AI-Assisted Design of Chemically Recyclable Polymers for Food Packaging
Abstract
1. Introduction
2. Methods
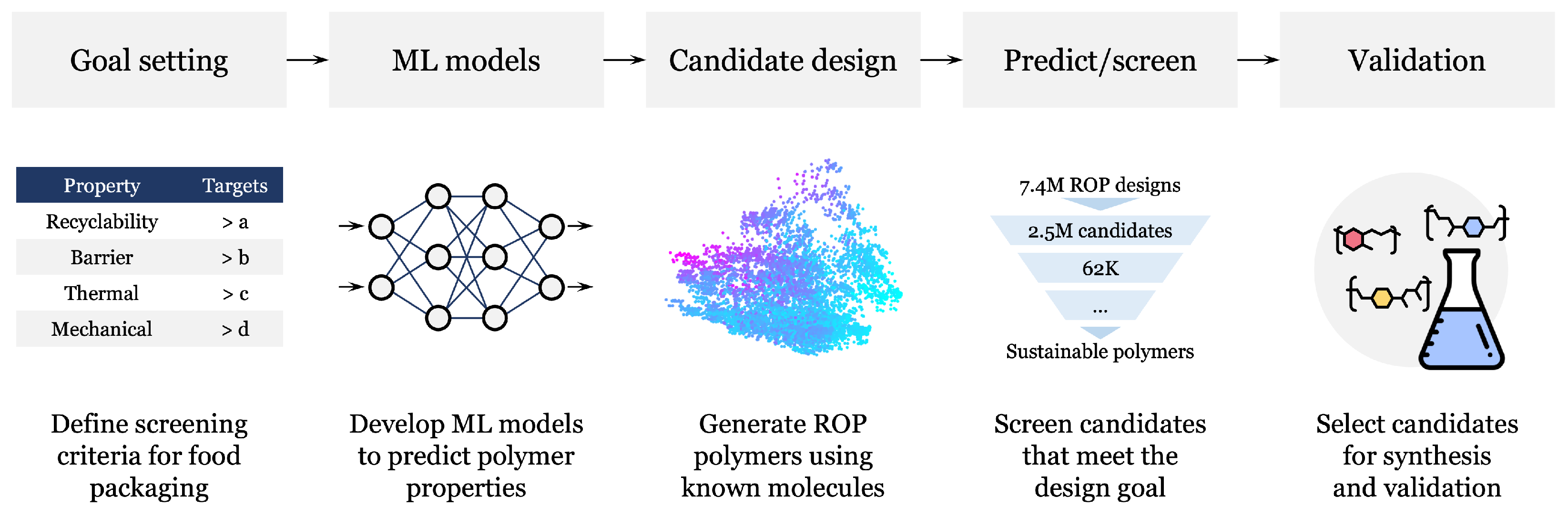
2.1. AI-Assisted Design Workflow
2.2. Synthesis of Poly-PDO from PDO
3. Results and Discussion
3.1. Screening of ROP Polymer Candidates
3.2. Experimental Validation of Film Performance
3.3. Chemical Recyclability
3.4. Practical Packaging Context and Structure–Property Considerations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Ebnesajjad, S. Plastic Films in Food Packaging: Materials, Technology and Applications; William Andrew: Norwich, NY, USA, 2012. [Google Scholar]
- Marsh, K.; Bugusu, B. Food Packaging: Roles, Materials, and Environmental Issues. J. Food Sci. 2007, 72, R39–R55. [Google Scholar] [CrossRef]
- Coles, R.; McDowell, D.; Kirwan, M. Food Packaging Technology, 2nd ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2011. [Google Scholar]
- Robertson, G.L. Food Packaging: Principles and Practice, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Kirwan, M.J. Handbook of Paper and Plastic Packaging Technology; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Verghese, K.; Lewis, H.; Lockrey, S.; Williams, H. Packaging’s role in minimizing food loss and waste across the supply chain. Packag. Technol. Sci. 2015, 28, 603–620. [Google Scholar] [CrossRef]
- Licciardello, F. Packaging, blessing in disguise. Review on its diverse contribution to food sustainability. Trends Food Sci. Technol. 2017, 65, 32–39. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Dalla Rosa, M. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2012, 19, 634–643. [Google Scholar] [CrossRef]
- Schmidt, J.; Grau, L.; Auer, M.; Maletz, R.; Woidasky, J. Multilayer Packaging in a Circular Economy. Polymers 2022, 14, 1825. [Google Scholar] [CrossRef]
- Hahladakis, J.N.; Iacovidou, E. Closing the loop on plastic packaging materials: What is quality and how does it affect recycling? Sci. Total Environ. 2018, 630, 1394–1400. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Hale, R.C.; Seeley, M.E.; La Guardia, M.J.; Mai, L.; Zeng, E.Y. A global perspective on microplastics. J. Geophys. Res. Ocean. 2020, 125, e2018JC014719. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.L. The plastic in microplastics: A review. Mar. Pollut. Bull. 2017, 119, 12–22. [Google Scholar] [CrossRef]
- Walker, T.W.; Frelka, N.; Shen, Z.; Chew, A.K.; Banick, J.; Grey, S.; Kim, M.S.; Dumesic, J.A.; Van Lehn, R.C.; Huber, G.W. Recycling of multilayer plastic packaging materials by solvent-targeted recovery and precipitation. Sci. Adv. 2020, 6, eaba7599. [Google Scholar] [CrossRef] [PubMed]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and chemical recycling of solid plastic waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef]
- Tamizhdurai, P.; Mangesh, V.L.; Santhosh, S.; Vedavalli, R.; Kavitha, C.; Bhutto, J.K.; Alreshidi, M.A.; Yadav, K.K.; Kumaran, R. A state-of-the-art review of multilayer packaging recycling: Challenges, alternatives, and outlook. J. Clean. Prod. 2024, 447, 141403. [Google Scholar] [CrossRef]
- Kern, J.; Su, Y.L.; Gutekunst, W.; Ramprasad, R. An informatics framework for the design of sustainable, chemically recyclable, synthetically accessible, and durable polymers. Npj Comput. Mater. 2025, 11, 182. [Google Scholar] [CrossRef]
- Xu, G.; Wang, Q. Chemically recyclable polymer materials: Polymerization and depolymerization cycles. Green Chem. 2022, 24, 2321. [Google Scholar] [CrossRef]
- Highmoore, J.F.; Kariyawasam, L.S.; Trenor, S.R.; Yang, Y. Design of depolymerizable polymers toward a circular economy. Green Chem. 2024, 26, 2384–2420. [Google Scholar] [CrossRef]
- Wimberger, L.; Ng, G.; Boyer, C. Light-driven polymer recycling to monomers and small molecules. Nat. Commun. 2024, 15, 2510. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.; Yaqoob, M.; Aggarwal, P. An overview of biodegradable packaging in food industry. J. Food Sci. Technol. 2021, 4, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Dhatt, P.S.; Hu, A.; Hu, C.; Huynh, V.; Dai, S.Y.; Yuan, J.S. Biomimetic layered, ecological, advanced, multi-functional film for sustainable packaging. Nat. Commun. 2025, 16, 6649. [Google Scholar] [CrossRef]
- Wu, F.; Misra, M.; Mohanty, A.K. Challenges and new opportunities on barrier performance of biodegradable polymers for sustainable packaging. Prog. Polym. Sci. 2021, 117, 101395. [Google Scholar] [CrossRef]
- Audus, D.J.; de Pablo, J.J. Polymer informatics: Opportunities and challenges. ACS Macro Lett. 2017, 6, 1078–1082. [Google Scholar] [CrossRef]
- Batra, R.; Song, L.; Ramprasad, R. Emerging materials intelligence ecosystems propelled by machine learning. Nat. Rev. Mater. 2021, 6, 655–678. [Google Scholar] [CrossRef]
- Tran, H.; Shen, K.H.; Shukla, S.; Kwon, H.K.; Ramprasad, R. Informatics-Driven Selection of Polymers for Fuel-Cell Applications. J. Phys. Chem. C 2023, 127, 977–986. [Google Scholar] [CrossRef]
- Zhu, G.; Kim, C.; Chandrasekarn, A.; Everett, J.D.; Ramprasad, R.; Lively, R.P. Polymer genome–based prediction of gas permeabilities in polymers. J. Polym. Eng. 2020, 40, 451–457. [Google Scholar] [CrossRef]
- Barnett, J.W.; Bilchak, C.R.; Wang, Y.; Benicewicz, B.C.; Murdock, L.A.; Bereau, T.; Kumar, S.K. Designing exceptional gas-separation polymer membranes using machine learning. Sci. Adv. 2020, 6, eaaz4301. [Google Scholar] [CrossRef]
- Wu, C.; Deshmukh, A.A.; Chen, L.; Ramprasad, R.; Sotzing, G.A.; Cao, Y. Rational design of all-organic flexible high-temperature polymer dielectrics. Matter 2022, 5, 2615–2623. [Google Scholar] [CrossRef]
- Nuyken, O.; Pask, S.D. Ring-Opening Polymerization—An Introductory Review. Polymers 2013, 5, 361–403. [Google Scholar] [CrossRef]
- Viacava, G.E.; Ansorena, M.R.; Marcovich, N.E. Chapter 18-Multilayered films for food packaging. In Nanostructured Materials for Food Packaging Applications; Jacob, J., Cacciotti, I., Thomas, S., Eds.; Micro and Nano Technologies, Elsevier: Amsterdam, The Netherlands, 2024; pp. 447–475. [Google Scholar] [CrossRef]
- Siracusa, V. Chapter 7—Packaging Material in the Food Industry. In Antimicrobial Food Packaging, 2nd ed.; Barros-Velázquez, J., Ed.; Academic Press: San Diego, CA, USA, 2025; pp. 111–126. [Google Scholar] [CrossRef]
- Saska, S.; Pilatti, L.; Silva, E.S.d.S.; Nagasawa, M.A.; Câmara, D.; Lizier, N.; Finger, E.; Dyszkiewicz Konwińska, M.; Kempisty, B.; Tunchel, S.; et al. Polydioxanone-based membranes for bone regeneration. Polymers 2021, 13, 1685. [Google Scholar] [CrossRef] [PubMed]
- Mathews, M.M. Polydioxanone: Applications in surgery and tissue engineering. In Synthetic Polymers in Drug and Biotherapeutics Delivery; Woodhead Publishing: Cambridge, UK, 2025; pp. 315–334. [Google Scholar]
- Goonoo, N.; Jeetah, R.; Bhaw-Luximon, A.; Jhurry, D. Polydioxanone-based bio-materials for tissue engineering and drug/gene delivery applications. Eur. J. Pharm. Biopharm. 2015, 97, 371–391. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.; Kim, C.; Gurnani, R.; Hvidsten, O.; DeSimpliciis, J.; Ramprasad, R.; Gadelrab, K.; Tuffile, C.; Molinari, N.; Kitchaev, D.; et al. Polymer composites informatics for flammability, thermal, mechanical and electrical property predictions. Polym. Chem. 2025, 16, 3459–3467. [Google Scholar] [CrossRef]
- Nistane, J.; Datta, R.; Lee, Y.J.; Sahu, H.; Jang, S.S.; Lively, R.; Ramprasad, R. Polymer design for solvent separations by integrating simulations, experiments and known physics via machine learning. Npj Comput. Mater. 2025, 11, 187. [Google Scholar] [CrossRef]
- Aklujkar, P.S.; Gurnani, R.; Rout, P.; Khomane, A.R.; Mutegi, I.; Desai, M.; Pollock, A.; Toribio, J.M.; Hao, J.; Cao, Y.; et al. Rationally designed high-temperature polymer dielectrics for capacitive energy storage: An experimental and computational alliance. Prog. Polym. Sci. 2025, 161, 101931. [Google Scholar] [CrossRef]
- Schertzer, W.; Shukla, S.; Sose, A.; Rafiq, R.; Al Otmi, M.; Sampath, J.; Lively, R.P.; Ramprasad, R. AI-driven design of fluorine-free polymers for sustainable and high-performance anion exchange membranes. J. Mater. Inform. 2025, 5, 5. [Google Scholar] [CrossRef]
- Atasi, C.; Kern, J.; Ramprasad, R. Design of recyclable plastics with machine learning and genetic algorithm. J. Chem. Inf. Model. 2024, 64, 9249–9259. [Google Scholar] [CrossRef]
- Matmerize, Inc. PolymRizeTM, version 0.27.0; A Standardized Software for Polymer Informatics; Matmerize, Inc.: Atlanta, GA, USA, 2021; Available online: https://polymrize.matmerize.com (accessed on 12 March 2026).
- Phan, B.K.; Shen, K.H.; Gurnani, R.; Tran, H.; Lively, R.; Ramprasad, R. Gas permeability, diffusivity, and solubility in polymers: Simulation-experiment data fusion and multi-task machine learning. Npj Comput. Mater. 2024, 10, 186. [Google Scholar] [CrossRef]
- Toland, A.; Tran, H.; Chen, L.; Li, Y.; Zhang, C.; Gutekunst, W.; Ramprasad, R. Accelerated scheme to predict ring-opening polymerization enthalpy: Simulation-experimental data fusion and multitask machine learning. J. Phys. Chem. A 2023, 127, 10709–10716. [Google Scholar] [CrossRef]
- Rasmussen, C.E.; Williams, C.K.I. Gaussian Processes for Machine Learning; MIT Press: Cambridge, MA, USA, 2005. [Google Scholar]
- Doan Tran, H.; Kim, C.; Chen, L.; Chandrasekaran, A.; Batra, R.; Venkatram, S.; Kamal, D.; Lightstone, J.P.; Gurnani, R.; Shetty, P.; et al. Machine-learning predictions of polymer properties with Polymer Genome. J. Appl. Phys. 2020, 128, 171104. [Google Scholar] [CrossRef]
- Ertl, P.; Schuffenhauer, A. Estimation of synthetic accessibility score of drug-like molecules based on molecular complexity and fragment contributions. J. Cheminform. 2009, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Virtual Forward Synthesis (VFS)-Ring Opening Polymerization (ROP) Results Data. 2025. Available online: https://github.com/Ramprasad-Group/polyVERSE/tree/main/Virtual-Polymer/VFS/ROP/packaging_replacements (accessed on 12 March 2026).
- Libiszowski, J.; Kowalski, A.; Szymanski, R.; Duda, A.; Raquez, J.M.; Degée, P.; Dubois, P. Monomer- linear macromolecules- cyclic oligomers equilibria in the polymerization of 1, 4-dioxan-2-one. Macromolecules 2004, 37, 52–59. [Google Scholar] [CrossRef]
- Lelkes, K.D.; Jezbera, D.; Svoboda, R.; Podzimek, Š.; Loskot, J.; Nalezinková, M.; Voda, P.; Duda, P.; Fučíková, A.M.; Hosszú, T.; et al. A comprehensive study on the degradation process of medical-grade polydioxanone at low pH. Polym. Test. 2024, 138, 108536. [Google Scholar] [CrossRef]
- Liu, X.; Hou, P.; Liu, S.; Qi, J.; Feng, S.; Zhang, L.; Ma, P.; Bai, W. Effect of poly (lactic-co-glycolic acid) blend ratios on the hydrolytic degradation of poly (para-dioxanone). J. Polym. Res. 2021, 28, 166. [Google Scholar] [CrossRef]
- Kang, H.J.; Song, Y.S. Effects of humidity and temperature on hydrolytic degradation of polydioxanone. Polym. Eng. Sci. 2022, 62, 2070–2078. [Google Scholar] [CrossRef]
- Choi, S.S.; Lee, J.H.; Lee, S.H. Physical nature of biodegradable polydioxanone filaments upon synthetic conditions. Polymer 2020, 44, 505–511. [Google Scholar]
- Nishida, H.; Konno, M.; Ikeda, A.; Tokiwa, Y. Microbial degradation of poly (p-dioxanone) I. Isolation of degrading microorganisms and microbial decomposition in pure culture. Polym. Degrad. Stab. 2000, 68, 205–217. [Google Scholar] [CrossRef]
- Jin, C.; Liang, B.; Li, J.; Li, F. Biodegradation behaviors of poly (p-dioxanone) in different environment media. J. Polym. Environ. 2013, 21, 1088–1099. [Google Scholar] [CrossRef]
- Sabino, M.A.; González, S.; Márquez, L.; Feijoo, J.L. Study of the hydrolytic degradation of polydioxanone PPDX. Polym. Degrad. Stab. 2000, 69, 209–216. [Google Scholar] [CrossRef]
- Ooi, C.P.; Cameron, R.E. he hydrolytic degradation of polydioxanone (PDSII) sutures. Part I: Morphological aspects. J. Biomed. Mater. Res. 2002, 63, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Weininger, D. SMILES, a chemical language and information system. 1. Introduction to methodology and encoding rules. J. Chem. Inf. Comput. Sci. 1988, 28, 31–36. [Google Scholar] [CrossRef]
- Lee, Y.J.; Robinson, A.M.; Jang, W.J.; Ye, Z.; Ren, Y.; Xia, Y.; Lively, R.P. Prediction-driven selection of microporous polymer membranes for organic solvent reverse osmosis. J. Membr. Sci. 2025, 734, 124475. [Google Scholar] [CrossRef]
- PACE. Partnership for an Advanced Computing Environment (PACE); Georgia Institute of Technology: Atlanta, GA, USA, 2017. [Google Scholar]




| Property | Desired Target |
|---|---|
| Enthalpy of polymerization | to kJ/mol |
| Water vapor permeability at 25 °C | < a |
| Oxygen permeability at 25 °C | < a |
| Glass transition temperature | <298 K |
| Melting temperature | >373 K |
| Degradation temperature | >473 K |
| Elongation at break | >% |
| Tensile strength | >20 MPa |
| Property | Datapoints | Model Type | Metric | CV Testset Error |
|---|---|---|---|---|
| Enthalpy of polymerization | 109 | MT-GPR | RMSE | 0.067 kJ/mol |
| Water vapor permeability | 36 | MT-NN | OME | 0.198 |
| Oxygen permeability | 747 | |||
| Glass transition temperature | 8962 | ST-NN | RMSE | 31 K |
| Melting temperature | 3938 | ST-NN | RMSE | 53 K |
| Degradation temperature | 4563 | ST-NN | RMSE | 72 K |
| Elongation at break | 1351 | MT-NN | OME | 0.37 |
| Tensile strength | 1023 | MT-NN | RMSE | 21 MPa |
| Polymer | C-1 | C-2 | C-3 | C-4 | C-5 |
|---|---|---|---|---|---|
| Enthalpy of polymerization (kJ/mol) | −12.7 | −17.08 | −16.17 | −16.93 | −16.47 |
| Water vapor permeability at 25 °C () | |||||
| Oxygen permeability at 25 °C () | |||||
| Glass transition temperature (K) | 261.9 | 238.6 | 269.1 | 272.4 | 203.8 |
| Melting temperature (K) | 360.5 | 414.7 | 385.4 | 474.3 | 376.1 |
| Degradation temperature (K) | 524 | 479.8 | 538.6 | 605.8 | 507.4 |
| Elongation at break (%) | 191 | 151 | 204 | 162 | 174 |
| Tensile strength (MPa) | 32.62 | 33.54 | 55.5 | 24.83 | 35.13 |
 | |||||
| Property | Prediction | Measured | |
|---|---|---|---|
| This Work | Literature | ||
| Enthalpy of polymerization (kJ/mol) | - | −13.8 a | |
| Water vapor permeability at 25 °C | - | ||
| ()) | |||
| Oxygen permeability at 25 °C | - | ||
| ()) | |||
| Glass transition temperature (K) | 257 | 261 to 263 b,c | |
| Melting temperature (K) | 378 | 363 to 397 b,c | |
| Degradation temperature (K) | 487 | - | |
| Elongation at break (%) | to 102.77 c,d | ||
| Tensile strength (MPa) | to 60 d,e | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Phan, B.K.; Kim, C.; Nistane, J.; Xiong, W.; Chen, H.; Jang, W.J.; Gholami, F.; Su, Y.; Qi, J.; Lively, R.; et al. AI-Assisted Design of Chemically Recyclable Polymers for Food Packaging. Polymers 2026, 18, 730. https://doi.org/10.3390/polym18060730
Phan BK, Kim C, Nistane J, Xiong W, Chen H, Jang WJ, Gholami F, Su Y, Qi J, Lively R, et al. AI-Assisted Design of Chemically Recyclable Polymers for Food Packaging. Polymers. 2026; 18(6):730. https://doi.org/10.3390/polym18060730
Chicago/Turabian StylePhan, Brandon K., Chiho Kim, Janhavi Nistane, Wei Xiong, Haoyu Chen, Woo Jin Jang, Farzad Gholami, Yongliang Su, Jerry Qi, Ryan Lively, and et al. 2026. "AI-Assisted Design of Chemically Recyclable Polymers for Food Packaging" Polymers 18, no. 6: 730. https://doi.org/10.3390/polym18060730
APA StylePhan, B. K., Kim, C., Nistane, J., Xiong, W., Chen, H., Jang, W. J., Gholami, F., Su, Y., Qi, J., Lively, R., Gutekunst, W., & Ramprasad, R. (2026). AI-Assisted Design of Chemically Recyclable Polymers for Food Packaging. Polymers, 18(6), 730. https://doi.org/10.3390/polym18060730








