Tuning Corn Zein-Chitosan Biocomposites via Mild Alkaline Treatment: Structural and Physicochemical Property Insights
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Materials
2.2. Corn Zein–Chitosan Composite Films
2.3. Fourier Transform Infrared Spectroscopy (FTIR)
2.4. Differential Scanning Calorimetry (DSC)
2.5. Thermal Gravitational Analysis (TGA)
2.6. Scanning Electron Microscopy (SEM)
3. Results and Discussion
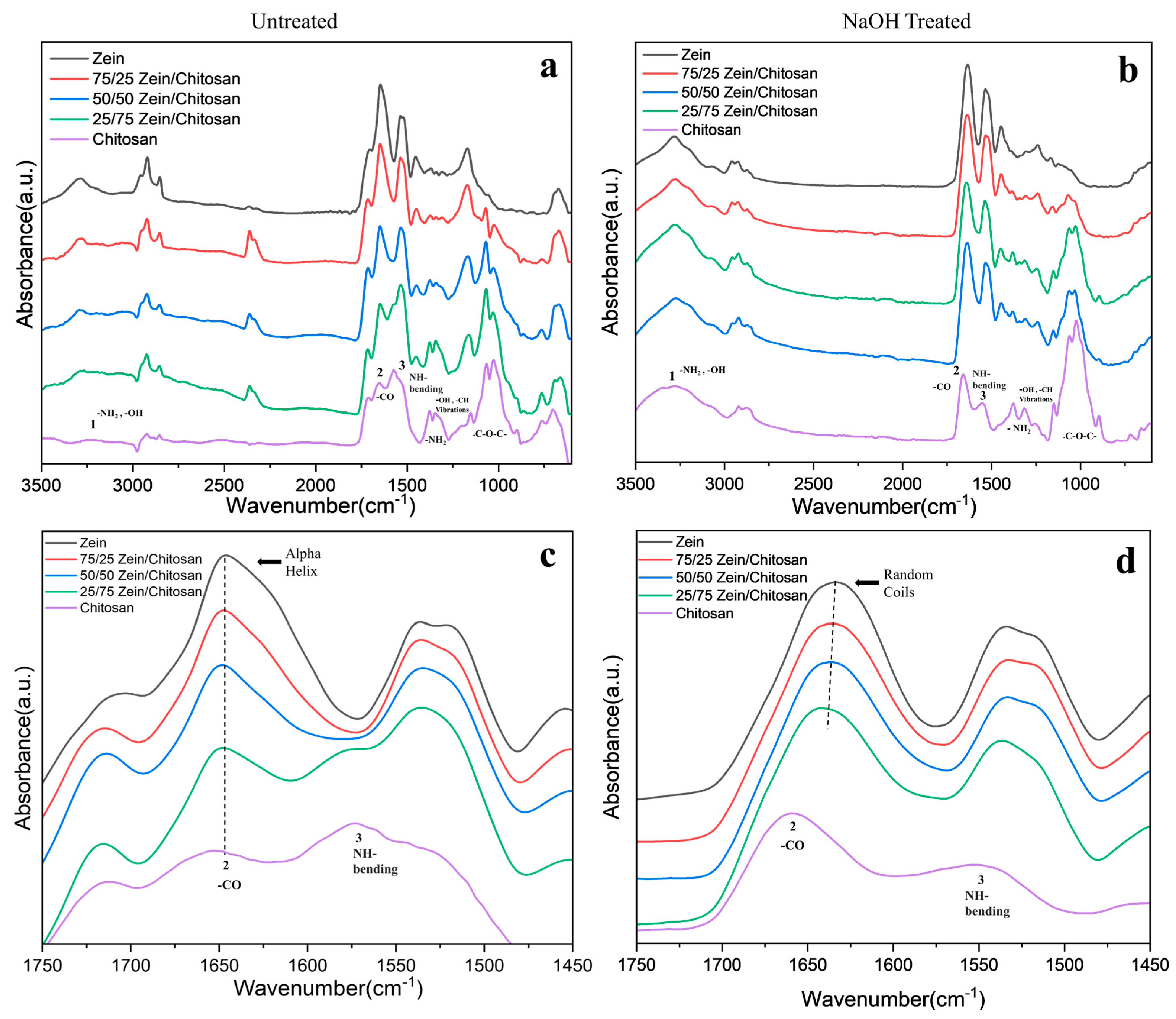
3.1. Structural Analysis
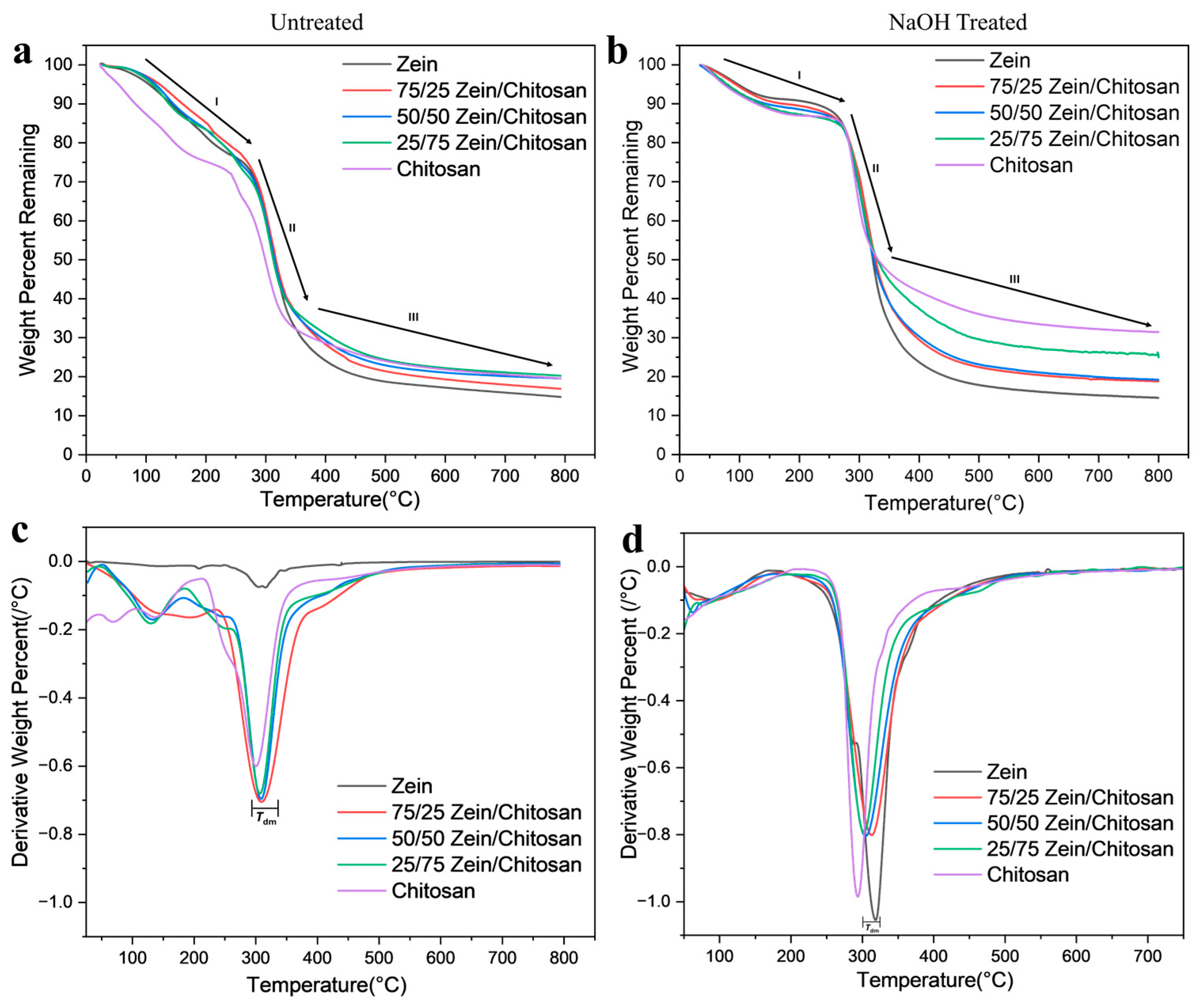
3.2. Thermogravimetric Analysis
3.3. Differential Scanning Calorimetry
3.4. Scanning Electron Microscopy
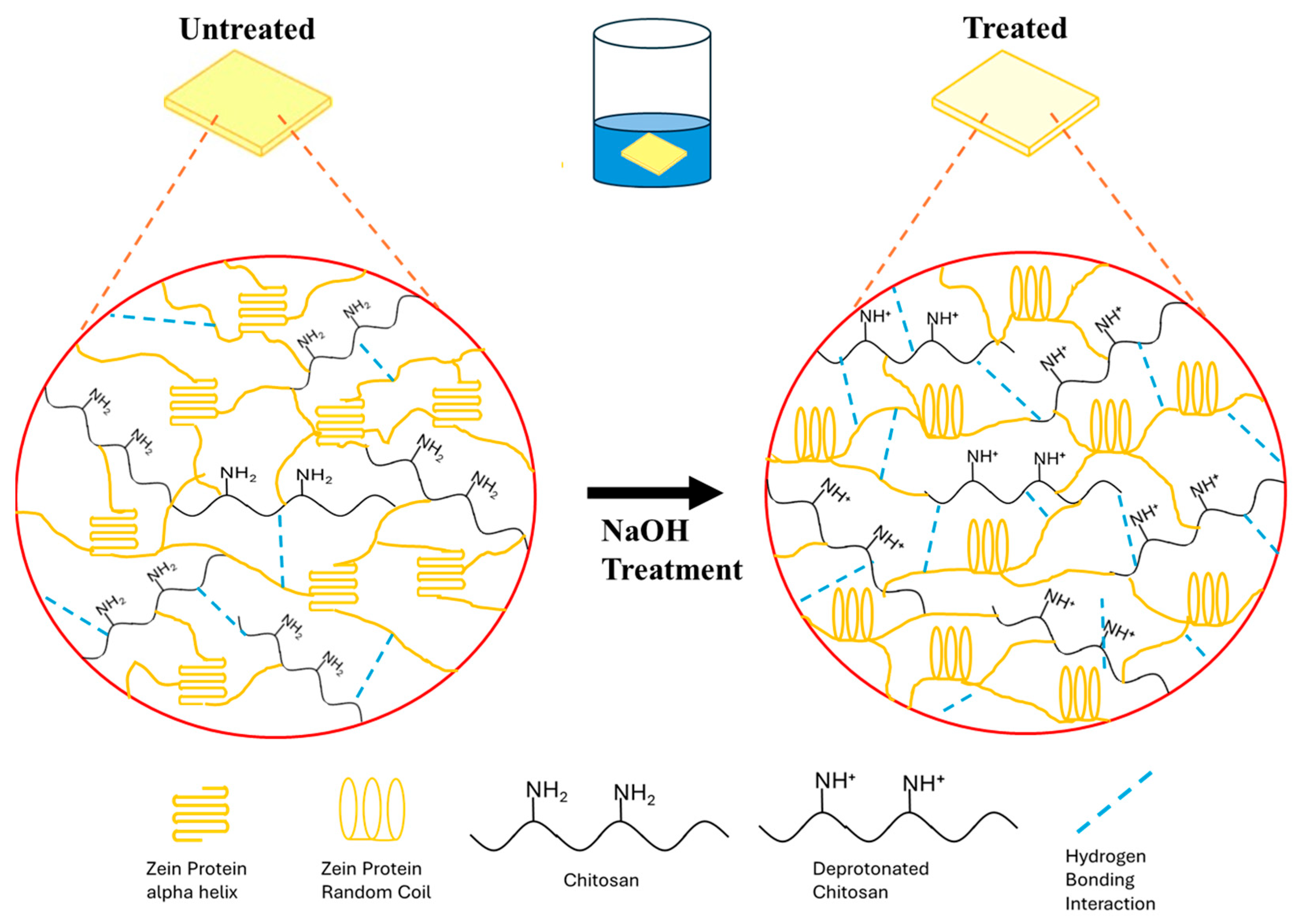
3.5. Mechanism of Zein–Chitosan Interaction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hu, X.; Cebe, P.; Weiss, A.S.; Omenetto, F.; Kaplan, D.L. Protein-based composite materials. Mater. Today 2012, 15, 208–215. [Google Scholar] [CrossRef]
- Hu, X.; Duki, S.; Forys, J.; Hettinger, J.; Buchicchio, J.; Dobbins, T.; Yang, C. Designing silk-silk protein alloy materials for biomedical applications. J. Vis. Exp. 2014, 90, e50891. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, L.; Che, X.; Zhang, H.; Shi, N.; Li, C.; Chen, Y.; Kong, W. Zein-based films and their usage for controlled delivery: Origin, classes and current landscape. J. Control. Release 2015, 206, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Argos, P.; Pedersen, K.; Marks, M.D.; Larkins, B.A.A. structural model for maize zein proteins. J. Biol. Chem. 1982, 257, 9984–9990. [Google Scholar] [CrossRef]
- Matsushima, N.; Danno, G.-I.; Takezawa, H.; Izumi, Y. Three-dimensional structure of maize α-zein proteins studied by small-angle X-ray scattering. Biochim. Biophys. Acta (BBA)-Protein Struct. Mol. Enzym. 1997, 1339, 14–22. [Google Scholar] [CrossRef]
- Poluri, N.; Gough, C.R.; Sanderlin, S.; Velardo, C.; Barca, A.; Pinto, J.; Perrotta, J.; Cohen, M.; Hu, X. Silk-Corn Zein Alloy Materials: Influence of Silk Types (Mori, Thai, Muga, Tussah, and Eri) on the Structure, Properties, and Functionality of Insect—Plant Protein Blends. Int. J. Mol. Sci. 2025, 26, 186. [Google Scholar] [CrossRef]
- Reboredo, C.; González-Navarro, C.J.; Martínez-López, A.L.; Martínez-Ohárriz, C.; Sarmento, B.; Irache, J.M. Zein-Based Nanoparticles as Oral Carriers for Insulin Delivery. Pharmaceutics 2021, 14, 39. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, H.; Su, J.; Brennan, C.S.; Van der Meeren, P.; Zhang, N.; Tong, Y.; Wang, P. Recent developments of electrospun zein nanofibres: Strategies, fabrication and therapeutic applications Recent developments of electrospun zein nanofibres: Strategies, fabrication and therapeutic applications. Mater. Today Adv. 2022, 16, 100307. [Google Scholar] [CrossRef]
- DeFrates, K.; Markiewicz, T.; Xue, Y.; Callaway, K.; Gough, C.; Moore, R.; Bessette, K.; Mou, X.; Hu, X. Air-jet spinning corn zein protein nanofibers for drug delivery: Effect of biomaterial structure and shape on release properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 118, 111419. [Google Scholar] [CrossRef] [PubMed]
- Mouzakitis, C.K.; Sereti, V.; Matsakidou, A.; Kotsiou, K.; Biliaderis, C.G.; Lazaridou, A. Physicochemical properties of zein-based edible films and coatings for extending wheat bread shelf life. Food Hydrocoll. 2022, 132, 107856. [Google Scholar] [CrossRef]
- Corradini, E.; Souto De Medeiros, E.; Carvalho, A.J.F.; Curvelo, A.A.S.; Mattoso, L.H.C. Mechanical and morphological characterization of starch/zein blends plasticized with glycerol. J. Appl. Polym. Sci. 2006, 101, 4133–4139. [Google Scholar] [CrossRef]
- Huang, W.; Zou, T.; Li, S.; Jing, J.; Xia, X.; Liu, X. Drug-Loaded Zein Nanofibers Prepared Using a Modified Coaxial Electrospinning Process. AAPS PharmSciTech 2013, 14, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Li, X.; Song, T.; Li, Y.; Pu, Y. Biodegradable nanofibrous membrane of zein/silk fibroin by electrospinning. Polym. Int. 2009, 58, 396–402. [Google Scholar] [CrossRef]
- Aranaz, I.; Alcántara, A.R.; Civera, M.C.; Arias, C.; Elorza, B.; Heras Caballero, A.; Acosta, N. Chitosan: An Overview of Its Properties and Applications. Polymers 2021, 13, 3256. [Google Scholar] [CrossRef] [PubMed]
- Mittal, H.; Ray, S.S.; Kaith, B.S.; Bhatia, J.K.; Sukriti; Sharma, J.; Alhassan, S.M. Recent progress in the structural modification of chitosan for applications in diversified biomedical fields. Eur. Polym. J. 2018, 109, 402–434. [Google Scholar] [CrossRef]
- Takara, E.A.; Marchese, J.; Ochoa, N.A. NaOH treatment of chitosan films: Impact on macromolecular structure and film properties. Carbohydr. Polym. 2015, 132, 25–30. [Google Scholar] [CrossRef]
- Schmuhl, R.; Krieg, H.M.; Keizer, K. Adsorption of Cu (II) and Cr (VI) ions by chitosan: Kinetics and equilibrium studies. Water SA 2001, 27, 1–8. [Google Scholar] [CrossRef]
- Moraes, M.A.d.; Nogueira, G.M.; Weska, R.F.; Beppu, M.M. Preparation and Characterization of Insoluble Silk Fibroin/Chitosan Blend Films. Polymers 2010, 2, 719–727. [Google Scholar] [CrossRef]
- Huang, J.; Qin, J.; Zhang, P.; Chen, X.; You, X.; Zhang, F.; Zuo, B.; Yao, M. Facile preparation of a strong chitosan-silk biocomposite film. Carbohydr. Polym. 2020, 229, 115515. [Google Scholar] [CrossRef]
- Li, B.; Qiu, L.; Zhang, J.; Liu, S.; Xu, M.; Wang, J.; Yang, H. Solubilization of chitosan in biologically relevant solvents by a low-temperature solvent-exchange method for developing biocompatible chitosan materials. Int. J. Biol. Macromol. 2023, 254 Pt 3, 127950. [Google Scholar] [CrossRef]
- Sakai, Y.; Hayano, K.; Yoshioka, H.; Yoshioka, H. A Novel Method of Dissolving Chitosan in Water for Industrial Application. Polym. J. 2001, 33, 640–642. [Google Scholar] [CrossRef]
- Sharkawy, A.; Barreiro, M.F.; Rodrigues, A.E. Chitosan-based Pickering emulsions and their applications: A review. Carbohydr. Polym. 2020, 250, 116885. [Google Scholar] [CrossRef] [PubMed]
- Loryuenyong, V.; Nakhlo, W.; Srikaenkaew, P.; Yaidee, P.; Eiad-Ua, A.; Buasri, A. Optimization and performance prediction of carbon dioxide adsorption on chitosan/ activated carbon/epichlorohydrin composite materials using Box–Behnken design and artificial neural network approaches. Case Stud. Chem. Environ. Eng. 2025, 11, 101144. [Google Scholar] [CrossRef]
- Oktay, B.; Ciftci, F.; Erarslan, A.; Ahlatcıoğlu Özerol, E. Dual-layer Natamycin and Boric-Acid-reinforced PVA/chitosan by 3D printing and electrospinning method: Characterization and In Vitro evaluation. Polymers 2025, 17, 1673. [Google Scholar] [CrossRef] [PubMed]
- Brito, G.B.; Pinho-Jr, J.d.S.; Guimarães, A.d.S.; Conte-Júnior, C.A.; Nele, M.; Perrone, D.; Castelo-Branco, V.N. Optimization and characterization of crosslinked chitosan-based oleogels based on mechanical properties of conventional solid fats. Polymers 2025, 17, 1526. [Google Scholar] [CrossRef]
- Yu, C.; Sun, H.; Yao, L.; Weng, Y. NaOH/Urea-Compatible Chitosan/Carboxymethylcellulose Films: Orthogonal Optimization of Packaging Properties. Molecules 2025, 30, 2279. [Google Scholar] [CrossRef]
- Masanabo, M.A.; Ray, S.S.; Emmambux, M.N. Properties of thermoplastic maize starch-zein composite films prepared by extrusion process under alkaline conditions. Int. J. Biol. Macromol. 2022, 208, 443–452. [Google Scholar] [CrossRef]
- Czechowska-Biskup, R.; Jarosinska, D.; Rokita, B.; Ulanski, P.; Rosiak, J.M. Determination of degree of deacetylation of chitosan-comparison of methods. Prog. Chem. Appl. Chitin Deriv. 2012, 17, 5–20. [Google Scholar]
- Sánchez-Machado, D.I.; López-Cervantes, J.; Escárcega-Galaz, A.A.; Campas-Baypoli, O.N.; Martínez-Ibarra, D.M.; Rascón-León, S. Measurement of the degree of deacetylation in chitosan films by FTIR, 1H NMR and UV spectrophotometry. MethodsX 2024, 12, 102583. [Google Scholar] [CrossRef]
- Gillgren, T.; Barker, S.A.; Belton, P.S.; Georget, D.M.R.; Stading, M. Plasticization of Zein: A Thermomechanical, FTIR, and Dielectric Study. Biomacromolecules 2009, 10, 1135–1139. [Google Scholar] [CrossRef]
- Gu, Z.; Xie, H.; Huang, C.; Li, L.; Yu, X. Preparation of chitosan/silk fibroin blending membrane fixed with alginate dialdehyde for wound dressing. Int. J. Biol. Macromol. 2013, 58, 121–126. [Google Scholar] [CrossRef]
- Fernandez, J.G.; Ingber, D.E. Unexpected Strength and Toughness in Chitosan-Fibroin Laminates Inspired by Insect Cuticle. Adv. Mater. 2012, 24, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jiang, X.; Huang, H.; Liu, A.; Liu, H.; Abid, N.; Ming, L. Chitosan/zein films incorporated with essential oil nanoparticles and nanoemulsions: Similarities and differences. Int. J. Biol. Macromol. 2022, 208, 983–994. [Google Scholar] [CrossRef]
- Magoshi, J.; Nakamura, S.; Murakami, K.I. Structure and physical properties of seed proteins. I. Glass transition and crystallization of zein protein from corn. J. Appl. Polym. Sci. 1992, 45, 2043–2048. [Google Scholar] [CrossRef]
- Shukla, R.; Cheryan, M. Zein: The industrial protein from corn. Ind. Crops Prod. 2001, 13, 171–192. [Google Scholar] [CrossRef]
- Kweon, H.Y.; Um, I.C.; Park, Y.H. Structural and thermal characteristics of Antheraea pernyi silk fibroin/chitosan blend film. Polymer 2001, 42, 6651–6656. [Google Scholar] [CrossRef]
- de Farias, B.S.; Grundmann, D.D.R.; Rizzi, F.Z.; Martins, N.S.S.; Sant’Anna Cadaval Junior, T.R.; de Almeida Pinto, L.A. Production of low molecular weight chitosan by acid and oxidative pathways: Effect on physicochemical properties. Food Res. Int. 2019, 123, 88–94. [Google Scholar] [CrossRef]
- Kumar, S.; Dutta, J.; Dutta, P.K. Preparation and characterization of N-heterocyclic chitosan derivative based gels for biomedical applications. Int. J. Biol. Macromol. 2009, 45, 330–337. [Google Scholar] [CrossRef]




| Film Composition | Untreated (%, DD) | NaOH Treated (%, DD) |
|---|---|---|
| Chitosan | 74.21 | 84.08 |
| 75/25 Chitosan/Zein | 70.63 | 73.44 |
| 50/50 Chitosan/Zein | 69.36 | 70.58 |
| 25/75 Chitosan/Zein | 60.56 | 67.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poluri, N.; Singer, C.; Salas-de la Cruz, D.; Hu, X. Tuning Corn Zein-Chitosan Biocomposites via Mild Alkaline Treatment: Structural and Physicochemical Property Insights. Polymers 2025, 17, 2161. https://doi.org/10.3390/polym17152161
Poluri N, Singer C, Salas-de la Cruz D, Hu X. Tuning Corn Zein-Chitosan Biocomposites via Mild Alkaline Treatment: Structural and Physicochemical Property Insights. Polymers. 2025; 17(15):2161. https://doi.org/10.3390/polym17152161
Chicago/Turabian StylePoluri, Nagireddy, Creston Singer, David Salas-de la Cruz, and Xiao Hu. 2025. "Tuning Corn Zein-Chitosan Biocomposites via Mild Alkaline Treatment: Structural and Physicochemical Property Insights" Polymers 17, no. 15: 2161. https://doi.org/10.3390/polym17152161
APA StylePoluri, N., Singer, C., Salas-de la Cruz, D., & Hu, X. (2025). Tuning Corn Zein-Chitosan Biocomposites via Mild Alkaline Treatment: Structural and Physicochemical Property Insights. Polymers, 17(15), 2161. https://doi.org/10.3390/polym17152161








