Dark Antimicrobial Mechanisms of Cationic Phenylene Ethynylene Polymers and Oligomers against Escherichia coli
Abstract
: The interactions of poly(phenylene ethynylene) (PPE)-based cationic conjugated polyelectrolytes (CPEs) and oligo-phenylene ethynylenes (OPEs) with E. coli cells are investigated to gain insights into the differences in the dark killing mechanisms between CPEs and OPEs. A laboratory strain of E. coli with antibiotic resistance is included in this work to study the influence of antibiotic resistance on the antimicrobial activity of the CPEs and OPEs. In agreement with our previous findings, these compounds can efficiently perturb the bacterial cell wall and cytoplasmic membrane, resulting in bacterial cell death. Electron microscopy imaging and cytoplasmic membrane permeability assays reveal that the oligomeric OPEs penetrate the bacterial outer membrane and interact efficiently with the bacterial cytoplasmic membrane. In contrast, the polymeric CPEs cause serious damage to the cell surface. In addition, the minimum inhibitory concentration (MIC) and hemolytic concentration (HC) of the CPEs and OPEs are also measured to compare their antimicrobial activities against two different strains of E. coli with the compounds' toxicity levels against human red blood cells (RBC). MIC and HC measurements are in good agreement with our previous model membrane perturbation study, which reveals that the different membrane perturbation abilities of the CPEs and OPEs are in part responsible for their selectivity towards bacteria compared to mammalian cells. Our study gives insight to several structural features of the PPE-based CPEs and OPEs that modulate their antimicrobial properties and that these features can serve as a basis for further tuning their structures to optimize antimicrobial properties.1. Introduction
Due in part to the misuse of antibiotics, pathogenic bacteria have evolved a number of resistance strategies to attenuate the effect of antibiotics, such as generating novel proteolytic enzymes, altering membrane protein and lipid components, and changing the role of transporters [1]. In recent years, the appearance of “superbugs”, which are resistant to almost all commercial antibiotics, has been a critical worldwide healthcare issue [2,3]. As a result, developing new classes of efficient antimicrobial agents has been the focus of significant research efforts during the last decade [4-9]. Since the discovery of the antimicrobial properties of poly(phenylene ethynylene) (PPE)-based cationic conjugated polyelectrolytes (CPEs) [10], a wide range of structurally similar polymeric CPEs and oligomeric OPEs (oligo-phenylene ethynylenes) have been synthesized [11-16]. These PPE-based CPE and OPE compounds are efficient broad-spectrum bactericides against both Gram-positive and Gram-negative bacteria.
Previously, we have demonstrated that the CPEs and OPEs exhibit remarkable light-activated biocidal activities and moderate killing efficiencies in the dark. The light-induced biocidal activity of these compounds has been attributed to the ability of their excited states to generate corrosive reactive oxygen species after activation with UV-visible light, which can strongly damage biomacromolecules [17,18]. Similar to naturally occurring antimicrobial peptides, one of the main targets of the compounds is believed to be the bacterial cell wall and cytoplasm membrane, so that their dark killing activity is linked to their ability to disrupt bacterial cell walls and membranes and subsequently cause bacterial cell death [19].
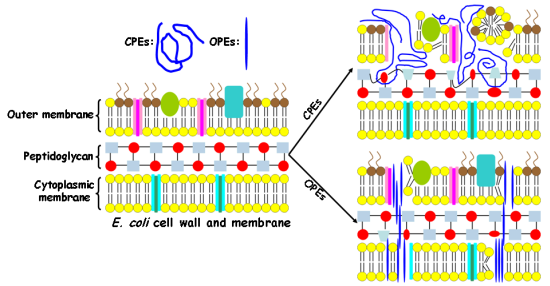
In this study, Gram-negative E. coli cells are used as a model pathogen. The E. coli cell wall contains an outer membrane composed of phospholipids, lipopolysaccharides, and proteins. In addition, a thin layer of crossed-linked peptidoglycan underneath the outer membrane is also a key component of the cell wall. The cytoplasmic membrane of the E. coli cell is composed of a phospholipid bilayer, which serves as a permeability barrier and matrix for ion channels, signaling proteins and other functional molecules. As the lipopolysaccharides and part of the phospholipids from both outer membrane and cytoplasmic membrane are highly negatively-charged, the E. coli cell has an overall negative surface charge [20]. In contrast, for human RBCs, only one cytoplasmic membrane separates the cytoplasm from the outside environment. In particular, the cytoplasmic membrane of the human RBC contains mostly zwitterionic phosphatidylcholine (PC) and 5–10% of negatively charged phosphatidylserine (PS) lipids. Due to the asymmetric distribution of erythrocyte membrane lipids, more than 95% of PS lipids reside on the inner leaflet of the membrane. As a result, the outer leaflet of the mammalian membrane is nearly neutral [20]. We hypothesize that the cationic CPEs and OPEs exert their toxicity by binding to and disrupting the bacterial cell wall and membrane in the dark [19]. To test our hypothesis, we investigate their interactions with intact E. coli cells.
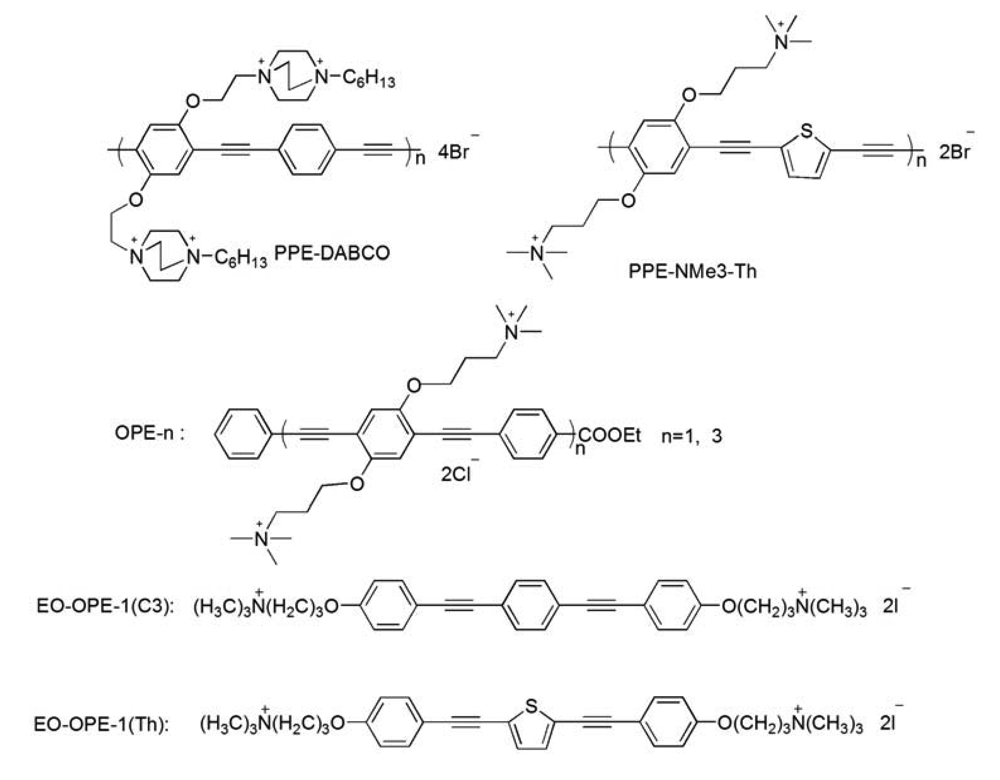
Previously, we have demonstrated that most of the CPEs and OPEs can significantly perturb model bacterial membranes made of total lipids extracted from E. coli cells (ATCC 11303); therefore, the same strain of E. coli cells is used in the current study. In addition, a laboratory strain, E. coli BL21(DE3)pLysS, with resistance to antibiotics carbenicillin and chloramphenicol, is also used to study the influence of antibiotic resistance on the antimicrobial activity of the CPEs and OPEs. As part of our investigation of the structure-function relationship of the CPEs and OPEs, several representative cationic CPEs and OPEs with the same backbones but different numbers of repeat units and/or side chains, are synthesized and used (Scheme 1). In order to compare the CPE or OPE's antimicrobial activity with other antimicrobial agents, the well-known cationic peptide, melittin, is employed herein as a reference. It has been well-documented that melittin exerts its cytotoxicity by interacting with the phospholipid membranes to induce the formation of transmembrane pores and cause the leakage of cytoplasm [21].
We report here a comprehensive investigation on the antibacterial activities of the CPEs and OPEs. In addition, the disruptive effect of the CPEs and OPEs on the morphology of E. coli cells and changes in cytoplasmic membrane permeability caused by OPEs and melittin are reported. The minimum inhibitory concentration (MIC) against E. coli cells and hemolytic concentration (HC) against human RBCs are also determined to compare the relative cytotoxicity of CPEs and OPEs against model bacterial and mammalian cells. In addition to lipids, proteins are also key membrane components for both prokaryotes and eukaryotes, which serve a wide range of functions, including selective ionic permeability, energy transduction, and structural functions [22]. Specifically, there are more than 200 proteins associated with the E. coli cell membrane and approximately 60 of them are believed to be involved with transport [22]. To further investigate the toxicity pathway of the CPEs and OPEs upon their association to the bacterial surface, we also evaluated whether CPEs and OPEs affect protein structure. In this study, we characterized the effect of CPEs and OPEs on the conformation of model proteins bovine serum albumin (BSA), lysozyme and Cytochrome C using circular dichroism. The results of this study provide insights into the CPEs and OPEs' antimicrobial mechanisms and will enable the design of more effective PPE-based antimicrobial agents.
2. Experimental Methods
Materials
The antimicrobial compounds (Scheme 1) used in this paper were synthesized as reported [12-14,16]. Melittin from honey bee venom (Gly-Ile-Gly-Ala-Val-Leu-Lys-Val-Leu-Thr-Thr-Gly-Leu-Pro-Ala-Leu-Ile-Ser-Trp-Ile-Lys-Arg-Lys-Arg-Gln-Gln-NH2, ∼70% purity) was obtained from Sigma-Aldrich (St. Louis, MO). Luria broth and Agar were purchased from BD Biosciences (Franklin Lakes, NJ). 3,5-Dipropylthiacarbocyanine (diSC3-5) was obtained from Molecular Probes (Eugene, OR). All other chemicals and antibiotics were purchased from Sigma-Aldrich. Fresh human whole blood was purchased from Bioreclamation (Westbury, NY); sodium citrate was used as anticoagulant. E. coli strain ATCC 11303 was obtained from American Type Culture Collection (ATCC, Manassas, VA) and E. coli strain BL21(DE3)pLysS was a stock from Dr. Graves' lab at the University of New Mexico. Ultrapure water was used throughout the study (Milli-Q, 18.2 MΩ cm−1 resistivity).
Bacterial Growth Conditions
E. coli (ATCC 11303) was grown in a standard Luria broth and E. coli BL21(DE3)pLysS was grown in the Luria broth with carbenicillin and chloramphenicol at concentrations of 50 and 34 μg/mL, respectively. A fresh E. coli culture was inoculated from an overnight culture followed by approximately three hours of incubation at 37 °C to the exponential growth phase (O.D.600∼0.5). At this growth phase, the E. coli cells were collected by centrifugation and washed twice with 10 mM PBS (138 mM NaCl and 2.7 mM KCl at pH 7.4). The cell pellet was resuspended with PBS buffer to O.D.600∼0.5.
Destruction of Cell Walls and Membranes
2 mL of E. coli suspension (∼108 colony forming units (CFU)/mL) was mixed with 10 μg/mL CPEs, OPEs or melittin followed by incubation at 37 °C for one hour in the dark. The mixture of E. coli cells and antimicrobial compound was centrifuged at 10,000 rpm for 10 minutes. The supernatant was decanted and its absorbance at 260 nm was measured (SpectroMax M2e microplate reader, Molecular Devices, Sunnyvale, CA). The cell pellets were resuspended with 2% glutaraldehyde and incubated at 4 °C for three hours to preserve the interface between E. coli cells and antimicrobial compounds, followed by washing with PBS buffer. Then, the fixed cells were dehydrated by sequential treatment with increasing concentrations of ethanol for 15 min and dehydrated with absolute ethanol twice. The dehydrated samples were dried at room temperature and transferred onto a piece of clean silicon wafer. The dried samples were sputtercoated with 10 nm thick gold/palladium. Morphologies of the E. coli cells were observed by SEM (Quanta 3D, Dual beam FEGSEM/FIB, FEI, Hillsboro, OR).
Cytoplasmic Membrane Permeability Assay
The effect of OPEs and melittin interaction with E. coli cytoplasmic membrane is investigated by a modified method [23]. Since the CPEs can strongly influence the fluorescence of the membrane potential-sensitive cyanine dye (data not shown), the effect of CPEs on membrane permeability was not determined. Thus, results of this assay are not conclusive. Experimental details, results, and discussion are presented in Supporting Information.
Circular Dichroism
An aliquot of a CPE or OPE was added to 3 mL of 0.1 mg/mL protein solution in 10 mM phosphate buffer (2 mM NaH2PO4 and 8 mM Na2HPO4 at pH 7.4) and incubated at 37 °C for one hour in the dark. The final concentration of CPE or OPE was 10 μg/mL. Circular dichroism spectra from 200 to 500 nm were recorded on an Aviv CD spectrometer (Model 420, Aviv Biomedical Inc.) in quartz cuvettes at room temperature.
Minimum Inhibitory Concentration (MIC) Determination
MIC values were determined by a modified method from literature [24,25]. The E. coli cells were diluted with a minimal medium (28 mM glucose, 42 mM Na2HPO4, 22 mM KH2PO4, 18.7 mM NH4Cl, 8.5 mM NaCl, 1 mM MgSO4, and 0.09 mM CaCl2 at pH 7.2. 50 μg/mL carbenicillin and 34 μg/mL chloramphenicol were added to the medium for E. coli BL21(DE3)pLysS) to ∼105 CFU/mL. The diluted cell solutions were then incubated with twofold serial dilutions of the antimicrobial compounds in a 96-well plate at 37 °C overnight. O.D.600 was obtained on a microplate reader (SpectroMax M2e, Molecular Devices, Sunnyvale, CA) to monitor cell growth. The MIC values (MIC90) reported herein are the minimum concentrations needed to inhibit 90% of the cell growth. Positive controls without antimicrobial compounds and negative controls without bacteria were also measured. The reported values are the averages of duplicate measurements.
Hemolysis Assay
The hemolytic activities of CPEs, OPEs and melittin were determined by the release of hemoglobin from human RBCs when incubated with the antimicrobial compounds [25,26]. Fresh human RBCs were obtained by centrifuging human whole blood at 2,000g for 5 min and washed with Tris buffer (10 mM Tris and 150 mM NaCl at pH 7.2). A RBC stock solution was made by a 200-fold dilution of the RBC suspension (0.5% red blood cell) with the Tris buffer. The RBC stock solutions along with various amounts of the antimicrobial compounds were incubated at 37 °C for 1 hour in microcentrifugation tubes. Then, the mixtures were centrifuged at 3500g for 10 minutes. 100 μL aliquots of the supernatant were transferred to a 96-well plate and mixed with 100 μL of Tris buffer. The hemolytic concentrations (HC50, concentrations of antimicrobial compounds that caused 50% cell hemolysis) were determined by measuring the absorbance of hemoglobins at 540 nm using a microplate reader (SpectroMax M5, Molecular Devices, Sunnyvale, CA). A positive control of cells incubated with 1% Triton-X100 was also prepared. A negative control of the RBC solution without antimicrobial compounds was also prepared. The reported values were the averages of duplicate measurements.
3. Results and Discussion
Previously, we have shown that the PPE-based cationic conjugated polymers (CPEs) and oligomers (OPEs) with different side chains and repeat units exhibit a range of toxicities against bacteria in the dark and different perturbation abilities against model membranes [14,19,27,28]. Generally, the amphipathic properties, molecular size, aggregation state, and charge density are factors modulating the interactions between CPEs and OPEs and their biological targets. The motivation of the current study is to elucidate the mechanistic origin of the different dark killing abilities among different CPEs and OPEs. SEM imaging and the cytoplasmic membrane permeability assays are used to elucidate the interactions between a CPE or an OPE with bacterial cell walls and membranes. The MIC and HC values serve as useful parameters to evaluate the selectivity the compound, i.e., antimicrobial activity against bacteria compared to cytotoxicity against mammalian cells.
CPEs and OPEs Can Disrupt Bacterial Cell Walls and Membranes
The addition of different CPE and OPE compounds caused different visual changes to ATCC 11303 E. coli cells. As shown in Figure 1, the addition of PPE-DABCO, PPE-Th and OPE-3 caused the E. coli cells to aggregate and precipitate (Figure 1(D,G,H)). No visible changes occurred with the addition of OPE-1 or melittin (Figure 1(B,C)). On the other hand, after the addition of two EO-OPE-1 compounds, the turbidity of the the E. coli cell suspension decreased (Figure 1(E,F)), implying that the EO-OPE-1s may have caused cell lysis. The same effects of the CPEs and OPEs on BL21(DE3)pLysS E. coli cells were observed (data not shown).
The effect of the different CPEs and OPEs on E. coli cell viability was determined by counting the colony forming units of E. coli cells after exposure to 10 μg/mL for one hour in the dark. Results are presented in Figures 2, S1 and S2. As shown, OPE-1 and melittin did not exert significant toxicity. In contrast, all other CPEs and OPEs show significant antimicrobial activities against the E. coli cells For example, the oligomer EO-OPE-1(C3) induced close to 100% cell death in both E. coli strains (Figure 2(B)).
Previously, we have shown that the dark biocidal activity of the CPE and OPE is associated with damages to bacterial cell walls and membranes by the direct contact between these compounds and bacteria. When the cell walls and membranes are perturbed, cytoplasm contents, such as nucleic acids and proteins can leak out and these materials can be detected by measuring the absorbance of the soluble fraction of the cell suspension at 260 nm [29]. Table 1 summarizes absorbance measurements of the supernatant of E. coli cells after incubating with the antimicrobial compounds. As shown, EO-OPE-1(C3) causes the highest level of leakage from both E. coli strains, which correlates with its bacteriolysis effect (Figure 1(E)) and high toxicity (Figure 2(A,B)). However, a direct correlation between toxicities and level of soluble cell content caused by the CPEs and OPEs cannot be drawn at this time primarily because the interaction between the antimicrobial compounds and E. coli cell content (i.e., DNA and proteins) is not fully understood. For example, polyvalent cations can readily precipitate DNA [30]. Therefore, low absorbance readings do not necessarily correlate with low levels of cell content leakage. Nonetheless, some meaningful comparisons can be made from the absorbance measurements. Figure 2, and results from our previous study, show that OPE-3 exhibits much higher dark antimicrobial activity than OPE-1 [28]. OPE-3 is also known to interact much more strongly with DNA compared to OPE-1 [31]. Therefore, DNA released from the E. coli cells may form stable complexes with OPE-3 that can be pelleted during centrifugation, resulting in a lower absorbance at 260 nm in the cell supernatant compared to OPE-1 (Table 1).
CPEs and OPEs Can Induce Changes to Bacterial Cell Morphology
In order to further elucidate the CPEs and OPEs' effects on bacterial cells, the morphological changes of E. coli cells with the addition of the different antimicrobial agents were examined by SEM imaging. As shown in Figures 3(A) and 4(A), the two strains of E. coli cells alone in PBS buffer maintain their integrity with a smooth cell surface. E. coli cells treated with melittin are still able to maintain the intact cell structures, but some cells now appear more rough and wrinkled (Figures 3(B) and 4(B), see arrows). E. coli cells treated with the polymers PPE-DABCO and PPE-Th exhibit obvious morphological changes compared to the untreated samples (Figures 3(C,D), 4(C,D)). The surfaces of polymer treated cells appear more rough, with possible formation of circular blebs (Figure 4(C), see arrows), and the cells appear to be agglomerated.
The addition of oligomeric EO-OPE-1(Th) and EO-OPE-1(C3) also caused changes to cell morphology. Most E. coli BL21(DE3)pLysS cells exposed to EO-OPE-1(C3) became completely disrupted, appearing as amorphous material rather than cells (Figure 4(E)). E. coli (ATCC 11303) cells exposed to EO-OPE-1(Th) showed roughening of the cell surface (Figure 3(D)) while E. coli BL21(DE3)pLysS cells, mostly maintained their integrity with a smooth cell surface (Figure 4(F)).
It is important to note that the molecular size of the antimicrobial compounds is one of the determining factors in their interactions with bacteria [32]. The relatively large sizes of the polymeric CPEs hinder their ability to penetrate into the cell wall and membrane, and as a result, they may only cause damages to the cell surfaces and cause cell aggregation. On the other hand, the smaller and unique linear structures of the oligomeric EO-OPE-1(C3) and EO-OPE-1(Th) compounds enable them to easily penetrate cell walls and membranes without at first causing serious morphological changes to the cell surface. These oligomers may then exert their cytotoxicity by inducing small membrane defects and inhibiting metabolic pathways. This proposed toxicity mechanism of the linear oligomers is supported by our observation that the addition of the two compounds significantly decreases the optical density of E. coli cell suspensions (Figure 1(E,F)). Disintegration of bacterial cells is likely caused by the insertion of the linear oligomers into the cell walls and membranes and subsequent disruption of these structures. The more linear of the two oligomers, EO-OPE-1(C3), showed the highest cell lysis activity, resulting in a large amount of cell debris that was both detected by absorbance measurements (Table 1) and visualized by SEM (Figure 4(E)). Due to the strong lytic activity of EO-OPE-1(C3) against E. coli (ATCC 11303), no sample can be collected or visualized with SEM imaging. In addition, it is worth mentioning that because of the resolution limit of the SEM instrument, neither individual antimicrobial molecules nor their aggregates could be clearly visualized.
The ability of the OPEs and melittin to depolarize the cytoplasmic membrane is determined by using the cationic membrane potential-sensitive cyanine dye diSC3-5. As found by Hancock et al., there is no correlation between the membrane permeability ability of antimicrobial compounds and their toxicities against bacteria [23]. Even though the results of this assay are not conclusive, it is clear that the four antimicrobial compounds (mellitin and three oligomers) used in this assay can interact with bacterial cytoplasmic membrane, although the extent of this interaction varies (see Supporting Information for detailed information).
CPEs Can Denature Native Protein Conformation
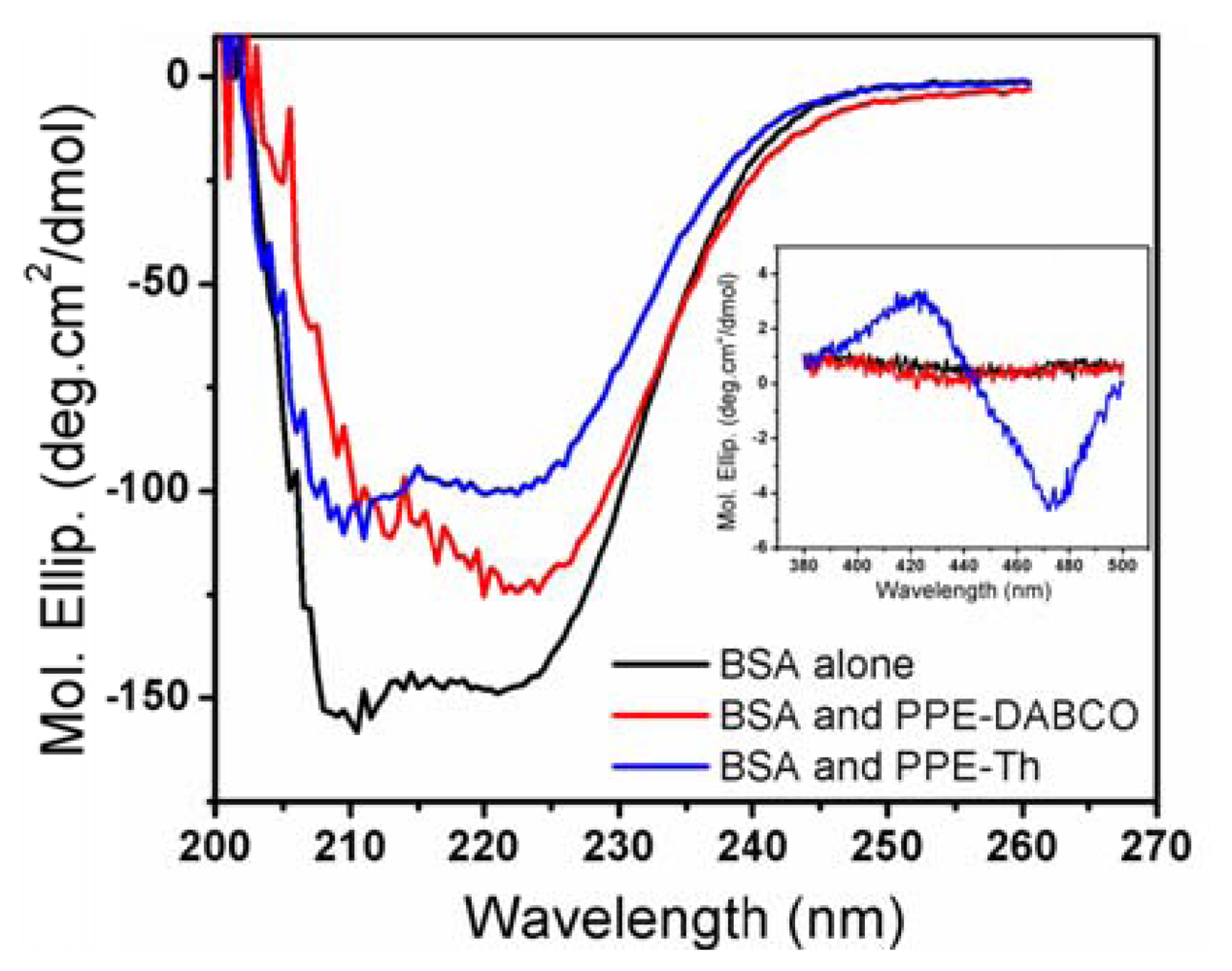
Since proteins are a key component of biological membranes [22], studying their interactions with CPEs and OPEs will provide further insight into CPEs and OPEs' toxicity mechanisms. In this study, the effect of CPEs and OPEs on the secondary structure of three model proteins, BSA, lysozyme, and cytochrome C were evaluated. Lysozyme and cytochrome C are well-folded small globular proteins that have been extensively studied. BSA possesses a high degree of homology with human serum albumin (HAS) [33,34]. Serum albumins are abundant in the mammalian circulatory system and carry out various important physiological functions [35]. More importantly, BSA is a more hydrophobic protein with high surface activity compared to the highly soluble and charged lysozyme and cytochrome C, and should serve as a better model for membrane proteins. The physicochemical properties of these model proteins are summarized in Table 2 [34,36-38]. The presence of CPEs and OPEs did not induce any changes in the CD spectra of lysozyme or cytochrome C (data not shown), indicating that no conformational changes to the native protein structures were induced. This is probably due to the electrostatic repulsion between the cationic CPEs and OPEs with the two positively charged proteins (see Table 2) that prevented their association. In contrast, some degree of structural perturbation was observed for BSA incubated with CPEs and OPEs in the dark (Figures 5 and S6). In particular, the addition of PPE-Th caused the most significant loss to the protein's secondary structures (200–260 nm) (Figure 5). Furthermore, BSA/PPE-Th mixture is also uniquely CD active in 400–500 nm region, where PPE-Th absorbs (inset of Figure 5). By itself, the PPE-Th polymer is not CD active in this region (data not shown). PPE-Th is known to be highly lipophilic and this property may be responsible for its ability to denature the relatively hydrophobic BSA [27]. Furthermore, complexing PPE-Th with BSA render the polymer CD-active, indicating that distinct structures are formed. PPE-DABCO also induces conformational change in BSA, albeit to a lesser extent compared to PPE-Th. In contrast, only small conformational changes are observed when BSA is incubated with the oligomers (Figure S6). Although none of the model proteins used in this study is a bacterial membrane protein, the result can provide some insights of to the interaction between our antimicrobial compounds and proteins. When a bacterial cell becomes associated with PPE-Th, one mode of its toxic pathway may be the denaturation of membrane proteins.
CPEs and OPEs Selectively Exert Toxicity Towards Bacterial Cells
We tested the inhibitory activities of CPEs and OPEs against E. coli cells in the dark. As shown in Table 3, the antimicrobial compounds exhibit very similar inhibitory effect on the two different strains of E. coli cells, implying that the physiological differences between these E. coli strains have limited influence on their susceptibilities towards CPEs, OPEs and melittin. Therefore, it is reasonable to conclude that the specific resistance strategies that the E. coli BL21(DE3)pLysS acquired against carbenicillin and chloramphenicol antibiotics are not effective against our novel antimicrobial agents. In particular, OPE-3 and PPE-Th exhibit excellent inhibitory activities against E. coli cells (Table 3). At relatively low concentrations, the two EO-OPE-1 compounds, PPE-DABCO, and melittin show efficient inhibitory activity against the E. coli cells. However, no inhibitory activity is observed for OPE-1 within the tested concentration range.
In order to evaluate the biocidal selectivities of the CPEs and OPEs, we tested their hemolytic activity against human RBCs. The concentrations necessary to cause 50% RBC hemolysis (HC50) of OPE-1 and OPE-3 are fairly high (Table 3). Thus, no significant hemolytic activity is observed for OPE-1 and OPE-3 within the tested concentration range. Under the same conditions, the two EO-OPE-1 compounds exhibited moderate hemolytic ability against RBC with HC50 values around 20 μg/mL. However, significant blood cell lysis was observed for polymer PPE-DABCO and peptide melittin at relatively low concentrations with HC50 values of around 5 μg/mL. We were unable to determine the HC50 value for PPE-Th using this method, probably due to the strong interaction of PPE-Th with hemoglobin that precipitated rather than lysed RBCs. A different method to measure hemolysis, based on Coulter Counter (Beckman Coulter, Miami, FL) measurements, was used to determine HC50 of PPE-Th and the value is about 1 μg/mL (data not shown).
Overall, the oligomer OPE-3 exhibited the highest selectivity towards bacterial cells while the polymeric PPE-DABCO and PPE-Th and antimicrobial peptide melittin showed poor biocidial selectivity. The latter three compounds showed efficient biocidal as well as hemolytic activities.
Dark Killing Mechanisms of CPEs and OPEs
Results from this study, combined with our previous work [19], reveal some mechanistic insights to the different biocidal efficiencies and selectivities of the CPE and OPE compounds. The following are three important modulating factors for the observed biocidal and hemolytic activities of the tested compounds. First, the molecular size, shape, and aggregation state determine whether the compounds can penetrate the bacterial outer membrane and reach the cytoplasmic membrane. This factor explains the toxicities and cell lytic activity of the CPEs and OPEs. In general, the oligomeric OPEs and oligo-peptide melitin appear to be small enough to penetrate the bacterial outer membrane once they bind to cell surfaces due to attractive electrostatic interactions. However, the penetrating ability of the polymers is compromised due to their large sizes [39]. Second, a compound's ability to perburb bacterial and mammalian cytoplasmic membranes determines its biocidal selectivity. Because OPE-3′s perturbation ability against model bacterial membrane is rather effective compared to model mammalian membrane (made of cholesterol and 1,2-dioleoyl-sn-glycero-3-phosphocholine) [19], OPE-3 possesses high biocidal selectivity. In contrast to OPE-3, PPE-DABCO perturbs both model bacterial and mammalian membranes [19], leading to a poor biocidal selectivity. Even though the two EO-OPE-1 compounds and OPE-3 can cause similar levels of damage to a model bacterial membrane [19], the high perturbation ability against model mammalian membranes endows the two EO-OPE-1 compounds relative high hemolytic activities. Third, the ability of a compound to interact and denature membrane proteins provides another pathway for toxicity. The ability of CPEs to complex and denature the native protein conformation of BSA gives us an explanation for its high antimicrobial activity when the compound does not exhibit significant lytic or membrane perturbation abilities. For example, the high inhibitory ability of PPE-Th against E. coli cells is believed to derive from its high lipophilicity property to efficiently damage the bacterial cell wall and membrane, including membrane proteins [27]. In addition, since the exponential growth phase E. coli cells, which are undergoing fast propagation, was employed in the antimicrobial investigations, other antimicrobial and inhibitory mechanisms may be involved, such as interference with bacterial metabolic pathways [40].
4. Conclusions
In this study, we selected several cationic CPEs and OPEs with the same backbones but different numbers of repeat units or side chains to investigate the relationships among antimicrobial activity, hemolytic activity, and molecular structure. Some insights into the origin of the different dark killing abilities among the CPEs/OPEs emerged. It is shown that the dark antimicrobial activity and hemolytic activity of these compounds correlates with their perturbation ability against bacterial and mammalian cytoplasm membranes. Molecular size, charge density and amphipathicity/hydrophobicity can influence the activities of these compounds. Linear oligomers exert their toxicities directly on disrupting bacterial cytoplasmic membrane but bulky polymers exert their toxicities bacteria through the destruction of outer membrane. Charge density is important for the initial binding step between antimicrobial compounds and bacteria. The effect of amphipathicity or hydrophobicity is not yet fully understood. However, it is clear that the spatial distribution of polar and nonpolar groups within a molecule strongly influences its interaction with phospholipid bilayer and protein [41]. Further experiments elucidating the molecular mechanisms of the CPEs and OPEs' membrane perturbation abilities and their interactions with lipopolysaccharides and peptidoglycans are currently underway.
Supplementary Material
polymers-03-01199-s001.pdf



| Antimicrobial Agents | Antimicrobial compounds alone | E. coli BL21(DE3)pLysS | E. coli (ATCC 11303) |
|---|---|---|---|
| Control | 0.06 * | 0.20 ** | 0.17 ** |
| Melittin | 0.06 | 0.63 | 0.46 |
| OPE-1 | 0.15 | 0.33 | 0.31 |
| OPE-3 | 0.14 | 0.20 | 0.19 |
| EO-OPE-1(C3) | 0.16 | 1.30 | 0.88 |
| EO-OPE-1(Th) | 0.19 | 0.26 | 0.31 |
| PPE-DABCO | 0.13 | 0.80 | 0.79 |
| PPE-Th | 0.17 | 0.20 | 0.22 |
*PBS buffer alone;**E. coli cells alone in the PBS buffer.
| Protein | BSA | Lysozyme | Cytochrome C |
|---|---|---|---|
| Isoelectric Point | 4.8 | 10.6 | 10∼10.5 |
| Net charge* | − | + | + |
| Molecular Weight (KDa) | 66 | 14 | 12 |
| Main Secondary Structure | α-helical | α-helical | α-helical and unordered structure |
*Net charge on protein under current experimental conditions.
| Antimicrobial Agents | MIC90 (μg/mL) | HC50 (μg/mL) | |
|---|---|---|---|
| BL21(DE3)pLysS | ATCC 11303 | ||
| OPE-1 | >30 | >30 | >100 |
| OPE-3 | 0.5 | 0.5 | >50 |
| EO-OPE-1(C3) | 2 | 2 | 24 |
| EO-OPE-1(Th) | 2 | 1 | 16 |
| PPE-DABCO | 2 | 2 | 4 |
| PPE-Th | 0.3 | 0.5 | N/D * |
| Melittin (70% purity) | 6 | 4 | 5 |
*unable to make measurement.
Acknowledgments
This research is financially supported by the Defense Threat Reduction Agency (Contract No. W911NF07-1-0079). The authors gratefully acknowledge help from Steven W. Graves of Department of Chemical and Nuclear Engineering at the University of New Mexico on the biocidal assays. We thank Yingbing Jiang from the Department of Earth & Planetary Science at the University of New Mexico for assistance with SEM imaging.
Reference
- Brogden, K.A. Antimicrobial Peptides: Pore Formers or Metabolic Inhibitors in Bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar]
- Klein, E.; Laxminarayan, R.; Smith, D.L.; Gilligan, C.A. Economic Incentives and Mathematical Models of Disease. Environ. Dev. Econ. 2007, 12, 707–732. [Google Scholar]
- Bryers, J.D. Medical Biofilms. Biotechnol. Bioeng. 2008, 100, 1–18. [Google Scholar]
- Zasloff, M. Antimicrobial Peptides of Multicellular Organisms. Nature 2002, 415, 389–395. [Google Scholar]
- Tew, G.N.; Scott, R.W.; Klein, M.L.; Degrado, W.F. De Novo Design of Antimicrobial Polymers, Foldamers, and Small Molecules: From Discovery to Practical Applications. Account. Chem. Res. 2010, 43, 30–39. [Google Scholar]
- Majumdar, P.; Lee, E.; Gubbins, N.; Stafslien, S.J.; Daniels, J.; Thorson, C.J.; Chisholm, B.J. Synthesis and Antimicrobial Activity of Quaternary Ammonium-Functionalized POSS (Q-POSS) and Polysiloxane Coatings Containing Q-POSS. Polymer 2009, 50, 1124–1133. [Google Scholar]
- Hancock, R.E.W.; Lehrer, R. Cationic Peptides: A New Source of Antibiotics. Trends Biotechnol. 1998, 16, 82–88. [Google Scholar]
- Xing, C.F.; Xu, Q.L.; Tang, H.W.; Liu, L.B.; Wang, S. Conjugated Polymer/Porphyrin Complexes for Efficient Energy Transfer and Improving Light-Activated Antibacterial Activity. J. Amer. Chem. Soc. 2009, 131, 13117–13124. [Google Scholar]
- Liu, S.; Sun, G. Functional Modification of Poly(ethylene terephthalate) with an Allyl Monomer: Chemistry and Structure Characterization. Polymer 2008, 49, 5225–5232. [Google Scholar]
- Lu, L.; Rininsl, F.H.; Wittenburg, S.K.; Achyuthan, K.E.; McBranch, D.W.; Whitten, D.G. Biocidal Activity of A Light-Absorbing Fluorescent Conjugated Polyelectrolyte. Langmuir 2005, 21, 10154–10159. [Google Scholar]
- Chemburu, S.; Corbitt, T.S.; Ista, L.K.; Ji, E.; Fulghum, J.; Lopez, G.P.; Ogawa, K.; Schanze, K.S.; Whitten, D.G. Light-Induced Biocidal Action of Conjugated Polyelectrolytes Supported on Colloids. Langmuir 2008, 24, 11053–11062. [Google Scholar]
- Tang, Y.L.; Zhou, Z.J.; Ogawa, K.; Lopez, G.P.; Schanze, K.S.; Whitten, D.G. Synthesis, Self-Assembly, and Photophysical Behavior of Oligo Phenylene Ethynylenes: From Molecular to Supramolecular Properties. Langmuir 2009, 25, 21–25. [Google Scholar]
- Tang, Y.L.; Hill, E.H.; Zhou, Z.J.; Evans, D.G.; Schanze, K.S.; Whitten, D.G. Synthesis, Self-Assembly, and Photophysical Properties of Cationic Oligo(p-phenyleneethynylene)s. Langmuir 2011, 27, 4945–4955. [Google Scholar]
- Zhou, Z.J.; Corbitt, T.S.; Parthasarathy, A.; Tang, Y.L.; Ista, L.F.; Schanze, K.S.; Whitten, D.G. “End-Only” Functionalized Oligo(phenylene ethynylene)s: Synthesis, Photophysical and Biocidal Activity. J. Phys. Chem. Lett. 2010, 1, 3207–3212. [Google Scholar]
- Corbitt, T.S.; Sommer, J.R.; Chemburu, S.; Ogawa, K.; Ista, L.K.; Lopez, G.P.; Whitten, D.G.; Schanze, K.S. Conjugated Polyelectrolyte Capsules: Light-Activated Antimicrobial Micro “Roach Motels”. ACS Appl. Mater. Interfaces 2009, 1, 48–52. [Google Scholar]
- Zhao, X.Y.; Pinto, M.R.; Hardison, L.M.; Mwaura, J.; Muller, J.; Jiang, H.; Witker, D.; Kleiman, V.D.; Reynolds, J.R.; Schanze, K.S. Variable Band Gap Poly(arylene ethynylene) Conjugated Polyelectrolytes. Macromolecules 2006, 39, 6355–6366. [Google Scholar]
- Imlay, J.A. Pathways of Oxidative Damage. Ann. Rev. Microbiol. 2003, 57, 395–418. [Google Scholar]
- Zheng, J.; Bizzozero, O.A. Traditional Reactive Carbonyl Scavengers Do Not Prevent the Carbonylation of Brain Proteins Induced by Acute Glutathione Depletion. Free Radical Res. 2010, 44, 258–266. [Google Scholar]
- Wang, Y.; Tang, Y.L.; Zhou, Z.J.; Ji, E.; Lopez, G.P.; Chi, E.Y.; Schanze, K.S.; Whitten, D.G. Membrane Perturbation Activity of Cationic Phenylene Ethynylene Oligomers and Polymers: Selectivity against Model Bacterial and Mammalian Membranes. Langmuir 2010, 26, 12509–12514. [Google Scholar]
- Som, A.; Tew, G.N. Influence of Lipid Composition on Membrane Activity of Antimicrobial Phenylene Ethynylene Oligomers. J. Phys. Chem. B 2008, 112, 3495–3502. [Google Scholar]
- Yang, L.; Harroun, T.A.; Weiss, T.M.; Ding, L.; Huang, H.W. Barrel-Stave Model or Toroidal Model? A Case Study on Melittin Pores. Biophys. J. 2001, 81, 1475–1485. [Google Scholar]
- Graham, J.M.; Higgins, J.A. Membrane Analysis; Springer: New York, NY, USA, 1997. [Google Scholar]
- Wu, M.H.; Maier, E.; Benz, R.; Hancock, R.E.W. Mechanism of Interaction of Different Classes of Cationic Antimicrobial Peptides with Planar Bilayers and with the Cytoplasmic Membrane of Escherichia coli. Biochemistry 1999, 38, 7235–7242. [Google Scholar]
- Rennie, J.; Arnt, L.; Tang, H.Z.; Nusslein, K.; Tew, G.N. Simple Oligomers as Antimicrobial Peptide Mimics. J. Ind. Microbiol. Biotechnol. 2005, 32, 296–300. [Google Scholar]
- Zhou, C.; Qi, X.; Li, P.; Chen, W.N.; Mouad, L.; Chang, M.W.; Leong, S.S.; Chan-Park, M.B. High Potency and Broad-Spectrum Antimicrobial Peptides Synthesized via Ring-Opening Polymerization of Alpha-aminoacid-N-carboxyanhydrides. Biomacromolecules 2010, 11, 60–67. [Google Scholar]
- Yang, L.H.; Gordon, V.D.; Mishra, A.; Sorn, A.; Purdy, K.R.; Davis, M.A.; Tew, G.N.; Wong, G.C.L. Synthetic Antimicrobial, Oligomers Induce A Composition-Dependent Topological Transition in Membranes. J. Amer. Chem. Soc. 2007, 129, 12141–12147. [Google Scholar]
- Corbitt, T.S.; Ding, L.P.; Ji, E.Y.; Ista, L.K.; Ogawa, K.; Lopez, G.P.; Schanze, K.S.; Whitten, D.G. Light and Dark Biocidal Activity of Cationic Poly(arylene ethynylene) Conjugated Polyelectrolytes. Photochem. Photobiol. Sci. 2009, 8, 998–1005. [Google Scholar]
- Tang, Y.L.; Corbitt, S.C.; Parthasarathy, A.; Zhou, Z.J.; Schanze, K.S.; Whitten, D.G. Light-Induced Antibacterial Activity of Symmetrical and Asymmetrical Oligophenylene Ethynylenes. Langmuir 2011, 27, 4856–4962. [Google Scholar]
- Broxton, P.; Woodcock, P.M.; Gilbert, P. A Study of the Anti-Bacterial Activity of Some Polyhexamethylene Biguanides Towards Escherichia-Coli Atcc-8739. J. Appl. Bact. 1983, 54, 345–353. [Google Scholar]
- Toma, A.C.; de Frutos, M.; Livolant, F.; Raspaud, E. DNA Condensed by Protamine: A “Short” or “Long” Polycation Behavior. Biomacromolecules 2009, 10, 2129–2134. [Google Scholar]
- Tang, Y.L.; Achyuthan, K.E.; Whitten, D.G. Label-Free and Real-Time Sequence Specific DNA Detection Based on Supramolecular Self-Assembly. Langmuir 2010, 26, 6832–6837. [Google Scholar]
- Lee, H.; Larson, R.G. Lipid Bilayer Curvature and Pore Formation Induced by Charged Linear Polymers and Dendrimers: The Effect of Molecular Shape. J. Phys. Chem. B 2008, 112, 12279–12285. [Google Scholar]
- He, X.M.; Carter, D.C. Atomic-Structure and Chemistry of Human Serum-Albumin. Nature 1992, 358, 209–215. [Google Scholar]
- Charbonneau, D.M.; Tajmir-Riahi, H.A. Study on the Interaction of Cationic Lipids with Bovine Serum Albumin. J. Phys. Chem. B 2010, 114, 1148–1155. [Google Scholar]
- Peters, T. All about Albumin. Biochemistry, Genetics and Medical Applications; Academic Press: San Diego, CA, USA, 1996. [Google Scholar]
- Razumovsky, L.; Damodaran, S. Surface Activity-Compressibility Relationship of Proteins at the Air-Water Interface. Langmuir 1999, 15, 1392–1399. [Google Scholar]
- White, F.H., Jr. Studies on Secondary Structure in Chicken Egg-White Lysozyme after Reductive Cleavage of Disulfide Bonds. Biochemistry 1976, 15, 2906–2912. [Google Scholar]
- Aravind, U.K.; Mathew, J.; Aravindakumar, C.T. Transport Studies of BSA, Lysozyme and Ovalbumin through Chitosan/Polystyrene Sulfonate Multilayer Membrane. J. Membr. Sci. 2007, 299, 146–155. [Google Scholar]
- Lienkamp, K.; Kumar, K.N.; Som, A.; Nusslein, K.; Tew, G.N. “Doubly Selective” Antimicrobial Polymers: How Do They Differentiate between Bacteria? Chem.-Eur. J. 2009, 15, 11710–11714. [Google Scholar]
- Boman, H.G.; Agerberth, B.; Boman, A. Mechanisms of Action on Escherichia coli of Cecropin P1 and PR-39, Two Antibacterial Peptides from Pig Intestine. Infect. Immun. 1993, 61, 2978–2984. [Google Scholar]
- Wimley, W.C. Describing the Mechanism of Antimicrobial Peptide Action with the Interfacial Activity Model. ACS Chem. Biol. 2010, 5, 905–917. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Y.; Zhou, Z.; Zhu, J.; Tang, Y.; Canady, T.D.; Chi, E.Y.; Schanze, K.S.; Whitten, D.G. Dark Antimicrobial Mechanisms of Cationic Phenylene Ethynylene Polymers and Oligomers against Escherichia coli. Polymers 2011, 3, 1199-1214. https://doi.org/10.3390/polym3031199
Wang Y, Zhou Z, Zhu J, Tang Y, Canady TD, Chi EY, Schanze KS, Whitten DG. Dark Antimicrobial Mechanisms of Cationic Phenylene Ethynylene Polymers and Oligomers against Escherichia coli. Polymers. 2011; 3(3):1199-1214. https://doi.org/10.3390/polym3031199
Chicago/Turabian StyleWang, Ying, Zhijun Zhou, Jingshu Zhu, Yanli Tang, Taylor D. Canady, Eva Y. Chi, Kirk S. Schanze, and David G. Whitten. 2011. "Dark Antimicrobial Mechanisms of Cationic Phenylene Ethynylene Polymers and Oligomers against Escherichia coli" Polymers 3, no. 3: 1199-1214. https://doi.org/10.3390/polym3031199
APA StyleWang, Y., Zhou, Z., Zhu, J., Tang, Y., Canady, T. D., Chi, E. Y., Schanze, K. S., & Whitten, D. G. (2011). Dark Antimicrobial Mechanisms of Cationic Phenylene Ethynylene Polymers and Oligomers against Escherichia coli. Polymers, 3(3), 1199-1214. https://doi.org/10.3390/polym3031199