Effects of Thermal Aging on Optical, Surface, Mechanical, and Biological Properties of CAD/CAM Polymer-Based Dental Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Specimen Preparation
2.3. Thermal Cycling
2.4. Color Stability Measurements
2.5. Surface Roughness Measurements
2.6. Surface Microhardness Measurement
2.7. Spectroscopic Analyses (FTIR and Raman)
2.8. Surface Morphology (SEM) Analysis
2.9. Cytotoxicity Analysis
2.10. Statistical Analysis
3. Results
3.1. Color Stability
3.2. Surface Roughness
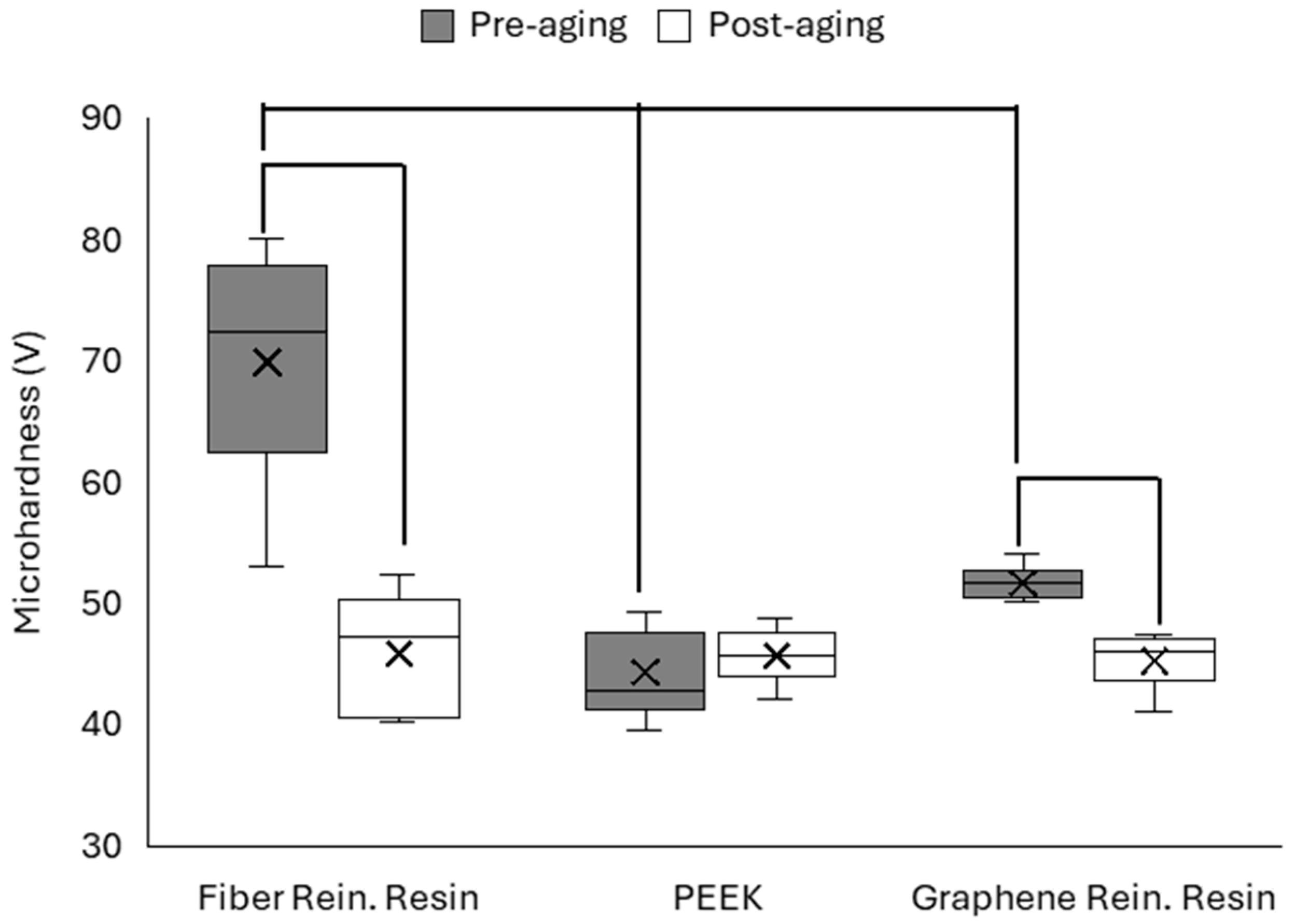
3.3. Surface Microhardness
3.4. Spectroscopic Analysis (FTIR and Raman)
3.5. Surface Morphology (SEM)
3.6. Cytotoxicity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marchesi, G.; Camurri Piloni, A.; Nicolin, V.; Turco, G.; Di Lenarda, R. Chairside CAD/CAM Materials: Current Trends of Clinical Uses. Biology 2021, 10, 1170. [Google Scholar] [CrossRef] [PubMed]
- Poggio, C.E.; Ercoli, C.; Rispoli, L.; Maiorana, C.; Esposito, M. Metal-free materials for fixed prosthodontic restorations. Cochrane Database Syst. Rev. 2017, 12, CD009606. [Google Scholar] [CrossRef]
- Jalkh, E.B.; Machado, C.; Gianinni, M.; Beltramini, I.; Piza, M.; Coelho, P.; Hirata, R.; Bonfante, E. Effect of thermocycling on biaxial flexural strength of CAD/CAM, bulk fill, and conventional resin composite materials. Oper. Dent. 2019, 44, E254–E262. [Google Scholar] [CrossRef]
- Porto, T.S.; Roperto, R.C.; Akkus, A.; Akkus, O.; Teich, S.; Faddoul, F.F.; Porto-Neto, S.d.T.; de Campos, E.A. Effect of thermal cycling on fracture toughness of CAD/CAM materials. Am. J. Dent. 2018, 31, 205–210. [Google Scholar]
- Fugolin, A.P.P.; Pfeifer, C.S. New resins for dental composites. J. Dent. Res. 2017, 96, 1085–1091. [Google Scholar] [CrossRef]
- Sachdev, H.; Soni, M.; Gupta, A.; Chansoria, H.; Patil, M.; Sharma, B.; Kar, K.; Sharma, R. Comparative evaluation of flexural strength of PMMA reinforced with graphene (G-CAM) and monolithic zirconia. An in vitro study. J. Pharm. Bioallied Sci. 2025, 17, S238–S241. [Google Scholar] [CrossRef] [PubMed]
- Yeslam, H.E. Flexural behavior of biocompatible high-performance polymer composites for CAD/CAM dentistry. J. Compos. Sci. 2023, 7, 270. [Google Scholar] [CrossRef]
- Schwitalla, A.D.; Spintig, T.; Kallage, I.; Müller, W.D. Flexural behavior of PEEK materials for dental application. Dent. Mater. 2015, 31, 1377–1384. [Google Scholar] [CrossRef] [PubMed]
- Porojan, L.; Toma, F.R.; Bîrdeanu, M.I.; Vasiliu, R.D.; Uțu, I.-D.; Matichescu, A. Surface Characteristics and Color Stability of Dental PEEK Related to Water Saturation and Thermal Cycling. Polymers 2022, 14, 2144. [Google Scholar] [CrossRef]
- Agarwalla, S.V.; Malhotra, R.; Rosa, V. Translucency, hardness and strength parameters of PMMA resin containing graphene-like material for CAD/CAM restorations. J. Mech. Behav. Biomed. Mater. 2019, 100, 103388. [Google Scholar] [CrossRef]
- Rajak, D.K.; Pagar, D.D.; Menezes, P.L.; Linul, E. Fiber-reinforced polymer composites: Manufacturing, properties, and applications. Polymers 2019, 11, 1667. [Google Scholar] [CrossRef]
- Petersen, R.; Liu, P.R. 3D-woven fiber-reinforced composite for CAD/CAM dental application. Soc. Adv. Mater. Process Eng. 2016, 2016, LB15–0138. [Google Scholar] [PubMed]
- Çakmak, G.; Donmez, M.B.; Akay, C.; Abou-Ayash, S.; Schimmel, M.; Yilmaz, B. Effect of thermal cycling on flexural strength and hardness of new-generation denture base materials. J. Prosthodont. 2023, 32, 81–86. [Google Scholar] [CrossRef]
- Lopez, D.; Ziada, H.; Abubakr, N.H. Influence of thermal aging on marginal integrity of CAD/CAM fabricated crowns. J. Dent. Sci. 2024, 19, 971. [Google Scholar] [CrossRef]
- Gómez-Polo, C.; Muñoz, M.P.; Luengo, M.C.L.; Vicente, P.; Galindo, P.; Casado, A.M.M. Comparison of the CIELab and CIEDE2000 color difference formulas. J. Prosthet. Dent. 2016, 115, 65–70. [Google Scholar] [CrossRef]
- Pecho, O.E.; Ghinea, R.; Alessandretti, R.; Pérez, M.M.; Della Bona, A. Visual and instrumental shade matching using CIELAB and CIEDE2000 color difference formulas. Dent. Mater. 2016, 32, 82–92. [Google Scholar] [CrossRef]
- Price, R.B.; Sullivan, B. Effect of Indenter Load on Vickers Microhardness and Indentation Depth of One Resin Composite. Materials 2024, 17, 6156. [Google Scholar] [CrossRef] [PubMed]
- ASTM E384; Standard Test Method for Microindentation Hardness of Materials. ASTM International: West Conshohocken, PA, USA, 2017.
- ISO 10993-12; Biological Evaluation of Medical Devices—Part 12: Sample Preparation and Reference Materials. International Organization for Standardization (ISO): Geneva, Switzerland, 2012.
- Luo, C.; Liu, Y.; Peng, B.; Chen, M.; Liu, Z.; Li, Z.; Kuang, H.; Gong, B.; Li, Z.; Sun, H. PEEK for Oral Applications: Recent Advances in Mechanical and Adhesive Properties. Polymers 2023, 15, 386. [Google Scholar] [CrossRef] [PubMed]
- DeAraújo Nobre, M.; Moura Guedes, C.; Almeida, R.; Silva, A. Poly-ether-ether-ketone and implant dentistry: The future of mimicking natural dentition is now! Polym. Int. 2021, 70, 999–1001. [Google Scholar] [CrossRef]
- Şahin, Z.; Ergun, G.; Ataol, A.S. Comparison of physical properties of CAD/CAM-produced PEEK and PMMA denture base materials. Mersin Univ. Saglik Bilim. Derg. 2021, 14, 484–494. [Google Scholar] [CrossRef]
- Qin, W.; Li, Y.; Ma, J.; Liang, Q.; Tang, B. Mechanical properties and cytotoxicity of hierarchical carbon fiber-reinforced PEEK composites. J. Mech. Behav. Biomed. Mater. 2019, 89, 227–233. [Google Scholar] [CrossRef]
- Di Carlo, S.; De Angelis, F.; Brauner, E.; Pranno, N.; Tassi, G.; Senatore, M.; Bossù, M. Flexural Strength and Elastic Modulus Evaluation of Structures Made by Conventional PMMA and PMMA Reinforced with Graphene. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 5201–5208. [Google Scholar] [CrossRef]
- Erdağ, Ü.H.; Şahin, O.; Köroğlu, A.; Özdemir, T.; Dede, D.Ö. Performance of PEEK for Dental Applications: Surface Roughness and Color Stability. Polym. Bull. 2023, 80, 6819–6834. [Google Scholar] [CrossRef]
- Çakmak, G.; Asadolahi, N.W.; Schimmel, M.; Molinero-Mourelle, P.; Akay, C.; Donmez, M.B.; Yilmaz, B. Effect of Coffee Thermal Cycling on the Surface Properties and Stainability of Additively Manufactured Denture Base Resins in Different Layer Thicknesses. J. Prosthodont. 2025, 34, 157–166. [Google Scholar] [CrossRef]
- Paolone, G.; Mandurino, M.; De Palma, F.; Mazzitelli, C.; Scotti, N.; Breschi, L.; Gherlone, E.; Cantatore, G.; Vichi, A. Color Stability of Polymer-Based Composite CAD/CAM Blocks: A Systematic Review. Polymers 2023, 15, 464. [Google Scholar] [CrossRef]
- Cinel Sahin, S.; Mutlu Sagesen, L. Effects of Polishing Protocols on the Surface Roughness and Color Stability of Polyetheretherketone (PEEK). Eur. Oral Res. 2024, 58, 1–7. [Google Scholar] [CrossRef]
- Paravina, R.D.; Ghinea, R.; Herrera, L.J.; Bona, A.D.; Igiel, C.; Linninger, M.; Sakai, M.; Takahashi, H.; Tashkandi, E.; Perez, M.M. Color Difference Thresholds in Dentistry. J. Esthet. Restor. Dent. 2015, 27, S1–S9. [Google Scholar] [CrossRef]
- Bollen, C.M.; Lambrechts, P.; Quirynen, M. Comparison of surface roughness threshold for bacterial plaque retention. Dent. Mater. 1997, 13, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Alnafaiy, S.M.; Labban, N.; Albaijan, R.; AlKahtani, R.N.; Al-Aali, K.A.; Abozaed, H.W.; Alturki, N.Y.; Alenezi, J.E. Evaluation of Shear Bond Strength and Failure Modes of Lithium Disilicate Ceramic Veneering Material to Different High-Performance Polymers. Polymers 2025, 17, 554. [Google Scholar] [CrossRef] [PubMed]
- Bathala, L.; Majeti, V.; Rachuri, N.; Singh, N.; Gedela, S. The Role of Polyether Ether Ketone (PEEK) in Dentistry—A Review. J. Med. Life 2019, 12, 5–9. [Google Scholar] [CrossRef]
- Schwitalla, A.; Müller, W.D. PEEK dental implants: A review of the literature. J. Oral Implantol. 2013, 39, 743–749. [Google Scholar] [CrossRef]
- Fajardo, J.I.; Paltán, C.A.; León, M.; Matute, A.Y.; Armas-Vega, A.; Puratambi, R.H.; Delgado-Gaete, B.A.; Requena, S.; Benalcazar, A. Impact of Thermal Cycling on the Vickers Microhardness of Dental CAD/CAM Materials: Greater Retention in Polymer-Infiltrated Ceramic Networks (PICNs) Compared to Nano-Filled Resin Composites. Ceramics 2025, 8, 125. [Google Scholar] [CrossRef]
- Saloni, P.; Sankaran, K.; Ganesh, S.B.; Jayalakshmi, S.; Priya, V.V.; Gayathri, R. Effect of Thermocycling on the Microhardness of Pre-Heated and Non-Heated Zirconium Composite Resin. J. Int. Oral Health 2025, 17, 304–309. [Google Scholar] [CrossRef]
- Akay, C.; Çakmak, G.; Donmez, M.B.; Abou-Ayash, S.; Mumcu, E.; Pat, S.; Yilmaz, B. Flexural Strength and Vickers Microhardness of Graphene-Doped SnO2 Thin-Film-Coated Polymethylmethacrylate after Thermocycling. Coatings 2023, 13, 1106. [Google Scholar] [CrossRef]
- Arafa, S.K.; Sherief, D.I.; Nassif, M.S. Effect of Aging on Mechanical and Antibacterial Properties of Fluorinated Graphene Reinforced Glass Ionomer: In Vitro Study. J. Mech. Behav. Biomed. Mater. 2023, 142, 105803. [Google Scholar] [CrossRef]
- Qasim, S.S.B.; Akbar, A.A.; Sadeqi, H.A.; Baig, M.R. Surface Characterization of Bone-Level and Tissue-Level PEEK and Titanium Dental Implant Scan Bodies After Repeated Autoclave Sterilization Cycles. Dent. J. 2024, 12, 392. [Google Scholar] [CrossRef] [PubMed]
- Badeghaish, W.; Wagih, A.; Rastogi, S.; Lubineau, G. Effect of High-Temperature Acid Aging on Microstructure and Mechanical Properties of PEEK. Polym. Test. 2024, 134, 108429. [Google Scholar] [CrossRef]
- Ruggiero, G.; Di Rosa, D.; Caso, F.; Sorrentino, R.; Zarone, F.; Caso, G. Synthesis and FT-IR/Raman Characterization of a Graphene Oxide–Methacrylamide Monomer for Dental Applications. Materials 2025, 18, 3550. [Google Scholar] [CrossRef] [PubMed]
- Bin-Shuwaish, M.S.; Maawadh, A.M.; Al-Hamdan, R.S.; Alresayes, S.; Almohareb, T.; Almutairi, B.; Vohra, F.; Abduljabbar, T. Influence of Graphene Oxide Filler Content on the Dentin Bond Integrity, Degree of Conversion and Bond Strength of Experimental Adhesive: A SEM, Micro-Raman, FTIR and Microtensile Study. Mater. Res. Express 2020, 7, 115403. [Google Scholar] [CrossRef]
- Silu, H.; Fu, Q.; Yan, L.; Kasal, B. Characterization of Interfacial Properties between Fibre and Polymer Matrix in Composite Materials—A Critical Review. J. Mater. Res. Technol. 2021, 13, 138–158. [Google Scholar] [CrossRef]
- ISO 10993-5; Biological Evaluation of Medical Devices Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization (ISO): Geneva, Switzerland, 2012.
- Moharil, S.; Reche, A.; Durge, K. Polyetheretherketone (PEEK) as a Biomaterial: An Overview. Cureus 2023, 15, e44307. [Google Scholar] [CrossRef]
- Zeroual, R.; Oussama, B. The Use of Polyetheretherketone (PEEK) in Implant Prosthetics: A Detailed Review of the Literature. J. Clin. Adv. Dent. 2023, 7, 34–40. [Google Scholar] [CrossRef]
- Alqutaibi, A.Y.; Alghauli, M.; Al-Gabri, R.S.; Fareed, M.A.; Alqutaibi, F.S.; Vohra, F.; Abduljabbar, T. Applications, Modifications, and Manufacturing of Polyetheretherketone (PEEK) in Dental Implantology: A Comprehensive Critical Review. Int. Mater. Rev. 2025, 70, 103–136. [Google Scholar] [CrossRef]
- Drummond, J.L.; Andronova, K.; Al-Turki, L.I.; Slaughter, L.D. Leaching and Mechanical Properties Characterization of Dental Composites. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 71, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Oja, J.; Lassila, L.; Vallittu, P.K.; Garoushi, S. Effect of Accelerated Aging on Some Mechanical Properties and Wear of Different Commercial Dental Resin Composites. Materials 2021, 14, 2769. [Google Scholar] [CrossRef]
- Meriç, G.; Dahl, J.E.; Ruyter, I.E. Cytotoxicity of Silica-Glass Fiber Reinforced Composites. Dent. Mater. 2008, 24, 1201–1206. [Google Scholar] [CrossRef]
- Williams, A.G.; Moore, E.; Thomas, A.; Johnson, J.A. Graphene-Based Materials in Dental Applications: Antibacterial, Biocompatible, and Bone Regenerative Properties. Int. J. Biomater. 2023, 2023, 8803283. [Google Scholar] [CrossRef]
- Dayan, C.; Guven, M.C.; Gencel, B.; Bural, C. A Comparison of the Color Stability of Conventional and CAD/CAM Polymethyl Methacrylate Denture Base Materials. Acta Stomatol. Croat. 2019, 53, 158–167. [Google Scholar] [CrossRef] [PubMed]







| Material Type | Commercial Name | Manufacturer |
|---|---|---|
| Fiber-reinforced composite (FRC) polymer | Trilor | Bioloren S.r.l., Saronno, Italy |
| Polyether ether ketone (PEEK) | JUVORA | JUVORA Ltd., Thornton-Cleveleys, UK |
| Graphene-reinforced PMMA-based acrylic resin G-CAM | G-CAM | Graphenano Dental, Valencia, Spain |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ateş, G.; Gungormus, M. Effects of Thermal Aging on Optical, Surface, Mechanical, and Biological Properties of CAD/CAM Polymer-Based Dental Materials. Polymers 2026, 18, 760. https://doi.org/10.3390/polym18060760
Ateş G, Gungormus M. Effects of Thermal Aging on Optical, Surface, Mechanical, and Biological Properties of CAD/CAM Polymer-Based Dental Materials. Polymers. 2026; 18(6):760. https://doi.org/10.3390/polym18060760
Chicago/Turabian StyleAteş, Gül, and Mustafa Gungormus. 2026. "Effects of Thermal Aging on Optical, Surface, Mechanical, and Biological Properties of CAD/CAM Polymer-Based Dental Materials" Polymers 18, no. 6: 760. https://doi.org/10.3390/polym18060760
APA StyleAteş, G., & Gungormus, M. (2026). Effects of Thermal Aging on Optical, Surface, Mechanical, and Biological Properties of CAD/CAM Polymer-Based Dental Materials. Polymers, 18(6), 760. https://doi.org/10.3390/polym18060760







