Sustained Delivery of Paliperidone Palmitate via Encapsulation in Bio-Based NIPU Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
2.2.1. Synthesis of Neat NIPU Nanoparticles Through Nanoemulsions
2.2.2. Synthesis of PP-Loaded NIPU Nanoparticles Through Nanoemulsions
2.3. Characterization
2.3.1. Characterization of Neat NIPU Nanoparticles
Fourier-Transform Infrared Spectroscopy (FTIR)
Differential Scanning Calorimetry (DSC)
X-Ray Diffraction Analysis (XRD)
Mechanical Tests
Dynamic Light Scattering (DLS)
Scanning Electron Microscopy (SEM)
Porosity Test
Hydrolysis and Enzymatic Hydrolysis
2.3.2. Characterization of PP-Loaded NIPU Nanoparticles
Water Contact Angle Analysis
Drug Loading
In Vitro Dissolution Studies
High-Pressure Liquid Chromatography (HPLC)
3. Results and Discussion
3.1. Synthesis of the Materials
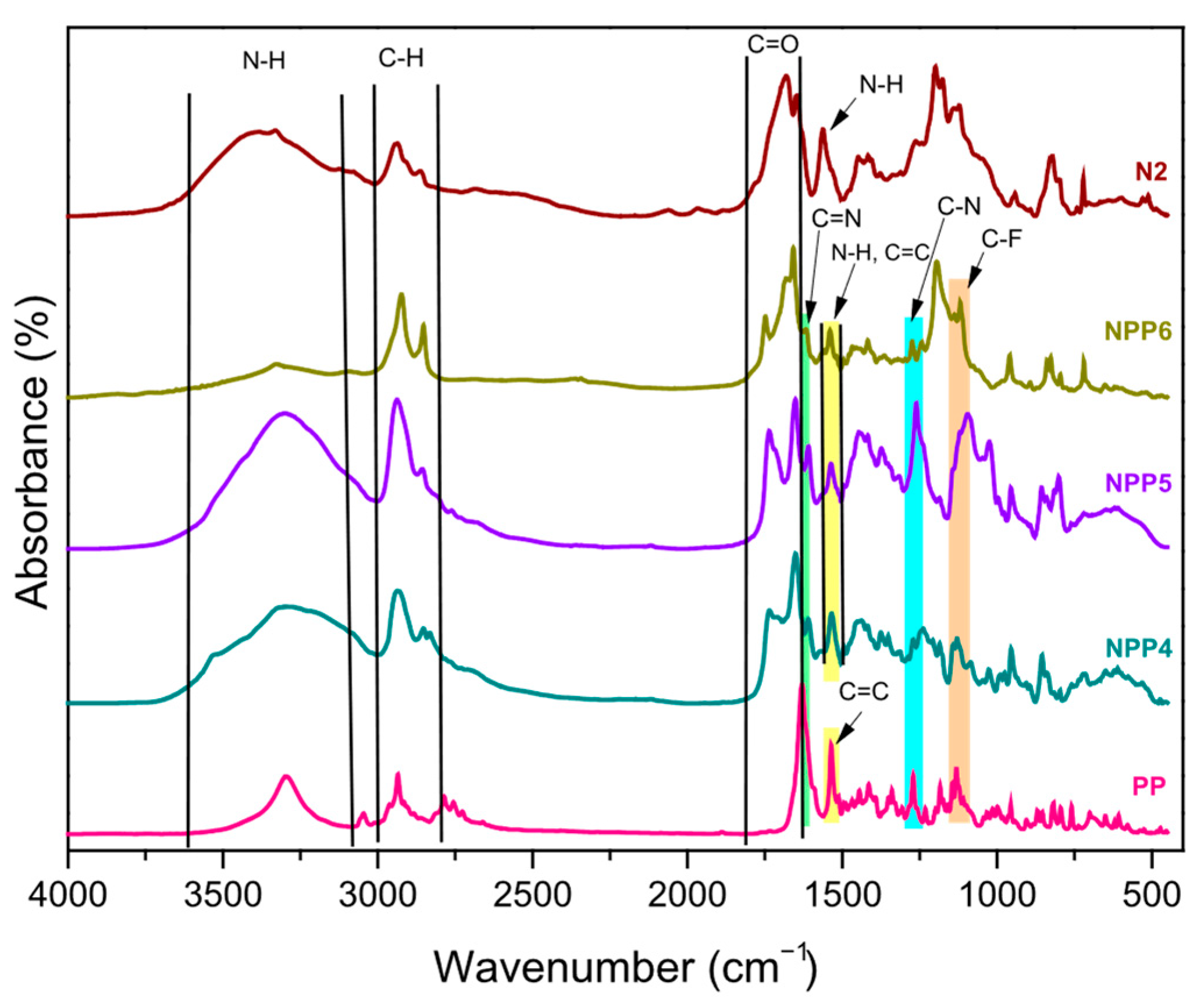
3.2. FTIR
3.3. Thermal Transitions (DSC)
3.4. XRD
3.5. Mechanical Tests
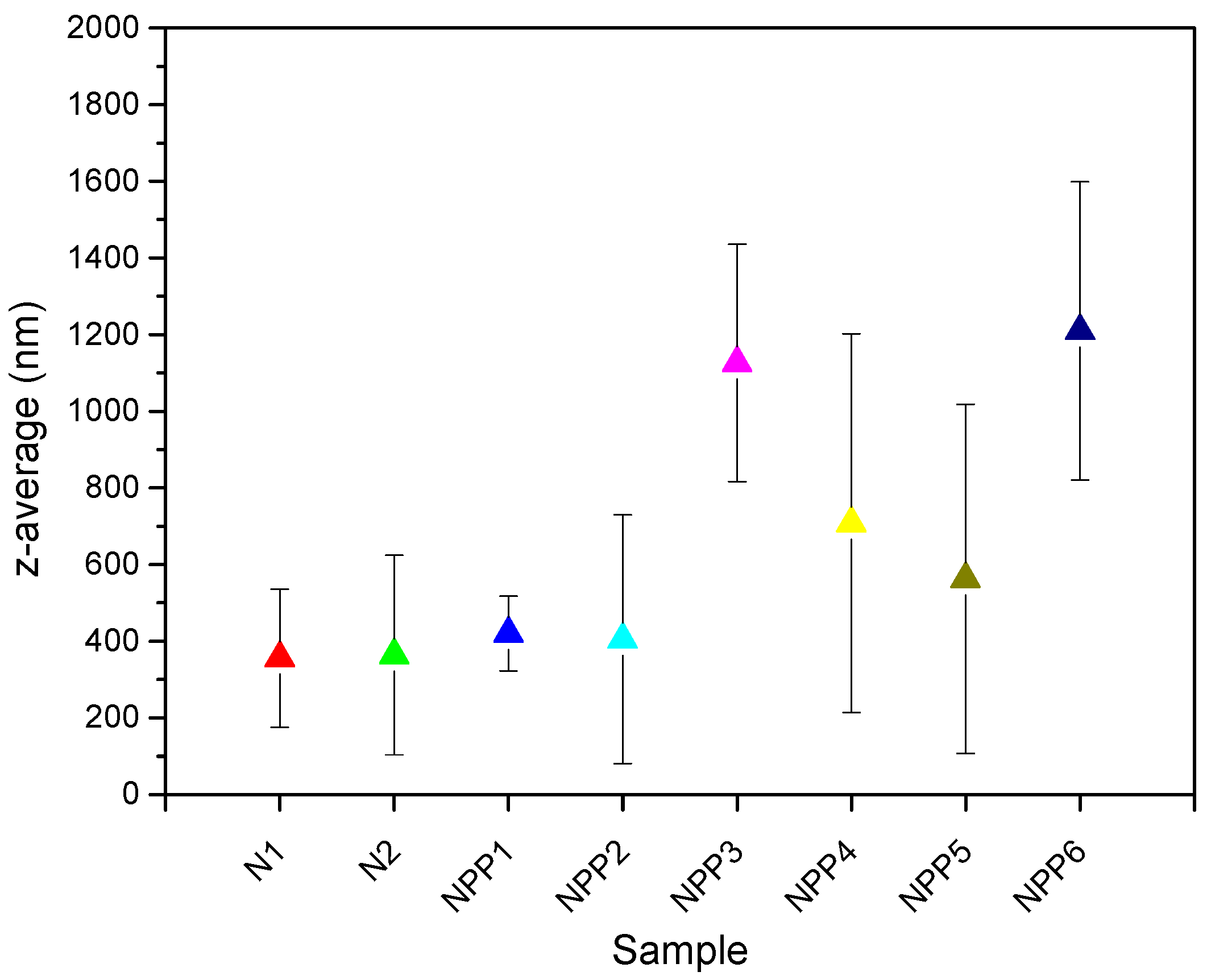
3.6. DLS
3.7. SEM
3.8. Porosity Test
3.9. Water Contact Angle
3.10. Hydrolysis and Enzymatic Hydrolysis
3.11. Drug Loading
3.12. In Vitro Dissolution
3.13. Modeling of Drug Release Profiles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Chue, P.; Chue, J. A Review of Paliperidone Palmitate. Expert Rev. Neurother. 2012, 12, 1383–1397. [Google Scholar] [CrossRef]
- Nanaki, S.; Tseklima, M.; Christodoulou, E.; Triantafyllidis, K.; Kostoglou, M.; Bikiaris, D.N. Thiolated Chitosan Masked Polymeric Microspheres with Incorporated Mesocellular Silica Foam (MCF) for Intranasal Delivery of Paliperidone. Polymers 2017, 9, 617. [Google Scholar] [CrossRef]
- Nanaki, S.; Tseklima, M.; Terzopoulou, Z.; Nerantzaki, M.; Giliopoulos, D.J.; Triantafyllidis, K.; Kostoglou, M.; Bikiaris, D.N. Use of Mesoporous Cellular Foam (MCF) in Preparation of Polymeric Microspheres for Long Acting Injectable Release Formulations of Paliperidone Antipsychotic Drug. Eur. J. Pharm. Biopharm. 2017, 117, 77–90. [Google Scholar] [CrossRef]
- Muthu, M.S.; Sahu, A.K.; Sonali; Abdulla, A.; Kaklotar, D.; Rajesh, C.V.; Singh, S.; Pandey, B.L. Solubilized Delivery of Paliperidone Palmitate by D- Alpha-Tocopheryl Polyethylene Glycol 1000 Succinate Micelles for Improved Short-Term Psychotic Management. Drug Deliv. 2016, 23, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Bikiaris, R.; Christodoulou, E.; Kostoglou, M.; Kasimatis, M.; Iatrou, H.; Nikolaidis, N. Paliperidone Palmitate Depot Microspheres Based on Biocompatible Poly(Alkylene Succinate) Polyesters as Long-Acting Injectable Formulations. J. Drug Deliv. Sci. Technol. 2021, 68, 103056. [Google Scholar] [CrossRef]
- Sangroniz, L.; Cavallo, D.; Müller, A.J. Self-Nucleation Effects on Polymer Crystallization. Macromolecules 2020, 53, 4581–4604. [Google Scholar] [CrossRef]
- Klonos, P.A.; Papadopoulos, L.; Kasimatis, M.; Iatrou, H.; Kyritsis, A.; Bikiaris, D.N. Synthesis, Crystallization, Structure Memory Effects, and Molecular Dynamics of Biobased and Renewable Poly(n-Alkylene Succinate)s with n from 2 to 10. Macromolecules 2021, 54, 1106–1119. [Google Scholar] [CrossRef]
- Balla, E.; Daniilidis, V.; Karlioti, G.; Kalamas, T.; Stefanidou, M.; Bikiaris, N.D.; Vlachopoulos, A.; Koumentakou, I.; Bikiaris, D.N. Poly(Lactic Acid): A Versatile Biobased Polymer for the Future with Multifunctional Properties-from Monomer Synthesis, Polymerization Techniques and Molecular Weight Increase to PLA Applications. Polymers 2021, 13, 1822. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, A.; Karlioti, G.; Balla, E.; Daniilidis, V.; Kalamas, T.; Stefanidou, M.; Bikiaris, N.D.; Christodoulou, E.; Koumentakou, I.; Karavas, E.; et al. Poly(Lactic Acid)-Based Microparticles for Drug Delivery Applications: An Overview of Recent Advances. Pharmaceutics 2022, 14, 359. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Zamboulis, A.; Bikiaris, N.D.; Xanthopoulou, E.; Ioannidis, R.O.; Bikiaris, D.N. A Decade of Innovation: Synthesis, Properties and Applications of PLA Copolymers. Prog. Polym. Sci. 2025, 167, 101991. [Google Scholar] [CrossRef]
- Zorkina, Y.; Abramova, O.; Ushakova, V.; Morozova, A.; Zubkov, E.; Valikhov, M.; Melnikov, P.; Majouga, A.; Chekhonin, V. Nano Carrier Drug Delivery Systems for the Treatment of Neuropsychiatric Disorders: Advantages and Limitations. Molecules 2020, 25, 5294. [Google Scholar] [CrossRef]
- Watanabe, T.; Ono, T.; Kimura, Y. Continuous Fabrication of Monodisperse Polylactide Microspheres by Droplet-to-Particle Technology Using Microfluidic Emulsification and Emulsion-Solvent Diffusion. Soft Matter 2011, 7, 9894–9897. [Google Scholar] [CrossRef]
- Lubrizol Pellethane® TPU. Available online: https://www.lubrizol.com/solutions/products/pellethane (accessed on 20 February 2026).
- Lubrizol TecoflexTM TPU. Available online: https://www.lubrizol.com/solutions/products/tecoflex (accessed on 20 February 2026).
- Lubrizol CarbothaneTM TPU. Available online: https://www.lubrizol.com/solutions/products/carbothane (accessed on 20 February 2026).
- Tan, R.Y.H.; Lee, C.S.; Pichika, M.R.; Cheng, S.F.; Lam, K.Y. PH-Responsive Polyurethane-Acrylate from Palm Olein and Sunflower Oil: Sustainable Synthesis, Enhanced Lovastatin Solubility, and Potential Drug Carrier for Targeted Delivery. J. Appl. Polym. Sci. 2024, 141, e55697. [Google Scholar] [CrossRef]
- Balla, E.; Bikiaris, D.N.; Pardalis, N.; Bikiaris, N.D. Toward Sustainable Polyurethane Alternatives: A Review of the Synthesis, Applications, and Lifecycle of Non-Isocyanate Polyurethanes (NIPUs). Polymers 2025, 17, 1364. [Google Scholar] [CrossRef]
- Velez-Pardo, A.; Díaz, L.E.; Valero, M.F. Functionalization Strategies of Non-Isocyanate Polyurethanes (NIPUs): A Systematic Review of Mechanical and Biological Advances. Polymers 2025, 17, 3255. [Google Scholar] [CrossRef]
- Aduba, D.C.; Zhang, K.; Kanitkar, A.; Sirrine, J.M.; Verbridge, S.S.; Long, T.E. Electrospinning of Plant Oil-Based, Non-Isocyanate Polyurethanes for Biomedical Applications. J. Appl. Polym. Sci. 2018, 135, 46464. [Google Scholar] [CrossRef]
- Favre, L.M.; Masurier, N.; Aubert-Pouëssel, A. Vegetable Oil-Based Materials for Drug Delivery Systems and Wound Dressings. Macromol. Biosci. 2025, 26, e00486. [Google Scholar] [CrossRef]
- Bhaliya, J.D.; Kutcherlapati, S.N.R.; Dhore, N.; Punugupati, N.; Sunkara, K.L.; Misra, S.; Joshi, S.S.K. Soybean Oil-Derived, Non-Isocyanate Polyurethane-TiO2 Nanocomposites with Enhanced Thermal, Mechanical, Hydrophobic and Antimicrobial Properties. RSC Sustain. 2025, 3, 1434–1447. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, Z.; Pathak, J.L.; Feng, W.; Wu, W.; Yang, C.; Wu, L.; Zheng, H. Fabrication and Characterization of Photosensitive Non-Isocyanate Polyurethane Acrylate Resin for 3D Printing of Customized Biocompatible Orthopedic Surgical Guides. Int. J. Bioprint. 2023, 9, 80–93. [Google Scholar] [CrossRef]
- Ntrivala, M.A.G.; Balla, E.D.; Klonos, P.A.; Kyritsis, A.; Bikiaris, D.N. Synthesis and Characterization of Novel Non-Isocyanate Polyurethanes Derived from Adipic Acid: A Comprehensive Study. Polymers 2025, 17, 728. [Google Scholar] [CrossRef]
- ASTM D882-18; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM: West Conshohocken, PA, USA, 2018.
- Wang, Z.; Gao, T.; Cui, L.; Wang, Y.; Zhang, P.; Chen, X. Electronic Supplementary Information for Improved Cellular Infiltration into 3D Interconnected Microchannel Scaffolds Formed by Using Melt-Spun Sacrificial Microfibers. RSC Adv. 2015, 6, 2131–2134. [Google Scholar] [CrossRef]
- Ramay, H.R.; Zhang, M. Preparation of Porous Hydroxyapatite Scaffolds by Combination of the Gel-Casting and Polymer Sponge Methods. Biomaterials 2003, 24, 3293–3302. [Google Scholar] [CrossRef]
- Nadendla, R.R.; Pinnamaneni, P.; Morla, S.P.; Abhinandana, P. Physico-Chemical Characterization of Paliperidone Palmitate and Compatibility Studies with Its Pharmaceutical Excipients. J. Pharm. Res. Int. 2021, 33, 85–91. [Google Scholar] [CrossRef]
- Parthiban, K.; George, J.K.; Ajayakumar, A. Formulation and Evaluation of Paliperidone Palmitate Loaded Nanoparticles Using Biodegradable Polymers. Int. J. Res. Sci. Innov. (IJRSI) 2019, 6, 1–6. [Google Scholar]
- Mali, S.; Oza, N. Central Composite Design for Formulation and Optimization of Long-Acting Injectable (LAI) Microspheres of Paliperidone Palmitate. Int. J. Appl. Pharm. 2021, 13, 87–98. [Google Scholar] [CrossRef]
- Maniyar, A.J.; Patel, G.M.; Shelat, P.K.; Lalwani, A.N. Development and Characterization of Thermosensitive Intranasal Gel Containing Paliperidone Loaded Microspheres. J. Young Pharm. 2016, 8, 368–377. [Google Scholar] [CrossRef]
- Shi, J.; Wang, D.; Tian, Y.; Wang, Z.; Gao, J.; Liu, N.; Gao, X.; Zheng, A.; Zhang, H.; Xiang, M. Comparison of Paliperidone Palmitate from Different Crystallization Processes and Effect on Formulations In Vitro and In Vivo. Pharmaceutics 2022, 14, 1094. [Google Scholar] [CrossRef]
- Hebda, E.; Wróbel, K.; Cieślik, A.; Szołdrowska, K.; Ozimek, J.; Zając, P.; Raftopoulos, K.N.; Pielichowski, K. Structurally Ordered NIPUs via Catalyst-Free Synthesis with Hard Segments Based on Erythritol and a Long-Chain Diamine. Molecules 2025, 30, 2912. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Nelson, A.M.; Talley, S.J.; Chen, M.; Margaretta, E.; Hudson, A.G.; Moore, R.B.; Long, T.E. Non-Isocyanate Poly(Amide-Hydroxyurethane)s from Sustainable Resources. Green Chem. 2016, 18, 4667–4681. [Google Scholar] [CrossRef]
- Yin, Q.; Xu, B.; Qin, Y.; Zhao, J.; Cheng, J.; Zhang, J. Biobased Linear and Crystallizable Polyhydroxy(Amide-Urethane)s from Diglycerol Bis(Cyclic Carbonate) and the Polyamides of Dimer Fatty Acids. ACS Appl. Polym. Mater. 2022, 4, 2116–2131. [Google Scholar] [CrossRef]
- Hsu, S.T.; Yao, Y.L. Effect of Drug Loading and Laser Surface Melting on Drug Release Profile from Biodegradable Polymer. J. Appl. Polym. Sci. 2013, 130, 4147–4156. [Google Scholar] [CrossRef]
- Balla, E.D.; Bikiaris, N.D.; Nanaki, S.G.; Papoulia, C.; Chrissafis, K.; Klonos, P.A.; Kyritsis, A.; Kostoglou, M.; Zamboulis, A.; Papageorgiou, G.Z. Chloramphenicol Loaded Sponges Based on Pva/Nanocellulose Nanocomposites for Topical Wound Delivery. J. Compos. Sci. 2021, 5, 208. [Google Scholar] [CrossRef]
- Liu, J.; Xiao, Y.; Allen, C. Polymer-Drug Compatibility: A Guide to the Development of Delivery Systems for the Anticancer Agent, Ellipticine. J. Pharm. Sci. 2004, 93, 132–143. [Google Scholar] [CrossRef]
- Zhou, G.; Zhu, J.; Inverarity, C.; Fang, Y.; Zhang, Z.; Ye, H.; Cui, Z.; Nguyen, L.; Wan, H.; Dye, J.F. Fabrication of Fibrin/Polyvinyl Alcohol Scaffolds for Skin Tissue Engineering via Emulsion Templating. Polymers 2023, 15, 1151. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.H.; Wei, D.X.; Ye, H.M.; Zhang, X.; Meng, X.; Zhou, Q. Development of Poly(Vinyl Alcohol) Porous Scaffold with High Strength and Well Ciprofloxacin Release Efficiency. Mater. Sci. Eng. C 2016, 67, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Jhon, Y.K.; Cheong, I.W.; Kim, J.H. Effect of Process Variables on Molecular Weight and Mechanical Properties of Water-Based Polyurethane Dispersion. Colloids Surf. A Physicochem. Eng. Asp. 2002, 196, 135–143. [Google Scholar] [CrossRef]
- Ozimek, J.; Pielichowski, K. Sustainability of Nonisocyanate Polyurethanes (NIPUs). Sustainability 2024, 16, 9911. [Google Scholar] [CrossRef]
- Tamami, B.; Sohn, S.; Wilkes, G.L. Incorporation of Carbon Dioxide into Soybean Oil and Subsequent Preparation and Studies of Nonisocyanate Polyurethane Networks. J. Appl. Polym. Sci. 2004, 92, 883–891. [Google Scholar] [CrossRef]
- Taghipour, B.; Yakhchali, M.; Haririan, I.; Tamaddon, A.M.; Samani, S.M. The Effects of Technical and Compositional Variables on the Size and Release Profile of Bovine Serum Albumin from PLGA Based Particulate Systems. Res. Pharm. Sci. 2014, 9, 407–420. [Google Scholar]
- Yenying, A.; Tangamatakul, K.; Supanchart, C.; Jenvoraphot, T.; Manokruang, K.; Worajittiphon, P.; Punyodom, W.; Daranarong, D. Preparation and Characterization of PLG Microparticles by the Multiple Emulsion Method for the Sustained Release of Proteins. Micromachines 2022, 13, 1761. [Google Scholar] [CrossRef]
- Budhian, A.; Siegel, S.J.; Winey, K.I. Haloperidol-Loaded PLGA Nanoparticles: Systematic Study of Particle Size and Drug Content. Int. J. Pharm. 2007, 336, 367–375. [Google Scholar] [CrossRef]
- Song, X.; Zhao, Y.; Hou, S.; Xu, F.; Zhao, R.; He, J.; Cai, Z.; Li, Y.; Chen, Q. Dual Agents Loaded PLGA Nanoparticles: Systematic Study of Particle Size and Drug Entrapment Efficiency. Eur. J. Pharm. Biopharm. 2008, 69, 445–453. [Google Scholar] [CrossRef]
- Madani, F.; Esnaashari, S.S.; Mujokoro, B.; Dorkoosh, F.; Khosravani, M.; Adabi, M. Investigation of Effective Parameters on Size of Paclitaxel Loaded PLGA Nanoparticles. Adv. Pharm. Bull. 2018, 8, 77–84. [Google Scholar] [CrossRef]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]
- Hoseini, B.; Jaafari, M.R.; Golabpour, A.; Momtazi-Borojeni, A.A.; Karimi, M.; Eslami, S. Application of Ensemble Machine Learning Approach to Assess the Factors Affecting Size and Polydispersity Index of Liposomal Nanoparticles. Sci. Rep. 2023, 13, 18012. [Google Scholar] [CrossRef]
- Ruiz, E.; Orozco, V.H.; Hoyos, L.M.; Giraldo, L.F. Study of Sonication Parameters on PLA Nanoparticles Preparation by Simple Emulsion-Evaporation Solvent Technique. Eur. Polym. J. 2022, 173, 111307. [Google Scholar] [CrossRef]
- Michen, B.; Geers, C.; Vanhecke, D.; Endes, C.; Rothen-Rutishauser, B.; Balog, S.; Petri-Fink, A. Avoiding Drying-Artifacts in Transmission Electron Microscopy: Characterizing the Size and Colloidal State of Nanoparticles. Sci. Rep. 2015, 5, 9793. [Google Scholar] [CrossRef]
- Udenni Gunathilake, T.M.S.; Ching, Y.C.; Ching, K.Y.; Chuah, C.H.; Abdullah, L.C. Biomedical and Microbiological Applications of Bio-Based Porous Materials: A Review. Polymers 2017, 9, 160. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Liu, Y.; Pan, Y.; Miszuk, J.M.; Sun, H. One-Pot Porogen Free Method Fabricated Porous Microsphere-Aggregated 3D PCL Scaffolds for Bone Tissue Engineering. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 2699–2710. [Google Scholar] [CrossRef]
- Chen, P.; Zhou, Z.; Liu, W.; Zhao, Y.; Huang, T.; Li, X.; Duan, J.; Fang, J. Preparation and Characterization of Poly(L-Lactide-Co-Glycolide-Co-ε-Caprolactone) Scaffolds by Thermally Induced Phase Separation. J. Macromol. Sci. Part B Phys. 2020, 59, 427–439. [Google Scholar] [CrossRef]
- Zeinali, R.; Del Valle, L.J.; Torras, J.; Puiggalí, J. Recent Progress on Biodegradable Tissue Engineering Scaffolds Prepared by Thermally-Induced Phase Separation (Tips). Int. J. Mol. Sci. 2021, 22, 3504. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Okpokwasili, C.; Huang, Y.; Shi, X.; Wu, J.; Liao, J.; Tang, L.; Hong, Y. Optimizing Anisotropic Polyurethane Scaffolds to Mechanically Match with Native Myocardium. ACS Biomater. Sci. Eng. 2020, 6, 2757–2769. [Google Scholar] [CrossRef] [PubMed]
- Hebbar, R.S.; Isloor, A.M.; Ismail, A.F. Contact Angle Measurements. In Membrane Characterization; Elsevier: Amsterdam, The Netherlands, 2017; pp. 219–255. ISBN 9780444637918. [Google Scholar]
- Grant, N.; Zhang, H. Poorly Water-Soluble Drug Nanoparticles via an Emulsion-Freeze-Drying Approach. J. Colloid Interface Sci. 2011, 356, 573–578. [Google Scholar] [CrossRef]
- Callies, M.; Quéré, D. On Water Repellency. Soft Matter 2005, 1, 55–61. [Google Scholar] [CrossRef]
- Lm, S.J.; Kim, D.; Kim, Y.; Jeong, S.; Pang, C.; Ryu, S.; Weon, B.M. Hydrophobicity Evolution on Rough Surfaces. Langmuir 2020, 36, 689–696. [Google Scholar] [CrossRef]
- Siegel, S.J.; Kahn, J.B.; Metzger, K.; Winey, K.I.; Werner, K.; Dan, N. Effect of Drug Type on the Degradation Rate of PLGA Matrices. Eur. J. Pharm. Biopharm. 2006, 64, 287–293. [Google Scholar] [CrossRef]
- Visan, A.I.; Popescu-Pelin, G.; Socol, G. Degradation Behavior of Polymers Used as Coating Materials for Drug Delivery—A Basic Review. Polymers 2021, 13, 1272. [Google Scholar] [CrossRef]
- Xu, Y.; Kim, C.S.; Saylor, D.M.; Koo, D. Polymer Degradation and Drug Delivery in PLGA-Based Drug–Polymer Applications: A Review of Experiments and Theories. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 1692–1716. [Google Scholar] [CrossRef] [PubMed]
- López-Fernández, J.; Benaiges, M.D.; Valero, F. Rhizopus Oryzae Lipase, a Promising Industrial Enzyme: Biochemical Characteristics, Production and Biocatalytic Applications. Catalysts 2020, 10, 1277. [Google Scholar] [CrossRef]
- Merck, S.A. Lipase from Pseudomonas Cepacia Powder, Light Beige, ≥30 U/Mg. Available online: https://www.sigmaaldrich.com/GR/en/product/sigma/62309 (accessed on 24 December 2025).
- Liu, Y.; Yang, G.; Jin, S.; Xu, L.; Zhao, C.X. Development of High-Drug-Loading Nanoparticles. Chempluschem 2020, 85, 2143–2157. [Google Scholar] [CrossRef]
- Gupta, N.; Kumar Ray, P.; Mandal, S.; Mangavade, A.; Sharma, J.; Sahu, B.; Aparna, T.N. Design and Formulation of Dual-Action Nanoparticles for Synergistic Cancer Therapy. Front. Health Inform. 2024, 13, 2117–2134. [Google Scholar]
- Hou, D.; Gui, R.; Hu, S.; Huang, Y.; Feng, Z.; Ping, Q. Preparation and Characterization of Novel Drug-Inserted-Montmorillonite Chitosan Carriers for Ocular Drug Delivery. Adv. Nanopart. 2015, 4, 70–84. [Google Scholar] [CrossRef]
- Huang, W.; Tsui, C.P.; Tang, C.Y.; Gu, L. Effects of Compositional Tailoring on Drug Delivery Behaviours of Silica Xerogel/Polymer Core-Shell Composite Nanoparticles. Sci. Rep. 2018, 8, 13002. [Google Scholar] [CrossRef] [PubMed]
- Prajapat, P.; Agrawal, D.; Bhaduka, G. A Brief Overview of Sustained Released Drug Delivery System. J. Appl. Pharm. Res. 2022, 10, 5–11. [Google Scholar] [CrossRef]
- Adepu, S.; Ramakrishna, S. Controlled Drug Delivery Systems: Current Status and Future Directions. Molecules 2021, 26, 5905. [Google Scholar] [CrossRef]
- Patiwala, M.S.M.; Jethara, S.I.; Patel, M.R. Recent Trends in Sustained Release Oral Drug Delivery System: A Promising Approach. World J. Pharm. Res. 2015, 4, 526–552. [Google Scholar]
- Beg, S.; Swain, S.; Rizwan, M.; Irfanuddin, M.; Shobha Malini, D. Bioavailability Enhancement Strategies: Basics, Formulation Approaches and Regulatory Considerations. Curr. Drug Deliv. 2011, 8, 691–702. [Google Scholar] [CrossRef]
- Yoo, J.; Won, Y.Y. Phenomenology of the Initial Burst Release of Drugs from PLGA Microparticles. ACS Biomater. Sci. Eng. 2020, 6, 6053–6062. [Google Scholar] [CrossRef]
- Rodrigues de Azevedo, C.; von Stosch, M.; Costa, M.S.; Ramos, A.M.; Cardoso, M.M.; Danhier, F.; Préat, V.; Oliveira, R. Modeling of the Burst Release from PLGA Micro- and Nanoparticles as Function of Physicochemical Parameters and Formulation Characteristics. Int. J. Pharm. 2017, 532, 229–240. [Google Scholar] [CrossRef]
- Chakrala, J. Formulation and Characterization of Trigonella Foenum Graecum (Fenugreek) Seed Polysaccharide Nanoparticles Using Diltiazem HCl as Model Drug. Jyothsna Chakrala. World J. Pharm. Res. 2020, 9, 830–844. [Google Scholar]
- Allison, S.D. Analysis of Initial Burst in PLGA Microparticles. Expert Opin. Drug Deliv. 2008, 5, 615–628. [Google Scholar] [CrossRef]
- Rothstein, S.N.; Little, S.R. A “Tool Box” for Rational Design of Degradable Controlled Release Formulations. J. Mater. Chem. 2011, 21, 29–39. [Google Scholar] [CrossRef]
- Surekha, B.; Misra, P.; Thippaiah, A.C.; Shamanna, B.R.; Madathil, A.; Rajadurai, M. A Microneedle Transdermal Patch Loaded with Iron(Ii) Nanoparticles for Non-Invasive Sustained Delivery to Combat Anemia. Mater. Adv. 2024, 5, 3247–3256. [Google Scholar] [CrossRef]
- Michaelides, K.; Al Tahan, M.A.; Zhou, Y.; Trindade, G.F.; Cant, D.J.H.; Pei, Y.; Dulal, P.; Al-Khattawi, A. New Insights on the Burst Release Kinetics of Spray-Dried PLGA Microspheres. Mol. Pharm. 2024, 21, 6245–6256. [Google Scholar] [CrossRef]
- Khalbas, A.H.; Albayati, T.M.; Ali, N.S.; Salih, I.K. Drug Loading Methods and Kinetic Release Models Using of Mesoporous Silica Nanoparticles as a Drug Delivery System: A Review. S. Afr. J. Chem. Eng. 2024, 50, 261–280. [Google Scholar] [CrossRef]
- Porbaha, P.; Ansari, R.; Kiafar, M.R.; Bashiry, R.; Khazaei, M.M.; Dadbakhsh, A.; Azadi, A. A Comparative Mathematical Analysis of Drug Release from Lipid-Based Nanoparticles. AAPS PharmSciTech 2024, 25, 208. [Google Scholar] [CrossRef] [PubMed]
- Al-Ali, M.; Al-Ali, L.I. Modeling Kinetics and Transport Mechanism Study of Poorly Soluble Drug Formulation in High Acidic Medium. Tikrit J. Eng. Sci. 2024, 31, 76–91. [Google Scholar] [CrossRef]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release i. fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J. Control. Release 1987, 5, 23–36. [Google Scholar] [CrossRef]
- Corrigan, O.I.; Li, X. Quantifying Drug Release from PLGA Nanoparticulates. Eur. J. Pharm. Sci. 2009, 37, 477–485. [Google Scholar] [CrossRef]
- Ignacio, M.; Chubynsky, M.V.; Slater, G.W. Interpreting the Weibull Fitting Parameters for Diffusion-Controlled Release Data. Phys. A Stat. Mech. Its Appl. 2017, 486, 486–496. [Google Scholar] [CrossRef]












| Concentrations of the Components in the Final Produced Formulations | Sample Name |
|---|---|
| NIPU 0.3%—PVA 0.5%—neat | N1 |
| NIPU 0.5%—PVA 0.5%—neat | Ν2 |
| 0.2% PP, NIPU 0.3%—PVA 0.5% | NPP1 |
| 0.5% PP, NIPU 0.3%—PVA 0.5% | NPP2 |
| 1% PP, NIPU 0.3%—PVA 0.5% | NPP3 |
| 0.2% PP, NIPU 0.5%—PVA 0.5% | NPP4 |
| 0.5% PP, NIPU 0.5%—PVA 0.5% | NPP5 |
| 1% PP, NIPU 0.5%—PVA 0.5% | NPP6 |
| Sample | 1st Heating Scan | Cooling | 2nd Heating Scan | ||
|---|---|---|---|---|---|
| Tg (°C) | Tm (°C) | Tc (°C) | Tg (°C) | Tm (°C) | |
| N1 | −59 | 107 | - | −42 | - |
| NPP1 | −51 | 12, 53, (88) * | −14 | −25 | 9 |
| NPP2 | −52 | 12, 53, (92) * | −10 | −24 | 9 |
| NPP3 | - | 48 | −1 | −34 | 13, 40 |
| N2 | −54 | - | - | −39 | 90 |
| NPP4 | −49 | 12, 53, (94) * | −16 | −32 | 11 |
| NPP5 | −54 | 13, 52, (78) * | −7 | −33 | 13, 42 |
| NPP6 | −55 | 13, 52, (86) * | −10 | −26 | 10 |
| Sample Name | Average Max Stress (MPa) |
|---|---|
| N1 | 0.08229 ± 0.05 |
| N2 | 0.08216 ± 0.05 |
| Sample Name | z-Average (nm) | PDI |
|---|---|---|
| Non-loaded NIPU nanoparticles | ||
| N1 | 356 ± 180.10 | 0.493 |
| N2 | 364 ± 260.19 | 0.381 |
| Nanoparticles loaded with Paliperidone Palmitate | ||
| NPP1 | 420 ± 97.61 | 0.370 |
| NPP2 | 405 ± 324.40 | 0.318 |
| NPP3 | 1126 ± 309.65 | 0.491 |
| NPP4 | 708 ± 493.76 | 0.314 |
| NPP5 | 563 ± 455.76 | 0.268 |
| NPP6 | 1210 ± 389.01 | 0.248 |
| Sample Name | Porosity (%) |
|---|---|
| N1 | 87.37 ± 4.37 |
| N2 | 86.81 ± 4.24 |
| NPP1 | 86.53 ± 4.18 |
| NPP2 | 85.64 ± 4.37 |
| NPP3 | 81.55 ± 3.91 |
| NPP4 | 84.75 ± 4.24 |
| NPP5 | 80.80 ± 3.96 |
| NPP6 | 79.89 ± 3.95 |
| Sample Name | Average Initial θ (°) (t0 = 0 s) | Average Final θ (°) (tf = 0.95 s) | Average Δθ (°) (Measure, °) |
|---|---|---|---|
| NIPU neat | 56.7 | 55.1 | 1.6 ± 0.2 |
| NPP1 | 52.9 | 19.5 | 33.4 ± 6.2 |
| NPP2 | 48.7 | 14.6 | 34.1 ± 3.1 |
| NPP3 | 55.1 | 22.5 | 32.6 ± 2.0 |
| NPP4 | 54.0 | 23.9 | 30.1 ± 1.9 |
| NPP5 | 69.4 | 43.0 | 26.4 ± 1.4 |
| NPP6 | 60.2 | 38.6 | 21.6 ± 3.2 |
| Sample Name | % Drug Loading |
|---|---|
| NPP1 | 56.489 ± 2.82 |
| NPP2 | 56.905 ± 2.84 |
| NPP3 | 76.681 ± 3.83 |
| NPP4 | 77.950 ± 3.9 |
| NPP5 | 48.383 ± 2.42 |
| NPP6 | 77.798 ± 3.89 |
| Formulation | R1 | R2 | k1 (1/Days) | k2 (1/Days) | tm (Days) |
|---|---|---|---|---|---|
| NPP1 | 33 | 65 | 10 | 0.8 | 0.75 |
| NPP2 | 40 | 60 | 12 | 0.6 | 0.333 |
| NPP3 | 75 | 23 | 14 | 1 | 0.333 |
| NPP4 | 52 | 48 | 7 | 0.8 | 0.333 |
| NPP5 | 35 | 63 | 20 | 0.7 | 0.167 |
| NPP6 | 70 | 25 | 45 | 3 | 0.0833 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ntrivala, M.A.; Balla, E.; Christodoulou, E.P.; Kostoglou, M.; Klonos, P.; Kyritsis, A.; Bikiaris, D.N. Sustained Delivery of Paliperidone Palmitate via Encapsulation in Bio-Based NIPU Nanoparticles. Polymers 2026, 18, 920. https://doi.org/10.3390/polym18080920
Ntrivala MA, Balla E, Christodoulou EP, Kostoglou M, Klonos P, Kyritsis A, Bikiaris DN. Sustained Delivery of Paliperidone Palmitate via Encapsulation in Bio-Based NIPU Nanoparticles. Polymers. 2026; 18(8):920. https://doi.org/10.3390/polym18080920
Chicago/Turabian StyleNtrivala, Maria Angeliki, Evangelia Balla, Ermis P. Christodoulou, Margaritis Kostoglou, Panagiotis Klonos, Apostolos Kyritsis, and Dimitrios N. Bikiaris. 2026. "Sustained Delivery of Paliperidone Palmitate via Encapsulation in Bio-Based NIPU Nanoparticles" Polymers 18, no. 8: 920. https://doi.org/10.3390/polym18080920
APA StyleNtrivala, M. A., Balla, E., Christodoulou, E. P., Kostoglou, M., Klonos, P., Kyritsis, A., & Bikiaris, D. N. (2026). Sustained Delivery of Paliperidone Palmitate via Encapsulation in Bio-Based NIPU Nanoparticles. Polymers, 18(8), 920. https://doi.org/10.3390/polym18080920










