Sustainable Thin-Film Composite Mixed-Matrix Membranes Based on Cellulose Acetate, Bimetallic ZIF-8-67, and Ionic Liquid for Enhanced Propene/Propane Separation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. MOF Synthesis
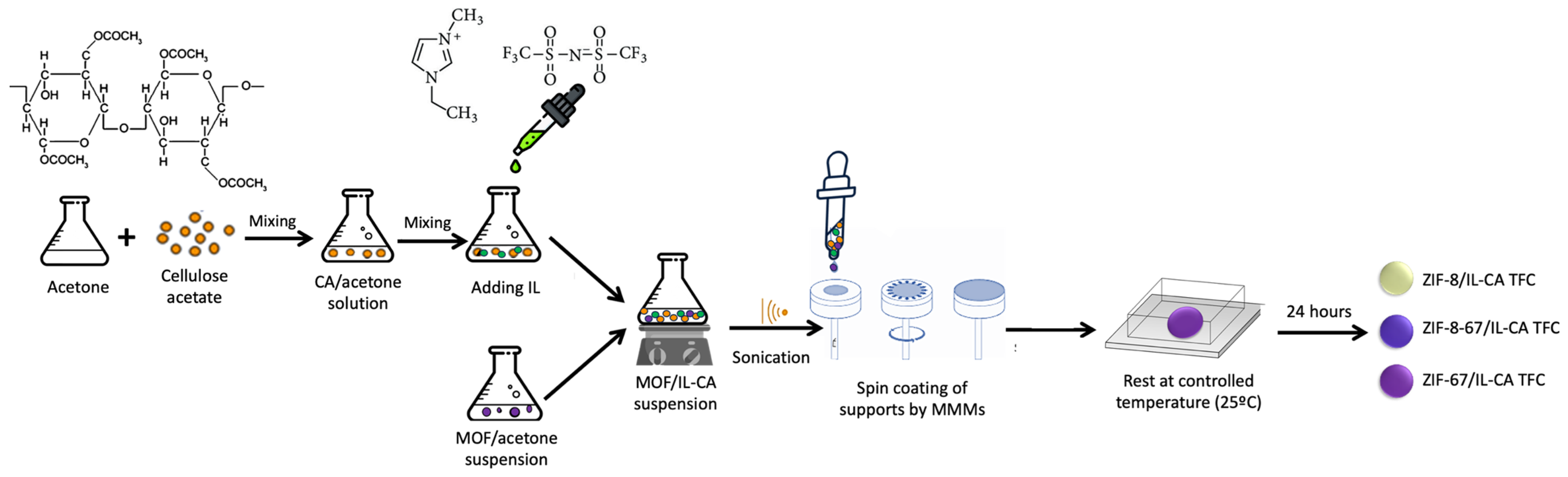
2.3. Membrane Preparation
2.4. Physicochemical Characterisations
2.5. Gas Transport Properties
2.5.1. Fixed-Volume Single-Gas Permeation Analyser
2.5.2. Mixed-Gas Permeation
3. Results and Discussions
3.1. Structural Analysis of ZIFs and MMMs
3.1.1. ZIF Porosimetry Analysis
3.1.2. MOF Structural Microanalysis
3.2. Crystallinity Identification of MOFs and MMMs (X-Ray Diffractometer (XRD) Analysis)
3.3. Fourier Transform Infrared Spectroscopy (FT-IR) Analysis
3.4. Membrane Fabrication and Characterisation
3.5. Gas Transport Studies
3.5.1. Single-Gas Permeation Properties of TFCs on PAN
CO2/CH4 and CO2/N2 Pairs

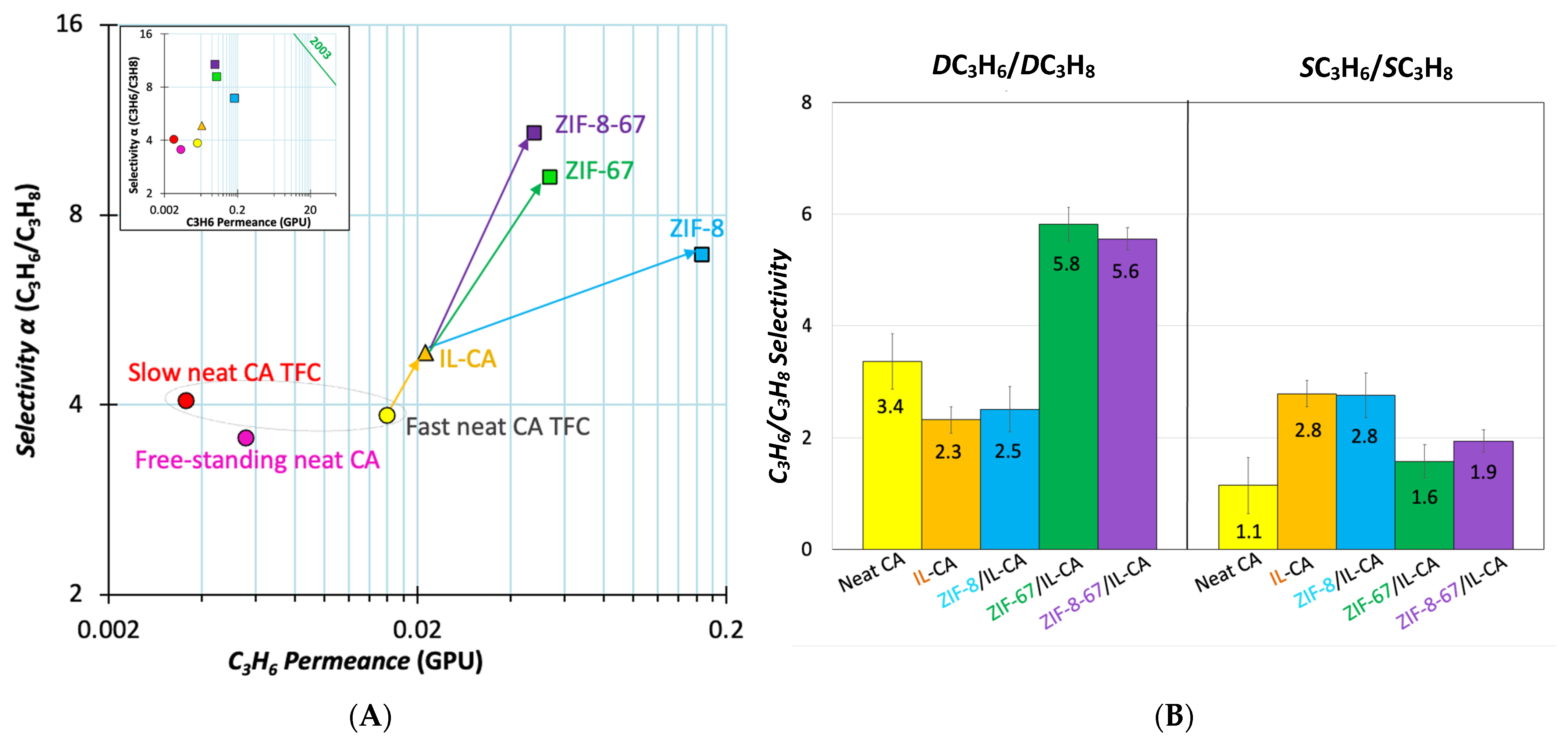
Permeation of the C3H6/C3H8 Gas Pair
Mechanistic Insight into ZIF-8, ZIF-67, and Synergistic Effect of Bimetallic ZIF-8-67 (MTV-ZIF)
3.5.2. Propene/Propane Mixture Separation
4. Conclusions and Outlook
- The transport properties of MMM TFCs on PAN and PTFE prioritised PAN support over PTFE.
- Higher coating speeds led to the formation of thinner selective layers and enhanced gas diffusion, benefiting CO2 and C3H6 separations.
- The presence of [BMIM]+[Tf2N]− enhanced ZIF and CA compatibility, facilitated CO2 and C3H6 permeation, and increased C3H6/C3H8 ideal selectivity, while CO2/CH4 and CO2/N2 separation showed a trade-off behaviour.
- The contribution of ZIFs further enhanced CO2 and C3H6 permeances, with ZIF-8-67 and ZIF-67 presenting the highest CO2/CH4 and CO2/N2 ideal selectivities. In C3H6/C3H8 separation, ZIF-8-67 exhibited the best ideal selectivity.
- Mixed-gas separation experiments also confirmed a high selectivity for propene over propane via the ZIF-8-67/IL-CA TFC, demonstrating the practical relevance of this membrane.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Bidwai, S.; Shivarkar, A. Propylene Market Size, Share, and Trends 2024 to 2034, Report Code: 2689, May 2025. Available online: https://www.precedenceresearch.com/propylene-market (accessed on 11 December 2025).
- Eldridge, R.B. Olefin/Paraffin Separation Technology: A Review. Ind. Eng. Chem. Res. 1993, 32, 2208–2212. [Google Scholar] [CrossRef]
- Moulijn, J.A.; Makkee, M.; Van Diepen, A.E. Chemical Process Technology; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Ma, X.; Williams, S.; Wei, X.; Kniep, J.; Lin, Y.S. Propylene/Propane Mixture Separation Characteristics and Stability of Carbon Molecular Sieve Membranes. Ind. Eng. Chem. Res. 2015, 54, 9824–9831. [Google Scholar] [CrossRef]
- Burns, R.L.; Koros, W.J. Defining the Challenges for C3H6/C3H8 Separation Using Polymeric Membranes. J. Membr. Sci. 2003, 211, 299. [Google Scholar] [CrossRef]
- Hou, J.; Liu, P.; Jiang, M.; Yu, L.; Li, L.; Tang, Z. Olefin/Paraffin Separation through Membranes: From Mechanisms to Critical Materials. J. Mater. Chem. A Mater. 2019, 7, 23489–23511. [Google Scholar] [CrossRef]
- Li, J.-R.; Kuppler, R.J.; Zhou, H.-C. Selective Gas Adsorption and Separation in Metal–Organic Frameworks. Chem. Soc. Rev. 2009, 38, 1477–1504. [Google Scholar] [CrossRef]
- Kuppler, R.J.; Timmons, D.J.; Fang, Q.-R.; Li, J.-R.; Makal, T.A.; Young, M.D.; Yuan, D.; Zhao, D.; Zhuang, W.; Zhou, H.-C. Potential Applications of Metal-Organic Frameworks. Coord. Chem. Rev. 2009, 253, 3042–3066. [Google Scholar] [CrossRef]
- Jiang, H.; Alezi, D.; Eddaoudi, M. A Reticular Chemistry Guide for the Design of Periodic Solids. Nat. Rev. Mater. 2021, 6, 466–487. [Google Scholar] [CrossRef]
- Lamia, N.; Jorge, M.; Granato, M.A.; Paz, F.A.A.; Chevreau, H.; Rodrigues, A.E. Adsorption of Propane, Propylene and Isobutane on a Metal–Organic Framework: Molecular Simulation and Experiment. Chem. Eng. Sci. 2009, 64, 3246–3259. [Google Scholar] [CrossRef]
- Wuttke, S.; Bazin, P.; Vimont, A.; Serre, C.; Seo, Y.; Hwang, Y.K.; Chang, J.; Férey, G.; Daturi, M. Discovering the Active Sites for C3 Separation in MIL-100 (Fe) by Using Operando IR Spectroscopy. Chem.–A Eur. J. 2012, 18, 11959–11967. [Google Scholar] [CrossRef]
- Bao, Z.; Alnemrat, S.; Yu, L.; Vasiliev, I.; Ren, Q.; Lu, X.; Deng, S. Adsorption of Ethane, Ethylene, Propane, and Propylene on a Magnesium-Based Metal–Organic Framework. Langmuir 2011, 27, 13554–13562. [Google Scholar] [CrossRef]
- Bloch, E.D.; Queen, W.L.; Krishna, R.; Zadrozny, J.M.; Brown, C.M.; Long, J.R. Hydrocarbon Separations in a Metal-Organic Framework with Open Iron (II) Coordination Sites. Science 2012, 335, 1606–1610. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Wang, C.; Xiang, L.; Xu, Y.; Pan, Y. Enhanced C3H6/C3H8 Separation Performance in Poly (Vinyl Acetate) Membrane Blended with ZIF-8 Nanocrystals. Chem. Eng. Sci. 2018, 179, 1–12. [Google Scholar] [CrossRef]
- Liu, D.; Xiang, L.; Chang, H.; Chen, K.; Wang, C.; Pan, Y.; Li, Y.; Jiang, Z. Rational Matching between MOFs and Polymers in Mixed Matrix Membranes for Propylene/Propane Separation. Chem. Eng. Sci. 2019, 204, 151–160. [Google Scholar] [CrossRef]
- Zhang, C.; Dai, Y.; Johnson, J.R.; Karvan, O.; Koros, W.J. High Performance ZIF-8/6FDA-DAM Mixed Matrix Membrane for Propylene/Propane Separations. J. Membr. Sci. 2012, 389, 34–42. [Google Scholar] [CrossRef]
- Ma, X.; Swaidan, R.J.; Wang, Y.; Hsiung, C.; Han, Y.; Pinnau, I. Highly Compatible Hydroxyl-Functionalized Microporous Polyimide-ZIF-8 Mixed Matrix Membranes for Energy Efficient Propylene/Propane Separation. ACS Appl. Nano Mater. 2018, 1, 3541–3547. [Google Scholar] [CrossRef]
- Japip, S.; Wang, H.; Xiao, Y.; Chung, T.S. Highly Permeable Zeolitic Imidazolate Framework (ZIF)-71 Nano-Particles Enhanced Polyimide Membranes for Gas Separation. J. Membr. Sci. 2014, 467, 162–174. [Google Scholar] [CrossRef]
- Hillman, F.; Zimmerman, J.M.; Paek, S.-M.; Hamid, M.R.A.; Lim, W.T.; Jeong, H.-K. Rapid Microwave-Assisted Synthesis of Hybrid Zeolitic–Imidazolate Frameworks with Mixed Metals and Mixed Linkers. J. Mater. Chem. A Mater. 2017, 5, 6090–6099. [Google Scholar] [CrossRef]
- Lee, M.J.; Kwon, H.T.; Jeong, H.K. High-Flux Zeolitic Imidazolate Framework Membranes for Propylene/Propane Separation by Postsynthetic Linker Exchange. Angew. Chem.-Int. Ed. 2018, 57, 156–161. [Google Scholar] [CrossRef]
- Askari, M.; Chung, T.-S. Natural Gas Purification and Olefin/Paraffin Separation Using Thermal Cross-Linkable Co-Polyimide/ZIF-8 Mixed Matrix Membranes. J. Membr. Sci. 2013, 444, 173–183. [Google Scholar] [CrossRef]
- Böhme, U.; Barth, B.; Paula, C.; Kuhnt, A.; Schwieger, W.; Mundstock, A.; Caro, J.; Hartmann, M. Ethene/Ethane and Propene/Propane Separation via the Olefin and Paraffin Selective Metal–Organic Framework Adsorbents CPO-27 and ZIF-8. Langmuir 2013, 29, 8592–8600. [Google Scholar] [CrossRef]
- Zhang, C.; Lively, R.P.; Zhang, K.; Johnson, J.R.; Karvan, O.; Koros, W.J. Unexpected Molecular Sieving Properties of Zeolitic Imidazolate Framework-8. J. Phys. Chem. Lett. 2012, 3, 2130–2134. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Olson, D.H.; Seidel, J.; Emge, T.J.; Gong, H.; Zeng, H.; Li, J. Zeolitic Imidazolate Frameworks for Kinetic Separation of Propane and Propene. J. Am. Chem. Soc. 2009, 131, 10368–10369. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Rong, Z.; Nguyen, H.L.; Yaghi, O.M. Structural Chemistry of Zeolitic Imidazolate Frameworks. Inorg. Chem. 2023, 62, 20861–20873. [Google Scholar] [CrossRef] [PubMed]
- Krokidas, P.; Castier, M.; Moncho, S.; Sredojevic, D.N.; Brothers, E.N.; Kwon, H.T.; Jeong, H.-K.; Lee, J.S.; Economou, I.G. ZIF-67 Framework: A Promising New Candidate for Propylene/Propane Separation. Experimental Data and Molecular Simulations. J. Phys. Chem. C 2016, 120, 8116–8124. [Google Scholar] [CrossRef]
- Ordonez, M.J.C.; Balkus, K.J., Jr.; Ferraris, J.P.; Musselman, I.H. Molecular Sieving Realized with ZIF-8/Matrimid® Mixed-Matrix Membranes. J. Membr. Sci. 2010, 361, 28–37. [Google Scholar] [CrossRef]
- Thompson, J.A.; Vaughn, J.T.; Brunelli, N.A.; Koros, W.J.; Jones, C.W.; Nair, S. Mixed-Linker Zeolitic Imidazolate Framework Mixed-Matrix Membranes for Aggressive CO2 Separation from Natural Gas. Microporous Mesoporous Mater. 2014, 192, 43–51. [Google Scholar] [CrossRef]
- An, H.; Park, S.; Kwon, H.T.; Jeong, H.-K.; Lee, J.S. A New Superior Competitor for Exceptional Propylene/Propane Separations: ZIF-67 Containing Mixed Matrix Membranes. J. Membr. Sci. 2017, 526, 367–376. [Google Scholar] [CrossRef]
- Oh, J.W.; Cho, K.Y.; Kan, M.-Y.; Yu, H.J.; Kang, D.-Y.; Lee, J.S. High-Flux Mixed Matrix Membranes Containing Bimetallic Zeolitic Imidazole Framework-8 for C3H6/C3H8 Separation. J. Membr. Sci. 2020, 596, 117735. [Google Scholar] [CrossRef]
- Loloei, M.; Kaliaguine, S.; Rodrigue, D. CO2-Selective Mixed Matrix Membranes of Bimetallic Zn/Co-ZIF vs. ZIF-8 and ZIF-67. Sep. Purif. Technol. 2022, 296, 121391. [Google Scholar] [CrossRef]
- Kaur, G.; Rai, R.K.; Tyagi, D.; Yao, X.; Li, P.-Z.; Yang, X.-C.; Zhao, Y.; Xu, Q.; Singh, S.K. Room-Temperature Synthesis of Bimetallic Co–Zn Based Zeolitic Imidazolate Frameworks in Water for Enhanced CO2 and H2 Uptakes. J. Mater. Chem. A Mater. 2016, 4, 14932–14938. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, Y.; Hu, L.; Liu, N.; Xu, J.; Zhou, J. Using Lantern Zn/Co-ZIF Nanoparticles to Provide Channels for CO2 Permeation through PEO-Based MMMs. J. Membr. Sci. 2020, 597, 117644. [Google Scholar] [CrossRef]
- Pan, Y.; Liu, Y.; Zeng, G.; Zhao, L.; Lai, Z. Rapid Synthesis of Zeolitic Imidazolate Framework-8 (ZIF-8) Nanocrystals in an Aqueous System. Chem. Commun. 2011, 47, 2071–2073. [Google Scholar] [CrossRef]
- Butova, V.V.; Budnyk, A.P.; Bulanova, E.A.; Lamberti, C.; Soldatov, A. V Hydrothermal Synthesis of High Surface Area ZIF-8 with Minimal Use of TEA. Solid State Sci. 2017, 69, 13–21. [Google Scholar] [CrossRef]
- Kida, K.; Okita, M.; Fujita, K.; Tanaka, S.; Miyake, Y. Formation of High Crystalline ZIF-8 in an Aqueous Solution. CrystEngComm 2013, 15, 1794–1801. [Google Scholar] [CrossRef]
- He, M.; Yao, J.; Liu, Q.; Wang, K.; Chen, F.; Wang, H. Facile Synthesis of Zeolitic Imidazolate Framework-8 from a Concentrated Aqueous Solution. Microporous Mesoporous Mater. 2014, 184, 55–60. [Google Scholar] [CrossRef]
- Gross, A.F.; Sherman, E.; Vajo, J.J. Aqueous Room Temperature Synthesis of Cobalt and Zinc Sodalite Zeolitic Imidizolate Frameworks. Dalton Trans. 2012, 41, 5458–5460. [Google Scholar] [CrossRef]
- McCarthy, M.C.; Varela-Guerrero, V.; Barnett, G.V.; Jeong, H.-K. Synthesis of Zeolitic Imidazolate Framework Films and Membranes with Controlled Microstructures. Langmuir 2010, 26, 14636–14641. [Google Scholar] [CrossRef]
- Yao, J.; He, M.; Wang, K.; Chen, R.; Zhong, Z.; Wang, H. High-Yield Synthesis of Zeolitic Imidazolate Frameworks from Stoichiometric Metal and Ligand Precursor Aqueous Solutions at Room Temperature. CrystEngComm 2013, 15, 3601–3606. [Google Scholar] [CrossRef]
- Fischer, S.; Thümmler, K.; Volkert, B.; Hettrich, K.; Schmidt, I.; Fischer, K. Properties and Applications of Cellulose Acetate. In Proceedings of the Macromolecular Symposia; Wiley Online Library: Hoboken, NJ, USA, 2008; Volume 262, pp. 89–96. [Google Scholar] [CrossRef]
- Akbarzadeh, E.; Shockravi, A.; Vatanpour, V. High Performance Compatible Thiazole-Based Polymeric Blend Cellulose Acetate Membrane as Selective CO2 Absorbent and Molecular Sieve. Carbohydr. Polym. 2021, 252, 117215. [Google Scholar] [CrossRef]
- Nikolaeva, D.; Verachtert, K.; Azcune, I.; Jansen, J.C.; Vankelecom, I.F.J. Influence of Ionic Liquid-like Cationic Pendants Composition in Cellulose Based Polyelectrolytes on Membrane-Based CO2 Separation. Carbohydr. Polym. 2021, 255, 117375. [Google Scholar] [CrossRef]
- Wei, D.W.; Wei, H.; Gauthier, A.C.; Song, J.; Jin, Y.; Xiao, H. Superhydrophobic Modification of Cellulose and Cotton Textiles: Methodologies and Applications. J. Bioresour. Bioprod. 2020, 5, 1–15. [Google Scholar] [CrossRef]
- Miao, X.; Lin, J.; Bian, F. Utilization of Discarded Crop Straw to Produce Cellulose Nanofibrils and Their Assemblies. J. Bioresour. Bioprod. 2020, 5, 26–36. [Google Scholar] [CrossRef]
- Asad, A.; Sameoto, D.; Sadrzadeh, M. Overview of Membrane Technology. In Nanocomposite Membranes for Water and Gas Separation; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–28. [Google Scholar]
- Li, H.; Xu, S.; Zhao, B.; Yu, Y.; Liu, Y. The Phase Structural Evolution and Gas Separation Performances of Cellulose Acetate/Polyimide Composite Membrane from Polymer to Carbon Stage. Membranes 2021, 11, 618. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Voicu, S.I. Recent Advances in Cellulose and Chitosan Based Membranes for Water Purification: A Concise Review. Carbohydr. Polym. 2016, 146, 148–165. [Google Scholar] [CrossRef]
- Doosti, M.; Abedini, R. Polyethyleneglycol-Modified Cellulose Acetate Membrane for Efficient Olefin/Paraffin Separation. Energy Fuels 2022, 36, 10082–10095. [Google Scholar] [CrossRef]
- Ito, A.; Hwang, S.-T. Permeation of Propane and Propylene Through Cellulosic Polymer Membranes. J. Appl. Polym. Sci. 1989, 38, 483–490. [Google Scholar] [CrossRef]
- Naghsh, M.; Sadeghi, M.; Moheb, A.; Chenar, M.P.; Mohagheghian, M. Separation of Ethylene/Ethane and Propylene/Propane by Cellulose Acetate–Silica Nanocomposite Membranes. J. Membr. Sci. 2012, 423, 97–106. [Google Scholar] [CrossRef]
- Rattanawong, W.; Osuwan, S.; Rirksomboon, T.; Kulprathipanja, S. The NaX-Zeolites/Cellulose Acetate Mixed Matrix Membrane for Olefin/Paraffin Separation: Membrane Technology in Petrolum, Petrochemical and Gas Processing. Prepr.-Am. Chem. Soc. Div. Pet. Chem. 2001, 46, 166–167. [Google Scholar]
- Sridhar, S.; Khan, A.A. Simulation Studies for the Separation of Propylene and Propane by Ethylcellulose Membrane. J. Membr. Sci. 1999, 159, 209–219. [Google Scholar] [CrossRef]
- Hajivand, P.; Longo, M.; Mastropietro, T.F.; Godbert, N.; Monteleone, M.; Bezzu, C.G.; Armentano, D.; Jansen, J.C. Tailoring the Thermal, Mechanical, and Gas Transport Properties of Cellulose Acetate Membranes with Ionic Liquids for Efficient Propene/Propane Separation. Polymer 2025, 333, 128679. [Google Scholar] [CrossRef]
- Fraga, S.C.; Monteleone, M.; Lanč, M.; Esposito, E.; Fuoco, A.; Giorno, L.; Pilnáček, K.; Friess, K.; Carta, M.; McKeown, N.B. A Novel Time Lag Method for the Analysis of Mixed Gas Diffusion in Polymeric Membranes by On-Line Mass Spectrometry: Method Development and Validation. J. Membr. Sci. 2018, 561, 39–58. [Google Scholar] [CrossRef]
- Monteleone, M.; Esposito, E.; Fuoco, A.; Lanč, M.; Pilnáček, K.; Friess, K.; Bezzu, C.; Carta, M.; McKeown, N.; Jansen, J.C. A Novel Time Lag Method for the Analysis of Mixed Gas Diffusion in Polymeric Membranes by On-Line Mass Spectrometry: Pressure Dependence of Transport Parameters. Membranes 2018, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Sonwane, C.G.; Bhatia, S.K. Characterization of Pore Size Distributions of Mesoporous Materials from Adsorption Isotherms. J. Phys. Chem. B 2000, 104, 9099–9110. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Cravillon, J.; Münzer, S.; Lohmeier, S.-J.; Feldhoff, A.; Huber, K.; Wiebcke, M. Rapid Room-Temperature Synthesis and Characterization of Nanocrystals of a Prototypical Zeolitic Imidazolate Framework. Chem. Mater. 2009, 21, 1410–1412. [Google Scholar] [CrossRef]
- Chang, P.-H.; Lee, Y.-T.; Peng, C.-H. Synthesis and Characterization of Hybrid Metal Zeolitic Imidazolate Framework Membrane for Efficient H2/CO2 Gas Separation. Materials 2020, 13, 5009. [Google Scholar] [CrossRef]
- Kwon, H.T.; Jeong, H.-K.; Lee, A.S.; An, H.S.; Lee, J.S. Heteroepitaxially Grown Zeolitic Imidazolate Framework Membranes with Unprecedented Propylene/Propane Separation Performances. J. Am. Chem. Soc. 2015, 137, 12304–12311. [Google Scholar] [CrossRef]
- Li, J.; Chang, H.; Li, Y.; Li, Q.; Shen, K.; Yi, H.; Zhang, J. Synthesis and Adsorption Performance of La@ ZIF-8 Composite Metal–Organic Frameworks. RSC Adv. 2020, 10, 3380–3390. [Google Scholar] [CrossRef]
- Guo, X.; Xing, T.; Lou, Y.; Chen, J. Controlling ZIF-67 Crystals Formation through Various Cobalt Sources in Aqueous Solution. J. Solid State Chem. 2016, 235, 107–112. [Google Scholar] [CrossRef]
- International Centre for Diffraction Data (ICDD). PDF® Card No. 00-062-1030, Zeolitic Imidazolate Framework-8 (ZIF-8); Powder Diffraction File; ICDD: Newtown Square, PA, USA.
- International Centre for Diffraction Data (ICDD). ZIF-67 (Cobalt Zeolitic Imidazolate Framework); Powder Diffraction File; ICDD: Newtown Square, PA, USA.
- Banerjee, R.; Phan, A.; Wang, B.; Knobler, C.; Furukawa, H.; O’Keeffe, M.; Yaghi, O.M. High-Throughput Synthesis of Zeolitic Imidazolate Frameworks and Application to CO2 Capture. Science 2008, 319, 939–943. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Côté, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional Chemical and Thermal Stability of Zeolitic Imidazolate Frameworks. Proc. Natl. Acad. Sci. USA 2006, 103, 10186–10191. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, S.; Shanti, R.; Morris, E. Characterization of Conducting Cellulose Acetate Based Polymer Electrolytes Doped with “Green” Ionic Mixture. Carbohydr. Polym. 2013, 91, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Okafuji, A.; Kato, T.; Ohno, H. Induction of an Infinite Periodic Minimal Surface by Endowing an Amphiphilic Zwitterion with Halogen-Bond Ability. ChemistryOpen 2016, 5, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Mizuno, M. Characteristic Spectroscopic Features Because of Cation–Anion Interactions Observed in the 700–950 cm−1 Range of Infrared Spectroscopy for Various Imidazolium-Based Ionic Liquids. ACS Omega 2018, 3, 8027–8035. [Google Scholar] [CrossRef]
- Zhang, Y.; Jia, Y.; Li, M.; Hou, L. Influence of the 2-Methylimidazole/Zinc Nitrate Hexahydrate Molar Ratio on the Synthesis of Zeolitic Imidazolate Framework-8 Crystals at Room Temperature. Sci. Rep. 2018, 8, 9597. [Google Scholar] [CrossRef]
- Wang, D.; Yang, H.; Wang, Q.; Lu, Y.; Yan, J.; Cheng, W.; Rojas, O.J.; Han, G. Composite Membranes of Polyacrylonitrile Cross-Linked with Cellulose Nanocrystals for Emulsion Separation and Regeneration. Compos. Part A Appl. Sci. Manuf. 2023, 164, 107300. [Google Scholar] [CrossRef]
- Burmann, P.; Zornoza, B.; Téllez, C.; Coronas, J. Mixed Matrix Membranes Comprising MOFs and Porous Silicate Fillers Prepared via Spin Coating for Gas Separation. Chem. Eng. Sci. 2014, 107, 66–75. [Google Scholar] [CrossRef]
- Jomekian, A.; Behbahani, R.M.; Mohammadi, T.; Kargari, A. High Speed Spin Coating in Fabrication of Pebax 1657 Based Mixed Matrix Membrane Filled with Ultra-Porous ZIF-8 Particles for CO2/CH4 Separation. Korean J. Chem. Eng. 2017, 34, 440–453. [Google Scholar] [CrossRef]
- Muldoon, M.J.; Aki, S.N.V.K.; Anderson, J.L.; Dixon, J.K.; Brennecke, J.F. Improving Carbon Dioxide Solubility in Ionic Liquids. J. Phys. Chem. B 2007, 111, 9001–9009. [Google Scholar] [CrossRef]
- Frentzel-Beyme, L.; Kloß, M.; Pallach, R.; Salamon, S.; Moldenhauer, H.; Landers, J.; Wende, H.; Debus, J.; Henke, S. Porous Purple Glass–a Cobalt Imidazolate Glass with Accessible Porosity from a Meltable Cobalt Imidazolate Framework. J. Mater. Chem. A Mater. 2019, 7, 985–990. [Google Scholar] [CrossRef]
- Henke, S.; Wharmby, M.T.; Kieslich, G.; Hante, I.; Schneemann, A.; Wu, Y.; Daisenberger, D.; Cheetham, A.K. Pore Closure in Zeolitic Imidazolate Frameworks under Mechanical Pressure. Chem. Sci. 2018, 9, 1654–1660. [Google Scholar] [CrossRef]
- Feng, S.; Bu, M.; Pang, J.; Fan, W.; Fan, L.; Zhao, H.; Yang, G.; Guo, H.; Kong, G.; Sun, H. Hydrothermal Stable ZIF-67 Nanosheets via Morphology Regulation Strategy to Construct Mixed-Matrix Membrane for Gas Separation. J. Membr. Sci. 2020, 593, 117404. [Google Scholar] [CrossRef]
- Chen, K.; Ni, L.; Zhang, H.; Xie, J.; Yan, X.; Chen, S.; Qi, J.; Wang, C.; Sun, X.; Li, J. Veiled Metal Organic Frameworks Nanofillers for Mixed Matrix Membranes with Enhanced CO2/CH4 Separation Performance. Sep. Purif. Technol. 2021, 279, 119707. [Google Scholar] [CrossRef]
- Song, Q.; Nataraj, S.K.; Roussenova, M.V.; Tan, J.C.; Hughes, D.J.; Li, W.; Bourgoin, P.; Alam, M.A.; Cheetham, A.K.; Al-Muhtaseb, S.A. Zeolitic Imidazolate Framework (ZIF-8) Based Polymer Nanocomposite Membranes for Gas Separation. Energy Environ. Sci. 2012, 5, 8359–8369. [Google Scholar] [CrossRef]
- Robeson, L.M. The Upper Bound Revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of Separation Factor versus Permeability for Polymeric Membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Singh, A.; Koros, W.J. Significance of Entropic Selectivity for Advanced Gas Separation Membranes. Ind. Eng. Chem. Res. 1996, 35, 1231–1234. [Google Scholar] [CrossRef]
- Viciano-Chumillas, M.; Liu, X.; Leyva-Pérez, A.; Armentano, D.; Ferrando-Soria, J.; Pardo, E. Mixed Component Metal-Organic Frameworks: Heterogeneity and Complexity at the Service of Application Performances. Coord. Chem. Rev. 2022, 451, 214273. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Wang, Z.; Chaemchuen, S.; Koo-amornpattana, W.; Qiao, A.; Bu, T.; Verpoort, F.; Wang, J.; Mu, S. Insights into Multivariate Zeolitic Imidazolate Frameworks. Chem. Synth. 2025, 5, 31. [Google Scholar] [CrossRef]
- Kharul, U.K. PPO-ZIF MMMs Possessing Metal-Polymer Interactions for Propane/Propylene Separation. J. Membr. Sci. 2023, 668, 121208. [Google Scholar]
- Moghadam, F.; Lee, T.H.; Park, I.; Park, H.B. Thermally Annealed Polyimide-Based Mixed Matrix Membrane Containing ZIF-67 Decorated Porous Graphene Oxide Nanosheets with Enhanced Propylene/Propane Selectivity. J. Membr. Sci. 2020, 603, 118019. [Google Scholar] [CrossRef]
- Beckman, I.N.; Syrtsova, D.A.; Shalygin, M.G.; Kandasamy, P.; Teplyakov, V.V. Transmembrane Gas Transfer: Mathematics of Diffusion and Experimental Practice. J. Membr. Sci. 2020, 601, 117737. [Google Scholar] [CrossRef]
- Monteleone, M.; Fuoco, A.; Esposito, E.; Rose, I.; Chen, J.; Comesaña-Gándara, B.; Bezzu, C.G.; Carta, M.; McKeown, N.B.; Shalygin, M.G. Advanced Methods for Analysis of Mixed Gas Diffusion in Polymeric Membranes. J. Membr. Sci. 2022, 648, 120356. [Google Scholar] [CrossRef]
- Teplyakov, V.; Meares, P. Correlation Aspects of the Selective Gas Permeabilities of Polymeric Materials and Membranes. Gas Sep. Purif. 1990, 4, 66–74. [Google Scholar] [CrossRef]
- Nordin, N.; Ismail, A.F.; Mustafa, A.; Goh, P.S.; Rana, D.; Matsuura, T. Aqueous Room Temper-ature Synthesis of Zeolitic Imidazole Framework 8 (ZIF-8) with Various Concentrations of Tri-ethylamine. RSC Adv. 2014, 4, 33292–33300. [Google Scholar] [CrossRef]
- Xiong, S.; Jia, X.; Mi, K.; Wang, Y. Upgrading Polytetrafluoroethylene Hollow-Fiber Membranes by CFD-Optimized Atomic Layer Deposition. J. Membr. Sci. 2021, 617, 118610. [Google Scholar] [CrossRef]
- Vu, M.-T.; Lin, R.; Diao, H.; Zhu, Z.; Bhatia, S.K.; Smart, S. Effect of Ionic Liquids (ILs) on MOFs/Polymer Interfacial Enhancement in Mixed Matrix Membranes. J. Membr. Sci. 2019, 587, 117157. [Google Scholar] [CrossRef]






| C3H6 | C3H8 | |
|---|---|---|
| Molecular formula |  |  |
| Molecular weight (g mol−1) | 42.08 | 44.1 |
| Normal boiling point (°C) | −47.69 | −42.13 |
| Kinetic diameter (nm) | 0.45 | 0.43 |
| Polarizability × [10−25 cm3] | 62.6 | 62.9–63.7 |
| Dipole moment × [1018 esu−1∙cm−2] | 0.366 | 0.084 |
| Critical temperature (°C) | 91.75 | 96.74 |
| Critical pressure (Bar) | 45.55 | 42.51 |
| Vapour pressure (Bar at (°C)) | 9.17 (21.1) | 8.41 (21.1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hajivand, P.; Longo, M.; Monteleone, M.; Fuoco, A.; Esposito, E.; Mastropietro, T.F.; Navarro-Alapont, J.; Armentano, D.; Jansen, J.C. Sustainable Thin-Film Composite Mixed-Matrix Membranes Based on Cellulose Acetate, Bimetallic ZIF-8-67, and Ionic Liquid for Enhanced Propene/Propane Separation. Polymers 2026, 18, 396. https://doi.org/10.3390/polym18030396
Hajivand P, Longo M, Monteleone M, Fuoco A, Esposito E, Mastropietro TF, Navarro-Alapont J, Armentano D, Jansen JC. Sustainable Thin-Film Composite Mixed-Matrix Membranes Based on Cellulose Acetate, Bimetallic ZIF-8-67, and Ionic Liquid for Enhanced Propene/Propane Separation. Polymers. 2026; 18(3):396. https://doi.org/10.3390/polym18030396
Chicago/Turabian StyleHajivand, Pegah, Mariagiulia Longo, Marcello Monteleone, Alessio Fuoco, Elisa Esposito, Teresa Fina Mastropietro, Javier Navarro-Alapont, Donatella Armentano, and Johannes Carolus Jansen. 2026. "Sustainable Thin-Film Composite Mixed-Matrix Membranes Based on Cellulose Acetate, Bimetallic ZIF-8-67, and Ionic Liquid for Enhanced Propene/Propane Separation" Polymers 18, no. 3: 396. https://doi.org/10.3390/polym18030396
APA StyleHajivand, P., Longo, M., Monteleone, M., Fuoco, A., Esposito, E., Mastropietro, T. F., Navarro-Alapont, J., Armentano, D., & Jansen, J. C. (2026). Sustainable Thin-Film Composite Mixed-Matrix Membranes Based on Cellulose Acetate, Bimetallic ZIF-8-67, and Ionic Liquid for Enhanced Propene/Propane Separation. Polymers, 18(3), 396. https://doi.org/10.3390/polym18030396












