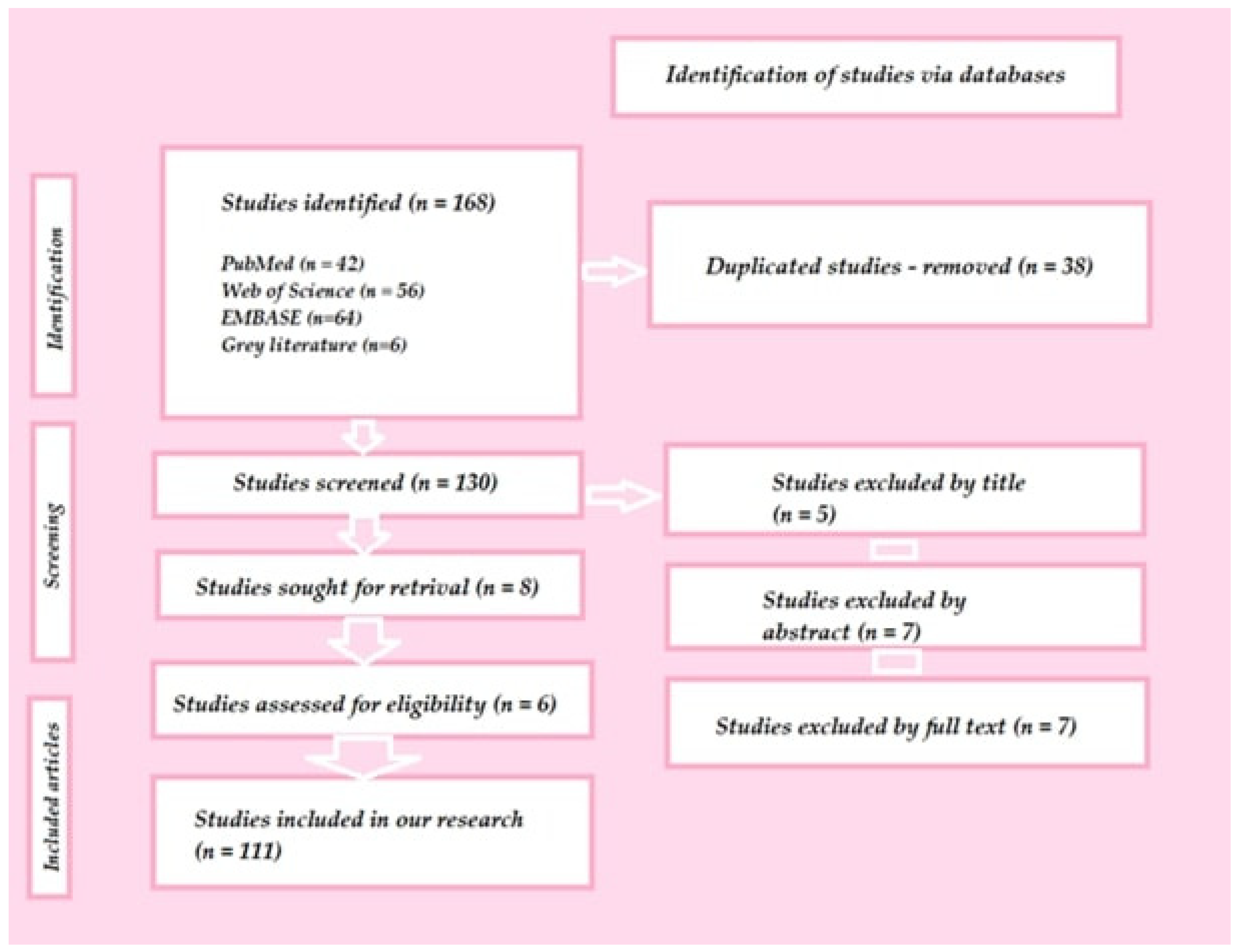
Recent Advances in 3D Printing of Polymers for Application in Prosthodontics
Abstract
:1. Introduction
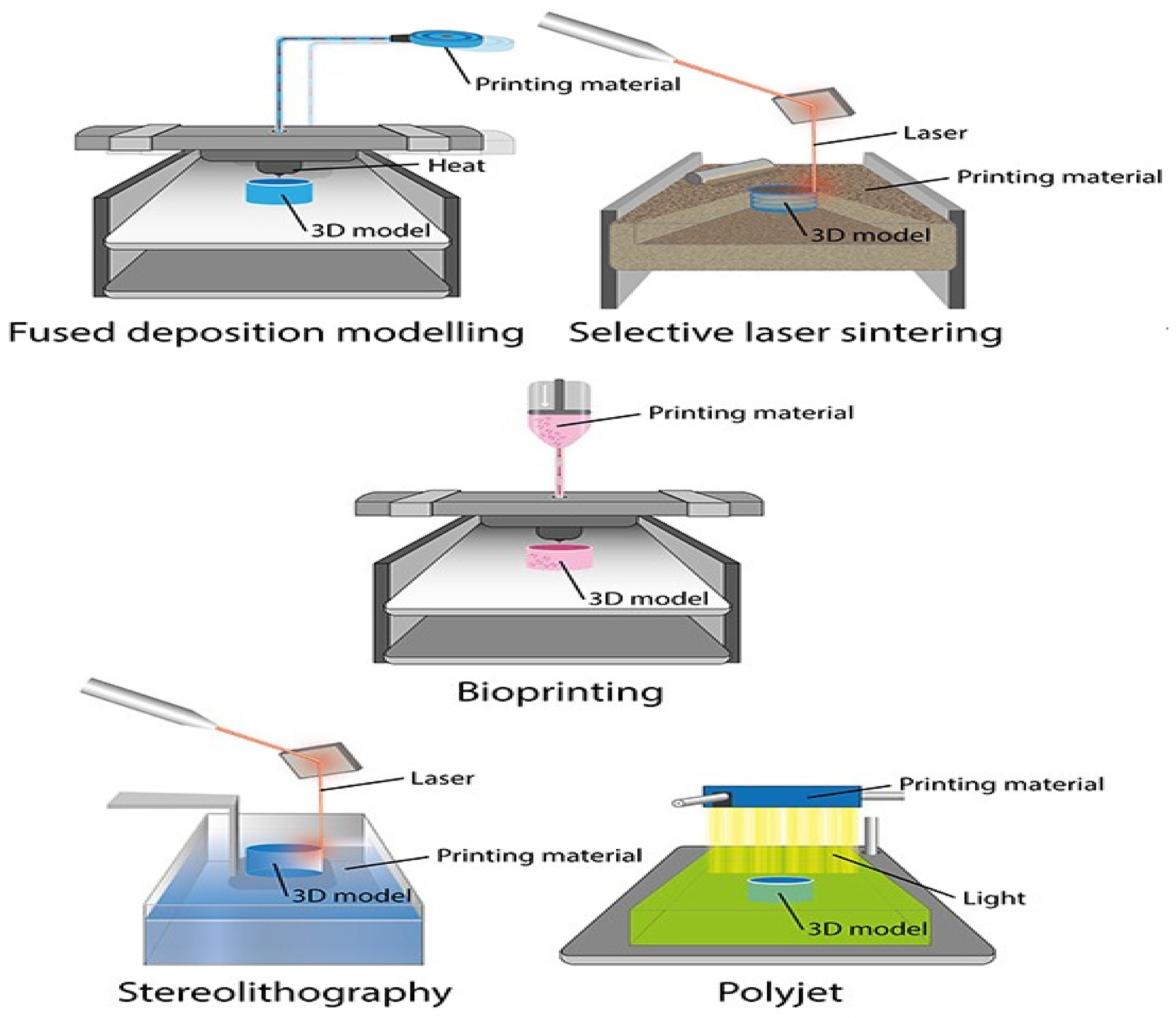
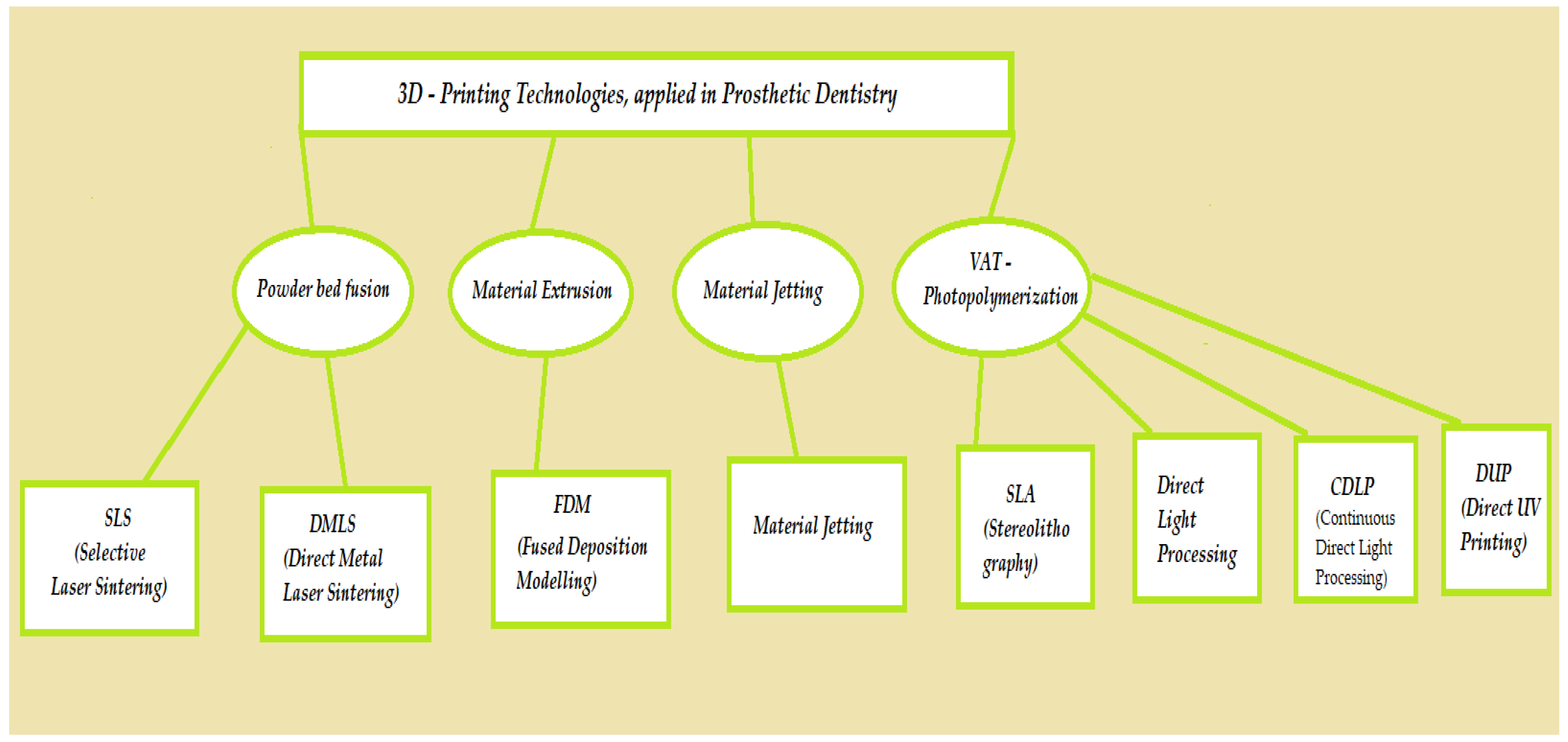
2. General Techniques for 3D Printing in Prosthetic Dentistry
3. Applications of 3D-Printed Polymers in Fixed Prosthodontics
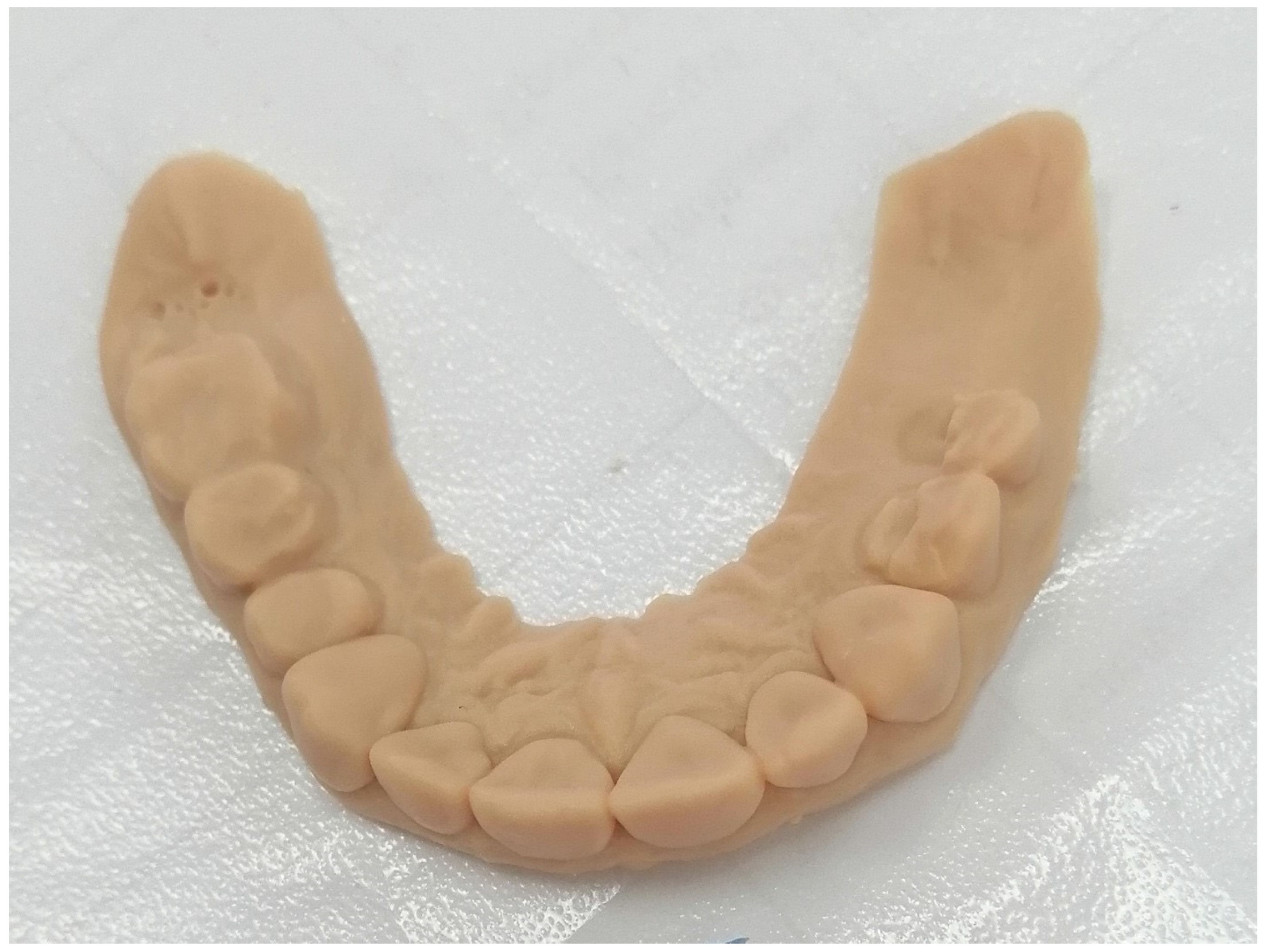
3.1. Fabricating of 3D Models Using Intraoral Scan
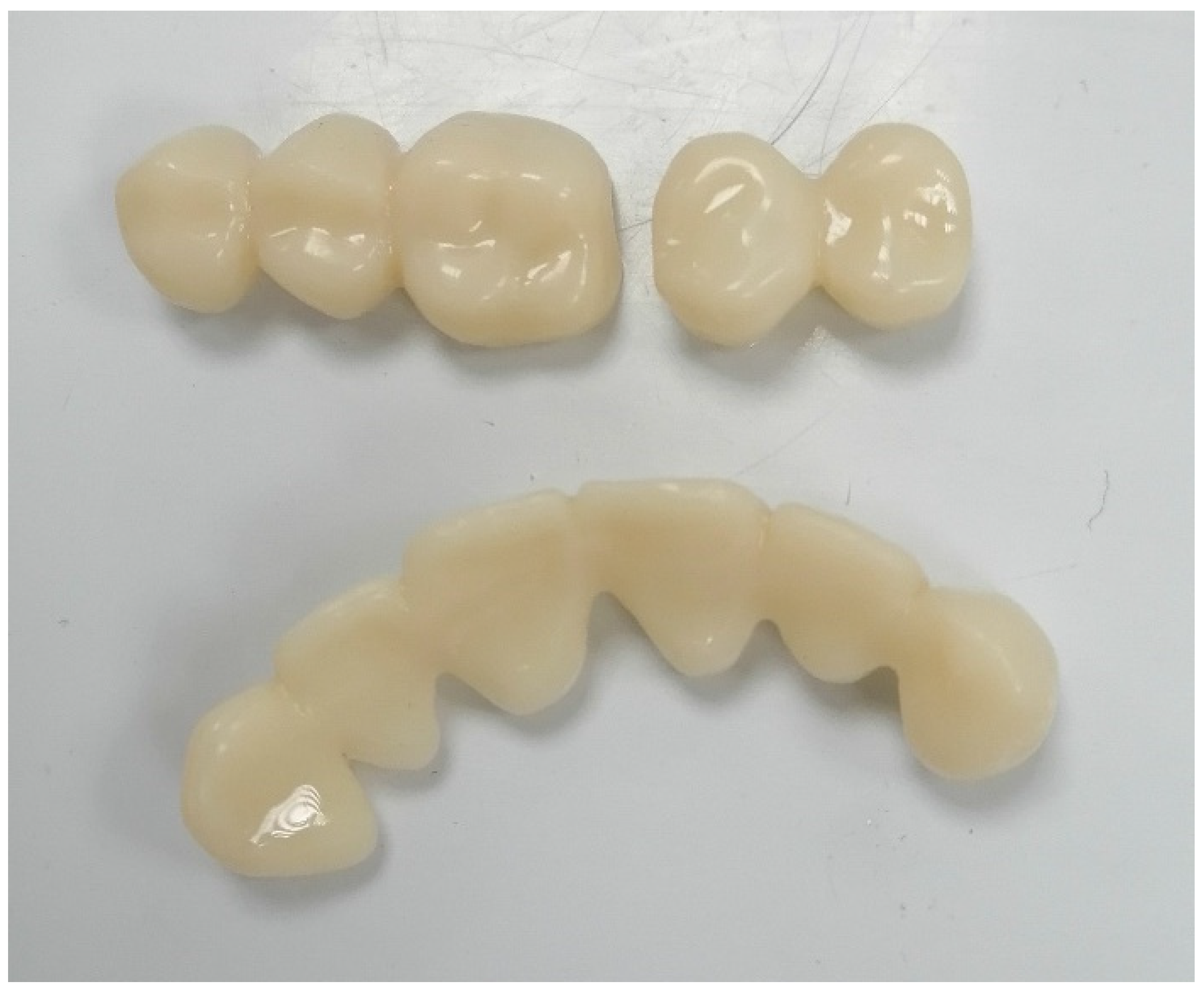
3.2. Fabricating of Temporary Crowns and Fixed Partial Dentures
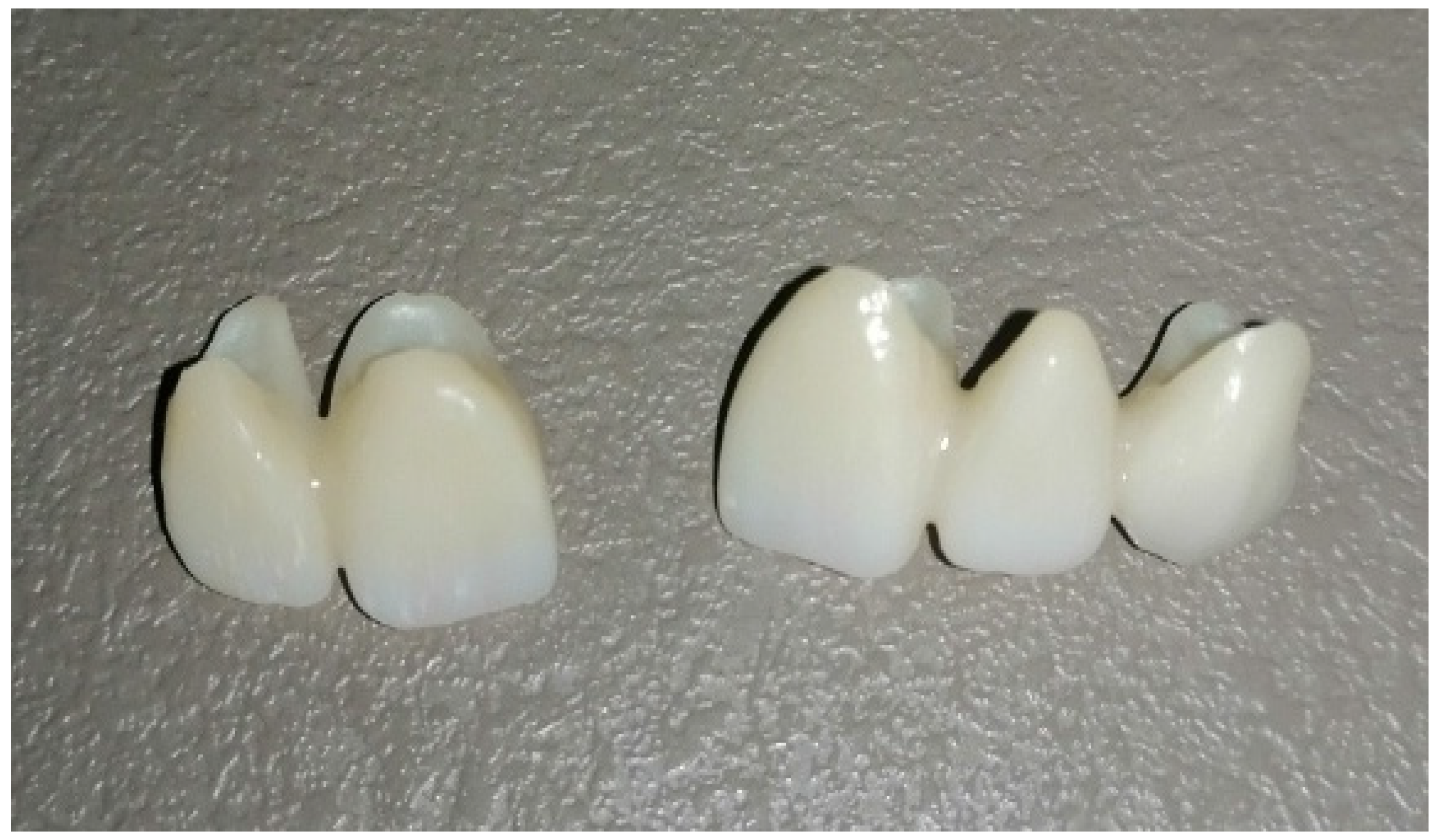
3.3. Fabricating Permanent Crowns, Inlays, Onlays, and Veneers
3.4. Fabricating of Permanent Post and Core Restorations
3.5. Fabricating of Drilling Stents for Guided Implantology
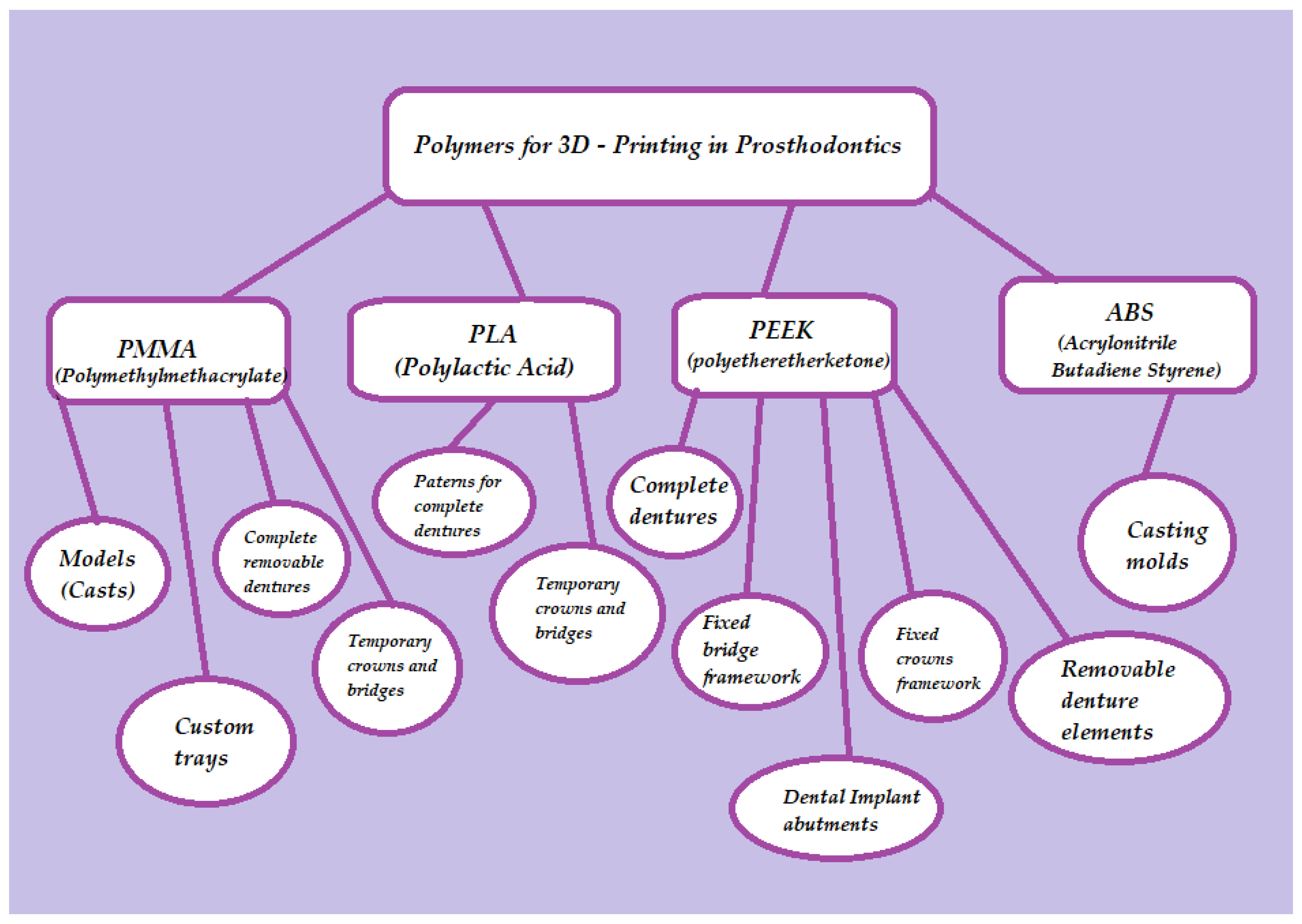
4. Applications of 3D-Printed Polymers in Removable Prosthodontics
4.1. Manufacturing of Custom Trays
4.2. Manufacturing of Denture Bases for Removable Dentures
4.3. Manufacturing of Artificial Teeth for Removable Dentures
4.4. Manufacturing of Occlusal Splints
5. Drawbacks of the Applications of Additive Manufacturing in Prosthetic Dentistry
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABS | acrylonitrile butadiene styrene |
| AM | additive manufacturing |
| CAD/CAM | computer-aided design/computer-aided manufacturing |
| CD | complete dentures |
| ISO | International Organization of Standardization |
| PC | polycarbonate |
| PCL | poly(e-caprolactone) |
| PDMS | polydimethylsiloxane |
| PE | polyethylene |
| PEEK | polyetheretherketone |
| PEG | polyethylene glycol |
| PLA | polylactic acid |
| PMMA | polymethyl methacrylate |
| PP | polypropylene |
| PU | polyurethane |
| STL | stereolithography, standard triangle language, standard tessellation language |
| 3D | three-dimensional |
References
- Thomé, T.; Erhardt, M.C.G.; Leme, A.A.; Al Bakri, I.; Bedran-Russo, A.K.; Bertassoni, L.E. Emerging Polymers in Dentistry. In Advanced Polymers in Medicine; Puoci, F., Ed.; Springer: Cham, Switzerland, 2015; pp. 265–296. [Google Scholar]
- Rokaya, D.; Srimaneepong, V.; Sapkota, J.; Qin, J.; Siraleartmukul, K.; Siriwongrungson, V. Polymeric materials and films in dentistry: An overview. J. Adv. Res. 2018, 14, 25–34. [Google Scholar] [CrossRef]
- Smith, J.R.; Lamprou, D.A. Polymer coatings for biomedical applications: A review. Trans. IMF 2014, 92, 9–19. [Google Scholar] [CrossRef]
- Nathanael, A.J.; Oh, T.H. Biopolymer Coatings for Biomedical Applications. Polymers 2020, 12, 3061. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Zafar, M.S.; Khurshid, Z.; Siddiqui, F. Applications of polyetheretherketone (PEEK) in oral implantology and prosthodontics. J. Prosthodont. Res. 2016, 60, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.; Upadhyay, A.; Khayambashi, P.; Farooq, I.; Sabri, H.; Tarar, M.; Lee, K.T.; Harb, I.; Zhou, S.; Wang, Y.; et al. Dental 3D-Printing: Transferring Art from the Laboratories to the Clinics. Polymers 2021, 13, 157. [Google Scholar] [CrossRef]
- Abduo, J.; Lyons, K.; Bennamoun, M. Trends in Computer-Aided Manufacturing in Prosthodontics: A Review of the Available Streams. Int. J. Dent. 2014, 2014, 783948. [Google Scholar] [CrossRef]
- Clark Ligon, S.; Liska, R.; Stampfl, J.; Gurr, M.; Mulhaupt, R. Polymers for 3D Printing and Customized Additive Manufacturing. Chem. Rev. 2017, 117, 10212–10290. [Google Scholar] [CrossRef]
- Dimitrova, M.; Chuchulska, B.; Zlatev, S.; Kazakova, R. Colour Stability of 3D-Printed and Prefabricated Denture Teeth after Immersion in Different Colouring Agents—An In Vitro Study. Polymers 2022, 14, 3125. [Google Scholar] [CrossRef]
- Tomova, Z.; Zhekov, Y.; Alexandrov, G.; Vlahova, A.; Vasileva, E. Application of CAD/CAM technologies and Materials for Prosthetic Restoration of Severely Damaged Teeth-Clinical Cases. Aust. Dent. J. 2023. ahead of print. [Google Scholar] [CrossRef]
- Ujfalusi, Z.; Pentek, A.; Told, R.; Schiffer, A.; Nyitrai, M.; Maroti, P. Detailed Thermal Characterization of Acrylonitrile Butadiene Styrene and Polylactic Acid Based Carbon Composites Used in Additive Manufacturing. Polymers 2020, 12, 2960. [Google Scholar] [CrossRef]
- Heo, H.; Jin, Y.; Yang, D.; Wier, C.; Minard, A.; Dahotre, N.B.; Neogi, A. Manufacturing and Characterization of Hybrid Bulk Voxelated Biomaterials Printed by Digital Anatomy 3D Printing. Polymers 2021, 13, 123. [Google Scholar] [CrossRef]
- Fully Guided Surgical Stent. Highly Accurate Guides for All Patient Cases and Implant Procedures. Available online: https://www.hiradentallab.com/fully-guided-surgical-stent/ (accessed on 11 November 2023).
- Torabi, K.; Farjood, E.; Hamedani, E. Rapid prototyping technologies and their applications in prosthodontics, a review of literature. J. Dent. 2015, 16, 1–9. [Google Scholar]
- Stansbury, J.W.; Idacavage, M.J. 3D printing with polymers: Challenges among expanding options and opportunities. Dent. Mater. 2016, 32, 54–64. [Google Scholar] [CrossRef]
- Theus, A.S.; Ning, L.; Hwang, B.; Gil, C.; Chen, S.; Wombwell, A.; Mehta, R.; Serpooshan, V. Bioprintability: Physiomechanical and Biological Requirements of Materials for 3D Bioprinting Processes. Polymers 2020, 12, 2262. [Google Scholar] [CrossRef]
- Georgiev, Z.; Alexandrov, S.; Tomova, Z. Experimental setting for testing the bond strength between metal alloy and PEEK. J. IMAB Annu. Proc. Sci. Pap. 2021, 27, 3881–3884. [Google Scholar] [CrossRef]
- Nold, J.; Wesemann, C.; Rieg, L.; Binder, L.; Witkowski, S.; Spies, B.C.; Kohal, R.J. Does Printing Orientation Matter? In-Vitro Fracture Strength of Temporary Fixed Dental Prostheses after a 1-Year Simulation in the Artificial Mouth. Materials 2021, 14, 259. [Google Scholar] [CrossRef] [PubMed]
- Groth, C.; Kravitz, N.D.; Jones, P.E.; Graham, J.W.; Redmond, W.R. Three-dimensional printing technology. J. Clin. Orthod. 2014, 48, 475–485. [Google Scholar] [PubMed]
- Etemad-Shahidi, Y.; Qallandar, O.B.; Evenden, J.; Alifui-Segbaya, F.; Ahmed, K.E. Accuracy of 3-Dimensionally Printed Full-Arch Dental Models: A Systematic Review. J. Clin. Med. 2020, 9, 3357. [Google Scholar] [CrossRef] [PubMed]
- Schweiger, J.; Edelhoff, D.; Güth, J.-F. 3D Printing in Digital Prosthetic Dentistry: An Overview of Recent Developments in Additive Manufacturing. J. Clin. Med. 2021, 10, 2010. [Google Scholar] [CrossRef] [PubMed]
- Herschdorfer, L.; Negreiros, W.; Gallucci, G.; Hamilton, A. Comparison of the accuracy of implants placed with CAD-CAM surgical templates manufactured with various 3D printers: An in vitro study. J. Prosthet. Dent. 2020, 125, 905–910. [Google Scholar] [CrossRef]
- Piedra Cascon, W.; Parra Nunez, A.; Charlen Diez, I.; Revilla León, M. Laboratory workflow to obtain long-term injected resin composite interim restorations from an additive manufactured esthetic diagnostic template. J. Esthet. Restor. Dent. 2019, 31, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Al Mortadi, N.; Jones, Q.; Eggbeer, D.; Lewis, J.; Williams, R.J. Fabrication of a resin appliance with alloy components using digital technology without an analog impression. Am. J. Orthod. Dentofac. Orthop. 2015, 148, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Elomaa, L.; Teixeira, S.; Hakala, R.; Korhonen, H.; Grijpma, D.W.; Seppala, J.V. Preparation of poly(epsilon-caprolactone)-based tissue engineering scaffolds by stereolithography. Acta Biomater. 2011, 7, 3850–3856. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Yeatts, A.; Dean, D.; Fisher, J.P. Stereolithographic bone scaffold design parameters: Osteogenic differentiation and signal expression. Tissue Eng. Part B Rev. 2010, 16, 523–539. [Google Scholar] [CrossRef]
- Skoog, S.A.; Goering, P.L.; Narayan, R.J. Stereolithography in tissue engineering. J. Mater. Sci. Mater. Med. 2014, 25, 845–856. [Google Scholar] [CrossRef]
- Arifin, N.; Sudin, I.; Ngadiman, N.H.A.; Ishak, M.S.A. A Comprehensive Review of Biopolymer Fabrication in Additive Manufacturing Processing for 3D-Tissue-Engineering Scaffolds. Polymers 2022, 14, 2119. [Google Scholar] [CrossRef] [PubMed]
- Osman, R.B.; Alharbi, N.; Wismeijer, D. Build angle: Does it influence the accuracy of 3D-printed dental restorations using digital light-processing technology? Int. J. Prosthodont. 2017, 30, 182–188. [Google Scholar] [CrossRef]
- Ausiello, P.; Gloria, A.; Maietta, S.; Watts, D.C.; Martorelli, M. Stress Distributions for Hybrid Composite Endodontic Post Designs with and without a Ferrule: FEA Study. Polymers 2020, 12, 1836. [Google Scholar] [CrossRef]
- de Andrade, G.-S.; Tribst, J.-P.-M.; Dal Piva, A.-M.O.; Bottino, M.-A.; Borges, A.-L.-S.; Valandro, L.-F.; Özcan, M. A Study on Stress Distribution to Cement Layer and Root Dentin for Post and Cores Made of CAD/CAM Materials with Different Elasticity Modulus in the Absence of Ferrule. J. Clin. Exp. Dent. 2019, 11, e1–e8. [Google Scholar] [CrossRef]
- Ruschel, G.H.; Gomes, É.A.; Silva-Sousa, Y.T.; Pinelli, R.G.P.; Sousa-Neto, M.D.; Pereira, G.K.R.; Spazzin, A.O. Mechanical Properties and Superficial Characterization of a Milled CAD-CAM Glass Fiber Post. J. Mech. Behav. Biomed. Mater. 2018, 82, 187–192. [Google Scholar] [CrossRef]
- Bilgin, M.S.; Erdem, A.; Dilber, E.; Ersoy, İ. Comparison of Fracture Resistance between Cast, CAD/CAM Milling, and Direct Metal Laser Sintering Metal Post Systems. J. Prosthodont. Res. 2016, 60, 23–28. [Google Scholar] [CrossRef]
- Lalama, M.; Rocha, M.G.; O’Neill, E.; Zoidis, P. Polyetheretherketone (PEEK) Post and Core Restorations: A 3D Accuracy Analysis between Heat-Pressed and CAD-CAM Fabrication Methods. J. Prosthodont. 2022, 31, 537–542. [Google Scholar] [CrossRef]
- Teixeira, K.N.; Duque, T.M.; Maia, H.P.; Gonçalves, T. Fracture Resistance and Failure Mode of Custom-Made Post-and-Cores of Polyetheretherketone and Nano-Ceramic Composite. Oper. Dent. 2020, 45, 506–515. [Google Scholar] [CrossRef]
- Uctasli, S.; Boz, Y.; Sungur, S.; Vallittu, P.K.; Garoushi, S.; Lassila, L. Influence of Post-Core and Crown Type on the Fracture Resistance of Incisors Submitted to Quasistatic Loading. Polymers 2021, 13, 1130. [Google Scholar] [CrossRef]
- Iemsaengchairat, R.; Aksornmuang, J. Fracture Resistance of Thin Wall Endodontically Treated Teeth without Ferrules Restored with Various Techniques. J. Esthet. Restor. Dent. 2022, 34, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Sadowsky, S.J. Comments Regarding: Batista VES, Bitencourt SB, Bastos NA, Pellizzer EP, Goiato MC, Dos Santos DM. Influence of the Ferrule Effect on the Failure of Fiber-Reinforced Composite Post-and-Core Restorations: A Systematic Review and Meta-Analysis. J Prosthet Dent 2020;123:239-45. J. Prosthet. Dent. 2021, 125, 554. [Google Scholar]
- Garcia, P.P.; Wambier, L.M.; de Geus, J.L.; da Cunha, L.F.; Correr, G.M.; Gonzaga, C.C. Do Anterior and Posterior Teeth Treated with Post-and-Core Restorations Have Similar Failure Rates? A Systematic Review and Meta-Analysis. J. Prosthet. Dent. 2019, 121, 887–894.e4. [Google Scholar] [CrossRef] [PubMed]
- Mosharraf, R.; Fathi, A.; Botshekan, S.S. Comparative Evaluation of the Effect of Core Type and Antirotational Post on Stress Distribution in an Endodontically Treated Maxillary First Molar: FEA. Int. J. Dent. 2022, 2022, 4336980. [Google Scholar] [CrossRef] [PubMed]
- Al-Qarni, F.D. Customized Post and Cores Fabricated with CAD/CAM Technology: A Literature Review. Int. J. Gen. Med. 2022, 15, 4771–4779. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-S.; Shin, J.-H.; Kim, J.-E.; Kim, J.-H.; Lee, W.-C.; Shin, S.-W.; Lee, J.-Y. Biomechanical Evaluation of a Tooth Restored with High Performance Polymer PEKK Post-Core System: A 3D Finite Element Analysis. Biomed Res. Int. 2017, 2017, 1373127. [Google Scholar] [CrossRef] [PubMed]
- Dimitrova, M.; Vlahova, A.; Hristov, I.; Kazakova, R.; Chuchulska, B.; Kazakov, S.; Forte, M.; Granberg, V.; Barile, G.; Capodiferro, S.; et al. Evaluation of Water Sorption and Solubility of 3D-Printed, CAD/CAM Milled, and PMMA Denture Base Materials Subjected to Artificial Aging. J. Compos. Sci. 2023, 7, 339. [Google Scholar] [CrossRef]
- Heboyan, A.; Zafar, M.S.; Karobari, M.I.; Tribst, J.P.M. Insights into Polymeric Materials for Prosthodontics and Dental Implantology. Materials 2022, 15, 5383. [Google Scholar] [CrossRef] [PubMed]
- Hadjieva, H.; Dimova, M.; Hadjieva, E.; Todorov, S. Changes in the vertical dimension of occlusion during the different periods of complete denture wear—A comparative study. J. IMAB Annu. Proc. Sci. Pap. 2014, 20, 546–549. [Google Scholar] [CrossRef]
- Falcão Spina, D.R.; da Costa, R.G.; Correr, G.M.; Rached, R.N. Scanning of Root Canal Impression for the Fabrication of a Resin CAD-CAM-Customized Post-and-Core. J. Prosthet. Dent. 2018, 120, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Sun, J.; Jiang, L.; Wu, Y.; He, J.; Ruan, W.; Yan, W. Influence of Margin Design and Restorative Material on the Stress Distribution of Endocrowns: A 3D Finite Element Analysis. BMC Oral Health 2022, 22, 30. [Google Scholar] [CrossRef]
- Lee, H.; Son, K.B.D.; Lee, D.-H.; Kim, S.-Y.; Lee, K.-B. Comparison of wear of interim crowns in accordance with the build angle of digital light processing 3-dimensional printing: A pilot in vivo study. Res. Sq. 2022, 9, 417. [Google Scholar] [CrossRef]
- Williams, R.J.; Bibb, R.; Rafik, T. A technique for fabricating patterns for removable partial denture frameworks using digitized casts and electronic surveying. J. Prosthet. Dent. 2004, 91, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-S.; Harris, B.T.; Pellerito, J.; Morton, D. Fabrication of an interim complete removable dental prosthesis with an in-office digital light processing three-dimensional printer: A proof-of-concept technique. J. Prosthet. Dent. 2018, 120, 331–334. [Google Scholar] [CrossRef]
- Bilgin, M.S.; Erdem, A.; Aglarci, O.S.; Dilber, E. Fabricating complete dentures with CAD/CAM and RP technologies. J. Prosthodont. 2015, 24, 576–579. [Google Scholar] [CrossRef]
- Revilla-León, M.; Besné-Torre, A.; Sánchez-Rubio, J.L.; Fábrega, J.J.; Özcan, M. Digital tools and 3D printing technologies integrated into the workflow of restorative treatment: A clinical report. J. Prosthet. Dent. 2019, 121, 3–8. [Google Scholar] [CrossRef]
- Revilla-León, M.; Sánchez-Rubio, J.L.; Oteo-Calatayud, J.; Özcan, M. Impression technique for a complete-arch prosthesis with multiple implants using additive manufacturing technologies. J. Prosthet. Dent. 2017, 117, 714–720. [Google Scholar] [CrossRef]
- Bagheri Saed, A.; Behravesh, A.H.; Hasannia, S.; Alavinasab Ardebili, S.A.; Akhoundi, B.; Pourghayoumi, M. Functionalized poly l-lactic acid synthesis and optimization of process parameters for 3D printing of porous scaffolds via digital light processing (DLP) method. J. Manuf. Process. 2020, 56, 550–561. [Google Scholar] [CrossRef]
- Prasad, A.; Kandasubramanian, B. Fused Deposition Processing Polycaprolactone of Composites for Biomedical Applications. Polym. Plast. Technol. Mater. 2019, 58, 1365–1398. [Google Scholar] [CrossRef]
- Rocha, C.R.; Perez, A.R.T.; Roberson, D.A.; Shemelya, C.M.; MacDonald, E.; Wicker, R.B. Novel ABS-based binary and ternary polymer blends for material extrusion 3D printing. J. Mater. Res. 2014, 29, 1859–1866. [Google Scholar] [CrossRef]
- Rebong, R.E.; Stewart, K.T.; Utreja, A.; Ghoneima, A.A. Accuracy of three-dimensional dental resin models created by fused deposition modeling, stereolithography, and Polyjet prototype technologies: A comparative study. Angle Orthod. 2018, 88, 363–369. [Google Scholar] [CrossRef]
- Pan, Y.; Zhou, C.; Chen, Y. A Fast Mask Projection Stereolithography Process for Fabricating Digital Models in Minutes. J. Manuf. Sci. Eng. 2012, 134, 051011. [Google Scholar] [CrossRef]
- Hazeveld, A.; Huddleston Slater, J.J.R.; Ren, Y. Accuracy and reproducibility of dental replica models reconstructed by different rapid prototyping techniques. Am. J. Orthod. Dentofac. Orthop. 2014, 145, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Greco, G.B.; Popi, D.; Di Stefano, D.A. Accuracy of 3-dimensional printing of dental casts: A proposal for quality standardization. J. Prosthet. Dent. 2022, 127, 899–910. [Google Scholar] [CrossRef]
- Alharbi, N.; Osman, R.; Wismeijer, D. Effects of build direction on the mechanical properties of 3D-printed complete coverage interim dental restorations. J. Prosthet. Dent. 2016, 115, 760–767. [Google Scholar] [CrossRef]
- Li, X.; Xie, B.; Jin, J.; Chai, Y.; Chen, Y. 3D Printing Temporary Crown and Bridge by Temperature Controlled Mask Image Projection Stereolithography. Procedia Manuf. 2018, 26, 1023–1033. [Google Scholar] [CrossRef]
- Dikova, T. Production of high-quality temporary crowns and bridges by stereolithography. Scr. Sci. Med. Dent. 2019, 5, 33–38. [Google Scholar] [CrossRef]
- Dimitrova, M.; Corsalini, M.; Kazakova, R.; Vlahova, A.; Chuchulska, B.; Barile, G.; Capodiferro, S.; Kazakov, S. Comparison between Conventional PMMA and 3D Printed Resins for Denture Bases: A Narrative Review. J. Compos. Sci. 2022, 6, 87. [Google Scholar] [CrossRef]
- Hada, T.; Kanazawa, M.; Iwaki, M.; Arakida, T.; Soeda, Y.; Katheng, A.; Otake, R.; Minakuchi, S. Effect of printing direction on the accuracy of 3D-printed dentures using stereolithography technology. Materials 2020, 13, 3405. [Google Scholar] [CrossRef] [PubMed]
- Piedra Cascon, W.; Revilla-León, M. Digital workflow for the design and additively manufacture of a splinted framework and custom tray for the impression of multiple implants: A dental technique. J. Prosthet. Dent. 2018, 120, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Makvandi, P.; Esposito Corcione, C.; Paladini, F.; Gallo, A.L.; Montagna, F.; Jamaledin, R.; Pollini, M.; Maffezzoli, A. Antimicrobial modified hydroxyapatite composite dental bite by stereolithography. Polym. Adv. Technol. 2018, 29, 364–371. [Google Scholar] [CrossRef]
- Yu, J.H.; Wang, Y.T.; Lin, C.L. Customized surgical template fabrication under biomechanical consideration by integrating CBCT image, CAD system and finite element analysis. Dent. Mater. J. 2018, 37, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Luebbers, H.; Agbaje, J.O.; Schepers, S.; Politis, C.; Van Slycke, S.; Vrielinck, L. Accuracy of dental implant placement using CBCT-derived mucosa-supported stereolithographic template. Clin. Implant Dent. Relat. Res. 2015, 17, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Ersoy, A.E.; Turkyilmaz, I.; Ozan, O.; McGlumphy, E.A. Reliability of implant placement with stereolithographic surgical guides generated from computed tomography: Clinical data from 94 implants. J. Periodontol. 2008, 79, 1339–1345. [Google Scholar] [CrossRef]
- Cassetta, M.; Di Carlo, S.; Pranno, N.; Sorrentino, V.; Di Giorgio, G.; Pompa, G. The use of stereolithographic surgical templates in oral implantology. Ann. Ital. Chir. 2013, 84, 589–593. [Google Scholar]
- Ozan, O.; Turkyilmaz, I.; Ersoy, A.E.; McGlumphy, E.A.; Rosenstiel, S.F. Clinical accuracy of 3 different types of computed tomography-derived stereolithographic surgical guides in implant placement. J. Oral Maxillofac. Surg. 2009, 67, 394–401. [Google Scholar] [CrossRef]
- D’haese, J.; Van De Velde, T.; Komiyama, A.; Hultin, M.; De Bruyn, H. Accuracy and complications using computer-designed stereolithographic surgical guides for oral rehabilitation by means of dental implants: A review of the literature. Clin. Implant Dent. Relat. Res. 2012, 14, 321–335. [Google Scholar] [CrossRef]
- Werz, S.M.; Zeichner, S.J.; Berg, B.I.; Zeilhofer, H.F.; Thieringer, F. 3D printed surgical simulation models as educational tool by maxillofacial surgeons. Eur. J. Dent. Educ. 2018, 22, e500–e505. [Google Scholar] [CrossRef] [PubMed]
- Park, M.E.; Shin, S.Y. Three-dimensional comparative study on the accuracy and reproducibility of dental casts fabricated by 3D printers. J. Prosthet. Dent. 2018, 119, 861.e1–861.e7. [Google Scholar] [CrossRef] [PubMed]
- Mai, H.-N.; Lee, K.-B.; Lee, D.-H. Fit of interim crowns fabricated using photopolymer-jetting 3D printing. J. Prosthet. Dent. 2017, 118, 208–215. [Google Scholar] [CrossRef]
- Bibb, R.; Eggbeer, D.; Williams, R. Rapid manufacture of removable partial denture frameworks. Rapid Prototyp. J. 2006, 12, 95–99. [Google Scholar] [CrossRef]
- Bibb, R.J.; Eggbeer, D.; Williams, R.; Woodward, A. Trial fitting of a removable partial denture framework made using computer-aided design and rapid prototyping techniques. Proc. Inst. Mech. Eng. H J. Eng. Med. 2006, 220, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Inokoshi, M.; Kanazawa, M.; Minakuchi, S. Evaluation of a complete denture trial method applying rapid prototyping. Dent. Mater. J. 2012, 31, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bei, H.-P.; Zhao, M.; Dong, Z.; Zhao, X. Shedding light on 3D printing: Printing photo-crosslinkable constructs for tissue engineering. Biomaterials 2022, 286, 121566. [Google Scholar] [CrossRef]
- Roi, A.; Ardelean, L.C.; Roi, C.I.; Boia, E.-R.; Boia, S.; Rusu, L.-C. Oral Bone Tissue Engineering: Advanced Biomaterials for Cell Adhesion, Proliferation and Differentiation. Materials 2019, 12, 2296. [Google Scholar] [CrossRef]
- Papaioannou, T.G.; Manolesou, D.; Dimakakos, E.; Tsoucalas, G.; Vavuranakis, M.; Tousoulis, D. 3D Bioprinting Methods and Techniques: Applications on Artificial Blood Vessel Fabrication. Acta Cardiol. Sin. 2019, 35, 284–289. [Google Scholar]
- Ozbolat, I.T.; Hospodiuk, M. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials 2016, 76, 321–343. [Google Scholar] [CrossRef]
- Kyle, S.; Jessop, Z.M.; Al-Sabah, A.; Whitaker, I.S. ‘Printability’ of Candidate Biomaterials for Extrusion Based 3D Printing: State-of-the-Art. Adv. Healthc. Mater. 2017, 6, 1700264. [Google Scholar] [CrossRef]
- He, Y.; Yang, F.; Zhao, H.; Gao, Q.; Xia, B.; Fu, J. Research on the printability of hydrogels in 3D bioprinting. Sci. Rep. 2016, 6, 29977. [Google Scholar] [CrossRef] [PubMed]
- Hölzl, K.; Lin, S.; Tytgat, L.; van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink properties before, during and after 3D bioprinting. Biofabrication 2016, 8, 032002. [Google Scholar] [CrossRef] [PubMed]
- New Shade NextDent Denture 3D+ Classic Pink is Now Available! Available online: https://nextdent.com/new-shade-nextdent-denture-3d-classic-pink-is-now-available/ (accessed on 7 November 2023).
- Ning, L.; Chen, X. A brief review of extrusion-based tissue scaffold bio-printing. Biotechnol. J. 2017, 12, 1600671. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.F.; Vaquette, C.; Zhang, Q.; Reis, R.L.; Ivanovski, S.; Hutmacher, D.W. Advanced tissue engineering scaffold design for regeneration of the complex hierarchical periodontal structure. J. Clin. Periodontol. 2014, 41, 283–294. [Google Scholar] [CrossRef]
- Sun, W.; Starly, B.; Daly, A.C.; Burdick, J.A.; Groll, J.; Skeldon, G.; Shu, W.; Sakai, Y.; Shinohara, M.; Nishikawa, M.; et al. The bioprinting roadmap. Biofabrication 2020, 12, 022002. [Google Scholar] [CrossRef]
- Raman, R.; Bashir, R. Stereolithographic 3D Bioprinting for Biomedical Applications. In Essentials of 3D Biofabrication and Translation; Atala, A., Yoo, J.J., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 89–121. [Google Scholar]
- Kim, S.H.; Yeon, Y.K.; Lee, J.M.; Chao, J.R.; Lee, Y.J.; Seo, Y.B.; Sultan, M.T.; Lee, O.J.; Lee, J.S.; Yoon, S.I.; et al. Precisely printable and biocompatible silk fibroin bioink for digital light processing 3D printing. Nat. Commun. 2018, 9, 1620. [Google Scholar] [CrossRef] [PubMed]
- Rider, P.; Kacarevic, Z.P.; Alkildani, S.; Retnasingh, S.; Barbeck, M. Bioprinting of tissue engineering scaffolds. J. Tissue Eng. 2018, 9, 2041731418802090. [Google Scholar] [CrossRef]
- Dimova-Gabrovska, M.; Dimitrova, D.; Mitronin, V. Removable Prosthetic Treatment in children—Literature review. J. IMAB Annu. Proc. Sci. Pap. 2018, 24, 2172–2176. [Google Scholar] [CrossRef]
- Saunders, R.E.; Derby, B. Inkjet printing biomaterials for tissue engineering: Bioprinting. Int. Mater. Rev. 2014, 59, 430–448. [Google Scholar] [CrossRef]
- Guillotin, B.; Souquet, A.; Catros, S.; Duocastella, M.; Pippenger, B.; Bellance, S.; Bareille, R.; Remy, M.; Bordenave, L.; Amedee, J.; et al. Laser-assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials 2010, 31, 7250–7256. [Google Scholar] [CrossRef]
- Guillotin, B.; Ali, M.; Ducom, A.; Catros, S.; Keriquel, V.; Souquet, A.; Remy, M.; Fricain, J.-C.; Guillemot, F. Laser-Assiste Bioprinting for Tissue Engineering. In Biofabrication Micro- and Nano-Fabrication, Printing, Patterning, and Assemblies; Forgacs, G., Sun, W., Eds.; William Andrew Publishing: Boston, MA, USA, 2013; pp. 95–118. [Google Scholar]
- An, J.; Teoh, J.E.M.; Suntornnond, R.; Chua, C.K. Design and 3D Printing of Scaffolds and Tissues. Engineering 2015, 1, 261–268. [Google Scholar] [CrossRef]
- Tzeng, J.-J.; Yang, T.-S.; Lee, W.-F.; Chen, H.; Chang, H.-M. Mechanical Properties and Biocompatibility of Urethane Acrylate-Based 3D-Printed Denture Base Resin. Polymers 2021, 13, 822. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Li, J.; He, Z.; Hong, J.; Bao, J. Urethane acrylate-based photosensitive resin for three-dimensional printing of stereolitho graphic elastomer. J. Appl. Polym. Sci. 2020, 137, 49294. [Google Scholar] [CrossRef]
- Permanent Crown Resin 1 kg (0.7 L). Available online: https://formlabs.com/store/materials/permanent-crown-resin-us/ (accessed on 8 November 2023).
- Khorsandi, D.; Fahimipour, A.; Abasian, P.; Saber, S.S.; Seyedi, M.; Ghanavati, S.; Ahmad, A.; De Stephanis, A.A.; Taghavinezhad-dilami, F.; Leonova, A.; et al. 3D and 4D printing in dentistry and maxillofacial surgery: Printing techniques, materials, and applications. Acta Biomater. 2021, 122, 26–49. [Google Scholar] [CrossRef] [PubMed]
- Réalisation D’un Inlay-Core en PEEK: Une Nouvelle Indication Pour ce Matériau. Available online: https://www.information-dentaire.fr/formations/realisation-d-un-inlay-core-en-peekune-nouvelle-indication-pour-ce-materiau/ (accessed on 10 November 2023).
- Rusu, L.-C.; Ardelean, L.C.; Jitariu, A.-A.; Miu, C.A.; Streian, C.G. An Insight into the Structural Diversity and Clinical Applicability of Polyurethanes in Biomedicine. Polymers 2020, 12, 1197. [Google Scholar] [CrossRef]
- Chen, H.; Lee, S.-Y.; Lin, Y.-M. Synthesis and formulation of PCL-based urethane acrylates for DLP 3D printers. Polymers 2020, 12, 1500. [Google Scholar] [CrossRef]
- Ngo, T.; Kashani, A.; Imbalzano, G.; Nguyen, K.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Molinero-Mourelle, P.; Canals, S.; Gómez-Polo, M.; Fernanda Solá-Ruiz, M.; del Río Highsmith, J.; Celemín Viñuela, A.; Solá-Ruiz, M.F.; Viñuela, A.C. Polylactic acid as a material for three-dimensional printing of provisional restorations. Int. J. Prosthodont. 2018, 31, 349–350. [Google Scholar] [CrossRef]
- Benli, M.; Eker-Gümü¸s, B.; Kahraman, Y.; Huck, O.; Özcan, M. Can polylactic acid be a CAD/CAM material for provisional crown restorations in terms of fit and fracture strength? Dent. Mater. J. 2021, 40, 772–780. [Google Scholar] [CrossRef]
- 3D Printed Splints vs. Thermoformed. Available online: https://www.designerdentallab.com/single-post/2018/01/09/3d-printed-splints-vs-thermoformed (accessed on 6 November 2023).
- Ilyas, R.A.; Zuhri, M.Y.M.; Aisyah, H.A.; Asyraf, M.R.M.; Hassan, S.A.; Zainudin, E.S.; Sapuan, S.M.; Sharma, S.; Bangar, S.P.; Jumaidin, R.; et al. Natural Fiber-Reinforced Polylactic Acid, Polylactic Acid Blends and Their Composites for Advanced Applications. Polymers 2022, 14, 202. [Google Scholar] [CrossRef] [PubMed]
- Bayraktar, G.; Guvener, B.; Bural, C.; Uresin, Y. Influence of polymerization method, curing process, and length of time of storage in water on the residual methyl methacrylate content in dental acrylic resins. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 76, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Braden, M. Some Aspects of the Chemistry and Physics of Dental Resins. Adv. Dent. Res. 1988, 2, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Gautam, R.; Singh, R.D.; Sharma, V.P.; Siddhartha, R.; Chand, P.; Kumar, R. Biocompatibility of polymethylmethacrylate resins used in dentistry. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1444–1450. [Google Scholar] [CrossRef] [PubMed]
- Goiato, M.C.; Freitas, E.; dos Santos, D.; de Medeiros, R.; Sonego, M. Acrylic resin cytotoxicity for denture base—Literature review. Adv. Clin. Exp. Med. 2015, 24, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.D.; Gautam, R.; Siddhartha, R.; Singh, B.P.; Chand, P.; Sharma, V.P.; Jurel, S.K. High performance liquid chromatographic determination of residual monomer released from heat-cured acrylic resin. An in vivo study. J. Prosthodont. 2013, 22, 358–361. [Google Scholar] [CrossRef]
- Wedekind, L.; Güth, J.-F.; Schweiger, J.; Kollmuss, M.; Reichl, F.-X.; Edelhoff, D.; Högg, C. Elution behavior of a 3D-printed, milled and conventional resin-based occlusal splint material. Dent. Mater. 2021, 37, 701–710. [Google Scholar] [CrossRef]
- Kurt, A.; Altintas, S.H.; Kiziltas, M.V.; Tekkeli, S.E.; Guler, E.M.; Kocyigit, A.; Usumez, A. Evaluation of residual monomer release and toxicity of self-adhesive resin cements. Dent. Mater. J. 2018, 37, 40–48. [Google Scholar] [CrossRef]
- Urban, V.M.; Machado, A.L.; Vergani, C.E.; Giampaolo, E.T.; Pavarina, A.C.; de Almeida, F.G.; Cass, Q.B. Effect of water-bath post-polymerization on the mechanical properties, degree of conversion, and leaching of residual compounds of hard chairside reline resins. Dent. Mater. 2009, 25, 662–671. [Google Scholar] [CrossRef]
- Steinmassl, P.-A.; Wiedemair, V.; Huck, C.; Klaunzer, F.; Steinmassl, O.; Grunert, I.; Dumfahrt, H. Do CAD/CAM dentures really release less monomer than conventional dentures? Clin. Oral Investig. 2017, 21, 1697–1705. [Google Scholar] [CrossRef]
- Zhang, C.-Y.; Cheng, Y.-L.; Tong, X.-W.; Yu, H.; Cheng, H. In vitro cytotoxicity of self-adhesive dual-cured resin cement polymerized beneath three different cusp inclinations of zirconia. BioMed Res. Int. 2019, 2019, 7404038. [Google Scholar] [CrossRef] [PubMed]
- Wataha, J.C. Predicting clinical biological responses to dental materials. Dent. Mater. 2012, 28, 23–40. [Google Scholar] [CrossRef] [PubMed]
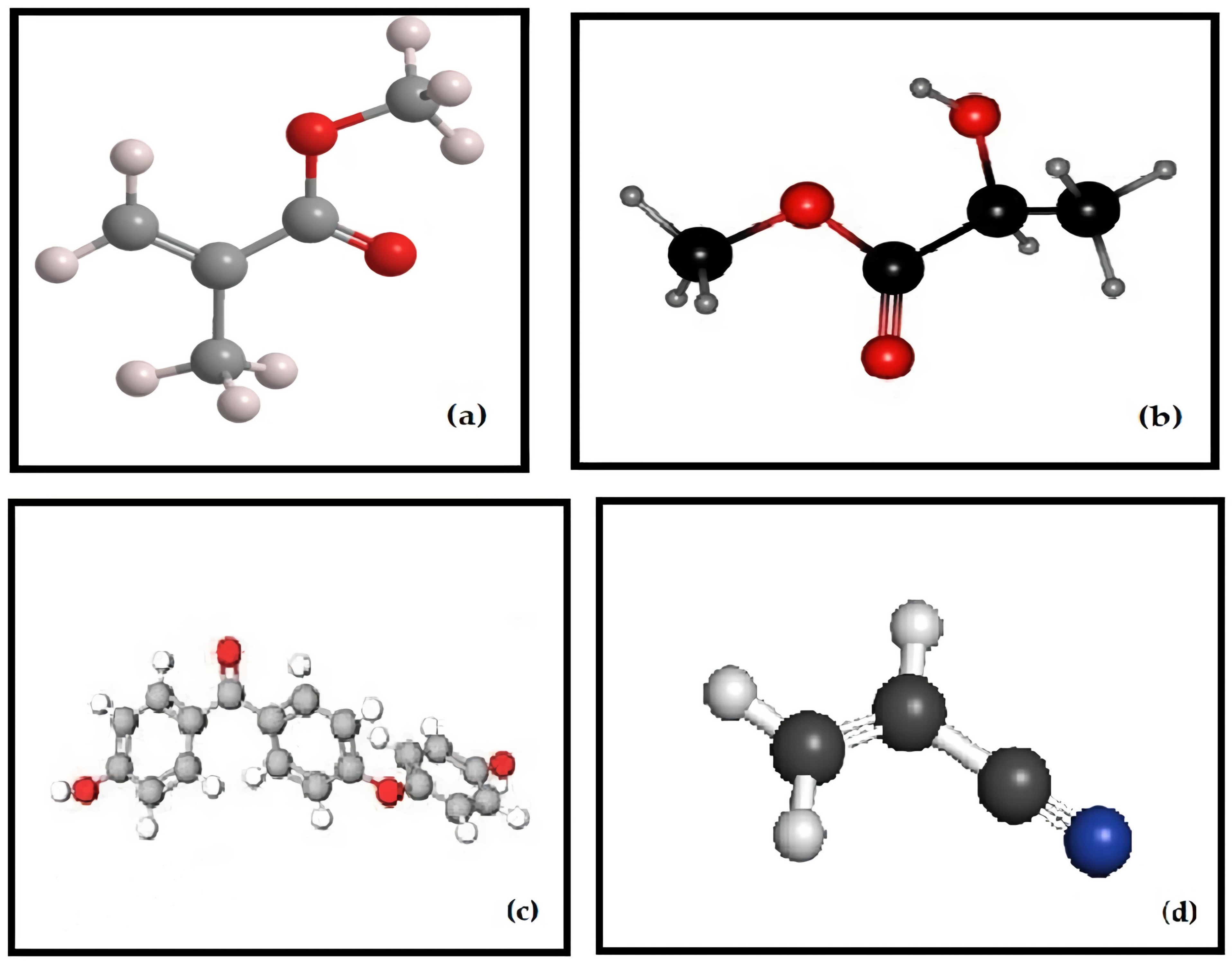
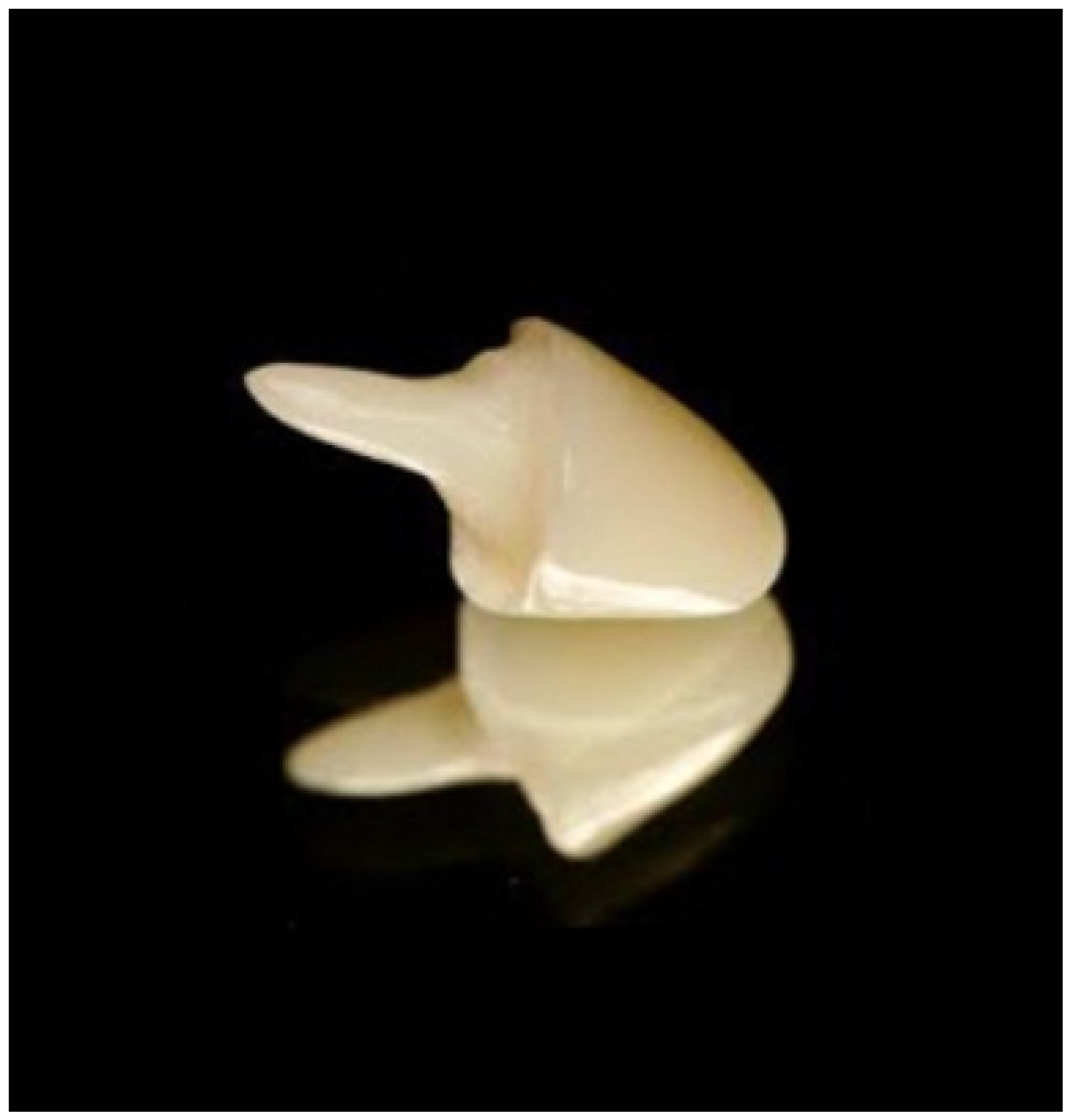
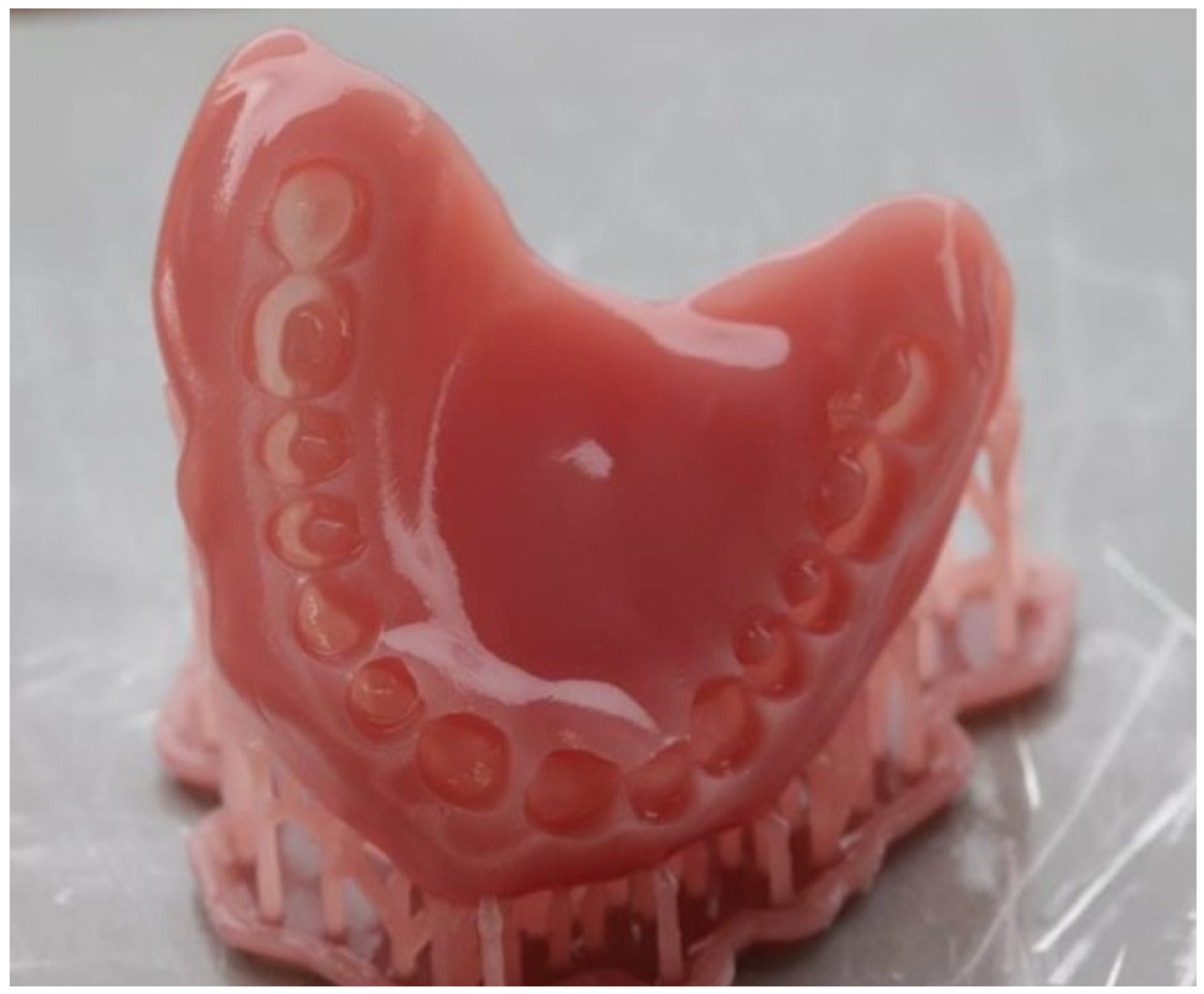

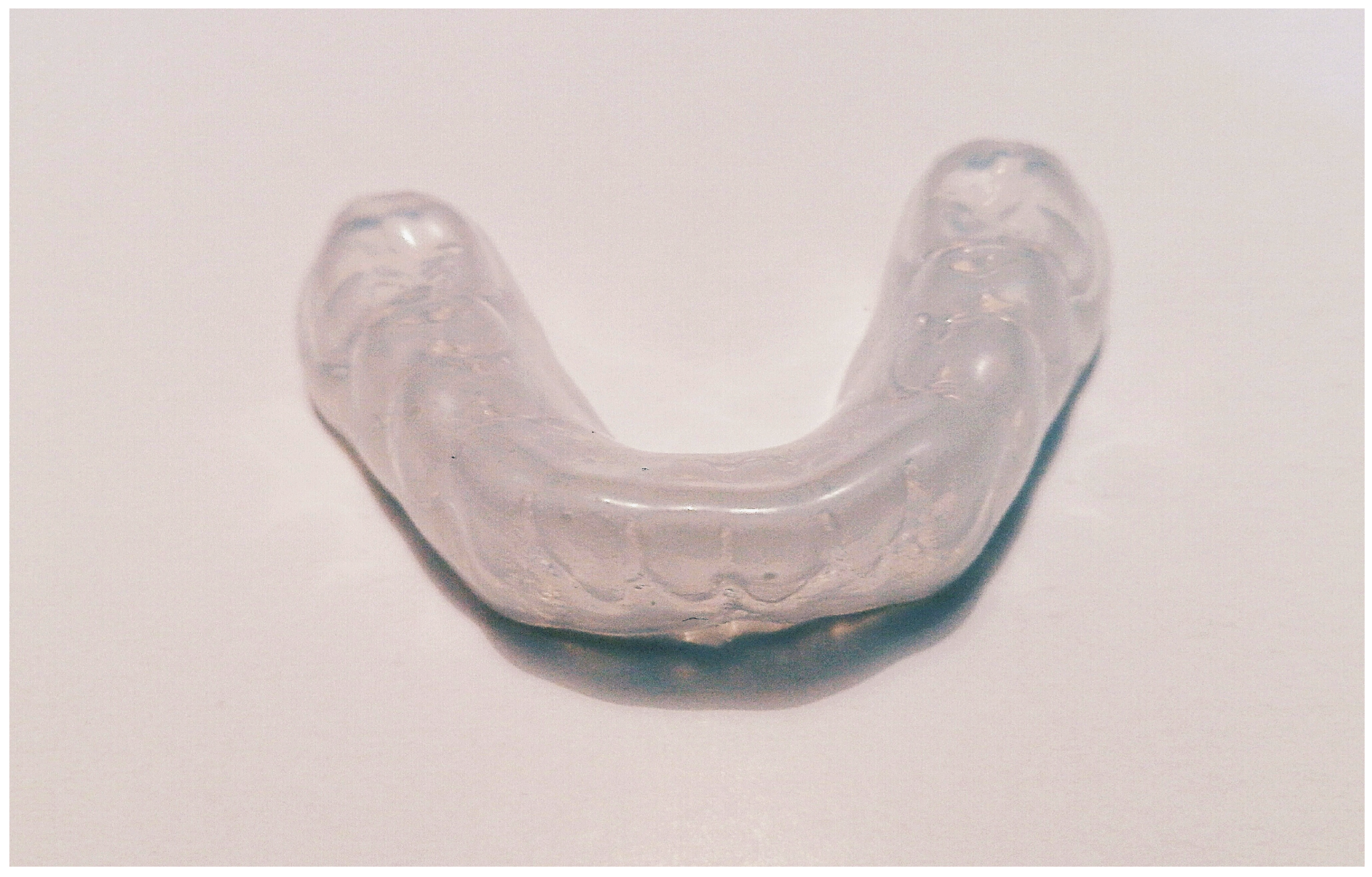
| Type of Polymer | Processing Method | Characteristics |
|---|---|---|
| Methacrylic Acid (Formlabs: Dental SG) | Stereolithography | Elastic Modulus: 1670 MPa; Orientations of 0° to 90°. |
| Polylactic acid (PLA) | Fused deposition modeling | Tensile Strength: 28–56 Mpa; Elastic Modulus: 2000 Mpa; Orientations of 0° to 90° |
| Polyether ether ketone (PEEK) | Fused deposition modeling | Tensile Strength: 58–85 Mpa Elastic Modulus: 3000–4100 Mpa; |
| Acrylonitrile butadiene styrene (ABS) | Fused deposition modeling | Tensile Strength: 27–31 Mpa; Layer height: 0.05–0.14 mm; Processed at 210–240 °C. data |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dimitrova, M.; Vlahova, A.; Kalachev, Y.; Zlatev, S.; Kazakova, R.; Capodiferro, S. Recent Advances in 3D Printing of Polymers for Application in Prosthodontics. Polymers 2023, 15, 4525. https://doi.org/10.3390/polym15234525
Dimitrova M, Vlahova A, Kalachev Y, Zlatev S, Kazakova R, Capodiferro S. Recent Advances in 3D Printing of Polymers for Application in Prosthodontics. Polymers. 2023; 15(23):4525. https://doi.org/10.3390/polym15234525
Chicago/Turabian StyleDimitrova, Mariya, Angelina Vlahova, Yavor Kalachev, Stefan Zlatev, Rada Kazakova, and Saverio Capodiferro. 2023. "Recent Advances in 3D Printing of Polymers for Application in Prosthodontics" Polymers 15, no. 23: 4525. https://doi.org/10.3390/polym15234525
APA StyleDimitrova, M., Vlahova, A., Kalachev, Y., Zlatev, S., Kazakova, R., & Capodiferro, S. (2023). Recent Advances in 3D Printing of Polymers for Application in Prosthodontics. Polymers, 15(23), 4525. https://doi.org/10.3390/polym15234525







