On the Fractionation and Physicochemical Characterisation of Self-Assembled Chitosan–DNA Polyelectrolyte Complexes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Depolymerisation of Chitosan
2.3. Characterisation of Chitosan: Degree of Acetylation and Molecular Weight Distribution
2.4. Formation of Polyelectrolyte Complexes
2.5. Characterisation of Chitosan–DNA Polyplexes
2.5.1. Zeta (ζ)-Potential Measurements
2.5.2. Batch-Mode Non-Invasive Back Scattering—Dynamic Light Scattering (NIBS-DLS)
2.5.3. Asymmetric Flow Field Flow Fractionation (AF4)
2.6. Negative Staining Transmission Electron Microscopy (TEM)
3. Results and Discussion
3.1. Spectrophotometric Determination of the Degree of N-Acetylation (DA) of Parent Chitosan
3.2. Molecular Weight Distribution of Chitosan
3.3. ζ-Potential of Polyelectrolyte Complexes
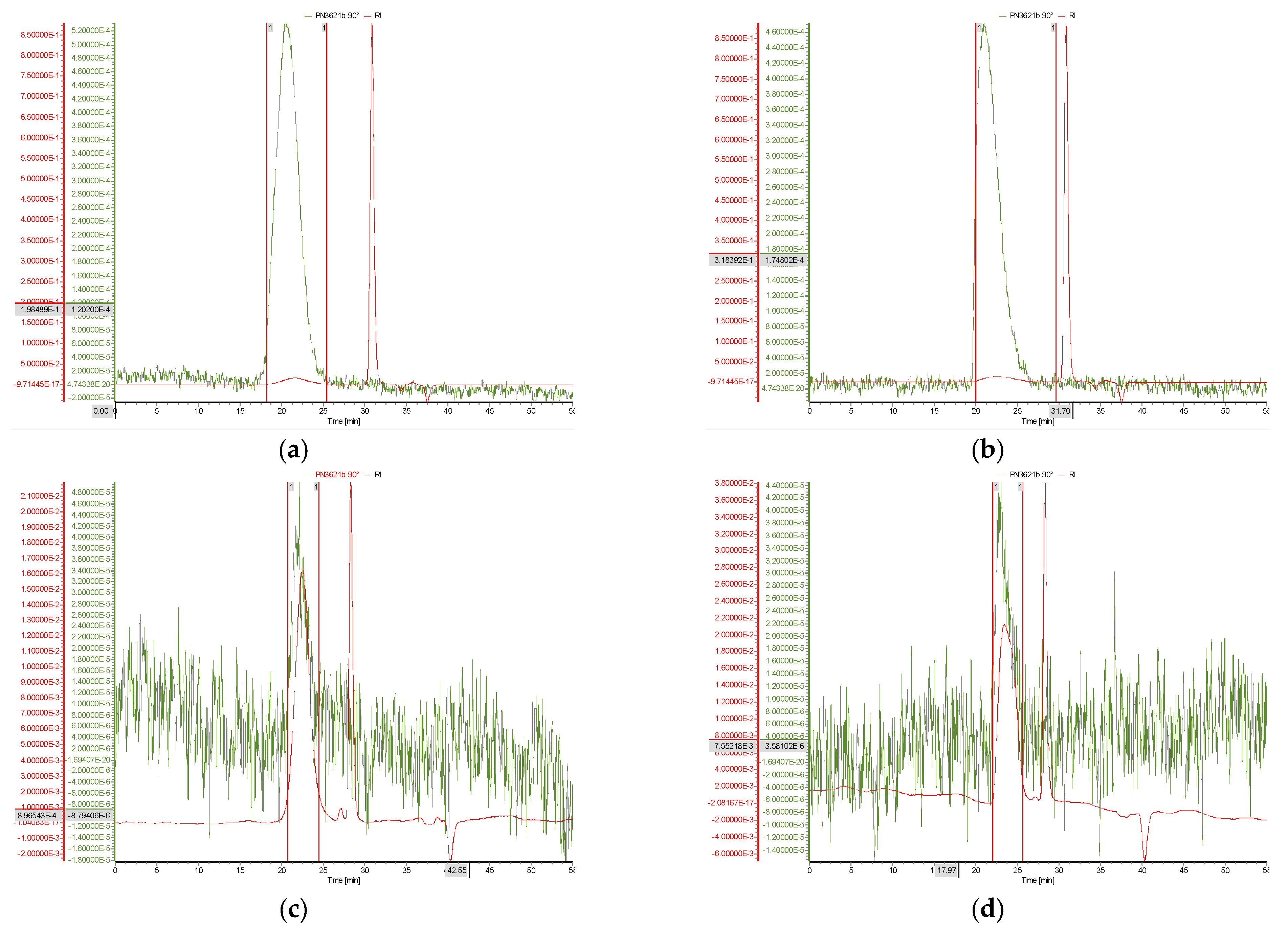
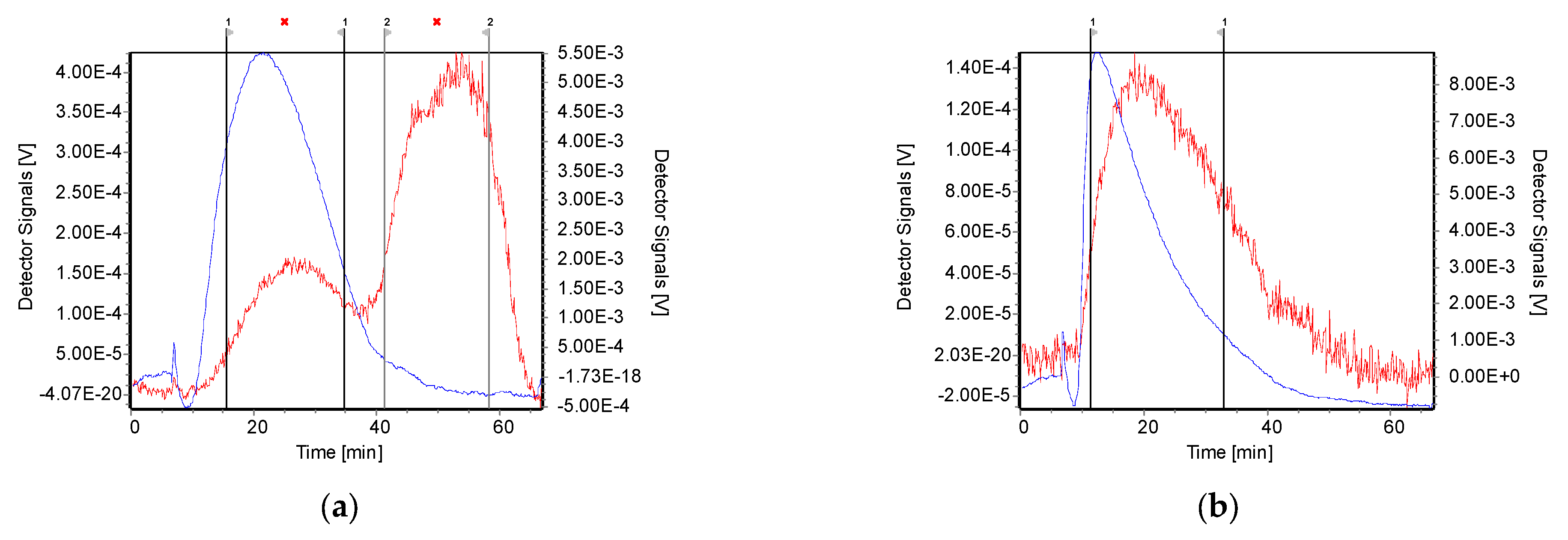
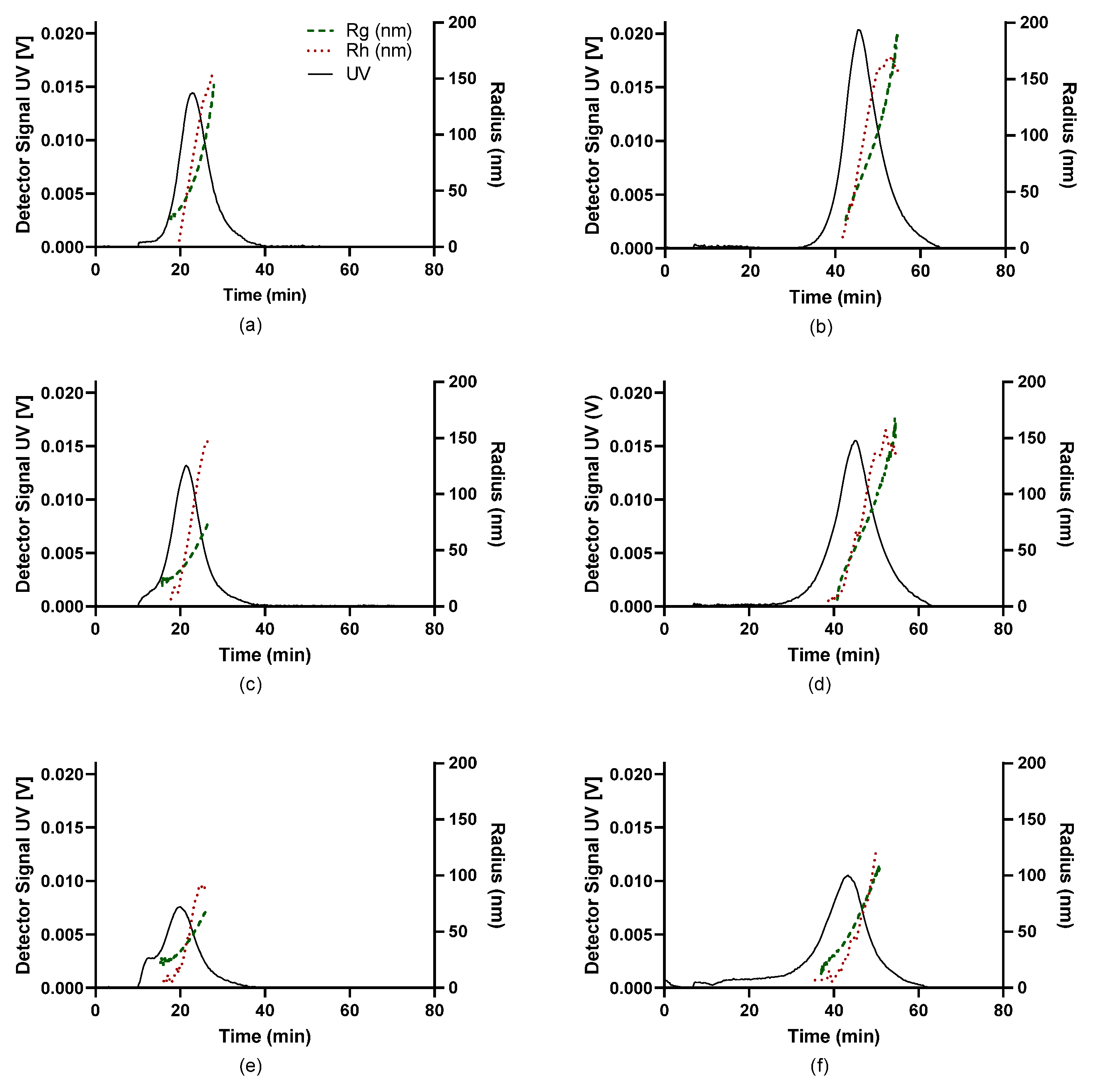
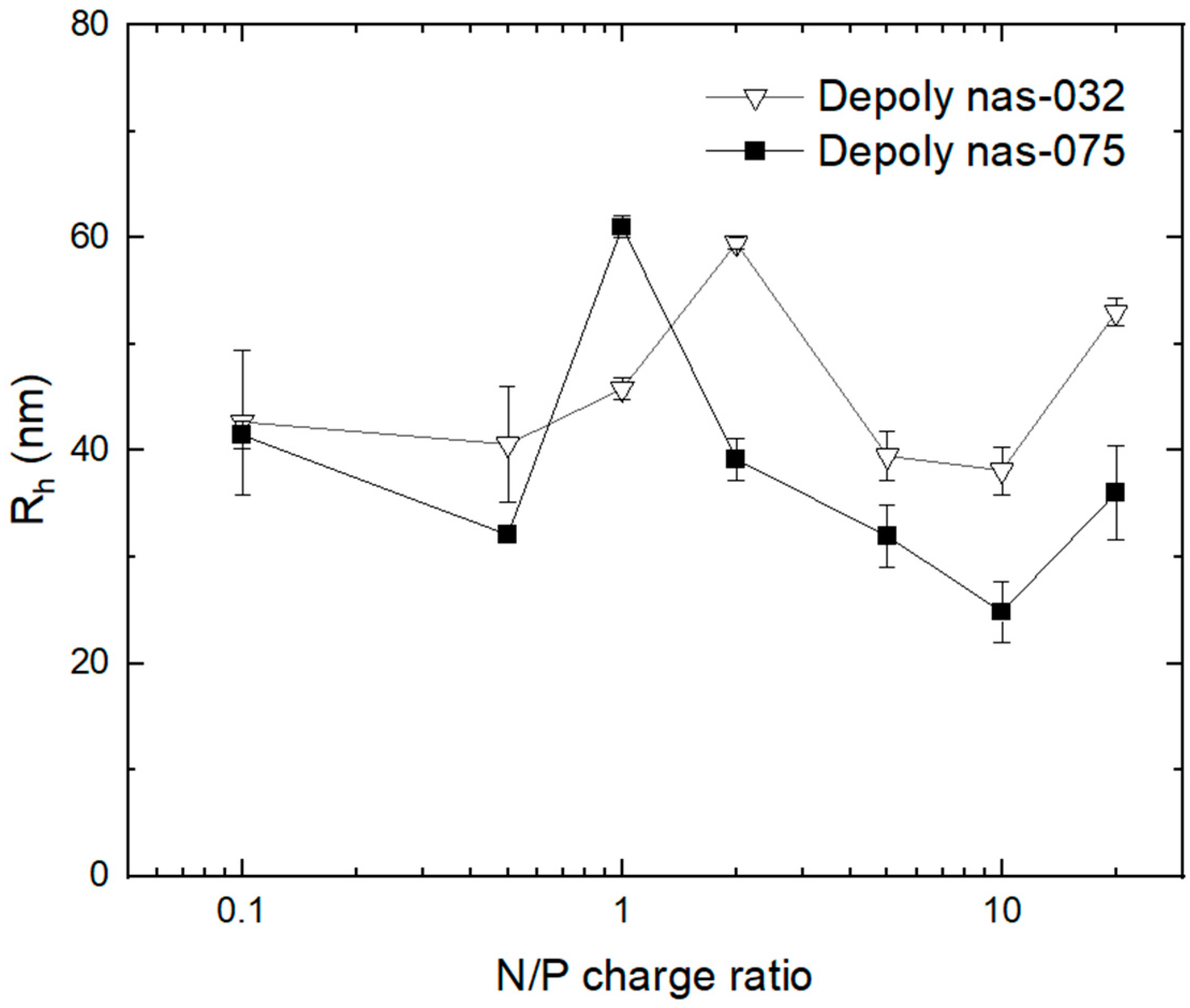
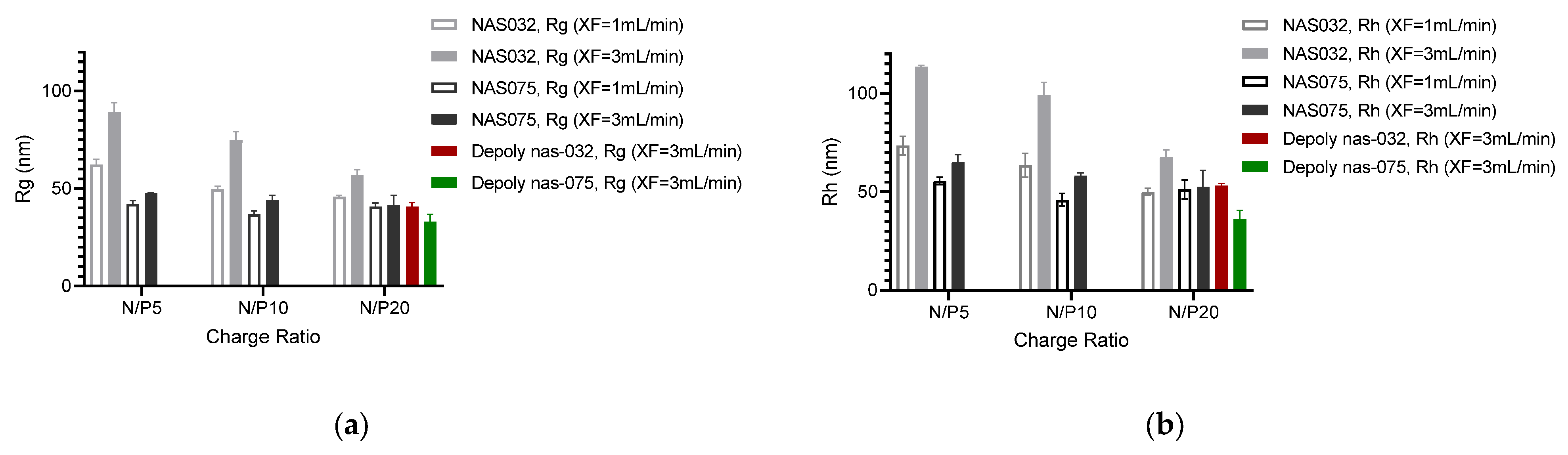
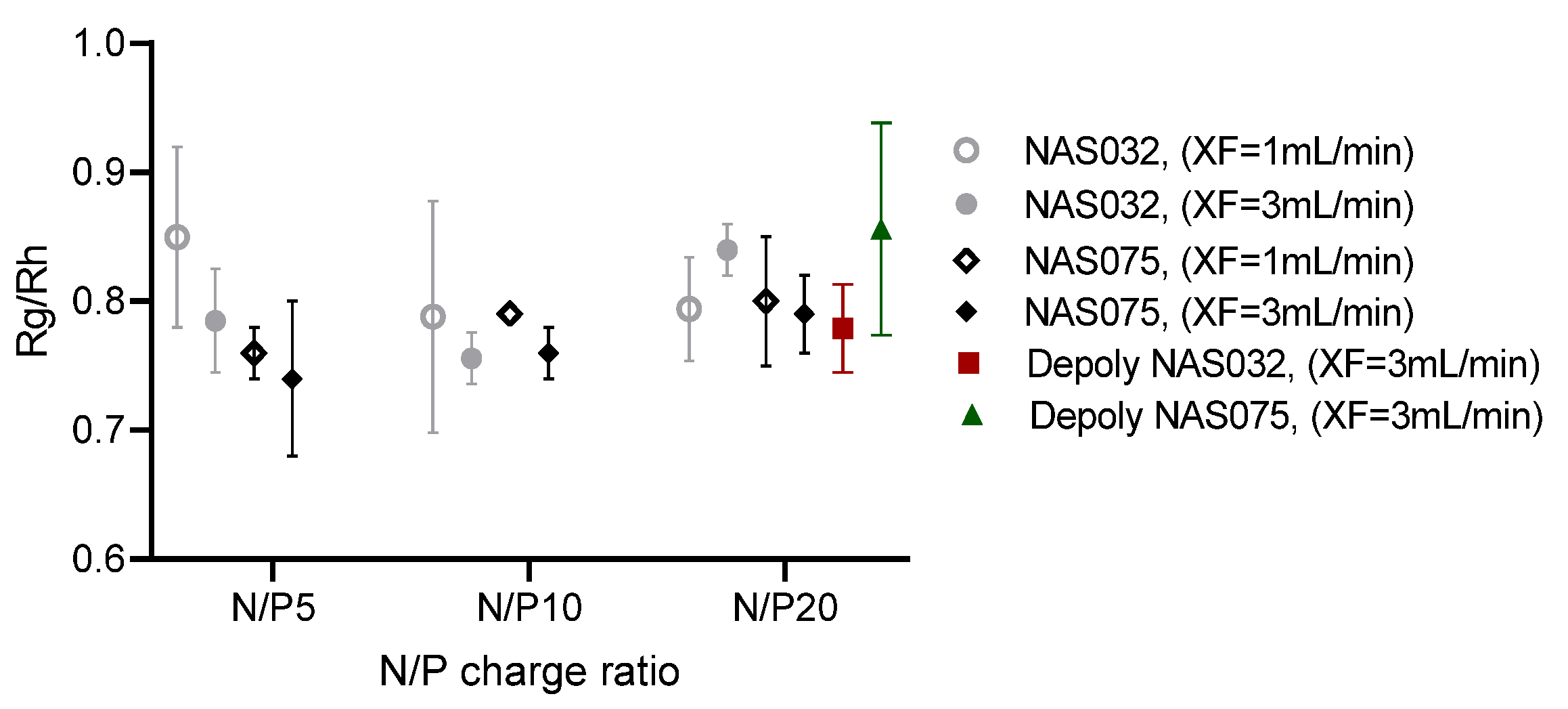
3.4. AF4 Characterisation of PEC
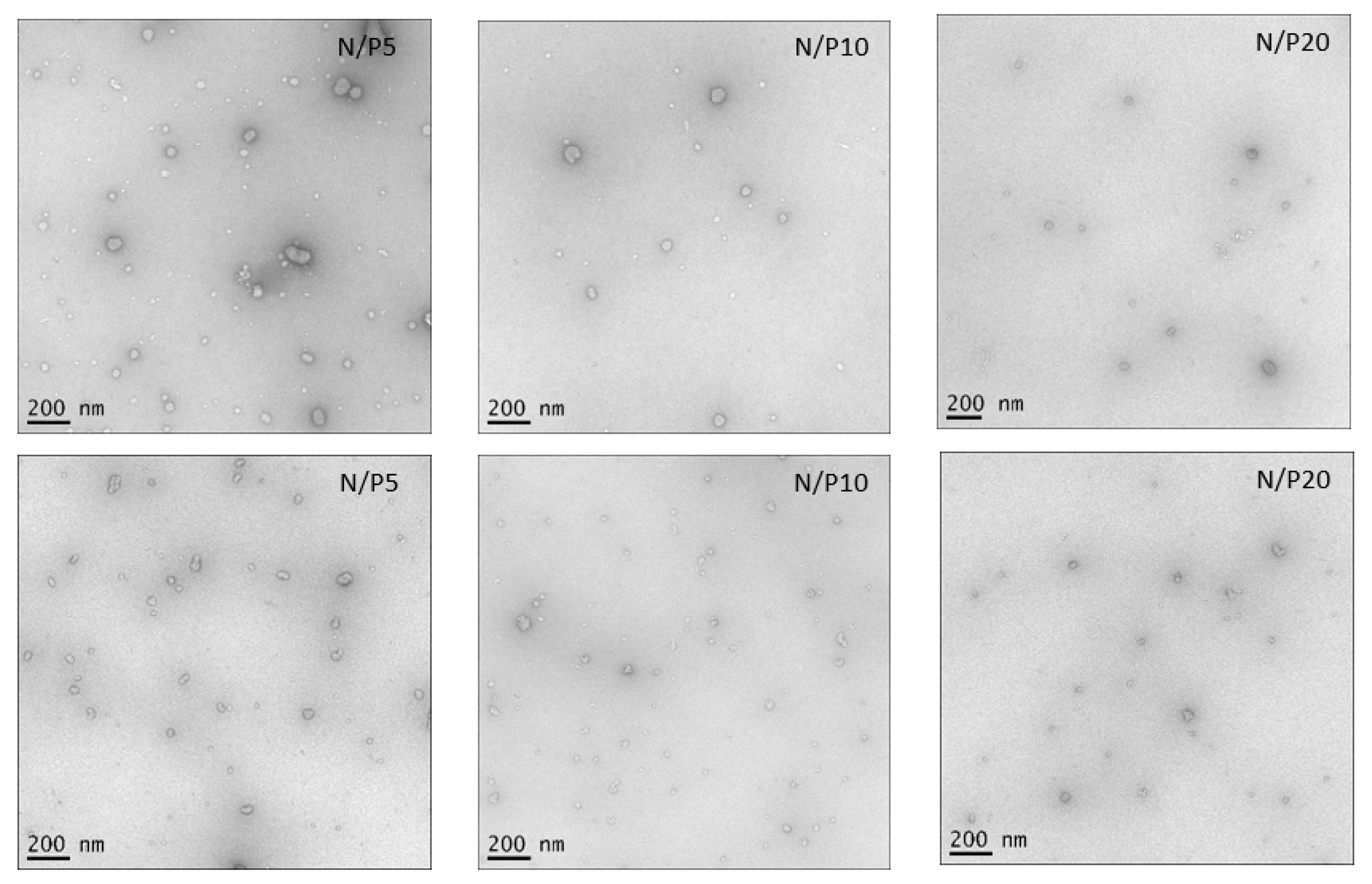
3.5. Negative Stain Transmission Electron Microscopy (TEM)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ban, E.; Kim, A. Coacervates: Recent developments as nanostructure delivery platforms for therapeutic biomolecules. Int. J. Pharm. 2022, 624, 122058. [Google Scholar] [CrossRef]
- Ishak, J.; Unsunnidhal, L.; Martien, R.; Kusumawati, A. In vitro evaluation of chitosan-DNA plasmid complex encoding Jembrana disease virus Env-TM protein as a vaccine candidate. J. Vet. Res. 2019, 63, 7–16. [Google Scholar] [CrossRef]
- Malmo, J.; Sorgard, H.; Varum, K.M.; Strand, S.P. siRNA delivery with chitosan nanoparticles: Molecular properties favoring efficient gene silencing. J. Control. Release 2012, 158, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Rashidpour, A.; Almajano, M.P.; Meton, I. Chitosan-based drug delivery system: Applications in fish biotechnology. Polymers 2020, 12, 1177. [Google Scholar] [CrossRef]
- Ali, A.; Ahmed, S. A review on chitosan and its nanocomposites in drug delivery. Int. J. Biol. Macromol. 2018, 109, 273–286. [Google Scholar] [CrossRef]
- Li, J.; Cai, C.; Li, J.; Li, J.; Li, J.; Sun, T.; Wang, L.; Wu, H.; Yu, G. Chitosan-based nanomaterials for drug delivery. Molecules 2018, 23, 2661. [Google Scholar] [CrossRef]
- Mohammadi, Z.; Eini, M.; Rastegari, A.; Tehrani, M.R. Chitosan as a machine for biomolecule delivery: A review. Carbohydr. Polym. 2021, 256, 117414. [Google Scholar] [CrossRef]
- Buschmann, M.D.; Merzouki, A.; Lavertu, M.; Thibault, M.; Jean, M.; Darras, V. Chitosans for delivery of nucleic acids. Adv. Drug Deliv. Rev. 2013, 65, 1234–1270. [Google Scholar] [CrossRef]
- Santos-Carballal, B.; Fernandez, E.F.; Goycoolea, F.M. Chitosan in non-viral gene delivery: Role of structure, characterisation methods, and insights in cancer and rare diseases therapies. Polymers 2018, 10, 444. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M.; Pavlov, G.; Desbrieres, J. Influence of acetic acid concentration on the solubilization of chitosan. Polymer 1999, 40, 7029–7032. [Google Scholar] [CrossRef]
- Il’ina, A.V.; Varlamov, V.P. Chitosan-based polyelectrolyte complexes: A review. Appl. Biochem. Microbiol. 2005, 41, 6–11. [Google Scholar]
- Cao, Y.; Tan, Y.F.; Wong, Y.S.; Liew, M.W.J.; Venkatraman, S. Recent Advances in Chitosan-Based Carriers for Gene Delivery. Mar. Drugs 2019, 17, 381. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Arora, S.; dos Santos Rodrigues, B.; Lakkadwala, S.; Banerjee, A.; Singh, J. Chitosan-based systems for gene delivery. In Functional Chitosan; Jana, S., Jana, S., Eds.; Springer: Singapore, 2019; pp. 229–267. [Google Scholar]
- Messai, I.; Lamalle, D.; Munier, S.; Verrier, B.; Ataman-Onal, Y.; Delair, T. Poly(D,L-lactic acid) and chitosan complexes: Interactions with plasmid DNA. Colloids Surf. A Physicochem. Eng. Asp. 2005, 255, 65–72. [Google Scholar] [CrossRef]
- Krayukhina, M.A.; Samoilova, N.A.; Yamskov, I.A. Polyelectrolyte complexes of chitosan: Formation, properties, and applications. Russ. Chem. Rev. 2008, 77, 799. [Google Scholar] [CrossRef]
- Mao, S.R.; Sun, W.; Kissel, T. Chitosan-based formulations for delivery of DNA and siRNA. Adv. Drug Deliv. Rev. 2010, 62, 12–27. [Google Scholar] [CrossRef]
- Costalat, M.; David, L.; Delair, T. Reversible controlled assembly of chitosan and dextran sulfate: A new method for nanoparticle elaboration. Carbohydr. Polym. 2014, 102, 717–726. [Google Scholar] [CrossRef]
- Carvalho, S.G.; Dos Santos, A.M.; Silvestre, A.L.P.; Meneguin, A.B.; Ferreira, L.M.B.; Chorilli, M.; Gremiao, M.P.D. New insights into physicochemical aspects involved in the formation of polyelectrolyte complexes based on chitosan and dextran sulfate. Carbohydr. Polym. 2021, 271, 118436. [Google Scholar] [CrossRef]
- Fernandez, E.F.; Santos-Carballal, B.; Weber, W.M.; Goycoolea, F.M. Chitosan as a non-viral co-transfection system in a cystic fibrosis cell line. Int. J. Pharm. 2016, 502, 1–9. [Google Scholar] [CrossRef]
- Poor, E.M.; Eslaminejad, M.B.; Gheibi, N.; Bagheri, F.; Atyabi, F. Chitosan-pDNA nanoparticle characteristics determine the transfection efficacy of gene delivery to human mesenchymal stem cells. Artif. Cells Nanomed. Biotechnol. 2014, 42, 376–384. [Google Scholar] [CrossRef]
- Koping-Hoggard, M.; Varum, K.M.; Issa, M.; Danielsen, S.; Christensen, B.E.; Stokke, B.T.; Artursson, P. Improved chitosan-mediated gene delivery based on easily dissociated chitosan polyplexes of highly defined chitosan oligomers. Gene Ther. 2004, 11, 1441–1452. [Google Scholar] [CrossRef]
- Koping-Hoggard, M.; Mel’nikova, Y.S.; Varum, K.M.; Lindman, B.; Artursson, P. Relationship between the physical shape and the efficiency of oligomeric chitosan as a gene delivery system in vitro and in vivo. J. Gene Med. 2003, 5, 130–141. [Google Scholar] [CrossRef]
- Lavertu, M.; Methot, S.; Tran-Khanh, N.; Buschmann, M.D. High efficiency gene transfer using chitosan/DNA nanoparticles with specific combinations of molecular weight and degree of deacetylation. Biomaterials 2006, 27, 4815–4824. [Google Scholar] [CrossRef] [PubMed]
- Zattoni, A.; Roda, B.; Borghi, F.; Marassi, V.; Reschiglian, P. Flow field-flow fractionation for the analysis of nanoparticles used in drug delivery. J. Pharm. Biomed. Anal. 2014, 87, 53–61. [Google Scholar] [CrossRef]
- Nilsson, L. Separation and characterisation of food macromolecules using field-flow fractionation: A review. Food Hydrocoll. 2013, 30, 1–11. [Google Scholar] [CrossRef]
- Messaud, F.A.; Sanderson, R.D.; Runyon, J.R.; Otte, T.; Pasch, H.; Williams, S.K.R. An overview on field-flow fractionation techniques and their applications in the separation and characterisation of polymers. Prog. Polym. Sci. 2009, 34, 351–368. [Google Scholar] [CrossRef]
- Quattrini, F.; Berrecoso, G.; Crecente-Campo, J.; Alonso, M.J. Asymmetric flow field-flow fractionation as a multifunctional technique for the characterisation of polymeric nanocarriers. Drug. Deliv. Transl. Res. 2021, 11, 396. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, W.; Dou, Y.; Song, T.; Shen, S.; Dou, H. Applications of asymmetrical flow field-flow fractionation for separation and characterisation of polysaccharides: A review. J. Chromatogr. A 2021, 1635, 461726. [Google Scholar] [CrossRef]
- Wahlund, K.G.; Nilsson, L. Flow FFF—Basics and key applications. In Field-Flow; Fractionation in Biopolymer Analysis; Williams, S., Caldwell, K., Eds.; Springer: Vienna, Austria, 2012. [Google Scholar]
- Juna, S.; Huber, A. Characterisation of normal corn starch using asymmetrical flow field-flow fractionation. Starch-Starke 2012, 64, 18–26. [Google Scholar] [CrossRef]
- Eskelin, K.; Poranen, M.M.; Oksanen, H.M. Asymmetrical flow field-flow fractionation on virus and virus-Like particle applications. Microorganisms 2019, 7, 555. [Google Scholar] [CrossRef]
- Wahlund, K.G.; Giddings, J.C. Properties of an Asymmetrical Flow Field-Flow Fractionation Channel Having One Permeable Wall. Anal. Chem. 1987, 59, 1332–1339. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.W.; Crist, R.M.; Clogston, J.D. The utility of asymmetric flow field-flow fractionation for preclinical characterisation of nanomedicines. Anal. Bioanal. Chem. 2020, 412, 425–438. [Google Scholar] [CrossRef]
- Ma, P.L.; Buschmann, M.D.; Winnik, F.M. One-Step Analysis of DNA/Chitosan Complexes by Field-Flow Fractionation Reveals Particle Size and Free Chitosan Content. Biomacromolecules 2010, 11, 549–554. [Google Scholar] [CrossRef]
- Ma, P.L.; Buschmann, M.D.; Winnik, F.M. Complete physicochemical characterization of DNA/chitosan complexes by multiple detection using asymmetrical flow field-flow fractionation. Anal. Chem. 2010, 82, 9636–9643. [Google Scholar] [CrossRef] [PubMed]
- Allan, G.G.; Peyron, M. Molecular-weight manipulation of chitosan. 1. kinetics of depolymerisation by nitrous-acid. Carbohydr. Res. 1995, 277, 257–272. [Google Scholar] [CrossRef]
- Allan, G.G.; Peyron, M. Molecular-weight manipulation of chitosan. 2. prediction and control of extent of depolymerisation by nitrous-acid. Carbohydr. Res. 1995, 277, 273–282. [Google Scholar] [CrossRef]
- Pedroni, V.I.; Gschaider, M.E.; Schulz, P.C. UV spectrophotometry: Improvements in the study of the degree of acetylation of chitosan. Macromol. Biosci. 2003, 3, 531–534. [Google Scholar] [CrossRef]
- Gonzalez-Espinosa, Y.; Sabagh, B.; Moldenhauer, E.; Clarke, P.; Goycoolea, F.M. Characterisation of chitosan molecular weight distribution by multi-detection asymmetric flow-field flow fractionation (AF4) and SEC. Int. J. Biol. Macromol. 2019, 136, 911–919. [Google Scholar] [CrossRef]
- Nguyen, S.; Winnik, F.M.; Buschmann, M.D. Improved reproducibility in the determination of the molecular weight of chitosan by analytical size exclusion chromatography. Carbohydr. Polym. 2009, 75, 528–533. [Google Scholar] [CrossRef]
- Bravo-Anaya, L.M.; Fernández-Solís, K.G.; Rosselgong, J.; Nano-Rodríguez, J.L.E.; Carvajal, F.; Rinaudo, M. Chitosan-DNA polyelectrolyte complex: Influence of chitosan characteristics and mechanism of complex formation. Int. J. Biol. Macromol. 2019, 126, 1037–1049. [Google Scholar] [CrossRef]
- Brugnerotto, J.; Lizardi, J.; Goycoolea, F.M.; Arguelles-Monal, W.; Desbrieres, J.; Rinaudo, M. An infrared investigation in relation with chitin and chitosan characterisation. Polymer 2001, 42, 3569–3580. [Google Scholar] [CrossRef]
- Varum, K.M.; Anthonsen, M.W.; Grasdalen, H.; Smidsrod, O. Determination of the degree of N-acetylation and the distribution of N-acetyl groups in partially N-deacetylated chitins (chitosans) by high-field n.m.r. spectroscopy. Carbohydr. Res. 1991, 211, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.A.; Rocchetti, R. determination of the degree of acetylation of chitosans by 1st derivative ultraviolet spectrophotometry. Carbohydr. Polym. 1985, 5, 461–472. [Google Scholar] [CrossRef]
- Liu, D.S.; Wei, Y.N.; Yao, P.J.; Jiang, L.B. Determination of the degree of acetylation of chitosan by UV spectrophotometry using dual standards. Carbohydr. Res. 2006, 341, 782–785. [Google Scholar] [CrossRef]
- Wu, T.; Zivanovic, S. Determination of the degree of acetylation (DA) of chitin and chitosan by an improved first derivative UV method. Carbohydr. Polym. 2008, 73, 248–253. [Google Scholar] [CrossRef]
- Hussain, I.; Singh, T.; Chittenden, C. Preparation of chitosan oligomers and characterisation: Their antifungal activities and decay resistance. Holzforschung 2012, 66, 119–125. [Google Scholar] [CrossRef]
- Affes, S.; Aranaz, I.; Acosta, N.; Heras, A.; Nasri, M.; Maalej, H. Physicochemical and biological properties of chitosan derivatives with varying molecular weight produced by chemical depolymerisation. In Biomass Conversion and Biorefinery; Springer: Berlin/Heidelberg, Germany, 2022. [Google Scholar] [CrossRef]
- O’Connell, A.; Gonzalez-Espinosa, Y.; Goycoolea, F.M.; Schuetz, P.; Mattsson, J. Characterisation of locust bean gum with asymmetric flow field-flow fractionation (AF4) and light scattering. Carbohydr. Polym. submitted.
- Augsten, C.; Mader, K. Characterising molar mass distributions and molecule structures of different chitosans using asymmetrical flow field-flow fractionation combined with multi-angle light scattering. Int. J. Pharm. 2008, 351, 23–30. [Google Scholar] [CrossRef]
- Ragelle, H.; Vandermeulen, G.; Preat, V. Chitosan-based siRNA delivery systems. J. Control. Release 2013, 172, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Alatorre-Meda, M.; Taboada, P.; Sabin, J.; Krajewska, B.; Varela, L.M.; Rodriguez, J.R. DNA-chitosan complexation: A dynamic light scattering study. Colloid. Surf. A Physicochem. Eng. Asp. 2009, 339, 145–152. [Google Scholar] [CrossRef]
- Richardson, S.C.W.; Kolbe, H.J.V.; Duncan, R. Potential of low molecular mass chitosan as a DNA delivery system: Biocompatibility, body distribution and ability to complex and protect DNA. Int. J. Pharm. 1999, 178, 231–243. [Google Scholar] [CrossRef]
- Wagner, M.; Holzschuh, S.; Traeger, A.; Fahr, A.; Schubert, U.S. Asymmetric flow field-flow fractionation in field of nanomedicine. Anal. Chem. 2014, 86, 5201–5210. [Google Scholar] [CrossRef]
- Alatorre-Meda, M.; Taboada, P.; Hardl, F.; Wagner, T.; Freis, M.; Rodriguez, J.R. The influence of chitosan valence on the complexation and transfection of DNA The weaker the DNA-chitosan binding the higher the transfection efficiency. Colloids Surf. B Biointerfaces 2011, 82, 54–62. [Google Scholar] [CrossRef]
- Tang, M.X.; Szoka, F.C. The influence of polymer structure on the interactions of cationic polymers with DNA and morphology of the resulting complexes. Gene Ther. 1997, 4, 823–832. [Google Scholar] [CrossRef] [PubMed]
- MacLaughlin, F.C.; Mumper, R.J.; Wang, J.J.; Tagliaferri, J.M.; Gill, I.; Hinchcliffe, M.; Rolland, A.P. Chitosan and depolymerised chitosan oligomers as condensing carriers for in vivo plasmid delivery. J. Control. Release 1998, 56, 259–272. [Google Scholar] [CrossRef]
- Danielsen, S.; Varum, K.M.; Stokke, B.T. Structural analysis of chitosan mediated DNA condensation by AFM: Influence of chitosan molecular parameters. Biomacromolecules 2004, 5, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Strand, S.P.; Danielsen, S.; Christensen, B.E.; Varum, K.M. Influence of chitosan structure on the formation and stability of DNA-chitosan polyelectrolyte complexes. Biomacromolecules 2005, 6, 3357–3366. [Google Scholar] [CrossRef] [PubMed]
- Koping-Hoggard, M.; Tubulekas, I.; Guan, H.; Edwards, K.; Nilsson, M.; Varum, K.M.; Artursson, P. Chitosan as a nonviral gene delivery system. Structure-property relationships and characteristics compared with polyethylenimine in vitro and after lung administration in vivo. Gene Ther. 2001, 8, 1108–1121. [Google Scholar] [CrossRef]
- Mao, H.Q.; Roy, K.; Troung-Le, V.L.; Janes, K.A.; Lin, K.Y.; Wang, Y.; August, J.T.; Leong, K.W. Chitosan-DNA nanoparticles as gene carriers: Synthesis, characterisation and transfection efficiency. J. Control. Release 2001, 70, 399–421. [Google Scholar] [CrossRef]
- Cui, Z.R.; Mumper, R.J. Chitosan-based nanoparticles for topical genetic immunisation. J. Control. Release 2001, 75, 409–419. [Google Scholar] [CrossRef]
- Valente, J.F.A.; Pereira, P.; Sousa, A.; Queiroz, J.A.; Sousa, F. Effect of plasmid DNA size on chitosan or polyethyleneimine polyplexes formulation. Polymers 2021, 13, 793. [Google Scholar] [CrossRef]
- von der Kammer, F.; Legros, S.; Larsen, E.H.; Loeschner, K.; Hofmann, T. Separation and characterisation of nanoparticles in complex food and environmental samples by field-flow fractionation. TrAC-Trends Anal. Chem. 2011, 30, 425–436. [Google Scholar] [CrossRef]
- Omar, J.; Boix, A.; Kerckhove, G.; von Holst, C. Optimisation of asymmetric flow field-flow fractionation for the characterisation of nanoparticles in coated polydisperse TiO2 with applications in food and feed. Food Addit. Contam. A 2016, 33, 1775–1784. [Google Scholar] [CrossRef]
- Niezabitowska, E.; Town, A.R.; Sabagh, B.; Moctezuma, M.D.M.; Kearns, V.R.; Spain, S.G.; Rannard, S.P.; McDonald, T.O. Insights into the internal structures of nanogels using a versatile asymmetric-flow field-flow fractionation method. Nanoscale Adv. 2020, 2, 4713–4721. [Google Scholar] [CrossRef]
- Bednar, A.J.; Poda, A.R.; Mitrano, D.M.; Kennedy, A.J.; Gray, E.P.; Ranville, J.F.; Hayes, C.A.; Crocker, F.H.; Steevens, J.A. Comparison of on-line detectors for field flow fractionation analysis of nanomaterials. Talanta 2013, 104, 140–148. [Google Scholar] [CrossRef]
- Gigault, J.; Pettibone, J.M.; Schmitt, C.; Hackley, V.A. Rational strategy for characterisation of nanoscale particles by asymmetric- flow field flow fractionation: A tutorial. Anal. Chim. Acta 2014, 809, 9–24. [Google Scholar] [CrossRef]
- Parot, J.; Caputo, F.; Mehn, D.; Hackley, V.A.; Calzolai, L. Physical characterisation of liposomal drug formulations using multi-detector asymmetrical-flow field flow fractionation. J. Control. Release 2020, 320, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Huglin, M.B. Ligt Scattering from Polymer Solutions; Academic Press: New York, NY, USA, 1972. [Google Scholar]
- Zhang, R.; Shklovskii, B.T. Phase diagram of solution of oppositely charged polyelectrolytes. Phys. A Stat. Mech. Appl. 2005, 352, 216–238. [Google Scholar] [CrossRef]
- Amaduzzi, F.; Bomboi, F.; Bonincontro, A.; Bordi, F.; Casciardi, S.; Chronopoulou, L.; Diociaiuti, M.; Mura, F.; Palocci, C.; Sennato, S. Chitosan-DNA complexes: Charge inversion and DNA condensation. Colloids Surf. B Biointerfaces 2014, 114, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bordi, F.; Chronopoulou, L.; Palocci, C.; Bomboi, F.; Di Martino, A.; Cifani, N.; Pompili, B.; Ascenzioni, F.; Sennato, S. Chitosan-DNA complexes: Effect of molecular parameters on the efficiency of delivery. Colloids Surf. A Physicochem. Eng. Asp. 2014, 460, 184–190. [Google Scholar] [CrossRef]
- Novoa-Carballal, R.; Fernandez-Megia, E.; Riguera, R. Dynamics of chitosan by H-1 NMR relaxation. Biomacromolecules 2010, 11, 2079–2086. [Google Scholar] [CrossRef]
- Menchicchi, B.; Fuenzalida, J.P.; Bobbili, K.B.; Hensel, A.; Swamy, M.J.; Goycoolea, F.M. Structure of chitosan determines its interactions with mucin. Biomacromolecules 2014, 15, 3550–3558. [Google Scholar] [CrossRef]
- Novoa-Carballal, R.; Riguera, R.; Fernandez-Megia, E. Chitosan hydrophobic domains are favoured at low degree of acetylation and molecular weight. Polymer 2013, 54, 2081–2087. [Google Scholar] [CrossRef]
- Boye, S.; Ennen, F.; Scharfenberg, L.; Appelhans, D.; Nilsson, L.; Lederer, A. From 1D rods to 3D networks: A biohybrid topological diversity investigated by asymmetrical flow field-flow fractionation. Macromolecules 2015, 48, 4607–4619. [Google Scholar] [CrossRef]
- Lobanov, M.Y.; Bogatyreva, N.S.; Galzitskaya, O.V. Radius of gyration as an indicator of protein structure compactness. Mol. Biol. 2008, 42, 623–628. [Google Scholar] [CrossRef]
- Lohrke, J.; Briel, A.; Mader, K. Characterization of superparamagnetic iron oxide nanoparticles by asymmetrical flow-field-flow-fractionation. Nanomedicine 2008, 3, 437–452. [Google Scholar] [CrossRef]
- Liu, W.G.; Sun, S.J.; Cao, Z.Q.; Xin, Z.; Yao, K.D.; Lu, W.W.; Luk, K.D.K. An investigation on the physicochemical properties of chitosan/DNA polyelectrolyte complexes. Biomaterials 2005, 26, 2705–2711. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, K.; Li, S. Nucleic Acids based polyelectrolyte complexes: Their complexation mechanism, morphology, and stability. Chem. Mater. 2021, 33, 7923–7943. [Google Scholar] [CrossRef]
- Hembram, K.C.; Prabha, S.; Chandra, R.; Ahmed, B.; Nimesh, S. Advances in preparation and characterisation of chitosan nanoparticles for therapeutics. Artif. Cell. Nanomed. Biotechnol. 2016, 44, 305–314. [Google Scholar] [CrossRef]
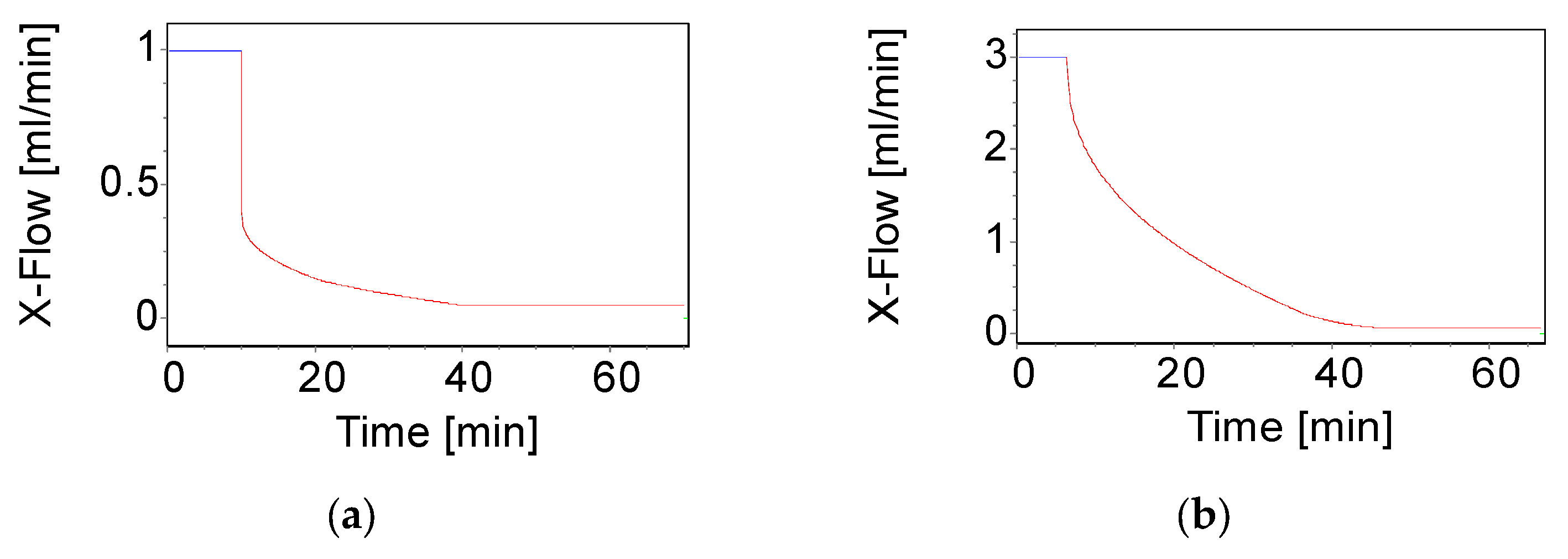





| Flow Setup | Method 1 | Method 2 | ||||
|---|---|---|---|---|---|---|
| Injection flow rate (mL/min) | 0.2 | 0.2 | ||||
| Injection/Focus time (min) | 9 @ flow rate 1.80 mL/min | 6 @ flow rate 3.30 mL/min | ||||
| Transition time (min) | 1 | 0.2 | ||||
| Cross-flow rate (mL/min) | 1.0 | 3.0 | ||||
| Elution steps | Time (min) | Flow rate (mL/min) | Type | Time (min) | Flow rate (mL/min) | Type |
| 10 | 0.4 | Exponent: 0.4 | 0.2 | 3 | Constant | |
| 20 | 0.15 | Exponent: 0.8 | 30 | 3 | Exponent: 0.4 | |
| 15 | 0.05 | Linear: 1 | 5 | 0.22 | Exponent: 0.8 | |
| 15 | 0.05 | Constant: 0.8 | 5 | 0.11 | Exponent: 0.8 | |
| 20 | 0.06 | Constant | ||||
| Rinse time (min) | 0.5 | 0.5 | ||||
| Total run time (min) | 70.5 | 66.9 | ||||
| Chitosan Sample | DA* (%) | DA** (%) |
|---|---|---|
| NAS-032 | 43.4 | 48.0 |
| NAS-075 | 4.7 | 0 |
| Chitosan Sample | Technique | Mn (g/mol) | Mw (g/mol) | Mz (g/mol) | Ð (=Mw/Mn) | Recovery (%) |
|---|---|---|---|---|---|---|
| NAS-032 | SEC | (9.18 ± 0.68) · 104 | (1.64 ± 0.01) · 105 | (3.01 ± 0.00) · 105 | 1.79 ± 0.11 | 88.1 |
| AF4 | (9.19 ± 0.79) · 104 | (1.42 ± 0.02) · 105 | (2.08 ± 0.11) · 105 | 1.55 ± 0.11 | 30.0 | |
| Deploy nas-032 | SEC | (5.41 ± 3.09) · 103 | (1.02 ± 0.21) · 104 | (1.38 ± 0.14) · 104 | 2.26 ± 0.97 | 51.0 |
| AF4 | (8.22 ± 0.49) · 103 | (8.39 ± 0.50) · 103 | (8.57 ± 0.52) · 103 | 1.02 ± 0.001 | 25.7 | |
| NAS-075 | SEC | (5.47 ± 0.50) · 104 | (1.18 ± 0.06) · 105 | (2.88 ± 0.19) · 105 | 2.17 ± 0.29 | 93.5 |
| AF4 | (6.42 ± 0.35) · 104 | (1.23 ± 0.05) · 105 | (2.49 ± 0.71) · 105 | 1.92 ± 0.17 | 38.7 | |
| Deploy nas-075 | SEC | (1.95 ± 1.51) · 103 | (6.12 ± 0.68) · 103 | (1.03 ± 0.31) · 104 | 4.68 ± 3.98 | 73 |
| AF4 | (1.42 ± 0.23) · 104 | (1.46 ± 0.22) · 104 | (1.51 ± 0.20) · 104 | 1.02 ± 0.01 | 18.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sajid, A.; Castronovo, M.; Goycoolea, F.M. On the Fractionation and Physicochemical Characterisation of Self-Assembled Chitosan–DNA Polyelectrolyte Complexes. Polymers 2023, 15, 2115. https://doi.org/10.3390/polym15092115
Sajid A, Castronovo M, Goycoolea FM. On the Fractionation and Physicochemical Characterisation of Self-Assembled Chitosan–DNA Polyelectrolyte Complexes. Polymers. 2023; 15(9):2115. https://doi.org/10.3390/polym15092115
Chicago/Turabian StyleSajid, Ayesha, Matteo Castronovo, and Francisco M. Goycoolea. 2023. "On the Fractionation and Physicochemical Characterisation of Self-Assembled Chitosan–DNA Polyelectrolyte Complexes" Polymers 15, no. 9: 2115. https://doi.org/10.3390/polym15092115
APA StyleSajid, A., Castronovo, M., & Goycoolea, F. M. (2023). On the Fractionation and Physicochemical Characterisation of Self-Assembled Chitosan–DNA Polyelectrolyte Complexes. Polymers, 15(9), 2115. https://doi.org/10.3390/polym15092115







