Microneedles Based on a Biodegradable Polymer—Hyaluronic Acid
Abstract
1. Introduction
2. Microneedles
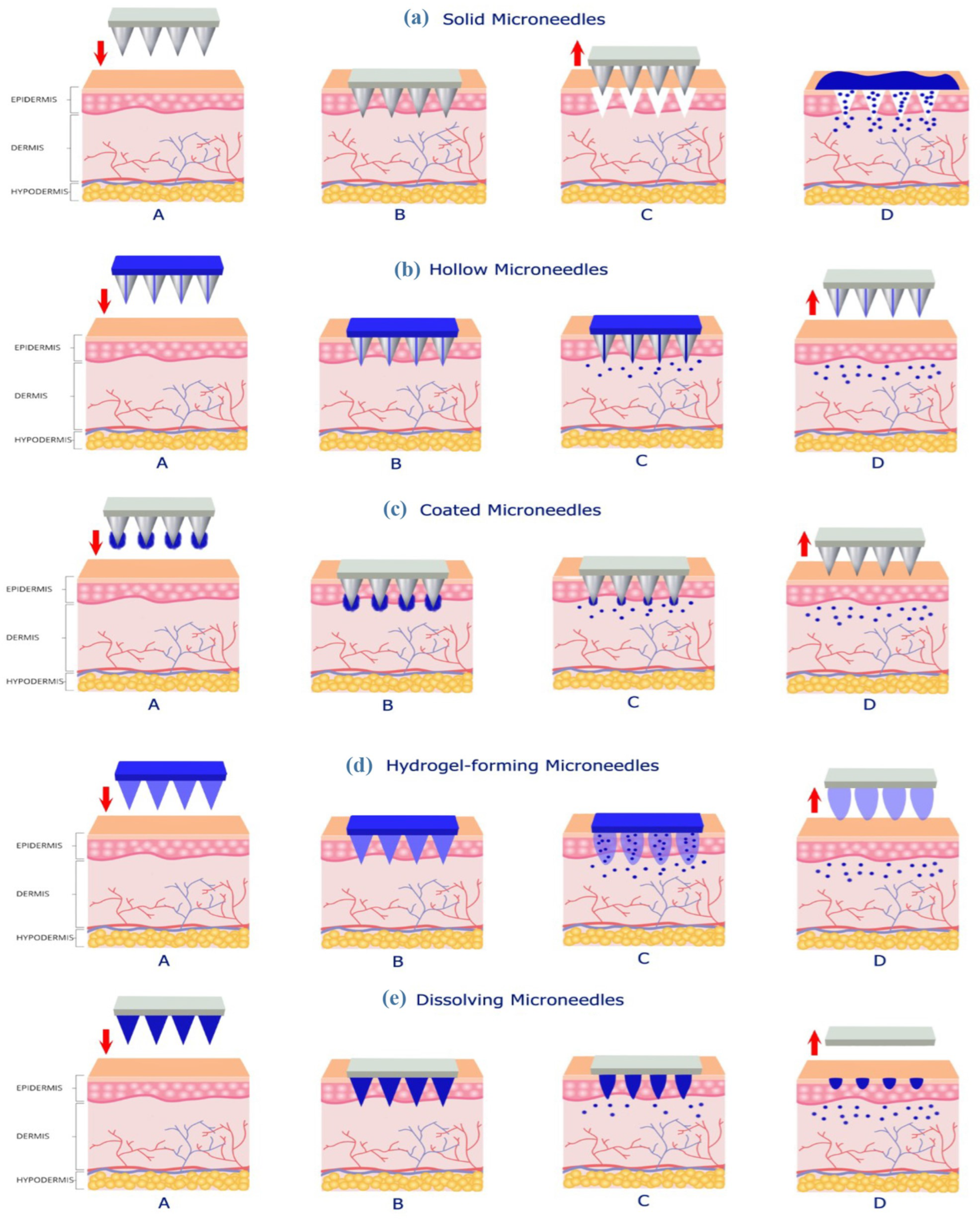
2.1. Types of Microneedles
2.1.1. Solid MNs
2.1.2. Hollow MNs
2.1.3. Coated MNs
2.1.4. Hydrogel-Forming MNs
2.1.5. Dissolving MNs
2.2. Shapes of Microneedles
- matrix—a flat element that carries the needles and remains on the surface of the skin;
- microneedles—protruding elements (protrusions), which, depending on the application, can have a length up to 2000 μm, diameter up to 250 μm, and a tip thickness of 1–25 μm [19].
2.3. Materials for Microneedles Preparation
2.3.1. Metal
2.3.2. Ceramic
2.3.3. Silicon and Glass
2.3.4. Polymer
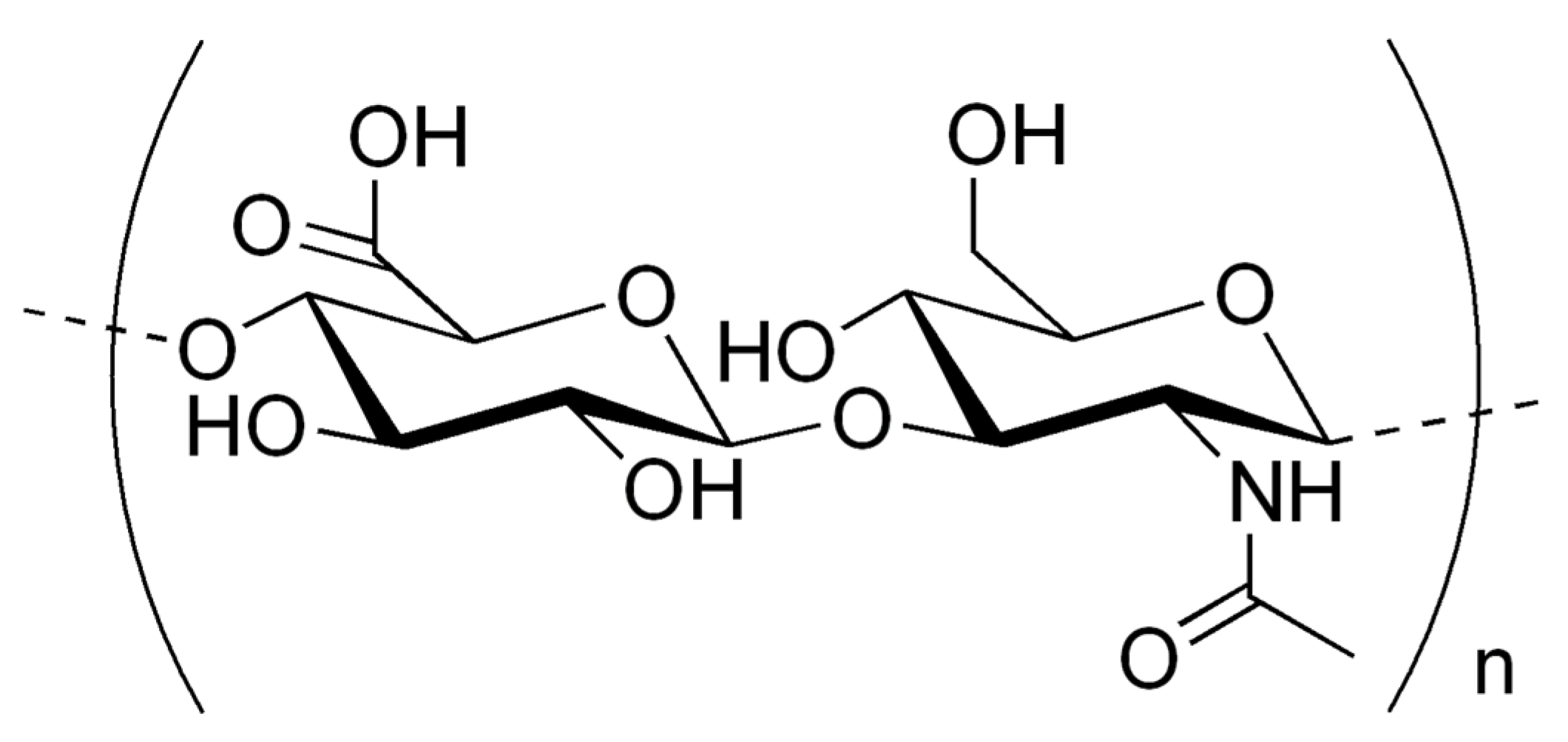
3. Hyaluronic Acid
- low molecular weight hyaluronic acid (LMW-HA) 0.5–1.5 × 106 Da;
- medium molecular hyaluronic acid (MMW-HA) 1.2–6 × 106 Da;
- high molecular weight hyaluronic acid (HMW-HA) 3–7 × 106 Da.
4. Fabrication of Hyaluronic Acid-Based Microneedles
4.1. Micro-Molding
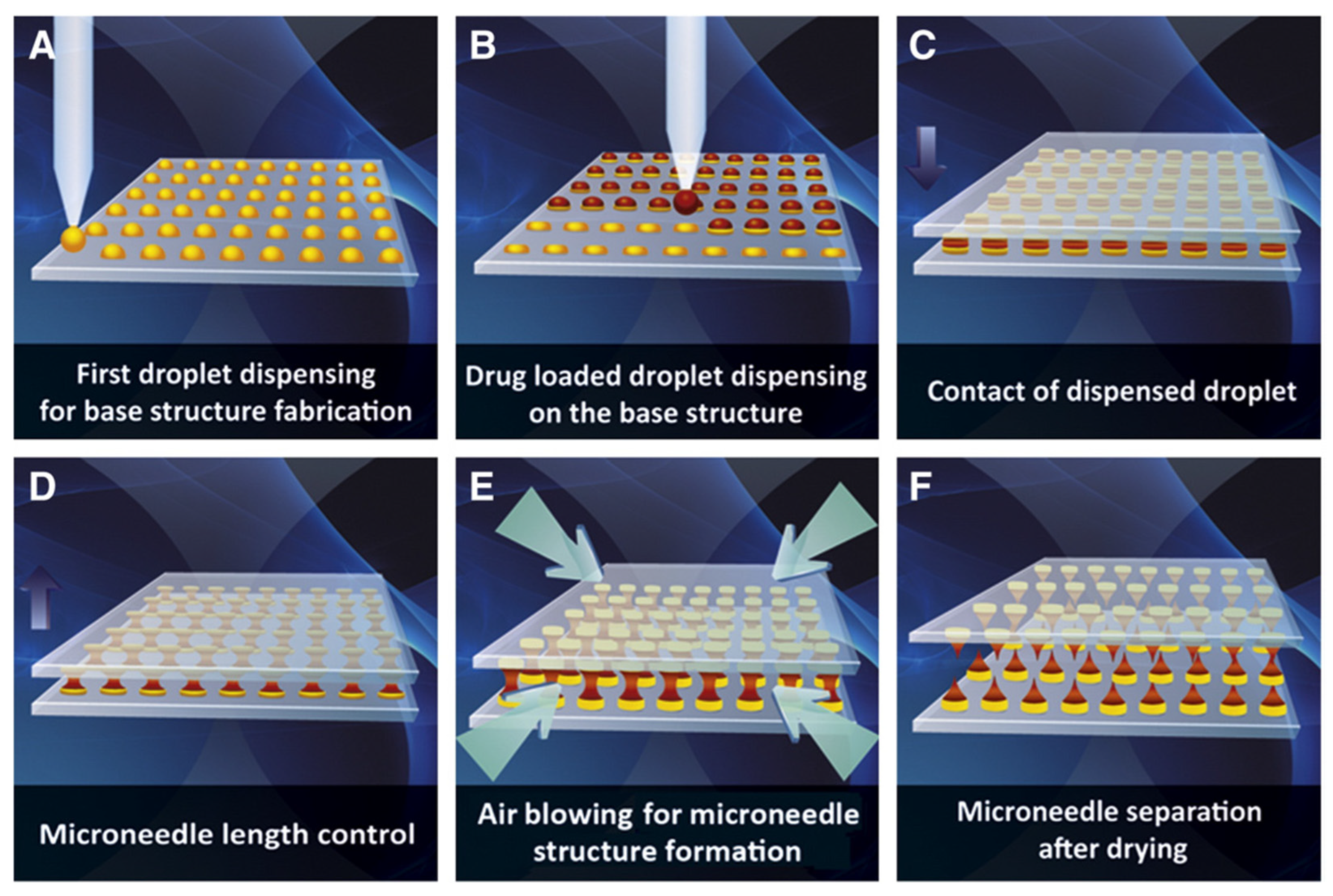
4.2. Droplet-Born Air Blowing
- applying single drops of a polymer solution to a flat wafer surface;
- dispersing droplets loaded with an active substance across the wafer surface;
- overlapping of two plates—the point of contact is the previously applied drop;
- controlled upward movement of the upper plate, which allows adjustment of the length of the microneedle;
- air blowing, which solidifies the droplets formed earlier;
- plate separation, which allows the recovery of the microneedles formed.
4.3. Photolithography
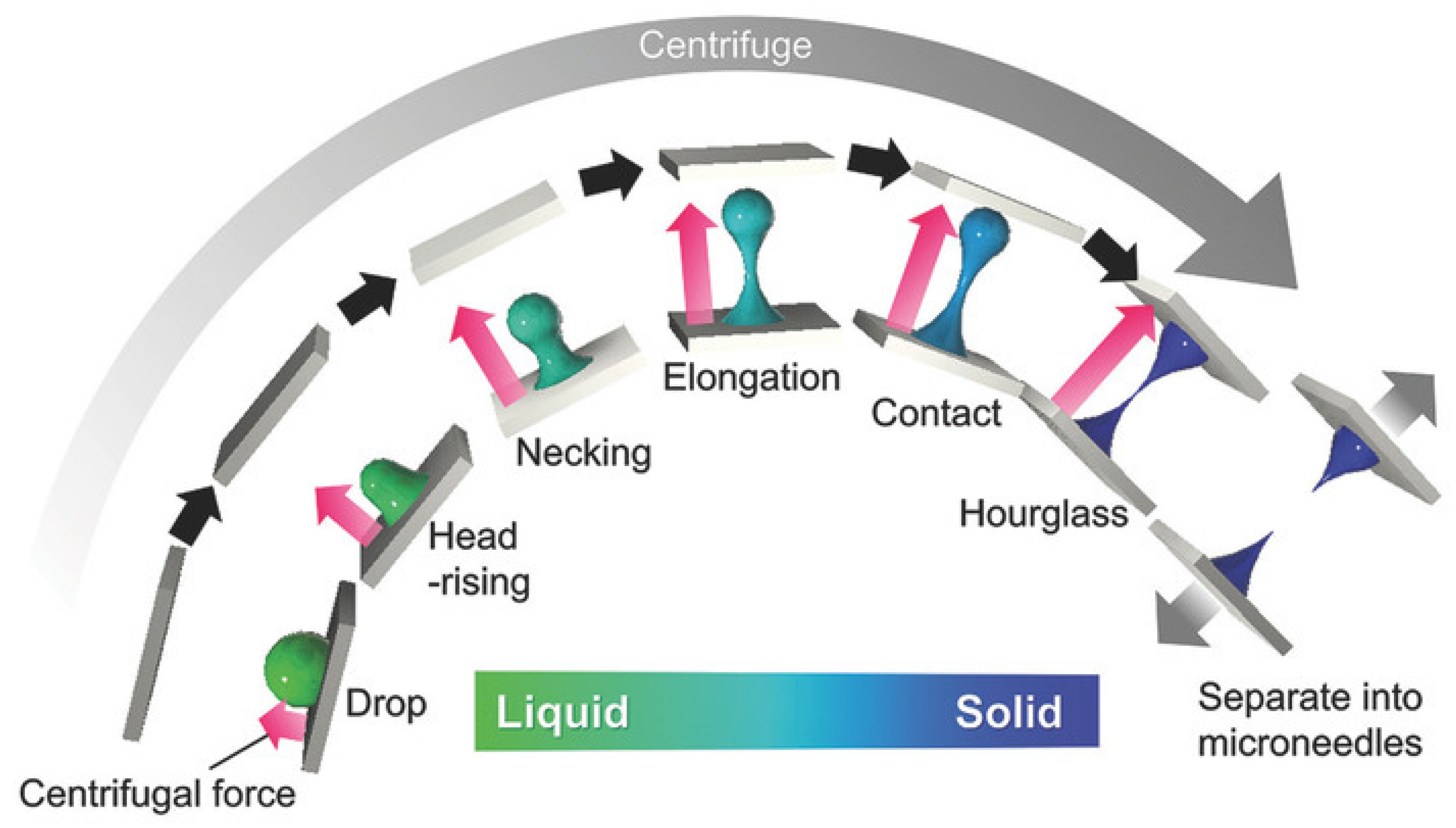
4.4. Centrifugal Lithography
4.5. Three-Dimensional Printing/Additive Manufacturing
- stereolithography (SLA);
- selective laser sintering (SLS);
- digital light processing (DLP);
- fused deposition modeling (FDM);
- two-photon polymerization (2PP);
- continuous liquid interface production (CLIP).
5. Future Directions
6. Conclusions
Funding
Conflicts of Interest
References
- Schafer, N.; Balwierz, R.; Biernat, P.; Ochędzan-Siodłak, W.; Lipok, J. Natural Ingredients of Transdermal Drug Delivery Systems as Permeation Enhancers of Active Substances through the Stratum Corneum. Mol. Pharm. 2023, 20, 3278–3297. [Google Scholar] [CrossRef] [PubMed]
- Noreen, S.; Ma, J.-X.; Saeed, M.; Pervaiz, F.; Hanif, M.F.; Ahmed, B.; Farooq, M.I.; Akram, F.; Safdar, M.; Madni, A.; et al. Natural Polysaccharide-Based Biodegradable Polymeric Platforms for Transdermal Drug Delivery System: A Critical Analysis. Drug Deliv. Transl. Res. 2022, 12, 2649–2666. [Google Scholar] [CrossRef] [PubMed]
- Zerbinati, N.; Esposito, C.; Cipolla, G.; Calligaro, A.; Monticelli, D.; Martina, V.; Golubovic, M.; Binic, I.; Sigova, J.; Gallo, A.L.; et al. Chemical and Mechanical Characterization of Hyaluronic Acid Hydrogel Cross-linked with Polyethylen Glycol and Its Use in Dermatology. Dermatol. Ther. 2020, 33, e13747. [Google Scholar] [CrossRef] [PubMed]
- Koyani, R.D. Synthetic Polymers for Microneedle Synthesis: From Then to Now. J. Drug Deliv. Sci. Technol. 2020, 60, 102071. [Google Scholar] [CrossRef]
- Aldawood, F.K.; Andar, A.; Desai, S. A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications. Polymers 2021, 13, 2815. [Google Scholar] [CrossRef] [PubMed]
- Sartawi, Z.; Blackshields, C.; Faisal, W. Dissolving Microneedles: Applications and Growing Therapeutic Potential. J. Control. Release 2022, 348, 186–205. [Google Scholar] [CrossRef] [PubMed]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A Smart Approach and Increasing Potential for Transdermal Drug Delivery System. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Alimardani, V.; Abolmaali, S.S.; Yousefi, G.; Rahiminezhad, Z.; Abedi, M.; Tamaddon, A.; Ahadian, S. Microneedle Arrays Combined with Nanomedicine Approaches for Transdermal Delivery of Therapeutics. J. Clin. Med. 2021, 10, 181. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.Z.; He, M.C.; Zhang, X.P.; Fei, W.M.; Cui, Y.; Guo, X.D. A Novel Method for Fabrication of Coated Microneedles with Homogeneous and Controllable Drug Dosage for Transdermal Drug Delivery. Drug Deliv. Transl. Res. 2022, 12, 2730–2739. [Google Scholar] [CrossRef]
- Ingrole, R.S.J.; Gill, H.S. Microneedle Coating Methods: A Review with a Perspective. J. Pharmacol. Exp. Ther. 2019, 370, 555–569. [Google Scholar] [CrossRef]
- Bhatnagar, S.; Gadeela, P.R.; Thathireddy, P.; Venuganti, V.V.K. Microneedle-Based Drug Delivery: Materials of Construction. J. Chem. Sci. 2019, 131, 90. [Google Scholar] [CrossRef]
- Li, S.; Li, W.; Prausnitz, M. Individually Coated Microneedles for Co-Delivery of Multiple Compounds with Different Properties. Drug Deliv. Transl. Res. 2018, 8, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.G.; White, L.R.; Estrela, P.; Leese, H.S. Hydrogel-Forming Microneedles: Current Advancements and Future Trends. Macromol. Biosci. 2021, 21, 2000307. [Google Scholar] [CrossRef] [PubMed]
- Courtenay, A.J.; McAlister, E.; McCrudden, M.T.C.; Vora, L.; Steiner, L.; Levin, G.; Levy-Nissenbaum, E.; Shterman, N.; Kearney, M.-C.; McCarthy, H.O.; et al. Hydrogel-Forming Microneedle Arrays as a Therapeutic Option for Transdermal Esketamine Delivery. J. Control. Release 2020, 322, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhao, J.; Fan, D. The Progress in the Application of Dissolving Microneedles in Biomedicine. Polymers 2023, 15, 4059. [Google Scholar] [CrossRef] [PubMed]
- Vora, L.K.; Moffatt, K.; Tekko, I.A.; Paredes, A.J.; Volpe-Zanutto, F.; Mishra, D.; Peng, K.; Raj Singh Thakur, R.; Donnelly, R.F. Microneedle Array Systems for Long-Acting Drug Delivery. Eur. J. Pharm. Biopharm. 2021, 159, 44–76. [Google Scholar] [CrossRef] [PubMed]
- Jamaledin, R.; Di Natale, C.; Onesto, V.; Taraghdari, Z.; Zare, E.; Makvandi, P.; Vecchione, R.; Netti, P. Progress in Microneedle-Mediated Protein Delivery. J. Clin. Med. 2020, 9, 542. [Google Scholar] [CrossRef] [PubMed]
- Dalvi, M.; Kharat, P.; Thakor, P.; Bhavana, V.; Singh, S.B.; Mehra, N.K. Panorama of Dissolving Microneedles for Transdermal Drug Delivery. Life Sci. 2021, 284, 119877. [Google Scholar] [CrossRef] [PubMed]
- Bhadale, R.S.; Londhe, V.Y. A Systematic Review of Carbohydrate-Based Microneedles: Current Status and Future Prospects. J. Mater. Sci. Mater. Med. 2021, 32, 89. [Google Scholar] [CrossRef]
- Cárcamo-Martínez, Á.; Mallon, B.; Domínguez-Robles, J.; Vora, L.K.; Anjani, Q.K.; Donnelly, R.F. Hollow Microneedles: A Perspective in Biomedical Applications. Int. J. Pharm. 2021, 599, 120455. [Google Scholar] [CrossRef]
- Min, H.S.; Kim, Y.; Nam, J.; Ahn, H.; Kim, M.; Kang, G.; Jang, M.; Yang, H.; Jung, H. Shape of Dissolving Microneedles Determines Skin Penetration Ability and Efficacy of Drug Delivery. Biomater. Adv. 2023, 145, 213248. [Google Scholar] [CrossRef] [PubMed]
- De Martino, S.; Battisti, M.; Napolitano, F.; Palladino, A.; Serpico, L.; Amendola, E.; Martone, A.; De Girolamo, P.; Squillace, A.; Dardano, P.; et al. Effect of Microneedles Shape on Skin Penetration and Transdermal Drug Administration. Biomater. Adv. 2022, 142, 213169. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, R.; Yuan, X.; Yang, L.; Luo, J.; Jiang, X.; Gou, Z.; Li, B.; Jiang, X.; Gou, M. Fast Customization of Microneedle Arrays by Static Optical Projection Lithography. ACS Appl. Mater. Interfaces 2021, 13, 60522–60530. [Google Scholar] [CrossRef] [PubMed]
- Faraji Rad, Z.; Prewett, P.D.; Davies, G.J. An Overview of Microneedle Applications, Materials, and Fabrication Methods. Beilstein J. Nanotechnol. 2021, 12, 1034–1046. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Yang, L.; Cui, Y. Microneedles: Materials, Fabrication, and Biomedical Applications. Biomed. Microdevices 2023, 25, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.P.; He, Y.T.; Li, W.X.; Chen, B.Z.; Zhang, C.Y.; Cui, Y.; Guo, X.D. An Update on Biomaterials as Microneedle Matrixes for Biomedical Applications. J. Mater. Chem. B 2022, 10, 6059–6077. [Google Scholar] [CrossRef] [PubMed]
- Howells, O.; Blayney, G.J.; Gualeni, B.; Birchall, J.C.; Eng, P.F.; Ashraf, H.; Sharma, S.; Guy, O.J. Design, Fabrication, and Characterisation of a Silicon Microneedle Array for Transdermal Therapeutic Delivery Using a Single Step Wet Etch Process. Eur. J. Pharm. Biopharm. 2022, 171, 19–28. [Google Scholar] [CrossRef]
- Ebrahiminejad, V.; Faraji Rad, Z.; Prewett, P.D.; Davies, G.J. Fabrication and Testing of Polymer Microneedles for Transdermal Drug Delivery. Beilstein J. Nanotechnol. 2022, 13, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Quan, G.; Sun, Y.; Yang, D.; Pan, X.; Wu, C. Nanoparticles-Encapsulated Polymeric Microneedles for Transdermal Drug Delivery. J. Control. Release 2020, 325, 163–175. [Google Scholar] [CrossRef]
- Azmana, M.; Mahmood, S.; Hilles, A.R.; Mandal, U.K.; Saeed Al-Japairai, K.A.; Raman, S. Transdermal Drug Delivery System through Polymeric Microneedle: A Recent Update. J. Drug Deliv. Sci. Technol. 2020, 60, 101877. [Google Scholar] [CrossRef]
- Yadav, P.R.; Munni, M.N.; Campbell, L.; Mostofa, G.; Dobson, L.; Shittu, M.; Pattanayek, S.K.; Uddin, M.J.; Das, D.B. Translation of Polymeric Microneedles for Treatment of Human Diseases: Recent Trends, Progress, and Challenges. Pharmaceutics 2021, 13, 1132. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.; Mehta, P.; Arshad, M.; Kucuk, I.; Chang, M.-W.; Ahmad, Z. Transdermal Microneedles—A Materials Perspective. AAPS PharmSciTech 2020, 21, 12. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; He, J.; Qi, J.; Zhu, Q.; Wu, W.; Lu, Y. Long-Acting Microneedles: A Progress Report of the State-of-the-Art Techniques. Drug Discov. Today 2020, 25, 1462–1468. [Google Scholar] [CrossRef] [PubMed]
- Malek-Khatabi, A.; Sadat Razavi, M.; Abdollahi, A.; Rahimzadeghan, M.; Moammeri, F.; Sheikhi, M.; Tavakoli, M.; Rad-Malekshahi, M.; Faraji Rad, Z. Recent Progress in PLGA-Based Microneedle-Mediated Transdermal Drug and Vaccine Delivery. Biomater. Sci. 2023, 11, 5390–5409. [Google Scholar] [CrossRef] [PubMed]
- Khosraviboroujeni, A.; Mirdamadian, S.Z.; Minaiyan, M.; Taheri, A. Preparation and Characterization of 3D Printed PLA Microneedle Arrays for Prolonged Transdermal Drug Delivery of Estradiol Valerate. Drug Deliv. Transl. Res. 2022, 12, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- George, A.; Sanjay, M.R.; Srisuk, R.; Parameswaranpillai, J.; Siengchin, S. A Comprehensive Review on Chemical Properties and Applications of Biopolymers and Their Composites. Int. J. Biol. Macromol. 2020, 154, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Güngör, S.; Kahraman, E.; Erdal, M.S.; Özsoy, Y. Recent Advances in Biopolymer-Based Transdermal Patches. In Biopolymer Membranes and Films; Elsevier: Amsterdam, The Netherlands, 2020; pp. 195–217. [Google Scholar]
- Mohammed, A.S.A.; Naveed, M.; Jost, N. Polysaccharides; Classification, Chemical Properties, and Future Perspective Applications in Fields of Pharmacology and Biological Medicine (A Review of Current Applications and Upcoming Potentialities). J. Polym. Environ. 2021, 29, 2359–2371. [Google Scholar] [CrossRef]
- Su, L.; Feng, Y.; Wei, K.; Xu, X.; Liu, R.; Chen, G. Carbohydrate-Based Macromolecular Biomaterials. Chem. Rev. 2021, 121, 10950–11029. [Google Scholar] [CrossRef] [PubMed]
- Abatangelo, G.; Vindigni, V.; Avruscio, G.; Pandis, L.; Brun, P. Hyaluronic Acid: Redefining Its Role. Cells 2020, 9, 1743. [Google Scholar] [CrossRef]
- Wawrzyńczak, A.; Nowak, I.; Woźniak, N.; Chudzińska, J.; Feliczak-Guzik, A. Synthesis and Characterization of Hierarchical Zeolites Modified with Polysaccharides and Its Potential Role as a Platform for Drug Delivery. Pharmaceutics 2023, 15, 535. [Google Scholar] [CrossRef]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 458280. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hu, L.; Huang, H.; Wang, H.; Zhang, T.; Chen, J.; Du, G.; Kang, Z. Eliminating the Capsule-like Layer to Promote Glucose Uptake for Hyaluronan Production by Engineered Corynebacterium Glutamicum. Nat. Commun. 2020, 11, 3120. [Google Scholar] [CrossRef] [PubMed]
- Iturriaga, V.; Vásquez, B.; Bornhardt, T.; del Sol, M. Effects of Low and High Molecular Weight Hyaluronic Acid on the Osteoarthritic Temporomandibular Joint in Rabbit. Clin. Oral Investig. 2021, 25, 4507–4518. [Google Scholar] [CrossRef] [PubMed]
- Hummer, C.D.; Angst, F.; Ngai, W.; Whittington, C.; Yoon, S.S.; Duarte, L.; Manitt, C.; Schemitsch, E. High Molecular Weight Intraarticular Hyaluronic Acid for the Treatment of Knee Osteoarthritis: A Network Meta-Analysis. BMC Musculoskelet. Disord. 2020, 21, 702. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Ma, Y.; Huang, Y.; Li, S.; Xu, H.; Su, E. Current Advances in the Biosynthesis of Hyaluronic Acid with Variable Molecular Weights. Carbohydr. Polym. 2021, 269, 118320. [Google Scholar] [CrossRef] [PubMed]
- Snetkov, P.; Zakharova, K.; Morozkina, S.; Olekhnovich, R.; Uspenskaya, M. Hyaluronic Acid: The Influence of Molecular Weight on Structural, Physical, Physico-Chemical, and Degradable Properties of Biopolymer. Polymers 2020, 12, 1800. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Zuo, Z.; Lin, R.; Yao, M.; Han, Y.; Han, J. The Most Promising Microneedle Device: Present and Future of Hyaluronic Acid Microneedle Patch. Drug Deliv. 2022, 29, 3087–3110. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.-W.; Wang, Z.-Y.; Ren, Z.-W.; Zhang, X.-W.; Wei, D.-X. Advances in Modified Hyaluronic Acid-Based Hydrogels for Skin Wound Healing. Biomater. Sci. 2022, 10, 3393–3409. [Google Scholar] [CrossRef] [PubMed]
- Saha, I.; Rai, V.K. Hyaluronic Acid Based Microneedle Array: Recent Applications in Drug Delivery and Cosmetology. Carbohydr. Polym. 2021, 267, 118168. [Google Scholar] [CrossRef]
- Damiri, F.; Kommineni, N.; Ebhodaghe, S.O.; Bulusu, R.; Jyothi, V.G.S.S.; Sayed, A.A.; Awaji, A.A.; Germoush, M.O.; Al-Malky, H.S.; Nasrullah, M.Z.; et al. Microneedle-Based Natural Polysaccharide for Drug Delivery Systems (DDS): Progress and Challenges. Pharmaceuticals 2022, 15, 190. [Google Scholar] [CrossRef]
- Kenchegowda, M.; Hani, U.; Al Fatease, A.; Haider, N.; Ramesh, K.V.R.N.S.; Talath, S.; Gangadharappa, H.V.; Kiran Raj, G.; Padmanabha, S.H.; Osmani, R.A.M. Tiny Titans-Unravelling the Potential of Polysaccharides and Proteins Based Dissolving Microneedles in Drug Delivery and Theranostics: A Comprehensive Review. Int. J. Biol. Macromol. 2023, 253, 127172. [Google Scholar] [CrossRef] [PubMed]
- Baruffi, F.; Gülçür, M.; Calaon, M.; Romano, J.-M.; Penchev, P.; Dimov, S.; Whiteside, B.; Tosello, G. Correlating Nano-Scale Surface Replication Accuracy and Cavity Temperature in Micro-Injection Moulding Using in-Line Process Control and High-Speed Thermal Imaging. J. Manuf. Process 2019, 47, 367–381. [Google Scholar] [CrossRef]
- Romano, J.-M.; Gulcur, M.; Garcia-Giron, A.; Martinez-Solanas, E.; Whiteside, B.R.; Dimov, S.S. Mechanical Durability of Hydrophobic Surfaces Fabricated by Injection Moulding of Laser-Induced Textures. Appl. Surf. Sci. 2019, 476, 850–860. [Google Scholar] [CrossRef]
- Smith, E.; Lau, W.M.; Abdelghany, T.M.; Vukajlovic, D.; Novakovic, K.; Ng, K.W. Vac-and-Fill: A Micromoulding Technique for Fabricating Microneedle Arrays with Vacuum-Activated, Hands-Free Mould-Filling. Int. J. Pharm. 2024, 650, 123706. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Jin, S.G. Microneedle for Transdermal Drug Delivery: Current Trends and Fabrication. J. Pharm. Investig. 2021, 51, 503–517. [Google Scholar] [CrossRef]
- Moore, L.E.; Vucen, S.; Moore, A.C. Trends in Drug- and Vaccine-Based Dissolvable Microneedle Materials and Methods of Fabrication. Eur. J. Pharm. Biopharm. 2022, 173, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, X.; Zhang, P.; Du, J.; Wang, Y. Tip-loaded fast-dissolving MN patches for photodynamic therapy of subcutaneous tumor. J. Control. Release 2018, 286, 201–209. [Google Scholar] [CrossRef]
- Zeng, Y.; Gao, Y.; He, L.; Ge, W.; Liu, J.; Yu, Y.; Xie, X. Multifunctional Polysaccharide Composited Microneedle for Oral Ulcers Healing. Mater. Today Bio 2023, 22, 100782. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Li, X.; Zhang, M.; Ling, G.; Zhang, P. Investigation of the Antibacterial Properties of Hyaluronic Acid Microneedles Based on Chitosan and MoS 2. J. Mater. Chem. B 2023, 11, 7169–7181. [Google Scholar] [CrossRef]
- Tay, J.H.; Lim, Y.H.; Zheng, M.; Zhao, Y.; Tan, W.S.; Xu, C.; Ramamurty, U.; Song, J. Development of Hyaluronic Acid-Silica Composites via in Situ Precipitation for Improved Penetration Efficiency in Fast-Dissolving Microneedle Systems. Acta Biomater. 2023, 172, 175–187. [Google Scholar] [CrossRef]
- Du, H.; Liu, P.; Zhu, J.; Lan, J.; Li, Y.; Zhang, L.; Zhu, J.; Tao, J. Hyaluronic Acid-Based Dissolving Microneedle Patch Loaded with Methotrexate for Improved Treatment of Psoriasis. ACS Appl. Mater. Interfaces 2019, 11, 43588–43598. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Gou, K.; Yue, X.; Zhao, S.; Zeng, R.; Qu, Y.; Zhang, C. A Novel Hyaluronic Acid-Based Dissolving Microneedle Patch Loaded with Ginsenoside Rg3 Liposome for Effectively Alleviate Psoriasis. Mater. Des. 2022, 224, 111363. [Google Scholar] [CrossRef]
- Cheng, Z.; Lin, H.; Wang, Z.; Yang, X.; Zhang, M.; Liu, X.; Wang, B.; Wu, Z.; Chen, D. Preparation and Characterization of Dissolving Hyaluronic Acid Composite Microneedles Loaded Micelles for Delivery of Curcumin. Drug Deliv. Transl. Res. 2020, 10, 1520–1530. [Google Scholar] [CrossRef]
- Lin, S.; Quan, G.; Hou, A.; Yang, P.; Peng, T.; Gu, Y.; Qin, W.; Liu, R.; Ma, X.; Pan, X.; et al. Strategy for Hypertrophic Scar Therapy: Improved Delivery of Triamcinolone Acetonide Using Mechanically Robust Tip-Concentrated Dissolving Microneedle Array. J. Control. Release 2019, 306, 69–82. [Google Scholar] [CrossRef]
- Ning, X.; Wiraja, C.; Chew, W.T.S.; Fan, C.; Xu, C. Transdermal Delivery of Chinese Herbal Medicine Extract Using Dissolvable Microneedles for Hypertrophic Scar Treatment. Acta Pharm. Sin. B 2021, 11, 2937–2944. [Google Scholar] [CrossRef]
- Champeau, M.; Jary, D.; Mortier, L.; Mordon, S.; Vignoud, S. A Facile Fabrication of Dissolving Microneedles Containing 5-Aminolevulinic Acid. Int. J. Pharm. 2020, 586, 119554. [Google Scholar] [CrossRef]
- Hao, Y.; Chen, Y.; He, X.; Yang, F.; Han, R.; Yang, C.; Li, W.; Qian, Z. Near-Infrared Responsive 5-Fluorouracil and Indocyanine Green Loaded MPEG-PCL Nanoparticle Integrated with Dissolvable Microneedle for Skin Cancer Therapy. Bioact. Mater. 2020, 5, 542–552. [Google Scholar] [CrossRef]
- Yang, H.; Wu, X.; Zhou, Z.; Chen, X.; Kong, M. Enhanced Transdermal Lymphatic Delivery of Doxorubicin via Hyaluronic Acid Based Transfersomes/Microneedle Complex for Tumor Metastasis Therapy. Int. J. Biol. Macromol. 2019, 125, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Lu, Z.; Shi, Y.; Du, Y.; Chen, X.; Kong, M. Systematic Comparisons of Dissolving and Swelling Hyaluronic Acid Microneedles in Transdermal Drug Delivery. Int. J. Biol. Macromol. 2021, 191, 783–791. [Google Scholar] [CrossRef]
- Xu, Q.; Li, X.; Zhang, P.; Wang, Y. Rapidly Dissolving Microneedle Patch for Synergistic Gene and Photothermal Therapy of Subcutaneous Tumor. J. Mater. Chem. B 2020, 8, 4331–4339. [Google Scholar] [CrossRef]
- Ouyang, M.; Wang, X.; Fu, Y.; Xie, G.; Du, S.; Li, Y.; Zhang, L.; Tao, J.; Zhu, J. Skin Optical Clearing Enabled by Dissolving Hyaluronic Acid Microneedle Patches. Int. J. Biol. Macromol. 2022, 220, 1188–1196. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Wang, W.; Weng, J.; Zhang, Z.; Yin, L.; Yang, Q.; Guo, F.; Wang, X.; Chen, F.; Yang, G. Dissolving Microneedles for Transdermal Delivery of Huperzine A for the Treatment of Alzheimer’s Disease. Drug Deliv. 2020, 27, 1147–1155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.P.; Wang, B.B.; Hu, L.F.; Fei, W.M.; Cui, Y.; Guo, X.D. Safety Evaluation of 3-Month Effects of Microneedle Patches Prepared from Hyaluronic Acid in Mice. Biochem. Eng. J. 2021, 176, 108157. [Google Scholar] [CrossRef]
- Kim, D.-S.; Choi, J.-T.; Kim, C.B.; Shin, Y.-R.; Park, P.; Kim, H.; Lee, J.M.; Park, J.-H. Microneedle Array Patch (MAP) Consisting of Crosslinked Hyaluronic Acid Nanoparticles for Processability and Sustained Release. Pharm. Res. 2020, 37, 50. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.; Huang, Y.; Kang, Y.; Dai, G.; Liu, Z.; Xu, K.; Zhong, W. The Effects of Molecular Weight of Hyaluronic Acid on Transdermal Delivery Efficiencies of Dissolving Microneedles. Eur. J. Pharm. Sci. 2022, 168, 106075. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Zheng, M.; Chew, S.W.T.; Xu, C. Advances in the Formulations of Microneedles for Manifold Biomedical Applications. Adv. Mater. Technol. 2020, 5, 1900552. [Google Scholar] [CrossRef]
- Chen, Z.; Ye, R.; Yang, J.; Lin, Y.; Lee, W.; Li, J.; Ren, L.; Liu, B.; Jiang, L. Rapidly Fabricated Microneedle Arrays Using Magnetorheological Drawing Lithography for Transdermal Drug Delivery. ACS Biomater. Sci. Eng. 2019, 5, 5506–5513. [Google Scholar] [CrossRef]
- Gera, A.K.; Burra, R.K. The Rise of Polymeric Microneedles: Recent Developments, Advances, Challenges, and Applications with Regard to Transdermal Drug Delivery. J. Funct. Biomater. 2022, 13, 81. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.D.; Kim, M.; Yang, H.; Lee, K.; Jung, H. Droplet-born air blowing: Novel dissolving microneedle fabrication. J. Control. Release 2013, 170, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Avcil, M.; Akman, G.; Klokkers, J.; Jeong, D.; Çelik, A. Efficacy of Bioactive Peptides Loaded on Hyaluronic Acid Microneedle Patches: A Monocentric Clinical Study. J. Cosmet. Dermatol. 2020, 19, 328–337. [Google Scholar] [CrossRef]
- Dharadhar, S.; Majumdar, A.; Dhoble, S.; Patravale, V. Microneedles for Transdermal Drug Delivery: A Systematic Review. Drug Dev. Ind. Pharm. 2019, 45, 188–201. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; You, X.; Chen, Z. Hollow Microneedles on a Paper Fabricated by Standard Photolithography for the Screening Test of Prediabetes. Sensors 2022, 22, 4253. [Google Scholar] [CrossRef] [PubMed]
- Dardano, P.; De Martino, S.; Battisti, M.; Miranda, B.; Rea, I.; De Stefano, L. One-Shot Fabrication of Polymeric Hollow Microneedles by Standard Photolithography. Polymers 2021, 13, 520. [Google Scholar] [CrossRef] [PubMed]
- Tucak, A.; Sirbubalo, M.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Muhamedagić, K.; Čekić, A.; Vranić, E. Microneedles: Characteristics, Materials, Production Methods and Commercial Development. Micromachines 2020, 11, 961. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Gu, S.; Zhang, Q.; Liu, T.; Liu, Z.; Kuang, T. A Comparison Study of Hyaluronic Acid Hydrogel Exquisite Micropatterns with Photolithography and Light-Cured Inkjet Printing Methods. e-Polymers 2022, 22, 332–341. [Google Scholar] [CrossRef]
- Zhang, Q.; Wei, X.; Ji, Y.; Yin, L.; Dong, Z.; Chen, F.; Zhong, M.; Shen, J.; Liu, Z.; Chang, L. Adjustable and Ultrafast Light-Cured Hyaluronic Acid Hydrogel: Promoting Biocompatibility and Cell Growth. J. Mater. Chem. B 2020, 8, 5441–5450. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, H.; Kang, K.; Cai, J.; Kang, L. Rapid Microneedle Fabrication by Heating and Photolithography. Int. J. Pharm. 2020, 575, 118992. [Google Scholar] [CrossRef] [PubMed]
- Juhng, S.; Song, J.; You, J.; Park, J.; Yang, H.; Jang, M.; Kang, G.; Shin, J.; Ko, H.W.; Jung, H. Fabrication of Liraglutide-Encapsulated Triple Layer Hyaluronic Acid Microneedles (TLMs) for the Treatment of Obesity. Lab. Chip 2023, 23, 2378–2388. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kim, S.; Kang, G.; Lahiji, S.F.; Jang, M.; Kim, Y.M.; Kim, J.; Cho, S.; Jung, H. Centrifugal Lithography: Self-Shaping of Polymer Microstructures Encapsulating Biopharmaceutics by Centrifuging Polymer Drops. Adv. Healthc. Mater. 2017, 6, 1700326. [Google Scholar] [CrossRef]
- Jang, M.; Baek, S.; Kang, G.; Yang, H.; Kim, S.; Jung, H. Dissolving Microneedle with High Molecular Weight Hyaluronic Acid to Improve Skin Wrinkles, Dermal Density and Elasticity. Int. J. Cosmet. Sci. 2020, 42, 302–309. [Google Scholar] [CrossRef]
- Johnson, A.R.; Procopio, A.T. Low Cost Additive Manufacturing of Microneedle Masters. 3D Print. Med. 2019, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Olowe, M.; Parupelli, S.K.; Desai, S. A Review of 3D-Printing of Microneedles. Pharmaceutics 2022, 14, 2693. [Google Scholar] [CrossRef] [PubMed]
- Detamornrat, U.; McAlister, E.; Hutton, A.R.J.; Larrañeta, E.; Donnelly, R.F. The Role of 3D Printing Technology in Microengineering of Microneedles. Small 2022, 18, 2106392. [Google Scholar] [CrossRef] [PubMed]
- Sirbubalo, M.; Tucak, A.; Muhamedagic, K.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Cekic, A.; Begic-Hajdarevic, D.; Cohodar Husic, M.; Dervišević, A.; et al. 3D Printing—A “Touch-Button” Approach to Manufacture Microneedles for Transdermal Drug Delivery. Pharmaceutics 2021, 13, 924. [Google Scholar] [CrossRef] [PubMed]
- Economidou, S.N.; Uddin, M.J.; Marques, M.J.; Douroumis, D.; Sow, W.T.; Li, H.; Reid, A.; Windmill, J.F.C.; Podoleanu, A. A Novel 3D Printed Hollow Microneedle Microelectromechanical System for Controlled, Personalized Transdermal Drug Delivery. Addit. Manuf. 2021, 38, 101815. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B.; Patel, V.; Shah, J. 3D Printing Technologies: Recent Development and Emerging Applications in Various Drug Delivery Systems. AAPS PharmSciTech 2020, 21, 220. [Google Scholar] [CrossRef] [PubMed]
- Quan, H.; Zhang, T.; Xu, H.; Luo, S.; Nie, J.; Zhu, X. Photo-Curing 3D Printing Technique and Its Challenges. Bioact. Mater. 2020, 5, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Plamadeala, C.; Gosain, S.R.; Hischen, F.; Buchroithner, B.; Puthukodan, S.; Jacak, J.; Bocchino, A.; Whelan, D.; O’Mahony, C.; Baumgartner, W.; et al. Bio-Inspired Microneedle Design for Efficient Drug/Vaccine Coating. Biomed. Microdevices 2020, 22, 8. [Google Scholar] [CrossRef]
- Dabbagh, S.R.; Sarabi, M.R.; Rahbarghazi, R.; Sokullu, E.; Yetisen, A.K.; Tasoglu, S. 3D-Printed Microneedles in Biomedical Applications. iScience 2021, 24, 102012. [Google Scholar] [CrossRef]
- Economidou, S.N.; Douroumis, D. 3D Printing as a Transformative Tool for Microneedle Systems: Recent Advances, Manufacturing Considerations and Market Potential. Adv. Drug Deliv. Rev. 2021, 173, 60–69. [Google Scholar] [CrossRef]
- Aldawood, F.K.; Parupelli, S.K.; Andar, A.; Desai, S. 3D Printing of Biodegradable Polymeric Microneedles for Transdermal Drug Delivery Applications. Pharmaceutics 2024, 16, 237. [Google Scholar] [CrossRef] [PubMed]
- Galarraga, J.H.; Dhand, A.P.; Enzmann, B.P.; Burdick, J.A. Synthesis, Characterization, and Digital Light Processing of a Hydrolytically Degradable Hyaluronic Acid Hydrogel. Biomacromolecules 2023, 24, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xiang, H.; Zhang, Q.; Miao, X. Polysaccharide-Based Transdermal Drug Delivery. Pharmaceuticals 2022, 15, 602. [Google Scholar] [CrossRef] [PubMed]
- Terashima, S.; Tatsukawa, C.; Takahashi, T.; Suzuki, M.; Aoyagi, S. Fabrication of Hyaluronic Acid Hollow Microneedle Array. Jpn. J. Appl. Phys. 2020, 59, SIIJ03. [Google Scholar] [CrossRef]
- Lv, J.; Zhao, J.; Li, X.; Ling, G.; Zhang, P. Preparation of a Novel Hyaluronic Acid-Based Separable Hydrogel Microneedle with Niacinamide to Treat Pigment Deposition Using Solvent-Free Solid-State Crosslinking Method. Eur. Polym. J. 2024, 210, 113003. [Google Scholar] [CrossRef]
- Huang, Y.; Lai, H.; Jiang, J.; Xu, X.; Zeng, Z.; Ren, L.; Liu, Q.; Chen, M.; Zhang, T.; Ding, X.; et al. PH-Activatable Oxidative Stress Amplifying Dissolving Microneedles for Combined Chemo-Photodynamic Therapy of Melanoma. Asian J. Pharm. Sci. 2022, 17, 679–696. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.L.; Vinayakumar, K.B.; Sillankorva, S. Polymeric Microneedles for Health Care Monitoring: An Emerging Trend. ACS Sensors 2024. [Google Scholar] [CrossRef]
- Zheng, H.; GhavamiNejad, A.; GhavamiNejad, P.; Samarikhalaj, M.; Giacca, A.; Poudineh, M. Hydrogel Microneedle-Assisted Assay Integrating Aptamer Probes and Fluorescence Detection for Reagentless Biomarker Quantification. ACS Sensors 2022, 7, 2387–2399. [Google Scholar] [CrossRef]
- Takeuchi, K.; Takama, N.; Kinoshita, R.; Okitsu, T.; Kim, B. Flexible and porous microneedles of PDMS for continuous glucose monitoring. Biomed. Microdevices 2020, 22, 79. [Google Scholar] [CrossRef]
- Biswas, A.A.; Dhondale, M.R.; Agrawal, A.K.; Serrano, D.R.; Mishra, B.; Kumar, D. Advancements in microneedle fabrication techniques: Artificial intelligence assisted 3D-printing technology. Drug Deliv. Transl. Res. 2024, 14, 1458–1479. [Google Scholar] [CrossRef]
- Jin, X.; Cai, A.; Xu, T.; Zhang, X. Artificial intelligence biosensors for continuous glucose monitoring. Interdiscip. Mater. 2023, 2, 290–307. [Google Scholar] [CrossRef]
- Luzuriaga, M.A.; Berry, D.R.; Reagan, J.C.; Smaldone, R.A.; Gassensmith, J.J. Biodegradable 3D printed polymer microneedles for transdermal drug delivery. Lab Chip 2018, 18, 1223–1230. [Google Scholar] [CrossRef] [PubMed]

| Source of HA | Molecular Weight [kDa] |
|---|---|
| Rooster combs | 1200 |
| Umbilical cord | 3400 |
| Bovine vitreous body | 770–1700 |
| Bacteria | 1000–4000 |
| Human joint fluid | 6000–7000 |
| Rheumatoid fluid | 3000–5000 |
| Sample Labeling | MNs Material | Active Substance | Mold Material | Synthesis Procedure | Potential Therapeutic Applications | Ref. |
|---|---|---|---|---|---|---|
| HA/HACC@DXMS&bFGF | Multifunctional dissolvable polysaccharide composite based on hyaluronic acid (HA) and hydroxypropyltrimethylammonium chitosan chloride (HACC) with dexamethasone (DXMS) and basic fibroblast growth factor (bFGF) | DXMS, bFGF | PDMS | Centrifuged at 12,000 rpm for 30 min | Oral ulcers healing | [59] |
| HA MNs containing CS-MoS2 | Soluble microneedles based on hyaluronic acid (HA) with a nanocomposite of chitosan and molybdenum sulfide (CS-MoS2) | Chitosan (CS)/molybdenum sulfide (MoS2) | Silicone/PDMS | 5 min under vacuum and centrifuged at 3000 rpm for 10 min, dried at room temperature for 24 h | Antibacterial | [60] |
| HA–Si MN | Fast-dissolving microneedles based on hyaluronic acid (HA) and in situ precipitated silica nanoparticles (Si) with insulin (pretest) | Insulin (pretest) | PDMS | Centrifuged (4000 rpm at 25 °C) for 10 min, dried at room temperature for 24 h | Diabetes | [61] |
| MTX-loaded HA-MN | Soluble microneedles based on hyaluronic acid (HA) loaded with methotrexate (MTX) | Methotrexate (MTX) | PDMS | Vacuum (∼0.08 MPa), dried at room temperature | Psoriasis | [62] |
| Rg3-MN | Soluble microneedles based on hyaluronic acid loaded with liposomes containing ginsenoside Rg3 | Ginsenoside Rg3 liposome | PDMS | Centrifuged in a swinging bucket rotor at 3500 g for 15 min at 25 °C | Psoriasis | [63] |
| Cur-loaded Que-DA-oHA MN | An amphiphilic polymer-based material composed of quercetin (Que), dithiodipropionic acid (DA), and oligomeric hyaluronic acid (oHA), containing curcumin-loaded micelles | Curcumin (Cur) | - | Frozen at −20 °C for 6 h and melted. The freezing-thawing process repeated three times | Skin diseases | [64] |
| TA-HA/HP-β-CD | Soluble microneedles based on hyaluronic acid (HA) with hydroxypropyl-β- cyclodextrin (HP-β CD) containing triamcinolone acetonide (TA) | Triamcinolone acetonide (TA) | PDMS | Centrifuged at 4000 rpm for 5 min at 2 °C, centrifuged at 4000 rpm for 60 min at 25 °C, centrifuged at 2000 rpm for another 3 min, dried in a desiccator at room temperature for 72 h | Hypertrophic scar therapy | [65] |
| Shikonin HA MN | Soluble microneedles based on hyaluronic acid loaded with shikonin | Shikonin (an active component extracted from the root of Lithospermum erythrorhizon) | PDMS | Centrifuged (3200× g, 5 min), dried in fume hood at room temperature | Hypertrophic scar therapy | [66] |
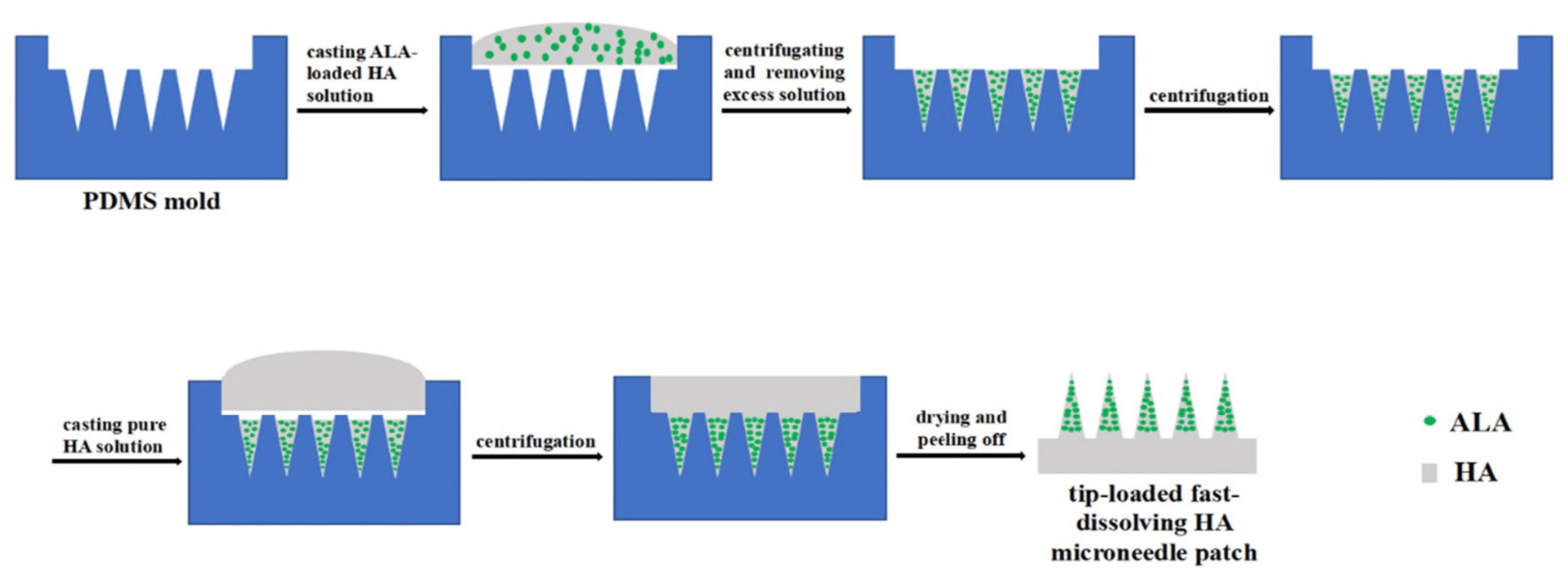
| 5-ALA-MN | Soluble microneedles based on hyaluronic acid with 5-aminolevulinic acid (5-ALA) | 5-aminolevulinic acid (5-ALA) | PDMS | Centrifuged at 4000 rpm for 35 min (21 °C), dried in an oven at 40 ± 1 °C for 24 h | Precancerous skin lesions | [67] |
| 5-Fu-ICGMPEG-PCL@HA MN | NIR-responsive hyaluronic acid-based microneedles integrated with monomethoxy-poly(ethylene glycol)-polycaprolactone (MPEG-PCL) nanoparticles loaded with 5-fluorouracil (5-Fu) and indocyanine green (ICG) | 5-fluorouracil (5-Fu), indocyanine green (ICG) | PDMS | Centrifuged, dried overnight in an oven at 45 °C | Skin cancer therapy | [68] |
| DOX-T/MNs | Soluble hyaluronic acid-based microneedles integrated with transferomes (T) loaded with doxorubicin hydrochloride (DOX) | Doxorubicin hydrochloride (DOX) | PDMS | Centrifuged, dried at room temperature | Tumor metastasis therapy | [69] |
| DOX/DMNs | Soluble hyaluronic acid-based microneedles loaded with doxorubicin (DOX) | Doxorubicin (DOX) | PDMS | Vacuum for 30 min, dried in a ventilated place for 12 h | Cancer treatment | [70] |
| DOX/SMNs | Swellable microneedles based on methacrylated hyaluronic acid loaded with doxorubicin (DOX) | Doxorubicin (DOX) | PDMS | Vacuum for 30 min, dried in a ventilated place for 12 h | Cancer treatment | [70] |
| p53 DNA/IR820 MN | Soluble hyaluronic acid-based microneedles loaded with p53 expression plasmid (p53 DNA) and indocyanine green derivative (IR820) | p53 DNA, IR820 | PDMS | Centrifuged at 5000 rpm for 15 min, dried overnight at 41 °C under vacuum | Synergistic combination of gene therapy and photothermal therapy of subcutaneous tumor | [71] |
| HA MN | Soluble hyaluronic acid-based microneedles | Hyaluronic acid (HA) | PDMS | Vacuum (~0.08 MPa), dried overnight in a sealed desiccator | Improving the permeability of skin tissues | [72] |
| DMNP loaded Hup A | Soluble hyaluronic acid-based microneedles containing dry huperzine A (Hup A) powder between layers of HA | huperzine A (Hup A) | PDMS | Centrifuged, dried overnight at room temperature | Alzheimer’s disease | [73] |
| HA MN | Soluble hyaluronic acid-based microneedles | Hyaluronic acid (HA) | PDMS | Vacuum for 45 min, dried overnight at room temperature | Safety evaluation | [74] |
| X-linked HA-NPs | Soluble microneedles based on crosslinked hyaluronic acid nanoparticles | Crosslinked hyaluronic acid nanoparticles (X-linked HA NPs) | PDMS | Centrifuged, dried at room temperature for 24 h | Sustained release test | [75] |
| RhB-loaded HA-MN | Soluble microneedles based on hyaluronic acid loaded with rhodamine B (RhB) | Rhodamine B (RhB) | PDMS | Centrifuged at 4000 rpm for 5 min, fan-dried for 48 h | Mechanical strength tests: the influence of HA molecular weight on the mechanical properties of HA-MNs; delivery efficiencies tests: transdermal delivery of rhodamine B (in vitro and in vivo) | [76] |
| Technology | Material | Microneedle Type | Power Source | Ref. |
|---|---|---|---|---|
| Stereolithography (SLA) | Liquid Polymers | Solid, Hollow, Coated | UV Light | [96] |
| Selective Laser Sintering (SLS) | Metals, Polymers, Ceramics, and Thermoplastics | Solid, Biocompatible, Hollow | Laser Beam | [97] |
| Digital Light Processing (DLP) | Epoxides, Acrylates | Solid, Hollow, Coated, Biodegradable, Hydrogel | UV Light | [98] |
| Fused Deposition Modeling (FDM) | Thermoplastic Polymers, Metal, Glass | Solid, Biodegradable | Heat | [95] |
| Two-Photon Polymerization (2PP) | UV-curable acrylates, ceramics, resins. | Solid, Hollow | UV Light | [99] |
| Continuous Liquid Interface Production (CLIP) | UV-curable resins, acrylates | Solid, Coated, Hydrogel, Biodegradable | UV Light | [100] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chudzińska, J.; Wawrzyńczak, A.; Feliczak-Guzik, A. Microneedles Based on a Biodegradable Polymer—Hyaluronic Acid. Polymers 2024, 16, 1396. https://doi.org/10.3390/polym16101396
Chudzińska J, Wawrzyńczak A, Feliczak-Guzik A. Microneedles Based on a Biodegradable Polymer—Hyaluronic Acid. Polymers. 2024; 16(10):1396. https://doi.org/10.3390/polym16101396
Chicago/Turabian StyleChudzińska, Jagoda, Agata Wawrzyńczak, and Agnieszka Feliczak-Guzik. 2024. "Microneedles Based on a Biodegradable Polymer—Hyaluronic Acid" Polymers 16, no. 10: 1396. https://doi.org/10.3390/polym16101396
APA StyleChudzińska, J., Wawrzyńczak, A., & Feliczak-Guzik, A. (2024). Microneedles Based on a Biodegradable Polymer—Hyaluronic Acid. Polymers, 16(10), 1396. https://doi.org/10.3390/polym16101396







