Atomic Layer Deposition of ZnO and ZnO/Cu Coatings for Fresh Food Packaging Application
Abstract
1. Introduction
2. Materials and Methods
2.1. ZnO and ZnO/Cu Film Preparation
2.2. Film Characterization
2.3. Burrata and Turkey Fillet Packaging
2.4. Microbiological Analyses and pH Determination
2.5. Sensory Analyses
2.6. Shelf Life Calculation
2.7. Statistical Analysis
3. Results and Discussion
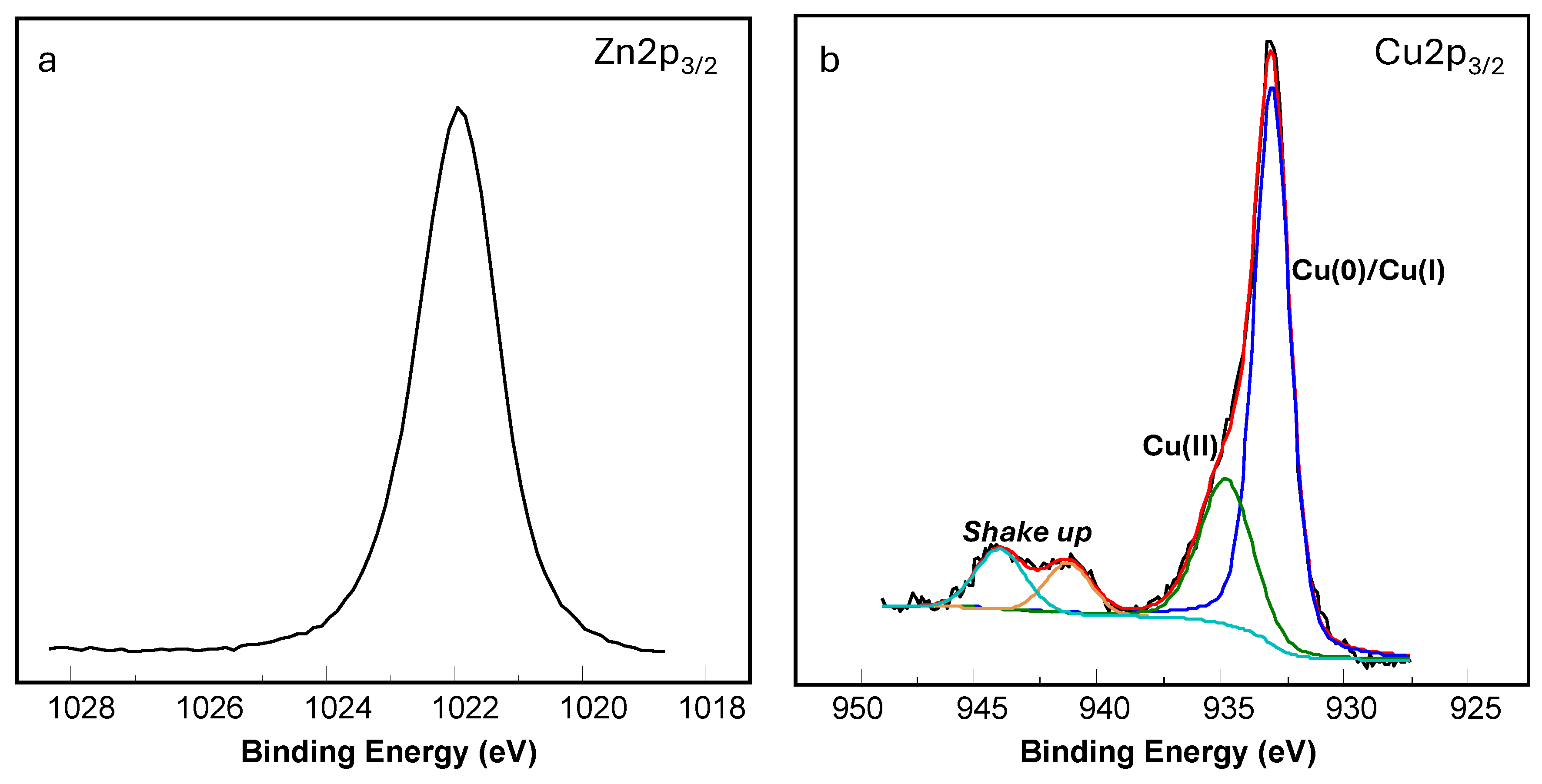
3.1. Chemical and Morphological Characterization of the Coatings
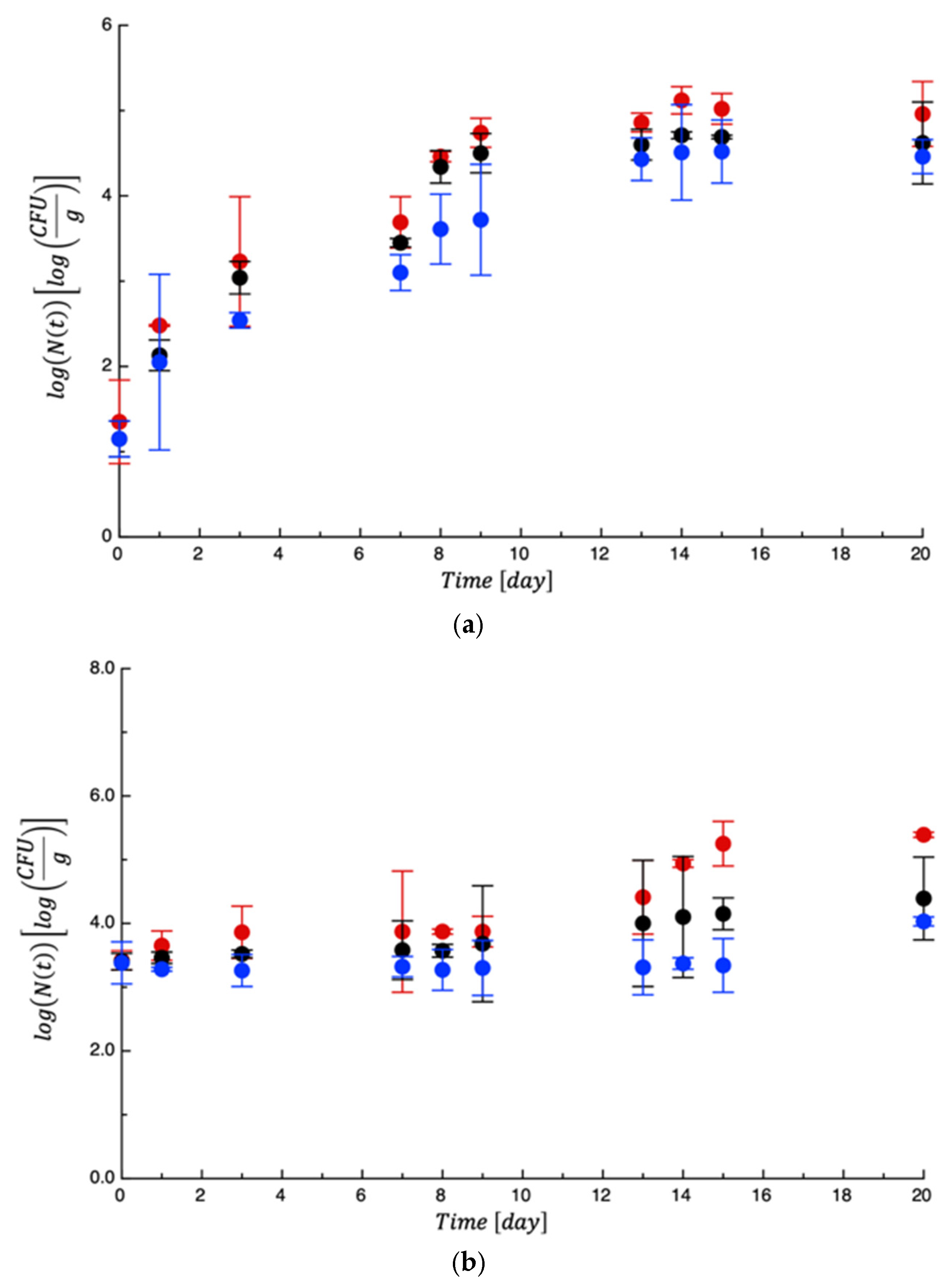
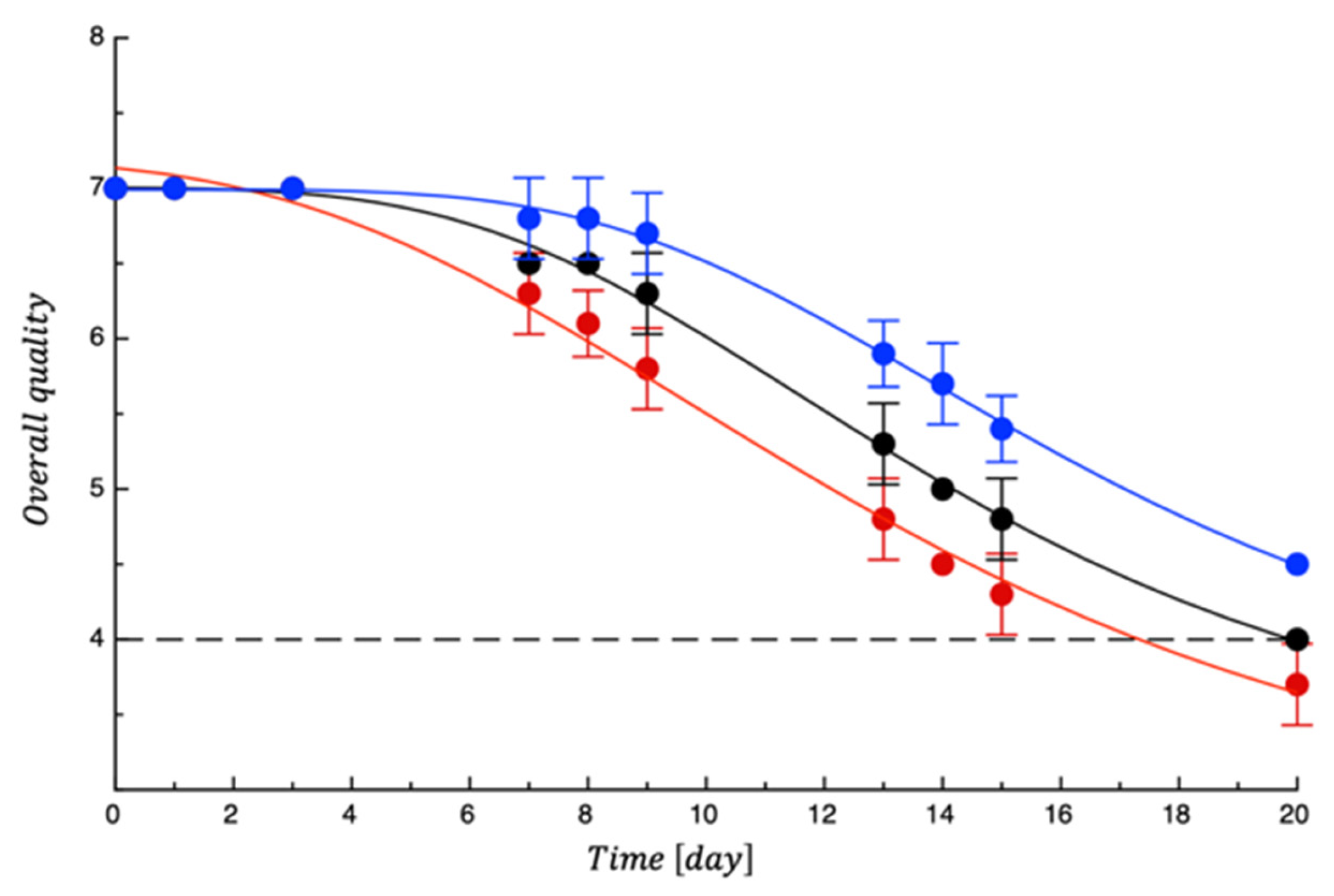
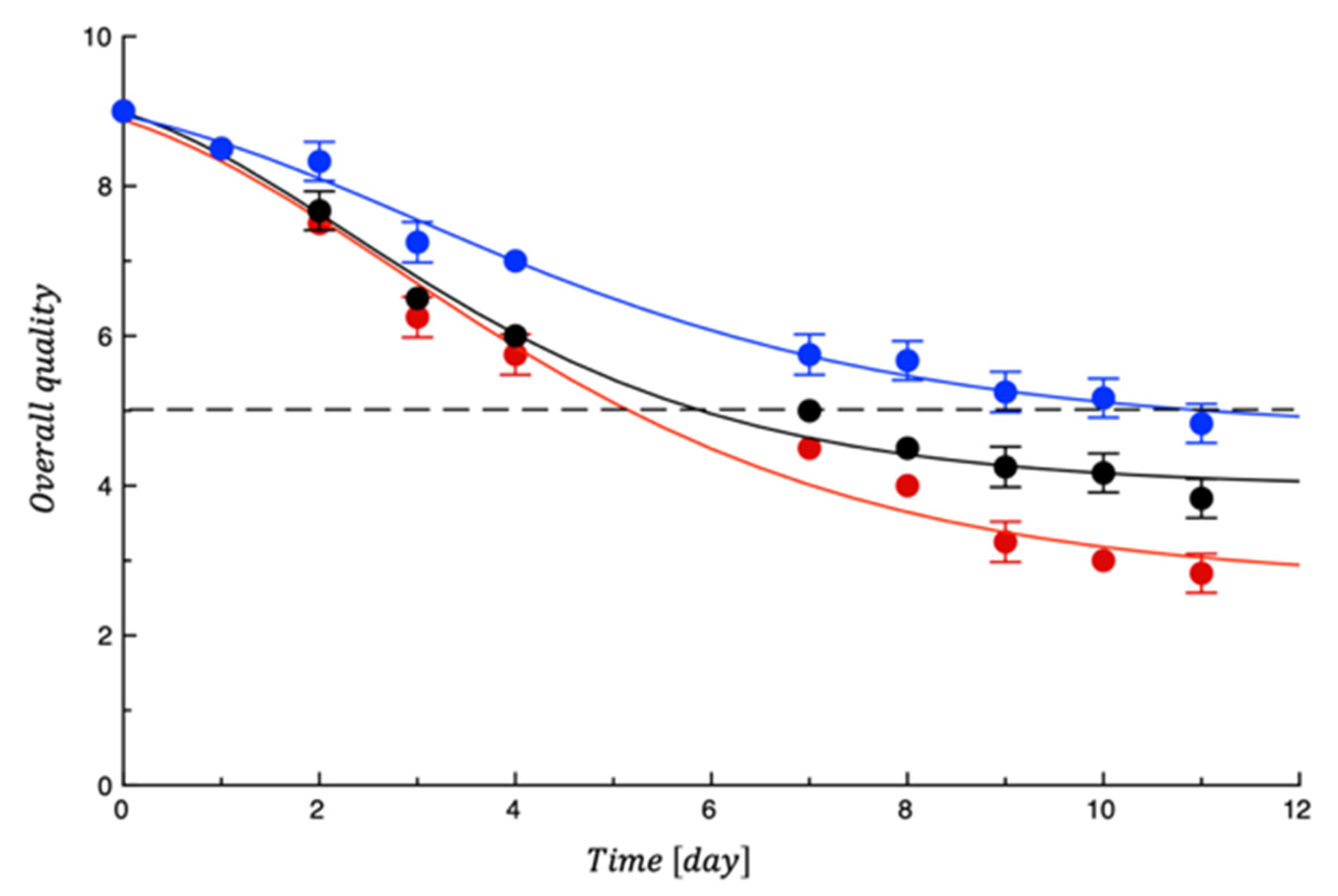
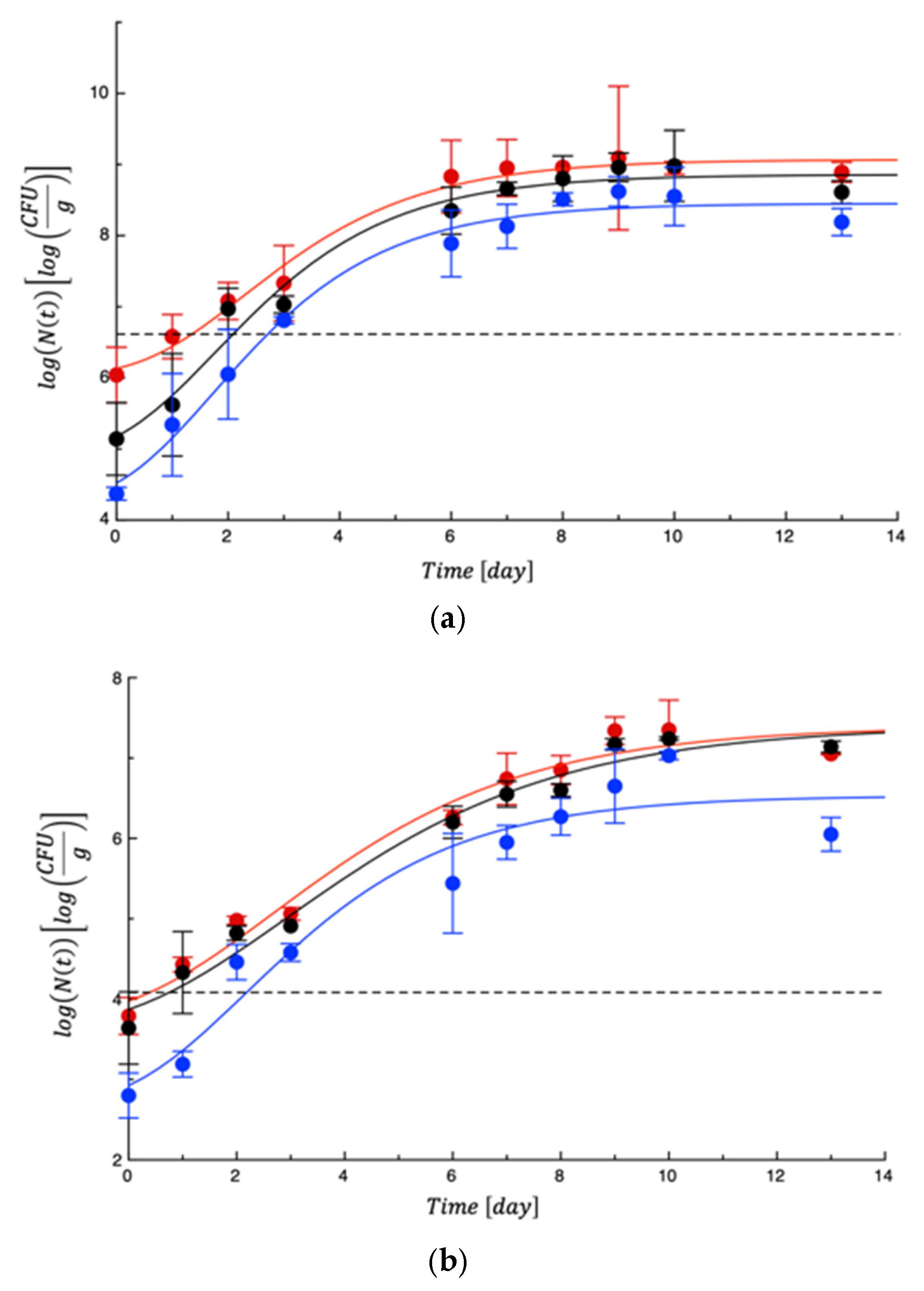
3.2. Shelf Life Tests on Burrata Cheese
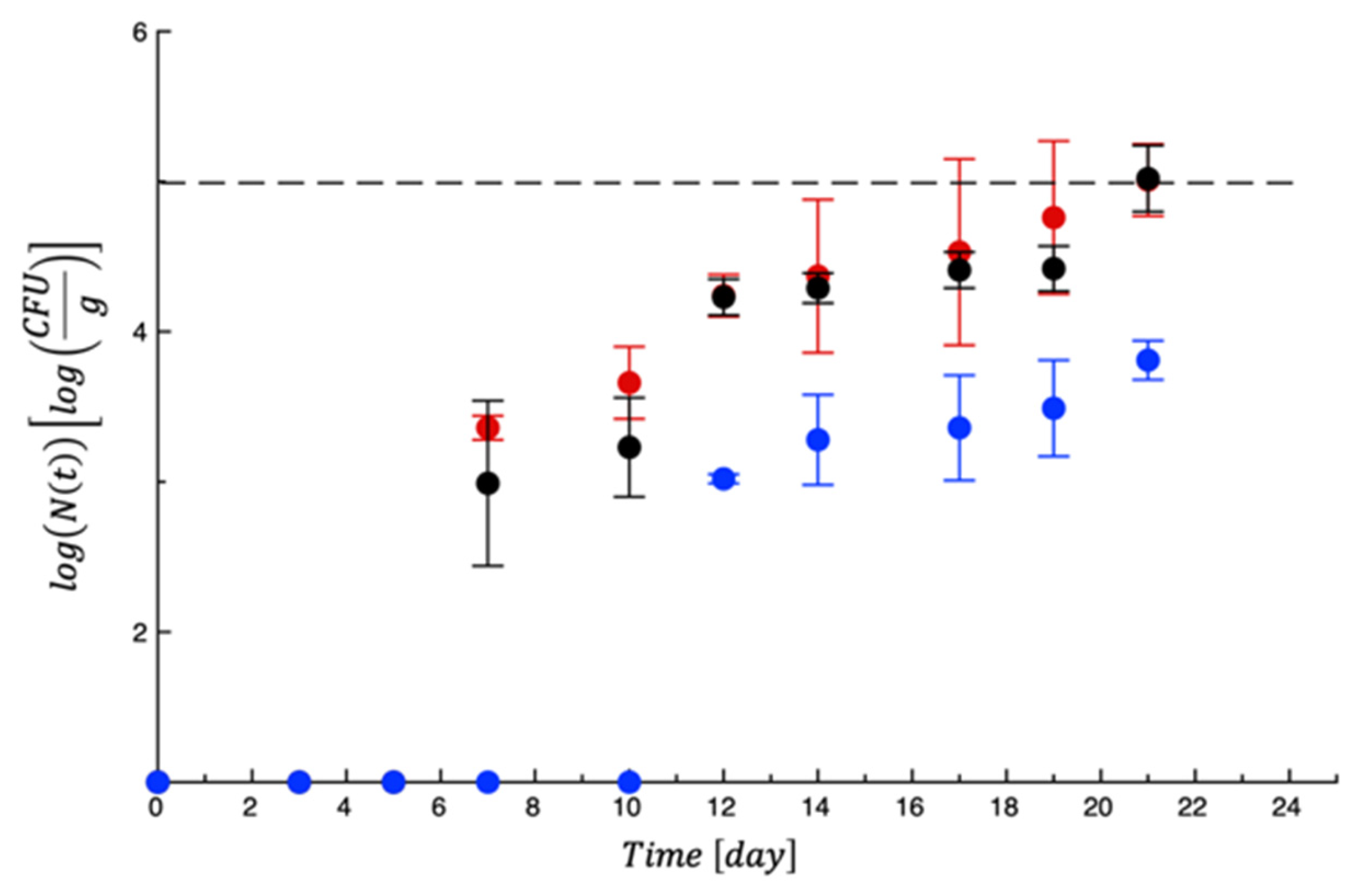
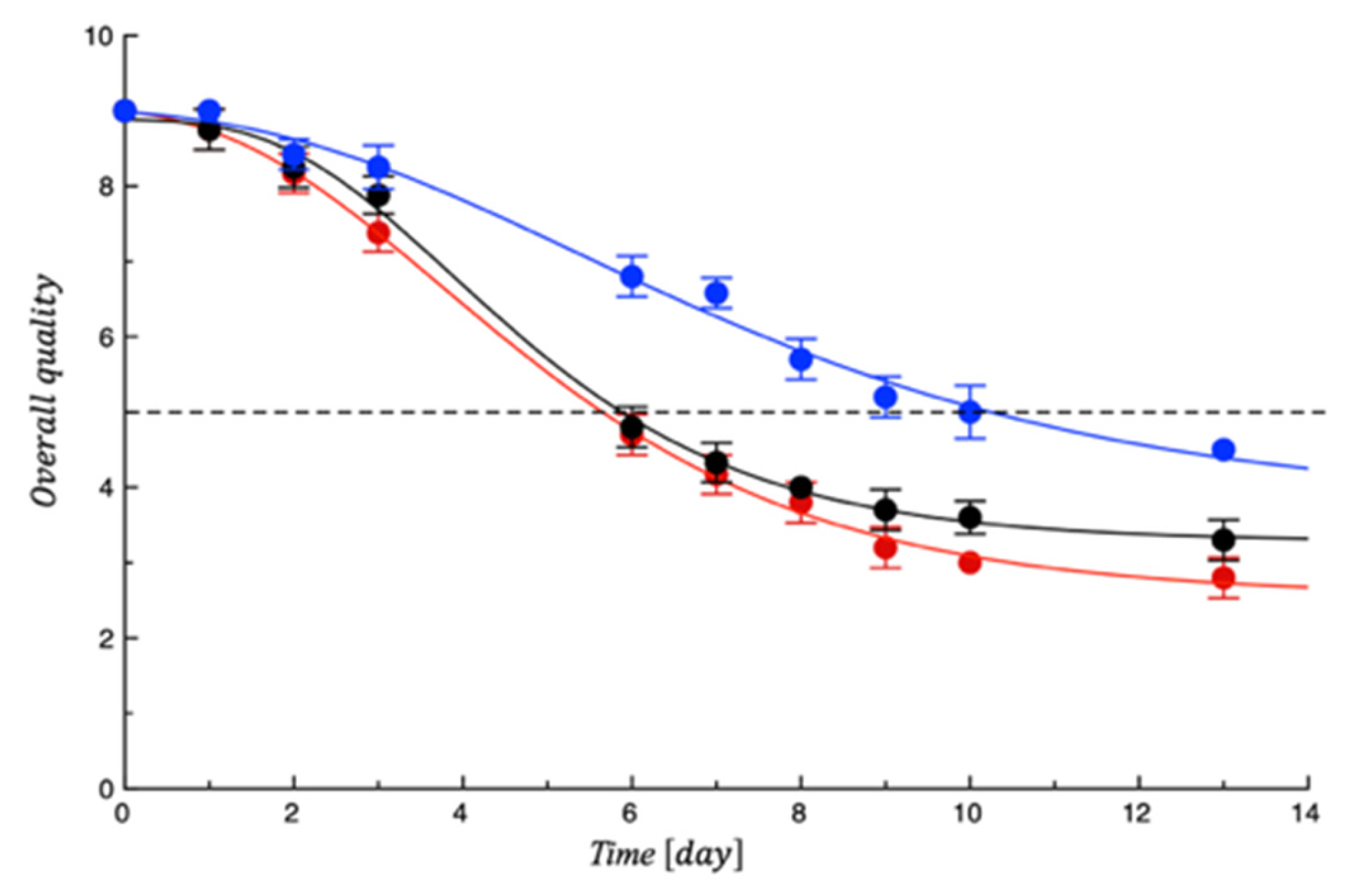
4. Shelf Life Tests on Turkey Fillet
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Cazon, P.; Mateus, A.R.; Silva, A.S. Advances in active packaging using natural biopolymers and fruit by-products for enhanced food preservation. Food Res. Int. 2025, 213, 116439. [Google Scholar] [CrossRef]
- Rodriguez, O.T.; Valero, M.F.; Gomez-Tejedor, J.A.; Diaz, L. Performance of biodegradable active packaging in the preservation of fresh-cut fruits: A systematic review. Polymers 2024, 16, 3518. [Google Scholar] [CrossRef]
- Azevedo, A.G.; Barros, C.; Miranda, S.; Machado, A.V.; Castro, O.; Silva, B.; Saraiva, M.; Silva, A.S.; Pastrana, L.; Carneiro, O.S.; et al. Active flexible films for food packaging: A review. Polymers 2022, 14, 2442. [Google Scholar] [CrossRef]
- Kamau, P.G.; Cruz-Romero, M.C.; Alzate, P.C.; Morris, M.A.; Kerry, J.P. Active packaging containing natural antimicrobials as a potential and innovative technology to extend shelf-life of fish products—A review. Food Packag. Shelf Life 2025, 49, 101500. [Google Scholar]
- Hoseinnejad, M.; Jafari, S.M.; Katouzian, I. Inorganic and metal nanoparticles and their antimicrobial activity in food packaging applications. Crit. Rev. Microbiol. 2018, 44, 161–181. [Google Scholar]
- Dash, K.K.; Deka, P.; Bangar, S.P.; Chaudhary, V.; Trif, M.; Rusu, A. Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review. Polymers 2022, 14, 521. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Ilyas, S.; Sultana, A.; Hezam, A.; Sunil, L.; Surmeneva, M.A.; Surmenev, R.A.; Nayan, M.B.; Ramakrishna, S.; et al. Emerging Trends for ZnO Nanoparticles and Their Applications in Food Packaging. ACS Food Sci. Technol. 2022, 2, 763–781. [Google Scholar] [CrossRef]
- Struller, C.F.; Kelly, P.J.; Copeland, N.J. Aluminum oxide barrier coatings on polymer films for food packaging applications. Surf. Coat. Technol. 2014, 241, 130–137. [Google Scholar]
- She, H.; Chen, P.; Yang, R. Advances in Antibacterial Polymer Coatings Synthesized via Chemical Vapor Deposition. Chem Bio Eng. 2024, 1, 516–534. [Google Scholar] [CrossRef]
- Kışla, D.; Gökmen, G.G.; Evrendilek, G.A.; Akan, T.; Vlčko, T.; Kulawik, P.; Režek Jambrak, A.; Ozogul, F. Recent developments in antimicrobial surface coatings: Various deposition techniques with nanosized particles, their application and environmental concerns. Trends Food Sci. Technol. 2023, 135, 144–172. [Google Scholar] [CrossRef]
- George, S.M. Atomic Layer Deposition: An Overview. Chem. Rev. 2009, 110, 111–131. [Google Scholar] [CrossRef]
- Abdulagatov, I.M.; Maksumova, A.M.; Magomedov, M.Z.; Tsakhaeva, R.O.; Khidirova, S.M.; Salikhov, A.M. Antibacterial Food Packaging Nanomaterial Based on Atomic Layer Deposition for Long-Term Food Storage. J. Food Sci. Technol. 2024, 61, 596–606. [Google Scholar]
- Benetti, G.; Cavaliere, E.; Banfi, F.; Gavioli, L. Antimicrobial Nanostructured Coatings: A Gas Phase Deposition and Magnetron Sputtering Perspective. Materials 2020, 13, 784. [Google Scholar] [CrossRef] [PubMed]
- Olawore, O.; Ogunmola, M.; Desai, S. Engineered Nanomaterial Coatings for Food Packaging: Design, Manufacturing, Regulatory, and Sustainability Implications. Micromachines 2024, 15, 245. [Google Scholar] [CrossRef] [PubMed]
- Nazarov, D.; Kozlova, L.; Rogacheva, E.; Kraeva, L.; Maximov, M. Atomic Layer Deposition of Antibacterial Nanocoatings: A Review. Antibiotics 2023, 12, 1656. [Google Scholar] [CrossRef]
- Markowska-Szczupak, A.; Paszkiewicz, O.; Michalkiewicz, B.; Kamińska, A.; Wróbel, R.J. Fabrication of Antibacterial Metal Surfaces Using Magnetron-Sputtering Method. Materials 2021, 14, 7301. [Google Scholar] [CrossRef]
- Vähä-Nissi, M.; Pitkänen, M.; Salo, E.; Kenttä, E.; Tanskanen, A.; Sajavaara, T.; Putkonen, M.; Sievänen, J.; Sneck, A.; Rättö, M.; et al. Antibacterial and barrier properties of oriented polymer films with ZnO thin films applied with atomic layer deposition at low temperatures. Thin Solid Films 2014, 562, 331–337. [Google Scholar] [CrossRef]
- Babaei-Ghazvini, A.; Acharya, B.; Korber, D.R. Antimicrobial Biodegradable Food Packaging Based on Chitosan and Metal/Metal-Oxide Bio-Nanocomposites: A Review. Polymers 2021, 13, 2790. [Google Scholar]
- De Vietro, N.; Conte, A.; Incoronato, A.L.; Del Nobile, M.A.; Fracassi, F. Aerosol-assisted low pressure plasma deposition of antimicrobial hybrid organic-inorganic Cu-composite thin films for food packaging applications. Innov. Food Sci. Emerg. Technol. 2017, 41, 130–134. [Google Scholar] [CrossRef]
- Medina-Ramírez, I.E.; Arzate-Cardenas, M.A.; Mojarro-Olmos, A.; Romo-López, M.A. Synthesis, characterization, toxicological and antibacterial activity evaluation of Cu@ZnO nanocomposites. Ceram. Int. 2019, 45, 17476–17488. [Google Scholar] [CrossRef]
- Kalia, A.; Kaur, M.; Shami, A.; Jawandha, S.K.; Alghuthaymi, M.A.; Thakur, A.; Abd-Elsalam, K.A. Nettle-Leaf Extract Derived ZnO/CuO Nanoparticle-Biopolymer-Based Antioxidant and Antimicrobial Nanocomposite Packaging Films and Their Impact on Extending the Post-Harvest Shelf Life of Guava Fruit. Biomolecules 2021, 11, 224. [Google Scholar]
- Costa, C.; Lucera, A.; Conte, A.; Zambrini, A.V.; Del Nobile, M.A. Technological Strategies to Preserve Burrata Cheese Quality. Coatings 2017, 7, 97. [Google Scholar] [CrossRef]
- UNI EN ISO 17294-2:2023; Water Quality—Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS)—Part 2: Determination of Selected Elements. Ente Nazionale Italiano di Unificazione (UNI): Milan, Italy, 2023.
- Incoronato, A.L.; Gammariello, D.; Conte, A.; Del Nobile, M.A. Technological solutions to increase shelf life of fresh meat burger. J. Food Process. Preserv. 2015, 39, 1324–1333. [Google Scholar] [CrossRef]
- Lordi, A.; Caro, D.; Le Rose, A.; Del Nobile, M.A.; Conte, A. Quality and environmental impact of meat burgers fortified with tomato by-products. LWT—Food Sci. Technol. 2025, 229, 118182. [Google Scholar] [CrossRef]
- Qu, B.; Xiao, Z.; Luo, Y. Facile in situ synthesis of ZnO-infused caseinate/carboxymethyl cellulose films with antibacterial properties for cheddar cheese packaging. Carbohydr. Polym. Technol. Appl. 2025, 10, 100779. [Google Scholar] [CrossRef]
- Qu, B.; Xiao, Z.; Luo, Y. Sustainable nanotechnology for food preservation: Synthesis, mechanisms, and applications of zinc oxide nanoparticles. J. Agric. Food Res. 2025, 19, 101743. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Soares, N.D.F.F.; Coimbra, D.R.; De Andrade, N.J.; Cruz, R.S.; Medeiros, E.A. Zinc oxide nanoparticles: Synthesis, antimicrobial activity and food packaging applications. Food Bioprocess Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Li, C.; Cao, J.; He, E.; Wu, X.; Wang, L. Facile preparation PCL/modified nano ZnO organic-inorganic composite and its application in antibacterial materials. J. Polym. Res. 2020, 27, 78. [Google Scholar]
- Amjadi, S.; Emaminia, S.; Nazari, M.; Davudian, S.H.; Roufegarinejad, L.; Hamishehkar, H. Application of Reinforced ZnO Nanoparticle-Incorporated Gelatin Bionanocomposite Film with Chitosan Nanofiber for Packaging of Chicken Fillet and Cheese as Food Models. Food Bioprocess Technol. 2019, 12, 1205–1219. [Google Scholar]
- Beigmohammadi, F.; Peighambardoust, S.H.; Hesari, J.; Azadmard-Damirchi, S.; Peighambardoust, S.J.; Khosrowshahi, N.K. Antibacterial properties of LDPE nanocomposite films in packaging of UF cheese. LWT—Food Sci. Technol. 2016, 65, 106–111. [Google Scholar] [CrossRef]
- Silva, F.A.S.; Carvalho, M.; de Carvalho, T.B.; Gama, M.; Poças, F.; Teixeira, P. Antimicrobial activity of in-situ bacterial nanocellulose-zinc oxide composites for food packaging. Food Packag. Shelf Life 2023, 40, 101201. [Google Scholar]
- Dermiki, M.; Ntzimani, A.; Badeka, A.; Savvaidis, I.N.; Kontominas, M.G. Shelf-life extension and quality attributes of the whey cheese “Myzithra Kalathaki” using modified atmosphere packaging. LWT—Food Sci. Technol. 2008, 41, 284–294. [Google Scholar] [CrossRef]
- Youssef, A.M.; El-Sayed, S.M.; El-Sayed, H.S.; Salama, H.H.; Assem, F.M.; Abd El-Salam, M.H. Novel bionanocomposite materials used for packaging skimmed milk acid coagulated cheese (Karish). Int. J. Biol. Macromol. 2018, 115, 1002–1011. [Google Scholar] [CrossRef]
- Youssef, A.M.; El-Sayed, S.M.; El-Sayed, H.S.; Salama, H.H.; Dufresne, A. Enhancement of Egyptian soft white cheese shelf life using a novel chitosan/carboxymethyl cellulose/zinc oxide bionanocomposite film. Carbohydr. Polym. 2016, 151, 9–19. [Google Scholar] [CrossRef]
- Korany, A.; Abdel-Atty, N.S.; Zeinhom, M.A.M.; Hassan, A.H.A. Application of gelatin-based zinc oxide nanoparticles bionanocomposite coatings to control Listeria monocytogenes in Talaga cheese and camel meat during refrigerated storage. Food Microbiol. 2024, 122, 104559. [Google Scholar] [CrossRef]
- Wang, W.; Gao, P.; Gui, H.; Wei, X.; Zhang, H.; Wang, X. Copper-based nanomaterials for the treatment of bacteria-infected wounds: Material classification, strategies and mechanisms. Coord. Chem. Rev. 2025, 522, 216205. [Google Scholar] [CrossRef]
- Abdallah, S.E.; Elmessery, W.M.; Elfallawi, F.E.; Taher, M.M.; El-Nawasany, L.I.; Shoueir, K.R. Bioinspired CuO nanoparticles from Melissa officinalis for antibacterial electrospun nanofiber food packaging. J. Food Meas. Charact. 2026, 1. [Google Scholar] [CrossRef]
- Longano, D.; Ditaranto, N.; Cioffi, N.; Di Niso, F.; Sibillano, T.; Ancona, A.; Conte, A.; Del Nobile, M.A.; Sabbatini, L.; Torsi, L. Analytical characterization of laser-generated copper nanoparticles for antibacterial composite food packaging. Anal. Bioanal. Chem. 2012, 403, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Conte, A.; Longano, D.; Costa, C.; Ditaranto, N.; Ancona, A.; Cioffi, N.; Scrocco, C.; Sabbatini, F.; Contò, F.; Del Nobile, M.A. A novel preservation technique applied to fiordilatte cheese. Innov. Food Sci. Emerg. Technol. 2013, 19, 158–165. [Google Scholar] [CrossRef]
- Ilg, Y.; Kreyenschmidt, J. Effects of food components on the antimicrobial activity of polypropylene surfaces containing silver ions (Ag+). Int. J. Food Sci. Technol. 2011, 46, 1469–1476. [Google Scholar] [CrossRef]
- Gvozdenko, A.A.; Siddiqui, S.A.; Blinov, A.V.; Golik, A.B.; Nagdalian, A.A.; Maglakelidze, D.G.; Statsenko, E.N.; Pirogov, M.A.; Blinova, A.A.; Sizonenko, M.N.; et al. Synthesis of CuO nanoparticles stabilized with gelatin for potential use in food packaging applications. Sci. Rep. 2022, 12, 12843. [Google Scholar] [CrossRef] [PubMed]
- Smaoui, S.; Cherif, I.; Hlima, H.B.; Khan, M.U.; Rebezov, M.; Thiruvengadam, M.; Sarkar, T.; Shariati, M.A.; Lorenzo, J.M. Zinc oxide nanoparticles in meat packaging: A systematic review of recent literature. Food Packag. Shelf Life 2023, 36, 101045. [Google Scholar] [CrossRef]
- Sasidharan, S.; Tey, L.H.; Djearamane, S.; Ab Rashid, N.K.M.; Pa, R.; Rajendran, V.; Syed, A.; Wong, L.S.; Santhanakrishnan, V.K.; Asirvadam, V.S.; et al. Innovative use of chitosan/ZnO NPs bio-nanocomposites for sustainable antimicrobial food packaging of poultry meat. Food Packag. Shelf Life 2024, 43, 101298. [Google Scholar] [CrossRef]
- Javan, A.J.; Baktash, S.; Yancheshmeh, B.S.; Parsaeimehr, M.; Madanchi, H.; Abdolshahi, A.; Marvdashti, L.M.; Shariatifar, N. Effect of Vicia villosa protein isolate-based edible coating incorporated with ZnO nanoparticles on the shelf-life of chicken breast meat during cold storage. Food Hydrocoll. Health 2024, 5, 100176. [Google Scholar] [CrossRef]
- Przybyszewska, A.; Barbosa, C.H.; Pires, F.; Pires, J.R.A.; Rodrigues, C.; Galus, S.; Souza, V.G.L.; Alves, M.M.; Santos, C.F.; Coelhoso, I.; et al. Packaging of Fresh Poultry Meat with Innovative and Sustainable ZnO/Pectin Bionanocomposite Films—A Contribution to the Bio and Circular Economy. Coatings 2023, 13, 1208. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, Y.; Gao, Y.; Liu, Y.; Gao, C. Poly (lactic acid)-based pH responsive membrane combined with chitosan and alizarin for food packaging. Int. J. Biol. Macromol. 2022, 214, 348–359. [Google Scholar] [CrossRef]
- Mohammadi, H.; Kamkar, A.; Misaghi, A.; Zunabovic-Pichler, M.; Fatehi, S. Nanocomposite films with CMC, okra mucilage, and ZnO nanoparticles: Extending the shelf-life of chicken breast meat. Food Packag. Shelf Life 2019, 21, 100330. [Google Scholar] [CrossRef]
- Vergara-Figueroa, J.; Cerda-Leal, F.; Alejandro-Martín, S.; Gacitúa, W. Evaluation of the PLA-nZH-Cu Nanocomposite Film on the Micro-Biological, Organoleptic and Physicochemical Qualities of Packed Chicken Meat. Foods 2022, 11, 546. [Google Scholar] [CrossRef]
- Bhuyan, T.; Khanuja, M.; Sharma, R.; Patel, S.; Reddy, M.R.; Anand, S.; Varma, A. A comparative study of pure and copper (Cu)-doped ZnO nanorods for antibacterial and photocatalytic applications with their mechanism of action. J. Nanopart. Res. 2015, 17, 288. [Google Scholar] [CrossRef]
- Roy, S.; Priyadarshi, R.; Rhim, J.W. Gelatin/agar-based multifunctional film integrated with copper-doped zinc oxide nanoparticles and clove essential oil Pickering emulsion for enhancing the shelf life of pork meat. Food Res. Int. 2022, 160, 111690. [Google Scholar]






| Zn (µg/cm2) | Cu (µg/cm2) | |
|---|---|---|
| Si | 0.020 ± 0.001 | 0.003 ± 0.001 |
| ZnO | 9.86 ± 0.05 | 0.003 ± 0.001 |
| ZnO/Cu | 10.20 ± 0.05 | 0.13 ± 0.01 |
| Soaking Time (Day) | Samples | Mass Released (µg) | |
|---|---|---|---|
| Cu | Zn | ||
| 4 | PET-ZnO | // | 0.103 |
| PET-ZnO/Cu | 0.033 | 0.73 | |
| 21 | PET-ZnO | // | 0.144 |
| PET-ZnO/Cu | 0.04 | 0.942 | |
| MAL [Day] | SAL [Day] | Shelf Life [Day] | |||
|---|---|---|---|---|---|
| Mesophilic Bacteria (1 × 107) | Pseudomonas spp. (1 × 106) | Coliforms (1 × 105) | Score > 4 | ||
| CNT | >20 | 10.6 ± 0.5 b | >20 | 17.1 ± 0.186 c | 10.6 ± 0.5 b |
| PET | >20 | 12.8 ± 0.6 b | >20 | 19.9 ± 0.510 b | 12.7 ± 0.6 b |
| PET-ZnO | >20 | 16.4 ± 2.1 a | >20 | 24.4 ± 1.133 a | 16.4 ± 2.1 a |
| MAL [Day] | SAL [Day] | Shelf Life [Day] | |||||
|---|---|---|---|---|---|---|---|
| Mesoph. (1 × 107) | Pseud. (1 × 107) | Enterob. (1 × 106) | LAB (1 × 107) | Staphyl. (1 × 104) | Score > 5 | ||
| CNT | >11 | 1.9 ± 0.1 b | >11 | >11 | 0.6 ± 0.2 | 5.1 ± 0.3 b | 0.6 ± 0.2b b |
| PET | >11 | 2.2 ± 0.2 a,b | >11 | >11 | >11 | 5.9 ± 0.3 b | 2.2 ± 0.2 a |
| PET-ZnO | >11 | 2.6 ± 0.2 a | >11 | >11 | >11 | 11.0 ± 1.4 a | 2.6 ± 0.2 a |
| MAL [Day] | SAL [Day] | Shelf Life [Day] | |||||
|---|---|---|---|---|---|---|---|
| Mesoph. (1 × 107) | Pseud. (1 × 107) | Enterob. (1 × 106) | LAB (1 × 107) | Staphyl. (1 × 104) | Score > 5 | ||
| CNT | >13 | 2.0 ± 0.2 b | 11.5 ± 18 | >13 | 0.1 ± 0.7 b | 5.7 ± 0.08 b | 0.1 ± 0.7 b |
| PET | >13 | 2.6 ± 0.2 b | >13 | >13 | 0.5 ± 0.5 b | 5.9 ± 0.1 b | 0.5 ± 0.5 b |
| PET-ZnO/Cu | >13 | 3.2 ± 0.2 a | >13 | >13 | 2.1 ± 0.4 a | 10.2 ± 0.4 a | 2.0 ± 0.3 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lordi, A.; Del Sole, R.; Palumbo, F.; Perrotta, A.; Fracassi, F.; Roggio, M.; Milella, A.; Conte, A.; Del Nobile, M.A. Atomic Layer Deposition of ZnO and ZnO/Cu Coatings for Fresh Food Packaging Application. Polymers 2026, 18, 751. https://doi.org/10.3390/polym18060751
Lordi A, Del Sole R, Palumbo F, Perrotta A, Fracassi F, Roggio M, Milella A, Conte A, Del Nobile MA. Atomic Layer Deposition of ZnO and ZnO/Cu Coatings for Fresh Food Packaging Application. Polymers. 2026; 18(6):751. https://doi.org/10.3390/polym18060751
Chicago/Turabian StyleLordi, Adriana, Regina Del Sole, Fabio Palumbo, Alberto Perrotta, Francesco Fracassi, Marianna Roggio, Antonella Milella, Amalia Conte, and Matteo Alessandro Del Nobile. 2026. "Atomic Layer Deposition of ZnO and ZnO/Cu Coatings for Fresh Food Packaging Application" Polymers 18, no. 6: 751. https://doi.org/10.3390/polym18060751
APA StyleLordi, A., Del Sole, R., Palumbo, F., Perrotta, A., Fracassi, F., Roggio, M., Milella, A., Conte, A., & Del Nobile, M. A. (2026). Atomic Layer Deposition of ZnO and ZnO/Cu Coatings for Fresh Food Packaging Application. Polymers, 18(6), 751. https://doi.org/10.3390/polym18060751











