behaviour of an epoxy resin-bisphenol—A polycarbonate blend. Eur. Polym. J. 1992, 28, 1471–1475. [Google Scholar] [CrossRef]
- Standard Test Methods for Evaluating Properties of Wood-Base Fiber and Particle Panel Materials; ASTM D1037-12; ASTM International: West Conshohocken, PA, USA, 2002.
- Standard Test Methods for Flexural Properties of Unreinforced and Reinforced Plastics and Electrical Insulating Materials; ASTM D790-10; ASTM International: West Conshohocken, PA, USA, 2002.
- Cheng, Q.; Wang, S.; Rials, T.G.; Lee, S.H. Physical and mechanical properties of polyvinyl alcohol and polypropylene composite materials reinforced with fibril aggregates isolated from regenerated cellulose fibers. Cellulose 2007, 14, 593–602. [Google Scholar] [CrossRef]
- Fei, X.; Zhao, F.; Wei, W.; Luo, J.; Chen, M.; Liu, X. Tannic acid as a bio-based modifier of epoxy/anhydride thermosets. Polymers 2016, 8, 314. [Google Scholar] [CrossRef]
- Zanjani, J.; Okan, B.; Menceloglua, Y.; Yildiz, M. Nano-engineered design and manufacturing of high-performance epoxy matrix composites with carbon fiber/selectively integrated graphene as multi-scale reinforcements. RSC Adv. 2016, 6, 9495–9506. [Google Scholar] [CrossRef]
- Capadona, J.R.; Van Den Berg, O.; Capadona, L.A.; Schroeter, M.; Rowan, S.J.; Tyler, D.J.; Weder, C. A versatile approach for the processing of polymer nanocomposites with self-assembled nanofibre templates. Nat. Nanotechnol. 2007, 2, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, H.; Morita, S. Identification of the epoxy curing mechanism under isothermal conditions by thermal analysis and infrared spectroscopy. J. Mol. Struct. 2014, 1069, 164–170. [Google Scholar] [CrossRef]
- Pramanik, M.; Mendon, S.K.; Rawlins, J.W. Determination of epoxy equivalent weight of glycidyl ether based epoxides via near infrared spectroscopy. Polym. Test. 2012, 31, 716–721. [Google Scholar] [CrossRef]
- Tfayli, A.; Piot, O.; Durlach, A.; Bernard, P.; Manfait, M. Discriminating nevus and melanoma on paraffin-embedded skin biopsies using FTIR microspectroscopy. Biochim. Biophys. Acta 2005, 1724, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Krump, H.; Alexy, P.; Luyt, A. Preparation of a maleated Fischer–Tropsch paraffin wax and FTIR analysis of grafted maleic anhydride. Polym. Test. 2005, 24, 129–135. [Google Scholar] [CrossRef]
- Atta-Obeng, E.; Via, B.K.; Fasina, O. Effect of microcrystalline cellulose, species, and particle size on mechanical and physical properties of particleboard. Wood Fiber Sci. 2012, 44, 227–235. [Google Scholar]
- Tang, L.; Weder, C. Cellulose whisker/epoxy resin nanocomposites. ACS Appl. Mater. Interfaces 2010, 2, 1073–1080. [Google Scholar] [CrossRef] [PubMed]

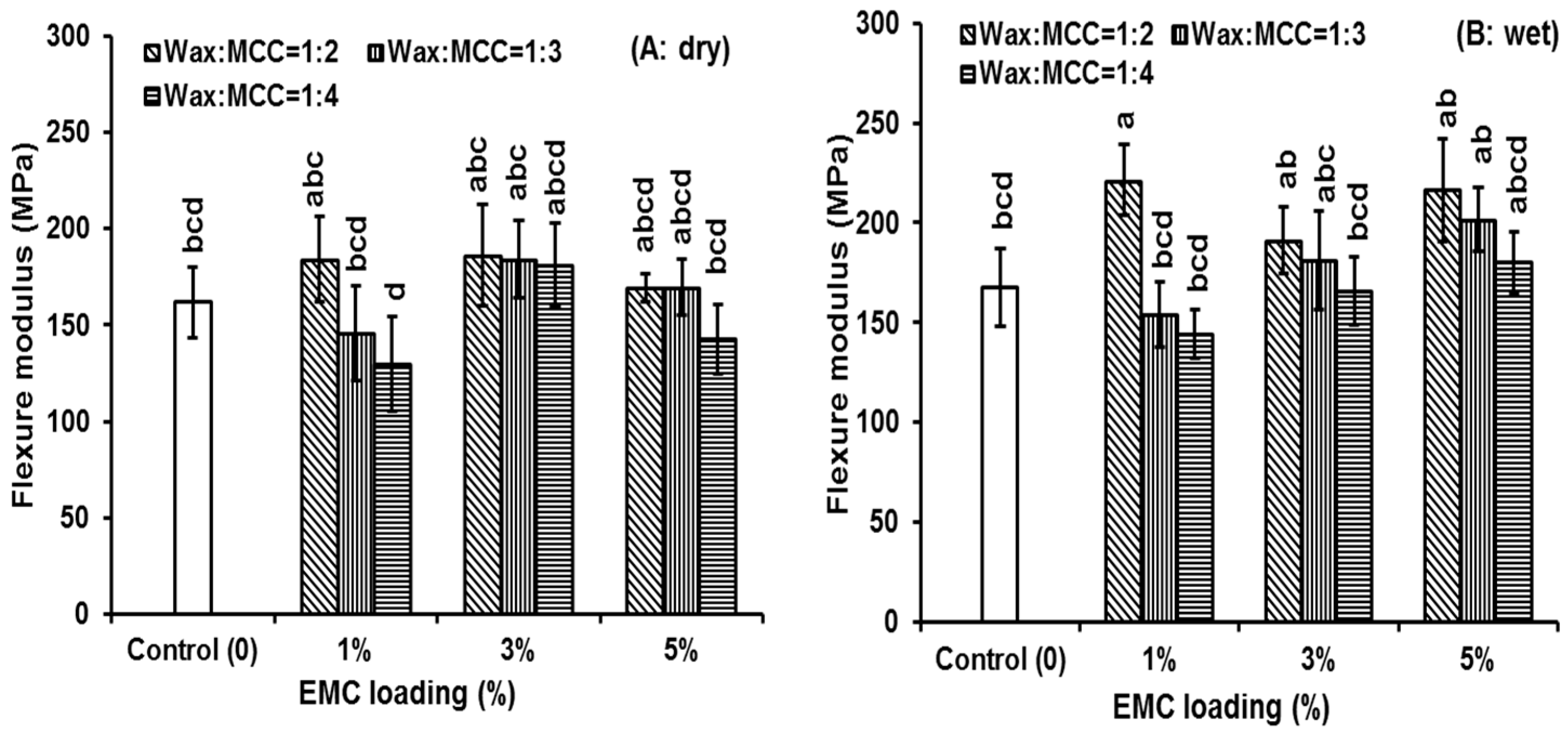

| Loading | Wax:MCC | Dry | Wet | ||
|---|---|---|---|---|---|
| MOE (MPa) 1 | Significance 2 | MOE (MPa) 1 | Significance 2 | ||
| Control (0) | 0:0 | 159.3 (39.1) | 161.7 (29.8) | ||
| 1% | 1:2 | 210.1 (35.9) | * | 221.3 (39.9) | * |
| 1:3 | 165.3 (33.2) | 153.9 (36.6) | |||
| 1:4 | 142.7 (31.2) | 144.4 (15.8) | |||
| 3% | 1:2 | 191.7 (46.9) | 191.4 (37.4) | ||
| 1:3 | 187.5 (39.5) | 185.0 (45.0) | |||
| 1:4 | 184.0 (44.6) | 165.8 (38.0) | |||
| 5% | 1:2 | 169.4 (16.2) | 216.5 (28.5) | * | |
| 1:3 | 169.6 (32.7) | 201.6 (20.5) | * | ||
| 1:4 | 146.8 (34.4) | 180.3 (25.3) | |||
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, Y.; Pan, Y.; Cheng, Q.; Liu, Y.; Essien, C.; Via, B.; Wang, X.; Sun, R.; Taylor, S. Characterization of Epoxy Composites Reinforced with Wax Encapsulated Microcrystalline Cellulose. Polymers 2016, 8, 415. https://doi.org/10.3390/polym8120415
Pan Y, Pan Y, Cheng Q, Liu Y, Essien C, Via B, Wang X, Sun R, Taylor S. Characterization of Epoxy Composites Reinforced with Wax Encapsulated Microcrystalline Cellulose. Polymers. 2016; 8(12):415. https://doi.org/10.3390/polym8120415
Chicago/Turabian StylePan, Yuanfeng, Ying Pan, Qingzheng Cheng, Yi Liu, Charles Essien, Brian Via, Xiaoying Wang, Runcang Sun, and Steven Taylor. 2016. "Characterization of Epoxy Composites Reinforced with Wax Encapsulated Microcrystalline Cellulose" Polymers 8, no. 12: 415. https://doi.org/10.3390/polym8120415
APA StylePan, Y., Pan, Y., Cheng, Q., Liu, Y., Essien, C., Via, B., Wang, X., Sun, R., & Taylor, S. (2016). Characterization of Epoxy Composites Reinforced with Wax Encapsulated Microcrystalline Cellulose. Polymers, 8(12), 415. https://doi.org/10.3390/polym8120415







