Bioprinting in Tissue Repair and Its ENT Applications
Abstract
1. Introduction
2. Biomaterials Used in ENT Tissue Regeneration
2.1. Carbohydrate Homopolymers
2.1.1. Cellulose
2.1.2. Starch
2.2. Carbohydrate Heteropolymers
2.2.1. Glycosaminoglycans (GAGs)
2.2.2. Alginate
2.2.3. Gellan Gum (GG)
2.2.4. Chitosan
2.3. Peptides and Proteins
2.3.1. Collagen
2.3.2. Gelatin
2.3.3. Silk Proteins
3. Cell Types in Tissue Regeneration
3.1. Stem Cells
3.2. Mature Cells
3.3. Synthetic (Artificial) Cells (SCs)
4. Bio-Printing and Engineering
4.1. Three-Dimensional Bioprinting
4.2. Four-Dimensional Bioprinting
4.3. Three-Dimensional/Four-Dimensional Bioprinting Applications in Otolaryngology
4.3.1. Nasal Reconstruction
4.3.2. Ear Reconstruction
4.3.3. Tracheal Reconstruction
5. Medical Applications in ENT Regenerative Medicine: Discussions and Future Perspectives
- Long-term viability of the bioimplant. While bioprinted tissue has generally demonstrated good viability in clinical settings, the current literature lacks reports of procedural failures or cases with graft necrosis resulting from viability loss. Consequently, the absence of negative data precludes a truly objective assessment of the long-term success rates of these neotransplants.
- Resistance to environmental factors (the extreme cold and heat in winter and summer) and minor trauma is a critical consideration, as the nose and auricles are anatomically prominent and frequently exposed to these stressors. These aspects were not quantitatively assessed in either successful clinical cases or animal models. Future research should prioritize evaluating the durability of bioprinted constructs, initially under controlled experimental conditions and subsequently in patients exposed to real-world environmental challenges.
- Age-related modeling and senescence of bioprinted tissues. The long-term behavior of neotissues during the natural biological aging process remains unknown. Due to the novelty of these procedures, sufficient longitudinal data are not yet available to draw definitive conclusions. Further research in this regard is essential to ensure their lifelong performance in pediatric and adult patients.
- Interaction with systemic pathologies and pharmacological treatments has not been reported in the literature. It is crucial to investigate how bioprinted neotissues respond to acute or chronic diseases compared to native tissue. Of particular interest are pathologies characterized by impaired perfusion accompanied by vasoconstriction and reduced local blood flow, as well as the effects of chronic hyperglycemia in diabetic patients. In these scenarios, a dual challenge arises ensuring the survival of native microvascularization while protecting the biomaterial from a potentially biochemically hostile microenvironment. In a specific pathological context, 3D bioprinted scaffolds can be tailored to better respond to these altered physiological conditions. Recent studies offer valuable insights into achieving this adaptation.
- −
- In diabetes mellitus (DM), the microenvironment characterized by hyperglycemia, inflammation, and mitochondrial dysfunction leads to the accumulation of ROS and advanced glycated end products (AGEs) [300,301]. Under these conditions, it is well established that, in addition to impairing microcirculation and promoting chronic inflammation, collagen glycation and the subsequent generation of AGEs are constant features in the progression of DM. This effect is also evident in collagen-based materials [302]. In patients with DM, several strategies exist for the management of 3D bioimplants. The use of decellularized ECM-based materials could prove effective under these altered conditions. Specifically, it is well established that chronic wounds are more alkaline than healthy skin, owing to bacterial contamination and an altered ECM. For instance, the development of pH-responsive hydrogels allows for the target release of vascular endothelial growth factor at alkaline pH (approximately 7.4) while inhibiting release at lower levels (5 or 6) [303]. One such 3D scaffold is composed of polyorganophosphazene polymers functionalized with antioxidant inclusions and fibroblasts. These scaffolds demonstrate enhanced cell viability and facilitate tissue regeneration in diabetic mouse models [304]. Recently, 3D GelMA-based scaffolds have been described that incorporate strontium-containing bioactive glass particles. These scaffolds are designed to remodel the diabetic tissue microenvironment, thereby enhancing angiogenesis, osteogenesis, and anti-inflammatory activity [305,306]. Certain hydrogels, such as those functionalized with glucose oxidase, have been designed to mitigate local hyperglycemia by catalyzing the conversion of glucose into hydrogen peroxide and glucuronic acid. This process subsequently lowers the local pH and exerts antimicrobial effects [307].
- −
- Hypoperfusion creates a chemically and metabolically hostile microenvironment. This leads to the accumulation of acidic metabolites, which accelerate the enzymatic degradation of polymers such as HA, alginate, and chitosan. Furthermore, the resulting reduction in local pH creates an environment that favors bacterial colonization [308]. In addition, the accumulation of acidic degradation products triggers the autocatalytic degradation of polylactic acid-based copolymers [239]. The reduction in local blood flow can induce ischemic conditions within the implanted biomaterial, leading to mechanical failure and the collapse of the scaffold [309].
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AGEs | Advanced glycated end products |
| CG | Carrageenan |
| c-MYC | Cellular-myelocytomatosis |
| DM | Diabetes mellitus |
| ECM | Extracellular matrix |
| ENT | Ear, nose, throat |
| ESC | Embryonic stem cell |
| EV | Extracellular vesicle |
| GAG | Glycosaminoglycan |
| Gal | Galactose |
| GelMA | Gelatin-methacrylate |
| GG | Gellan gum |
| Glc | Glucose |
| GlcA | Glucuronic acid |
| GlcNAc | N-acetylated glucosamine |
| HA | Hyaluronic acid |
| hNC | Human nasoseptal chondrocytes |
| hNEC | Human nasal epithelial cell |
| IEO | Inner ear organoids |
| KLF4 | Kruppel-like factor 4 |
| iPSC | Induced pluripotent stem cell |
| MMP | Matrix metalloproteinase |
| MSC | Mesenchymal stem cell |
| OCT4 | Octamer-binding transcription factor 4 |
| PEG | Polyethylene glycol |
| PEGDA | Polyethylene glycol diacrylate |
| PNIPAM | Poly-N-isopropylamide |
| ROS | Reactive oxygen species |
| SC | Synthetic cell |
| SF | Silk fibroin |
| SOX2 | Sex determining region Y-box2 |
| TM | Tympanic membrane |
References
- Jangan, A.; Minhas, S.; Diakos, E.; Simmons, M.; Mughal, Z. Evolving Trends and Future Demands in ENT Procedures: A Nationwide 10-Year Analysis. J. Clin. Med. 2024, 13, 7850. [Google Scholar] [CrossRef]
- Vrinceanu, D.; Sarafoleanu, C.; Kalcioglu, M.T.; Neagos, A. Multidisciplinarity and Transdisciplinarity as Current Trends in Otorhinolaryngology and Head and Neck Pathology. Medicina 2022, 58, 1661. [Google Scholar] [CrossRef]
- Massoud, S.; Farhat, R.; Abd Elhadi, U.; Awawde, R.; Merchavy, S.; Safia, A. Incidence Rate and Determinants of Recurrent Cholesteatoma Following Surgical Management: A Systematic Review, Subgroup, and Meta-Regression Analysis. Biomedicines 2025, 13, 2506. [Google Scholar] [CrossRef]
- de Lima, J.M.; Castellano, L.R.C.; Bonan, P.R.F.; de Medeiros, E.S.; Hier, M.; Bijian, K.; Alaoui-Jamali, M.A.; da Cruz Perez, D.E.; da Silva, S.D. Chitosan/PCL nanoparticles can improve anti-neoplastic activity of 5-fluorouracil in head and neck cancer through autophagy activation. Int. J. Biochem. Cell Biol. 2021, 134, 105964. [Google Scholar] [CrossRef]
- Huang, Z.; Hwang, P.; Sun, Y.; Zhou, B. Steroid-eluting sinus stents for improving symptoms in chronic rhinosinusitis patients undergoing functional endoscopic sinus surgery. Cochrane Database Syst. Rev. 2015, 2015, CD010436. [Google Scholar] [CrossRef] [PubMed]
- Gocal, W.A.; Tong, J.Y.; Maxwell, P.J.; Sataloff, R.T. Systematic Review of Recurrence Rates of Benign Vocal Fold Lesions Following Surgery. J. Voice 2025, 39, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Sudha, T.Y.S.; Sasanka, K.S.B.S.K.; Kumar, H.; Varshney, S. Unveiling the safety landscape: A comprehensive review of the toxicological profile of facial aesthetic implants and biomaterials. Natl. J. Maxillofac. Surg. 2024, 15, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Spałek, J.; Ociepa, P.; Deptuła, P.; Piktel, E.; Daniluk, T.; Król, G.; Góźdź, S.; Bucki, R.; Okła, S. Biocompatible Materials in Otorhinolaryngology and Their Antibacterial Properties. Int. J. Mol. Sci. 2022, 23, 2575. [Google Scholar] [CrossRef]
- Niermeyer, W.L.; Rodman, C.; Li, M.M.; Chiang, T. Tissue engineering applications in otolaryngology-The state of translation. Laryngoscope Investig. Otolaryngol. 2020, 5, 630–648. [Google Scholar] [CrossRef]
- Li, G.; Sun, S. Silk Fibroin-Based Biomaterials for Tissue Engineering Applications. Molecules 2022, 27, 2757. [Google Scholar] [CrossRef]
- Salary, M.; Mohebbi, S.; Ahmadi, A.; Bagher, Z.; Pezeshki-Modaress, M.; Aminianfar, H.; Farzad-Mohajeri, S.; Samiei, N.; Taghizadeh-Hesary, F.; Ghanbari, H. Utilizing biomaterials for laryngeal respiratory mucosal tissue repair in an animal model. Biomater. Biosyst. 2025, 19, 100120. [Google Scholar] [CrossRef]
- Li, Y.; Xun, X.; Duan, L.; Gao, E.; Li, J.; Lin, L.; Li, X.; He, A.; Ao, H.; Xu, Y.; et al. Cartilage structure-inspired nanofiber-hydrogel composite with robust proliferation and stable chondral lineage-specific differentiation function to orchestrate cartilage regeneration for artificial tracheal construction. Bioact. Mater. 2025, 47, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.F. Using platelet-rich fibrin scaffolds with diced cartilage graft in the treatment of empty nose syndrome. Ear Nose Throat J. 2024, 103, NP168–NP172. [Google Scholar] [CrossRef] [PubMed]
- Aydin, A.H.; Kaya, E.; Arik, D.; Canaz, F.; Pinarbasli, M.O.; Gurbuz, M.K.; Cakli, H.; Cingi, C. Experimental Investigation of the Effect of Polydioxanone Plate and Platelet-Rich Plasma on Cartilage Regeneration. Ear Nose Throat J. 2024, 103, 45S–57S. [Google Scholar] [CrossRef] [PubMed]
- McMillan, A.; McMillan, N.; Gupta, N.; Kanotra, S.P.; Salem, A.K. 3D Bioprinting in Otolaryngology: A Review. Adv. Healthc. Mater. 2023, 12, e2203268. [Google Scholar] [CrossRef]
- Svistushkin, M.V.; Kotova, S.; Shpichka, A.; Starostina, S.; Shekhter, A.; Bikmulina, P.; Nikiforov, A.; Zolotova, A.; Royuk, V.; Kochetkov, P.A.; et al. Stem cell therapy for vocal fold regeneration after scarring: A review of experimental approaches. Stem Cell Res. Ther. 2022, 13, 176. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, W.; Seunggi, C.; Li, Z.; Huang, Y.; Zhou, K.; Wang, B.; Chen, Z.; Zhang, Z. The application and progress of stem cells in auricular cartilage regeneration: A systematic review. Front. Cell Dev. Biol. 2023, 11, 1204050. [Google Scholar] [CrossRef]
- Liu, S.; Yu, J.M.; Gan, Y.C.; Qiu, X.Z.; Gao, Z.C.; Wang, H.; Chen, S.X.; Xiong, Y.; Liu, G.H.; Lin, S.E.; et al. Biomimetic natural biomaterials for tissue engineering and regenerative medicine: New biosynthesis methods, recent advances, and emerging applications. Mil. Med. Res. 2023, 10, 16. [Google Scholar] [CrossRef]
- Shokrani, H.; Shokrani, A.; Seidi, F.; Mashayekhi, M.; Kar, S.; Nedeljkovic, D.; Kuang, T.; Saeb, M.R.; Mozafari, M. Polysaccharide-based biomaterials in a journey from 3D to 4D printing. Bioeng. Transl. Med. 2023, 8, e10503. [Google Scholar] [CrossRef]
- Sternberg, K. Current requirements for polymeric biomaterials in otolaryngology. GMS Curr. Top. Otorhinolaryngol. Head Neck Surg. 2009, 8, Doc11. [Google Scholar]
- Iravani, S.; Varma, R.S. Cellulose-Based Composites as Scaffolds for Tissue Engineering: Recent Advances. Molecules 2022, 27, 8830. [Google Scholar] [CrossRef] [PubMed]
- Janmohammadi, M.; Nazemi, Z.; Salehi, A.O.M.; Seyfoori, A.; John, J.V.; Nourbakhsh, M.S.; Akbari, M. Cellulose-based composite scaffolds for bone tissue engineering and localized drug delivery. Bioact. Mater. 2022, 20, 137–163. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamid, H.N.; Mathew, A.P. Cellulose-Based Nanomaterials Advance Biomedicine: A Review. Int. J. Mol. Sci. 2022, 23, 5405. [Google Scholar] [CrossRef]
- Tyshkunova, I.V.; Poshina, D.N.; Skorik, Y.A. Cellulose Cryogels as Promising Materials for Biomedical Applications. Int. J. Mol. Sci. 2022, 23, 2037. [Google Scholar] [CrossRef]
- Du, Z.; Wang, N.; Du, J. Recent Progress in Cellulose-Based Conductive Hydrogels. Polymers 2025, 17, 1089. [Google Scholar] [CrossRef]
- Courtenay, J.C.; Sharma, R.I.; Scott, J.L. Recent Advances in Modified Cellulose for Tissue Culture Applications. Molecules 2018, 23, 654. [Google Scholar] [CrossRef]
- Laboy-López, S.; Méndez Fernández, P.O.; Padilla-Zayas, J.G.; Nicolau, E. Bioactive Cellulose Acetate Electrospun Mats as Scaffolds for Bone Tissue Regeneration. Int. J. Biomater. 2022, 2022, 3255039. [Google Scholar] [CrossRef]
- Hasanin, M.S. Cellulose-Based Biomaterials: Chemistry and Biomedical Applications. Starch 2022, 74, 2200060. [Google Scholar] [CrossRef]
- Vázquez-Rivas, E.; Desales-Guzmán, L.A.; Pacheco-Sánchez, J.H.; Burillo-Amezcua, S.G. Cellulose-Based Hybrid Hydrogels for Tissue Engineering Applications: A Sustainable Approach. Gels 2025, 11, 438. [Google Scholar] [CrossRef]
- Chen, C.; Xi, Y.; Weng, Y. Recent Advances in Cellulose-Based Hydrogels for Tissue Engineering Applications. Polymers 2022, 14, 3335. [Google Scholar] [CrossRef]
- Utoiu, E.; Manoiu, V.S.; Oprita, E.I.; Craciunescu, O. Bacterial Cellulose: A Sustainable Source for Hydrogels and 3D-Printed Scaffolds for Tissue Engineering. Gels 2024, 10, 387. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bai, J.; Tian, P.; Xie, R.; Duan, Z.; Lv, Q.; Tao, Y. The Application Status of Nanoscale Cellulose-Based Hydrogels in Tissue Engineering and Regenerative Biomedicine. Front. Bioeng. Biotechnol. 2021, 9, 732513. [Google Scholar] [CrossRef] [PubMed]
- Xiaorui, L.; Fuyin, Z.; Xudong, W.; Xuezheng, G.; Shudong, Z.; Hui, L.; Dandan, D.; Yubing, L.; Lizhen, W.; Yubo, F. Biomaterial inks for extrusion-based 3D bioprinting: Property, classification, modification, and selection. Int. J. Bioprint. 2022, 9, 649. [Google Scholar]
- Sainsbury, E.; Amaral, R.D.; Blayney, A.W.; Walsh, R.M.; O’Brien, F.J.; O’Leary, C. Tissue engineering and regenerative medicine strategies for the repair of tympanic membrane perforations. Biomater. Biosyst. 2022, 6, 100046. [Google Scholar] [CrossRef]
- Jiang, L.; Cherif, C.; Wöltje, M. Fibrous Biomaterial Scaffold for Tympanic Membrane Repair: Microarchitectural Engineering and Structure Function Performance. J. Funct. Biomater. 2026, 17, 53. [Google Scholar] [CrossRef]
- Vyas, J.; Shah, I.; Singh, S.; Prajapati, B.G. Biomaterials-based additive manufacturing for customized bioengineering in management of otolaryngology: A comprehensive review. Front. Bioeng. Biotechnol. 2023, 11, 1234340. [Google Scholar] [CrossRef]
- Biskin, S.; Damar, M.; Oktem, S.N.; Sakalli, E.; Erdem, D.; Pakir, O. A new graft material for myringoplasty: Bacterial cellulose. Eur. Arch. Otorhinolaryngol. 2016, 273, 3561–3565. [Google Scholar] [CrossRef]
- Azimi, B.; Milazzo, M.; Danti, S. Cellulose-Based Fibrous Materials from Bacteria to Repair Tympanic Membrane Perforations. Front. Bioeng. Biotechnol. 2021, 9, 669863. [Google Scholar] [CrossRef]
- Anand, S.; Danti, S.; Moroni, L.; Mota, C. Regenerative therapies for tympanic membrane. Prog. Mater. Sci. 2022, 127, 100942. [Google Scholar] [CrossRef]
- Kaboodkhani, R.; Mehrabani, D.; Karimi-Busheri, F. Achievements and Challenges in Transplantation of Mesenchymal Stem Cells in Otorhinolaryngology. J. Clin. Med. 2021, 10, 2940. [Google Scholar] [CrossRef]
- Falua, K.J.; Pokharel, A.; Babaei-Ghazvini, A.; Ai, Y.; Acharya, B. Valorization of Starch to Biobased Materials: A Review. Polymers 2022, 14, 2215. [Google Scholar] [CrossRef]
- Vardhan, H.; Singhal, N.; Vashistha, P.; Jain, R.; Bist, Y.; Gaur, A.; Wagri, K.N. Starch–biomacromolecule complexes: A comprehensive review of interactions, functional materials, and applications in food, pharma, and packaging. Carbohydr. Polym. Technol. Appl. 2025, 11, 101001. [Google Scholar] [CrossRef]
- Sponchiado, P.A.I.; Melo, M.T.; Cominal, J.G.; Martelli Tosi, M.; Ciancaglini, P.; Ramos, A.P.; Maniglia, B.C. Biomembranes Based on Potato Starch Modified by Dry Heating Treatment: One Sustainable Strategy to Amplify the Use of Starch as a Biomaterial. Biomacromolecules 2025, 26, 1530–1540. [Google Scholar] [CrossRef] [PubMed]
- Massey, C.J.; Suh, J.D.; Tessema, B.; Gray, S.T.; Singh, A. Biomaterials in Rhinology. Otolaryngol. Head Neck Surg. 2016, 154, 606–617. [Google Scholar] [CrossRef]
- Yu, T.-C.; Huang, T.-C.; Chen, P.-Y.; Shih, C.-C.; Chang, W.-W. Biomaterial Material Applications in Postoperative Surgical Fields of Uvulopalatopharyngoplasty: A Comparative Study. J. Funct. Biomater. 2023, 14, 337. [Google Scholar] [CrossRef]
- Razali, R.A.; Vijakumaran, U.; Fauzi, M.B.; Lokanathan, Y. Maximizing Postoperative Recovery: The Role of Functional Biomaterials as Nasal Packs—A Comprehensive Systematic Review without Meta-Analysis (SWiM). Pharmaceutics 2023, 15, 1534. [Google Scholar] [CrossRef]
- LyBarger, K.S. Review of Evidence Supporting the Arista™ Absorbable Powder Hemostat. Med. Devices 2024, 17, 173–188. [Google Scholar] [CrossRef]
- Iaconisi, G.N.; Lunetti, P.; Gallo, N.; Cappello, A.R.; Fiermonte, G.; Dolce, V.; Capobianco, L. Hyaluronic Acid: A Powerful Biomolecule with Wide-Ranging Applications—A Comprehensive Review. Int. J. Mol. Sci. 2023, 24, 10296. [Google Scholar] [CrossRef]
- Di Mola, A.; Landi, M.R.; Massa, A.; D’Amora, U.; Guarino, V. Hyaluronic Acid in Biomedical Fields: New Trends from Chemistry to Biomaterial Applications. Int. J. Mol. Sci. 2022, 23, 14372. [Google Scholar] [CrossRef]
- Grabowski, M.; Gmyrek, D.; Żurawska, M.; Trusek, A. Hyaluronic Acid: Production Strategies, Gel-Forming Properties, and Advances in Drug Delivery Systems. Gels 2025, 11, 424. [Google Scholar] [CrossRef]
- Pashkina, E.; Bykova, M.; Berishvili, M.; Lazarev, Y.; Kozlov, V. Hyaluronic Acid-Based Drug Delivery Systems for Cancer Therapy. Cells 2025, 14, 61. [Google Scholar] [CrossRef] [PubMed]
- Ricard-Blum, S.; Vivès, R.R.; Schaefer, L.; Götte, M.; Merline, R.; Passi, A.; Heldin, P.; Magalhães, A.; Reis, C.A.; Skandalis, S.S.; et al. A biological guide to glycosaminoglycans: Current perspectives and pending questions. FEBS J. 2024, 291, 3331–3366. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Dong, J.; Pan, R.; Xu, Z.; Li, M.; Zang, R. Structures, Properties, and Bioengineering Applications of Alginates and Hyaluronic Acid. Polymers 2023, 15, 2149. [Google Scholar] [CrossRef]
- Jiang, T.; Yang, Y.; Lin, Z.; Hong, Y.; Luo, Z. Modified Polysaccharides: Potential Biomaterials for Bioprinting. J. Funct. Biomater. 2025, 16, 338. [Google Scholar] [CrossRef]
- Magdziarz, S.; Boguń, M.; Frączyk, J. Coating Methods of Carbon Nonwovens with Cross-Linked Hyaluronic Acid and Its Conjugates with BMP Fragments. Polymers 2023, 15, 1551. [Google Scholar] [CrossRef]
- Karam, J.; Singer, B.J.; Miwa, H.; Chen, L.H.; Maran, K.; Hasani, M.; Garza, S.; Onyekwere, B.; Yeh, H.C.; Li, S.; et al. Molecular weight of hyaluronic acid crosslinked into biomaterial scaffolds affects angiogenic potential. Acta Biomater. 2023, 169, 228–242. [Google Scholar] [CrossRef]
- Sikkema, R.; Keohan, B.; Zhitomirsky, I. Hyaluronic-Acid-Based Organic-Inorganic Composites for Biomedical Applications. Materials 2021, 14, 4982. [Google Scholar] [CrossRef]
- Shi, W.; Zhang, J.; Gao, Z.; Hu, F.; Kong, S.; Hu, X.; Zhao, F.; Ao, Y.; Shao, Z. Three-Dimensional Printed Silk Fibroin/Hyaluronic Acid Scaffold with Functionalized Modification Results in Excellent Mechanical Strength and Efficient Endogenous Cell Recruitment for Articular Cartilage Regeneration. Int. J. Mol. Sci. 2024, 25, 10523. [Google Scholar] [CrossRef]
- Ou, H.; Yang, Q.; Zhang, Y.; Tang, X.; Xiao, M.; Li, S.; Lei, L.; Xie, Z. The role of cells and their derivatives in otorhinolaryngologic diseases treatment. Life Sci. 2024, 352, 122898. [Google Scholar] [CrossRef]
- Dinaki, K.; Grigoriadis, N.; Vizirianakis, I.; Constantinidis, J.; Triaridis, S.; Karkos, P. Tissue engineering and regenerative medicine in otorhinolaryngology. J. Clin. Trans. Res. 2024, 10, 99–111. [Google Scholar] [CrossRef]
- Oh, S.Y.; Kim, H.Y.; Jung, S.Y.; Kim, H.S. Tissue Engineering and Regenerative Medicine in the Field of Otorhinolaryngology. Tissue Eng. Regen. Med. 2024, 21, 969–984. [Google Scholar] [CrossRef] [PubMed]
- Lott, D.G.; Janus, J.R. Tissue engineering for otorhinolaryngology-head and neck surgery. Mayo Clin. Proc. 2014, 89, 1722–1733. [Google Scholar] [CrossRef] [PubMed]
- Gaston, J.; Thibeault, S.L. Hyaluronic acid hydrogels for vocal fold wound healing. Biomatter 2013, 3, e23799. [Google Scholar] [CrossRef]
- Wang, Y.; Wen, F.; Yao, X.; Zeng, L.; Wu, J.; He, Q.; Li, H.; Fang, L. Hybrid Hydrogel Composed of Hyaluronic Acid, Gelatin, and Extracellular Cartilage Matrix for Perforated TM Repair. Front. Bioeng. Biotechnol. 2021, 9, 811652. [Google Scholar] [CrossRef]
- Pacheco-Quito, E.-M.; Ruiz-Caro, R.; Veiga, M.-D. Carrageenan: Drug Delivery Systems and Other Biomedical Applications. Mar. Drugs 2020, 18, 583. [Google Scholar] [CrossRef]
- Neamtu, B.; Barbu, A.; Negrea, M.O.; Berghea-Neamțu, C.Ș.; Popescu, D.; Zăhan, M.; Mireșan, V. Carrageenan-Based Compounds as Wound Healing Materials. Int. J. Mol. Sci. 2022, 23, 9117. [Google Scholar] [CrossRef]
- Lipinska, A.P.; Collén, J.; Krueger-Hadfield, S.A.; Mora, T.; Ficko-Blean, E. To gel or not to gel: Differential expression of carrageenan-related genes between the gametophyte and tetasporophyte life cycle stages of the red alga Chondruscrispus. Sci. Rep. 2020, 10, 11498. [Google Scholar] [CrossRef]
- Hale, J.; Gerhäuser, J.; Gaukel, V.; Wefers, D. Commercially available carrageenans show broad variation in their structure, composition, and functionality. Eur. Food Res. Technol. 2024, 250, 2989–3003. [Google Scholar] [CrossRef]
- Gubaidullin, A.T.; Makarova, A.O.; Derkach, S.R.; Voron’ko, N.G.; Kadyirov, A.I.; Ziganshina, S.A.; Salnikov, V.V.; Zueva, O.S.; Zuev, Y.F. Modulation of Molecular Structure and Mechanical Properties of κ-Carrageenan-Gelatin Hydrogel with Multi-Walled Carbon Nanotubes. Polymers 2022, 14, 2346. [Google Scholar] [CrossRef]
- Yermak, I.; Anastyuk, S.; Kravchenko, A.; Helbert, W.; Glazunov, V.; Shulgin, A.; Spirin, P.; Prassolov, V. New Insights into the Structure of Kappa/Beta-Carrageenan: A Novel Potential Inhibitor of HIV-1. Int. J. Mol. Sci. 2021, 22, 12905. [Google Scholar] [CrossRef]
- Kravchenko, A.O.; Menchinskaya, E.S.; Isakov, V.V.; Glazunov, V.P.; Yermak, I.M. Carrageenans and Their Oligosaccharides from Red Seaweeds Ahnfeltiopsisflabelliformis and Mastocarpuspacificus (Phyllophoraceae) and Their Antiproliferative Activity. Int. J. Mol. Sci. 2023, 24, 7657. [Google Scholar] [CrossRef]
- Hilliou, L. Structure–Elastic Properties Relationships in Gelling Carrageenans. Polymers 2021, 13, 4120. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Lee, Y.W.; Jung, W.K.; Oh, J.; Nam, S.Y. Enhanced rheological behaviors of alginate hydrogels with carrageenan for extrusion-based bioprinting. J. Mech. Behav. Biomed. Mater. 2019, 98, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Stathis, C.; Victoria, N.; Loomis, K.; Nguyen, S.A.; Eggers, M.; Septimus, E.; Safdar, N. Review of the use of nasal and oral antiseptics during a global pandemic. Future Microbiol. 2021, 16, 119–130. [Google Scholar] [CrossRef]
- Schütz, D.; Conzelmann, C.; Fois, G.; Groß, R.; Weil, T.; Wettstein, L.; Stenger, S.; Zelikin, A.; Hoffmann, T.K.; Frick, M.; et al. Carrageenan-containing over-the-counter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures. Am. J. Physiol. Lung Cell. Mol. Physiol. 2021, 320, L750–L756. [Google Scholar] [CrossRef]
- Figueroa, J.M.; Lombardo, M.E.; Dogliotti, A.; Flynn, L.P.; Giugliano, R.; Simonelli, G.; Valentini, R.; Ramos, A.; Romano, P.; Marcote, M.; et al. Efficacy of a Nasal Spray Containing Iota-Carrageenan in the Postexposure Prophylaxis of COVID-19 in Hospital Personnel Dedicated to Patients Care with COVID-19 Disease. Int. J. Gen. Med. 2021, 14, 6277–6286. [Google Scholar] [CrossRef]
- Razali, R.A.; Muhammad Firdaus, F.I.; Fauzi, M.B.; Mobarak, N.N.; Aminuddin, S.; Lokanathan, Y. Biodegradable Gelatin–Carrageenan Sponges: High-Potential Functional Nasal Packs for Efficient Secretome Delivery. Polymers 2024, 16, 3387. [Google Scholar] [CrossRef]
- Muscolino, E.; Di Stefano, A.B.; Trapani, M.; Sabatino, M.A.; Giacomazza, D.; Alessi, S.; Cammarata, E.; Moschella, F.; Cordova, A.; Toia, F.; et al. κ-Carrageenan and PVA blends as bioinks to 3D print scaffolds for cartilage reconstruction. Int. J. Biol. Macromol. 2022, 222, 1861–1875. [Google Scholar] [CrossRef]
- Zhang, H.; Cheng, J.; Ao, Q. Preparation of Alginate-Based Biomaterials and Their Applications in Biomedicine. Mar. Drugs 2021, 19, 264. [Google Scholar] [CrossRef]
- Soleimanpour, M.; Mirhaji, S.S.; Jafari, S.; Derakhshankhah, H.; Mamashli, F.; Nedaei, H.; Karimi, M.R.; Motasadizadeh, H.; Fatahi, Y.; Ghasemi, A.; et al. Designing a new alginate-fibrinogen biomaterial composite hydrogel for wound healing. Sci. Rep. 2022, 12, 7213. [Google Scholar] [CrossRef]
- He, Q.; Tong, T.; Yu, C.; Wang, Q. Advances in Algin and Alginate-Hybrid Materials for Drug Delivery and Tissue Engineering. Mar. Drugs 2023, 21, 14. [Google Scholar] [CrossRef]
- Hurtado, A.; Aljabali, A.A.A.; Mishra, V.; Tambuwala, M.M.; Serrano-Aroca, Á. Alginate: Enhancement Strategies for Advanced Applications. Int. J. Mol. Sci. 2022, 23, 4486. [Google Scholar] [CrossRef] [PubMed]
- Najafi, R.; Chahsetareh, H.; Pezeshki-Modaress, M.; Aleemardani, M.; Simorgh, S.; Davachi, S.M.; Alizadeh, R.; Asghari, A.; Hassanzadeh, S.; Bagher, Z. Alginate sulfate/ECM composite hydrogel containing electrospun nanofiber with encapsulated human adipose-derived stem cells for cartilage tissue engineering. Int. J. Biol. Macromol. 2023, 238, 124098. [Google Scholar] [CrossRef] [PubMed]
- Tomić, S.L.; BabićRadić, M.M.; Vuković, J.S.; Filipović, V.V.; Nikodinovic-Runic, J.; Vukomanović, M. Alginate-Based Hydrogels and Scaffolds for Biomedical Applications. Mar. Drugs 2023, 21, 177. [Google Scholar] [CrossRef] [PubMed]
- Adamiak, K.; Sionkowska, A. State of Innovation in Alginate-Based Materials. Mar. Drugs 2023, 21, 353. [Google Scholar] [CrossRef]
- Rossi, A.; Pescara, T.; Gambelli, A.M.; Gaggia, F.; Asthana, A.; Perrier, Q.; Basta, G.; Moretti, M.; Senin, N.; Rossi, F.; et al. Biomaterials for extrusion-based bioprinting and biomedical applications. Front. Bioeng. Biotechnol. 2024, 12, 1393641. [Google Scholar] [CrossRef]
- Brown, M.; Okuyama, H.; Li, L.; Yang, Z.; Li, J.; Tabrizian, M.; Li-Jessen, N.Y.K. Clicktetrazine dECM-alginate hydrogels for injectable, mechanically mimetic, and biologically active vocal fold biomaterials. Biomaterials 2026, 325, 123590. [Google Scholar] [CrossRef]
- Ilhan, E.; Ulag, S.; Sahin, A.; Yilmaz, B.K.; Ekren, N.; Kilic, O.; Sengor, M.; Kalaskar, D.M.; Oktar, F.N.; Gunduz, O. Fabrication of tissue-engineered tympanic membrane patches using 3D-Printing technology. J. Mech. Behav. Biomed. Mater. 2021, 114, 104219. [Google Scholar] [CrossRef]
- AbdlAali, R.A.K.; Al-Sahlany, S.T.G. Gellan Gum as a Unique Microbial Polysaccharide: Its Characteristics, Synthesis, and Current Application Trends. Gels 2024, 10, 183. [Google Scholar] [CrossRef]
- Gomes, D.; Batista-Silva, J.P.; Sousa, A.; Passarinha, L.A. Progress and opportunities in Gellan gum-based materials: A review of preparation, characterization and emerging applications. Carbohydr. Polym. 2023, 311, 120782. [Google Scholar] [CrossRef]
- Li, H.; Gao, K.; Guo, H.; Li, R.; Li, G. Advancements in Gellan Gum-Based Films and Coatings for Active and Intelligent Packaging. Polymers 2024, 16, 2402. [Google Scholar] [CrossRef]
- D’Amora, U.; Ronca, A.; Scialla, S.; Soriente, A.; Manini, P.; Phua, J.W.; Ottenheim, C.; Pezzella, A.; Calabrese, G.; Raucci, M.G.; et al. Bioactive Composite Methacrylated Gellan Gum for 3D-Printed Bone Tissue-Engineered Scaffolds. Nanomaterials 2023, 13, 772. [Google Scholar] [CrossRef] [PubMed]
- Croft, A.S.; Ćorluka, S.; Fuhrer, J.; Wöltje, M.; Silva-Correia, J.; Oliveira, J.M.; Erbach, G.F.; Reis, R.L.; Gantenbein, B. Repairing Annulus Fibrosus Fissures Using Methacrylated Gellan Gum Combined with Novel Silk. Materials 2023, 16, 3173. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Yu, Y.; Tan, J.; Huang, L.; Luo, B.; Lu, L.; Zhou, C. 3D bioprinting of gellan gum and poly (ethylene glycol) diacrylate based hydrogels to produce human-scale constructs with high-fidelity. Mater. Des. 2018, 160, 486–495. [Google Scholar] [CrossRef]
- Wojtyłko, M.; Froelich, A.; Jadach, B. Hypromellose-, Gelatin- and Gellan Gum-Based Gel Films with Chlorhexidine for Potential Application in Oral Inflammatory Diseases. Gels 2024, 10, 265. [Google Scholar] [CrossRef]
- Ganesan, N.; Felix, L.O.; Mishra, B.; Zhang, L.; Dellis, C.; Shehadeh, F.; Wu, D.; Cruz, L.A.; Arce, R.M.; Mylonakis, E. Gellan gum formulations containing natural polyphenolic compounds to treat oral candidiasis. Microbiol. Spectr. 2025, 13, e0079825. [Google Scholar] [CrossRef]
- Garcia, M.T.; Carmo, P.H.F.d.; Figueiredo-Godoi, L.M.A.; Gonçalves, N.I.; Lima, P.M.N.d.; Ramos, L.d.P.; Oliveira, L.D.d.; Borges, A.L.S.; Shukla, A.; Junqueira, J.C. Gellan-Based Hydrogel as a Drug Delivery System for Caffeic Acid Phenethyl Ester in the Treatment of Oral Candida albicans Infections. Pharmaceutics 2024, 16, 298. [Google Scholar] [CrossRef]
- Marina, C.D.; Puscasiu, D.; Flangea, C.; Vlad, T.; Cimporescu, A.; Popescu, R.; Moatar, A.E.; Vlad, D.C. Adipo-Modulation by Turmeric Bioactive Phenolic Components: From Curcuma Plant to Effects. Int. J. Mol. Sci. 2025, 26, 6880. [Google Scholar] [CrossRef]
- Nan, A.; Dumitrascu, V.; Flangea, C.; Dumitrescu, G.; Puscasiu, D.; Vlad, T.; Popescu, R.; Vlad, C. From Chemical Composition to Antiproliferative Effects Through In Vitro Studies: Honey, an Ancient and Modern Hot Topic Remedy. Nutrients 2025, 17, 1595. [Google Scholar] [CrossRef]
- Popescu, R.; Flangea, C.; Vlad, D.C.; Cobec, I.M.; Seropian, P.; Marina, C.D.; Vlad, T.; Dumitrascu, A.L.; Puscasiu, D. Nutritional Impact on Breast Cancer in Menopausal and Post-Menopausal Patients Treated with Aromatase Inhibitors. Cancers 2026, 18, 73. [Google Scholar] [CrossRef]
- Jafernik, K.; Ładniak, A.; Blicharska, E.; Czarnek, K.; Ekiert, H.; Wiącek, A.E.; Szopa, A. Chitosan-Based Nanoparticles as Effective Drug Delivery Systems—A review. Molecules 2023, 28, 1963. [Google Scholar] [CrossRef]
- Satitsri, S.; Muanprasat, C. Chitin and Chitosan Derivatives as Biomaterial Resources for Biological and Biomedical Applications. Molecules 2020, 25, 5961. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int. J. Mol. Sci. 2020, 21, 9656. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.; Poverenov, E. Hydrophilic Chitosan Derivatives: Synthesis and Applications. Chemistry 2022, 28, e202202156. [Google Scholar] [CrossRef] [PubMed]
- Kulka, K.; Sionkowska, A. Chitosan Based Materials in Cosmetic Applications: A Review. Molecules 2023, 28, 1817. [Google Scholar] [CrossRef]
- Petroni, S.; Tagliaro, I.; Antonini, C.; D’Arienzo, M.; Orsini, S.F.; Mano, J.F.; Brancato, V.; Borges, J.; Cipolla, L. Chitosan-Based Biomaterials: Insights into Chemistry, Properties, Devices, and Their Biomedical Applications. Mar. Drugs 2023, 21, 147. [Google Scholar] [CrossRef]
- Notario-Pérez, F.; Martín-Illana, A.; Cazorla-Luna, R.; Ruiz-Caro, R.; Veiga, M.D. Applications of Chitosan in Surgical and Post-Surgical Materials. Mar. Drugs 2022, 20, 396. [Google Scholar] [CrossRef]
- Becerra, J.; Rodriguez, M.; Leal, D.; Noris-Suarez, K.; Gonzalez, G. Chitosan-collagen-hydroxyapatite membranes for tissue engineering. J. Mater. Sci. Mater. Med. 2022, 33, 18. [Google Scholar] [CrossRef]
- Hemmingsen, L.M.; Škalko-Basnet, N.; Jøraholmen, M.W. The Expanded Role of Chitosan in Localized Antimicrobial Therapy. Mar. Drugs 2021, 19, 697. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.Y.; Han, H.; Cho, W.W.; Han, H.; Choi, A.; Hong, H.; Kim, J.Y.; Park, J.H.; Park, S.H.; et al. Improved chondrogenic performance with protective tracheal design of Chitosan membrane surrounding 3D-printed trachea. Sci. Rep. 2021, 11, 9258. [Google Scholar] [CrossRef]
- Spencer, H.; Moshkbouymatin, N.; Webb, W.R.; Joshi, A.; D’Souza, A. Update on the role of emerging stem cell technology in head and neck medicine. Head Neck 2021, 43, 1928–1938. [Google Scholar] [CrossRef] [PubMed]
- Coburn, P.T.; Herbay, A.C.; Berrini, M.; Li-Jessen, N.Y.K. An in vitro assessment of the response of THP-1 macrophages to varying stiffness of a glycol-chitosan hydrogel for vocal fold tissue engineering applications. J. Biomed. Mater. Res. A 2021, 109, 1337–1352. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y.; Hosoya, K.; Fujiwara, Y.; Saito, Y.; Yoshida, M.; Matsune, S.; Okubo, K.; Takei, T. Injectable Chitosan Hydrogel Particles as Nasal Packing Materials After Endoscopic Sinus Surgery for Treatment of Chronic Sinusitis. Gels 2025, 11, 60. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; So, E.; Mustafa, F.E.; Jeong, J.-h.; Lee, B.-K. Preliminary Study on the Development of a Biodegradable Functional Nasal Packing Material. Polymers 2025, 17, 1878. [Google Scholar] [CrossRef]
- Pham, H.K.; Nguyen, T.; Tran, T.T.; Thai, T.T. A comparison of effectiveness between Posisep and Merocel nasal packing after endoscopic sinus surgery: Findings from a randomized, double-blinded, controlled trial. Medicine 2023, 102, e34782. [Google Scholar] [CrossRef]
- Elden, H.; Yilmaz, M.S.; Cihan, A.; Guven, E.M.; Kara, A. Effect of Chitosan-Based Biodegradable Nasal Packs on Eustachian Dysfunction after Septoplasty. Int. Arch. Otorhinolaryngol. 2025, 29, s00451809433. [Google Scholar] [CrossRef]
- Chen, V.H.E.; Tan, L.S.H. Gelatin versus chitosan as packing materials in endoscopic myringoplasty for chronic suppurative otitis media. Am. J. Otolaryngol. 2025, 46, 104654. [Google Scholar] [CrossRef]
- Biswal, S.; Agmon, N. Collagen Structured Hydration. Biomolecules 2023, 13, 1744. [Google Scholar] [CrossRef]
- Wang, J.; Hu, J.; Yuan, X.; Li, Y.; Song, L.; Xu, F. Recombinant collagen hydrogels induced by disulfide bonds. J. Biomed. Mater. Res. A 2022, 110, 1774–1785. [Google Scholar] [CrossRef]
- Giannetti, G.; Matsumura, F.; Caporaletti, F.; Micha, D.; Koenderink, G.H.; Ilie, I.M.; Bonn, M.; Woutersen, S.; Giubertoni, G. Water and Collagen: A Mystery Yet to Unfold. Biomacromolecules 2025, 26, 2784–2799. [Google Scholar] [CrossRef]
- Guo, L.; Xiang, W.; Pan, Z.; Gu, H.; Jiang, X. Post-translational modifications of collagen and its related diseases in metabolic pathways. Acta Pharm. Sin. B 2025, 15, 1773–1795. [Google Scholar] [CrossRef]
- Qi, Y.; Zhou, D.; Kessler, J.L.; Qiu, R.; Yu, S.M.; Li, G.; Qin, Z.; Li, Y. Terminal repeats impact collagen triple-helix stability through hydrogen bonding. Chem. Sci. 2022, 13, 12567–12576. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, Y.; Han, Y.; Cui, J.; Jing, Z.; Li, D.; Liu, J.; Xiao, C.; Li, D.; Cai, B. Collagen-Based Hydrogels for Cartilage Regeneration. Orthop. Surg. 2023, 15, 3026–3045. [Google Scholar] [CrossRef] [PubMed]
- Shaikenov, R.O.; Serbun, P.G.; Zhang, J.; Wu, H.; Wang, Z.; Snetkov, P.P.; Morozkina, S.N. Natural Polymers, Their Modifications and Composites with Synthetic Polymers for Tympanic Membrane Regeneration. J. Funct. Biomater. 2025, 16, 384. [Google Scholar] [CrossRef] [PubMed]
- Svistushkin, M.; Kotova, S.; Zolotova, A.; Fayzullin, A.; Antoshin, A.; Serejnikova, N.; Shekhter, A.; Voloshin, S.; Giliazova, A.; Istranova, E.; et al. Collagen Matrix to Restore the Tympanic Membrane: Developing a Novel Platform to Treat Perforations. Polymers 2024, 16, 248. [Google Scholar] [CrossRef]
- Ajaiy, M.; Sankaranarayanan, G.; Jayaprakash, A.; Deva Sooria, N. A Comparative Study of Using Collagen Patch or Fat Plug in Myringoplasty for Small Tympanic Membrane Perforations. Indian J. Otolaryngol. Head Neck Surg. 2022, 74, 4081–4086. [Google Scholar] [CrossRef]
- Tanasie, G.; Bojin, F.; Tatu, R.F.; Sisu, A.M.; Cristea, M.; Puscasiu, D.A.; Nemes, E.A.; Tatu, C.A. In vitro effects of biomaterials on mesenchymal stem cells viability and proliferation. Mater. Plast. 2017, 54, 523–526. [Google Scholar] [CrossRef]
- Hurmuz, M.; Bojin, F.; Ionac, M.; Tatu, F.; Puscasiu, D.; Tatu, C. Plastic adherence method for isolation of stem cells derived from infrapatellar fat pad. Mater. Plast. 2016, 53, 553–556. [Google Scholar]
- Zou, C.Y.; Hu, J.J.; Lu, D.; Li, Q.J.; Jiang, Y.L.; Wang, R.; Wang, H.Y.; Lei, X.X.; Li-Ling, J.; Yang, H.; et al. A self-fused hydrogel for the treatment of glottic insufficiency through outstanding durability, extracellular matrix-inducing bioactivity and function preservation. Bioact. Mater. 2022, 24, 54–68. [Google Scholar] [CrossRef]
- Chiesa-Estomba, C.M.; Aiastui, A.; González-Fernández, I.; Hernáez-Moya, R.; Rodiño, C.; Delgado, A.; Garces, J.P.; Paredes-Puente, J.; Aldazabal, J.; Altuna, X.; et al. Three-Dimensional Bioprinting Scaffolding for Nasal Cartilage Defects: A Systematic Review. Tissue Eng. Regen. Med. 2021, 18, 343–353. [Google Scholar] [CrossRef]
- Pandini, F.E.; Kubo, F.M.M.; Plepis, A.M.d.G.; Martins, V.d.C.A.; da Cunha, M.R.; Silva, V.R.; Hirota, V.B.; Lopes, E.; Menezes, M.A.; Pelegrine, A.A.; et al. In Vivo Study of Nasal Bone Reconstruction with Collagen, Elastin and Chitosan Membranes in Abstainer and Alcoholic Rats. Polymers 2022, 14, 188. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Wang, J.; Hao, Z.; Zhao, D. Gelatin-based biomaterials and gelatin as an additive for chronic wound repair. Front. Pharmacol. 2024, 15, 1398939. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, O.V. Gelatin as It Is: History and Modernity. Int. J. Mol. Sci. 2023, 24, 3583. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wan, J.; Zhang, Z.; Hou, R. Recent advances on 3D-bioprinted gelatin methacrylate hydrogels for tissue engineering in wound healing: A review of current applications and future prospects. Int. Wound J. 2024, 21, e14533. [Google Scholar] [CrossRef]
- Bupphathong, S.; Quiroz, C.; Huang, W.; Chung, P.-F.; Tao, H.-Y.; Lin, C.-H. Gelatin Methacrylate Hydrogel for Tissue Engineering Applications—A Review on Material Modifications. Pharmaceuticals 2022, 15, 171. [Google Scholar] [CrossRef]
- Nedunchezian, S.; Wu, C.-W.; Wu, S.-C.; Chen, C.-H.; Chang, J.-K.; Wang, C.-K. Characteristic and Chondrogenic Differentiation Analysis of Hybrid Hydrogels Comprised of Hyaluronic Acid Methacryloyl (HAMA), Gelatin Methacryloyl (GelMA), and the Acrylate-Functionalized Nano-Silica Crosslinker. Polymers 2022, 14, 2003. [Google Scholar] [CrossRef]
- Paul, S.; Schrobback, K.; Tran, P.A.; Meinert, C.; Davern, J.W.; Weekes, A.; Nedunchezhiyan, U.; Klein, T.J. GelMA-glycol chitosan hydrogels for cartilage regeneration: The role of uniaxial mechanical stimulation in enhancing mechanical, adhesive, and biochemical properties. APL Bioeng. 2023, 7, 036114. [Google Scholar] [CrossRef]
- Zertuche-Arias, T.; Alatorre-Meda, M.; Rivero, I.A.; Juárez, P.; Castro-Ceseña, A.B. N-Acetylcysteine and pro-adrenomedullin dual-crosslinked gelatin-chitosan hydrogels with enhanced mechanical and mineralization performance. RSC Adv. 2025, 15, 22524–22533. [Google Scholar] [CrossRef]
- Marfoglia, A.; Tibourtine, F.; Pilloux, L.; Cazalbou, S. Tunable Double-Network GelMA/Alginate Hydrogels for Platelet Lysate-Derived Protein Delivery. Bioengineering 2023, 10, 1044. [Google Scholar] [CrossRef]
- Kadri, R.; Elkhoury, K.; Ben Messaoud, G.; Kahn, C.; Tamayol, A.; Mano, J.F.; Arab-Tehrany, E.; Sánchez-González, L. Physicochemical Interactions in Nanofunctionalized Alginate/GelMA IPN Hydrogels. Nanomaterials 2021, 11, 2256. [Google Scholar] [CrossRef]
- Sreelesh, L.S.; Aswathi, T.V. Post dermoid excision nasal tip deformity correction using gelatin sponge. Indian J. Otolaryngol. Head Neck Surg. 2022, 74, 4831–4834. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Gregory, D.A.; Tomeh, M.A.; Zhao, X. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 1499. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Cui, J.; Wu, S.; Geng, Z.; Su, J. Silk fibroin-based biomaterials for cartilage/osteochondral repair. Theranostics 2022, 12, 5103–5124. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Dong, W.; Lai, E.; Wang, J. Silk fibroin-based scaffolds for tissue engineering. Front. Bioeng. Biotechnol. 2024, 12, 1381838. [Google Scholar] [CrossRef]
- Lyu, Y.; Liu, Y.; He, H.; Wang, H. Application of Silk-Fibroin-Based Hydrogels in Tissue Engineering. Gels 2023, 9, 431. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Q.; Wan, H.; Zhu, G.; Xiao, Z.; Zhang, Y.; Lei, L.; Li, S. 3D bioprinting in otorhinolaryngology: From bench to clinical application. Int. J. Bioprint. 2024, 10, 3006. [Google Scholar] [CrossRef]
- Benecke, L.; Chen, Z.; Zeidler-Rentzsch, I.; von Witzleben, M.; Bornitz, M.; Zahnert, T.; Neudert, M.; Cherif, C.; Aibibu, D. Development of electrospun, biomimetic tympanic membrane implants with tunable mechanical and oscillatory properties for myringoplasty. Biomater. Sci. 2022, 10, 2287–2301. [Google Scholar] [CrossRef]
- Weatherbee, B.A.T.; Cui, T.; Zernicka-Goetz, M. Modeling human embryo development with embryonic and extra-embryonic stem cells. Dev. Biol. 2021, 474, 91–99. [Google Scholar] [CrossRef]
- Varzideh, F.; Gambardella, J.; Kansakar, U.; Jankauskas, S.S.; Santulli, G. Molecular Mechanisms Underlying Pluripotency and Self-Renewal of Embryonic Stem Cells. Int. J. Mol. Sci. 2023, 24, 8386. [Google Scholar] [CrossRef]
- Lodewijk, G.A.; Kozuki, S.; Han, C.J.; Topacio, B.R.; Lee, S.; Nixon, L.; Zargari, A.; Knight, G.; Ashton, R.; Qi, L.S.; et al. Self-organization of mouse embryonic stem cells into reproducible pre-gastrulation embryo models via CRISPRa programming. Cell Stem Cell 2025, 32, 895–913.e8. [Google Scholar] [CrossRef]
- Wu, J.; Fu, J. Toward developing human organs via embryo models and chimeras. Cell 2024, 187, 3194–3219. [Google Scholar] [CrossRef] [PubMed]
- Mim, T.J.; Ahmad, I.; Basher, S.R.; Rahman, M.F.; Ambati, S.R.; Venkatagiri, K.; Seher, N.; Kumar, D.; Choudhary, N.; Kondaveeti, S.B. Stem cells in organogenesis and regeneration. Stem Cell Res. Ther. 2026, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Zamponi, M.; Mollica, P.A.; Khodour, Y.; Bjerring, J.S.; Bruno, R.D.; Sachs, P.C. Combined 3D bioprinting and tissue-specific ECM system reveals the influence of brain matrix on stem cell differentiation. Front. Cell Dev. Biol. 2023, 11, 1258993. [Google Scholar] [CrossRef] [PubMed]
- de Groot, S.C.; Sliedregt, K.; van Benthem, P.P.G.; Rivolta, M.N.; Huisman, M.A. Building an Artificial Stem Cell Niche: Prerequisites for Future 3D-Formation of Inner Ear Structures—Toward 3D Inner Ear Biotechnology. Anat. Rec. 2020, 303, 408–426. [Google Scholar] [CrossRef]
- Carpena, N.T.; Chang, S.Y.; Abueva, C.D.G.; Jung, J.Y.; Lee, M.Y. Differentiation of embryonic stem cells into a putative hair cell-progenitor cells via co-culture with HEI-OC1 cells. Sci. Rep. 2021, 11, 13893. [Google Scholar] [CrossRef]
- Chang, H.T.; Heuer, R.A.; Oleksijew, A.M.; Coots, K.S.; Roque, C.B.; Nella, K.T.; McGuire, T.L.; Matsuoka, A.J. An engineered three-dimensional stem cell niche in the inner ear by applying a nanofibrillar cellulose hydrogel with a sustained-release neurotrophic factor delivery system. Acta Biomater. 2020, 108, 111–127. [Google Scholar] [CrossRef]
- Aboul-Soud, M.A.M.; Alzahrani, A.J.; Mahmoud, A. Induced Pluripotent Stem Cells (iPSCs)—Roles in Regenerative Therapies, Disease Modelling and Drug Screening. Cells 2021, 10, 2319. [Google Scholar] [CrossRef]
- Owaidah, A.Y. Induced pluripotent stem cells in cartilage tissue engineering: A literature review. Biosci. Rep. 2024, 44, BSR20232102. [Google Scholar] [CrossRef]
- Trillhaase, A.; Maertens, M.; Aherrahrou, Z.; Erdmann, J. Induced Pluripotent Stem Cells (iPSCs) in Vascular Research: From Two- to Three-Dimensional Organoids. Stem Cell Rev. Rep. 2021, 17, 1741–1753. [Google Scholar] [CrossRef]
- Sarker, D.B.; Xue, Y.; Mahmud, F.; Jocelyn, J.A.; Sang, Q.-X.A. Interconversion of Cancer Cells and Induced Pluripotent Stem Cells. Cells 2024, 13, 125. [Google Scholar] [CrossRef]
- Zhao, S.; Chen, J.; Wu, L.; Tao, X.; Yaqub, N.; Chang, J. Induced Pluripotent Stem Cells for Tissue-Engineered Skeletal Muscles. Int. J. Mol. Sci. 2023, 24, 11520. [Google Scholar] [CrossRef] [PubMed]
- Caron, L.; Testa, S.; Magdinier, F. Induced Pluripotent Stem Cells for Modeling Physiological and Pathological Striated Muscle Complexity. J. Neuromuscul. Dis. 2023, 10, 761–776. [Google Scholar] [CrossRef] [PubMed]
- Albert, K.; Niskanen, J.; Kälvälä, S.; Lehtonen, Š. Utilising Induced Pluripotent Stem Cells in Neurodegenerative Disease Research: Focus on Glia. Int. J. Mol. Sci. 2021, 22, 4334. [Google Scholar] [CrossRef] [PubMed]
- Hadzimustafic, N.; D’Elia, A.; Shamoun, V.; Haykal, S. Human-Induced Pluripotent Stem Cells in Plastic and Reconstructive Surgery. Int. J. Mol. Sci. 2024, 25, 1863. [Google Scholar] [CrossRef]
- Reza, H.A.; Santangelo, C.; Iwasawa, K.; Reza, A.A.; Sekiya, S.; Glaser, K.; Bondoc, A.; Merola, J.; Takebe, T. Multi-zonal liver organoids from human pluripotent stem cells. Nature 2025, 641, 1258–1267. [Google Scholar] [CrossRef]
- Zhang, T.; Qian, C.; Song, M.; Tang, Y.; Zhou, Y.; Dong, G.; Shen, Q.; Chen, W.; Wang, A.; Shen, S.; et al. Application Prospect of Induced Pluripotent Stem Cells in Organoids and Cell Therapy. Int. J. Mol. Sci. 2024, 25, 2680. [Google Scholar] [CrossRef]
- van der Valk, W.H.; van Beelen, E.S.A.; Steinhart, M.R.; Nist-Lund, C.; Osorio, D.; de Groot, J.C.M.J.; Sun, L.; van Benthem, P.P.G.; Koehler, K.R.; Locher, H. A single-cell level comparison of human inner ear organoids with the human cochlea and vestibular organs. Cell Rep. 2023, 42, 112623. [Google Scholar] [CrossRef]
- Jeong, M.; Kurihara, S.; Stankovic, K.M. An In Vitro Oxidative Stress Model of the Human Inner Ear Using Human-Induced Pluripotent Stem Cell-Derived Otic Progenitor Cells. Antioxidants 2024, 13, 1407. [Google Scholar] [CrossRef]
- Arkenberg, M.R.; Koehler, K.; Lin, C.C. Heparinized Gelatin-Based Hydrogels for Differentiation of Induced Pluripotent Stem Cells. Biomacromolecules 2022, 23, 4141–4152. [Google Scholar] [CrossRef]
- Cao, Y.; Liu, X.; Chai, R.; He, Z. Inner Ear Organoids: A Hydrogel-Based Platform for Drug Screening and Deafness Modeling. Neurosci. Bull. 2026, 42, 221–232. [Google Scholar] [CrossRef]
- Sun, G.; Tang, M.; Wang, X.; Li, D.; Liu, W.; Qi, J.; Wang, H.; Hu, B. Generation of human otic neuronal organoids using pluripotent stem cells. Cell Prolif. 2023, 56, e13434. [Google Scholar] [CrossRef] [PubMed]
- Doda, D.; Alonso Jimenez, S.; Rehrauer, H.; Carreño, J.F.; Valsamides, V.; Di Santo, S.; Widmer, H.R.; Edge, A.; Locher, H.; van der Valk, W.H.; et al. Human pluripotent stem cell-derived inner ear organoids recapitulate otic development in vitro. Development 2023, 150, dev201865. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gao, J.; Liang, Z.; Gao, C.; Niu, Q.; Wu, F.; Zhang, L. Mesenchymal stem cells and their microenvironment. Stem Cell Res. Ther. 2022, 13, 429. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wu, Q.; Tam, P.K.H. Immunomodulatory Mechanisms of Mesenchymal Stem Cells and Their Potential Clinical Applications. Int. J. Mol. Sci. 2022, 23, 10023. [Google Scholar] [CrossRef]
- Jo, H.; Brito, S.; Kwak, B.M.; Park, S.; Lee, M.-G.; Bin, B.-H. Applications of Mesenchymal Stem Cells in Skin Regeneration and Rejuvenation. Int. J. Mol. Sci. 2021, 22, 2410. [Google Scholar] [CrossRef]
- Zhidu, S.; Ying, T.; Rui, J.; Chao, Z. Translational potential of mesenchymal stem cells in regenerative therapies for human diseases: Challenges and opportunities. Stem Cell Res. Ther. 2024, 15, 266. [Google Scholar] [CrossRef]
- Perde-Schrepler, M.; Brie, I.; Cenariu, M.; Chira, S.; Raduly, L.; Budisan, L.; Berindan-Neagoe, I.; Stiufiuc, R.; Dindelegan, M.; Blebea, C.; et al. Protective Effect of Mesenchymal Stem Cell-Derived Extracellular Vesicles on Inner Ear Sensorineural Cells Affected by Cisplatin. Medicina 2025, 61, 1042. [Google Scholar] [CrossRef]
- Zielinska, D.; Micka-Michalak, K.; Ademi, H.; Fisch, P.; Boeni, R.; Linder, T.; Moehrlen, U.; Biedermann, T.; Klar, A.S. Adipose-mesenchymal stem cells enhance the formation of auricular cartilage in vitro and in vivo. Stem Cells Transl. Med. 2025, 14, szae098. [Google Scholar] [CrossRef]
- Mizuno, K.; Ohnishi, H.; Yoshimatsu, M.; Zhao, C.; Hayashi, Y.; Kuwata, F.; Kaba, S.; Okuyama, H.; Kawai, Y.; Hiwatashi, N.; et al. Laryngeal Cartilage Regeneration of Nude Rats by Transplantation of Mesenchymal Stem Cells Derived from Human-Induced Pluripotent Stem Cells. Cell Transplant. 2023, 32, 9636897231178460. [Google Scholar] [CrossRef]
- Yoshimatsu, M.; Ohnishi, H.; Zhao, C.; Hayashi, Y.; Kuwata, F.; Kaba, S.; Okuyama, H.; Kawai, Y.; Hiwatashi, N.; Kishimoto, Y.; et al. In vivo regeneration of rat laryngeal cartilage with mesenchymal stem cells derived from human induced pluripotent stem cells via neural crest cells. Stem Cell Res. 2021, 52, 102233. [Google Scholar] [CrossRef]
- Iravani, K.; Mehrabani, D.; Doostkam, A.; Azarpira, N.; Iranpour, P.; Bahador, M.; Mehravar, S. Use of Magnetic Resonance Imaging to Assess the Regenerative Effects of Adipose Tissue-Derived Mesenchymal Stem Cells in a Rabbit Cartilaginous Laryngeal Defect Model. Curr. Ther. Res. Clin. Exp. 2022, 97, 100682. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.Z.A.; Muhilan, Y.; Nordin, F.; Ng, M.H.; Law, J.X.; Imran, S.A.M.; Idris, I.M.; Tye, G.J. Stem cell derived exosome trilogy: An epic comparison of human MSCs, ESCs and iPSCs. Stem Cell Res. Ther. 2025, 16, 318. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Ghosh, D.; Otsuka, T.; Laurencin, C.T. Classes of Stem Cells: From Biology to Engineering. Regen. Eng. Transl. Med. 2024, 10, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Tsou, Y.H.; Khoneisser, J.; Huang, P.C.; Xu, X. Hydrogel as a bioactive material to regulate stem cell fate. Bioact. Mater. 2016, 1, 39–55. [Google Scholar] [CrossRef]
- Guvendiren, M.; Burdick, J.A. Engineering synthetic hydrogel microenvironments to instruct stem cells. Curr. Opin. Biotechnol. 2013, 24, 841–846. [Google Scholar] [CrossRef]
- Ma, J.; Huang, C. Composition and Mechanism of Three-Dimensional Hydrogel System in Regulating Stem Cell Fate. Tissue Eng. Part B Rev. 2020, 26, 498–518. [Google Scholar] [CrossRef]
- Heinzelmann, E.; Piraino, F.; Costa, M.; Roch, A.; Norkin, M.; Garnier, V.; Homicsko, K.; Brandenberg, N. iPSC-derived and Patient-Derived Organoids: Applications and challenges in scalability and reproducibility as pre-clinical models. Curr. Res. Toxicol. 2024, 7, 100197. [Google Scholar] [CrossRef]
- Cheng, W.; Fan, C.; Song, Q.; Chen, P.; Peng, H.; Lin, L.; Liu, C.; Wang, B.; Zhou, Z. Induced pluripotent stem cell-based therapies for organ fibrosis. Front. Bioeng. Biotechnol. 2023, 11, 1119606. [Google Scholar] [CrossRef]
- Zhang, X.; Liang, Y.; Luo, D.; Li, P.; Chen, Y.; Fu, X.; Yue, Y.; Hou, R.; Liu, J.; Wang, X. Advantages and disadvantages of various hydrogel scaffold types: A research to improve the clinical conversion rate of loaded MSCs-Exos hydrogel scaffolds. Biomed. Pharmacother. 2024, 179, 117386. [Google Scholar] [CrossRef]
- Wong, R.S.Y.; Tan, E.W.; Goh, B.H. Mesenchymal Stem Cell-Based Therapies: Challenges and Enhancement Strategies. Cell Biochem. Biophys. 2026, 84, 131–147. [Google Scholar] [CrossRef]
- Borciani, G.; Montalbano, G.; Baldini, N.; Vitale-Brovarone, C.; Ciapetti, G. Protocol of Co-Culture of Human Osteoblasts and Osteoclasts to Test Biomaterials for Bone Tissue Engineering. Methods Protoc. 2022, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Brun, P.; Zamuner, A.; Battocchio, C.; Cassari, L.; Todesco, M.; Graziani, V.; Iucci, G.; Marsotto, M.; Tortora, L.; Secchi, V.; et al. Bio-Functionalized Chitosan for Bone Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 5916. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.Y.; Hikita, A.; Asawa, Y.; Hoshi, K. Advancements in chondrocyte 3-dimensional embedded culture: Implications for tissue engineering and regenerative medicine. Biomed. J. 2025, 48, 100786. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Lindsay, S.; Gurbaxani, S.; Crawford, A.; Claeyssens, F. Elastomeric Porous Poly(glycerol sebacate) Methacrylate (PGSm) Microspheres as 3D Scaffolds for Chondrocyte Culture and Cartilage Tissue Engineering. Int. J. Mol. Sci. 2023, 24, 10445. [Google Scholar] [CrossRef]
- Zhou, G.; Jiang, H.; Yin, Z.; Liu, Y.; Zhang, Q.; Zhang, C.; Pan, B.; Zhou, J.; Zhou, X.; Sun, H.; et al. In Vitro Regeneration of Patient-specific Ear-shaped Cartilage and Its First Clinical Application for Auricular Reconstruction. EBioMedicine 2018, 28, 287–302. [Google Scholar] [CrossRef]
- Kaiser, B.; Miot, S.; Wixmerten, A.; Pullig, O.; Eyrich, M.; Fulco, I.; Vavrina, J.; Schaefer, D.J.; Martin, I.; Barbero, A.; et al. Engineered autologous nasal cartilage for repair of nasal septal perforations: A case series. Int. J. Surg. 2024, 110, 6573–6580. [Google Scholar] [CrossRef]
- Sato, W.; Zajkowski, T.; Moser, F.; Adamala, K.P. Synthetic cells in biomedical applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2022, 14, e1761. [Google Scholar] [CrossRef]
- Guindani, C.; da Silva, L.C.; Cao, S.; Ivanov, T.; Landfester, K. Synthetic Cells: From Simple Bio-Inspired Modules to Sophisticated Integrated Systems. Angew. Chem. Int. Ed. Engl. 2022, 61, e202110855. [Google Scholar] [CrossRef]
- Chen, G.; Levin, R.; Landau, S.; Kaduri, M.; Adir, O.; Ianovici, I.; Krinsky, N.; Doppelt-Flikshtain, O.; Shklover, J.; Shainsky-Roitman, J.; et al. Implanted synthetic cells trigger tissue angiogenesis through de novo production of recombinant growth factors. Proc. Natl. Acad. Sci. USA 2022, 119, e2207525119. [Google Scholar] [CrossRef]
- Shim, J.; Zhou, C.; Gong, T.; Iserlis, D.A.; Linjawi, H.A.; Wong, M.; Pan, T.; Tan, C. Building protein networks in synthetic systems from the bottom-up. Biotechnol. Adv. 2021, 49, 107753. [Google Scholar] [CrossRef]
- Maia, R.F.; Vaziri, A.S.; Shahbazi, M.A.; Santos, H.A. Artificial cells and biomimicry cells: A rising star in the fight against cancer. Mater. Today Bio 2025, 32, 101723. [Google Scholar] [CrossRef]
- Reyes Valenzuela, A.; Bao, G.; Vikstrom, A.; Kost, K.M.; Prakash, S.; Mongeau, L. Polymeric Microspheres Containing Human Vocal Fold Fibroblasts for Vocal Fold Regeneration. Laryngoscope 2021, 131, 1828–1834. [Google Scholar] [CrossRef] [PubMed]
- Yeou, S.H.; Shin, Y.S. Regenerative Strategies for Vocal Fold Repair Using Injectable Materials. Biomimetics 2025, 10, 748. [Google Scholar] [CrossRef] [PubMed]
- Rook, O.; Zwart, H.; Dogterom, M. Public attitudes to potential synthetic cells applications: Pragmatic support and ethical acceptance. PLoS ONE 2025, 20, e0319337. [Google Scholar] [CrossRef] [PubMed]
- Kurtoğlu, A.; Yıldız, A.; Arda, B. The view of synthetic biology in the field of ethics: A thematic systematic review. Front. Bioeng. Biotechnol. 2024, 12, 1397796. [Google Scholar] [CrossRef]
- Bohua, L.; Yuexin, W.; Yakun, O.; Kunlan, Z.; Huan, L.; Ruipeng, L. Ethical framework on risk governance of synthetic biology. J. Biosaf. Biosecur. 2023, 5, 45–56. [Google Scholar] [CrossRef]
- Zhang, J.; Wehrle, E.; Rubert, M.; Müller, R. 3D Bioprinting of Human Tissues: Biofabrication, Bioinks, and Bioreactors. Int. J. Mol. Sci. 2021, 22, 3971. [Google Scholar] [CrossRef]
- Panja, N.; Maji, S.; Choudhuri, S.; Ali, K.A.; Hossain, C.M. 3D Bioprinting of Human Hollow Organs. AAPS PharmSciTech 2022, 23, 139. [Google Scholar] [CrossRef]
- Zennifer, A.; Manivannan, S.; Sethuraman, S.; Kumbar, S.G.; Sundaramurthi, D. 3D bioprinting and photocrosslinking: Emerging strategies & future perspectives. Biomater. Adv. 2022, 134, 112576. [Google Scholar]
- Lam, E.H.Y.; Yu, F.; Zhu, S.; Wang, Z. 3D Bioprinting for Next-Generation Personalized Medicine. Int. J. Mol. Sci. 2023, 24, 6357. [Google Scholar] [CrossRef]
- Mladenovska, T.; Choong, P.F.; Wallace, G.G.; O’Connell, C.D. The regulatory challenge of 3D bioprinting. Regen. Med. 2023, 18, 659–674. [Google Scholar] [CrossRef]
- Xie, M.; Su, J.; Zhou, S.; Li, J.; Zhang, K. Application of Hydrogels as Three-Dimensional Bioprinting Ink for Tissue Engineering. Gels 2023, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Dell, A.C.; Wagner, G.; Own, J.; Geibel, J.P. 3D Bioprinting Using Hydrogels: Cell Inks and Tissue Engineering Applications. Pharmaceutics 2022, 14, 2596. [Google Scholar] [CrossRef] [PubMed]
- Hull, S.M.; Brunel, L.G.; Heilshorn, S.C. 3D Bioprinting of Cell-Laden Hydrogels for Improved Biological Functionality. Adv. Mater. 2022, 34, e2103691. [Google Scholar] [CrossRef] [PubMed]
- Loukelis, K.; Helal, Z.A.; Mikos, A.G.; Chatzinikolaidou, M. Nanocomposite Bioprinting for Tissue Engineering Applications. Gels 2023, 9, 103. [Google Scholar] [CrossRef]
- Al Qassab, M.; Merheb, M.; Sayadi, S.; Salloum, P.; Dabbousi, Z.; Bayeh, A.; Harb, F.; Azar, S.; Ghadieh, H.E. Organ-Specific Strategies in Bioprinting: Addressing Translational Challenges in the Heart, Liver, Kidney, and Pancreas. J. Funct. Biomater. 2025, 16, 356. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, X.; Shan, M.; Hao, Z.; Zhang, X.; Meng, L.; Zhai, Z.; Zhang, L.; Liu, X.; Wang, X. Convergence of 3D Bioprinting and Nanotechnology in Tissue Engineering Scaffolds. Biomimetics 2023, 8, 94. [Google Scholar] [CrossRef]
- Finny, A.S. 3D bioprinting in bioremediation: A comprehensive review of principles, applications, and future directions. PeerJ 2024, 12, e16897. [Google Scholar] [CrossRef]
- Kantaros, A. 3D Printing in Regenerative Medicine: Technologies and Resources Utilized. Int. J. Mol. Sci. 2022, 23, 14621. [Google Scholar] [CrossRef]
- Jeon, Y.; Kim, M.; Song, K.H. Development of Hydrogels Fabricated via Stereolithography for Bioengineering Applications. Polymers 2025, 17, 765. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, Y.; Zhong, L.; Pan, F.; Wang, J. Advances in tissue engineering of vasculature through three-dimensional bioprinting. Dev. Dyn. 2021, 250, 1717–1738. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, X.; Li, Y.; Zhang, Y. Applications of Light-Based 3D Bioprinting and Photoactive Biomaterials for Tissue Engineering. Materials 2023, 16, 7461. [Google Scholar] [CrossRef] [PubMed]
- Khalak, F.A.; Decuyper, J.M.; Khalak, K.A.; Alonso, S.R.; Saenz-Del-Burgo, L.; Pedraz Muñoz, J.L. 3D bioprinting approaches for musculoskeletal interfaces in tissue engineering. Int. J. Pharm. 2025, 682, 125939. [Google Scholar] [CrossRef] [PubMed]
- Tharakan, S.; Khondkar, S.; Ilyas, A. Bioprinting of Stem Cells in Multimaterial Scaffolds and Their Applications in Bone Tissue Engineering. Sensors 2021, 21, 7477. [Google Scholar] [CrossRef]
- Taylor, S.; Mueller, E.; Jones, L.R.; Makela, A.V.; Ashammakhi, N. Translational Aspects of 3D and 4D Printing and Bioprinting. Adv. Healthc. Mater. 2024, 13, e2400463. [Google Scholar] [CrossRef]
- Lai, J.; Liu, Y.; Lu, G.; Yung, P.; Wang, X.; Tuan, R.S.; Li, Z.A. 4D bioprinting of programmed dynamic tissues. Bioact. Mater. 2024, 37, 348–377. [Google Scholar] [CrossRef]
- Kang, X.; Zhang, X.B.; Gao, X.D.; Hao, D.J.; Li, T.; Xu, Z.W. Bioprinting for bone tissue engineering. Front. Bioeng. Biotechnol. 2022, 10, 1036375. [Google Scholar] [CrossRef]
- Mathur, V.; Agarwal, P.; Kasturi, M.; Varadharajan, S.; Devi, E.S.; Vasanthan, K.S. Transformative bioprinting: 4D printing and its role in the evolution of engineering and personalized medicine. Discov. Nano 2025, 20, 118. [Google Scholar] [CrossRef]
- Kim, J.; D A, G.; Debnath, P.; Saha, P. Smart Multi-Responsive Biomaterials and Their Applications for 4D Bioprinting. Biomimetics 2024, 9, 484. [Google Scholar] [CrossRef]
- Faber, L.; Yau, A.; Chen, Y. Translational biomaterials of four-dimensional bioprinting for tissue regeneration. Biofabrication 2023, 16, 012001. [Google Scholar] [CrossRef]
- Vyas, J.; Raytthatha, N.; Vyas, P.; Patel, J. Personalized solutions for ENT implants: The role of 3D/4D printing. Braz. J. Pharm. Sci. 2025, 61, e24308. [Google Scholar] [CrossRef]
- Nazir, A.; Nazir, A.; Khan, N.; Jamal, M.S.W.; Shubietah, A.; Naqvi, I.; Rafay Hussain Zaidi, S.; Khan, B.; Ghumman, G.M.; Jamal Chaudhary, A.; et al. Smart bioactive hydrogels for myocardial infarction repair: A multifunctional approach integrating stimuli-responsive drug delivery, electroconductivity, and real-time biosensing. Ann. Med. Surg. 2025, 88, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Wistner, S.C.; Rashad, L.; Slaughter, G. Advances in tissue engineering and biofabrication for in vitro skin modeling. Bioprinting 2023, 35, e00306. [Google Scholar] [CrossRef] [PubMed]
- Malekmohammadi, S.; Sedghi Aminabad, N.; Sabzi, A.; Zarebkohan, A.; Razavi, M.; Vosough, M.; Bodaghi, M.; Maleki, H. Smart and Biomimetic 3D and 4D Printed Composite Hydrogels: Opportunities for Different Biomedical Applications. Biomedicines 2021, 9, 1537. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, Y.S.; Chaudhari, M.Y.; Gholap, A.D.; Alam, M.I.; Khalid, M.; Webster, T.J.; Gowri, S.; Faiyazuddin, M. Surface engineering of nano magnesium alloys for orthopedic implants: A systematic review of strategies to mitigate corrosion and promote bone regeneration. Front. Bioeng. Biotechnol. 2025, 13, 1617585. [Google Scholar] [CrossRef]
- Bhandari, A.; Ghosh, R.N.; Namboothiri, P.K.; Peter, M. A review of stimuli-responsive materials in 4D bioprinting for biomedical applications. Mater. Adv. 2025, 7, 17–39. [Google Scholar] [CrossRef]
- Aftab, M.; Ikram, S.; Ullah, M.; Khan, S.U.; Wahab, A.; Naeem, M. Advancement of 3D Bioprinting Towards 4D Bioprinting for Sustained Drug Delivery and Tissue Engineering from Biopolymers. J. Manuf. Mater. Process. 2025, 9, 285. [Google Scholar] [CrossRef]
- Li, X.; Ren, J.; Huang, Y.; Cheng, L.; Gu, Z. Applications and Recent Advances in 3D Bioprinting Sustainable Scaffolding Techniques. Molecules 2025, 30, 3027. [Google Scholar] [CrossRef]
- Bakhtiari, H.; Nouri, A.; Aamir, M.; Najafi, M.; Tolouei-Rad, M. Impact of biodegradation on the mechanical and fatigue properties of 3D-printed PLA bone scaffolds. J. Mech. Behav. Biomed. Mater. 2025, 168, 107039. [Google Scholar] [CrossRef]
- Qi, H.; Zhang, B.; Lian, F. 3D-printed bioceramic scaffolds for bone defect repair: Bone aging and immune regulation. Front. Bioeng. Biotechnol. 2025, 13, 1557203. [Google Scholar] [CrossRef]
- Yang, D.; Xiao, J.; Wang, B.; Li, L.; Kong, X.; Liao, J. The immune reaction and degradation fate of scaffold in cartilage/bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 109927. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yuan, B.; Yin, R.; Zhang, H. Inflammation Responses to Bone Scaffolds under Mechanical Stimuli in Bone Regeneration. J. Funct. Biomater. 2023, 14, 169. [Google Scholar] [CrossRef] [PubMed]
- Najafi, N.; Vartanian, K.B.; Eskandar, T.; Ghookas, K.; Rostomian, E.; Agrawal, D.K. Bioprinting for craniofacial reconstruction: A review of advancements, clinical use, and challenges. J. Craniomaxillofac. Surg. 2025, 53, 2255–2269. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, G.; Lin, H.; Shang, Y.; Liu, N.; Zhen, Y.; An, Y. Cartilage 3D bioprinting for rhinoplasty using adipose-derived stem cells as seed cells: Review and recent advances. Cell Prolif. 2023, 56, e13417. [Google Scholar] [CrossRef]
- Chowdhury, R.; Al-Musaileem, N.; Raman, K.S.; Al-Majid, D.; Solomon, P.; Rival, R. 3D Printing in Nasal Reconstruction: Application-Based Evidence on What Works, When, and Why. Biomedicines 2025, 13, 1434. [Google Scholar] [CrossRef]
- Pham, T.B.; Sah, R.L.; Masuda, K.; Watson, D. Human Septal Cartilage Tissue Engineering: Current Methodologies and Future Directions. Bioengineering 2024, 11, 1123. [Google Scholar] [CrossRef]
- Farahani, P.K. Application of Tissue Engineering and Biomaterials in Nose Surgery. JPRAS Open 2023, 40, 262–272. [Google Scholar] [CrossRef]
- Lan, X.; Liang, Y.; Erkut, E.J.N.; Kunze, M.; Mulet-Sierra, A.; Gong, T.; Osswald, M.; Ansari, K.; Seikaly, H.; Boluk, Y.; et al. Bioprinting of human nasoseptal chondrocytes-laden collagen hydrogel for cartilage tissue engineering. FASEB J. 2021, 35, e21191. [Google Scholar] [CrossRef]
- Lan, X.; Liang, Y.; Vyhlidal, M.; Erkut, E.J.; Kunze, M.; Mulet-Sierra, A.; Osswald, M.; Ansari, K.; Seikaly, H.; Boluk, Y.; et al. In vitro maturation and in vivo stability of bioprinted human nasal cartilage. J. Tissue Eng. 2022, 13, 20417314221086368. [Google Scholar] [CrossRef]
- Deniz Derman, I.; Yeo, M.; Castaneda, D.C.; Callender, M.; Horvath, M.; Mo, Z.; Xiong, R.; Fleming, E.; Chen, P.; Peeples, M.E.; et al. High-throughput bioprinting of the nasal epithelium using patient-derived nasal epithelial cells. Biofabrication 2023, 15, 044103. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Y.; Yu, Y. Establishment of nasal and olfactory epithelium organoids for unveiling mechanism of tissue regeneration and pathogenesis of nasal diseases. Cell. Mol. Life Sci. 2025, 82, 33. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Li, W.; Zhuang, L.; Liu, J.; Wang, P.; Gu, Y.; Liu, Y.; Yu, Y. Deficiency of Aging-Related Gene Chitinase-Like 4 Impairs Olfactory Epithelium Homeostasis. Cell Prolif. 2025, 58, e70055. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, T.; Zhu, K.; Ba, G.; Liu, J.; Zhou, P.; Li, S.; Wang, L.; Liu, H.; Ren, W.; et al. A single-cell transcriptomic census of mammalian olfactory epithelium aging. Dev. Cell. 2024, 59, 3043–3058.e8. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, R.; Yadav, P.K.; Pandey, R.; Mehrotra, D. Auricular reconstruction via 3D bioprinting strategies: An update. J. Oral Biol. Craniofac. Res. 2022, 12, 580–588. [Google Scholar] [CrossRef]
- Lu, W.; Yang, M.; Zhang, Y.; Meng, B.; Ma, F.; Wang, W.; Guo, T. Characterization of Acellular Cartilage Matrix-Sodium Alginate Scaffolds in Various Proportions. Tissue Eng. Part C Methods 2024, 30, 170–182. [Google Scholar] [CrossRef]
- Kong, Y.; Lu, Z.; Zhan, J.; Zhou, X.; Chen, S.; Chen, Q.; Gong, H.; Zhang, X.; Mao, X.; Wang, Y.; et al. Enhancing auricular reconstruction: A biomimetic scaffold with 3D-printed multiscale porous structure utilizing chondrogenic activity ink. Mater. Today Bio 2025, 31, 101516. [Google Scholar] [CrossRef]
- Gvaramia, D.; Fisch, P.; Flégeau, K.; Huber, L.; Kern, J.; Jakob, Y.; Hirsch, D.; Rotter, N. Evaluation of Bioprinted Autologous Cartilage Grafts in an Immunocompetent Rabbit Model. Adv. Ther. 2024, 7, adtp.202300441. [Google Scholar] [CrossRef]
- Posniak, S.; Chung, J.H.Y.; Liu, X.; Mukherjee, P.; Gambhir, S.; Khansari, A.; Wallace, G.G. Bioprinting of Chondrocyte Stem Cell Co-Cultures for Auricular Cartilage Regeneration. ACS Omega 2022, 7, 5908–5920. [Google Scholar] [CrossRef]
- Jia, L.; Liu, S.; Gu, L.; Liu, X.; Sun, K.; Chu, F.; Zeng, J.; Liu, W.; Jiang, H.; Liu, X. Integrated biomimetic bioprinting of perichondrium with cartilage for auricle reconstruction. Bioact. Mater. 2025, 48, 100–117. [Google Scholar] [CrossRef]
- Jia, L.; Hua, Y.; Zeng, J.; Liu, W.; Wang, D.; Zhou, G.; Liu, X.; Jiang, H. Bioprinting and regeneration of auricular cartilage using a bioactive bioink based on microporous photocrosslinkable acellular cartilage matrix. Bioact. Mater. 2022, 16, 66–81. [Google Scholar] [CrossRef]
- Chen, X.; Hu, H.; Yang, J.; Wang, Y.; Yue, W.; Xing, P.; Zhang, Y.; Chen, J.; Xu, T.; Li, Y. 3D bioprinted composite scaffold incorporating microfluidics-derived chondrocyte microspheroids promotes auricular cartilage regeneration. Mater. Today Bio 2026, 37, 102826. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Jia, L.; Wang, D.; Chen, Z.; Liu, W.; Yang, Q.; Liu, X.; Jiang, H. Bacterial nanocellulose-reinforced gelatin methacryloyl hydrogel enhances biomechanical property and glycosaminoglycan content of 3D-bioprinted cartilage. Int. J. Bioprint. 2022, 9, 631. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Wu, S.; Mou, S.; Guo, N.; Wang, Z.; Sun, J. Microtissue-Based Bioink as a Chondrocyte Microshelter for DLP Bioprinting. Adv. Healthc. Mater. 2022, 11, e2201877. [Google Scholar] [CrossRef] [PubMed]
- Xue, H.; Chen, S.; Hu, Y.; Huang, J.; Shen, Y. Advances in 3D printing for the repair of tympanic membrane perforation: A comprehensive review. Front. Bioeng. Biotechnol. 2024, 12, 1439499. [Google Scholar] [CrossRef]
- Timofticiuc, I.-A.; Caruntu, A.; Dragosloveanu, C.D.M.; Scheau, A.-E.; Badarau, I.A.; Periferakis, A.; Dragosloveanu, S.; Didilescu, A.C.; Caruntu, C.; Scheau, C. Head and Neck 3D Bioprinting—A Review on Recent Advancements in Soft Tissue 3D Bioprinting and Medical Applications. J. Funct. Biomater. 2025, 16, 240. [Google Scholar] [CrossRef]
- Wang, Y.; Li, H.; Zhang, J.; Chen, M.; Pan, Y.; Lou, X. 3D Bioprinting Inner Ear Organ of Corti Organoids Induce Hair Cell Regeneration. J. Biomed. Mater. Res. A 2025, 113, e37892. [Google Scholar] [CrossRef]
- Xia, M.; Wu, M.; Li, Y.; Liu, Y.; Jia, G.; Lou, Y.; Ma, J.; Gao, Q.; Xie, M.; Chen, Y.; et al. Varying mechanical forces drive sensory epithelium formation. Sci. Adv. 2023, 9, eadf2664. [Google Scholar] [CrossRef]
- Lou, Y.; Liu, Y.; Wu, M.; Jia, G.; Xia, M.; Li, W. Inner ear organoids: Recent progress and potential applications. Fundam. Res. 2023, 5, 2926–2936. [Google Scholar] [CrossRef]
- Wu, M.; Jia, G.; Liu, Y.; Lou, Y.; Li, Y.; Xia, M.; Li, H.; Li, W. PKM2 controls cochlear development through lactate-dependent transcriptional regulation. Proc. Natl. Acad. Sci. USA 2025, 122, e2410829122. [Google Scholar] [CrossRef]
- Xia, M.; Ma, J.; Wu, M.; Guo, L.; Chen, Y.; Li, G.L.; Sun, S.; Chai, R.; Li, H.; Li, W. Generation of innervated cochlear organoid recapitulates early development of auditory unit. Stem Cell Rep. 2023, 18, 319–336. [Google Scholar] [CrossRef]
- Yeou, S.H.; Shin, Y.S. Tissue-Engineered Tracheal Reconstruction. Biomimetics 2025, 10, 457. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, A.C.; Machado Holzlsauer, G.; Ruiz Lucio de Lima Parra, J.P.; Querino Candelária, R.A.; Santos da Silva, T.; da Silva Nunes Barreto, R.; Miglino, M.A. Organic and Synthetic Substitutes in Tracheal Reconstruction: A Scoping Review (2015–2025). Bioengineering 2025, 12, 704. [Google Scholar] [CrossRef] [PubMed]
- Lee, O.J.; Lee, H.J.; Lee, J.S.; Oh, M.S.; Jung, H.; Kim, H.; Park, C.H.; Lee, I.; Yang, J.; Lee, J.J.; et al. Evaluation Strategies for Tissue-engineered Tracheas: From In Vitro Characterization to In Vivo Assessment. In Vivo 2025, 39, 2490–2504. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Wei, S.; Zhang, A. Bioengineered tracheal graft with enhanced vascularization and mechanical stability for functional airway reconstruction. Regen. Ther. 2025, 29, 364–380. [Google Scholar] [CrossRef]
- Lee, H.-Y.; Lee, J.W. Current Status and Future Outlook of Additive Manufacturing Technologies for the Reconstruction of the Trachea. J. Funct. Biomater. 2023, 14, 196. [Google Scholar] [CrossRef]
- Yu, Y.S.; Ahn, C.B.; Son, K.H.; Lee, J.W. Motility Improvement of Biomimetic Trachea Scaffold via Hybrid 3D-Bioprinting Technology. Polymers 2021, 13, 971. [Google Scholar] [CrossRef]
- Park, J.H.; Ahn, M.; Park, S.H.; Kim, H.; Bae, M.; Park, W.; Hollister, S.J.; Kim, S.W.; Cho, D.W. 3D bioprinting of a trachea-mimetic cellular construct of a clinically relevant size. Biomaterials 2021, 279, 121246. [Google Scholar] [CrossRef]
- Sun, Y.; Huo, Y.; Ran, X.; Chen, H.; Pan, Q.; Chen, Y.; Zhang, Y.; Ren, W.; Wang, X.; Zhou, G.; et al. Instant trachea reconstruction using 3D-bioprinted C-shape biomimetic trachea based on tissue-specific matrix hydrogels. Bioact. Mater. 2023, 32, 52–65. [Google Scholar] [CrossRef]
- Huo, Y.; Xu, Y.; Wu, X.; Gao, E.; Zhan, A.; Chen, Y.; Zhang, Y.; Hua, Y.; Swieszkowski, W.; Zhang, Y.S.; et al. Functional Trachea Reconstruction Using 3D-Bioprinted Native-Like Tissue Architecture Based on Designable Tissue-Specific Bioinks. Adv. Sci. 2022, 9, e2202181. [Google Scholar] [CrossRef]
- Zheng, T.; Liu, W.; Liu, S.; Tan, S.; Wang, P.; Li, T.; Chu, F.; Liu, J.; Jiang, H.; Liu, X. Integrated bioprinting of trachea-like structures based on tissue-specific bioink. Mater. Today Bio 2025, 34, 102105. [Google Scholar] [CrossRef]
- McMillan, A.; Hoffman, M.R.; Xu, Y.; Wu, Z.; Thayer, E.; Peel, A.; Guymon, A.; Kanotra, S.; Salem, A.K. 3D bioprinted ferret mesenchymal stem cell-laden cartilage grafts for laryngotracheal reconstruction in a ferret surgical model. Biomater. Sci. 2025, 13, 1304–1322. [Google Scholar] [CrossRef]
- Jessop, Z.M.; Hague, A.; Dobbs, T.D.; Stewart, K.J.; Whitaker, I.S. Facial Cartilaginous Reconstruction-A Historical Perspective, State-of-the-Art, and Future Directions. Front. Surg. 2021, 8, 680186. [Google Scholar] [CrossRef] [PubMed]
- Tanveer, W.; Ridwan-Pramana, A.; Molinero-Mourelle, P.; Koolstra, J.H.; Forouzanfar, T. Systematic Review of Clinical Applications of CAD/CAM Technology for Craniofacial Implants Placement and Manufacturing of Nasal Prostheses. Int. J. Environ. Res. Public Health 2021, 18, 3756. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.A.; Baptista, B.; Damaso, R.; Castanheira, A. Staged Paramedian Forehead Flap Reconstruction of a Large Oncologic Nasal Defect: A Case Report. Cureus 2025, 17, e90925. [Google Scholar] [CrossRef] [PubMed]
- Gigov, K.; Ginev, I.; Minev, I.; Kavradzhieva, P. Three-Layer Reconstruction of a Full-Thickness Nasal Alar Defect after Basal-Cell Carcinoma Removal. Reports 2024, 7, 75. [Google Scholar] [CrossRef]
- Pereira, F.; Martins, S.; Cebotari, M.; Coelho, L. Paramedian Frontal Flap Reconstruction for Nasal Defect Following an Accidental Amputation. Cureus 2024, 16, e61167. [Google Scholar] [CrossRef]
- Ureel, M.; Dadjam, D.; Dhooghe, N.; De Jong, M.; Coopman, R. Case Report: Pediatric alloplastic nose reconstruction with a 3D printed patient specific titanium implant. Front. Surg. 2024, 11, 1330889. [Google Scholar] [CrossRef]
- Mayo, W.; Mohamad, A.H.; Zazo, H.; Zazo, A.; Alhashemi, M.; Meslmany, A.; Haddad, B. Facial defects reconstruction by titanium mesh bending using 3D printing technology: A report of two cases. Ann. Med. Surg. 2022, 78, 103837. [Google Scholar] [CrossRef]
- Scott, B.L.; Wax, M.K.; Gerecci, D.; Loyo, M. Case Report of Delayed Auricular Reconstruction Using a Porous Polyethylene Implant for an Advanced Skin Cancer. Ear Nose Throat J. 2022, 101, 165–166. [Google Scholar] [CrossRef]
- Khurayzi, T.; Abdalla, H.K. Vibrant Soundbridge and Porous Polyethylene Auricular Reconstruction in a Single-Stage Procedure. Sci. Prog. 2026, 109, 368504251412579. [Google Scholar] [CrossRef]
- Ahmad, A.F. Reconstruction of Nasal Deformity Resulting from Mucormycosis Using a Chin Silicone Implant. Plast. Reconstr. Surg. Glob. Open 2024, 12, e6088. [Google Scholar] [CrossRef]
- Lee, S.B.; Min, H.J. Spontaneous Fracture of a Silicone Implant: A Delayed Complication of Rhinoplasty. Ear Nose Throat J. 2025, 104, 355S–358S. [Google Scholar] [CrossRef] [PubMed]
- Moris, V.; Zwetyenga, N.; Habre, S.B. Biomechanical and biological advantages of retaining the silicone-induced capsule for cartilage graft survival in revision rhinoplasty. J. Stomatol. Oral Maxillofac. Surg. 2024, 125, 101935. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Wang, C.; Luan, F.; Pan, B. Comparison of auricle reconstruction with expanded flaps and auricle reconstruction with non-expanded flaps in patients with microtia: A meta-analysis. Ear Nose Throat J. 2024, 103, NP351–NP359. [Google Scholar] [CrossRef] [PubMed]
- Behar, B.J.; Mackay, D.R. Brent technique for microtia reconstruction. Oper. Techn. Otolaryngol. Head Neck Surg. 2017, 28, 77–83. [Google Scholar] [CrossRef]
- Rendón-Medina, M.A.; Hanson-Viana, E.; Arias-Salazar, L.; Rojas-Ortiz, J.A.; Mendoza-Velez, M.L.A.; Hernandez-Ordoñez, R.; Vázquez-Morales, H.L.; Pacheco-López, R.C. Auricular Total Reconstruction with Radial Forearm Prelaminated Flap Assisted by 3D Surface Imaging and 3D Printing. Plast. Reconstr. Surg. Glob. Open 2022, 10, e4580. [Google Scholar] [CrossRef]
- Walton, R.L.; Seelaus, R.; Robinson, B.R. Subtotal Nasal Reconstruction Using a Custom 3-Dimensional Porous Polyethylene Construct. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2568. [Google Scholar] [CrossRef]
- Ziegler, J.P.; Oyer, S.L. Prelaminated paramedian forehead flap for subtotal nasal reconstruction using three-dimensional printing. BMJ Case Rep. 2021, 14, e238146. [Google Scholar] [CrossRef]
- Borghi, A.; Ruggiero, F.; Tenhagen, M.; Schievano, S.; Ponniah, A.; Dunaway, D.; O’Hara, J.; Ong, J.; Britto, J.A. Design and manufacturing of a patient-specific nasal implant for congenital arhinia: Case report. JPRAS Open 2019, 21, 28–34. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z.; Tu, C.; Chen, X.; He, R. Advanced Glycation End Products in Disease Development and Potential Interventions. Antioxidants 2025, 14, 492. [Google Scholar] [CrossRef]
- Kammona, O.; Tsanaktsidou, E.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10, 147. [Google Scholar] [CrossRef] [PubMed]
- Diamantides, N.; Slyker, L.; Martin, S.; Rodriguez, M.R.; Bonassar, L.J. Pre-glycation impairs gelation of high concentration collagen solutions. J. Biomed. Mater. Res. A 2022, 110, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.T.; Liang, K.; Ngo, Z.H.; Dube, C.T.; Lim, C.Y. Application of 3D Bioprinting Technologies to the Management and Treatment of Diabetic Foot Ulcers. Biomedicines 2020, 8, 441. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.; Cho, J.K.; Bong, K.W.; Song, S.C. Thermo-responsive Bioink for Personalized 3D Printed Scaffolds with Antioxidant and Fibroblast Delivery to Accelerate Diabetic Wound Healing. Biomater. Res. 2025, 29, 0216. [Google Scholar] [CrossRef]
- Jiang, P.; Li, Q.; Luo, Y.; Luo, F.; Che, Q.; Lu, Z.; Yang, S.; Yang, Y.; Chen, X.; Cai, Y. Current status and progress in research on dressing management for diabetic foot ulcer. Front. Endocrinol. 2023, 14, 1221705. [Google Scholar] [CrossRef]
- Sathisaran, I. 3D printing and bioprinting in the battle against diabetes and its chronic complications. Front. Bioeng. Biotechnol. 2024, 12, 1363483. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, Y.; Xu, B.; Liu, H.; Chang, Q. Microenvironmental dynamics of diabetic wounds and insights for hydrogel-based therapeutics. J. Tissue Eng. 2024, 15, 20417314241253290. [Google Scholar] [CrossRef]
- Echeverria Molina, M.I.; Malollari, K.G.; Komvopoulos, K. Design Challenges in Polymeric Scaffolds for Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 9, 617141. [Google Scholar] [CrossRef]
- Becquart, P.; Cambon-Binder, A.; Monfoulet, L.E.; Bourguignon, M.; Vandamme, K.; Bensidhoum, M.; Petite, H.; Logeart-Avramoglou, D. Ischemia is the prime but not the only cause of human multipotent stromal cell death in tissue-engineered constructs in vivo. Tissue Eng. Part A 2012, 18, 2084–2094. [Google Scholar] [CrossRef]
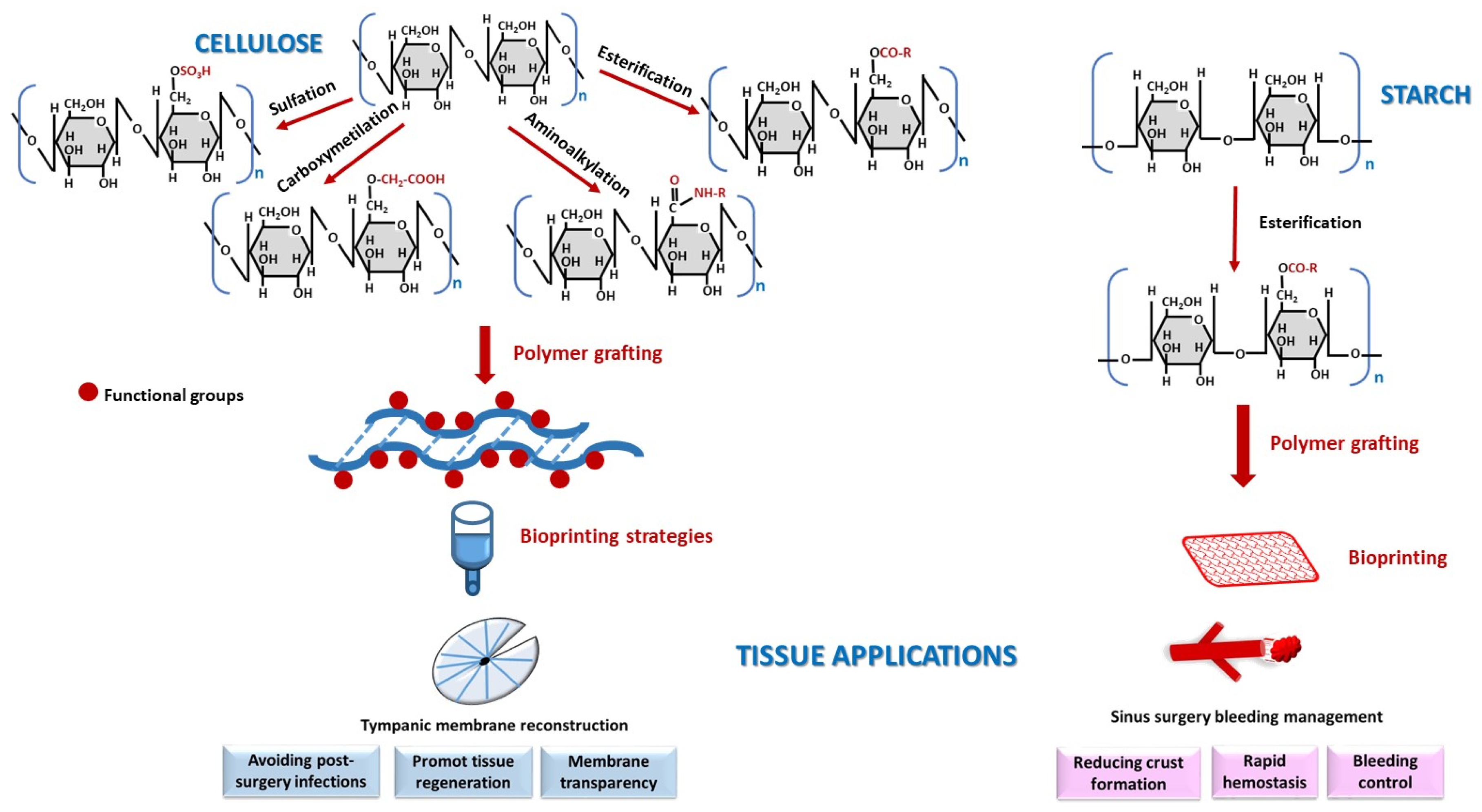
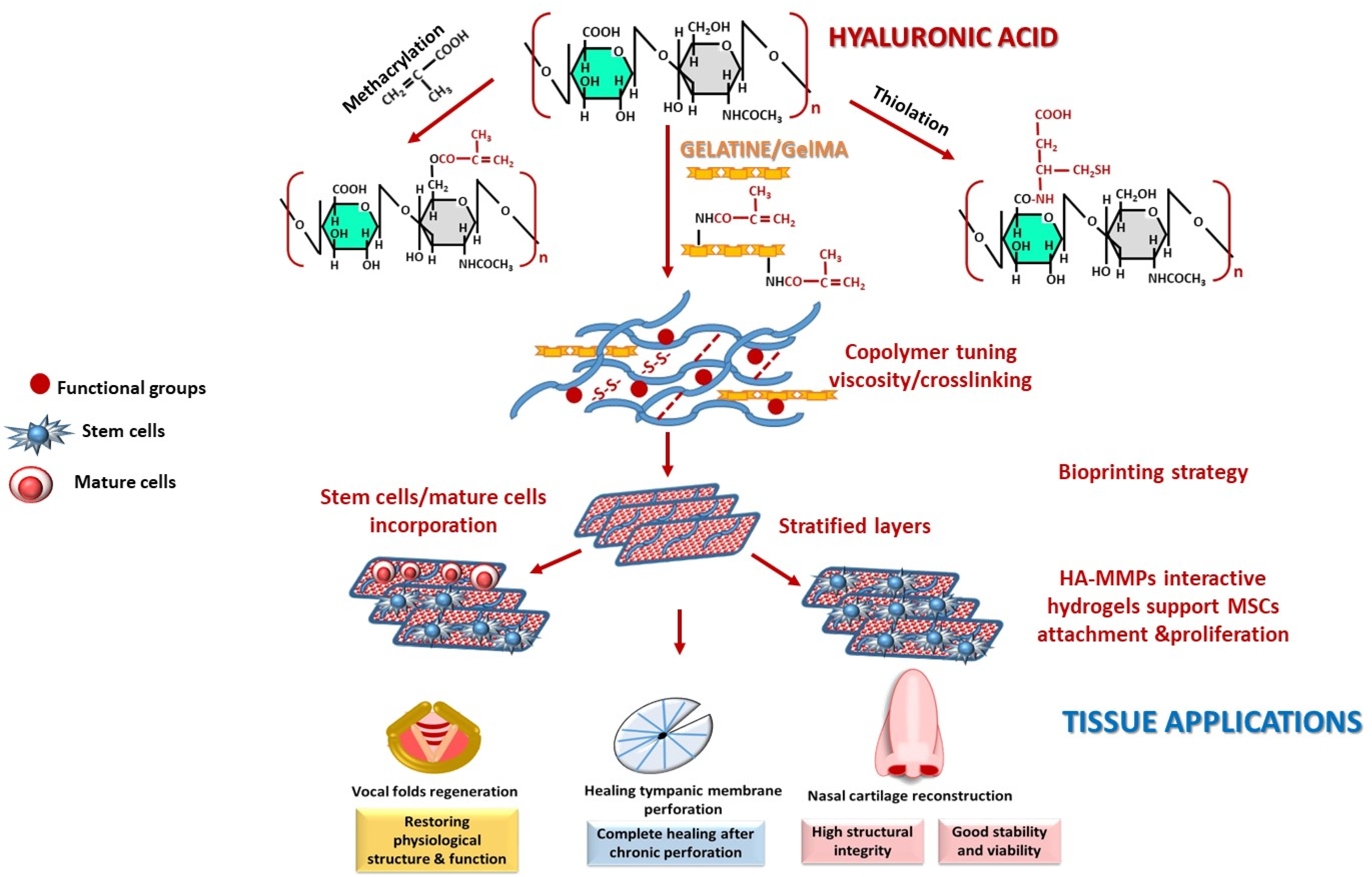
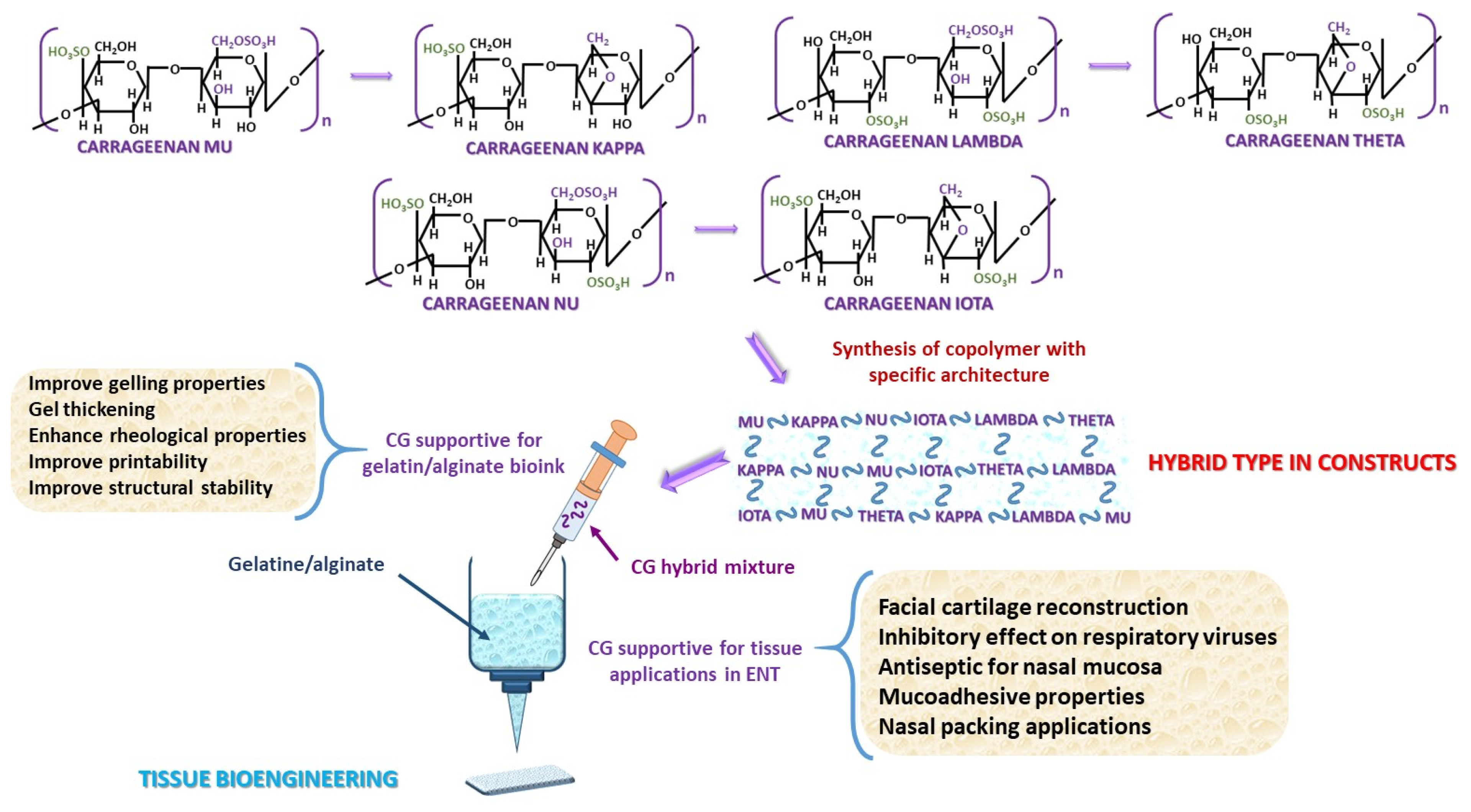




| Stem Cells | Applications | Advantages | Limitations | Cell-Scaffold Interactions |
|---|---|---|---|---|
| ESCs | Enable reconstructive surgery of the vocal folds using epithelial cells derived from ESCs [111]; Support reconstructive surgery of the inner ear [154]; Treat hearing loss through the differentiation of ESCs into inner ear hair cells [155]; Allow for the in vivo transplantation of ESCs-derived otic neuronal progenitors within cellulose hydrogels [156]. | Possess the capacity to differentiate into any somatic cell [151,152]; Functionally create architecture with vascular network and complex neuronal connections [153]; Express pluripotent factors such as OCT4, SOX2 [182]; Exhibit self-renewal capacity [183]. | Carry a risk of rejection or tumor formation [40]; Require appropriate growth media and modulation with transcription factors [150]; Involve ethical controversies that lead to restrictive use, depending on legislation [182]. | Modulate the self-renewal properties of ESCs using HA-Tyr hydrogels [184]; Maintain ESCs in an undifferentiated state through encapsulation in HA hydrogels, and HA binding sites influence ESC receptors [185]; Promote the differentiation of ESCs into neural precursor cells using Matrigel [186]; Induce the transformation of ESCs into iPSCs using gelatin gels [169]. |
| iPSCs | Differentiate into a wide range of tissues, including cartilage [158], blood vessels [159], skeletal muscle [161,162], glial cells [163], and skin [164]; Support the development of complex organoids [165,166]; Modulate the ototoxic effects of gentamicin and cisplatin on the inner ear [168]; Treat hearing loss using IEOs derived from iPSCs [170]. | Differentiation into specific mature cells through induction with OCT4, SOX2, KLF4 and c-MYC [157,158,159]; Avoid the ethical controversies associated with ESCs [159,160]; Allow for self-assembly into embryoid bodies [187]; Serve as a source for exosomes production [182]; Exhibit anti-fibrotic effects [188]. | Carry a risk of malignant transformation during handling [159,160]; Requires adherence to strict regulatory requirements and safety standards for clinical use [187]; Exhibit high variability across methods, resulting in low reproducibility and reliability [187]. | Develop IEOs using hydrogels—such as norbornene-functionalized gelatin cross-linked with thiolated PEG, or heparin-functionalized HA—to induce differentiation into ectoderm, mesoderm, and endoderm [169]; Supplement with Matrigel to map the developmental stages of IEOs [172] and produce human neuronal organoids [171]. |
| MSCs | Protect inner ear cells against the ototoxic effects of chemotherapy using MSC-derived EV [177]; Support auricular cartilage regeneration [17]; Promote laryngeal cartilage and vocal cord regeneration [179,180,181]. | Possess self-renewal ability [173,174]; Differentiate into organ-specific cell type [173,174]; Diminish ethical concerns compared to ESCs [175]; Reduce the risk of malignant transformation [175]; Serve as a source of exosomes [189]; Reduce inflammation [189]. | Exhibit significant heterogeneity, complicated standardization; this is further influenced by donor-to-donor variability, including factors such as species, gender, and health status [190]; Possess limited capacity, perpetual self-renewal, and replication [190]; Show limited potential to differentiate into derivatives of the endoderm, mesoderm, and ectoderm [191]. | Facilitate auricular cartilage construction through the interaction of adipose-derived MSCs with type 1 collagen [178]; Promote wound repair and cell proliferation using umbilical cord MSCs-derived exosomes encapsulated in SF/sericin hydrogel dressing [189]; Prolong the therapeutic duration of bone marrow MSCs using alginate hydrogels, which function as reservoirs for sustained release [189]. |
| Printing Technology | Advantages | Drawbacks | References |
|---|---|---|---|
| Inkjet bioprinting | High resolution; High throughput; High water content to facilitate nutrient diffusion; Excellent reproducibility; Good cytocompatibility; High cell viability (>90%); Cost-effective; Scalable for multiple printheads. | Direct cellular stress that could lead to membrane damage and irreversible cell alterations; Unsuitable for large-scale constructs; Limited bioink reservoir volume; Incompatible with high viscosity bioinks. | [207,212,213,214,215,221,222,223,224] |
| Extruded bioprinting | Large-scale tissue engineering; Compatible with a range of materials and viscosities; User-friendly operation. | Direct cellular stress that could compromise viability; Low resolution; Fine structures cannot be accurately reproduced; Filament size is limited by the nozzlediameter. | [207,216,221] |
| Laser-assisted bioprinting | Minimal cellular stress; Suitable for biolayers or single-layer printing; High cell viability (>90%); High resolution; Precise control ofcell distribution within the bioink. | Prohibitively expensive; Complex setup and maintenance; Cell density below 108 cells/mL. | [207,216,217,223,224] |
| Stereolithography | High spatial resolution; High precision and high speed printing; Rapid layer-by-layer solidification; High cell viability. | UV-induced cell damage; requires optical transparent materials for light penetration; The photopolymer substrate cannot be changed during the printing process. | [210,221,222] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Vlad, T.; Mituletu, M.; Flangea, C.; Marina, C.D.; Caraba, M.N.; Balica, N.C.; Vlad, C.S.; Popescu, R. Bioprinting in Tissue Repair and Its ENT Applications. Polymers 2026, 18, 821. https://doi.org/10.3390/polym18070821
Vlad T, Mituletu M, Flangea C, Marina CD, Caraba MN, Balica NC, Vlad CS, Popescu R. Bioprinting in Tissue Repair and Its ENT Applications. Polymers. 2026; 18(7):821. https://doi.org/10.3390/polym18070821
Chicago/Turabian StyleVlad, Tania, Mihai Mituletu, Corina Flangea, Cristina Doriana Marina, Marioara Nicoleta Caraba, Nicolae Constantin Balica, Cristian Sebastian Vlad, and Roxana Popescu. 2026. "Bioprinting in Tissue Repair and Its ENT Applications" Polymers 18, no. 7: 821. https://doi.org/10.3390/polym18070821
APA StyleVlad, T., Mituletu, M., Flangea, C., Marina, C. D., Caraba, M. N., Balica, N. C., Vlad, C. S., & Popescu, R. (2026). Bioprinting in Tissue Repair and Its ENT Applications. Polymers, 18(7), 821. https://doi.org/10.3390/polym18070821









