Electrochemically Deposited Ag/PANI on ITO: Non-Monotonic Disorder–Dispersion Coupling and Enhanced Third-Order Optical Nonlinearity
Abstract
1. Introduction
2. Experimental Methods
2.1. Chemicals
2.2. Thin-Film Depositions
2.3. Evaluation of Optical and Structural Properties
3. Results and Discussion
3.1. Nanocomposites and Film Deposition
3.2. XRF Characterization
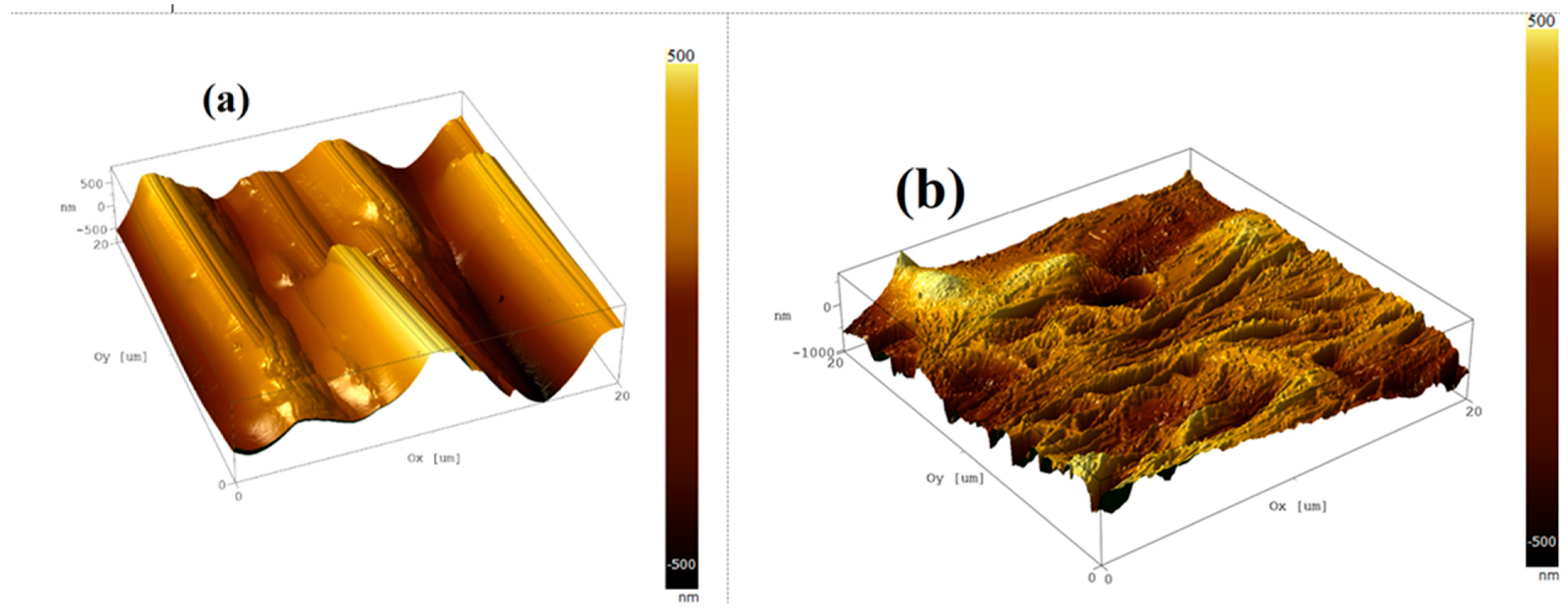
3.3. Atomic Force Microscopy (AFM) Characterization
3.4. Optical Properties
- Optical constants: Extinction coefficient (K) and refractive index (n)
- Optical Anisotropy and Birefringence Analysis
- The optical conductivity
3.5. Dielectric Properties
3.5.1. Wavelength-Dependent Dielectric Response
3.5.2. The Dielectric Loss Tangent (tan δ)
3.5.3. Nonlinear Optical Parameters
3.6. FTIR Spectra
3.7. Electrical Conductivity
3.8. Surface Morphology
3.9. XRD Analysis
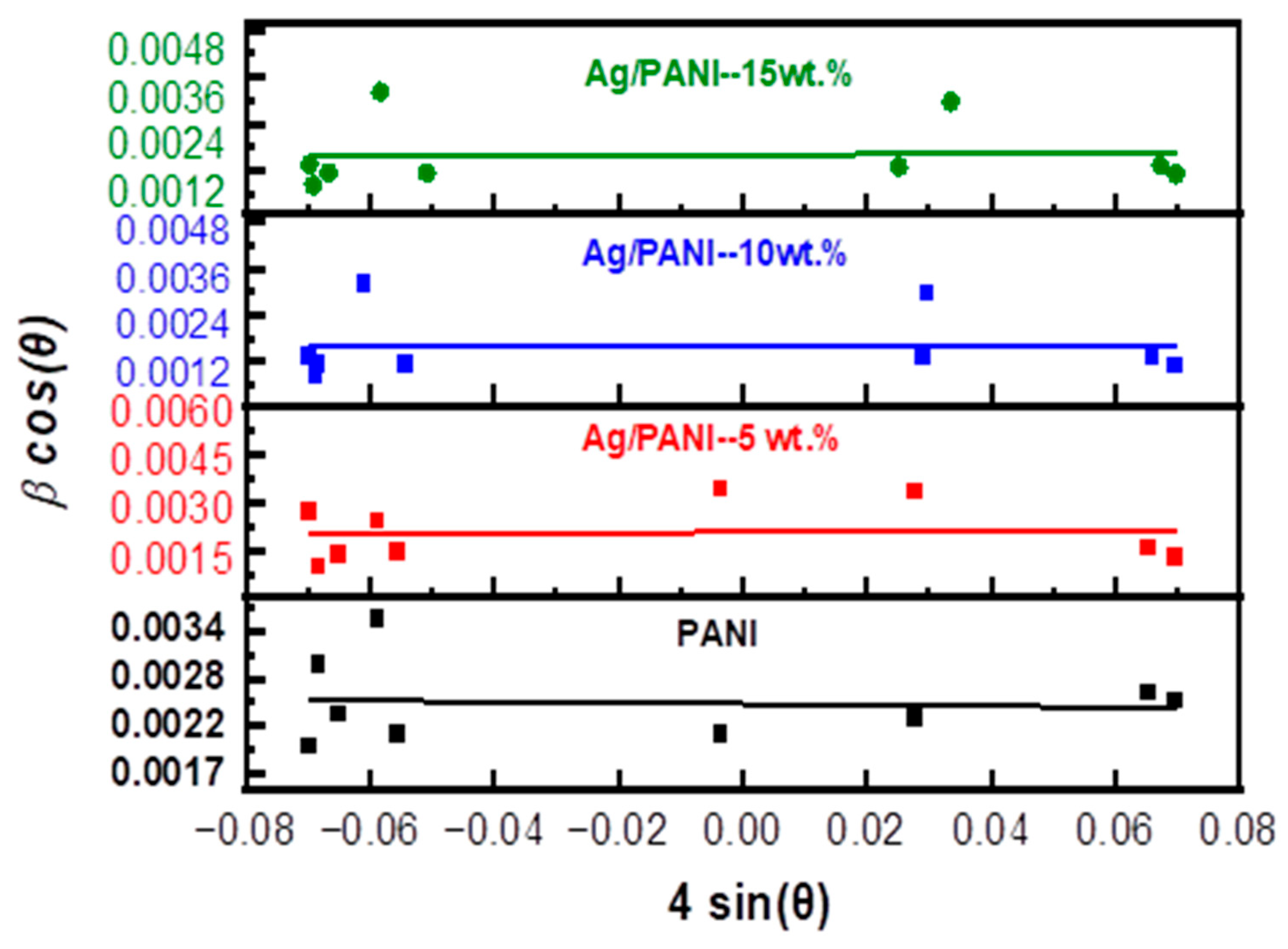
Williamson–Hall (W–H) Microstructural Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A. Supporting Information and Data
Appendix A.1. Atomic Force Microscopy (AFM)
| Sample | Ra (nm) | Rq (nm) | Rz (nm) |
|---|---|---|---|
| PANI (pristine) | 240.088 | 303.869 | 2283.39 |
| Ag/PANI (15 wt.%) | 637 | 830 | 4740 |
Appendix A.2. AFM Height-Distribution Histogram of the Pristine PANI Ag/PANI (15 wt.%) Nanocomposite Film

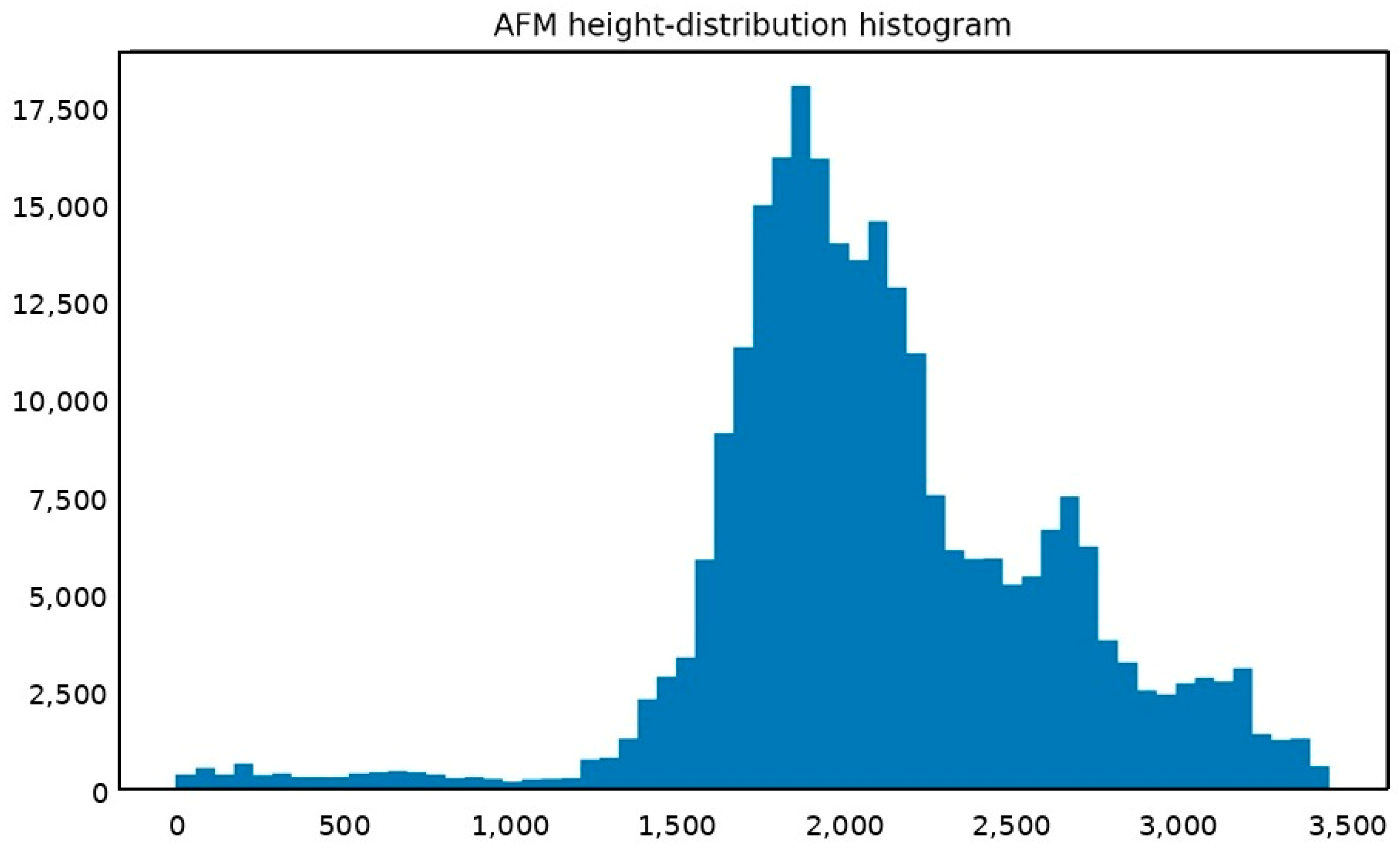
Appendix A.3. XRD Analysis
| Sample | 2θ (deg) | d (Å) | FWHM (deg) | Crystallite Size D (nm) | Phase | hkl |
|---|---|---|---|---|---|---|
| PANI film | 9.29 | 9.513 | 0.0920 | 86.6 | PANI (emeraldine salt) | |
| PANI film | 15.63 | 5.665 | 0.1332 | 60.2 | PANI (emeraldine salt) | |
| PANI film | 18.28 | 4.848 | 0.1151 | 73.3 | PANI (emeraldine salt) | (110) |
| PANI film | 22.56 | 3.938 | 0.1361 | 62.3 | PANI (emeraldine salt) | (200) |
| PANI film | 27.43 | 3.250 | 0.1288 | 63.5 | PANI (emeraldine salt) | |
| Ag/PANI 5 wt.% | 9.44 | 9.365 | 0.0900 | 88.6 | PANI (emeraldine salt) | |
| Ag/PANI 5 wt.% | 15.63 | 5.665 | 0.1310 | 61.2 | PANI (emeraldine salt) | |
| Ag/PANI 5 wt.% | 18.43 | 4.810 | 0.0803 | 100.2 | PANI (emeraldine salt) | (110) |
| Ag/PANI 5 wt.% | 22.56 | 3.938 | 0.1537 | 52.7 | PANI (emeraldine salt) | (200) |
| Ag/PANI 5 wt.% | 38.63 | 2.329 | 0.0744 | 113.2 | fcc Ag | (111) |
| Ag/PANI 5 wt.% | 44.82 | 2.020 | 0.0948 | 90.6 | fcc Ag | (200) |
| Ag/PANI 10 wt.% | 9.29 | 9.513 | 0.0936 | 85.2 | PANI (emeraldine salt) | |
| Ag/PANI 10 wt.% | 15.78 | 5.613 | 0.1146 | 70.0 | PANI (emeraldine salt) | |
| Ag/PANI 10 wt.% | 18.43 | 4.810 | 0.1677 | 48.0 | PANI (emeraldine salt) | (110) |
| Ag/PANI 10 wt.% | 22.56 | 3.938 | 0.1750 | 46.3 | PANI (emeraldine salt) | (200) |
| Ag/PANI 10 wt.% | 27.57 | 3.232 | 0.1623 | 50.4 | PANI (emeraldine salt) | |
| Ag/PANI 10 wt.% | 38.63 | 2.329 | 0.0866 | 97.2 | fcc Ag | (111) |
| Ag/PANI 10 wt.% | 44.82 | 2.020 | 0.0846 | 101.6 | fcc Ag | (200) |
| Ag/PANI 15 wt.% | 9.44 | 9.365 | 0.0877 | 90.9 | PANI (emeraldine salt) | |
| Ag/PANI 15 wt.% | 15.78 | 5.613 | 0.1204 | 66.6 | PANI (emeraldine salt) | |
| Ag/PANI 15 wt.% | 18.58 | 4.772 | 0.1078 | 74.7 | PANI (emeraldine salt) | (110) |
| Ag/PANI 15 wt.% | 22.85 | 3.888 | 0.1326 | 61.1 | PANI (emeraldine salt) | (200) |
| Ag/PANI 15 wt.% | 27.72 | 3.216 | 0.0908 | 90.1 | PANI (emeraldine salt) | |
| Ag/PANI 15 wt.% | 38.78 | 2.320 | 0.0762 | 110.5 | fcc Ag | (111) |
| Ag/PANI 15 wt.% | 44.97 | 2.014 | 0.0810 | 106.1 | fcc Ag | (200) |
References
- Grosso, D.; Ribot, F.; Boissiere, C.; Sanchez, C. Molecular and supramolecular dynamics of hybrid organic-inorganic interfaces for the rational construction of advanced hybrid nanomaterials. Chem. Soc. Rev. 2011, 40, 829–848. [Google Scholar] [CrossRef] [PubMed]
- Algidsawi, A.J.K.; Hashim, A.; Hadi, A.; Habeeb, M.A. Exploring the characteristics of SnO2 nanoparticles doped organic blend for low cost nanoelectronics applications. Semicond. Phys. Quantum Electron. Optoelectron. 2021, 24, 472–477. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Kumar, B.G.; Rajendran, S.; Qin, J.; Vadivel, S.; Durgalakshmi, D.; Karimi, F. Tuning of metal oxides photocatalytic performance using Ag nanoparticles integration. J. Mol. Liq. 2020, 314, 113588. [Google Scholar] [CrossRef]
- AlZoubi, T.; Kadhem, W.J.; Al Gharram, M.; Makhadmeh, G.; Abdelfattah, M.A.; Abuelsamen, A.; Mourched, B. Advanced Optoelectronic Modeling and Optimization of HTL-Free FASnI3/C60 Perovskite Solar Cell Architecture for Superior Performance. Nanomaterials 2024, 14, 1062. [Google Scholar] [CrossRef]
- AlZoubi, T.; Mourched, B.; Al Gharram, M.; Makhadmeh, G.; Abu Noqta, O. Improving photovoltaic performance of hybrid organic-inorganic MAGeI3 perovskite solar cells via numerical optimization of carrier transport materials (HTLs/ETLs). Nanomaterials 2023, 13, 2221. [Google Scholar] [CrossRef] [PubMed]
- Suriya, P.; Prabhu, M.; Jagannathan, K. Synthesis and structural, optical and photovoltaic characteristics of pure and Ag doped TiO2 nanoparticles for dye sensitized solar cell application. Mater. Today Proc. 2022, 65, 100–105. [Google Scholar] [CrossRef]
- Al Zoubi, T.; Al-Gharram, M.; Moustafa, M. Insights into the impact of defect states and temperature on the performance of kesterite-based thin-film solar cells. Optik 2022, 264, 169442. [Google Scholar] [CrossRef]
- Oreibi, I.; Ali Habeeb, M.; Abdul Hamza, R.S. Polymer nanocomposites comprising PVA matrix and Ag–BaTiO3 nanofillers: A comparative study of structural, dielectric and optical characteristics for optics and quantum nanoelectronic applications. Opt. Quantum Electron. 2024, 56, 119. [Google Scholar] [CrossRef]
- Parvatikar, N.; Jain, S.; Bhoraskar, S.V.; Ambika Prasad, M.V.N. Spectroscopic and electrical properties of polyaniline/CeO2 composites and their application as humidity sensor. J. Appl. Polym. Sci. 2006, 102, 5533–5537. [Google Scholar] [CrossRef]
- Al-Gharram, M.; Uhlmann, P.; Al-Hussein, M. Highly dispersed crystalline magnetic and conductive polyaniline/iron oxide nanocomposite films. Colloids Surf. A Physicochem. Eng. Asp. 2024, 684, 133212. [Google Scholar] [CrossRef]
- Frackowiak, E.; Khomenko, V.; Jurewicz, K.; Lota, K.; Béguin, F. Supercapacitors based on conducting polymers/nanotubes composites. J. Power Sources 2006, 153, 413–418. [Google Scholar] [CrossRef]
- Wang, L.; Wu, H.; Chen, L. Polyaniline-Al2O3 nanocomposites for electronic applications. J. Mater. Sci. 2018, 53, 10946–10955. [Google Scholar] [CrossRef]
- Dey, A.; De, S.; De, A.; De, S.K. Characterization and dielectric properties of polyaniline–TiO2 nanocomposites. Nanotechnology 2004, 15, 1277. [Google Scholar] [CrossRef]
- Ahmadi, F. Silver Nanoparticle Enhanced the Optical Properties of the Rare Earth Doped Megnesium Zinc Sulfophosphate Glass. Ph.D. Thesis, University of Technology Malaysia, Johor, Malaysia, 2017. [Google Scholar]
- Somani, P.R.; Marimuthu, R.; Mandale, A.B. Synthesis, characterization and charge transport mechanism in conducting polyaniline/V2O5 composites. Polymer 2001, 42, 2991–3001. [Google Scholar] [CrossRef]
- Bhadra, S.; Singha, N.K.; Khastgir, D. Polyaniline by new miniemulsion polymerization and the effect of reducing agent on conductivity. Synth. Met. 2006, 156, 1148–1154. [Google Scholar] [CrossRef]
- Majeed Khan, M.A.; Kumar, S.; Ahamed, M.; Alrokayan, S.A.; AlSalhi, M.S. Structural and thermal studies of silver nanoparticles and electrical transport study of their thin films. Nanoscale Res. Lett. 2011, 6, 434. [Google Scholar] [CrossRef]
- Basavaiah, K.; Kumar, Y.P.; Rao, A.V.P. A facile one-pot synthesis of polyaniline/magnetite nanocomposites by micelles-assisted method. Appl. Nanosci. 2012, 3, 409–415. [Google Scholar] [CrossRef]
- AlGharram, M.; AlZoubi, T. Controlled Electrochemical Deposition of Mo/PANI Nanocomposites: Insights into Bandgap Narrowing and DC Charge-Transport in MoOx/Polyaniline Thin Films. Ceram. Int. 2025, in press. [Google Scholar]
- Alhazime, A.A.; Benthami, K.; Alsobhi, B.O.; Ali, G.W.; Nouh, S.A. Pani-Ag/PVA nanocomposite: Gamma induced changes in the thermal and optical characteristics. J. Vinyl Addit. Technol. 2020, 27, 47–53. [Google Scholar] [CrossRef]
- Allen, J.P.; Scanlon, D.O.; Watson, G.W. Electronic structures of silver oxides. Phys. Rev. B 2011, 84, 115141. [Google Scholar] [CrossRef]
- Xiao, Q.; Fan, L.H.; Ma, Q.; Ning, Y.M.; Gu, Z.; Chen, L.; Cui, T.J. Secure wireless communication of brain–computer interface and mind control of smart devices enabled by space-time-coding metasurface. Nat. Commun. 2025, 16, 7914. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Wang, Z.; Fan, Y.; Zhang, K.; Lin, R.; Wu, J.; Tsai, D.P. Intensity-asymmetric wavefront shaping in nonlocal meta-lens. Nat. Commun. 2026. [Google Scholar] [CrossRef]
- Tamboli, M.S.; Kulkarni, M.V.; Patil, R.H.; Gade, W.N.; Navale, S.C.; Kale, B.B. Nanowires of silver–polyaniline nanocomposite synthesized via in situ polymerization and its novel functionality as an antibacterial agent. Colloids Surf. B Biointerfaces 2012, 92, 35–41. [Google Scholar] [CrossRef]
- Al-Gharram, M.; AlZoubi, T. Electrochemical synthesis of a novel hybrid nanocomposite based on Co3O4 nanoparticles embedded in PANI-camphor sulfonic acid matrix for optoelectronic applications. Ceram. Int. 2024, 50, 5473–5482. [Google Scholar] [CrossRef]
- Alzoubi, F.; Al-Gharram, M.; AlZoubi, T.; Noqta, O.A.; Makhadmeh, G.; Al-Khateeb, H.; Al-Qadi, M. Advanced electrochemical synthesis and characterization of Al2O3 nanoparticles embedded in polyaniline matrix for optoelectronic applications. Ceram. Int. 2024, 50, 37968–37977. [Google Scholar] [CrossRef]
- Al-Gharram, M.; Jum’h, I.; Telfah, A.; Al-Hussein, M. Highly crystalline conductive electrodeposited films of PANI-CSA/CoFe2O4 nanocomposites. Colloids Surf. A Physicochem. Eng. Asp. 2021, 628, 127342. [Google Scholar] [CrossRef]
- Revenko, A.G.; Tsvetyansky, A.L.; Eritenko, A.N. X-ray fluorescence analysis of solid-state films, layers, and coatings. Radiat. Phys. Chem. 2022, 197, 110157. [Google Scholar] [CrossRef]
- Al-Gharram, M.; AlZoubi, T.; Mandumpal, J. Electrochemical Fabrication and Characterization of CeO2/PANI Nanocomposite with Enhanced Optoelectronic Performance. Results Eng. 2025, 27, 106305. [Google Scholar] [CrossRef]
- Tauc, J. Amorphous and Liquid Semiconductors; Springer: Boston, MA, USA, 1974. [Google Scholar]
- Duan, C.; Wang, H.; Ou, X.; Li, F.; Zhang, X. Efficient visible light photocatalyst fabricated by depositing plasmonic Ag nanoparticles on conductive polymer-protected Si nanowire arrays for photoelectrochemical hydrogen generation. ACS Appl. Mater. Interfaces 2014, 6, 9742–9750. [Google Scholar] [CrossRef]
- Kwon, J.; Shin, Y.; Sung, Y.; Doh, H.; Kim, S. Silver sulfide nanocrystals and their photodetector applications. Accounts Mater. Res. 2024, 5, 1097–1108. [Google Scholar] [CrossRef]
- Kanis, D.R.; Ratner, M.A.; Marks, T.J.; Zerner, M.C. Nonlinear optical characteristics of novel inorganic chromophores using the Zindo formalism. Chem. Mater. 1991, 3, 19–22. [Google Scholar] [CrossRef]
- Henaish, A.M.A.; Salem, B.I.; Meaz, T.M.; Alibwaini, Y.A.; Ajlouni, A.W.; Hemeda, O.M.; Arrasheed, E.A. Synthesize, characterization, dielectric, linear and nonlinear optical properties of Ni–Al ferrite/PANI nanocomposite film. Opt. Mater. 2021, 119, 111397. [Google Scholar] [CrossRef]
- Jilani, A.; Othman, M.H.D.; Ansari, M.O.; Khan, I.U.; Hussain, S.Z. Linear/nonlinear optical susceptibility spectroscopic constants of polyaniline@graphene oxide nanocomposite thin films. Synth. Met. 2019, 251, 30–39. [Google Scholar] [CrossRef]
- He, K.; Lu, J.; Ma, X.; Ju, Y.; Xie, L.; Pang, L.; Chen, J. Effect of Maxwell–Wagner relaxation on field charging of particles. Aerosol Sci. Technol. 2015, 49, 1210–1221. [Google Scholar] [CrossRef]
- Palaniappan, S. Chemical and electrochemical polymerization of aniline using tartaric acid. Eur. Polym. J. 2001, 37, 975–981. [Google Scholar] [CrossRef]
- Gupta, K.; Jana, P.C.; Meikap, A.K. Optical and electrical transport properties of polyaniline–silver nanocomposite. Synth. Met. 2010, 160, 1566–1573. [Google Scholar] [CrossRef]
- Verma, A.; Kumar, T. Ag/Cu doped polyaniline hybrid nanocomposite-based novel gas sensor for enhanced ammonia gas sensing performance at room temperature. RSC Adv. 2024, 14, 25093–25107. [Google Scholar] [CrossRef]
- Al-Gharram, M.; AlZoubi, T. Linear and nonlinear optical properties of hybrid Polyaniline/CoFe2O4 nanocomposites: Electrochemical synthesis, characterization, and analysis. Ceram. Int. 2024, 50, 32841–32852. [Google Scholar] [CrossRef]
- Sakr, G.B.; Yahia, I.S.; Fadel, M.; Fouad, S.S.; Romcevic, N. Optical spectroscopy, optical conductivity, dielectric properties and new methods for determining the gap states of CuSe thin films. J. Alloys Compd. 2010, 507, 557–562. [Google Scholar] [CrossRef]
- Alibwaini, Y.A.; Hemeda, O.M.; El-Shater, R.; Sharshar, T.; Ashour, A.H.; Ajlouni, A.W.; Henaish, A.M.A. Synthesis, characterizations, optical and photoluminescence properties of polymer blend PVA/PEG films doped eosin Y (EY) dye. Opt. Mater. 2021, 111, 110600. [Google Scholar] [CrossRef]
- Benyoucef, M.; Usman, M.A.; Alzoubi, T.; Reithmaier, J.P. Pre-patterned silicon substrates for the growth of III–V nanostructures. Phys. Status Solidi A 2012, 209, 2402–2410. [Google Scholar] [CrossRef]
- Paulraj, P.; Umar, A.; Rajendran, K.; Manikandan, A.; Kumar, R.; Manikandan, E.; Baskoutas, S. Solid-state synthesis of Ag-doped PANI nanocomposites for their end-use as an electrochemical sensor for hydrogen peroxide and dopamine. Electrochim. Acta 2020, 363, 137158. [Google Scholar] [CrossRef]
- Moustafa, M.; Laouini, G.; Alzoubi, T. Finite element analysis of SAW sensor with ZnO substrate for dichloromethane (DCM) gas detection. Arch. Acoust. 2021, 46, 419–426. [Google Scholar] [CrossRef]
- Alzoubi, F.; Al-Gharram, M.; AlZoubi, T.; Al-Khateeb, H.; Al-Qadi, M.; Abu Noqta, O.; Mandumpal, J. Tailoring CuO/Polyaniline Nanocomposites for Optoelectronic Applications: Synthesis, Characterization, and Performance Analysis. Polymers 2025, 17, 1423. [Google Scholar] [CrossRef]
- Tichá, H.; Tichy, L. Semiempirical relation between non-linear susceptibility (refractive index), linear refractive index and optical gap and its application to amorphous chalcogenides. J. Optoelectron. Adv. Mater. 2002, 4, 381–386. [Google Scholar]
- Al-Gharram, M.; AlZoubi, T. Innovative quartz balance technique for vacancies analysis in palladium-hydrogen system. Ceram. Int. 2025, 51, 31150–31161. [Google Scholar] [CrossRef]
- Al-Gharram, M.; Jum’h, I.; Telfah, A.; Al-Hussein, M. PANI-CSA/Co3O4 nanocomposite films: Optical, morphological, and structural properties. In Proceedings of the 2nd International Conference on Industry 4.0 and Artificial Intelligence (ICIAI 2021); Atlantis Press: Dordrecht, The Netherlands, 2022; pp. 48–52. [Google Scholar]
- AlZoubi, T.; Al-Gharram, M.; Makhadmeh, G.; Noqta, O.A. Comparative analysis of optoelectronic and structural characteristics in electrochemically synthesized hybrid-nanocomposites based on PANI-CSA/metal oxide nanoparticles. Phys. B Condens. Matter 2025, 713, 417380. [Google Scholar] [CrossRef]
- Sathiyanarayanan, S.; Muthukrishnan, S.; Venkatachari, G.; Trivedi, D.C. Corrosion protection of steel by polyaniline (PANI) pigmented paint coating. Prog. Org. Coat. 2005, 53, 297–301. [Google Scholar] [CrossRef]
- Trchová, M.; Stejskal, J. Polyaniline: The infrared spectroscopy of conducting polymer nanotubes (IUPAC Technical Report). Pure Appl. Chem. 2011, 83, 1803–1817. [Google Scholar] [CrossRef]
- Azhakanantham, D.; Al Zoubi, T.; AlGharram, M.; Al-Hmoud, M.; Ibnauof, K.H.; Aldaghri, O.; Kesavan, A.V. Forthright synthesis of titanium-based MXene nanosheets for bio-synaptic device applications: A novel approach. Ceram. Int. 2025, 51, 259–266. [Google Scholar] [CrossRef]
- Nagaraja, M.; Pattar, J.; Shashank, N.; Manjanna, J.; Kamada, Y.; Rajanna, K.; Mahesh, H.M. Electrical, structural and magnetic properties of polyaniline/pTSA-TiO2 nanocomposites. Synth. Met. 2009, 159, 718–722. [Google Scholar] [CrossRef]
- Mišurović, J.; Mojović, M.; Marjanović, B.; Vulić, P.; Ćirić-Marjanović, G. Magnetite nanoparticles-catalysed synthesis of conductive polyaniline. Synth. Met. 2019, 257, 116174. [Google Scholar] [CrossRef]
- Al-Gharram, M.; AlZoubi, T.; Noqta, O.A.; Makhadmeh, G.N. Comprehensive Study of Linear and Nonlinear Optical Properties of Polyaniline-Al2O3 Nanocomposites for Optoelectronic Applications. Surf. Interfaces 2024, 52, 104939. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Li, X.; Yang, B. Nanostructured conducting polymers: Synthesis, morphology, and applications. Adv. Mater. 2018, 30, 1706574. [Google Scholar]
- Al-Gharram, M.; AlZoubi, T. Exploring dual optical responses of polyaniline-Fe2O3 nanocomposites for advanced optoelectronic and supercapacitor applications. Ceram. Int. 2025, in press. [Google Scholar] [CrossRef]
- Lee, Y.H.; Kim, S.H. Enhanced electrochemical performance of conducting polymer nanocomposites through nanoparticle dispersion. Electrochim. Acta 2019, 306, 606–615. [Google Scholar]
- Burdușel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoantă, L.; Ficai, A.; Andronescu, E. Biomedical applications of silver nanoparticles: An up-to-date overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef]
- Ji, X.; Xu, Y.; Zhang, W.; Cui, L.; Liu, J. Review of functionalization, structure and properties of graphene/polymer composite fibers. Compos. Part A Appl. Sci. Manuf. 2016, 87, 29–45. [Google Scholar] [CrossRef]
- Qiu, L.; Zhang, J.; Yang, X. Structural control and enhanced charge transport in nanocomposite materials for energy storage. J. Mater. Chem. A 2021, 9, 14444–14458. [Google Scholar]
- Al-Zoubi, T.; Usman, M.A.; Benyoucef, M.; Reithmaier, J.P. Growth of InAs quantum dots and dashes on silicon substrates: Formation and characterization. J. Cryst. Growth 2011, 323, 422–425. [Google Scholar] [CrossRef]
- Al-Hmoud, M.; Gul, B.; Khan, M.S.; AlGharram, M.; Aziz, S.M.; Al Zoubi, T.; Ahmed, A.M. Insight into novel ErMI (M = S, Se) chalcohalide materials for next-generation energy applications. RSC Adv. 2025, 15, 34808–34820. [Google Scholar] [CrossRef]
- Khairy, M. Synthesis, characterization, magnetic and electrical properties of polyaniline/NiFe2O4 nanocompo-site. Synth. Met. 2014, 189, 34–41. [Google Scholar] [CrossRef]














| Sample | Indirect Energy Gap (Eg) (eV) | Urbach Energy (meV) | Steepness Parameter (γ) (eV−1) | Strength of Electron–Phonon (Ee_p) (eV) |
|---|---|---|---|---|
| PANI | 1.98 | 377 | 0.06 | 10.7 |
| Ag/PANI—5 wt.% | 1.81 | 1280 | 0.02 | 36.2 |
| Ag/PANI—10 wt.% | 1.38 | 1640 | 0.01 | 46.4 |
| Ag/PANI—15 wt.% | 1.19 | Not reliable | *** | *** |
| Sample | Average Refractive Index (n̄ ) | Birefringence (Δn) | Estimated ne (max n) | Estimated n0 (min n) |
|---|---|---|---|---|
| PANI | 2.228 | 0.475 | 2.545 | 2.070 |
| Ag/PANI—5 wt.% | 5.133 | 4.591 | 8.194 | 3.603 |
| Ag/PANI—10 wt.% | 2.924 | 2.975 | 4.907 | 1.932 |
| Ag/PANI—15 wt.% | 6.556 | 9.410 | 12.829 | 3.419 |
| Sample | Ed (eV) | E0 (eV) | f (eV)2 | n0 | εs | M−1 (ev)−2 | M−3 (ev)−2 |
|---|---|---|---|---|---|---|---|
| PANI | 4.81 | 1.92 | 9.23 | 1.18 | 1.40 | 2.51 | 0.68 |
| Ag/PANI—5 wt.% | 7.19 | 1.56 | 11.25 | 1.10 | 1.22 | 4.60 | 1.88 |
| Ag/PANI—10 wt.% | 2.58 | 1.79 | 4.63 | 1.30 | 1.69 | 1.44 | 0.45 |
| Ag/PANI—15 wt.% | 24.3 | 5.27 | 128.21 | 1.10 | 1.22 | 4.60 | 0.17 |
| Sample | χ1 | χ3 (e.s.u. × 10−9) | n2 (e.s.u. × 10−7) |
|---|---|---|---|
| PANI | 0.2 | 6.73 | 2.15 |
| Ag/PANI—5 wt.% | 0.4 | 75.82 | 25.91 |
| Ag/PANI—10 wt.% | 0.1 | 0.73 | 0.21 |
| Ag/PANI—15 wt.% | 0.4 | 76.56 | 0.26 |
| Sample | (nm) | ||||
|---|---|---|---|---|---|
| PANI | 1.6 | 8.2 | 4.8 | 9.1 | 220 |
| Ag/PANI—5 wt.% | 27.0 | 193.1 | 78.2 | 0.7 | 790 |
| Ag/PANI—10 wt.% | 21.7 | 108.6 | 62.9 | 0.3 | 690 |
| Ag/PANI—15 wt.% | 98.0 | 469.8 | 248 | 4.8 | 440 |
| Sample | Average Crystalline Grain Size (D) (nm) | Strain (ε × 10−4) | Dislocation Density (δ × 1013 m−2) | Strain Energy Density (Us) (J/m3) [62,63] |
|---|---|---|---|---|
| PANI | 58 | 6.47 | 29.7 | 5.23 × 102 |
| Ag/PANI—5 wt.% | 69 | 10.8 | 21.9 | 1.46 × 103 |
| Ag/PANI—10 wt.% | 91 | 2.8 | 12.2 | 9.80 × 101 |
| Ag/PANI—15 wt.% | 92 | 5.6 | 12.1 | 3.92 × 102 |
| Sample | Ref. | Indirect Energy Gap (Eg) (eV) | Urbach Energy (meV) | Strain (ε) |
|---|---|---|---|---|
| PANI | This work | 1.98 | 377 | 6.47 × 10−4 |
| Ag/PANI—5 wt.% | This work | 1.81 | 1280 | 10.8 × 10−4 |
| Ag/PANI—10 wt.% | This work | 1.38 | 1640 | 2.8 × 10−4 |
| Ag/PANI—15 wt.% | This work | 1.19 | Not reliable | 5.6 × 10−4 |
| PANI/NiO—5 wt.% | [64] | 3.45 | 290 | 13.5 × 10−4 |
| PANI/TiO2—10 wt.% | [64] | 3.53 | 240 | 5.90 × 10−4 |
| PANI/NiFe2O4—15 wt.% | [65] | 3.60 | 220 | 5.00 × 10−4 |
| PANI/Al2O3—5 wt.% | [56] | 3.76 | 463 | 3.6 × 10−4 |
| PANI/Al2O3—10 wt.% | [56] | 3.79 | 470 | 4.9 × 10−5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
AlGharram, M.; AlZoubi, T.; Makableh, Y.; Mouhtady, O. Electrochemically Deposited Ag/PANI on ITO: Non-Monotonic Disorder–Dispersion Coupling and Enhanced Third-Order Optical Nonlinearity. Polymers 2026, 18, 864. https://doi.org/10.3390/polym18070864
AlGharram M, AlZoubi T, Makableh Y, Mouhtady O. Electrochemically Deposited Ag/PANI on ITO: Non-Monotonic Disorder–Dispersion Coupling and Enhanced Third-Order Optical Nonlinearity. Polymers. 2026; 18(7):864. https://doi.org/10.3390/polym18070864
Chicago/Turabian StyleAlGharram, Mahmoud, Tariq AlZoubi, Yahia Makableh, and Omar Mouhtady. 2026. "Electrochemically Deposited Ag/PANI on ITO: Non-Monotonic Disorder–Dispersion Coupling and Enhanced Third-Order Optical Nonlinearity" Polymers 18, no. 7: 864. https://doi.org/10.3390/polym18070864
APA StyleAlGharram, M., AlZoubi, T., Makableh, Y., & Mouhtady, O. (2026). Electrochemically Deposited Ag/PANI on ITO: Non-Monotonic Disorder–Dispersion Coupling and Enhanced Third-Order Optical Nonlinearity. Polymers, 18(7), 864. https://doi.org/10.3390/polym18070864






