Accelerating Payload Release from Complex Coacervates through Mechanical Stimulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Coacervate Preparation
2.3. Release Experiments
2.4. Gravimetric Analysis
3. Results and Discussion
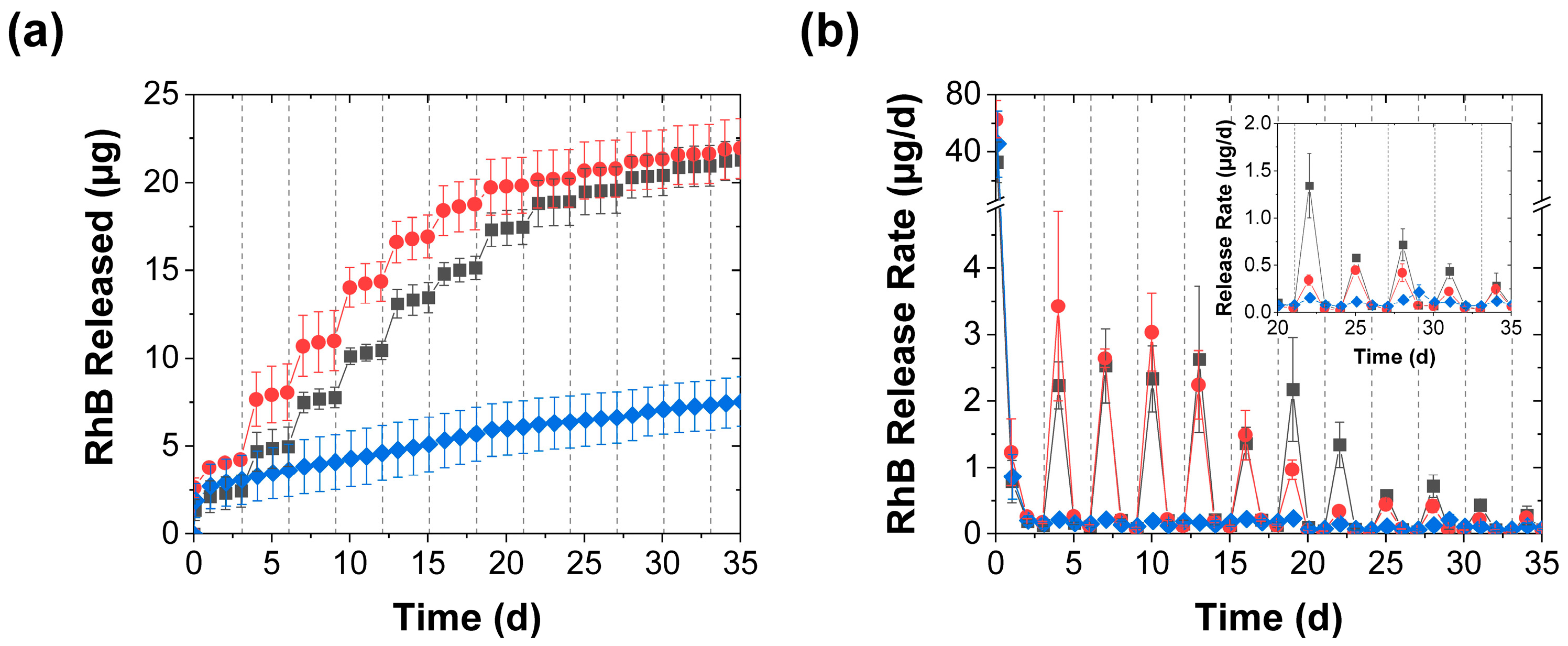
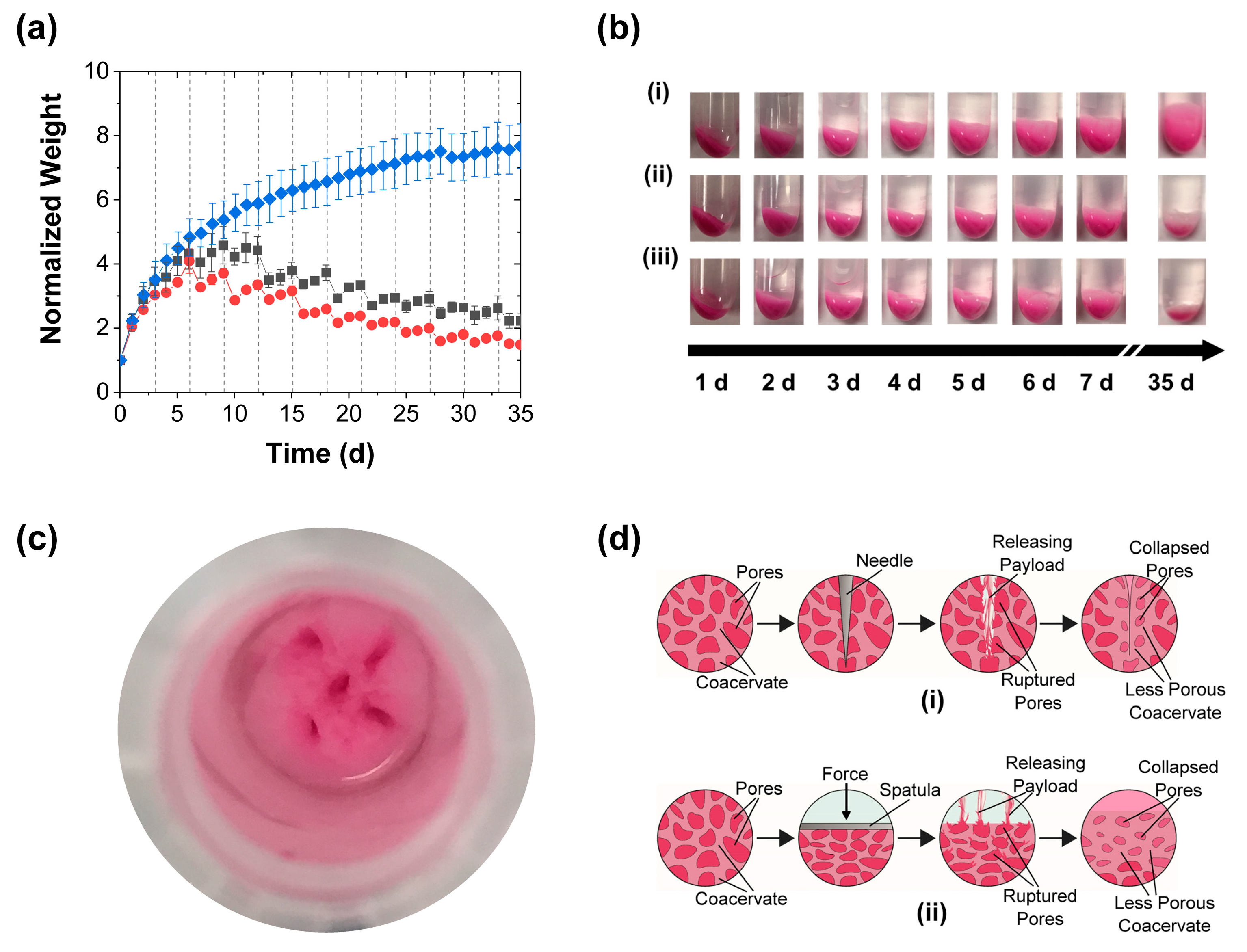
3.1. Effect of Mechanical Stimulation
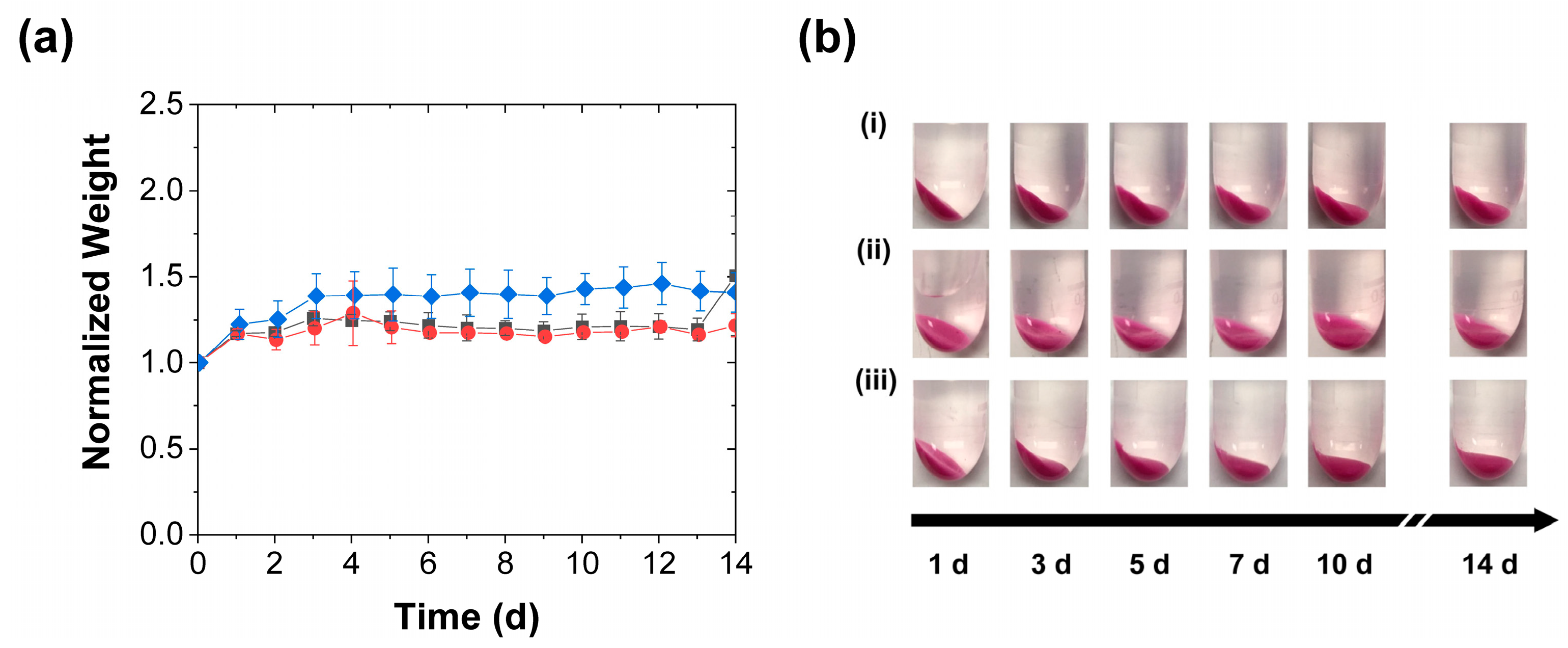
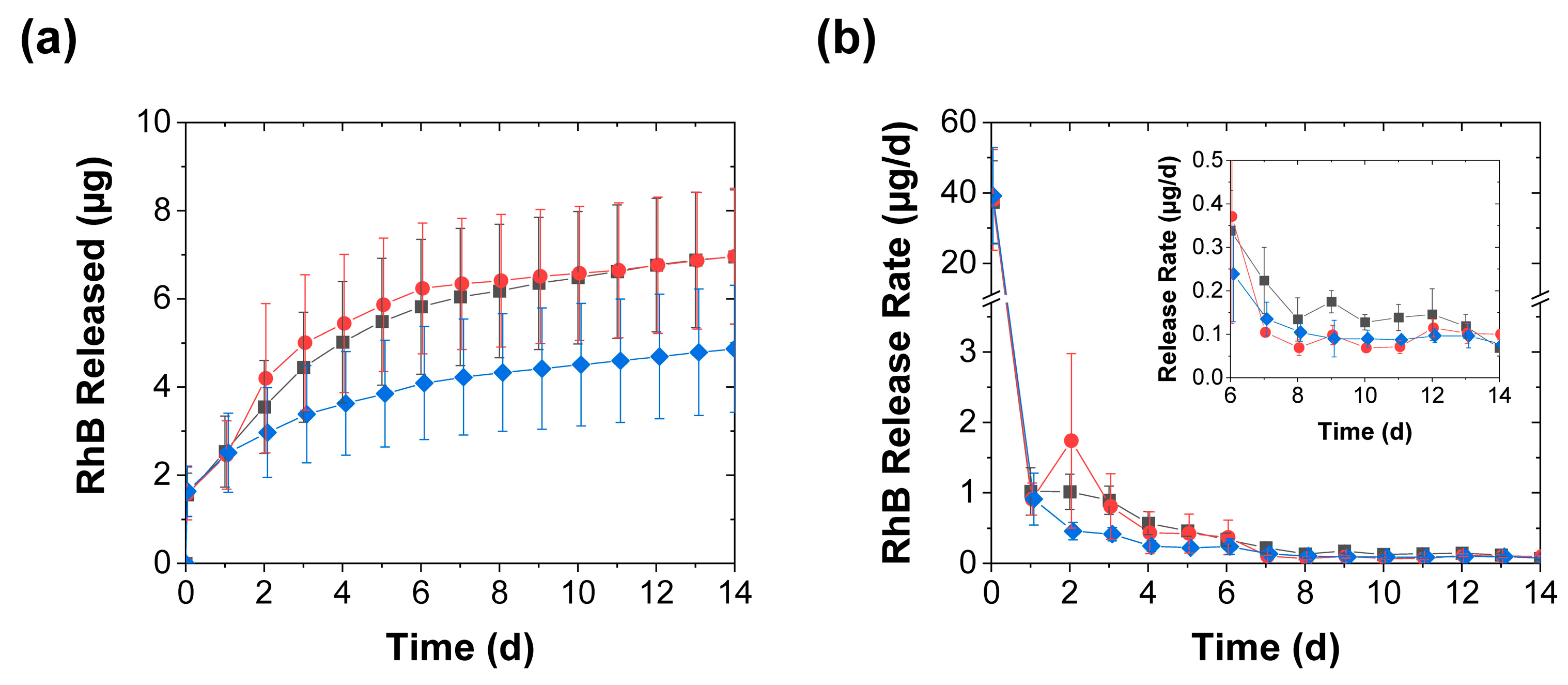
3.2. Effect of Mechanical Stimulation Frequency
3.3. Effect of Release Media
3.4. Further Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- De Jong, H.G.B. Complex colloid systems. In Colloid Science; Kruyt, H.R., Ed.; Elsevier: Amsterdam, The Netherlands, 1949; Volume II, pp. 335–432. [Google Scholar]
- Menger, F.M.; Sykes, B.M. Anatomy of a coacervate. Langmuir 1998, 14, 4131–4137. [Google Scholar] [CrossRef]
- Kizilay, E.; Kayitmazer, A.B.; Dubin, P.L. Complexation and coacervation of polyelectrolytes with oppositely charged colloids. Adv. Colloid Interface Sci. 2011, 167, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Momeni, A.; Filiaggi, M.J. Rheology of polyphosphate coacervates. J. Rheol. 2016, 60, 25–34. [Google Scholar] [CrossRef]
- Wang, X.; Lee, J.; Wang, Y.-W.; Huang, Q. Composition and rheological properties of β-lactoglobulin/pectin coacervates: Effects of salt concentration and initial protein/polysaccharide ratio. Biomacromolecules 2007, 8, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Winter, H.H.; Perry, S.L. Linear viscoelasticity of complex coacervates. Adv. Colloid Interface Sci. 2017, 239, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Blocher, W.C.; Perry, S.L. Complex coacervate-based materials for biomedicine. WIREs Nanomed. Nanobiotechnol. 2017, 9, e1442. [Google Scholar] [CrossRef]
- Johnson, N.R.; Wang, Y. Coacervate delivery systems for proteins and small molecule drugs. Expert Opin. Drug Deliv. 2014, 11, 1829–1832. [Google Scholar] [CrossRef]
- Lawrence, P.G.; Patil, P.S.; Leipzig, N.D.; Lapitsky, Y. Ionically cross-linked polymer networks for the multiple-month release of small molecules. ACS Appl. Mater. Interfaces 2016, 8, 4323–4335. [Google Scholar] [CrossRef]
- Black, K.A.; Priftis, D.; Perry, S.L.; Yip, J.; Byun, W.Y.; Tirrell, M. Protein encapsulation via polypeptide complex coacervation. ACS Macro Lett. 2014, 3, 1088–1091. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, P.G.; Lapitsky, Y. Ionically crosslinked poly(allylamine) as a stimulus-responsive underwater adhesive: Ionic strength and pH effects. Langmuir 2015, 31, 1564–1574. [Google Scholar] [CrossRef]
- Scott, W.A.; Gharakhanian, E.G.; Bell, A.G.; Evans, D.; Barun, E.; Houk, K.; Deming, T.J. Active Controlled and Tunable Coacervation Using Side-Chain Functional α-Helical Homopolypeptides. J. Am. Chem. Soc. 2021, 143, 18196–18203. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Schlenoff, J.B. The polyelectrolyte complex/coacervate continuum. Macromolecules 2014, 47, 3108–3116. [Google Scholar] [CrossRef]
- Bagaria, H.G.; Wong, M.S. Polyamine-salt aggregate assembly of capsules as responsive drug delivery vehicles. J. Mater. Chem. 2011, 21, 9454–9466. [Google Scholar] [CrossRef]
- De Silva, U.K.; Brown, J.L.; Lapitsky, Y. Poly(allylamine)/tripolyphosphate coacervates enable high loading and multiple-month release of weakly amphiphilic anionic drugs: A case study with ibuprofen. RSC Adv. 2018, 8, 19409–19419. [Google Scholar] [CrossRef]
- Zhao, M.; Zacharia, N.S. Sequestration of Methylene Blue into polyelectrolyte complex coacervates. Macromol. Rapid Commun. 2016, 37, 1249–1255. [Google Scholar] [CrossRef]
- Wang, Y.f.; Gao, J.Y.; Dubin, P.L. Protein separation via polyelectrolyte coacervation: Selectivity and efficiency. Biotechnol. Prog. 1996, 12, 356–362. [Google Scholar] [CrossRef]
- Schmitt, C.; Turgeon, S.L. Protein/polysaccharide complexes and coacervates in food systems. Adv. Colloid Interface Sci. 2011, 167, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Stewart, R.J. Biomimetic underwater adhesives with environmentally triggered setting mechanisms. Adv. Mater. 2010, 22, 729–733. [Google Scholar] [CrossRef]
- Stewart, R.J.; Wang, C.S.; Shao, H. Complex coacervates as a foundation for synthetic underwater adhesives. Adv. Colloid Interface Sci. 2011, 167, 85–93. [Google Scholar] [CrossRef]
- Alam, S.S.; Seo, Y.; Lapitsky, Y. Highly sustained release of bactericides from complex coacervates. ACS Appl. Bio Mater. 2020, 3, 8427–8437. [Google Scholar] [CrossRef]
- Yeo, Y.; Bellas, E.; Firestone, W.; Langer, R. Complex coacervates for thermally sensitive controlled release of flavor compounds. J. Agric. Food Chem. 2005, 53, 7518–7525. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.M.; Barreiro, M.F.; Coelho, M.; Rodrigues, A.E. Microencapsulation of essential oils with biodegradable polymeric carriers for cosmetic applications. Chem. Eng. J. 2014, 245, 191–200. [Google Scholar] [CrossRef]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Tavera, E.M.; Kadali, S.B.; Bagaria, H.G.; Liu, A.W.; Wong, M.S. Experimental and modeling analysis of diffusive release from single-shell microcapsules. AIChE J. 2009, 55, 2950–2965. [Google Scholar] [CrossRef]
- Jacobs, M.I.; Jira, E.R.; Schroeder, C.M. Understanding How Coacervates Drive Reversible Small Molecule Reactions to Promote Molecular Complexity. Langmuir 2021, 37, 14323–14335. [Google Scholar] [CrossRef]
- Alam, S.S.; Mather, C.B.; Seo, Y.; Lapitsky, Y. Poly(allylamine)/tripolyphosphate coacervates for encapsulation and long-term release of cetylpyridinium chloride. Colloids Surf. A. 2021, 629, 127490. [Google Scholar] [CrossRef]
- Green, B.K.; Schleicher, L. Oil-containing microscopic capsules and method of making them. U.S. Patent 2,800,457, 1957. [Google Scholar]
- Uhlemann, J.; Schleifenbaum, B.; Bertram, H.-J. Flavor encapsulation technologies: An overview including recent developments. Perfum. Flavor. 2002, 27, 52–61. [Google Scholar]
- Rungwasantisuk, A.; Raibhu, S. Application of encapsulating lavender essential oil in gelatin/gum-arabic complex coacervate and varnish screen-printing in making fragrant gift-wrapping paper. Prog. Org. Coat. 2020, 149, 105924. [Google Scholar] [CrossRef]
- Weinbreck, F.; Minor, M.; De Kruif, C. Microencapsulation of oils using whey protein/gum arabic coacervates. J. Microencapsul. 2004, 21, 667–679. [Google Scholar] [CrossRef]
- Fares, H.M.; Wang, Q.; Yang, M.; Schlenoff, J.B. Swelling and inflation in polyelectrolyte complexes. Macromolecules 2018, 52, 610–619. [Google Scholar] [CrossRef]
- Hamad, F.G.; Chen, Q.; Colby, R.H. Linear viscoelasticity and swelling of polyelectrolyte complex coacervates. Macromolecules 2018, 51, 5547–5555. [Google Scholar] [CrossRef]
- Scranton, A.B.; Klier, J.; Peppas, N.A. Soluble chain fractions in hydrophilic polymer networks: Origin and effect on dynamic uptake overshoots. Polymer 1990, 31, 1288–1293. [Google Scholar] [CrossRef]
- Lapitsky, Y.; Eskuchen, W.J.; Kaler, E.W. Surfactant and polyelectrolyte gel particles that swell reversibly. Langmuir 2006, 22, 6375–6379. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.I. Kinetics of drug release from hydrogel matrices. J. Conrolled Release 1985, 2, 277–288. [Google Scholar] [CrossRef]
- Wood, J.M.; Attwood, D.; Collett, J.H. The swelling properties of poly(2-hydroxyethyl methacrylate) hydrogels polymerized by gamma-irradiation and chemical initiation. Int. J. Pharm. 1981, 7, 189. [Google Scholar] [CrossRef]
- Marmisollé, W.A.; Irigoyen, J.; Gregurec, D.; Moya, S.; Azzaroni, O. Supramolecular surface chemistry: Substrate-independent, phosphate-driven growth of polyamine-based multifunctional thin films. Adv. Funct. Mater. 2015, 25, 4144–4152. [Google Scholar] [CrossRef]
- Yu, J.; Murthy, V.S.; Rana, R.K.; Wong, M.S. Synthesis of nanoparticle-assembled tin oxide/polymer microcapsules. Chem. Commun. 2006, 10, 1097–1099. [Google Scholar] [CrossRef]
- Kikuchi, A.; Okano, T. Pulsatile drug release control using hydrogels. Adv. Drug Deliv. Rev. 2002, 54, 53–77. [Google Scholar] [CrossRef]
- Bansal, M.; Dravid, A.; Aqrawe, Z.; Montgomery, J.; Wu, Z.; Svirskis, D. Conducting polymer hydrogels for electrically responsive drug delivery. J. Control. Release 2020, 328, 192–209. [Google Scholar] [CrossRef]
- Mertz, D.; Harlepp, S.; Goetz, J.; Bégin, D.; Schlatter, G.; Bégin-Colin, S.; Hébraud, A. Nanocomposite polymer scaffolds responding under external stimuli for drug delivery and tissue engineering applications. Adv. Therap. 2020, 3, 1900143. [Google Scholar] [CrossRef]
- Feazel, L.M.; Baumgartner, L.K.; Peterson, K.L.; Frank, D.N.; Harris, J.K.; Pace, N.R. Opportunistic pathogens enriched in showerhead biofilms. Proc. Natl. Acad. Sci. USA 2009, 106, 16393–16399. [Google Scholar] [CrossRef]
- Moat, J.; Rizoulis, A.; Fox, G.; Upton, M. Domestic shower hose biofilms contain fungal species capable of causing opportunistic infection. J. Water Health 2016, 14, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.-Y.; Fei, C.-N.; Zhang, Y.; Liu, J.; Liu, H.; Song, J. Three key factors influencing the bacterial contamination of dental unit waterlines: A 6-year survey from 2012 to 2017. Int. Dent. J. 2019, 69, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.L.; Fontana, S.; Pinci, F.; Fiumana, E.; Pedna, M.F.; Farolfi, P.; Sabattini, M.A.B.; Scaturro, M. Pneumonia associated with a dental unit waterline. Lancet 2012, 379, 684. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hatem, W.A.; Lapitsky, Y. Accelerating Payload Release from Complex Coacervates through Mechanical Stimulation. Polymers 2023, 15, 586. https://doi.org/10.3390/polym15030586
Hatem WA, Lapitsky Y. Accelerating Payload Release from Complex Coacervates through Mechanical Stimulation. Polymers. 2023; 15(3):586. https://doi.org/10.3390/polym15030586
Chicago/Turabian StyleHatem, Wesam A., and Yakov Lapitsky. 2023. "Accelerating Payload Release from Complex Coacervates through Mechanical Stimulation" Polymers 15, no. 3: 586. https://doi.org/10.3390/polym15030586
APA StyleHatem, W. A., & Lapitsky, Y. (2023). Accelerating Payload Release from Complex Coacervates through Mechanical Stimulation. Polymers, 15(3), 586. https://doi.org/10.3390/polym15030586







