From Lignocellulosic Residues to Protein Sources: Insights into Biomass Pre-Treatments and Conversion
Abstract
1. Introduction
2. Lignocellulosic Biomass
2.1. Composition and Structure of Lignocellulosic Biomass
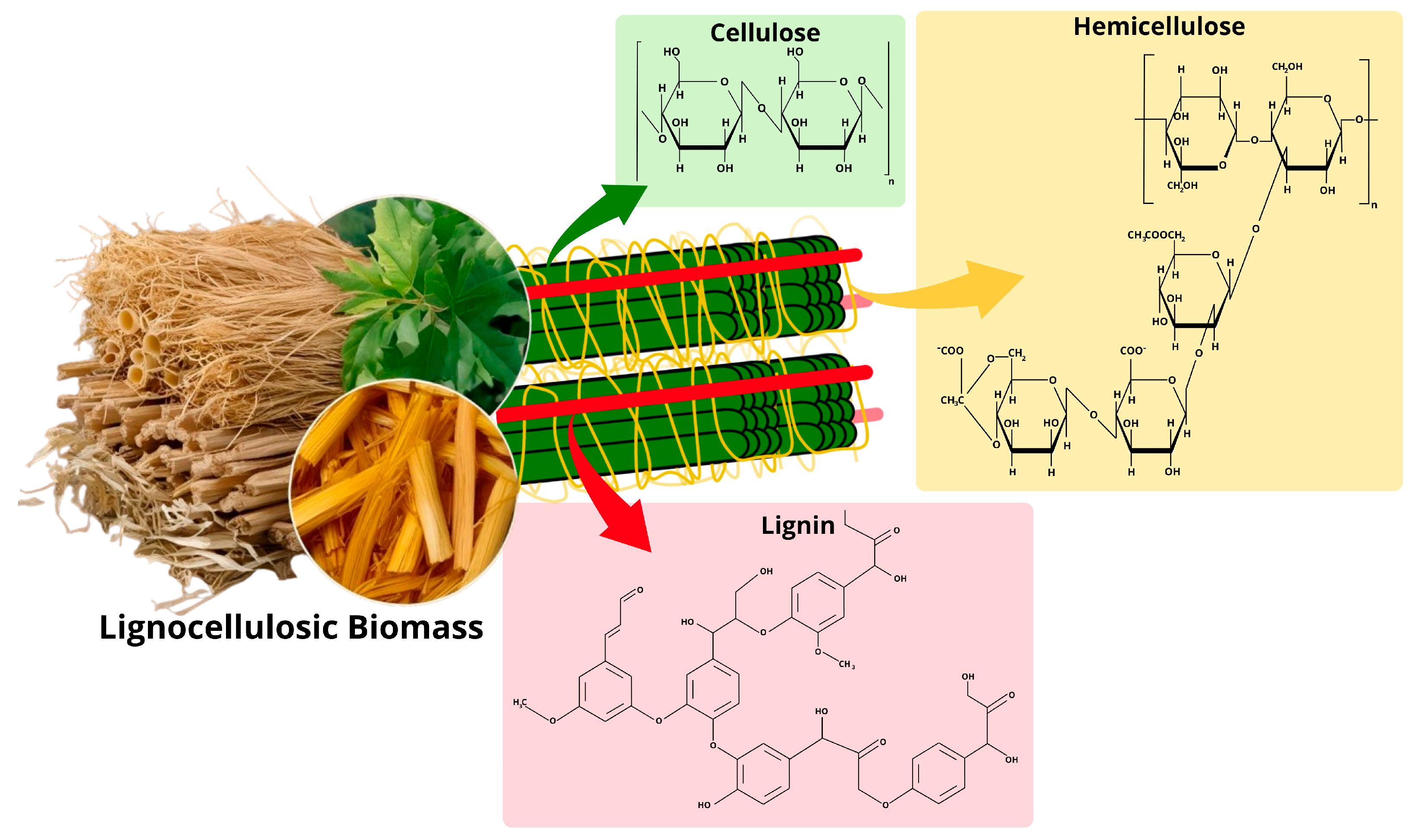
2.2. Bioconversion of Lignocellulosic Biomass
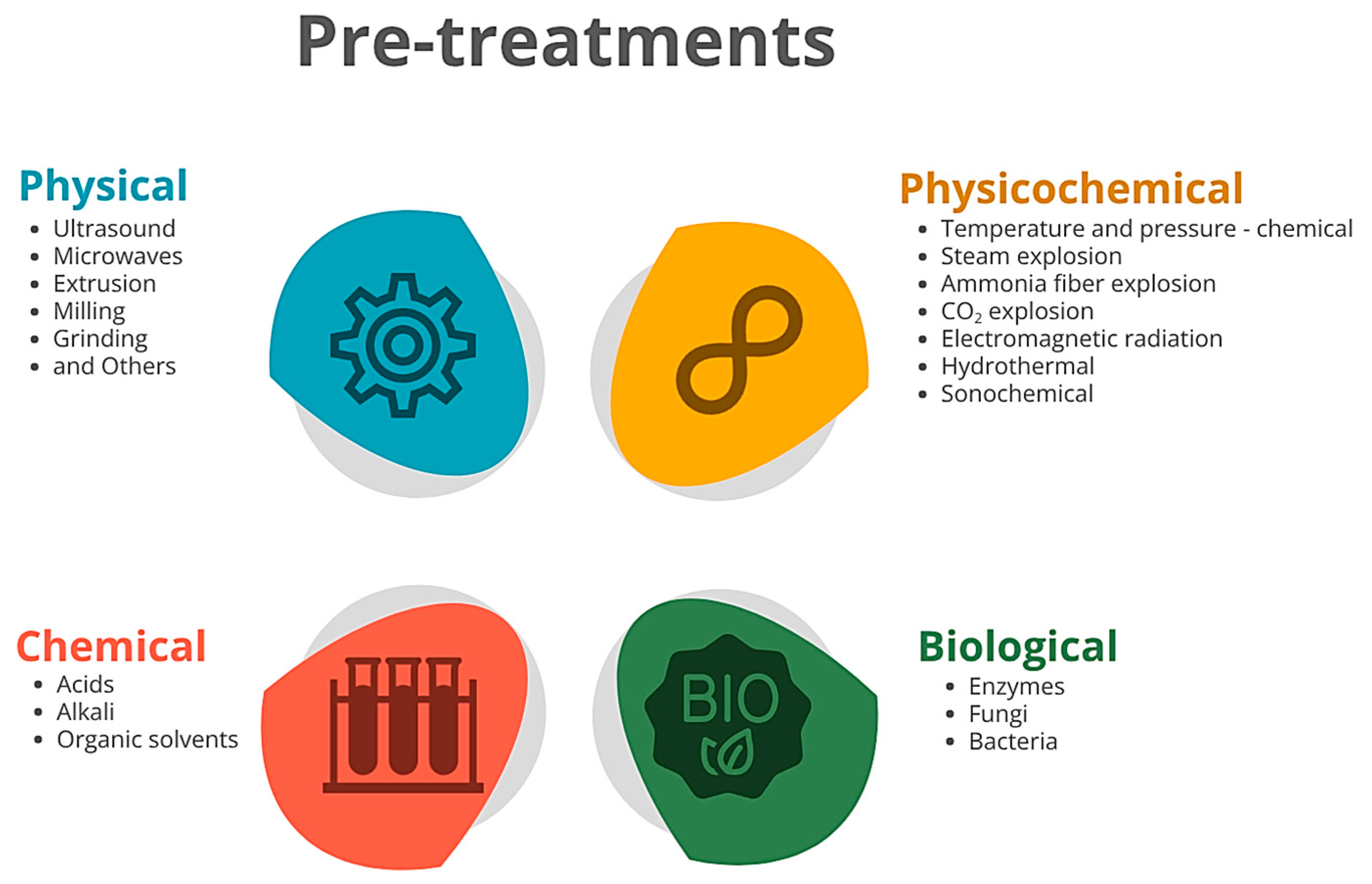
2.2.1. Lignocellulose Pre-Treatments
2.2.2. SCP Production Methods
2.3. Microorganisms Used in Single-Cell Protein Production

2.4. Nutritional Benefits of SCP
| Type of SCP | Protein Content (% Dry Weight) | Essential Amino Acids | Other Nutrients | Benefits | References |
|---|---|---|---|---|---|
| Fungal | 30–50% | High in lysine and threonine, complete essential amino acid profile (including methionine) | Fibres (chitin, β-glucans), unsaturated fatty acids, vitamins B9, B12, choline, minerals (Ca, Mg, Zn) | Reduces LDL cholesterol, increases satiety, lowers glycaemic response | Rajput et al. [86]; Finnigan et al. [107]; Wiedeman et al. [108]; Derbyshire & Delange [109]; Nyyssölä et al. [110]; Turnbull et al. [111] |
| Yeast | 40–55% | Contains all essential amino acids | B-complex vitamins, minerals, fibres, antioxidants | Antioxidant potential, functional food, food additive | Jach et al. [95]; Timira et al. [112]; Saravanan et al. [113] |
| Bacterial | 50–80% | Variable, rich in histidine, valine, and leucine | Carbohydrates, fibres, fats, high nucleic acid content (may limit direct human use in large quantities, but can be reduced by processing) | Grows on diverse substrates | Bratosin et al. [12]; Sharif et al. [16]; Kaur & Chavan [114]; Zha et al. [115]; Hadi & Brightwell [116] |
| Microalgae | 30–80% | Complete, species-dependent | Vitamins, minerals, fibres, low nucleic acid content | Immune support, cardiovascular and chronic disease prevention | Janssen et al. [117]; Montenegro Herrera et al. [118]; Grosshagaue et al. [119]; Koyande et al. [120]; Geada et al. [121] |
| Mixed | 40–70% | Balanced, depending on the combination | Combined nutrients from various sources | Optimised nutritional profile, cost-effective production | Wang et al. [122]; Rasouli et al. [123]; Hülsen et al. [124] |
3. Challenges and Future Directions in SCP Production
3.1. Challenges and Limitations
- Complexity of lignocellulosic biomass processing: The conversion of lignocellulosic biomass, a renewable and abundant feedstock, into fermentable substrates for SCP production involves multiple complex steps. These include size reduction and various physical, chemical, and biochemical pre-treatments aimed at breaking down plant cell wall components into fermentable sugars. Such processes often require specific conditions (e.g., pH, temperature and chemical concentrations) that differ from those used in traditional SCP production. Furthermore, these pre-treatments may generate inhibitory compounds that can negatively affect microbial fermentation, necessitating additional biomass conditioning steps [128].
- Nutrient limitations: Lignocellulosic substrates are deficient in nitrogen and other essential nutrients required for microbial growth, and therefore supplementation is often required [128].
- Enzymatic hydrolysis requirements: Even after pre-treatment, microorganisms are typically unable to directly metabolize complex polymers such as cellulose, hemicellulose, pectin and starch. These compounds must first be hydrolyzed into simpler sugars using enzymatic saccharification, requiring a suite of enzymes such as endo-glucanases, exo-glucanases, β-glucanases, and xylanases. These additional enzymatic steps significantly increase the cost of production when using agroforestry-derived biomass [128].
- Consumer acceptance: SCP products, particularly those intended for human consumption, often have undesirable flavors, odors, and colors. Additionally, obtaining pure SCP can be difficult due to residual lignocellulose particles that remain after fermentation. While purification techniques can be developed, they may increase the overall production costs [129].
- Safety concerns: Although most microorganisms used in SCP production are generally recognized as safe, contamination by pathogenic microbes can occur at various stages along the production and supply chain. This necessitates strict hygiene protocols and quality control standards to ensure product safety and protect public health. Moreover, the safety and sensory characteristics of SCP products depend on the microbial source. For instance, microalgae may pose a risk of toxin contamination; mycoproteins may trigger allergic reactions; and bacterial SCP requires careful strain selection to avoid pathogenic variants [116].
- Lack of industrial-scale validation: Current knowledge regarding the nutritional value of SCP is promising, but most evidence is based on laboratory-scale studies. Additional data from industrial-scale operations are needed to validate its safety, nutritional composition, and health impacts in real-world settings [130].
- Economic viability: The need for extensive pre-treatment, enzymatic hydrolysis, and purification steps increases the overall cost of SCP production, especially when using lignocellulosic biomass. These costs limit its competitiveness with conventional protein sources and represent a major barrier to commercial scalability [128,129].
3.2. Future Directions
- Safety and regulation: Advances in microbial engineering and synthetic biology, particularly the development of microbial cell factories, have the potential to enhance SCP’s cost-efficiency, nutritional properties, and functional versatility, further improving its market competitiveness [131]. Adding to this, factors such as affordability, cultural barriers and relevant legislation need to be considered. Regarding the principles for food and feed safety, and the way specific co- and by-products might be used, the key regulatory frameworks in the EU have been defined by the General Food Law Regulation (EC) No 178/2002 and the Regulation (EU) No 68/2013. Approval processes usually involve a comprehensive safety assessment of allergens, toxicity, nutritional composition, and contaminants. Given this, SCP approval is subject to several regulations depending on its intended use, as feed and food legislation is determinant in shaping the development path of alternative protein sources. Therefore, it is essential to comply with regulations to allow for a safe, sustainable and transparent development of the global protein supply chain [4,6,14].
- Industrial investment and slow progress: Several companies worldwide are already investing in SCP technologies, gradually expanding their applications and commercial viability. However, despite its potential to help meet global protein demand, progress in SCP production has been relatively slow over the past six decades [87].
- Need for standardization: SCP remains at an early stage of development, and there is a pressing need to establish standardized methodologies that ensure consistent yields, quality, and safety. Standardization efforts will require close collaboration among academic researchers, industry stakeholders, and funding agencies [79].
- Importance of commercial scaling: Commercial expansion scaling is essential not only to meet immediate protein supply demands but also to advance the broader goals of the circular economy.
- Environmental advantages of lignocellulosic SCP: SCP produced from lignocellulosic biomass offers clear environmental advantages, such as reducing greenhouse gas emissions and minimizing land use. Beyond the microbial conversion of cellulose and hemicellulose into fermentable sugars for SCP production, future research should emphasize integrated biorefinery strategies that valorise all polymeric fractions present in lignocellulosic biomass. This includes not only the saccharification of structural polysaccharides (cellulose and hemicellulose), but also the conversion of residual hemicellulose and lignin into high-value polymeric materials. For instance, hemicellulose can be used to produce functional oligosaccharides and biodegradable films [132], while lignin can serve as a renewable polyol source in the synthesis of bio-based polyurethanes [133]. These valorisation pathways contribute to a circular bioeconomy by maximizing resource efficiency and minimizing waste [134].
- Product development and sensory improvement: To fully realize these benefits, increased focus must be placed on the development of SCP-based products, improving their sensory appeal, and evaluating their long-term health effects.
- Contribution to food system sustainability: Ultimately, SCP has the potential to promote both physical and economic access to nutritious food, contributing significantly to the global transition toward sustainable food systems.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.-P.; Maubois, J.-L.; Beaufrère, B. Slow and Fast Dietary Proteins Differently Modulate Postprandial Protein Accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Q.; Qu, Y.; Gras, S.L.; Kentish, S.E. Separation Technologies for Whey Protein Fractionation. Food Eng. Rev. 2023, 15, 438–465. [Google Scholar] [CrossRef]
- Zeng, X.; Wang, Y.; Yang, S.; Liu, Y.; Li, X.; Liu, D. The Functionalities and Applications of Whey/Whey Protein in Fermented Foods: A Review. Food Sci. Biotechnol. 2024, 33, 769–790. [Google Scholar] [CrossRef]
- Nirmal, N.; Anyimadu, C.F.; Khanashyam, A.C.; Bekhit, A.E.A.; Dhar, B.K. Alternative Protein Sources: Addressing Global Food Security and Environmental Sustainability. Sustain. Dev. 2025, 33, 3958–3969. [Google Scholar] [CrossRef]
- Henchion, M.; Moloney, A.P.; Hyland, J.; Zimmermann, J.; McCarthy, S. Review: Trends for Meat, Milk and Egg Consumption for the next Decades and the Role Played by Livestock Systems in the Global Production of Proteins. Animal 2021, 15, 100287. [Google Scholar] [CrossRef]
- Salter, A.M.; Lopez-Viso, C. Role of Novel Protein Sources in Sustainably Meeting Future Global Requirements. Proc. Nutr. Soc. 2021, 80, 186–194. [Google Scholar] [CrossRef]
- Hayes, M. Challenges Regarding Protein Provision for the Growing Global Population: Improving the Environmental Impact of Traditional Protein Supply Chains and Maximising Use of Coproducts and Alternative, New Resources. Glob. Chall. 2023, 7, 2300059. [Google Scholar] [CrossRef]
- Talwar, R.; Freymond, M.; Beesabathuni, K.; Lingala, S. Current and Future Market Opportunities for Alternative Proteins in Low- and Middle-Income Countries. Curr. Dev. Nutr. 2024, 8, 102035. [Google Scholar] [CrossRef] [PubMed]
- Dunuweera, A.N.; Nikagolla, D.N.; Ranganathan, K. Fruit Waste Substrates to Produce Single-Cell Proteins as Alternative Human Food Supplements and Animal Feeds Using Baker’s Yeast (Saccharomyces cerevisiae). J. Food Qual. 2021, 2021, 1–6. [Google Scholar] [CrossRef]
- Spalvins, K.; Zihare, L.; Blumberga, D. Single Cell Protein Production from Waste Biomass: Comparison of Various Industrial by-Products. Energy Procedia 2018, 147, 409–418. [Google Scholar] [CrossRef]
- Tropea, A.; Ferracane, A.; Albergamo, A.; Potortì, A.G.; Lo Turco, V.; Di Bella, G. Single Cell Protein Production through Multi Food-Waste Substrate Fermentation. Fermentation 2022, 8, 91. [Google Scholar] [CrossRef]
- Bratosin, B.C.; Darjan, S.; Vodnar, D.C. Single Cell Protein: A Potential Substitute in Human and Animal Nutrition. Sustainability 2021, 13, 9284. [Google Scholar] [CrossRef]
- Mondal, A.K.; Sengupta, S.; Bhowal, J.; Bhattacharya, D.K. Utilization of fruit wastes in producing single cell protein. Int. J. Sci. Environ. Technol. 2012, 1, 430–438. [Google Scholar]
- Li, Y.P.; Ahmadi, F.; Kariman, K.; Lackner, M. Recent Advances and Challenges in Single Cell Protein (SCP) Technologies for Food and Feed Production. npj Sci. Food 2024, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, P. Single Cell Protein Production from Lignocellulosic Biomass; SpringerBriefs in Molecular Science; Springer: Singapore, 2017; ISBN 978-981-10-5872-1. [Google Scholar]
- Sharif, M.; Zafar, M.H.; Aqib, A.I.; Saeed, M.; Farag, M.R.; Alagawany, M. Single Cell Protein: Sources, Mechanism of Production, Nutritional Value and Its Uses in Aquaculture Nutrition. Aquaculture 2021, 531, 735885. [Google Scholar] [CrossRef]
- Ukaegbu-Obi, K.M. Single Cell Protein: A Resort to Global Protein Challenge and Waste Management. J. Microbiol. Microb. Technol. 2016, 1, 5. [Google Scholar] [CrossRef]
- Kumar, R.; Raj, T.; Næss, G.; Sørensen, M.; Dhawan, V. Opportunities and Challenges in Single-cell Protein Production Using Lignocellulosic Material. Biofuels Bioprod. Biorefin. 2024, 18, 310–321. [Google Scholar] [CrossRef]
- Alawad, I.; Ibrahim, H. Pretreatment of Agricultural Lignocellulosic Biomass for Fermentable Sugar: Opportunities, Challenges, and Future Trends. Biomass Convers. Biorefin. 2024, 14, 6155–6183. [Google Scholar] [CrossRef]
- Kumar, A.; Gautam, A.; Dutt, D. Biotechnological Transformation of Lignocellulosic Biomass in to Industrial Products: An Overview. Adv. Biosci. Biotechnol. 2016, 7, 149–168. [Google Scholar] [CrossRef]
- Segers, B.; Nimmegeers, P.; Spiller, M.; Tofani, G.; Jasiukaitytė-Grojzdek, E.; Dace, E.; Kikas, T.; Marchetti, J.M.; Rajić, M.; Yildiz, G.; et al. Lignocellulosic Biomass Valorisation: A Review of Feedstocks, Processes and Potential Value Chains and Their Implications for the Decision-Making Process. RSC Sustain. 2024, 2, 3730–3749. [Google Scholar] [CrossRef]
- Demain, A.L.; Newcomb, M.; Wu, J.H.D. Cellulase, Clostridia, and Ethanol. Microbiol. Mol. Biol. Rev. 2005, 69, 124–154. [Google Scholar] [CrossRef] [PubMed]
- Ashokkumar, V.; Venkatkarthick, R.; Jayashree, S.; Chuetor, S.; Dharmaraj, S.; Kumar, G.; Chen, W.-H.; Ngamcharussrivichai, C. Recent Advances in Lignocellulosic Biomass for Biofuels and Value-Added Bioproducts—A Critical Review. Bioresour. Technol. 2022, 344, 126195. [Google Scholar] [CrossRef]
- Paul, S.; Dutta, A. Challenges and Opportunities of Lignocellulosic Biomass for Anaerobic Digestion. Resour. Conserv. Recycl. 2018, 130, 164–174. [Google Scholar] [CrossRef]
- Yousuf, A.; Pirozzi, D.; Sannino, F. Fundamentals of Lignocellulosic Biomass. In Lignocellulosic Biomass to Liquid Biofuels; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–15. ISBN 978-0-12-815936-1. [Google Scholar]
- Frassoldati, A.; Ranzi, E. Modeling of Thermochemical Conversion of Biomasses. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 978-0-12-409547-2. [Google Scholar]
- Zabed, H.; Sahu, J.N.; Boyce, A.N.; Faruq, G. Fuel Ethanol Production from Lignocellulosic Biomass: An Overview on Feedstocks and Technological Approaches. Renew. Sustain. Energy Rev. 2016, 66, 751–774. [Google Scholar] [CrossRef]
- Machmudah, S.; Wahyudiono; Kanda, H.; Goto, M. Chapter 3—Hydrolysis of Biopolymers in Near-Critical and Subcritical Water. In Water Extraction of Bioactive Compounds; Dominguez González, H., González Muñoz, M.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 69–107. ISBN 978-0-12-809380-1. [Google Scholar]
- Khaire, K.C.; Moholkar, V.S.; Goyal, A. Bioconversion of Sugarcane Tops to Bioethanol and Other Value Added Products: An Overview. Mater. Sci. Energy Technol. 2021, 4, 54–68. [Google Scholar] [CrossRef]
- Abdel-Azeem, A.M.; Sheir, D.H. Bioconversion of Lignocellulosic Residues into Single-Cell Protein (SCP) by Chaetomium. In Recent Developments on Genus Chaetomium. Fungal Biology; Abdel-Azeem, A., Ed.; Springer: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Huang, C.; Li, R.; Tang, W.; Zheng, Y.; Meng, X. Improve Enzymatic Hydrolysis of Lignocellulosic Biomass by Modifying Lignin Structure via Sulfite Pretreatment and Using Lignin Blockers. Fermentation 2022, 8, 558. [Google Scholar] [CrossRef]
- Tian, S.-Q.; Zhao, R.-Y.; Chen, Z.-C. Review of the Pretreatment and Bioconversion of Lignocellulosic Biomass from Wheat Straw Materials. Renew. Sustain. Energy Rev. 2018, 91, 483–489. [Google Scholar] [CrossRef]
- Xu, N.; Liu, S.; Xin, F.; Zhou, J.; Jia, H.; Xu, J.; Jiang, M.; Dong, W. Biomethane Production From Lignocellulose: Biomass Recalcitrance and Its Impacts on Anaerobic Digestion. Front. Bioeng. Biotechnol. 2019, 7, 191. [Google Scholar] [CrossRef]
- Banu Jamaldheen, S.; Kurade, M.B.; Basak, B.; Yoo, C.G.; Oh, K.K.; Jeon, B.-H.; Kim, T.H. A Review on Physico-Chemical Delignification as a Pretreatment of Lignocellulosic Biomass for Enhanced Bioconversion. Bioresour. Technol. 2022, 346, 126591. [Google Scholar] [CrossRef]
- Arora, A.; Nandal, P.; Singh, J.; Verma, M.L. Nanobiotechnological Advancements in Lignocellulosic Biomass Pretreatment. Mater. Sci. Energy Technol. 2020, 3, 308–318. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, X.; Gao, L. New Strategy for the Biosynthesis of Alternative Feed Protein: Single-Cell Protein Production from Straw-Based Biomass. GCB Bioenergy 2024, 16, e13120. [Google Scholar] [CrossRef]
- Zhou, M.; Tian, X. Development of Different Pretreatments and Related Technologies for Efficient Biomass Conversion of Lignocellulose. Int. J. Biol. Macromol. 2022, 202, 256–268. [Google Scholar] [CrossRef]
- Baksi, S.; Saha, D.; Saha, S.; Sarkar, U.; Basu, D.; Kuniyal, J.C. Pre-Treatment of Lignocellulosic Biomass: Review of Various Physico-Chemical and Biological Methods Influencing the Extent of Biomass Depolymerization. Int. J. Environ. Sci. Technol. 2023, 20, 13895–13922. [Google Scholar] [CrossRef]
- Taylor, M.; Alabdrabalameer, H.; Skoulou, V. Choosing Physical, Physicochemical and Chemical Methods of Pre-Treating Lignocellulosic Wastes to Repurpose into Solid Fuels. Sustainability 2019, 11, 3604. [Google Scholar] [CrossRef]
- Haghighi Mood, S.; Hossein Golfeshan, A.; Tabatabaei, M.; Salehi Jouzani, G.; Najafi, G.H.; Gholami, M.; Ardjmand, M. Lignocellulosic Biomass to Bioethanol, a Comprehensive Review with a Focus on Pretreatment. Renew. Sustain. Energy Rev. 2013, 27, 77–93. [Google Scholar] [CrossRef]
- Hoang, A.T.; Nižetić, S.; Ong, H.C.; Mofijur, M.; Ahmed, S.F.; Ashok, B.; Bui, V.T.V.; Chau, M.Q. Insight into the Recent Advances of Microwave Pretreatment Technologies for the Conversion of Lignocellulosic Biomass into Sustainable Biofuel. Chemosphere 2021, 281, 130878. [Google Scholar] [CrossRef]
- Chen, W.-H.; Nižetić, S.; Sirohi, R.; Huang, Z.; Luque, R.; M.Papadopoulos, A.; Sakthivel, R.; Phuong Nguyen, X.; Tuan Hoang, A. Liquid Hot Water as Sustainable Biomass Pretreatment Technique for Bioenergy Production: A Review. Bioresour. Technol. 2022, 344, 126207. [Google Scholar] [CrossRef]
- Antczak, A.; Szadkowski, J.; Szadkowska, D.; Zawadzki, J. Assessment of the Effectiveness of Liquid Hot Water and Steam Explosion Pretreatments of Fast-Growing Poplar (Populus trichocarpa) Wood. Wood Sci. Technol. 2022, 56, 87–109. [Google Scholar] [CrossRef]
- Gundupalli, M.P.; Anne Sahithi, S.T.; Jayex, E.P.; Asavasanti, S.; Yasurin, P.; Cheng, Y.-S.; Sriariyanun, M. Combined Effect of Hot Water and Deep Eutectic Solvent (DES) Pretreatment on a Lignocellulosic Biomass Mixture for Improved Saccharification Efficiency. Bioresour. Technol. Rep. 2022, 17, 100986. [Google Scholar] [CrossRef]
- Mankar, A.R.; Pandey, A.; Modak, A.; Pant, K.K. Pretreatment of Lignocellulosic Biomass: A Review on Recent Advances. Bioresour. Technol. 2021, 334, 125235. [Google Scholar] [CrossRef]
- Sidana, A.; Yadav, S.K. Recent Developments in Lignocellulosic Biomass Pretreatment with a Focus on Eco-Friendly, Non-Conventional Methods. J. Clean. Prod. 2022, 335, 130286. [Google Scholar] [CrossRef]
- Ramirez Cabrera, P.A.; Lozano Pérez, A.S.; Guerrero Fajardo, C.A. Innovative Design of a Continuous Ultrasound Bath for Effective Lignocellulosic Biomass Pretreatment Based on a Theorical Method. Inventions 2024, 9, 105. [Google Scholar] [CrossRef]
- Calcio Gaudino, E.; Grillo, G.; Tabasso, S.; Stevanato, L.; Cravotto, G.; Marjamaa, K.; Pihlajaniemi, V.; Koivula, A.; Aro, N.; Uusitalo, J.; et al. Optimization of Ultrasound Pretreatment and Enzymatic Hydrolysis of Wheat Straw: From Lab to Semi-Industrial Scale. J. Clean. Prod. 2022, 380, 134897. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhao, J.; Xu, F.; Li, Y. Pretreatment of Lignocellulosic Biomass for Enhanced Biogas Production. Prog. Energy Combust. Sci. 2014, 42, 35–53. [Google Scholar] [CrossRef]
- Rodriguez, C.; Alaswad, A.; Benyounis, K.Y.; Olabi, A.G. Pretreatment Techniques Used in Biogas Production from Grass. Renew. Sustain. Energy Rev. 2017, 68, 1193–1204. [Google Scholar] [CrossRef]
- Singh, S.K. Biological Treatment of Plant Biomass and Factors Affecting Bioactivity. J. Clean. Prod. 2021, 279, 123546. [Google Scholar] [CrossRef]
- Saratale, R.G.; Ponnusamy, V.K.; Piechota, G.; Igliński, B.; Shobana, S.; Park, J.-H.; Saratale, G.D.; Shin, H.S.; Banu, J.R.; Kumar, V.; et al. Green Chemical and Hybrid Enzymatic Pretreatments for Lignocellulosic Biorefineries: Mechanism and Challenges. Bioresour. Technol. 2023, 387, 129560. [Google Scholar] [CrossRef]
- Tamilselvan, R.; Immanuel Selwynraj, A. Enhancing Biogas Generation from Lignocellulosic Biomass through Biological Pretreatment: Exploring the Role of Ruminant Microbes and Anaerobic Fungi. Anaerobe 2024, 85, 102815. [Google Scholar] [CrossRef]
- Bhushan, S.; Jayakrishnan, U.; Shree, B.; Bhatt, P.; Eshkabilov, S.; Simsek, H. Biological Pretreatment for Algal Biomass Feedstock for Biofuel Production. J. Environ. Chem. Eng. 2023, 11, 109870. [Google Scholar] [CrossRef]
- Abbas, M.; Ejaz, U.; Shafique, M.; Naz, S.A.; Sohail, M. Biological Pretreatment of Sugarcane Bagasse for the Production of Fungal Laccase and Bacterial Cellulase. J. Basic Microbiol. 2023, 63, 722–733. [Google Scholar] [CrossRef]
- Meenakshisundaram, S.; Fayeulle, A.; Léonard, E.; Ceballos, C.; Liu, X.; Pauss, A. Combined Biological and Chemical/Physicochemical Pretreatment Methods of Lignocellulosic Biomass for Bioethanol and Biomethane Energy Production—A Review. Appl. Microbiol. 2022, 2, 716–734. [Google Scholar] [CrossRef]
- Nargotra, P.; Sharma, V.; Lee, Y.-C.; Tsai, Y.-H.; Liu, Y.-C.; Shieh, C.-J.; Tsai, M.-L.; Dong, C.-D.; Kuo, C.-H. Microbial Lignocellulolytic Enzymes for the Effective Valorization of Lignocellulosic Biomass: A Review. Catalysts 2023, 13, 83. [Google Scholar] [CrossRef]
- Balla, A.; Silini, A.; Cherif-Silini, H.; Bouket, A.C.; Boudechicha, A.; Luptakova, L.; Alenezi, F.N.; Belbahri, L. Screening of Cellulolytic Bacteria from Various Ecosystems and Their Cellulases Production under Multi-Stress Conditions. Catalysts 2022, 12, 769. [Google Scholar] [CrossRef]
- Tang, H.; Li, Y.-Q.; Zheng, L.; Wang, M.-J.; Luo, C.-B. Efficient Saccharification of Bamboo Biomass by Secretome Protein of the Cellulolytic Bacterium Serratia Marcescens LY1 Based on Whole-Genome and Secretome Analysis. Renew. Energy 2022, 193, 32–40. [Google Scholar] [CrossRef]
- Sethupathy, S.; Morales, G.M.; Li, Y.; Wang, Y.; Jiang, J.; Sun, J.; Zhu, D. Harnessing Microbial Wealth for Lignocellulose Biomass Valorization through Secretomics: A Review. Biotechnol. Biofuels 2021, 14, 154. [Google Scholar] [CrossRef]
- Cheng, H.-H.; Whang, L.-M. Resource Recovery from Lignocellulosic Wastes via Biological Technologies: Advancements and Prospects. Bioresour. Technol. 2022, 343, 126097. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Saini, S.; Sharma, K.K. Fungal-Integrated Second-Generation Lignocellulosic Biorefinery: Utilization of Agricultural Biomass for Co-Production of Lignocellulolytic Enzymes, Mushroom, Fungal Polysaccharides, and Bioethanol. Biomass Convers. Biorefin. 2024, 14, 1117–1131. [Google Scholar] [CrossRef]
- Mohammad Rahmani, A.; Gahlot, P.; Moustakas, K.; Kazmi, A.A.; Shekhar Prasad Ojha, C.; Tyagi, V.K. Pretreatment Methods to Enhance Solubilization and Anaerobic Biodegradability of Lignocellulosic Biomass (Wheat Straw): Progress and Challenges. Fuel 2022, 319, 123726. [Google Scholar] [CrossRef]
- Suryadi, H.; Judono, J.J.; Putri, M.R.; Eclessia, A.D.; Ulhaq, J.M.; Agustina, D.N.; Sumiati, T. Biodelignification of Lignocellulose Using Ligninolytic Enzymes from White-Rot Fungi. Heliyon 2022, 8, e08865. [Google Scholar] [CrossRef]
- Li, X.; Shi, Y.; Kong, W.; Wei, J.; Song, W.; Wang, S. Improving Enzymatic Hydrolysis of Lignocellulosic Biomass by Bio-Coordinated Physicochemical Pretreatment—A Review. Energy Rep. 2022, 8, 696–709. [Google Scholar] [CrossRef]
- Sajid, S.; Kudakwashe Zveushe, O.; Resco de Dios, V.; Nabi, F.; Lee, Y.K.; Kaleri, A.R.; Ma, L.; Zhou, L.; Zhang, W.; Dong, F.; et al. Pretreatment of Rice Straw by Newly Isolated Fungal Consortium Enhanced Lignocellulose Degradation and Humification during Composting. Bioresour. Technol. 2022, 354, 127150. [Google Scholar] [CrossRef]
- Suthar, S.; Kishore Singh, N. Fungal Pretreatment Facilitates the Rapid and Valuable Composting of Waste Cardboard. Bioresour. Technol. 2022, 344, 126178. [Google Scholar] [CrossRef]
- Kuhad, R.C.; Rapoport, A.; Kumar, V.; Singh, D.; Kumar, V.; Tiwari, S.K.; Ahlawat, S.; Singh, B. Biological Pretreatment of Lignocellulosic Biomass: An Environment-Benign and Sustainable Approach for Conversion of Solid Waste into Value-Added Products. Crit. Rev. Environ. Sci. Technol. 2024, 54, 771–796. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Jagtap, S.S.; Bedekar, A.A.; Bhatia, R.K.; Patel, A.K.; Pant, D.; Rajesh Banu, J.; Rao, C.V.; Kim, Y.-G.; Yang, Y.-H. Recent Developments in Pretreatment Technologies on Lignocellulosic Biomass: Effect of Key Parameters, Technological Improvements, and Challenges. Bioresour. Technol. 2020, 300, 122724. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, B.; Sarker, S.; Lamb, J.J.; Lien, K.M. Yield Improvements in Anaerobic Digestion of Lignocellulosic Feedstocks. J. Clean. Prod. 2021, 288, 125447. [Google Scholar] [CrossRef]
- Joshi, M.; Manjare, S. Chemical Approaches for the Biomass Valorisation: A Comprehensive Review of Pretreatment Strategies. Environ. Sci. Pollut. Res. 2024, 31, 48928–48954. [Google Scholar] [CrossRef]
- Paudel, P.P.; Kafle, S.; Park, S.; Kim, S.J.; Cho, L.; Kim, D.H. Advancements in Sustainable Thermochemical Conversion of Agricultural Crop Residues: A Systematic Review of Technical Progress, Applications, Perspectives, and Challenges. Renew. Sustain. Energy Rev. 2024, 202, 114723. [Google Scholar] [CrossRef]
- Kululo, W.W.; Habtu, N.G.; Abera, M.K.; Sendekie, Z.B.; Fanta, S.W.; Yemata, T.A. Advances in Various Pretreatment Strategies of Lignocellulosic Substrates for the Production of Bioethanol: A Comprehensive Review. Discov. Appl. Sci. 2025, 7, 476. [Google Scholar] [CrossRef]
- Prasad, B.R.; Padhi, R.K.; Ghosh, G. A Review on Key Pretreatment Approaches for Lignocellulosic Biomass to Produce Biofuel and Value-Added Products. Int. J. Environ. Sci. Technol. 2023, 20, 6929–6944. [Google Scholar] [CrossRef]
- Gervasi, T.; Mandalari, G. Valorization of Agro-Industrial Orange Peel By-Products through Fermentation Strategies. Fermentation 2024, 10, 224. [Google Scholar] [CrossRef]
- Reihani, S.F.S.; Khosravi-Darani, K. Influencing Factors on Single-Cell Protein Production by Submerged Fermentation: A Review. Electron. J. Biotechnol. 2019, 37, 34–40. [Google Scholar] [CrossRef]
- Raziq, A.; Lateef, M.; Ullah, A.; Ullah, H.; Khan, M.W. Single Cell Protein (SCP) Production and Potential Substrates: A Comprehensive Review. Pure Appl. Biol. 2020, 9, 1743–1754. [Google Scholar] [CrossRef]
- Kieliszek, M.; Kot, A.M.; Bzducha-Wróbel, A.; BŁażejak, S.; Gientka, I.; Kurcz, A. Biotechnological Use of Candida Yeasts in the Food Industry: A Review. Fungal Biol. Rev. 2017, 31, 185–198. [Google Scholar] [CrossRef]
- Thiviya, P.; Gamage, A.; Kapilan, R.; Merah, O.; Madhujith, T. Single Cell Protein Production Using Different Fruit Waste: A Review. Separations 2022, 9, 178. [Google Scholar] [CrossRef]
- Ribeiro, G.O.; Rodrigues, L.d.A.P.; dos Santos, T.B.S.; Alves, J.P.S.; Oliveira, R.S.; Nery, T.B.R.; Barbosa, J.D.V.; Soares, M.B.P. Innovations and Developments in Single Cell Protein: Bibliometric Review and Patents Analysis. Front. Microbiol. 2023, 13, 1093464. [Google Scholar] [CrossRef] [PubMed]
- Adedayo, M.; Ajiboye, E.; Akintunde, J.; Odaibo, A. Single Cell Proteins: As Nutritional Enhancer. Adv. Appl. Sci. Res. 2011, 2, 396–409. [Google Scholar]
- Musoni, M.; Destain, J.; Thonart, P.; Bahama, J.-B.; Delvigne, F. Bioreactor Design and Implementation Strategies for the Cultivation of Filamentous Fungi and the Production of Fungal Metabolites: From Traditional Methods to Engineered Systems. Biotechnol. Agron. Soc. Environ. 2015, 19, 430–442. [Google Scholar]
- Amara, A.A.; El-Baky, N.A. Fungi as a Source of Edible Proteins and Animal Feed. JoF 2023, 9, 73. [Google Scholar] [CrossRef] [PubMed]
- Verduzco-Oliva, R.; Gutierrez-Uribe, J.A. Beyond Enzyme Production: Solid State Fermentation (SSF) as an Alternative Approach to Produce Antioxidant Polysaccharides. Sustainability 2020, 12, 495. [Google Scholar] [CrossRef]
- Wu, J.; Hu, J.; Zhao, S.; He, M.; Hu, G.; Ge, X.; Peng, N. Single-Cell Protein and Xylitol Production by a Novel Yeast Strain Candida Intermedia FL023 from Lignocellulosic Hydrolysates and Xylose. Appl. Biochem. Biotechnol. 2018, 185, 163–178. [Google Scholar] [CrossRef]
- Rajput, S.D.; Pandey, N.; Sahu, K. A Comprehensive Report on Valorization of Waste to Single Cell Protein: Strategies, Challenges, and Future Prospects. Environ. Sci. Pollut. Res. 2024, 31, 26378–26414. [Google Scholar] [CrossRef]
- Onyeaka, H.; Anumudu, C.K.; Okpe, C.; Okafor, A.; Ihenetu, F.; Miri, T.; Odeyemi, O.A.; Anyogu, A. Single Cell Protein for Foods and Feeds: A Review of Trends. Open Microbiol. J. 2022, 16, e187428582206160. [Google Scholar] [CrossRef]
- Ahmed, M.G.; Gouda, S.A.; Donia, S.; Hassanein, N.M. Production of Single Cell Protein by Fungi from Different Food Wastes. Biomass Convers. Biorefin. 2025, 15, 5447–5462. [Google Scholar] [CrossRef]
- Oshoma, C.; Eguakun-Owie, S. Conversion of Food Waste to Single Cell Protein Using Aspergillus niger. J. Appl. Sci. Environ. Manag. 2018, 22, 350. [Google Scholar] [CrossRef]
- Edelman, J.; Fewell, A.; Solomons, G. Myco-Protein-a New Food. Nutr. Abstr. Rev. Ser. A Hum. Exp. 1983, 53, 471–480. [Google Scholar]
- Nasseri, A.T.; Rasoul-Amini, S.; Morowvat, M.H.; Ghasemi, Y. Single Cell Protein: Production and Process. Am. J. Food Technol. 2011, 6, 103–116. [Google Scholar] [CrossRef]
- Ritala, A.; Häkkinen, S.T.; Toivari, M.; Wiebe, M.G. Single Cell Protein—State-of-the-Art, Industrial Landscape and Patents 2001–2016. Front. Microbiol. 2017, 8, 2009. [Google Scholar] [CrossRef] [PubMed]
- Tamang, J.P.; Watanabe, K.; Holzapfel, W.H. Review: Diversity of Microorganisms in Global Fermented Foods and Beverages. Front. Microbiol. 2016, 7, 377. [Google Scholar] [CrossRef]
- Niego, A.G.; Rapior, S.; Thongklang, N.; Raspé, O.; Jaidee, W.; Lumyong, S.; Hyde, K.D. Macrofungi as a Nutraceutical Source: Promising Bioactive Compounds and Market Value. J. Fungi 2021, 7, 397. [Google Scholar] [CrossRef]
- Jach, M.E.; Serefko, A.; Ziaja, M.; Kieliszek, M. Yeast Protein as an Easily Accessible Food Source. Metabolites 2022, 12, 63. [Google Scholar] [CrossRef]
- Zheng, S.; Yang, M.; Yang, Z. Biomass Production of Yeast Isolate from Salad Oil Manufacturing Wastewater. Bioresour. Technol. 2005, 96, 1183–1187. [Google Scholar] [CrossRef]
- Thiviya, P.; Gamage, A.; Kapilan, R.; Merah, O.; Madhujith, T. Production of Single-Cell Protein from Fruit Peel Wastes Using Palmyrah Toddy Yeast. Fermentation 2022, 8, 355. [Google Scholar] [CrossRef]
- Devillers, H.; Brunel, F.; Połomska, X.; Sarilar, V.; Lazar, Z.; Robak, M.; Neuvéglise, C. Draft Genome Sequence of Yarrowia Lipolytica Strain A-101 Isolated from Polluted Soil in Poland. Genome Announc. 2016, 4, e01094-16. [Google Scholar] [CrossRef] [PubMed]
- Drzymała, K.; Mirończuk, A.M.; Pietrzak, W.; Dobrowolski, A. Rye and Oat Agricultural Wastes as Substrate Candidates for Biomass Production of the Non-Conventional Yeast Yarrowia Lipolytica. Sustainability 2020, 12, 7704. [Google Scholar] [CrossRef]
- Dobrowolski, A.; Drzymała, K.; Mituła, P.; Mirończuk, A.M. Production of Tailor-Made Fatty Acids from Crude Glycerol at Low pH by Yarrowia lipolytica. Bioresour. Technol. 2020, 314, 123746. [Google Scholar] [CrossRef]
- Lapeña, D.; Olsen, P.M.; Arntzen, M.Ø.; Kosa, G.; Passoth, V.; Eijsink, V.G.H.; Horn, S.J. Spruce Sugars and Poultry Hydrolysate as Growth Medium in Repeated Fed-Batch Fermentation Processes for Production of Yeast Biomass. Bioprocess. Biosyst. Eng. 2020, 43, 723–736. [Google Scholar] [CrossRef]
- Sharma, S.; Hansen, L.D.; Hansen, J.Ø.; Mydland, L.T.; Horn, S.J.; Øverland, M.; Eijsink, V.G.H.; Vuoristo, K.S. Microbial Protein Produced from Brown Seaweed and Spruce Wood as a Feed Ingredient. J. Agric. Food Chem. 2018, 66, 8328–8335. [Google Scholar] [CrossRef]
- Diwan, B.; Gupta, P. Conversion of Rice Straw to Caprylic Acid-Rich Microbial Oils by Oleaginous Yeast Isolates. Biomass Convers. Biorefin. 2022, 12, 5901–5914. [Google Scholar] [CrossRef]
- Yang, F.; Jin, Z.; Nawaz, M.; Xiao, Y.; Jiang, Y.; Hu, J.; Li, J.; Gao, M.-T. Oligosaccharides in Straw Hydrolysate Could Improve the Production of Single-Cell Protein with Saccharomyces cerevisiae. J. Sci. Food Agric. 2022, 102, 2928–2936. [Google Scholar] [CrossRef]
- Bonan, C.I.D.G.; Tramontina, R.; dos Santos, M.W.; Biazi, L.E.; Soares, L.B.; Pereira, I.O.; Hoffmam, Z.B.; Coutouné, N.; Squina, F.M.; Robl, D.; et al. Biorefinery Platform for Spathaspora Passalidarum NRRL Y-27907 in the Production of Ethanol, Xylitol, and Single Cell Protein from Sugarcane Bagasse. Bioenergy Res. 2022, 15, 1169–1181. [Google Scholar] [CrossRef]
- Pereira, A.G.; Fraga-Corral, M.; Garcia-Oliveira, P.; Otero, P.; Soria-Lopez, A.; Cassani, L.; Cao, H.; Xiao, J.; Prieto, M.A.; Simal-Gandara, J. Single-Cell Proteins Obtained by Circular Economy Intended as a Feed Ingredient in Aquaculture. Foods 2022, 11, 2831. [Google Scholar] [CrossRef]
- Finnigan, T.; Needham, L.; Abbott, C. Mycoprotein: A Healthy New Protein with a Low Environmental Impact. In Sustainable Protein Sources; Nadathur, S.R., Wanasundara, J.P.D., Scanlin, L., Eds.; Academic Press: San Diego, CA, USA, 2017; pp. 305–325. ISBN 978-0-12-802778-3. [Google Scholar]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef]
- Derbyshire, E.J.; Delange, J. Fungal Protein—What Is It and What Is the Health Evidence? A Systematic Review Focusing on Mycoprotein. Front. Sustain. Food Syst. 2021, 5, 581682. [Google Scholar] [CrossRef]
- Nyyssölä, A.; Suhonen, A.; Ritala, A.; Oksman-Caldentey, K.-M. The Role of Single Cell Protein in Cellular Agriculture. Curr. Opin. Biotechnol. 2022, 75, 102686. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, W.; Leeds, A.; Edwards, D. Mycoprotein Reduces Blood Lipids in Free-Living Subjects. Am. J. Clin. Nutr. 1992, 55, 415–419. [Google Scholar] [CrossRef]
- Timira, V.; Chen, X.; Zhou, P.; Wu, J.; Wang, T. Potential Use of Yeast Protein in Terms of Biorefinery, Functionality, and Sustainability in Food Industry. Comp. Rev. Food Sci. Food Safe 2024, 23, e13326. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, K.; Rajendran, M.; Kathirvel, P. Utilization of Water Hyacinth (Eichhornia crassipes) as a Feasible Substrate for the Production of Single Cell Protein Using Saccharomyces cerevisiae (Baker’s Yeast). Waste Biomass Valorization 2023, 14, 4231–4242. [Google Scholar] [CrossRef]
- Kaur, N.; Chavan, A. Production of Single-Cell Protein from Fruit and Vegetable Waste Using an Isolated Strain of Lactobacillus sp. Ind. Biotechnol. 2022, 18, 304–313. [Google Scholar] [CrossRef]
- Zha, X.; Tsapekos, P.; Zhu, X.; Khoshnevisan, B.; Lu, X.; Angelidaki, I. Bioconversion of Wastewater to Single Cell Protein by Methanotrophic Bacteria. Bioresour. Technol. 2021, 320, 124351. [Google Scholar] [CrossRef]
- Hadi, J.; Brightwell, G. Safety of Alternative Proteins: Technological, Environmental and Regulatory Aspects of Cultured Meat, Plant-Based Meat, Insect Protein and Single-Cell Protein. Foods 2021, 10, 1226. [Google Scholar] [CrossRef]
- Janssen, M.; Wijffels, R.H.; Barbosa, M.J. Microalgae Based Production of Single-Cell Protein. Curr. Opin. Biotechnol. 2022, 75, 102705. [Google Scholar] [CrossRef]
- Montenegro-Herrera, C.A.; Vera-López Portillo, F.; Hernández-Chávez, G.T.; Martinez, A. Single-Cell Protein Production Potential with the Extremophilic Red Microalgae Galdieria Sulphuraria: Growth and Biochemical Characterization. J. Appl. Phycol. 2022, 34, 1341–1352. [Google Scholar] [CrossRef]
- Grosshagauer, S.; Kraemer, K.; Somoza, V. The True Value of Spirulina. J. Agric. Food Chem. 2020, 68, 4109–4115. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A Potential Alternative to Health Supplementation for Humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Geada, P.; Moreira, C.; Silva, M.; Nunes, R.; Madureira, L.; Rocha, C.M.R.; Pereira, R.N.; Vicente, A.A.; Teixeira, J.A. Algal Proteins: Production Strategies and Nutritional and Functional Properties. Bioresour. Technol. 2021, 332, 125125. [Google Scholar] [CrossRef]
- Wang, D.-H.; Zhu, M.-Y.; Lian, S.-J.; Zou, H.; Fu, S.-F.; Guo, R.-B. Conversion of Renewable Biogas into Single-Cell Protein Using a Combined Microalga- and Methane-Oxidizing Bacterial System. ACS EST Eng. 2022, 2, 2317–2325. [Google Scholar] [CrossRef]
- Rasouli, Z.; Valverde-Pérez, B.; D’Este, M.; De Francisci, D.; Angelidaki, I. Nutrient Recovery from Industrial Wastewater as Single Cell Protein by a Co-Culture of Green Microalgae and Methanotrophs. Biochem. Eng. J. 2018, 134, 129–135. [Google Scholar] [CrossRef]
- Hülsen, T.; Hsieh, K.; Lu, Y.; Tait, S.; Batstone, D.J. Simultaneous Treatment and Single Cell Protein Production from Agri-Industrial Wastewaters Using Purple Phototrophic Bacteria or Microalgae—A Comparison. Bioresour. Technol. 2018, 254, 214–223. [Google Scholar] [CrossRef]
- Anupama; Ravindra, P. Value-Added Food: Single Cell Protein. Biotechnol. Adv. 2000, 18, 459–479. [Google Scholar] [CrossRef]
- Du, L.; Zong, Y.; Li, H.; Wang, Q.; Xie, L.; Yang, B.; Pang, Y.; Zhang, C.; Zhong, Z.; Gao, J. Hyperuricemia and Its Related Diseases: Mechanisms and Advances in Therapy. Signal Transduct. Target. Ther. 2024, 9, 212. [Google Scholar] [CrossRef]
- Sekoai, P.T.; Roets-Dlamini, Y.; O’Brien, F.; Ramchuran, S.; Chunilall, V. Valorization of Food Waste into Single-Cell Protein: An Innovative Technological Strategy for Sustainable Protein Production. Microorganisms 2024, 12, 166. [Google Scholar] [CrossRef]
- Aidoo, R.; Kwofie, E.M.; Adewale, P.; Lam, E.; Ngadi, M. Overview of Single Cell Protein: Production Pathway, Sustainability Outlook, and Digital Twin Potentials. Trends Food Sci. Technol. 2023, 138, 577–598. [Google Scholar] [CrossRef]
- Najafpour, G.D. Single-Cell Protein. In Biochemical Engineering and Biotechnology; Elsevier: Amsterdam, The Netherlands, 2015; pp. 417–434. ISBN 978-0-444-63357-6. [Google Scholar]
- Salazar-López, N.J.; Barco-Mendoza, G.A.; Zuñiga-Martínez, B.S.; Domínguez-Avila, J.A.; Robles-Sánchez, R.M.; Ochoa, M.A.V.; González-Aguilar, G.A. Single-Cell Protein Production as a Strategy to Reincorporate Food Waste and Agro By-Products Back into the Processing Chain. Bioengineering 2022, 9, 623. [Google Scholar] [CrossRef]
- Balagurunathan, B.; Ling, H.; Choi, W.J.; Chang, M.W. Potential Use of Microbial Engineering in Single-Cell Protein Production. Curr. Opin. Biotechnol. 2022, 76, 102740. [Google Scholar] [CrossRef]
- Wang, L.; Li, G.; Chen, X.; Yang, Y.; Liew, R.K.; Abo-Dief, H.M.; Lam, S.S.; Sellami, R.; Peng, W.; Li, H. Extraction Strategies for Lignin, Cellulose, and Hemicellulose to Obtain Valuable Products from Biomass. Adv. Compos. Hybrid Mater. 2024, 7, 219. [Google Scholar] [CrossRef]
- Kim, B.; Lee, J.; Jang, S.; Park, J.; Choi, J.; Lee, S.; Jung, J.; Park, J. Exploring the Effect of the Polyol Structure and the Incorporation of Lignin on the Properties of Bio-Based Polyurethane. Polymers 2025, 17, 604. [Google Scholar] [CrossRef]
- Gao, L.; Jiang, F.; Zhang, Z.; Bao, T.; Zhu, D.; Wu, X. Unlocking Lignin Valorization and Harnessing Lignin-Based Raw Materials for Bio-Manufacturing. Sci. China Life Sci. 2025, 68, 994–1009. [Google Scholar] [CrossRef]
- Putri, D.; Ulhidayati, A.; Musthofa, I.A.; Wardani, A.K. Single Cell Protein Production of Chlorella sp. Using Food Processing Waste as a Cultivation Medium. IOP Conf. Ser. Earth Environ. Sci. 2018, 131, 012052. [Google Scholar] [CrossRef]
- Pruksasri, S.; Wollinger, K.K.; Novalin, S. Transformation of Rice Bran into Single-Cell Protein, Extracted Protein, Soluble and Insoluble Dietary Fiber, and Minerals. J. Sci. Food Agric. 2019, 99, 5044–5049. [Google Scholar] [CrossRef]
- Chen, Y.; Song, C.; Cui, X.; Han, J.; Paithoonrangsarid, K.; Gan, Q.; Lu, Y. Halophilic Microalga-Based Circular Economy Producing Functional Food by Reclaiming High-Salinity Seafood Processing Sewage. Biomass Bioenergy 2023, 178, 106952. [Google Scholar] [CrossRef]
- Dou, J.; Huang, Y.; Ren, H.; Li, Z.; Cao, Q.; Liu, X.; Li, D. Autotrophic, Heterotrophic, and Mixotrophic Nitrogen Assimilation for Single-Cell Protein Production by Two Hydrogen-Oxidizing Bacterial Strains. Appl. Biochem. Biotechnol. 2019, 187, 338–351. [Google Scholar] [CrossRef]
- Khoshnevisan, B.; Tsapekos, P.; Zhang, Y.; Valverde-Pérez, B.; Angelidaki, I. Urban Biowaste Valorization by Coupling Anaerobic Digestion and Single Cell Protein Production. Bioresour. Technol. 2019, 290, 121743. [Google Scholar] [CrossRef]
- Patthawaro, S.; Saejung, C. Production of Single Cell Protein from Manure as Animal Feed by Using Photosynthetic Bacteria. MicrobiologyOpen 2019, 8, e913. [Google Scholar] [CrossRef]
- Rashid, N.; Abdelnabi, M.N.A.; Vincent, A.S.; Mackey, H.R. Simultaneous Treatment of Fruit Juice Industry Wastewater and Single-Cell Protein Synthesis Using Purple Non-Sulfur Bacteria. Biomass Convers. Biorefinery 2023, 13, 16321–16332. [Google Scholar] [CrossRef]
- Kumari, P.V.K.; Rao, Y.S.; Vaishnavi, V.R.; Sowjanya, D. Production of single cell protein from sugarcane using fungi. Int. J. Pharm. Sci. Res. 2018, 9, 1213–1217. [Google Scholar] [CrossRef]
- Alankar, S.S.L.; Sajesh, N.; Rastogi, S.; Sakhuja, S.; Rajeswari, G.; Kumar, V.; Chandel, A.K.; Jacob, S. Bioprocessing of Fermentable Sugars Derived from Water Hyacinth into Microbial Lipids and Single Cell Proteins by Oleaginous Yeast Rhodosporidium Toruloides NCIM 3547. Biomass Convers. Biorefin. 2023, 13, 15435–15449. [Google Scholar] [CrossRef]
- Bakratsas, G.; Polydera, A.; Nilson, O.; Kossatz, L.; Xiros, C.; Katapodis, P.; Stamatis, H. Single-Cell Protein Production by Pleurotus Ostreatus in Submerged Fermentation. Sustain. Food Technol. 2023, 1, 377–389. [Google Scholar] [CrossRef]
- Gervasi, T.; Pellizzeri, V.; Calabrese, G.; Di Bella, G.; Cicero, N.; Dugo, G. Production of Single Cell Protein (SCP) from Food and Agricultural Waste by Using Saccharomyces cerevisiae. Nat. Prod. Res. 2018, 32, 648–653. [Google Scholar] [CrossRef]
- Razzaq, Z.U.; Khan, M.K.I.; Maan, A.A.; Rahman, S.U. Characterization of Single Cell Protein from Saccharomyces cerevisiae for Nutritional, Functional and Antioxidant Properties. Food Meas. 2020, 14, 2520–2528. [Google Scholar] [CrossRef]
- Khan, S.; Qurat-Ul-Ain; Ishaq, M.; Salman, M.; Rehman, A.; Ullah, I.; Wazir, K.; Jamil, J. Production of Single Cell Microbial Protein and Its Use as Protein Source in Broiler Ration. Pak. J. Zool. 2020, 53, 875. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, P.; Chen, Y.C.; Cao, Q.; Liu, X.F.; Li, D. The Production of Single Cell Protein from Biogas Slurry with High Ammonia-Nitrogen Content by Screened Nectaromyces Rattus. Poult. Sci. 2021, 100, 101334. [Google Scholar] [CrossRef] [PubMed]
- Bertasini, D.; Binati, R.L.; Bolzonella, D.; Battista, F. Single Cell Proteins Production from Food Processing Effluents and Digestate. Chemosphere 2022, 296, 134076. [Google Scholar] [CrossRef]
- Carranza-Méndez, R.C.; Chávez-González, M.L.; Sepúlveda-Torre, L.; Aguilar, C.N.; Govea-Salas, M.; Ramos-González, R. Production of Single Cell Protein from Orange Peel Residues by Candida utilis. Biocatal. Agric. Biotechnol. 2022, 40, 102298. [Google Scholar] [CrossRef]
- Hashem, M.; Al-Qahtani, M.S.; Alamri, S.A.; Moustafa, Y.S.; Lyberatos, G.; Ntaikou, I. Valorizing Food Wastes: Assessment of Novel Yeast Strains for Enhanced Production of Single-Cell Protein from Wasted Date Molasses. Biomass Convers. Biorefin. 2022, 12, 4491–4502. [Google Scholar] [CrossRef]
- Pillaca-Pullo, O.S.; Lopes, A.M.; Rodriguez-Portilla, L.M.I.; Estela-Escalante, W. Optimizing Medium Composition with Wastewater from Coffea Arabica Processing to Produce Single-Cell Protein Using Candida Sorboxylosa. J. Chem. Technol. Biotechnol. 2023, 98, 106–116. [Google Scholar] [CrossRef]
- Zeng, D.; Jiang, Y.; Su, Y.; Zhang, Y. Upcycling Waste Organic Acids and Nitrogen into Single Cell Protein via Brewer’s Yeast. J. Clean. Prod. 2022, 369, 133279. [Google Scholar] [CrossRef]
- Abodunde, C.A.; Akin-Osanaiye, B.C. Conversion of Orange and Pineapple Fruit Peel Waste into Single Cell Protein Using Saccharomyces cerevisiae. IJ-FANRes 2023, 4, 14–20. [Google Scholar] [CrossRef]
- Tian, Y.; Li, J.; Meng, J.; Li, J. High-Yield Production of Single-Cell Protein from Starch Processing Wastewater Using Co-Cultivation of Yeasts. Bioresour. Technol. 2023, 370, 128527. [Google Scholar] [CrossRef]
- Zhang, B.; Ren, D.; Liu, Q.; Liu, X.; Bao, J. Coproduction of Single Cell Protein and Lipid from Lignocellulose Derived Carbohydrates and Inorganic Ammonia Salt with Soluble Ammonia Recycling. Bioresour. Technol. 2023, 384, 129345. [Google Scholar] [CrossRef]
- Liu, K.; Huang, S.; Zhang, L.; Xiong, Y.; Wang, X.; Bao, Y.; Li, D.; Li, J. Efficient Production of Single Cell Protein from Biogas Slurry Using Screened Alkali-Salt-Tolerant Debaryomyces hansenii. Bioresour. Technol. 2024, 393, 130119. [Google Scholar] [CrossRef]
| Fermentation Methods | Advantages | Disadvantages |
|---|---|---|
| Submerged (LSF) |
|
|
| Semi-solid |
|
|
| Solid-state (SSF) |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
dos Anjos, I.V.; Coelho, N.; Duarte, H.; Proença, D.N.; Duarte, M.F.; Barros, R.; Raposo, S.; Gonçalves, S.; Romano, A.; Medronho, B. From Lignocellulosic Residues to Protein Sources: Insights into Biomass Pre-Treatments and Conversion. Polymers 2025, 17, 2251. https://doi.org/10.3390/polym17162251
dos Anjos IV, Coelho N, Duarte H, Proença DN, Duarte MF, Barros R, Raposo S, Gonçalves S, Romano A, Medronho B. From Lignocellulosic Residues to Protein Sources: Insights into Biomass Pre-Treatments and Conversion. Polymers. 2025; 17(16):2251. https://doi.org/10.3390/polym17162251
Chicago/Turabian Styledos Anjos, Isabela Vera, Natacha Coelho, Hugo Duarte, Diogo Neves Proença, Maria F. Duarte, Raul Barros, Sara Raposo, Sandra Gonçalves, Anabela Romano, and Bruno Medronho. 2025. "From Lignocellulosic Residues to Protein Sources: Insights into Biomass Pre-Treatments and Conversion" Polymers 17, no. 16: 2251. https://doi.org/10.3390/polym17162251
APA Styledos Anjos, I. V., Coelho, N., Duarte, H., Proença, D. N., Duarte, M. F., Barros, R., Raposo, S., Gonçalves, S., Romano, A., & Medronho, B. (2025). From Lignocellulosic Residues to Protein Sources: Insights into Biomass Pre-Treatments and Conversion. Polymers, 17(16), 2251. https://doi.org/10.3390/polym17162251













