Hydrophobic Surface Treatment for the Protection of Carparo Stone
Abstract
1. Introduction
2. Materials and Methods
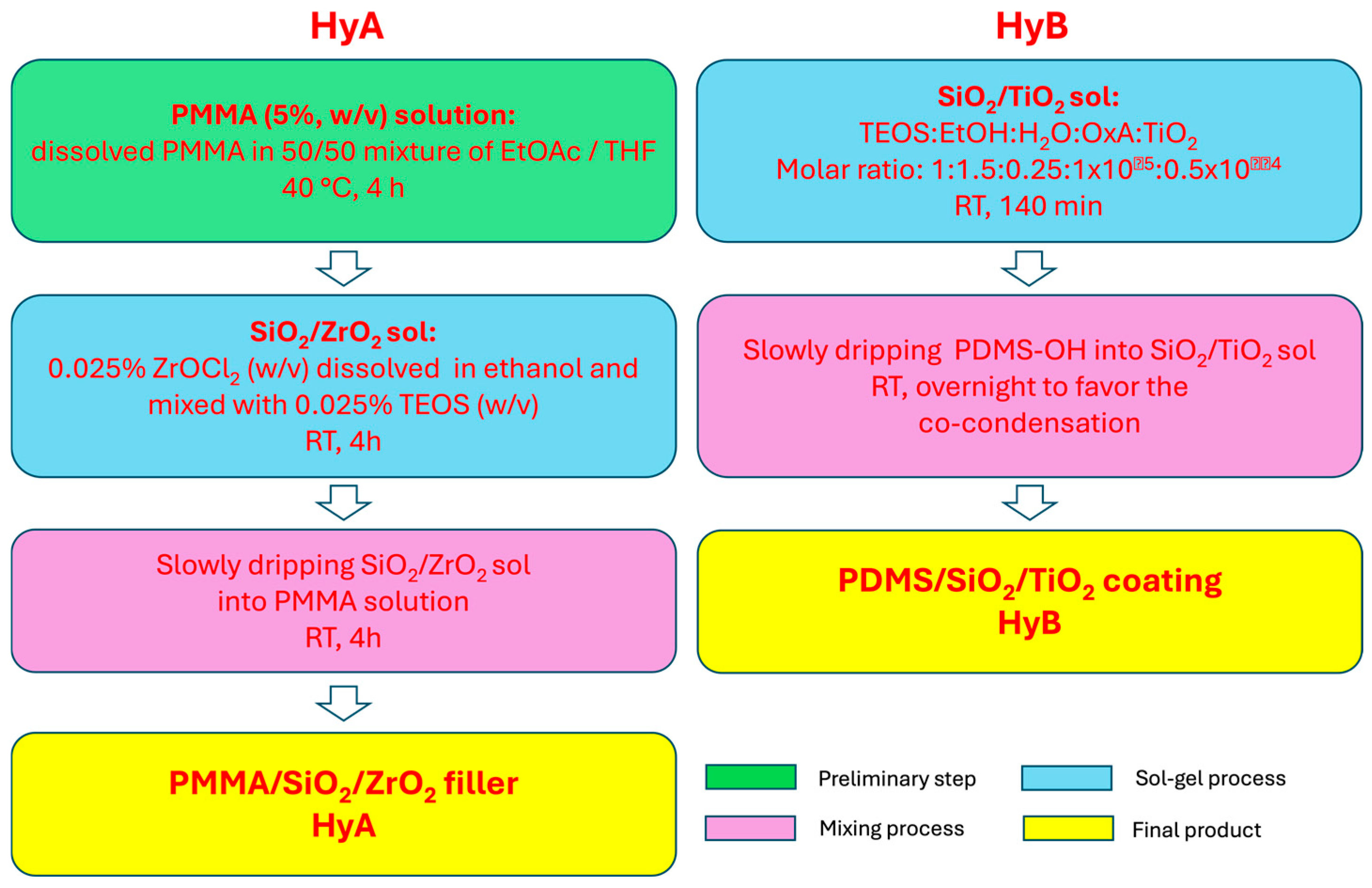
2.1. Synthesis of the Coatings
2.2. Carparo Sample Preparation
2.3. Characterizations
2.4. Artificial Aging
3. Results
3.1. Coating Characterization
3.1.1. Fourier Transform Infrared (FTIR) Spectroscopy
3.1.2. Thermal Analysis (TGA/DTG)
3.2. Characterization of the Stone Samples
3.2.1. Transmitted-Light Polarized Optical Microscopy
3.2.2. Raman and FTIR Spectroscopies
3.3. Characterization of Coated Stones and Effects of the Coatings
3.3.1. FTIR Spectroscopy
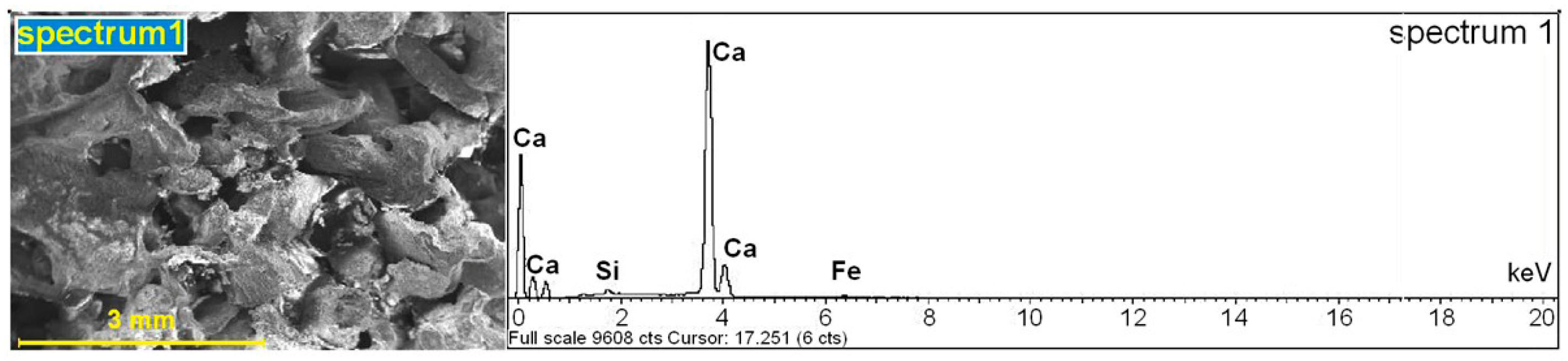
3.3.2. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS)
3.3.3. Microcomputed Tomography (Micro-CT)
3.3.4. Measurement of Color Surface
3.3.5. Determination of the Static Contact Angle
3.3.6. Water Absorption by Capillarity
3.3.7. Water Vapor Permeability
3.4. Artificial Aging
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De la Torre, M. Values and heritage conservation. Herit. Soc. 2013, 6, 155–166. [Google Scholar] [CrossRef]
- Artesani, A.; Di Turo, F.; Zucchelli, M.; Traviglia, A. Recent advances in protective coatings for cultural heritage–an overview. Coatings 2020, 10, 217. [Google Scholar] [CrossRef]
- Lettieri, M.; Masieri, M.; Aquaro, M.; Dilorenzo, D.; Frigione, M. Eco-Friendly Protective Coating to Extend the Life of Art-Works and Structures Made in Porous Stone Materials. Coatings 2021, 11, 1270. [Google Scholar] [CrossRef]
- Hosseini, M.; Karapanagiotis, I. (Eds.) Advanced Materials for the Conservation of Stone; Springer International Publishing: Cham, Switzerland, 2018. [Google Scholar]
- Raneri, S.; Barone, G.; Mazzoleni, P.; Alfieri, I.; Bergamonti, L.; De Kock, T.; Cnudde, V.; Lottici, P.P.; Lorenzi, A.; Predieri, G.; et al. Efficiency assessment of hybrid coatings for natural building stones: Advanced and multi-scale laboratory investigation. Constr. Build. Mater. 2018, 180, 412–424. [Google Scholar] [CrossRef]
- Ruffolo, S.A.; La Russa, M.F. Nanostructured coatings for stone protection: An overview. Front. Mater. 2019, 6, 147. [Google Scholar] [CrossRef]
- Yan, Y.; Wang, Y. A Review of Atmospheric Deterioration and Sustainable Conservation of Calcareous Stone in Historical Buildings and Monuments. Sustainability 2024, 16, 10751. [Google Scholar] [CrossRef]
- Benavente, D.; Cultrone, G.; Gómez-Heras, M. The combined influence of mineralogical, hygric and thermal properties on the durability of porous building stones. Eur. J. Mineral. 2008, 20, 673–685. [Google Scholar] [CrossRef]
- Salvini, S.; Coletti, C.; Maritan, L.; Massironi, M.; Pieropan, A.; Spiess, R.; Mazzoli, C. Petrographic characterization and durability of carbonate stones used in UNESCO World Heritage sites in northeastern Italy. Environ. Earth Sci. 2023, 82, 49. [Google Scholar] [CrossRef]
- Calvo, J.P.; Regueiro, M. Carbonate rocks in the Mediterranean region—From classical to innovative uses of building stone. Geol. Soc. Lond. Spéc. Publ. 2010, 331, 27–35. [Google Scholar] [CrossRef]
- Cioban, L.A.; Dochia, M.; Muresan, C.; Chambre, D.R. Weathering and deterioration of carbonate stones from historical monuments: A review. Sci. Tech. Bull. Ser. Chem. Food Sci. Eng. 2022, 19, 15–33. [Google Scholar]
- Benavente, D.; de Jongh, M.; Cañaveras, J.C. Weathering Processes and Mechanisms Caused by Capillary Waters and Pigeon Droppings on Porous Limestones. Minerals 2021, 11, 18. [Google Scholar] [CrossRef]
- Isola, D.; Bartoli, F.; Meloni, P.; Caneva, G.; Zucconi, L. Black Fungi and Stone Heritage Conservation: Ecological and Metabolic Assays for Evaluating Colonization Potential and Responses to Traditional Biocides. Appl. Sci. 2022, 12, 2038. [Google Scholar] [CrossRef]
- Cardell, C.; Benavente, D.; Rodríguez-Gordillo, J. Weathering of limestone building material by mixed sulfate solutions. Characterization of stone microstructure, reaction products and decay forms. Mater. Charact. 2008, 59, 1371–1385. [Google Scholar] [CrossRef]
- Eyssautier-Chuine, S.; Marin, B.; Thomachot-Schneider, C.; Fronteau, G.; Schneider, A.; Gibeaux, S.; Vazquez, P. Simulation of acid rain weathering effect on natural and artificial carbonate stones. Environ. Earth Sci. 2016, 75, 748. [Google Scholar] [CrossRef]
- Doehne, E.; Price, C.A. Stone Conservation—An Overview of Current Research, 2nd ed.; The Getty Conservation Institute: Los Angeles, CA, USA, 2010. [Google Scholar]
- Fassina, V. Environmental pollution in relation to stone decay. Durab. Build. Mater. 1988, 5, 317–358. [Google Scholar]
- Ruffolo, S.A.; La Russa, M.F.; Rovella, N.; Ricca, M. The Impact of Air Pollution on Stone Materials. Environments 2023, 10, 119. [Google Scholar] [CrossRef]
- Maravelaki-Kalaitzaki, P.; Kallithrakas-Kontos, N.; Agioutantis, Z.; Maurigiannakis, S.; Korakaki, D. A comparative study of porous limestones treated with silicon-based strengthening agents. Prog. Org. Coat. 2008, 62, 49–60. [Google Scholar] [CrossRef]
- Lettieri, M.; Masieri, M. Performances and coating morphology of a siloxane-based hydrophobic product applied in different concentrations on a highly porous stone. Coatings 2016, 6, 60. [Google Scholar] [CrossRef]
- Corcione, C.E.; De Simone, N.; Santarelli, M.L.; Frigione, M. Protective properties and durability characteristics of experimental and commercial organic coatings for the preservation of porous stone. Prog. Org. Coat. 2017, 103, 193–203. [Google Scholar] [CrossRef]
- Calò, G.; Di Pierro, M.; Federico, A.; Mongelli, G. Caratteri geologici petrografici mineralogici e meccanici dei carpari della provincia di Lecce. In Quarry and Construction; Ed. PEI: Parma, Italy, 1985. [Google Scholar]
- La Verde, G.; Raulo, A.; D’Avino, V.; Paternoster, G.; Roca, V.; La Commara, M.; Pugliese, M. Pietra leccese and other natural stones in puglia region: A new category of building materials for radiation protection? Int. J. Environ. Res. Public Health 2021, 18, 11213. [Google Scholar] [CrossRef] [PubMed]
- Congedo, P.M.; Baglivo, C.; D’Agostino, D.; Quarta, G.; Di Gloria, P. Rising Damp in Building Stones: Numerical and Experimental Comparison in Lecce Stone and Carparo under Controlled Microclimatic Conditions. Constr. Build. Mater. 2021, 296, 123713. [Google Scholar] [CrossRef]
- Folk, R.L. Practical petrographic classification of limestones. Bull. Am. Assoc. Petrol. Geol. 1959, 43, 1–38. [Google Scholar] [CrossRef]
- Calia, A.; Lettieri, M.; Mecchi, A.; Quarta, G. The role of the petrophysical characteristics on the durability and conservation of some porous calcarenites from Southern Italy. Geol. Soc. Spec. Publ. 2016, 416, 183–201. [Google Scholar] [CrossRef]
- Fico, D.; De Benedetto, G.E. Characterisation of surface finishes on ancient historical buildings in Salento (Southern Italy): An integrated analytical approach to establish the secrets of artisans. J. Cult. Herit. 2019, 35, 99–107. [Google Scholar] [CrossRef]
- Andriani, G.F.; Walsh, N. Petrophysical and mechanical properties of soft and porous building rocks used in Apulian monuments (south Italy). Geol. Soc. Lond. Spec. Publ. 2010, 333, 129–141. [Google Scholar] [CrossRef]
- Fico, D. Càrparu e Leccisu: Tecniche di Finitura e Deterioramento Degli Edifici Storici del Salento. Ph.D. Thesis, Università del Salento, Lecce, Italy, 2015. [Google Scholar]
- Licchelli, M.; Malagodi, M.; Weththimuni, M.L.; Zanchi, C. Water-repellent properties of fluoroelastomers on a very porous stone: Effect of the application procedure. Prog. Org. Coat. 2013, 76, 495–503. [Google Scholar] [CrossRef]
- Bergamonti, L.; Potenza, M.; Scigliuzzo, F.; Meli, S.; Casoli, A.; Lottici, P.P.; Graiff, C. Hydrophobic and Photocatalytic Treatment for the Conservation of Painted Lecce stone in Outdoor Conditions: A New Cleaning Approach. Appl. Sci. 2024, 14, 1261. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Q.; Liu, T.; Zhang, B. The preservation damage of hydrophobic polymer coating materials in conservation of stone relics. Prog. Org. Coat. 2013, 76, 1127–1134. [Google Scholar] [CrossRef]
- Sabatini, V.; Pargoletti, E.; Comite, V.; Ortenzi, M.A.; Fermo, P.; Gulotta, D.; Cappelletti, G. Towards Novel Fluorinated Methacrylic Coatings for Cultural Heritage: A Combined Polymers and Surfaces Chemistry Study. Polymers 2019, 11, 1190. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wei, W.; Li, Z.; Duan, W.; Han, H.; Xie, Q. A superhydrophobic fluorinated PDMS composite as a wearable strain sensor with excellent mechanical robustness and liquid impalement resistance. J. Mater. Chem. A 2020, 8, 3509–3516. [Google Scholar] [CrossRef]
- Petcu, C.; Alexandrescu, E.; Bălan, A.; Tănase, M.A.; Cinteză, L.O. Synthesis and characterisation of organo-modified silica nanostructured films for the water-repellent treatment of historic stone buildings. Coatings 2020, 10, 1010. [Google Scholar] [CrossRef]
- Henry, B.J.; Carlin, J.P.; Hammerschmidt, J.A.; Buck, R.C.; Buxton, L.W.; Fiedler, H.; Seed, J.; Hernandez, O. A critical review of the application of polymer of low concern and regulatory criteria to fluoropolymers. Integr. Environ. Assess. Manag. 2018, 14, 316–334. [Google Scholar] [CrossRef] [PubMed]
- Kapridaki, C.; Maravelaki-Kalaitzaki, P. TiO2–SiO2–PDMS nano-composite hydrophobic coating with self-cleaning properties for marble protection. Prog. Org. Coat. 2013, 76, 400–410. [Google Scholar] [CrossRef]
- Manoudis, P.N.; Karapanagiotis, I. Modification of the wettability of polymer surfaces using nanoparticles. Prog. Org. Coat. 2014, 77, 331–338. [Google Scholar] [CrossRef]
- Manoudis, P.N.; Tsakalof, A.; Karapanagiotis, I.; Zuburtikudis, I.; Panayiotou, C. Fabrication of super-hydrophobic surfaces for enhanced stone protection. Surf. Coat. Technol. 2009, 203, 1322–1328. [Google Scholar] [CrossRef]
- Karapanagiotis, I.; Pavlou, A.; Manoudis, P.N.; Aifantis, K.E. Water repellent ORMOSIL films for the protection of stone and other materials. Mater. Lett. 2014, 131, 276–279. [Google Scholar] [CrossRef]
- Mosquera, M.J.; de los Santos, D.M.; Rivas, T. Surfactant-synthesized ormosils with application to stone restoration. Langmuir 2010, 26, 6737–6745. [Google Scholar] [CrossRef]
- Li, D.; Xu, F.; Liu, Z.; Zhu, J.; Zhang, Q.; Shao, L. The effect of adding PDMS-OH and silica nanoparticles on sol–gel properties and effectiveness in stone protection. Appl. Surf. Sci. 2013, 266, 368–374. [Google Scholar] [CrossRef]
- Kapridaki, C.; Pinho, L.; Mosquera, M.J.; Maravelaki-Kalaitzaki, P. Producing photoactive, transparent and hydrophobic SiO2-crystalline TiO2 nanocomposites at ambient conditions with application as self-cleaning coatings. Appl. Catal. B 2014, 156, 416–427. [Google Scholar] [CrossRef]
- Zarzuela, R.; Carbú, M.; Gil, A.; Cantoral, J.; Mosquera, M.J. Incorporation of functionalized Ag-TiO2NPs to ormosil-based coatings as multifunctional biocide, superhydrophobic and photocatalytic surface treatments for porous ceramic materials. Surf. Interfaces 2021, 25, 101257. [Google Scholar] [CrossRef]
- Nguyen, T.V.; Nguyen, T.A.; Dao, P.H.; Mac, V.P.; Nguyen, A.H.; Do, M.T. Effect of rutile titania dioxide nanoparticles on the mechanical property, thermal stability, weathering resistance and antibacterial property of styrene acrylic polyurethane coating. Adv. Nat. Sci. Nanosci. Nanotechnol. 2016, 7, 045015. [Google Scholar] [CrossRef]
- Bergamonti, L.; Bondioli, F.; Alfieri, I.; Alinovi, S.; Lorenzi, A.; Predieri, G.; Lottici, P.P. Weathering resistance of PMMA/SiO2/ZrO2 hybrid coatings for sandstone conservation. Polym. Degrad. Stab. 2018, 147, 274–283. [Google Scholar] [CrossRef]
- Lacan, P.; Guizard, C.; Cot, L. Chemical and rheological investigations of the sol-gel transition in organically-modified siloxanes. J. Sol-Gel Sci. Technol. 1995, 4, 151–162. [Google Scholar] [CrossRef]
- UNI 10921:2001; Conservation of Cultural Property—Test Methods—Natural and Artificial Stone Materials—Hydrophobic Products. UNI Ente Nazionale Italiano di Unificazione: Milan, Italy, 2001.
- UNI EN 15886:2010; Conservation of Cultural Property—Test Methods—Color Measurement of Surfaces. UNI Ente Nazionale Italiano di Unificazione: Milan, Italy, 2010.
- UNI EN 15802: 2010; Conservation of Cultural Property—Test Methods—Determination of Static Contact Angle. UNI Ente Nazionale Italiano di Unificazione: Milan, Italy, 2010.
- UNI EN 15801:2010; Conservation of Cultural Property—Test Methods—Determination of Water Absorption by Capillarity. UNI Ente Nazionale Italiano di Unificazione: Milan, Italy, 2010.
- UNI EN 15803:2010; Conservation of Cultural Property—Test Methods—Determination of Water Vapour Permeability. UNI Ente Nazionale Italiano di Unificazione: Milan, Italy, 2010.
- Washburn, E.W. The dynamics of capillary flow. Phys. Rev. 1921, 17, 273–283. [Google Scholar] [CrossRef]
- Wang, H.; Xu, P.; Zhong, W.; Shen, L.; Du, Q. Transparent poly (methyl methacrylate)/silica/zirconia nanocomposites with excellent thermal stabilities. Polym. Degrad. Stab. 2005, 87, 319–327. [Google Scholar] [CrossRef]
- Wang, X.; Wu, L.; Li, J. Influence of nanozirconia on the thermal stability of poly (methyl methacrylate) prepared by in situ bulk polymerization. J. Appl. Polym. Sci. 2010, 117, 163–170. [Google Scholar] [CrossRef]
- Bergamonti, L.; Verza, E.; Magnani, R.; Michelini, E.; Ferretti, D.; Lottici, P.P.; Graiff, C. Protection of gypsum artifacts by Mg(OH)2 based super-hydrophobic nanocomposite. Constr. Build. Mater. 2025, 490, 142532. [Google Scholar] [CrossRef]
- Téllez, L.; Rubio, J.; Rubio, F.; Morales, E.; Oteo, J.L. FT-IR study of the hydrolysis and polymerization of tetraethyl orthosilicate and polydimethyl siloxane in the presence of tetrabutyl orthotitanate. Spectrosc. Lett. 2004, 37, 11–31. [Google Scholar] [CrossRef]
- Xu, F.; Liu, H.; Li, D. Synthesis of PDMS-SiO2 hybrids using different templates. J. Porous Mater. 2016, 23, 1133–1141. [Google Scholar] [CrossRef]
- Bellido-Aguilar, D.A.; Zheng, S.; Zhan, X.; Huang, Y.; Zhao, X.; Zeng, X.; Pallathadka, P.K.; Zhang, Q.; Chen, Z. Effect of a fluoroalkyl-functional curing agent on the wettability, thermal and mechanical properties of hydrophobic biobased epoxy coatings. Surf. Coat. Technol. 2019, 362, 274–281. [Google Scholar] [CrossRef]
- Schramm, C. High temperature ATR-FTIR characterization of the interaction of polycarboxylic acids and organotrialkoxysilanes with cellulosic material. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 243, 118815. [Google Scholar] [CrossRef]
- Wang, Y.T.; Chang, T.C.; Hong, Y.S.; Chen, H.B. Effect of the interfacial structure on the thermal stability of poly (methyl methacrylate)–silica hybrids. Thermochim. Acta 2003, 397, 219–226. [Google Scholar] [CrossRef]
- Criado, M.; Sobrados, I.; Sanz, J. Polymerization of hybrid organic–inorganic materials from several silicon compounds followed by TGA/DTA, FTIR and NMR techniques. Prog. Org. Coat. 2014, 77, 880–891. [Google Scholar] [CrossRef]
- Camino, G.; Lomakin, S.M.; Lazzari, M. Polydimethylsiloxane thermal degradation Part 1. Kinetic aspects. Polymer 2001, 42, 2395–2402. [Google Scholar] [CrossRef]
- Camino, G.; Lomakin, S.M.; Lageard, M. Thermal polydimethylsiloxane degradation. Part 2. The degradation mechanisms. Polymer 2002, 43, 2011–2015. [Google Scholar] [CrossRef]
- Wen, J.; Mark, J.E. Sol–gel preparation of composites of poly (dimethylsiloxane) with SiO2 and SiO2/TiO2, and their mechanical properties. Polym. J. 1995, 27, 492–502. [Google Scholar] [CrossRef]
- Karlina, L.; Azmiyawati, C.; Darmawan, A. Synthesis and characterization of hydrophobic silica prepared by different acid catalysts. IOP Conf. Ser. Mater. Sci. Eng. 2019, 509, 012065. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Anbalagan, G.; Pandi, S. Raman and infrared spectra of carbonates of calcite structure. J. Raman Spectrosc. 2006, 37, 892–899. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Anbalagan, G. Spectroscopic characterization of natural calcite minerals. Spectrochim. Acta Mol. Biomol. Spectrosc. 2007, 68, 656–664. [Google Scholar] [CrossRef]
- Bugani, S.; Camaiti, M.; Morselli, L.; Van de Casteele, E.; Janssens, K. Investigating morphological changes in treated vs. untreated stone building materials by x-ray micro-CT. Anal. Bioanal. Chem. 2008, 391, 1343–1350. [Google Scholar] [CrossRef]
- Callow, B.; Falcon-Suarez, I.; Marin-Moreno, H.; Bull, J.M.; Ahmed, S. Optimal X-ray micro-CT image based methods for porosity and permeability quantification in heterogeneous sandstones. Geophys. J. Int. 2020, 223, 1210–1229. [Google Scholar] [CrossRef]
- Toniolo, L.; Poli, T.; Castelvetro, V.; Manariti, A.; Chiantore, O.; Lazzari, M. Tailoring new fluorinated acrylic copolymers as protective coatings for marble. J. Cult. Herit. 2002, 3, 309–316. [Google Scholar] [CrossRef]
- Corcione, C.E.; Striani, R.; Frigione, M. Novel hydrophobic free-solvent UV-cured hybrid organic–inorganic methacrylic-based coatings for porous stones. Prog. Org. Coat. 2014, 77, 803–812. [Google Scholar] [CrossRef]
- García, O.; Malaga, K. Definition of the procedure to determine the suitability and durability of an anti-graffiti product for application on cultural heritage porous materials. J. Cult. Herit. 2012, 13, 77–82. [Google Scholar] [CrossRef]















| Analyses/Tests | Number of Samples | Dimensions (cm) | Measurements per Sample | Standard Rules |
|---|---|---|---|---|
| Petrographic analysis | 1 untreated | 3 × 1.5 × 0.003 | 1 | |
| SEM-EDS | 1 untreated 1 HyB 1 HyC | 1.5 × 1.5 × 0.5 | 1 | |
| Raman spectroscopy | 1 untreated | 1.5 × 1.5 × 1 | 5 | |
| FTIR spectroscopy | 1 untreated | powder | 5 | |
| X-ray micro-tomography | ** 1 HyA + BaCl2 | 1.5 × 1.5 × 1.5 | 1 | |
| * Colorimetric test | ** 5 HyB ** 5 HyC | 5 × 5 × 1 | 6 | UNI EN 15886:2010 [49] |
| * Static contact angle | ** 5 HyB ** 5 HyC | 5 × 5 × 1 | 6 | UNI EN 15802:2010 [50] |
| * Capillary water absorption | 5 untreated 5 HyB 5 HyC | 5 × 5 × 1 | 1 | UNI EN 15801:2010 [51] |
| * Water vapor permeability | 5 untreated 5 HyB 5 HyC | 5 × 5 × 1 | 1 | UNI EN 15803:2010 [52] |
| * Samples | Number of Samples | Measurements per Sample | Tests |
|---|---|---|---|
| HyB | 3 | 6 | Colorimetric measurements |
| HyC | 3 | 6 | |
| HyB | 3 | 6 | Static contact angle |
| HyC | 3 | 6 |
| Samples | ΔE* (Averaged from 6 Points per Sample Before and After Treatment) | Contact Angle (°) (Averaged from 6 Drops per Sample Before and After Treatment) | AC (kg/(m2s1/2)) (Averaged from 5 Samples) | WVTR (g/(m2d)) (Averaged from 5 Samples) | RVP (%) (Averaged from 5 Samples) |
|---|---|---|---|---|---|
| Untreated | - | Not measurable | 0.029 ± 0.001 | 130 ± 2 | - |
| HyB | 4.4 ± 1 | 140 ± 2 | 6.3 × 10−4 ± 2 × 10−5 | 76 ± 3 | 40 ± 2 |
| HyC | 3.7 ± 1 | 141 ± 2 | 1.0 × 10−3 ± 5 × 10−4 | 68 ± 2 | 48 ± 2 |
| Aged Samples | ||
|---|---|---|
| Samples | ΔE* Before and After Aging (Averaged from 6 Points per Sample) | Contact Angle (°) (Averaged from 6 Drops per Sample) |
| HyB | 2.1 ± 0.5 | 129 ± 2 |
| HyC | 1.5 ± 0.5 | 126 ± 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Potenza, M.; Verza, E.; Scigliuzzo, F.; Meli, S.; Casoli, A.; Lottici, P.P.; Graiff, C.; Bergamonti, L. Hydrophobic Surface Treatment for the Protection of Carparo Stone. Polymers 2026, 18, 237. https://doi.org/10.3390/polym18020237
Potenza M, Verza E, Scigliuzzo F, Meli S, Casoli A, Lottici PP, Graiff C, Bergamonti L. Hydrophobic Surface Treatment for the Protection of Carparo Stone. Polymers. 2026; 18(2):237. https://doi.org/10.3390/polym18020237
Chicago/Turabian StylePotenza, Marianna, Edoardo Verza, Federica Scigliuzzo, Sandro Meli, Antonella Casoli, Pier Paolo Lottici, Claudia Graiff, and Laura Bergamonti. 2026. "Hydrophobic Surface Treatment for the Protection of Carparo Stone" Polymers 18, no. 2: 237. https://doi.org/10.3390/polym18020237
APA StylePotenza, M., Verza, E., Scigliuzzo, F., Meli, S., Casoli, A., Lottici, P. P., Graiff, C., & Bergamonti, L. (2026). Hydrophobic Surface Treatment for the Protection of Carparo Stone. Polymers, 18(2), 237. https://doi.org/10.3390/polym18020237











