Exploring 1,3-Dioxolane Extraction of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from Methylocystis hirsuta and Mixed Methanotrophic Strain: Effect of Biomass-to-Solvent Ratio and Extraction Time
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals, NMS Medium and Biopolymer Production
2.1.1. Chemicals
2.1.2. Culture Medium
2.1.3. Biopolymer Production
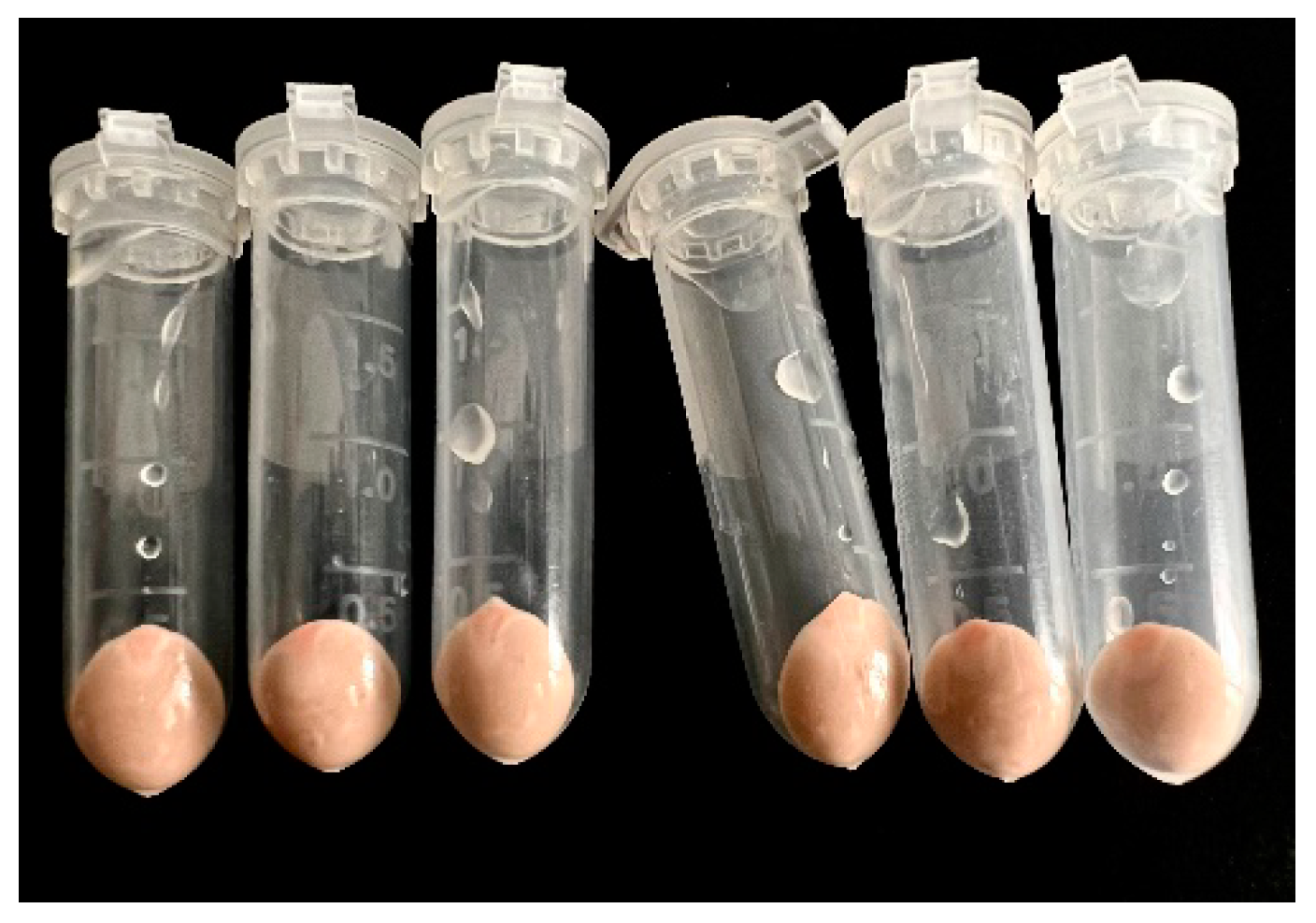
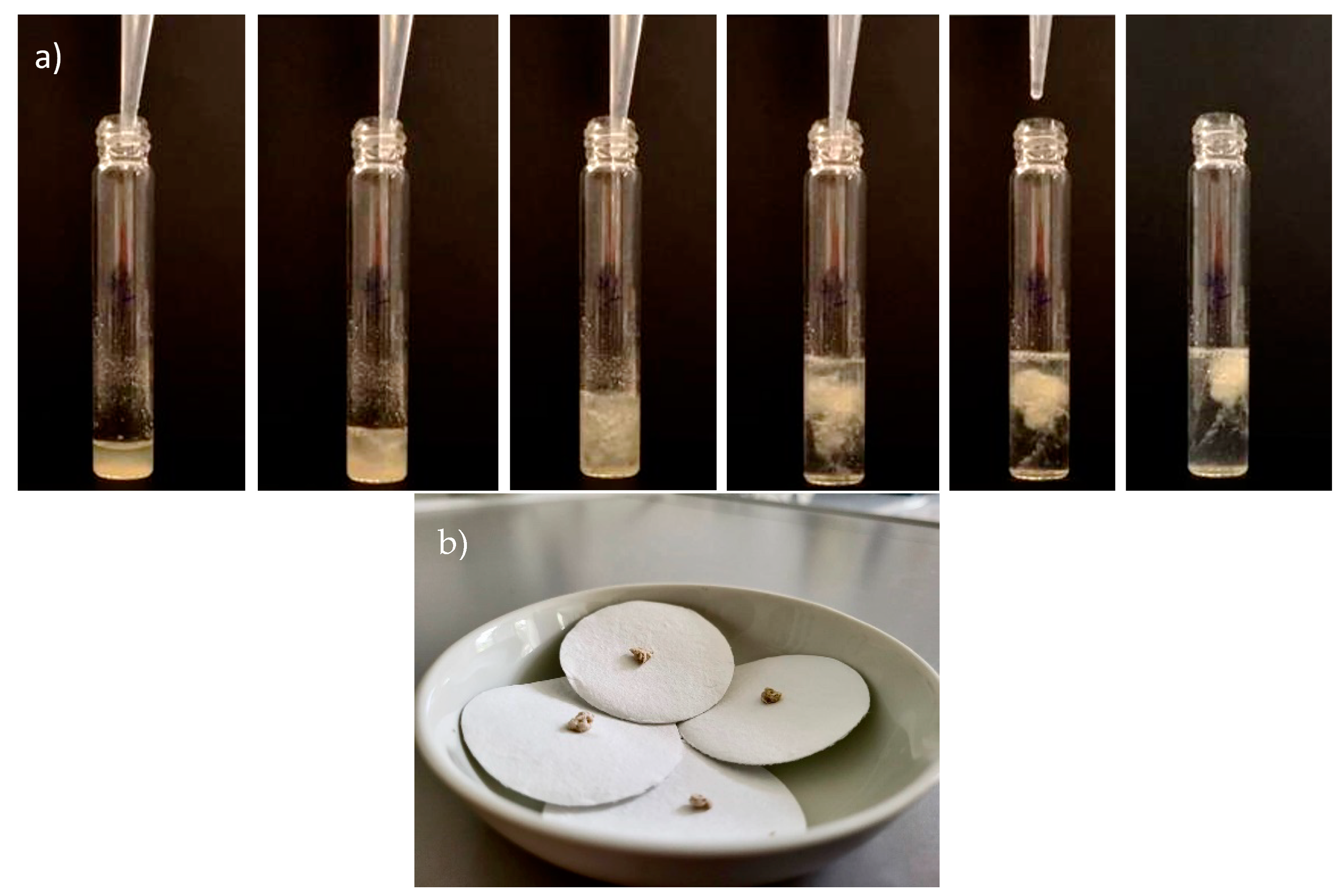
2.2. PHB and PHB-co-HV Extraction with 1,3-Dioxolane
2.3. Extraction Kinetics of PHB and PHB-co-HV
2.4. Recovery and Purity Assessment
2.5. Analytical Procedures
3. Results
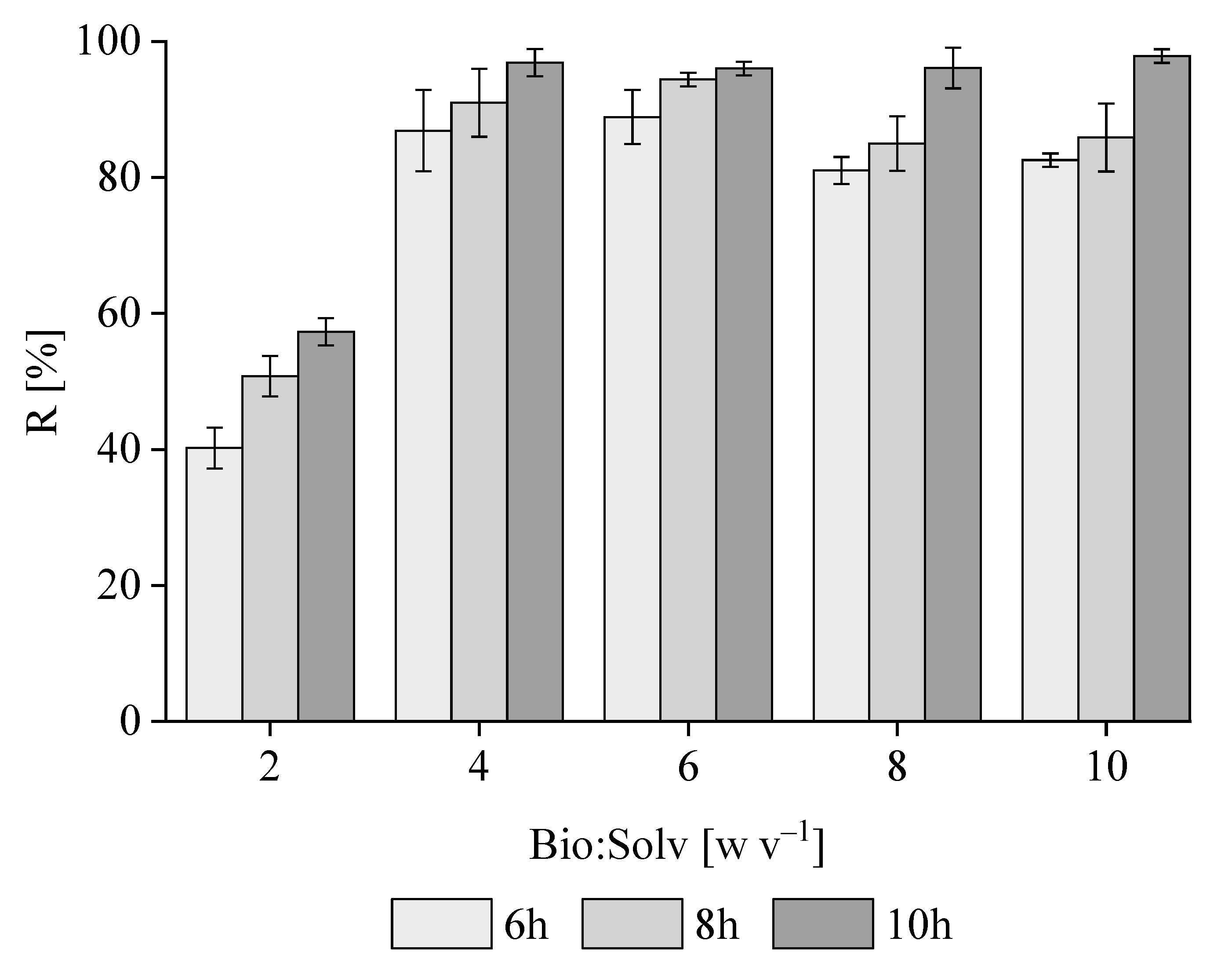
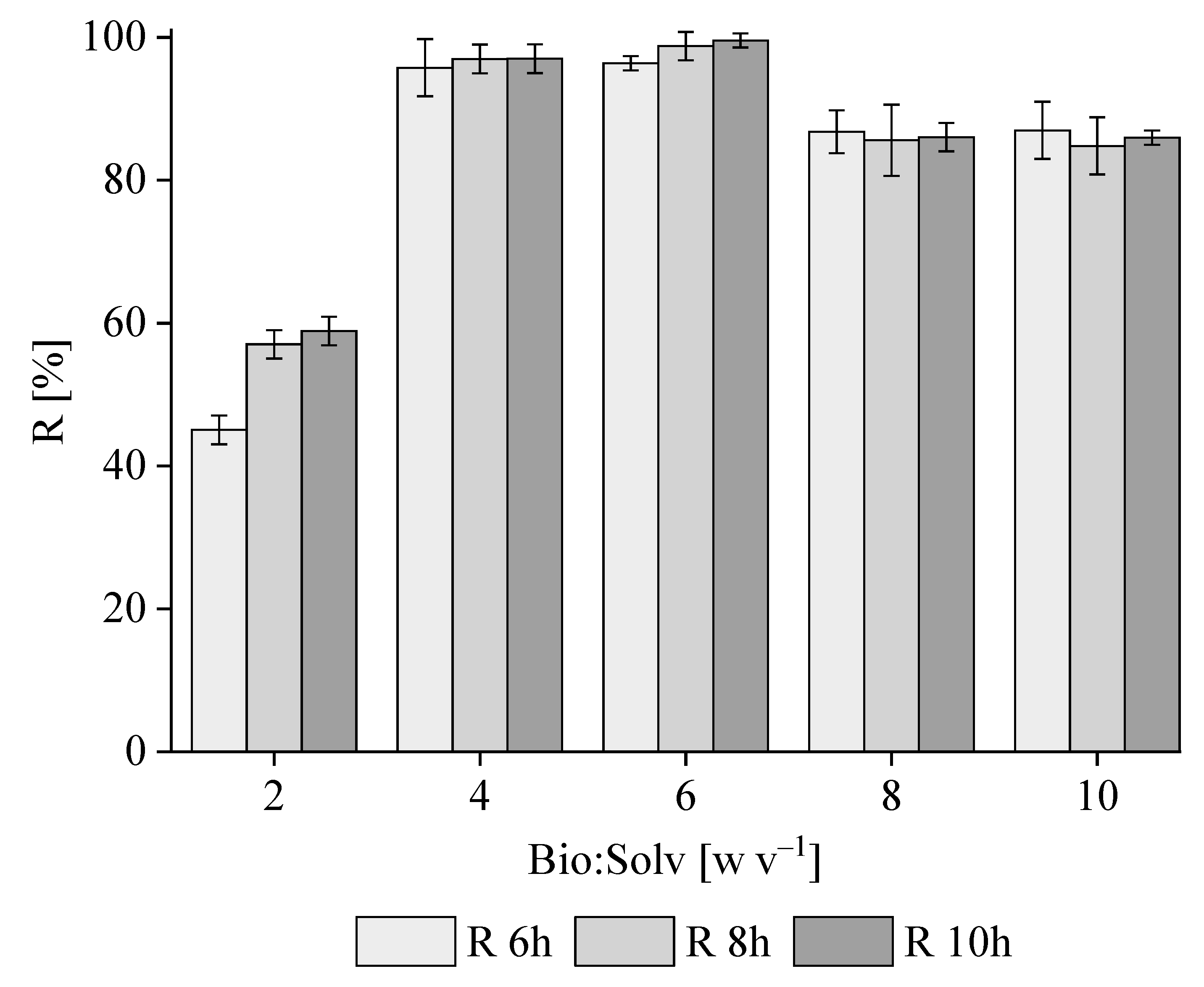
3.1. PHB Extraction with 1,3-Dioxolane from M. hirsuta
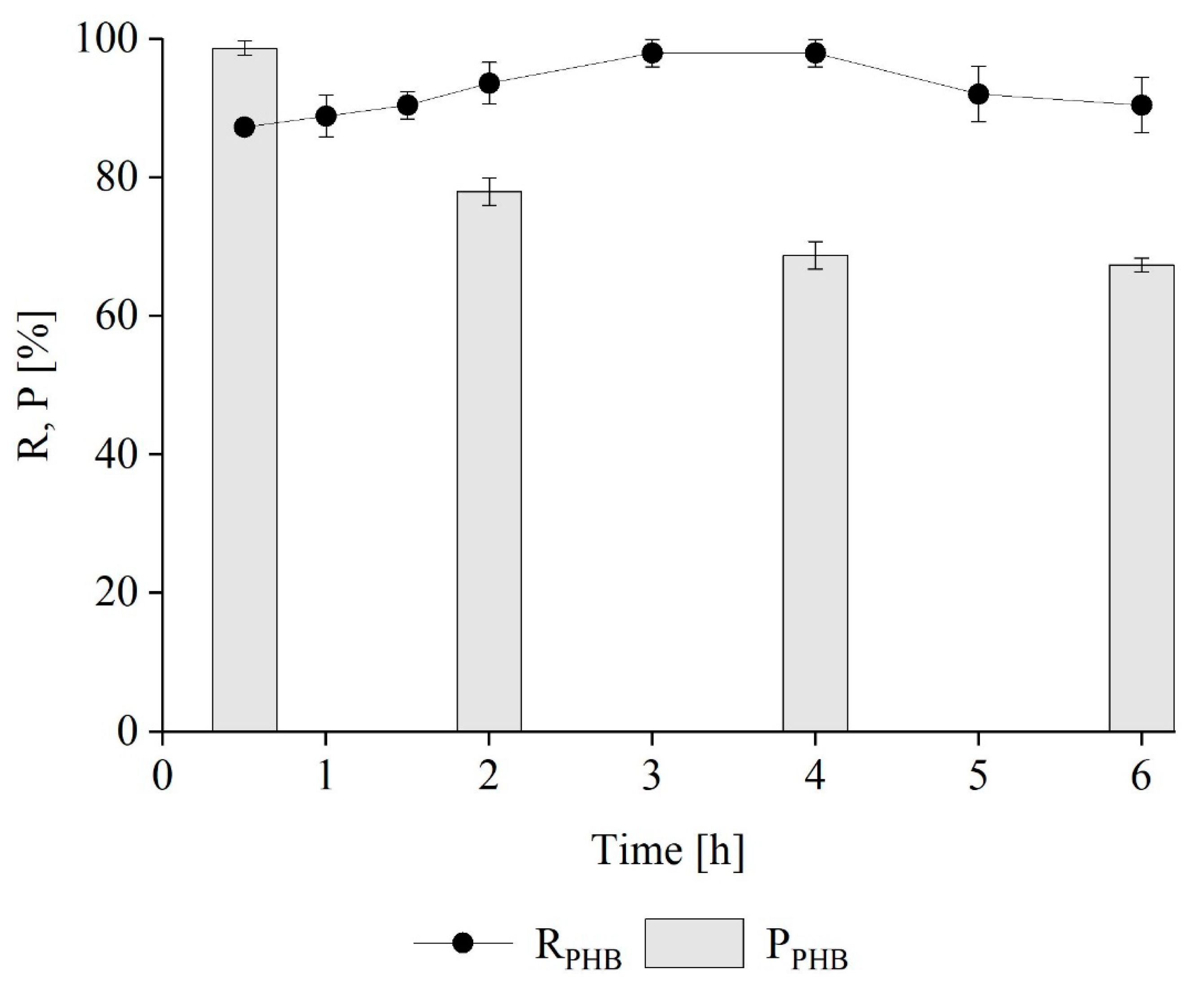
Kinetics of PHB Extraction from M. hirsuta
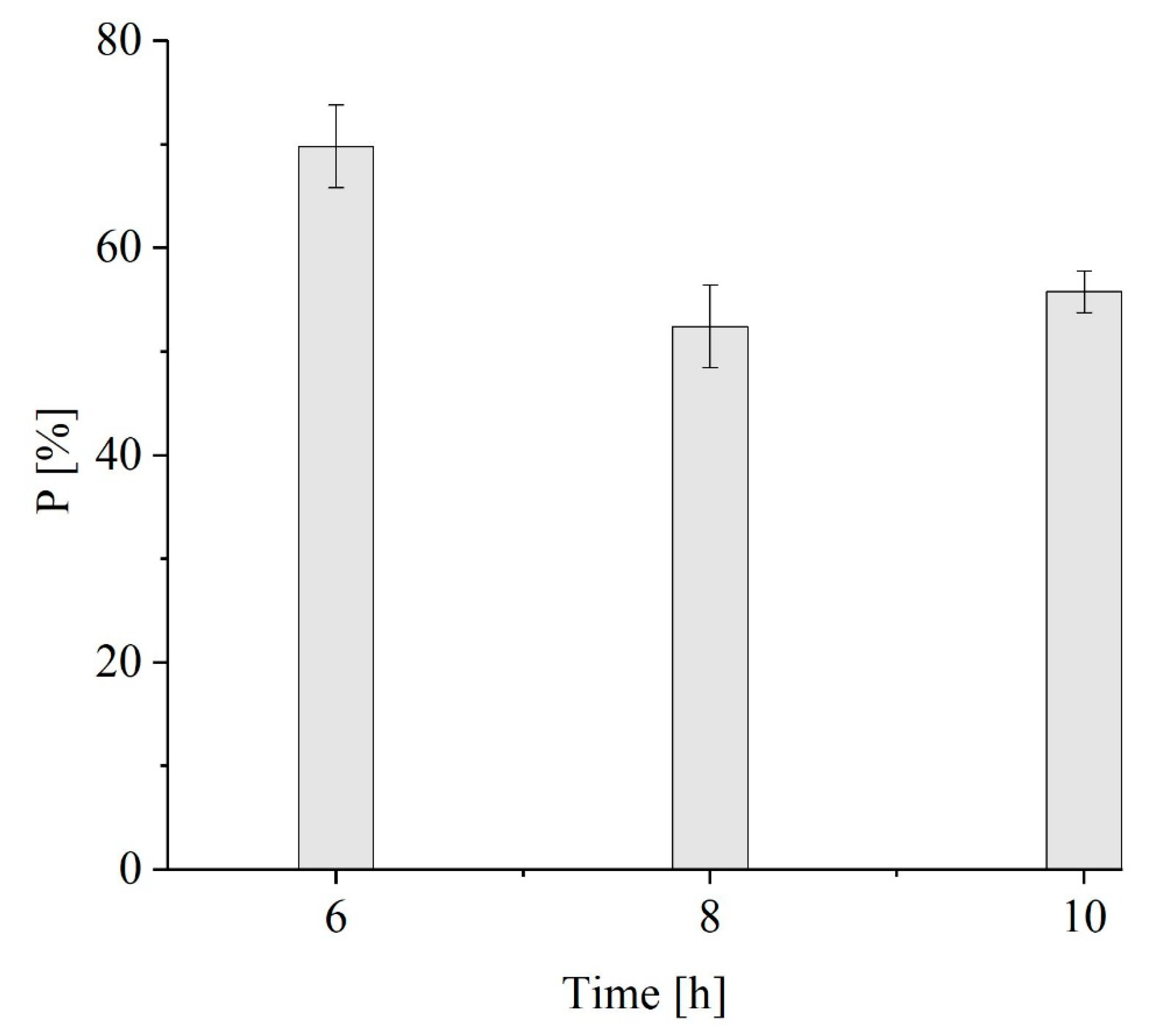
3.2. PHB-co-HV Extraction with 1,3-Dioxolane from M. hirsuta
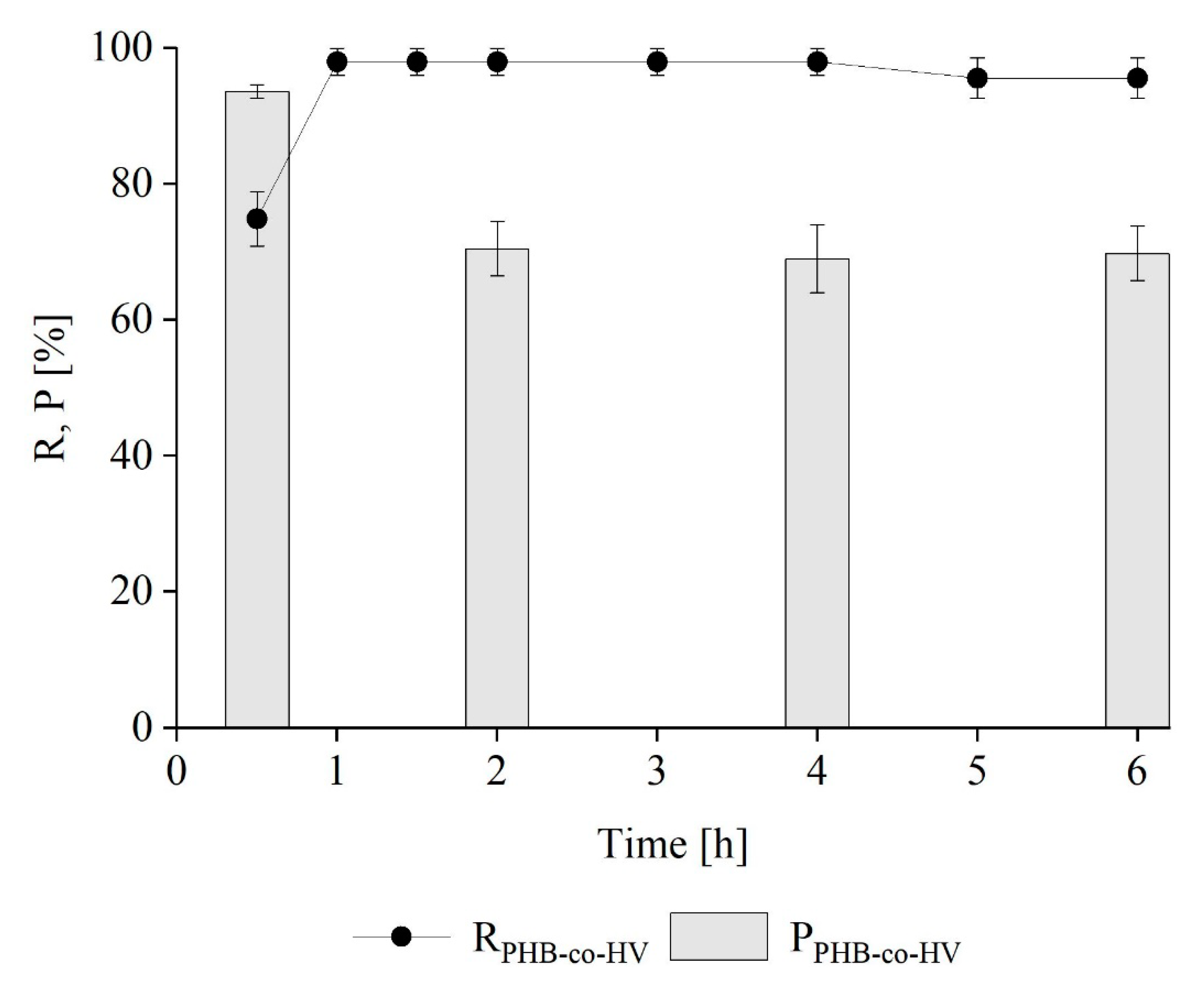
Kinetics of PHB-co-HV Extraction from M. hirsuta
3.3. PHB and PHB-co-HV Extraction from a Mixed Methanotrophic Culture
3.4. Comparison of Extraction Yields for Different Contents of PHB and PHB-co-HV
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Organisation for Economic Co-Operation and Development (OECD), Plastic Pollution is Growing Relentlessly as Waste Management and Recycling Fall Short, Says OECD, OECD Better Policies Better Lives. 29–31. 2022. Available online: https://www.oecd.org/environment/plastic-pollution-is-growing-relentlessly-as-waste-management-and-recycling-fall-short.htm (accessed on 22 February 2022).
- Khanna, S.; Srivastava, A.K. Statistical media optimization studies for growth and PHB production by Ralstonia eutropha. Process. Biochem. 2004, 40, 2173–2182. [Google Scholar] [CrossRef]
- Raberg, M.; Voigt, B.; Hecker, M.; Steinbüchel, A. A Closer Look on the Polyhydroxybutyrate- (PHB-) Negative Phenotype of Ralstonia eutropha PHB-4. PLoS ONE 2014, 9, e95907. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Q.; Wu, Q. The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials 2005, 26, 6565–6578. [Google Scholar] [CrossRef] [PubMed]
- Bonartsev, A.P.; Myshkina, V.L.; Nikolaeva, D.A.; Furina, E.K.; Makhina, T.A. Biosynthesis, biodegradation, and application of poly (3- hydroxybutyrate) and its copolymers—Natural polyesters produced by diazotrophic bacteria. Commun. Curr. Res. Educ. Top. Trends Appl. Microbiol. 2007, 1, 295–307. [Google Scholar]
- Aramvash, A.; Shahabi, Z.A.; Aghjeh, S.D.; Ghafari, M.D. Statistical physical and nutrient optimization of bioplastic polyhydroxybutyrate production by Cupriavidus necator. Int. J. Environ. Sci. Technol. 2015, 12, 2307–2316. [Google Scholar] [CrossRef]
- Koller, M.; Salerno, A.; Dias, M.; Reiterer, A.; Braunegg, G. Modern biotechnological polymer synthesis: A review. Food Technol. Biotechnol. 2010, 48, 255–269. [Google Scholar]
- Shang, L.; Fei, Q.; Zhang, Y.H.; Wang, X.Z.; Fan, D.-D.; Chang, H.N. Thermal Properties and Biodegradability Studies of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate). J. Polym. Environ. 2012, 20, 23–28. [Google Scholar] [CrossRef]
- Kunasundari, B.; Sudesh, K. Isolation and recovery of microbial polyhydroxyalkanoates. Express Polym. Lett. 2011, 5, 620–634. [Google Scholar] [CrossRef]
- Hahn, S.K.; Chang, Y.K.; Kim, B.S.; Chang, H.N. Optimization of microbial poly(3-hydroxybutyrate) recover using dispersions of sodium hypochlorite solution and chloroform. Biotechnol. Bioeng. 1994, 44, 256–261. [Google Scholar] [CrossRef]
- Kapritchkoff, F.M.; Viotti, A.P.; Alli, R.C.; Zuccolo, M.; Pradella, J.G.; Maiorano, A.E.; Miranda, E.A.; Bonomi, A. Enzymatic recovery and purification of polyhydroxybutyrate produced by Ralstonia eutropha. J. Biotechnol. 2006, 122, 453–462. [Google Scholar] [CrossRef]
- Bensch, M.; Selbach, B.; Hubbuch, J. High throughput screening techniques in downstream processing: Preparation, characterization and optimization of aqueous two-phase systems. Chem. Eng. Sci. 2007, 62, 2011–2021. [Google Scholar] [CrossRef]
- Anis, S.N.S.; Iqbal, N.M.; Kumar, S.; Al-Ashraf, A. Increased recovery and improved purity of PHA from recombinant Cupriavidus necator. Bioengineered 2013, 4, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Budde, C.F. Production of Polyhydroxyalkanoate Copolymers from Plant Oil; Massachusetts Institute of Technology: Cambridge, MA, USA, 2010; pp. 1–221. [Google Scholar]
- Wongmoon, C.; Napathorn, S.C. Optimization for the efficient recovery of poly(3-hydroxybutyrate) using the green solvent 1,3-dioxolane. Front. Bioeng. Biotechnol. 2022, 10, 1086636. [Google Scholar] [CrossRef] [PubMed]
- Abate, T.; Amabile, C.; De Crescenzo, C.; Sabbarese, S.; Capece, G.; Ciampa, R.; Chianese, S.; Musmarra, D. Alternative Polyhydroxybutyrate Solvent Extraction: A Process Simulation Study. Chem. Eng. Trans. 2022, 92, 265–270. [Google Scholar] [CrossRef]
- Yang, Y.-H.; Jeon, J.-M.; Yi, D.H.; Kim, J.-H.; Seo, H.-M.; Rha, C.; Sinskey, A.J.; Brigham, C.J. Application of a non-halogenated solvent, methyl ethyl ketone (MEK) for recovery of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) [P(HB-co-HV)] from bacterial cells. Biotechnol. Bioprocess Eng. 2015, 20, 291–297. [Google Scholar] [CrossRef]
- Montiel-Jarillo, G.; Morales-Urrea, D.A.; Contreras, E.M.; López-Córdoba, A.; Gómez-Pachón, E.Y.; Carrera, J.; Suárez-Ojeda, M.E. Improvement of the Polyhydroxyalkanoates Recovery from Mixed Microbial Cultures Using Sodium Hypochlorite Pre-Treatment Coupled with Solvent Extraction. Polymers 2022, 14, 3938. [Google Scholar] [CrossRef] [PubMed]
- Aramvash, A.; Gholami-Banadkuki, N.; Moazzeni-Zavareh, F.; Hajizadeh-Turchi, S. An Environmentally friendly and efficient method for extraction of PHB biopolymer with non-halogenated solvents. J. Microbiol. Biotechnol. 2015, 25, 1936–1943. [Google Scholar] [CrossRef] [PubMed]
- Aramvash, A.; Moazzeni Zavareh, F.; GholaM Banadkuki, N. Comparison of different solvents for extraction of polyhydroxybutyrate from Cupriavidus necator. Eng. Life Sci. 2017, 18, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Yabueng, N.; Napathorn, S.C. Toward non-toxic and simple recovery process of poly(3-hydroxybutyrate) using the green solvent 1,3-dioxolane. Process. Biochem. 2018, 69, 197–207. [Google Scholar] [CrossRef]
- Moscoso, R.; Carbajo, J.; Squella, J. 1,3-Dioxolane: A green solvent for the preparation of carbon nanotube-modified electrodes. Electrochem. Commun. 2014, 48, 69–72. [Google Scholar] [CrossRef]
- Ramsay, J.A.; Berger, E.; Voyer, R.; Chavarie, C.; Ramsay, B.A. Extraction of PHB using chlorinated solvents. Biotechnol. Tech. 1994, 8, 589–594. [Google Scholar] [CrossRef]
- Alder, C.M.; Hayler, J.D.; Henderson, R.K.; Redman, A.M.; Shukla, L.; Shuster, L.E.; Sneddon, H.F. Updating and further expanding GSK’s solvent sustainability guide. Green Chem. 2016, 18, 3879–3890. [Google Scholar] [CrossRef]
- Koller, M. Established and advanced approaches for recovery of microbial polyhydroxyalkanoate (PHA) biopolyesters from surrounding microbial biomass. EuroBiotech J. 2020, 4, 113–126. [Google Scholar] [CrossRef]
- Jacquel, N.; Lo, C.-W.; Wei, Y.-H.; Wu, H.-S.; Wang, S.S. Isolation and purification of bacterial poly(3-hydroxyalkanoates). Biochem. Eng. J. 2008, 39, 15–27. [Google Scholar] [CrossRef]
- Samorì, C.; Abbondanzi, F.; Galletti, P.; Giorgini, L.; Mazzocchetti, L.; Torri, C.; Tagliavini, E. Extraction of polyhydroxyalkanoates from mixed microbial cultures: Impact on polymer quality and recovery. Bioresour. Technol. 2015, 189, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Pérez, R.; Casal, J.; Muñoz, R.; Lebrero, R. Polyhydroxyalkanoates production from methane emissions in Sphagnum mosses: Assessing the effect of temperature and phosphorus limitation. Sci. Total. Environ. 2019, 688, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, Y.; Firmino, P.I.M.; Pérez, V.; Lebrero, R.; Muñoz, R. Biogas valorization via continuous polyhydroxybutyrate production by Methylocystis hirsuta in a bubble column bioreactor. Waste Manag. 2020, 113, 395–403. [Google Scholar] [CrossRef] [PubMed]
- López, J.C.; Arnáiz, E.; Merchán, L.; Lebrero, R.; Muñoz, R. Biogas-based polyhydroxyalkanoates production by Methylocystis hirsuta: A step further in anaerobic digestion biorefineries. Chem. Eng. J. 2018, 333, 529–536. [Google Scholar] [CrossRef]
- Fei, T.; Cazeneuve, S.; Wen, Z.; Wu, L.; Wang, T. Effective recovery of poly-β-hydroxybutyrate (PHB) biopolymer from Cupriavidus necator using a novel and environmentally friendly solvent system. Biotechnol. Prog. 2016, 32, 678–685. [Google Scholar] [CrossRef]
- Braunegg, G.; Sonnleitner, B.; Lafferty, R.M. A Rapid Gas Chromatographic Method for the Determination of Poly-?-Hydroxybutyric Acid in Microbial Biomass. Appl. Microbiol. Biotechnol. 1978, 6, 29–37. [Google Scholar] [CrossRef]
- Zúñiga, C.; Morales, M.; Le Borgne, S.; Revah, S. Production of poly-β-hydroxybutyrate (PHB) by Methylobacterium organophilum isolated from a methanotrophic consortium in a two-phase partition bioreactor. J. Hazard. Mater. 2011, 190, 876–882. [Google Scholar] [CrossRef]
- Rodríguez, Y.; Firmino, P.I.M.; Arnáiz, E.; Lebrero, R.; Muñoz, R. Elucidating the influence of environmental factors on biogas-based polyhydroxybutyrate production by Methylocystis hirsuta CSC. Sci. Total. Environ. 2020, 706, 135136. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Waste Water, 22nd ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2012. [Google Scholar]
- Road, S.; Lincolnshire, N. Safety Data Sheet, 1,3-Dioxolane SOLVAGREEN ≥90% for Synthesis; Replaces Version of: 22 April 2022, Date of Compilation: 14 June 2017; Carl Roth GmbH: Karlsruhe, Germany, 2024. [Google Scholar]
- Filippi, S.; Cinelli, P.; Mezzetta, A.; Carlozzi, P.; Seggiani, M. Extraction of polyhydroxyalkanoates from purple non-sulfur bacteria by non-chlorinated solvents. Polymers 2021, 13, 4163. [Google Scholar] [CrossRef] [PubMed]
- Kurian, N.S.; Das, B. Comparative analysis of various extraction processes based on economy, eco-friendly, purity and recovery of polyhydroxyalkanoate: A review. Int. J. Biol. Macromol. 2021, 183, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Alfano, S.; Lorini, L.; Majone, M.; Sciubba, F.; Valentino, F.; Martinelli, A. Ethylic esters as green solvents for the extraction of intracellular polyhydroxyalkanoates produced by mixed microbial culture. Polymers 2021, 13, 2789. [Google Scholar] [CrossRef] [PubMed]
- Fiorese, M.L.; Freitas, F.; Pais, J.; Ramos, A.M.; De Aragã£O, G.M.F.; Reis, M.A.M. Recovery of polyhydroxybutyrate (PHB) from Cupriavidus necator biomass by solvent extraction with 1,2-propylene carbonate. Eng. Life Sci. 2009, 9, 454–461. [Google Scholar] [CrossRef]
- Lafferty, R.M.; Heinzle, E. Use of Cyclic Carbonic Acid Esters as Solvents for Poly-(β-hydroxybutyric acid). US4101533A, 18 July 1978. [Google Scholar]
- Mohammadi, M.; Hassan, M.A.; Phang, L.-Y.; Ariffin, H.; Shirai, Y.; Ando, Y. Recovery and purification of intracellular polyhydroxyalkanoates from recombinant Cupriavidus necator using water and ethanol. Biotechnol. Lett. 2012, 34, 253–259. [Google Scholar] [CrossRef]
- Mohammadi, M.; Hassan, M.A.; Shirai, Y.; Man, H.C.; Ariffin, H.; Yee, L.-N.; Mumtaz, T.; Chong, M.-L.; Phang, L.-Y. Separation and Purification of Polyhydroxyalkanoates from Newly Isolated Comamonas sp. EB172 by Simple Digestion with Sodium Hydroxide. Sep. Sci. Technol. 2012, 47, 534–541. [Google Scholar] [CrossRef]
- Narasimhan, K.; Yee, K.; Cearley, A.C.; Levengood, D.; Chen, G.-Q. Solvent Extraction of Polyhydroxyalkanoates from Biomass. US Patent 7226765, 5 June 2007. [Google Scholar]


| Polymer | PHB | PHB-co-HV | ||
|---|---|---|---|---|
| Biomass | M. hirsuta | Mixed Consortium | M. hirsuta | Mixed Consortium |
| Content of PHAs [% w w−1] | 62 | 48 | 60 | 35 |
| R [%] | 87 ± 1 | 88.4 ± 3 | 75 ± 4 | 70 ± 4.5 |
| P [%] | 99 ± 0.9 | 96 ± 2 | 94 ± 1 | 97 ± 2.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amabile, C.; Abate, T.; Chianese, S.; Musmarra, D.; Muñoz, R. Exploring 1,3-Dioxolane Extraction of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from Methylocystis hirsuta and Mixed Methanotrophic Strain: Effect of Biomass-to-Solvent Ratio and Extraction Time. Polymers 2024, 16, 1910. https://doi.org/10.3390/polym16131910
Amabile C, Abate T, Chianese S, Musmarra D, Muñoz R. Exploring 1,3-Dioxolane Extraction of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from Methylocystis hirsuta and Mixed Methanotrophic Strain: Effect of Biomass-to-Solvent Ratio and Extraction Time. Polymers. 2024; 16(13):1910. https://doi.org/10.3390/polym16131910
Chicago/Turabian StyleAmabile, Claudia, Teresa Abate, Simeone Chianese, Dino Musmarra, and Raul Muñoz. 2024. "Exploring 1,3-Dioxolane Extraction of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from Methylocystis hirsuta and Mixed Methanotrophic Strain: Effect of Biomass-to-Solvent Ratio and Extraction Time" Polymers 16, no. 13: 1910. https://doi.org/10.3390/polym16131910
APA StyleAmabile, C., Abate, T., Chianese, S., Musmarra, D., & Muñoz, R. (2024). Exploring 1,3-Dioxolane Extraction of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from Methylocystis hirsuta and Mixed Methanotrophic Strain: Effect of Biomass-to-Solvent Ratio and Extraction Time. Polymers, 16(13), 1910. https://doi.org/10.3390/polym16131910








