Walls That Grow: Fungal Species-Driven Performance of Mycelium-Based Composites Grown on Rice-Husk Waste
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Species and Cultures
2.2. Substrate Preparation and Inoculation
2.3. Material Fabrication
2.4. Sample Preparation
2.5. Morphological and Microstructural Analysis
2.6. Chemical Characterisation
2.7. Thermal Degradation Analysis
2.8. Physical and Mechanical Analysis
2.8.1. Tensile Strength
2.8.2. Compressive Strength
2.8.3. Flexural Strength
2.8.4. Density
2.9. Wettability Performance
2.10. Statistical Analysis
3. Results and Discussion
3.1. Morphological and Structural Analysis
3.2. Chemical Characterisation
3.3. Thermal Degradation Analysis
3.4. Physical and Mechanical Analysis
3.4.1. Tensile Strength
3.4.2. Compressive Strength
3.4.3. Flexural Strength
3.4.4. Density
3.5. Wettability Performance
3.6. Statistical Comparison of Fabricated MBCs
3.6.1. Flexural Strength
3.6.2. Tensile Strength
3.6.3. Compressive Strength
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| EPS | Expanded Polystyrene |
| FTIR | Fourier Transform Infrared |
| GWP | Global Warming Potential |
| HSD | Honestly Significant Difference |
| MBC | Mycelium-Based Composite |
| MOE | Modulus of Elasticity |
| MOR | Modulus of Rupture |
| PDA | Potato Dextrose Agar |
| SEM | Scanning Electron Microscopy |
| TGA | Thermogravimetric Analysis |
| WCA | Water Contact Angle |
References
- Ajayi, S.O.; Oyedele, L.O.; Akinade, O.O.; Bilal, M.; Owolabi, H.A.; Alaka, H.A.; Kadiri, K.O. Reducing waste to landfill: A need for cultural change in the UK construction industry. J. Build. Eng. 2016, 5, 185–193. [Google Scholar] [CrossRef]
- United Nations Environment Programme; Global Alliance for Buildings and Construction. Not just another brick in the wall: The solutions exist—Scaling them will build on progress and cut emissions fast. In Global Status Report for Buildings and Construction 2024/2025; United Nations Environment Programme: Nairobi, Kenya, 2025. [Google Scholar]
- Ghazvinian, A.; Gürsoy, B. Mycelium-based composite graded materials: Assessing the effects of time and substrate mixture on mechanical properties. Biomimetics 2022, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Chu, J.; Ma, L.; Qi, X.; Kumar, A. Life cycle assessment of plywood manufacturing process in China. Int. J. Environ. Res. Public Health 2019, 16, 2037. [Google Scholar] [CrossRef] [PubMed]
- Parhizi, Z.; Dearnaley, J.; Kauter, K.; Mikkelsen, D.; Pal, P.; Shelley, T.; Burey, P.P. The Fungus Among Us: Innovations and Applications of Mycelium-Based Composites. J. Fungi 2025, 11, 549. [Google Scholar] [CrossRef]
- IPCC. Sixth Assessment Report 2022; The Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2022. [Google Scholar]
- Bhuvaneshwari, S.; Hettiarachchi, H.; Meegoda, J.N. Crop residue burning in India: Policy challenges and potential solutions. Int. J. Environ. Res. Public Health 2019, 16, 832. [Google Scholar] [CrossRef] [PubMed]
- Defonseka, C. Polymeric Composites with Rice Hulls: An Introduction; Walter de Gruyter GmbH & Co. KG: Berlin, Germany, 2019. [Google Scholar]
- Karana, E.; Blauwhoff, D.; Hultink, E.-J.; Camere, S. When the material grows: A case study on designing (with) mycelium-based materials. Int. J. Des. 2018, 12, 119–136. [Google Scholar]
- Appels, F.V.W. The Use of Fungal Mycelium for the Production of Bio-Based Materials. Ph.D. Thesis, Universiteit Utrecht, Utrecht, The Netherlands, 2020. [Google Scholar]
- Yang, Z.; Zhang, F.; Still, B.; White, M.; Amstislavski, P. Physical and mechanical properties of fungal mycelium-based biofoam. J. Mater. Civ. Eng. 2017, 29, 04017030. [Google Scholar] [CrossRef]
- Attias, N.; Danai, O.; Tarazi, E.; Pereman, I.; Grobman, Y.J. Implementing bio-design tools to develop mycelium-based products. Des. J. 2019, 22, 1647–1657. [Google Scholar] [CrossRef]
- Shakir, M.A.; Azahari, B.; Yusup, Y.; Yhaya, M.F.; Salehabadi, A.; Ahmad, M.I. Preparation and characterization of mycelium as a bio-matrix in fabrication of bio-composite. J. Adv. Res. Fluid Mech. Therm. Sci. 2020, 65, 253–263. [Google Scholar]
- Jones, M.; Mautner, A.; Luenco, S.; Bismarck, A.; John, S. Engineered mycelium composite construction materials from fungal biorefineries: A critical review. Mater. Des. 2020, 187, 108397. [Google Scholar] [CrossRef]
- Attias, N.; Danai, O.; Ezov, N.; Tarazi, E.; Grobman, Y.J. Developing novel applications of mycelium based bio-composite materials for design and architecture. Proc. Build. Biobased Mater. Best Pract. Perform. Specif. 2017, 1, 4945479. [Google Scholar]
- Javadian, A.; Le Ferrand, H.; Hebel, D.E.; Saeidi, N. Application of mycelium-bound composite materials in construction industry: A short review. SOJ Mater. Sci. Eng. 2020, 7, 1–9. [Google Scholar] [CrossRef]
- Gezer, E.D.; Gümüşkaya, E.; Uçar, E.; Ustaömer, D. Mechanical properties of mycelium based MDF. Sigma J. Eng. Nat. Sci. 2020, 11, 135–140. [Google Scholar]
- Elsacker, E.; Vandelook, S.; Van Wylick, A.; Ruytinx, J.; De Laet, L.; Peeters, E. A comprehensive framework for the production of mycelium-based lignocellulosic composites. Sci. Total Environ. 2020, 725, 138431. [Google Scholar] [CrossRef] [PubMed]
- Holt, G.A.; Mcintyre, G.; Flagg, D.; Bayer, E.; Wanjura, J.; Pelletier, M. Fungal mycelium and cotton plant materials in the manufacture of biodegradable molded packaging material: Evaluation study of select blends of cotton byproducts. J. Biobased Mater. Bioenergy 2012, 6, 431–439. [Google Scholar] [CrossRef]
- Jones, M.; Huynh, T.; Dekiwadia, C.; Daver, F.; John, S. Mycelium composites: A review of engineering characteristics and growth kinetics. J. Bionanosci. 2017, 11, 241–257. [Google Scholar] [CrossRef]
- Pelkmans, J.F.; Lugones, L.G.; Wösten, H.A. 15 fruiting body formation in basidiomycetes. In Growth, Differentiation and Sexuality; Springer: Berlin/Heidelberg, Germany, 2016; pp. 387–405. [Google Scholar]
- Deacon, J.W. Introduction to Modern Mycology; Basic Microbiology; Halsted Press: New York, NY, USA, 1980; Volume 7. [Google Scholar]
- Kavanagh, K. Fungi: Biology and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Lelivelt, R. The Mechanical Possibilities of Mycelium Materials; Eindhoven University of Technology (TU/e): Eindhoven, The Netherlands, 2015; Volume 682. [Google Scholar]
- Appels, F.V.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.; Dijksterhuis, J.; Krijgsheld, P.; Wösten, H.A. Fabrication factors influencing mechanical, moisture-and water-related properties of mycelium-based composites. Mater. Des. 2019, 161, 64–71. [Google Scholar] [CrossRef]
- Kundanati, L. Fungi-based Biomimetic Approach to Address Plastic Pollution: A Developing Nation’s Perspective. Preprint 2022. [Google Scholar] [CrossRef]
- Worrall, J.J. Media for selective isolation of hymenomycetes. Mycologia 1991, 83, 296–302. [Google Scholar] [CrossRef]
- ASTM D1037-12; Standard Test Methods for Evaluating Properties of Wood-Base Fiber and Particle Panel Materials. ASTM: Conshohocken, PA, USA, 2020.
- Gou, L.; Li, S.; Yin, J.; Li, T.; Liu, X. Morphological and physico-mechanical properties of mycelium biocomposites with natural reinforcement particles. Constr. Build. Mater. 2021, 304, 124656. [Google Scholar] [CrossRef]
- Amstislavski, P.; Pöhler, T.; Valtonen, A.; Wikström, L.; Harlin, A.; Salo, S.; Jetsu, P.; Szilvay, G.R. Low-density, water-repellent, and thermally insulating cellulose-mycelium foams. Cellulose 2024, 31, 8769–8785. [Google Scholar] [CrossRef]
- Elsacker, E.; Vandelook, S.; Brancart, J.; Peeters, E.; De Laet, L. Mechanical, physical and chemical characterisation of mycelium-based composites with different types of lignocellulosic substrates. PLoS ONE 2019, 14, e0213954. [Google Scholar] [CrossRef]
- Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A.; Athanassiou, A. Advanced materials from fungal mycelium: Fabrication and tuning of physical properties. Sci. Rep. 2017, 7, 41292. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.; Pereira, H.; Hinterstoisser, B. Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib. Spectrosc. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Mohebby, B. Attenuated total reflection infrared spectroscopy of white-rot decayed beech wood. Int. Biodeterior. Biodegrad. 2005, 55, 247–251. [Google Scholar] [CrossRef]
- Pandey, K.; Pitman, A. FTIR studies of the changes in wood chemistry following decay by brown-rot and white-rot fungi. Int. Biodeterior. Biodegrad. 2003, 52, 151–160. [Google Scholar] [CrossRef]
- Jones, M.; Bhat, T.; Huynh, T.; Kandare, E.; Yuen, R.; Wang, C.H.; John, S. Waste-derived low-cost mycelium composite construction materials with improved fire safety. Fire Mater. 2018, 42, 816–825. [Google Scholar] [CrossRef]
- Sayfutdinova, A.R.; Cherednichenko, K.A.; Rakitina, M.A.; Dubinich, V.N.; Bardina, K.A.; Rubtsova, M.I.; Petrova, D.A.; Vinokurov, V.A.; Voronin, D.V. Natural fibrous materials based on fungal mycelium hyphae as porous supports for shape-stable phase-change composites. Polymers 2023, 15, 4504. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Bhat, T.; Wang, C.H.; Moinuddin, K.; John, S. Thermal degradation and fire reaction properties of mycelium composites. In Proceedings of the 21st International Conference on Composite Materials, Xi’an, China, 20–25 August 2017; pp. 20–25. [Google Scholar]
- Lingam, D.; Narayan, S.; Mamun, K.; Charan, D. Engineered mycelium-based composite materials: Comprehensive study of various properties and applications. Constr. Build. Mater. 2023, 391, 131841. [Google Scholar] [CrossRef]
- Houette, T.; Maurer, C.; Niewiarowski, R.; Gruber, P. Growth and mechanical characterization of mycelium-based composites towards future bioremediation and food production in the material manufacturing cycle. Biomimetics 2022, 7, 103. [Google Scholar] [CrossRef]
- van Empelen, J.C. A Study into More Sustainable, Alternative Building Materials as A Substitute for Concrete in Tropical Climates; Delft University of Technology: Delft, The Netherlands, 2018; pp. 1–26. [Google Scholar]
- Shakir, M.A.; Ahmad, M.I.; Yusup, Y.; Rafatullah, M. From waste to wealth: Converting rubber wood sawdust into green mycelium-based composite. Biomass Convers. Biorefin. 2025, 15, 739–757. [Google Scholar] [CrossRef]
- Ghazanfari, A.; Malzl, L.; Pramreiter, M.; Konnerth, J.; Kromoser, B. A systematic review of strand-based engineered wood products for construction: Standard test methods and mechanical properties. Wood Mater. Sci. Eng. 2025, 21, 1012–1034. [Google Scholar] [CrossRef]
- Sood, M.; Dwivedi, G. Effect of fiber treatment on flexural properties of natural fiber reinforced composites: A review. Egypt. J. Pet. 2018, 27, 775–783. [Google Scholar] [CrossRef]
- Boon, J.G.; Hashim, R.; Danish, M.; Nadhari, W.N.A.W. Physical and mechanical properties of binderless particleboard made from steam-pretreated oil palm trunk particles. J. Compos. Sci. 2019, 3, 46. [Google Scholar] [CrossRef]
- Elsacker, E.; Søndergaard, A.; Van Wylick, A.; Peeters, E.; De Laet, L. Growing living and multifunctional mycelium composites for large-scale formwork applications using robotic abrasive wire-cutting. Constr. Build. Mater. 2021, 283, 122732. [Google Scholar] [CrossRef]
- Sydor, M.; Cofta, G.; Doczekalska, B.; Bonenberg, A. Fungi in mycelium-based composites: Usage and recommendations. Materials 2022, 15, 6283. [Google Scholar] [CrossRef]
- Alemu, D.; Tafesse, M.; Mondal, A.K. Mycelium-based composite: The future sustainable biomaterial. Int. J. Biomater. 2022, 2022, 8401528. [Google Scholar] [CrossRef]
- Aiduang, W.; Chanthaluck, A.; Kumla, J.; Jatuwong, K.; Srinuanpan, S.; Waroonkun, T.; Oranratmanee, R.; Lumyong, S.; Suwannarach, N. Amazing fungi for eco-friendly composite materials: A comprehensive review. J. Fungi 2022, 8, 842. [Google Scholar] [CrossRef]
- Aiduang, W.; Kumla, J.; Srinuanpan, S.; Thamjaree, W.; Lumyong, S.; Suwannarach, N. Mechanical, physical, and chemical properties of mycelium-based composites produced from various lignocellulosic residues and fungal species. J. Fungi 2022, 8, 1125. [Google Scholar] [CrossRef] [PubMed]
- Aiduang, W.; Jatuwong, K.; Jinanukul, P.; Suwannarach, N.; Kumla, J.; Thamjaree, W.; Teeraphantuvat, T.; Waroonkun, T.; Oranratmanee, R.; Lumyong, S. Sustainable Innovation: Fabrication and characterization of mycelium-based green composites for modern interior materials using agro-industrial wastes and different species of fungi. Polymers 2024, 16, 550. [Google Scholar] [CrossRef] [PubMed]
- Birinci, A.U.; Demir, A.; Ozturk, H. Comparison of thermal performances of plywood shear walls produced with different thermal insulation materials. Maderas. Cienc. Tecnol. 2022, 24. [Google Scholar] [CrossRef]
- Ashby, M.F.; Cebon, D. Materials selection in mechanical design. J. Phys. IV 1993, 3, C7-1–C7-9. [Google Scholar] [CrossRef]
- Sun, W.; Tajvidi, M.; Hunt, C.G.; Howell, C. All-natural smart mycelium surface with tunable wettability. ACS Appl. Bio Mater. 2020, 4, 1015–1022. [Google Scholar] [CrossRef]




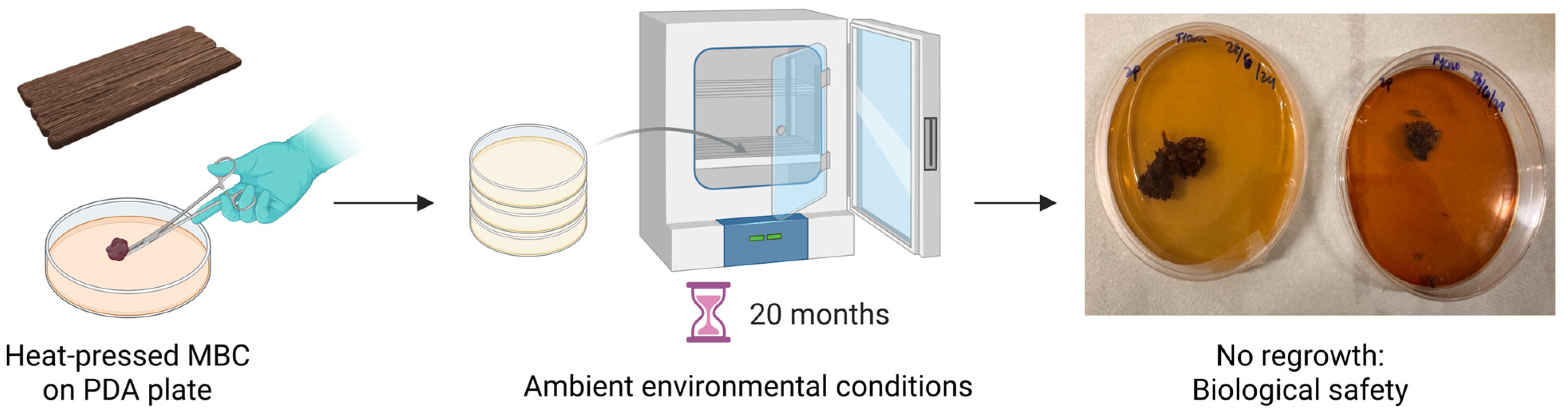
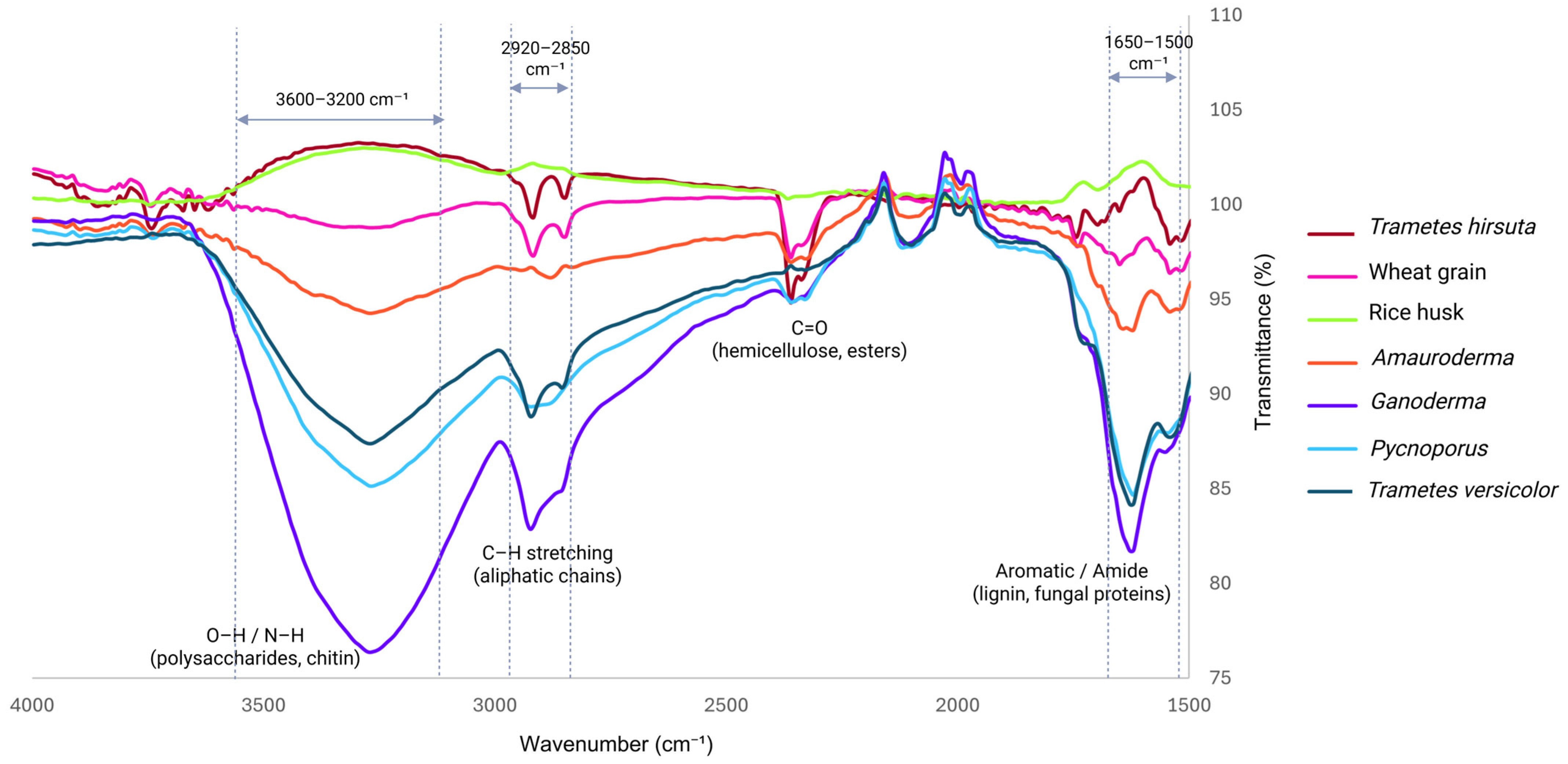
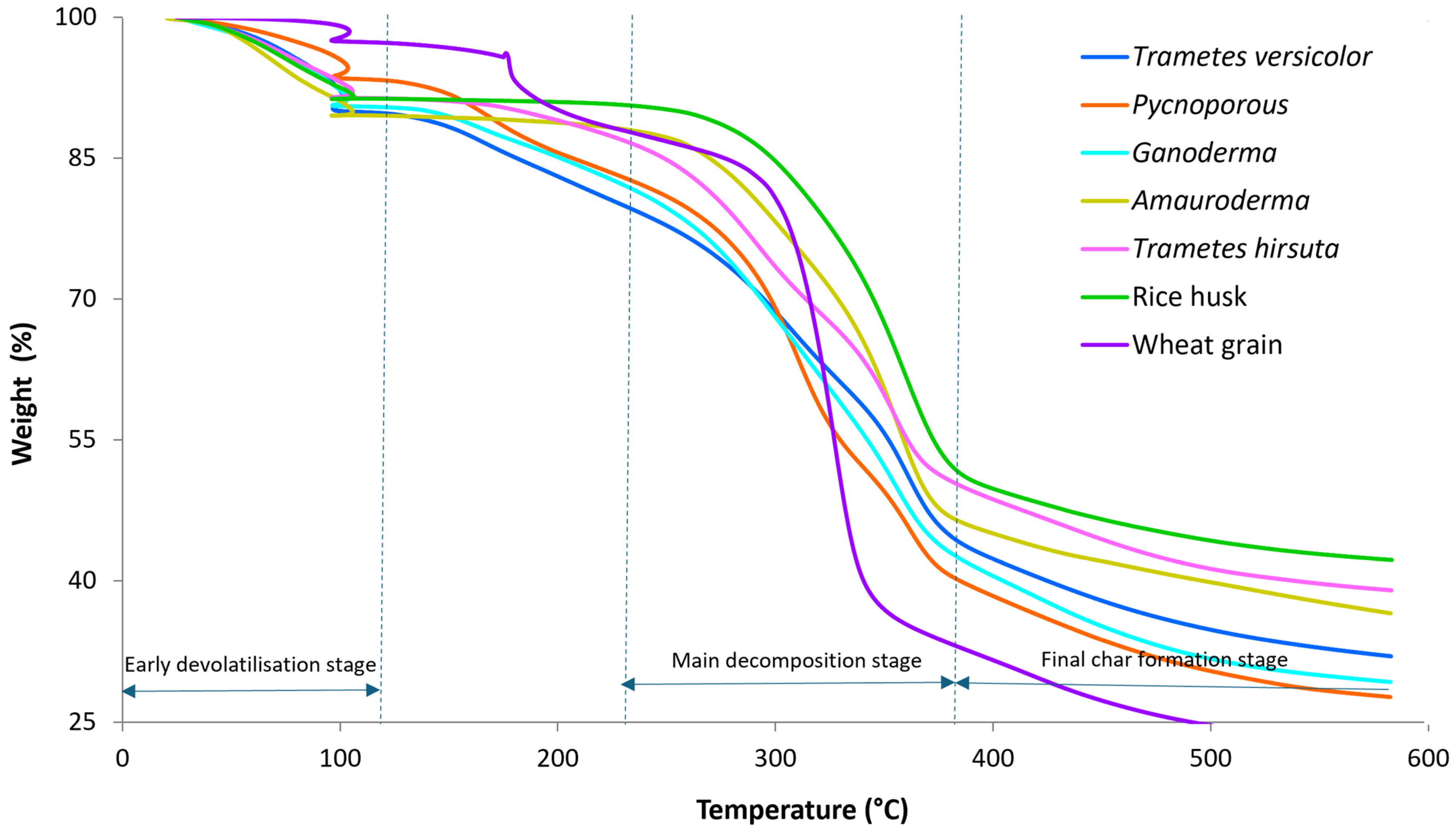

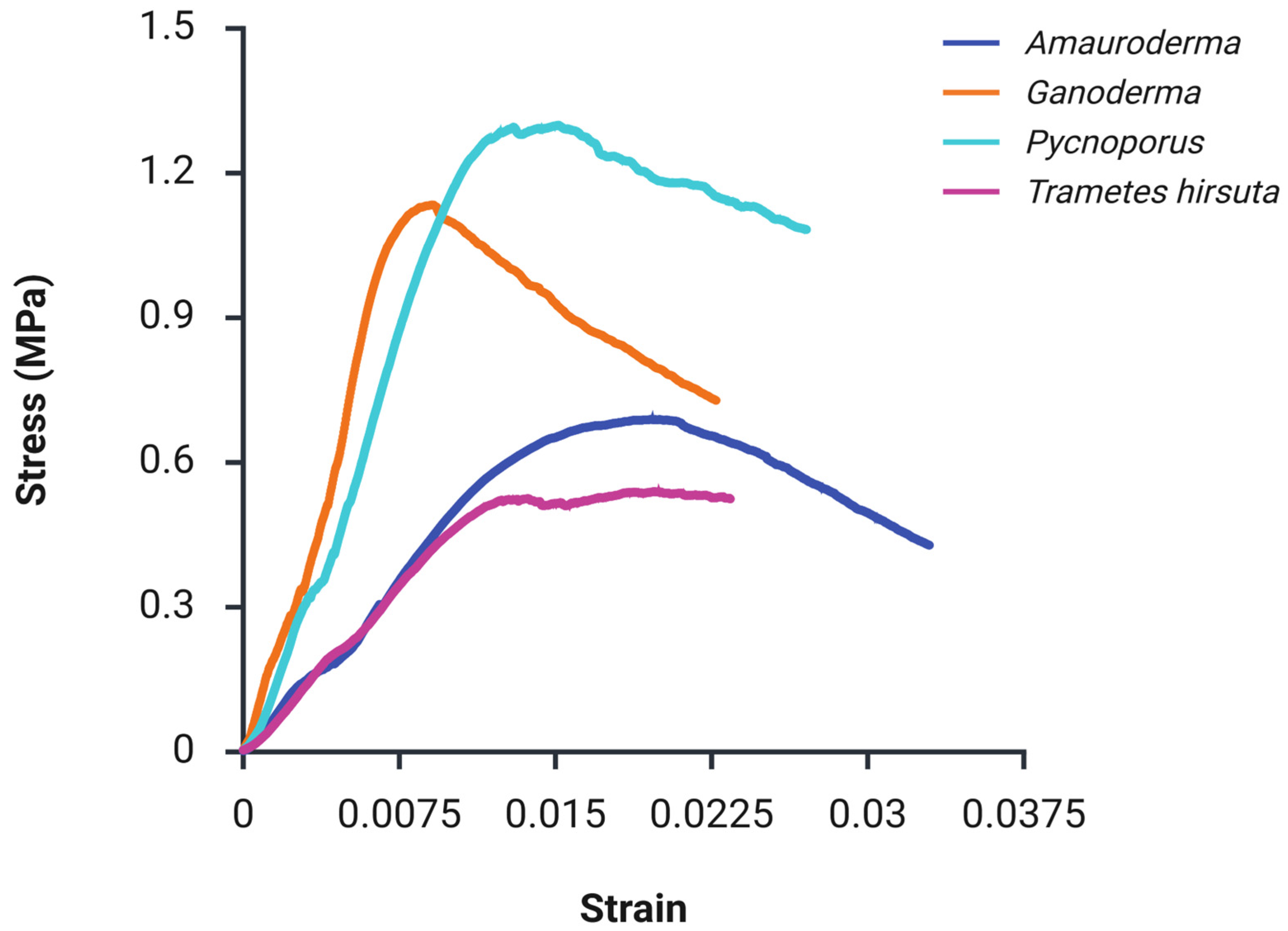

| Species | Thickness (mm) | Tensile Strength (MPa) | Young’s Modulus (MPa) | Compression Strength (MPa) | Elastic Modulus (MPa) | Flexural Strength (MPa) | Flexural Modulus (MPa) |
|---|---|---|---|---|---|---|---|
| Amauroderma sp. | 6.33 ± 1.75 | 0.52 ± 0.12 | 294.75 ± 184.42 | 0.35 ± 0.44 | 11.46 ± 22.30 | 0.69 ± 0.39 | 270.21 |
| Ganoderma sp. | 5.88 ± 0.25 | 0.27 ± 0.18 | 28.66 ± 12.01 | 0.21 ± 0.15 | 10.84 ± 7.04 | 1.19 ± 0.14 | 352.84 |
| Pycnoporus coccineus | 5.77 ± 0.26 | 0.45 ± 0.02 | 102.14 ± 15.87 | 0.38 ± 0.19 | 22.45 ± 13.19 | 1.38 ± 0.11 | 532.91 |
| Trametes hirsuta | 5.45 ± 1.26 | 0.16 ± 0.07 | 17.84 ± 59.77 | 0.13 ± 0.16 | 6.09 ± 9.65 | 0.60 ± 0.20 | 172.95 |
| Sample | Average Density (kg/m3) | Sample Weight (g) | Sample Volume (cm3) |
|---|---|---|---|
| Pycnoporus coccineus | 250.137 ± 0.0005 | 0.5097 | 0.3390 ± 0.0001 |
| Ganoderma sp. | 251.730 ± 0.0010 | 0.2984 | 0.1967 ± 0.0001 |
| Trametes hirsuta | 250.410 ± 0.0010 | 0.3783 | 0.2515 ± 0.0002 |
| Amauroderma sp. | 259.140 ± 0.0023 | 0.3133 | 0.1969 ± 0.0003 |
| Trametes versicolor | 258.870 ± 0.0005 | 0.3484 | 0.2193 ± 0.0001 |
| Wheat grain | 245.290 ± 0.0001 | 1.4529 | 1.1000 ± 0.0001 |
| Rice husk | 235.153 ± 0.0020 | 0.2184 | 0.1611 ± 0.0002 |
| Fungal Species | Biomass Contact Angle (°) | Wettability Status | Heat-Pressed MBC Contact Angle (°) | Wettability Status |
|---|---|---|---|---|
| Amauroderma sp. | 107.2 | Hydrophobic (>90°) | 140.6 | Hydrophobic (>90°) |
| Ganoderma sp. | 115.7 | Hydrophobic (>90°) | 138.3 | Hydrophobic (>90°) |
| Trametes versicolor | 84.9 | More wettable (<90°) | 81.3 | More wettable (<90°) |
| Trametes hirsuta | 114.6 | Hydrophobic (>90°) | 132.2 | Hydrophobic (>90°) |
| Pycnoporus coccineus | 112.4 | Hydrophobic (>90°) | 134.5 | Hydrophobic (>90°) |
| Property | F-Value | p-Value | Significant (α = 0.05) |
|---|---|---|---|
| Flexural | 2.40 | 0.14 | No |
| Tensile | 6.28 | 0.017 | Yes |
| Compression | 0.58 | 0.64 | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Parhizi, Z.; Jadwiszczak, E.; Dearnaley, J.; Mikkelsen, D.; Burey, P. Walls That Grow: Fungal Species-Driven Performance of Mycelium-Based Composites Grown on Rice-Husk Waste. Polymers 2026, 18, 932. https://doi.org/10.3390/polym18080932
Parhizi Z, Jadwiszczak E, Dearnaley J, Mikkelsen D, Burey P. Walls That Grow: Fungal Species-Driven Performance of Mycelium-Based Composites Grown on Rice-Husk Waste. Polymers. 2026; 18(8):932. https://doi.org/10.3390/polym18080932
Chicago/Turabian StyleParhizi, Zahra, Ewa Jadwiszczak, John Dearnaley, Deirdre Mikkelsen, and Paulomi (Polly) Burey. 2026. "Walls That Grow: Fungal Species-Driven Performance of Mycelium-Based Composites Grown on Rice-Husk Waste" Polymers 18, no. 8: 932. https://doi.org/10.3390/polym18080932
APA StyleParhizi, Z., Jadwiszczak, E., Dearnaley, J., Mikkelsen, D., & Burey, P. (2026). Walls That Grow: Fungal Species-Driven Performance of Mycelium-Based Composites Grown on Rice-Husk Waste. Polymers, 18(8), 932. https://doi.org/10.3390/polym18080932






