Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives
Abstract
1. Introduction
2. Adsorption
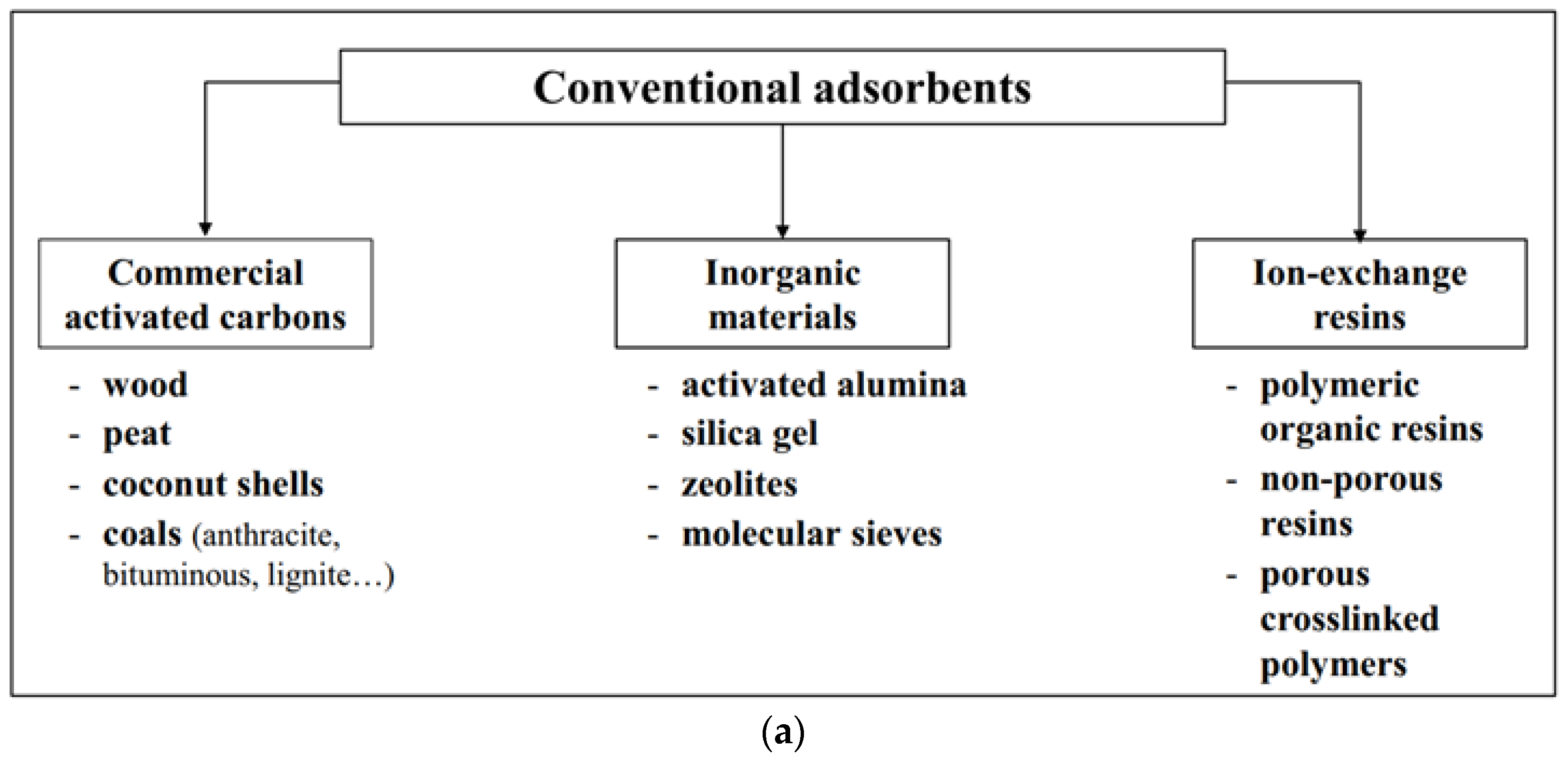
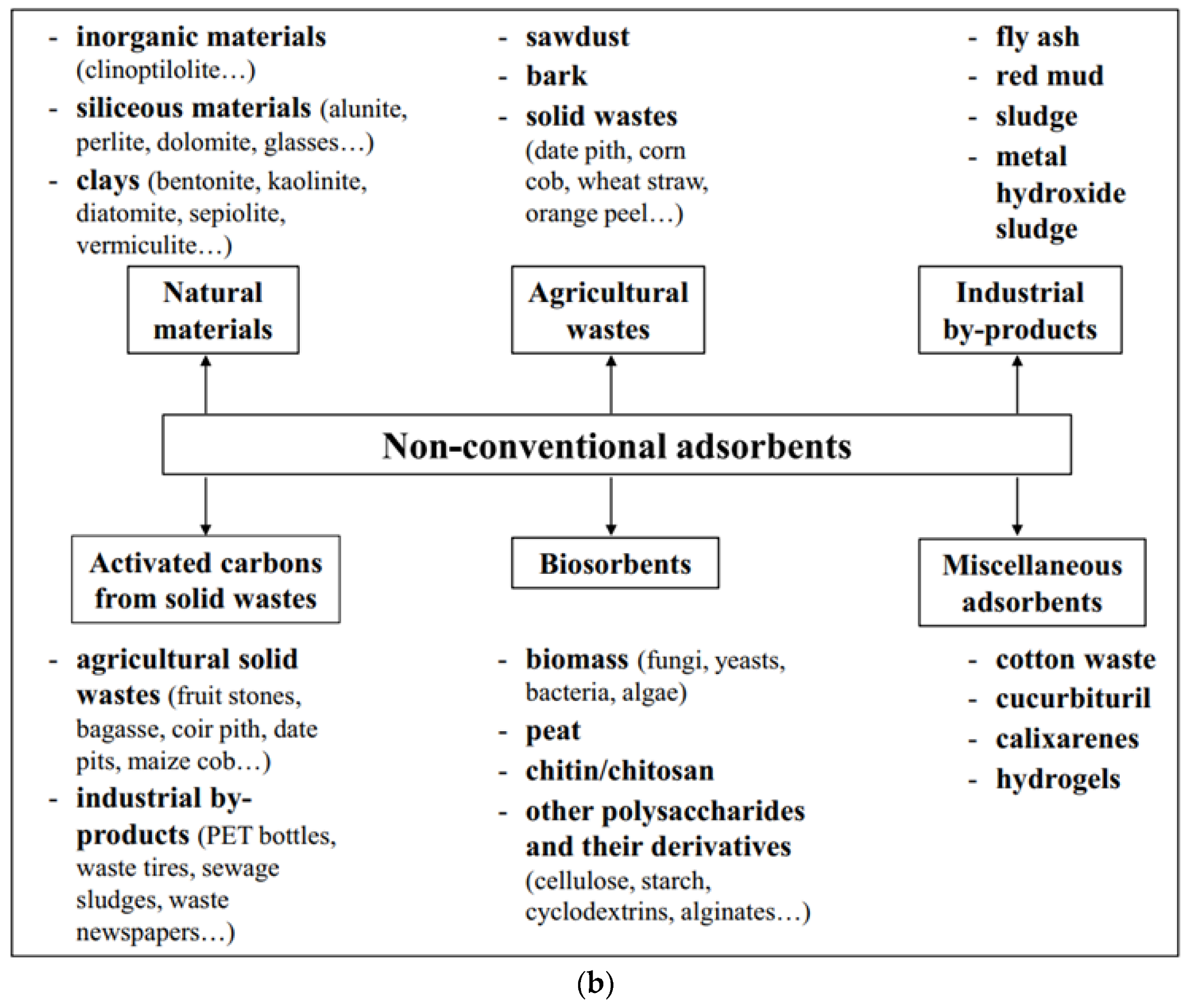
2.1. Adsorbents
2.2. Starch-Based Adsorbents
2.3. Starch-Based Adsorbents for In situ and Ex Situ Water Remediation
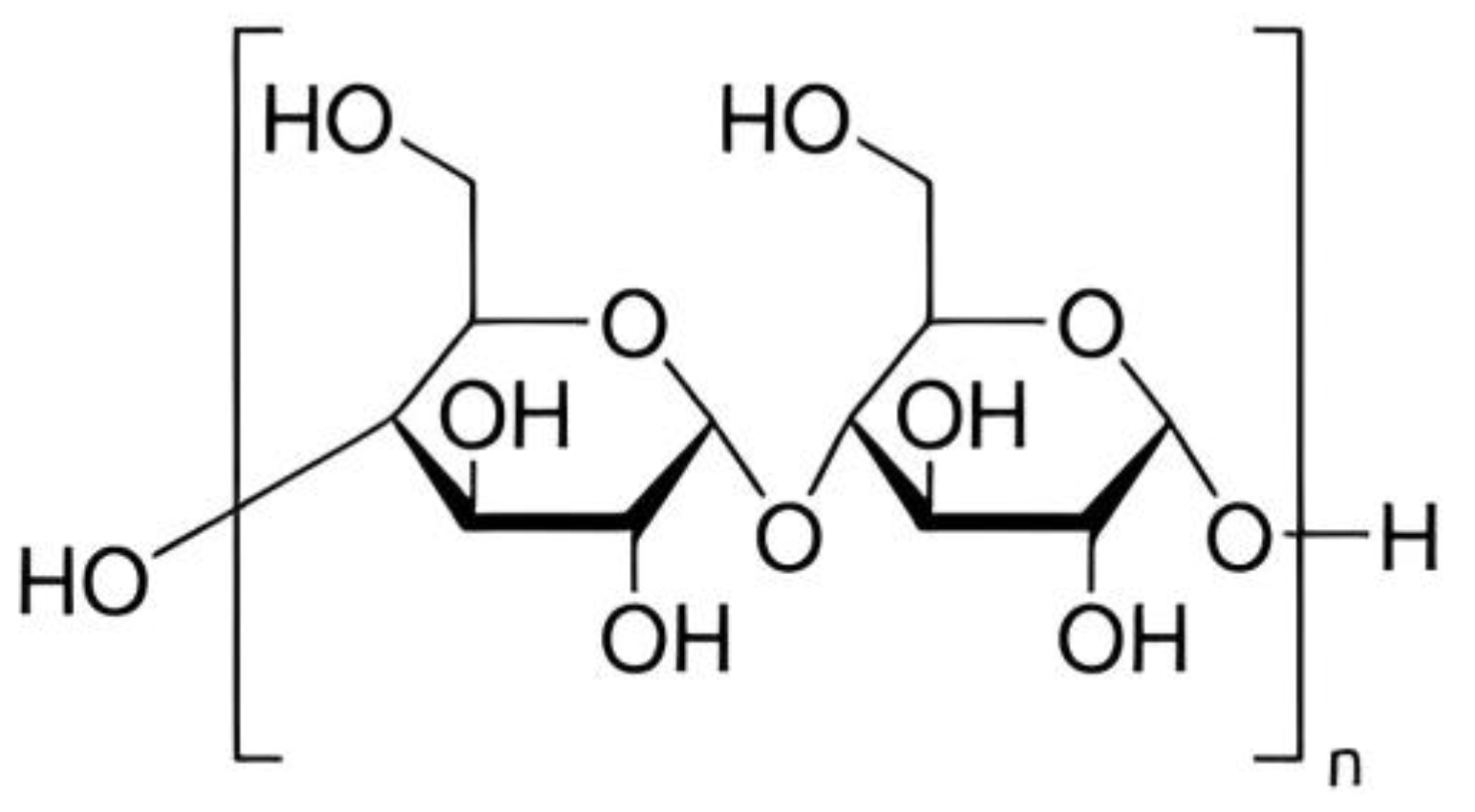
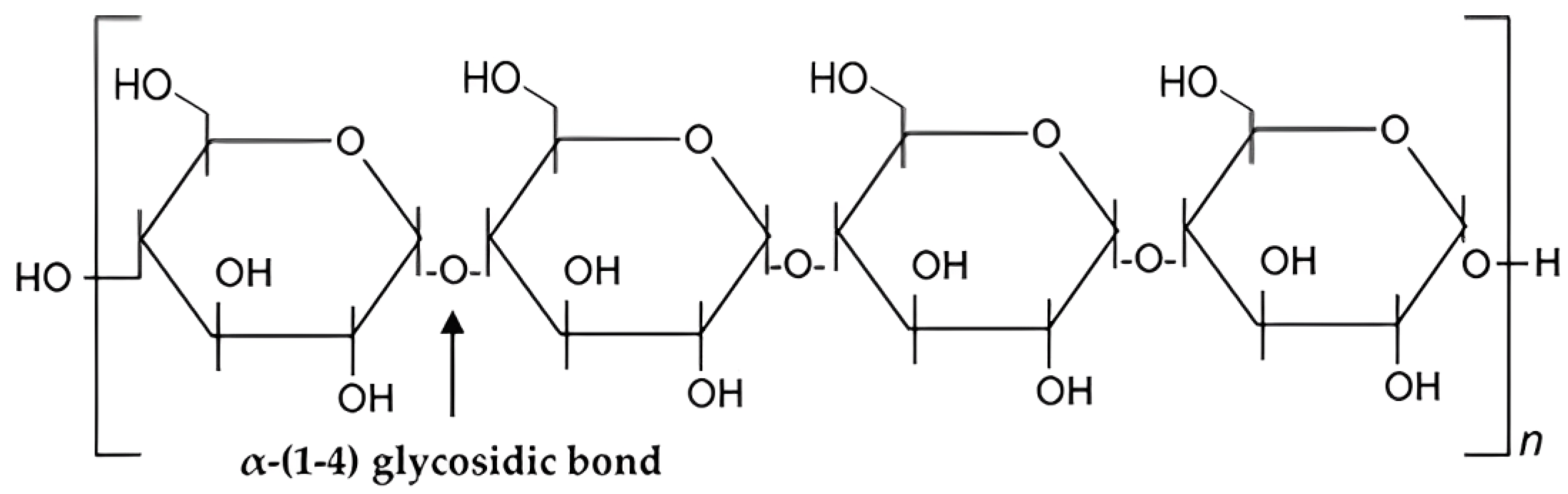
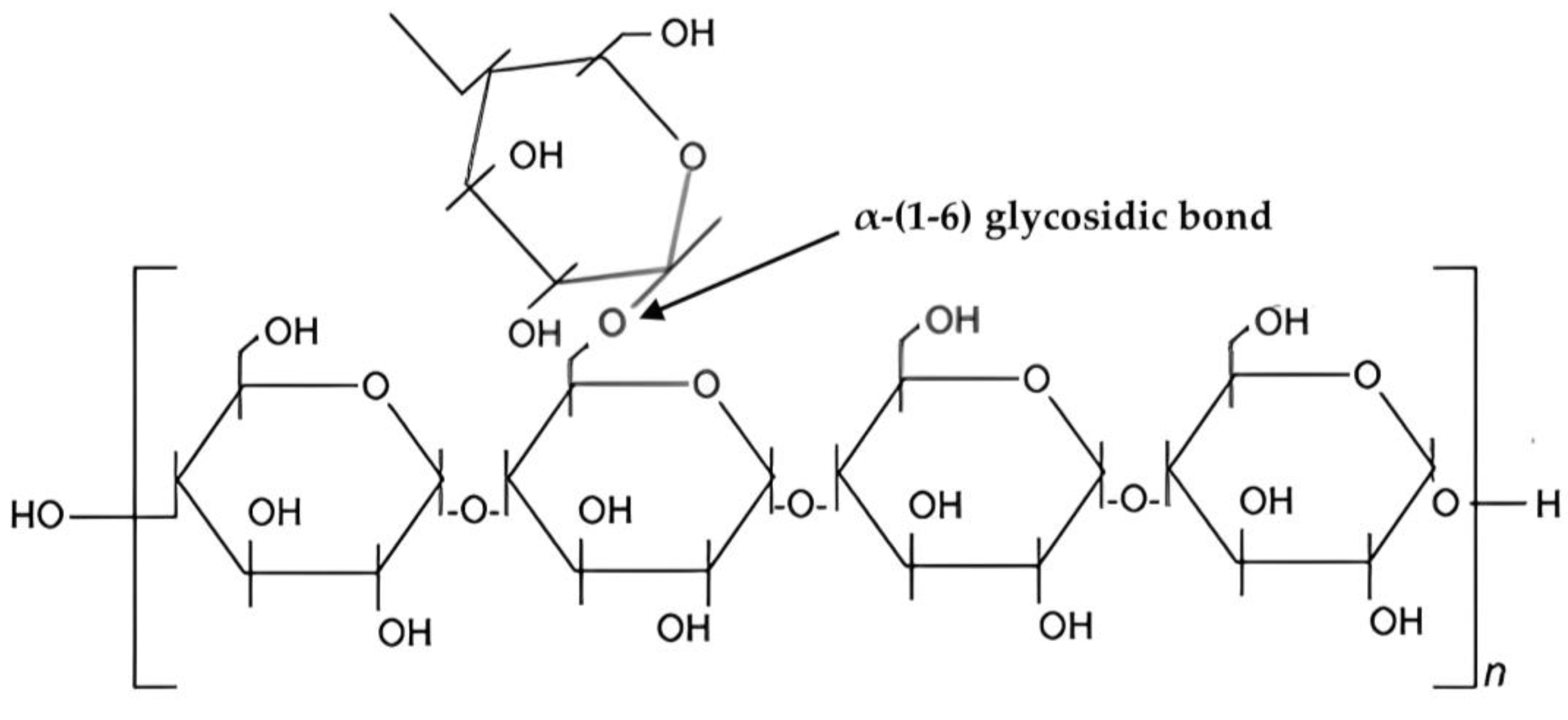
3. Chemical Structure and Properties of Starch
Chemistry and Properties
4. Applications for Water Treatment
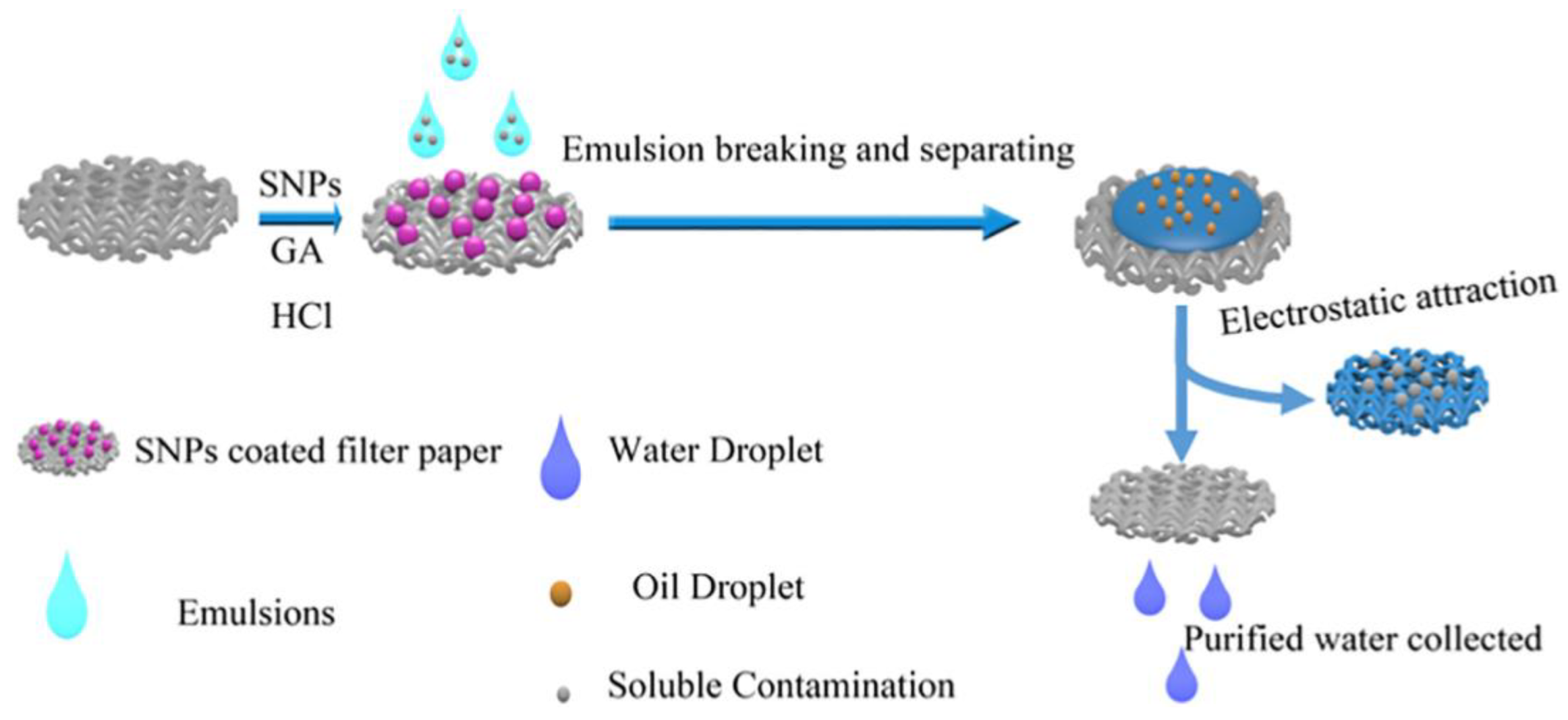
4.1. Removal of Oil and Organic Solvent
4.2. Removal of Pesticides
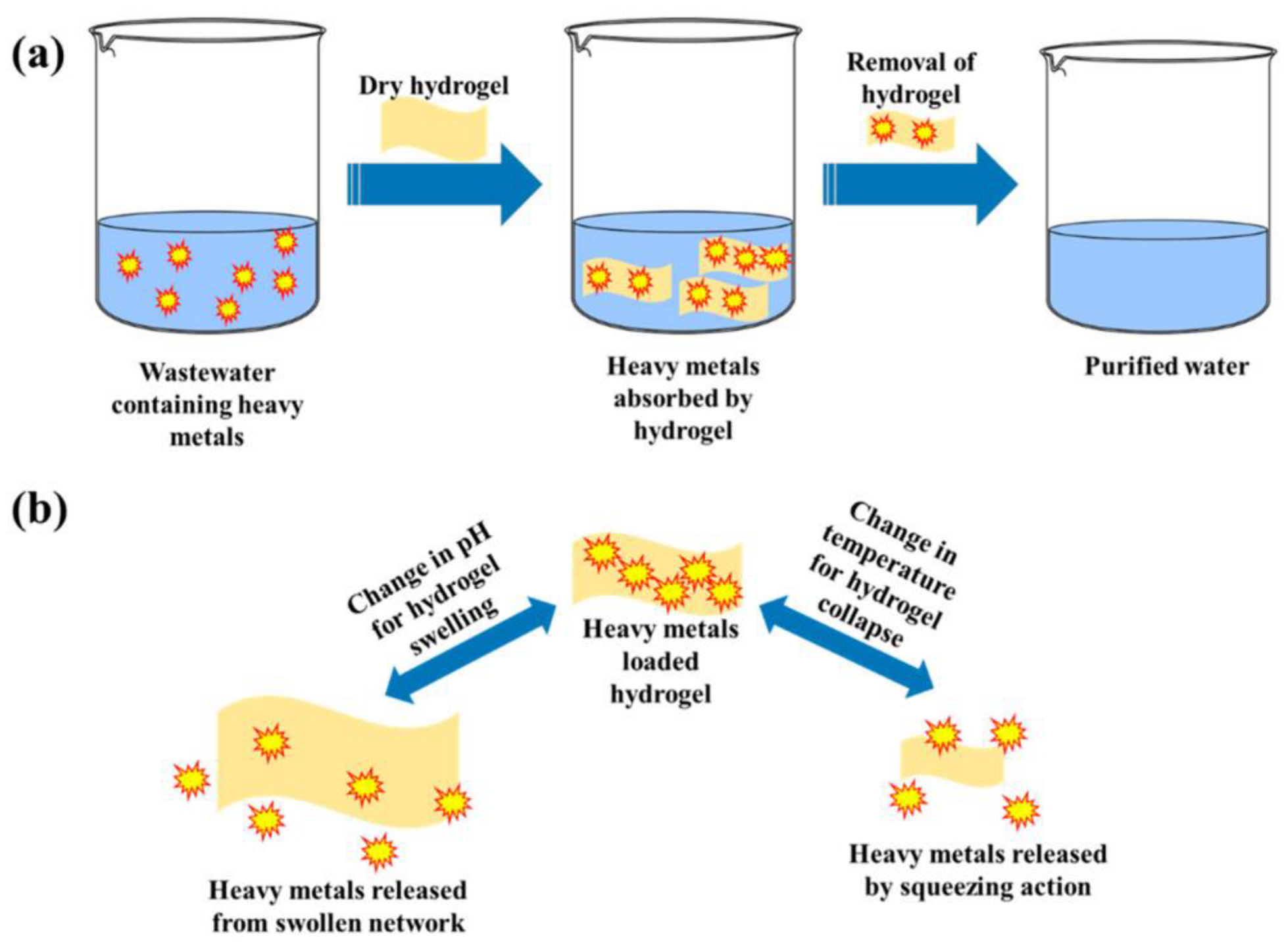
4.3. Removal of Heavy Metal Ions
4.4. Removal of Dye
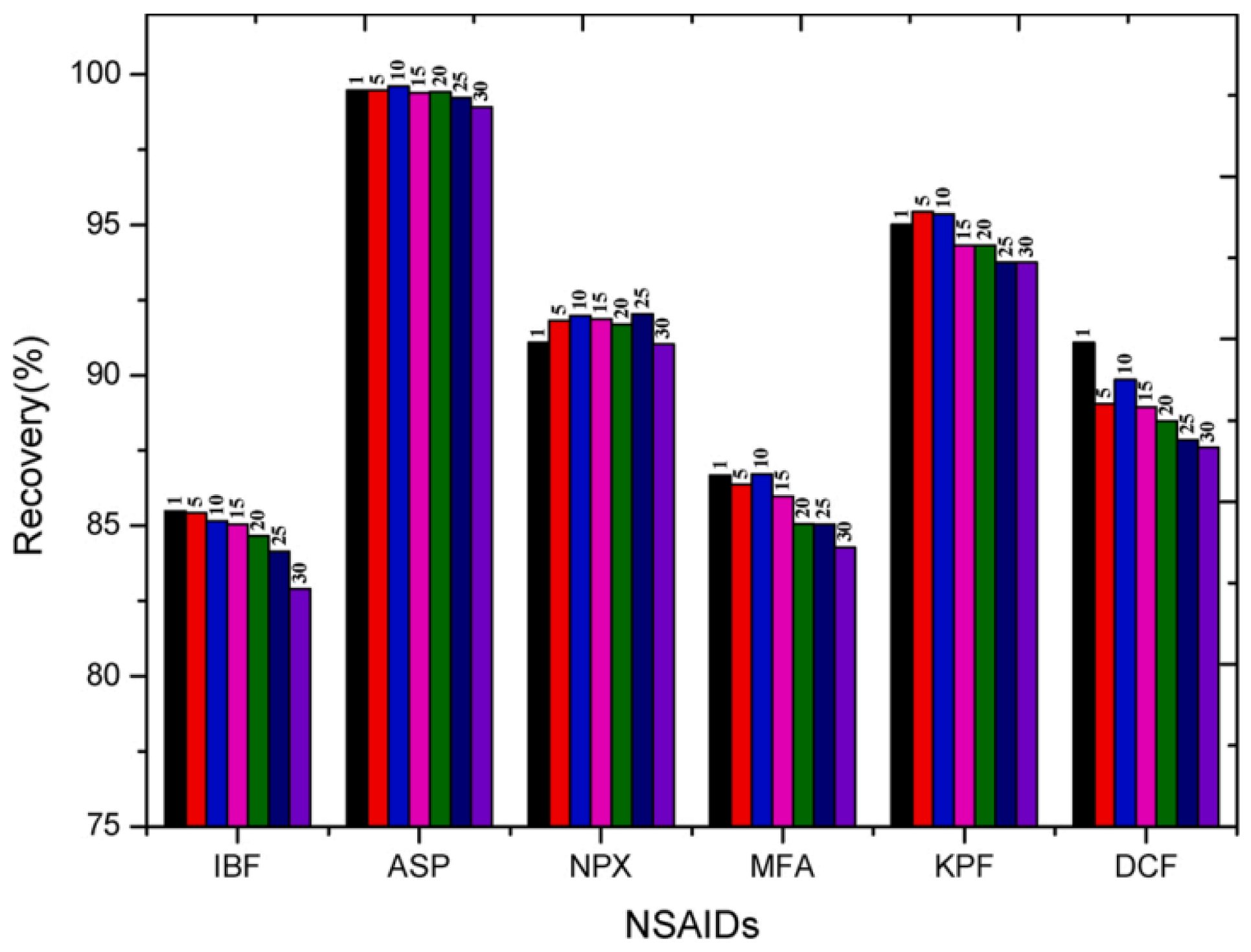
4.5. Removal of Pharmaceutical Pollutants
5. Outlook and Challenges
6. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations. The 17 Goals. Available online: https://sdgs.un.org/goals (accessed on 29 June 2023).
- World Health Organization. Drinking-Water. Available online: https://www.who.int/news-room/fact-sheets/detail/drinking-water (accessed on 29 June 2023).
- Cai, H.; Mei, Y.; Chen, J.; Wu, Z.; Lan, L.; Zhu, D. An Analysis of the Relation between Water Pollution and Economic Growth in China by Considering the Contemporaneous Correlation of Water Pollutants. J. Clean. Prod. 2020, 276, 122783. [Google Scholar] [CrossRef]
- Ezbakhe, F. Addressing Water Pollution as a Means to Achieving the Sustainable Development Goals. J. Water Pollut. Control 2018, 1, 6. [Google Scholar]
- Deletic, A.; Wang, H. Water Pollution Control for Sustainable Development. Engineering 2019, 5, 839–840. [Google Scholar] [CrossRef]
- Wang, M.; Janssen, A.B.G.; Bazin, J.; Strokal, M.; Ma, L.; Kroeze, C. Accounting for Interactions between Sustainable Development Goals Is Essential for Water Pollution Control in China. Nat. Commun. 2022, 13, 730. [Google Scholar] [CrossRef] [PubMed]
- Ekere, N.R.; Agbazue, V.E.; Ngang, B.U.; Ihedioha, J.N. Hydrochemistry and Water Quality Index of Groundwater Resources in Enugu North District, Enugu, Nigeria. Environ. Monit. Assess. 2019, 191, 150. [Google Scholar] [CrossRef] [PubMed]
- Olukanni, D.O.; Ebuetse, M.A.; Anake, W.U. Drinking Water Quality and Sanitation Issues: A Survey of a Semi-Urban Setting in Nigeria. Int. J. Res. Eng. Sci. 2014, 2, 58–65. [Google Scholar]
- Ighalo, J.O.; Adeniyi, A.G. A Comprehensive Review of Water Quality Monitoring and Assessment in Nigeria. Chemosphere 2020, 260, 127569. [Google Scholar] [CrossRef]
- Jain, K.; Patel, A.S.; Pardhi, V.P.; Flora, J.S.S. Nanotechnology in Wastewater Management: A New Paradigm towards Wastewater Treatment. Molecules 2021, 26, 1797. [Google Scholar] [CrossRef]
- Ezugbe, E.O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef]
- Chaudhry, F.N.; Malik, M.F. Factors Affecting Water Pollution: A Review. J. Ecosyst. Ecography 2017, 7, 5–8. [Google Scholar] [CrossRef]
- Owa, F.D. Water Pollution: Sources, Effects, Control and Management. Mediterr. J. Soc. Sci. 2013, 4, 65–68. [Google Scholar] [CrossRef]
- Halder, J.N.; Islam, M.N. Water Pollution and Its Impact on the Human Health. J. Environ. Hum. 2015, 2, 36–46. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; Von Gunten, U.; Wehrli, B. Global Water Pollution and Human Health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Chen, B.; Yang, S.; Cao, Q.; Qian, Y. Life Cycle Economic Assessment of Coal Chemical Wastewater Treatment Facing the ‘Zero Liquid Discharge’ Industrial Water Policies in China: Discharge or Reuse? Energy Policy 2020, 137, 111107. [Google Scholar] [CrossRef]
- Qin, Y.; Curmi, E.; Kopec, G.M.; Allwood, J.M.; Richards, K.S. China’s Energy-Water Nexus—Assessment of the Energy Sector’s Compliance with the ‘3 Red Lines’ Industrial Water Policy. Energy Policy 2015, 82, 131–143. [Google Scholar] [CrossRef]
- Kallis, G.; Butler, D. The EU Water Framework Directive: Measures and Implications. Water Policy 2001, 3, 125–142. [Google Scholar] [CrossRef]
- Carere, M.; Dulio, V.; Hanke, G.; Polesello, S. Guidance for Sediment and Biota Monitoring under the Common Implementation Strategy for the Water Framework Directive. Trends Anal. Chem. 2012, 36, 15–24. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and Disadvantages of Techniques Used for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Leslie Grady, C.P., Jr.; Daigger, G.T.; Love, N.G.; Filipe, C.D.M. Biological Wastewater Treatment, 3rd ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2011. [Google Scholar]
- Qu, X.; Brame, J.; Li, Q.; Alvarez, P.J.J. Nanotechnology for a Safe and Sustainable Water Supply: Enabling Integrated Water Treatment and Reuse. Acc. Chem. Res. 2013, 46, 834–843. [Google Scholar] [CrossRef]
- Bădescu, I.S.; Bulgariu, D.; Ahmad, I.; Bulgariu, L. Valorisation Possibilities of Exhausted Biosorbents Loaded with Metal Ions—A Review. J. Environ. Manag. 2018, 224, 288–297. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Sajjadi, M.; Iravani, S.; Rajender, S. Varma Starch, Cellulose, Pectin, Gum, Alginate, Chitin and Chitosan Derived (Nano) Materials for Sustainable Water Treatment: A Review. Carbohydr. Polym. 2021, 251, 116986. [Google Scholar] [CrossRef]
- Yusof, Y.M.; Kadir, M.F.Z. Electrochemical Characterizations and the Effect of Glycerol in Biopolymer Electrolytes Based on Methylcellulose-Potato Starch Blend. Mol. Cryst. Liq. Cryst. 2016, 627, 220–233. [Google Scholar] [CrossRef]
- Ahamad, T.; Naushad, M.; Mousa, R.H.; Alshehri, S.M. Fabrication of Starch-Salicylaldehyde Based Polymer Nanocomposite (PNC) for the Removal of Pollutants from Contaminated Water. Int. J. Biol. Macromol. 2020, 165, 2731–2738. [Google Scholar] [CrossRef] [PubMed]
- Amaraweera, S.M.; Gunathilake, C.; Gunawardene, O.H.P.; Fernando, N.M.L.; Wanninayaka, D.B.; Dassanayake, R.S.; Rajapaksha, S.M.; Manamperi, A.; Fernando, C.A.N.; Kulatunga, A.K.; et al. Development of Starch-Based Materials Using Current Modification Techniques and Their Applications: A Review. Molecules 2021, 26, 6880. [Google Scholar] [CrossRef]
- Zhang, D.; Mu, T.; Sun, H. Effects of Starch from Five Different Botanical Sources on the Rheological and Structural Properties of Starch-Gluten Model Doughs. Food Res. Int. 2018, 103, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Liu, S. An Overview of Biological Basics. In Bioprocess Engineering: Kinetics, Sustainability, and Reactor Design; Elsevier: Amsterdam, The Netherlands, 2017; pp. 21–80. ISBN 978-0-444-63783-3. [Google Scholar]
- Wang, S. Starch Structure, Functionality and Application in Foods; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Abd El-Ghany, N.A.; Elella, M.H.A.; Abdallah, H.M.; Mostafa, M.S.; Samy, M. Recent Advances in Various Starch Formulation for Wastewater Purification via Adsorption Technique: A Review; Springer: Berlin/Heidelberg, Germany, 2023; ISBN 0123456789. [Google Scholar]
- Fang, K.; Deng, L.; Yin, J.; Yang, T.; Li, J.; He, W. Recent Advances in Starch-Based Magnetic Adsorbents for the Removal of Contaminants from Wastewater: A Review. Int. J. Biol. Macromol. 2022, 218, 909–929. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, H.; Wang, L.; Ul Abdin, Z.; Chen, Y.; Wang, J.; Zhou, W.; Yang, X.; Khan, R.U.; Zhang, H.; et al. Recent Progress in Chemical Modification of Starch and Its Applications. RSC Adv. 2015, 5, 67459–67474. [Google Scholar] [CrossRef]
- Uda, M.N.A.; Gopinath, S.C.B.; Hashim, U.; Uda, M.N.A.; Adam, T.; Parmin, N.A.; Subramaniam, S.; Chinni, S.V.; Lebaka, V.R.; Gobinath, R. Controlling Arsenic Accumulation in Rice Grain under Nanomaterials-Assisted Optimal Greenhouse Set-Up. Sustainability 2023, 15, 2633. [Google Scholar] [CrossRef]
- Aly, A.A.; Hebeish, A.A. Development of Starch-Based Cationic Adsorbents for Removal of Anionic Dyes from Aqueous Systems. Int. J. Sci. Res. 2015, 4, 4–12. [Google Scholar]
- Kecili, R.; Hussain, C.M. Mechanism of Adsorption on Nanomaterials. In Nanomaterials in Chromatography: Current Trends in Chromatographic Research Technology and Techniques; Elsevier: Amsterdam, The Netherlands, 2018; pp. 89–115. [Google Scholar]
- Rathi, B.S.; Kumar, P.S.; Show, P.L. A Review on Effective Removal of Emerging Contaminants from Aquatic Systems: Current Trends and Scope for Further Research. J. Hazard. Mater. 2021, 409, 124413. [Google Scholar] [CrossRef]
- Saif, S.; Adil, S.F.; Khan, M.; Hatshan, M.R.; Khan, M.; Bashir, F. Adsorption Studies of Arsenic(V) by Cuo Nanoparticles Synthesized by Phyllanthus Emblica Leaf-Extract-Fueled Solution Combustion Synthesis. Sustainability 2021, 13, 2017. [Google Scholar] [CrossRef]
- Ali, I.; Arsh, A.; Mbianda, X.Y.; Burakov, A.; Galunin, E.; Burakova, I.; Mkrtchyan, E.; Tkachev, A.; Grachev, V. Graphene Based Adsorbents for Remediation of Noxious Pollutants from Wastewater. Environ. Int. 2019, 127, 160–180. [Google Scholar] [CrossRef] [PubMed]
- McKay, G. Use of Adsorbents for the Removal of Pollutants from Wastewaters; CRC Press: Boca Raton, FL, USA, 1996. [Google Scholar]
- Park, D.; Yun, Y.S.; Park, J.M. The Past, Present, and Future Trends of Biosorption. Biotechnol. Bioprocess Eng. 2010, 15, 86–102. [Google Scholar] [CrossRef]
- Crini, G. Recent Developments in Polysaccharide-Based Materials Used as Adsorbents in Wastewater Treatment. Prog. Polym. Sci. 2005, 30, 38–70. [Google Scholar] [CrossRef]
- Crini, G. Non-Conventional Low-Cost Adsorbents for Dye Removal: A Review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and Non-Conventional Adsorbents for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Anirudhan, T.S.; Shainy, F. Effective Removal of Mercury(II) Ions from Chlor-Alkali Industrial Wastewater Using 2-Mercaptobenzamide Modified Itaconic Acid-Grafted-Magnetite Nanocellulose Composite. J. Colloid Interface Sci. 2015, 456, 22–31. [Google Scholar] [CrossRef]
- Ali, I.; Gupta, V.K. Advances in Water Treatment by Adsorption Technology. Nat. Protoc. 2006, 1, 2661–2667. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-Cost Adsorbents for Heavy Metals Uptake from Contaminated Water: A Review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Varma, A.J.; Deshpande, S.V.; Kennedy, J.F. Metal Complexation by Chitosan and Its Derivatives: A Review. Carbohydr. Polym. 2004, 55, 77–93. [Google Scholar] [CrossRef]
- Cheng, R.; Ou, S. Application of Modified Starches in Wastewater Treatment. In Polymer Science: Research Advances, Practical Applications and Educational Aspects; Méndez-Vilas, A., Solano, A., Eds.; Formatex Research Center S.L.: Madrid, Spain, 2011. [Google Scholar]
- Haq, F.; Mehmood, S.; Haroon, M.; Kiran, M.; Waseem, K.; Aziz, T.; Farid, A. Role of Starch Based Materials as a Bio-Sorbents for the Removal of Dyes and Heavy Metals from Wastewater. J. Polym. Environ. 2022, 30, 1730–1748. [Google Scholar] [CrossRef]
- Kim, S.; Chu, K.H.; Al-Hamadani, Y.A.J.; Park, C.M.; Jang, M.; Kim, D.H.; Yu, M.; Heo, J.; Yoon, Y. Removal of Contaminants of Emerging Concern by Membranes in Water and Wastewater: A Review. Chem. Eng. J. 2018, 335, 896–914. [Google Scholar] [CrossRef]
- Zafar, R.; Zia, K.M.; Tabasum, S.; Jabeen, F.; Noreen, A.; Zuber, M. Polysaccharide Based Bionanocomposites, Properties and Applications: A Review. Int. J. Biol. Macromol. 2016, 92, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wei, H.; Liu, Y.; Li, S.; Wang, G.; Guo, T.; Han, H. An in Situ Reactive Spray-Drying Strategy for Facile Preparation of Starch-Chitosan Based Hydrogel Microspheres for Water Treatment Application. Chem. Eng. Process. Process Intensif. 2021, 168, 108548. [Google Scholar] [CrossRef]
- Zhang, B.; Li, X.; Xie, Q.; Tao, H.; Wang, W.; Chen, H.Q. Preparation and Characterization of Non-Crystalline Granular Starch and Corresponding Carboxymethyl Starch. Int. J. Biol. Macromol. 2017, 103, 656–662. [Google Scholar] [CrossRef]
- Haq, F.; Yu, H.; Wang, L.; Teng, L.; Haroon, M.; Khan, R.U.; Mehmood, S.; Bilal-Ul-Amin; Ullah, R.S.; Khan, A.; et al. Advances in Chemical Modifications of Starches and Their Applications. Carbohydr. Res. 2019, 476, 12–35. [Google Scholar] [CrossRef]
- Schwantes, D.; Gonçalves, A.C.; Junior Miola, A.; Coelho, G.F.; Dos Santos, M.G.; Leismann, E.A.V. Removal of Cu (II) and Zn (II) from Water with Natural Adsorbents from Cassava Agroindustry Residues. Acta Sci. Technol. 2015, 37, 409–417. [Google Scholar] [CrossRef]
- Musarurwa, H.; Tavengwa, N.T. Application of Carboxymethyl Polysaccharides as Bio-Sorbents for the Sequestration of Heavy Metals in Aquatic Environments. Carbohydr. Polym. 2020, 237, 116142. [Google Scholar] [CrossRef]
- Luo, H.; Dong, F.; Wang, Q.; Li, Y.; Xiong, Y. Construction of Porous Starch-Based Hydrogel via Regulating the Ratio of Amylopectin/Amylose for Enhanced Water-Retention. Molecules 2021, 26, 3999. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Hou, C.; Qi, Z.; He, X.; Li, Y. Facile Synthesis of Monodisperse Functional Magnetic Dialdehyde Starch Nano-Composite and Used for Highly Effective Recovery of Hg(II). Chemosphere 2015, 141, 26–33. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, C.; Wang, X.; Wang, J.; Jiang, H.; Xu, K.; Bai, Y.; Wang, P. Fabrication of Raspberry-like Starch-Based Polymer Microspheres for W/O and O/W Emulsions Separation and Purification. J. Environ. Chem. Eng. 2023, 11, 109260. [Google Scholar] [CrossRef]
- Irani, M.; Ismail, H.; Ahmad, Z.; Fan, M. Synthesis of Linear Low-Density Polyethylene-g-Poly (Acrylic Acid)-Co-Starch/Organo-Montmorillonite Hydrogel Composite as an Adsorbent for Removal of Pb(ΙΙ) from Aqueous Solutions. J. Environ. Sci. 2015, 27, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Keirudin, A.A.; Zainuddin, N.; Yusof, N.A. Crosslinked Carboxymethyl Sago Starch/Citric Acid Hydrogel for Sorption of Pb2+, Cu2+, Ni2+ and Zn2+ from Aqueous Solution. Polymers 2020, 12, 2465. [Google Scholar] [CrossRef] [PubMed]
- Haroon, M.; Wang, L.; Yu, H.; Abbasi, N.M.; Zain-Ul-Abdin; Saleem, M.; Khan, R.U.; Ullah, R.S.; Chen, Q.; Wu, J. Chemical Modification of Starch and Its Application as an Adsorbent Material. RSC Adv. 2016, 6, 78264–78285. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Nabil, G.M.; Zaki, M.M.; Saleh, M.M. Starch Functionalization of Iron Oxide By-Product from Steel Industry as a Sustainable Low Cost Nanocomposite for Removal of Divalent Toxic Metal Ions from Water. Int. J. Biol. Macromol. 2019, 137, 455–468. [Google Scholar] [CrossRef]
- Ebadollahzadeh, H.; Zabihi, M. Competitive Adsorption of Methylene Blue and Pb (II) Ions on the Nano-Magnetic Activated Carbon and Alumina. Mater. Chem. Phys. 2020, 248, 122893. [Google Scholar] [CrossRef]
- Reed, B.E.; Arunachalam, S. Use of Granular Activated Carbon Columns for Lead Removal. J. Environ. Eng. 1994, 120, 416–436. [Google Scholar] [CrossRef]
- McKay, G.; Bino, M.J.; Altamemi, A.R. The Adsorption of Various Pollutants from Aqueous Solutions on to Activated Carbon. Water Res. 1985, 19, 491–495. [Google Scholar] [CrossRef]
- Sharma, D.; Forster, C. Removal of Hexavalent Chromium from Aqueous Solutions by Granular Activated Carbon. Water 1996, 22, 153–160. [Google Scholar]
- Hegazy, S.H.; Mohamed, S.K. Starch-Graft-Polyacrylamide Copolymer/Fe3O4/Graphene Oxide Nanocomposite: Synthesis, Characterization, and Application as a Low-Cost Adsorbent for Ni (II) from Aqueous Solutions. J. Polym. Res. 2021, 28, 49. [Google Scholar] [CrossRef]
- Kadirvelu, K.; Thamaraiselvi, K.; Namasivayam, C. Adsorption of Nickel (II) from Aqueous Solution onto Activated Carbon Prepared from Coirpith. Sep. Purif. Technol. 2001, 24, 497–505. [Google Scholar] [CrossRef]
- Priyanka, M.; Saravanakumar, M.P. Ultrahigh Adsorption Capacity of Starch Derived Zinc Based Carbon Foam for Adsorption of Toxic Dyes and Its Preliminary Investigation on Oil-Water Separation. J. Clean. Prod. 2018, 197, 511–524. [Google Scholar] [CrossRef]
- Inbaraj, B.S.; Sulochana, N. Basic Dye Adsorption on a Low Cost Carbonaceous Sorbent- Kinetic and Equilibrium Studies. Indian J. Chem. Technol. 2002, 9, 201–208. [Google Scholar]
- Tamura, T.; Miyoshi, T.; Boki, K.; Tanada, S. Adsorption of Indigo Carmine by Activated Carbon. Environ. Technol. Lett. 1988, 9, 281–286. [Google Scholar] [CrossRef]
- Mahmoodi-Babolan, N.; Nematollahzadeh, A.; Heydari, A.; Merikhy, A. Bioinspired Catecholamine/Starch Composites as Superadsorbent for the Environmental Remediation. Int. J. Biol. Macromol. 2019, 125, 690–699. [Google Scholar] [CrossRef]
- Vanjara, A.K. Colour Removal from Textile Effluent Using Refuse Derived Fuel as an Adsorbent. Indian J. Chem. Technol. 1998, 5, 53–55. [Google Scholar]
- Gunawardene, O.H.P.; Gunathilake, C.A.; Amaraweera, A.P.S.M.; Fernando, N.M.L.; Manipura, A.; Manamperi, W.A.; Kulatunga, K.M.A.K.; Rajapaksha, S.M.; Gamage, A.; Dassanayake, R.S.; et al. Removal of Pb(II) Ions from Aqueous Solution Using Modified Starch. J. Compos. Sci. 2021, 5, 46. [Google Scholar] [CrossRef]
- Dang, X.; Yu, Z.; Yang, M.; Wai, M.; Song, Y.; Wang, X.; Zhang, H. Sustainable Electrochemical Synthesis of Natural Starch-Based Biomass Adsorbent with Ultrahigh Adsorption Capacity for Cr(VI) and Dyes Removal. Sep. Purif. Technol. 2022, 288, 120668. [Google Scholar] [CrossRef]
- Mittal, H.; Alhassan, S.M.; Ray, S.S. Efficient Organic Dye Removal from Wastewater by Magnetic Carbonaceous Adsorbent Prepared from Corn Starch. J. Environ. Chem. Eng. 2018, 6, 7119–7131. [Google Scholar] [CrossRef]
- Yu, M.; Zheng, Y.; Tian, J. Study on the Biodegradability of Modified Starch/Polylactic Acid (PLA) Composite Materials. RSC Adv. 2020, 10, 26298–26307. [Google Scholar] [CrossRef]
- Ju, B.; Yan, D.; Zhang, S. Micelles Self-Assembled from Thermoresponsive 2-Hydroxy-3-Butoxypropyl Starches for Drug Delivery. Carbohydr. Polym. 2012, 87, 1404–1409. [Google Scholar] [CrossRef]
- Ogunsona, E.; Ojogbo, E.; Mekonnen, T. Advanced Material Applications of Starch and Its Derivatives. Eur. Polym. J. 2018, 108, 570–581. [Google Scholar] [CrossRef]
- Qamruzzaman, M.; Ahmed, F.; Mondal, M.I.H. An Overview on Starch-Based Sustainable Hydrogels: Potential Applications and Aspects; Springer: Berlin/Heidelberg, Germany, 2022; Volume 30, ISBN 0123456789. [Google Scholar]
- Junlapong, K.; Maijan, P.; Chaibundit, C.; Chantarak, S. Effective Adsorption of Methylene Blue by Biodegradable Superabsorbent Cassava Starch-Based Hydrogel. Int. J. Biol. Macromol. 2020, 158, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Posada-Velez, M.C.; Pineda-Gomez, P.; Martinez-Hernandez, H.D. Acetylated Corn and Potato Starches as an Alternative to the Toxic Inorganic Coagulants/Flocculants for Wastewater Treatment. Environ. Nanotechnol. Monit. Manag. 2023, 20, 100786. [Google Scholar] [CrossRef]
- Sun, J.; Shi, M.; Wang, W. Ethanol-Water near-Azeotropic Mixture Dehydration by Compound Starch-Based Adsorbent. Trans. Tianjin Univ. 2015, 21, 427–432. [Google Scholar] [CrossRef]
- Suo, F.; Liu, X.; Li, C.; Yuan, M.; Zhang, B.; Wang, J.; Ma, Y.; Lai, Z.; Ji, M. Mesoporous Activated Carbon from Starch for Superior Rapid Pesticides Removal. Int. J. Biol. Macromol. 2019, 121, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhang, Q.; Zhou, J.; Fang, H.; Yang, H.; Wang, F. Investigation of Pesticide Residue Removal Effect of Gelatinized Starch Using Surface-Enhanced Raman Scattering Mapping. Food Chem. 2021, 365, 130448. [Google Scholar] [CrossRef]
- Md. Munjur, H.; Hasan, M.N.; Awual, M.R.; Islam, M.M.; Shenashen, M.A.; Iqbal, J. Biodegradable Natural Carbohydrate Polymeric Sustainable Adsorbents for Efficient Toxic Dye Removal from Wastewater. J. Mol. Liq. 2020, 319, 114356. [Google Scholar] [CrossRef]
- Bai, W.; Fan, L.; Zhou, Y.; Zhang, Y.; Shi, J.; Lv, G.; Wu, Y.; Liu, Q.; Song, J. Removal of Cd2+ Ions from Aqueous Solution Using Cassava Starch–based Superabsorbent Polymers. J. Appl. Polym. Sci. 2017, 134, 44758. [Google Scholar] [CrossRef]
- Fang, K.; Li, K.; Yang, T.; Li, J.; He, W. Starch-Based Magnetic Nanocomposite as an Efficient Absorbent for Anticancer Drug Removal from Aqueous Solution. Int. J. Biol. Macromol. 2021, 184, 509–521. [Google Scholar] [CrossRef]
- Sun, J.; Wang, W.; Wang, P.; Lv, H.; Luo, X.; Gao, H. Characterization of a Compound Starch-Based Adsorbent for Alcohol-Water Azeotrope Dehydration. Adsorpt. Sci. Technol. 2013, 31, 829–844. [Google Scholar] [CrossRef]
- Wu, P.; Gao, H.; Sun, J.; Ma, T.; Liu, Y.; Wang, F. Biosorptive Dehydration of Tert-Butyl Alcohol Using a Starch-Based Adsorbent: Characterization and Thermodynamics. Bioresour. Technol. 2012, 107, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, T.; Neelwarne, B.; Aswathanarayana, R.G. In Situ and Ex Situ Adsorption and Recovery of Betalains from Hairy Root Cultures of Beta Vulgaris. Biotechnol. Prog. 2004, 20, 777–785. [Google Scholar] [CrossRef]
- Kim, Y.S.; Nakano, K.; Lee, T.J.; Kanchanatawee, S.; Matsumura, M. On-Site Nitrate Removal of Groundwater by an Immobilized Psychrophilic Denitrifier Using Soluble Starch as a Carbon Source. J. Biosci. Bioeng. 2002, 93, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Foroutan, R.; Peighambardoust, S.J.; Mohammadi, R.; Peighambardoust, S.H.; Ramavandi, B. Development of New Magnetic Adsorbent of Walnut Shell Ash/Starch/Fe3O4 for Effective Copper Ions Removal: Treatment of Groundwater Samples. Chemosphere 2022, 296, 133978. [Google Scholar] [CrossRef] [PubMed]
- Jiryaei Sharahi, F.; Shahbazi, A. Melamine-Based Dendrimer Amine-Modified Magnetic Nanoparticles as an Efficient Pb(II) Adsorbent for Wastewater Treatment: Adsorption Optimization by Response Surface Methodology. Chemosphere 2017, 189, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, G.A.; Abdel-Aal, S.E.; Badway, N.A.; Abo Farha, S.A.; Alshafei, E.A. Radiation Synthesis and Characterization of Starch-Based Hydrogels for Removal of Acid Dye. Starch Staerke 2014, 66, 400–408. [Google Scholar] [CrossRef]
- Gadd, G.M. Biosorption: Critical Review of Scientific Rationale, Environmental Importance and Significance for Pollution Treatment. J. Chem. Technol. Biotechnol. 2009, 84, 13–28. [Google Scholar] [CrossRef]
- Crini, G.; Badot, P.-M. Sorption Processes and Pollution: Conventional and Non-Conventional Sorbents for Pollutant Removal from Wastewaters; PUFC: Besançon, France, 2010. [Google Scholar]
- Hoover, R. Composition, Molecular Structure, and Physicochemical Properties of Tuber and Root Starches: A Review. Carbohydr. Polym. 2001, 45, 253–267. [Google Scholar] [CrossRef]
- Denyer, K.A.Y.; Johnson, P.; Zeeman, S.; Smith, A.M. The Control of Amylose Synthesis. J. Plant Physiol. 2001, 158, 479–487. [Google Scholar] [CrossRef]
- Pacsu, E.; Lejaren Arthur Hiller, J. Cellulose Studies IV. The Chemical Structure of Cellulose and Starch. Text. Res. J. 1946, 16, 243–248. [Google Scholar] [CrossRef]
- Morrison, W.R.; Karkalas, J. Starch. In Methods in Plant Biochemistry; Academic Press: Cambridge, MA, USA, 1990; Volume 2, pp. 323–352. ISBN 0124610129. [Google Scholar]
- Liu, X.; Chao, C.; Yu, J.; Copeland, L.; Wang, S. Mechanistic Studies of Starch Retrogradation and Its Effects on Starch Gel Properties. Food Hydrocoll. 2021, 120, 106914. [Google Scholar] [CrossRef]
- French, D. Organization of Starch Granules. In Starch: Chemistry and Technology; Whistler, R.L., Bemiller, J.N., Paschall, E.F., Eds.; Academic Press: Cambridge, MA, USA, 1984; pp. 183–247. [Google Scholar]
- Buléon, A.; Colonna, P.; Planchot, V.; Ball, S. Starch Granules: Structure and Biosynthesis. Int. J. Biol. Macromol. 1998, 23, 85–112. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Hussain, C.M. Green and Sustainable Advanced Materials, Volume 2: Applications; John Wiley and Sons Inc.: Hoboken, NJ, USA, 2018. [Google Scholar]
- Saurav, K.; Majhi, M.R.; Singh, V.K. Preperation of Porous Magenesia by Decomposing an Ex-Potato Known as Starch Soluble (C6H10O5)N. Am. J. Sci. Ind. Res. 2014, 5, 120–125. [Google Scholar]
- Copeland, L.; Blazek, J.; Salman, H.; Tang, M.C. Form and Functionality of Starch. Food Hydrocoll. 2009, 23, 1527–1534. [Google Scholar] [CrossRef]
- Xu, J.; Sagnelli, D.; Faisal, M.; Perzon, A.; Taresco, V.; Mais, M.; Giosafatto, C.V.L.; Hebelstrup, K.H.; Ulvskov, P.; Jørgensen, B.; et al. Amylose/Cellulose Nanofiber Composites for All-Natural, Fully Biodegradable and Flexible Bioplastics. Carbohydr. Polym. 2021, 253, 117277. [Google Scholar] [CrossRef]
- Nakanishi, Y.; Norisuye, T.; Teramoto, A.; Kitamura, S. Conformation of Amylose in Dimethyl Sulfoxide. Macromolecules 1993, 26, 4220–4225. [Google Scholar] [CrossRef]
- Mendes, J.F.; Paschoalin, R.T.; Carmona, V.B.; Sena Neto, A.R.; Marques, A.C.P.; Marconcini, J.M.; Mattoso, L.H.C.; Medeiros, E.S.; Oliveira, J.E. Biodegradable Polymer Blends Based on Corn Starch and Thermoplastic Chitosan Processed by Extrusion. Carbohydr. Polym. 2016, 137, 452–458. [Google Scholar] [CrossRef]
- Obadi, M.; Qi, Y.; Xu, B. High-Amylose Maize Starch: Structure, Properties, Modifications and Industrial Applications. Carbohydr. Polym. 2023, 299, 120185. [Google Scholar] [CrossRef]
- Chi, C.; Guo, X.; Zhou, Y.; Chen, B.; He, Y. A Facile Method for Isolating Long Branch-Chains of Amylopectin from Starch by Preheating and Pullulanase Treatment. Ind. Crops Prod. 2023, 191, 115987. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, H.; Chen, Z.; Zhong, Q. Structural Basis for the Low Digestibility of Starches Recrystallized from Side Chains of Amylopectin Modified by Amylosucrase to Different Chain Lengths. Carbohydr. Polym. 2020, 241, 116352. [Google Scholar] [CrossRef]
- Kim, B.S.; Kim, H.S.; Yoo, S.H. Characterization of Enzymatically Modified Rice and Barley Starches with Amylosucrase at Scale-up Production. Carbohydr. Polym. 2015, 125, 61–68. [Google Scholar] [CrossRef]
- Ratnavathi, C.V.; Komala, V.V. Sorghum Grain Quality. In Sorghum Biochemistry: An Industrial Perspective; Ratnavathi, C.V., Patil, J.V., Chavan, U.D., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 1–61. [Google Scholar]
- Akter, M.; Bhattacharjee, M.; Dhar, A.K.; Rahman, F.B.A.; Haque, S.; Ur Rashid, T.U.; Kabir, S.M.F. Cellulose-Based Hydrogels for Wastewater Treatment: A Concise Review. Gels 2021, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- García-Padilla, Á.; Moreno-Sader, K.A.; Realpe, Á.; Acevedo-Morantes, M.; Soares, J.B.P. Evaluation of Adsorption Capacities of Nanocomposites Prepared from Bean Starch and Montmorillonite. Sustain. Chem. Pharm. 2020, 17, 100292. [Google Scholar] [CrossRef]
- Fan, Y.; Picchioni, F. Modification of Starch: A Review on the Application of “Green” Solvents and Controlled Functionalization. Carbohydr. Polym. 2020, 241, 116350. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, N.; Abarca, R.L.; Urdaneta, J.; Romero, J.; Galotto, M.J.; Guarda, A. Cassava Starch: Structural Modification for Development of a Bio-Adsorber for Aqueous Pollutants. Characterization and Adsorption Studies on Methylene Blue. Polym. Bull. 2021, 78, 1087–1107. [Google Scholar] [CrossRef]
- Gupta, A.D.; Rawat, K.P.; Bhadauria, V.; Singh, H. Recent Trends in the Application of Modified Starch in the Adsorption of Heavy Metals from Water: A Review. Carbohydr. Polym. 2021, 269, 117763. [Google Scholar] [CrossRef]
- Haq, F.; Yu, H.; Wang, L.; Liu, J.; Mehmood, S.; Haroon, M.; Amin, B.U.; Fahad, S.; Uddin, M.A. Dual Modification of Starches by Phosphorylation and Grafting and Their Application as Adsorbents for the Removal of Phenol. J. Polym. Res. 2020, 27, 313. [Google Scholar] [CrossRef]
- Kemas, C.U.; Ngwuluka, N.C.; Ochekpe, N.A.; Nep, E.I. Starch-Based Xerogels: Effect of Acetylation on Physicochemical and Rheological Properties. Int. J. Biol. Macromol. 2017, 98, 94–102. [Google Scholar] [CrossRef]
- Kaul, S.; Kaur, K.; Kaur, J.; Mehta, N.; Kennedy, J.F. Properties of Potato Starch as Influenced by Microwave, Ultrasonication, Alcoholic-Alkali and Pre-Gelatinization Treatments. Int. J. Biol. Macromol. 2023, 226, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Repo, E.; Yin, D.; Chen, L.; Kalliola, S.; Tang, J.; Iakovleva, E.; Tam, K.C.; Sillanpää, M. One-Pot Synthesis of Trifunctional Chitosan-EDTA-β-Cyclodextrin Polymer for Simultaneous Removal of Metals and Organic Micropollutants. Sci. Rep. 2017, 7, 15811. [Google Scholar] [CrossRef] [PubMed]
- Cova, T.F.; Murtinho, D.; Aguado, R.; Pais, A.A.C.C.; Valente, A.J.M. Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation. Polysaccharides 2021, 2, 16–38. [Google Scholar] [CrossRef]
- Wu, G.; Liu, Q.; Wang, J.; Xia, S.; Wu, H.; Zong, J.; Han, J.; Xing, W. Facile Fabrication of Rape Straw Biomass Fiber/β-CD/Fe3O4 as Adsorbent for Effective Removal of Ibuprofen. Ind. Crops Prod. 2021, 173, 114150. [Google Scholar] [CrossRef]
- Skwierawska, A.M.; Nowacka, D.; Nowicka, P.; Rosa, S.; Kozłowska-Tylingo, K. Structural Adaptive, Self-Separating Material for Removing Ibuprofen from Waters and Sewage. Materials 2021, 14, 7697. [Google Scholar] [CrossRef]
- Rafati, L.; Ehrampoush, M.H.; Rafati, A.A.; Mokhtari, M.; Mahvi, A.H. Nanocomposite Adsorbent Based on β-Cyclodextrin-PVP-Clay for the Removal of Naproxen from Aqueous Solution: Fixed-Bed Column and Modeling Studies. Desalin. Water Treat. 2018, 132, 63–74. [Google Scholar] [CrossRef]
- Rahman, N.; Nasir, M. Effective Removal of Acetaminophen from Aqueous Solution Using Ca (II)-Doped Chitosan/β-Cyclodextrin Composite. J. Mol. Liq. 2020, 301, 112454. [Google Scholar] [CrossRef]
- Duan, C.; Wang, J.; Liu, Q.; Zhou, Y.; Zhou, Y. Efficient Removal of Salbutamol and Atenolol by an Electronegative Silanized β-Cyclodextrin Adsorbent. Sep. Purif. Technol. 2022, 282, 120013. [Google Scholar] [CrossRef]
- Schäfer, A.I.; Stelzl, K.; Faghih, M.; Sen Gupta, S.; Krishnadas, K.R.; Heißler, S.; Pradeep, T. Polyethersulfone Nanofibers Impregnated with β-Cyclodextrin for Increased Micropollutant Removal from Water. ACS Sustain. Chem. Eng. 2018, 6, 2942–2953. [Google Scholar] [CrossRef]
- Jiang, L.; Liu, Y.; Liu, S.; Hu, X.; Zeng, G.; Hu, X.; Liu, S.; Liu, S.; Huang, B.; Li, M. Fabrication of β-Cyclodextrin/Poly (L-Glutamic Acid) Supported Magnetic Graphene Oxide and Its Adsorption Behavior for 17β-Estradiol. Chem. Eng. J. 2017, 308, 597–605. [Google Scholar] [CrossRef]
- Sandilya, A.A.; Natarajan, U.; Priya, M.H. Molecular View into the Cyclodextrin Cavity: Structure and Hydration. ACS Omega 2020, 5, 25655–25667. [Google Scholar] [CrossRef]
- Przybyla, M.A.; Yilmaz, G.; Remzi Becer, C. Natural Cyclodextrins and Their Derivatives for Polymer Synthesis. Polym. Chem. 2020, 11, 7582–7602. [Google Scholar] [CrossRef]
- Xia, D.; Wang, P.; Ji, X.; Khashab, N.M.; Sessler, J.L.; Huang, F. Functional Supramolecular Polymeric Networks: The Marriage of Covalent Polymers and Macrocycle-Based Host-Guest Interactions. Chem. Rev. 2020, 120, 6070–6123. [Google Scholar] [CrossRef]
- Hu, X.; Xu, G.; Zhang, H.; Li, M.; Tu, Y.; Xie, X.; Zhu, Y.; Jiang, L.; Zhu, X.; Ji, X.; et al. Multifunctional β-Cyclodextrin Polymer for Simultaneous Removal of Natural Organic Matter and Organic Micropollutants and Detrimental Microorganisms from Water. ACS Appl. Mater. Interfaces 2020, 12, 12165–12175. [Google Scholar] [CrossRef] [PubMed]
- Szejtli, J. Utilization of Cyclodextrins in Industrial Products and Processes. J. Mater. Chem. 1997, 7, 575–587. [Google Scholar] [CrossRef]
- Wang, F.; Ma, R.; Tian, Y. Superhydrophobic Starch-Based Adsorbent with Honeycomb Coral-like Surface Fabricated via Facile Immersion Process for Removing Oil from Water. Int. J. Biol. Macromol. 2022, 207, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Ma, R.; Tian, Y. Superhydrophobic Starch-Based Nanocomposite Cryogel for Oil Removal Underwater and Magnetically Guided Oil Slick Cleanup. Carbohydr. Polym. 2022, 287, 119297. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.T.; Nguyen, S.T.; Do, N.D.; Thai, N.N.T.; Thai, Q.B.; Huynh, H.K.P.; Nguyen, V.T.T.; Phan, A.N. Green Aerogels from Rice Straw for Thermal, Acoustic Insulation and Oil Spill Cleaning Applications. Mater. Chem. Phys. 2020, 253, 123363. [Google Scholar] [CrossRef]
- Wang, F.; Ma, R.; Tian, Y. Fabrication of Superhydrophobic/Oleophilic Starch Cryogel via a Simple Sol-Gel Immersion Process for Removing Oil from Water. Ind. Crops Prod. 2022, 184, 115010. [Google Scholar] [CrossRef]
- Wang, F.; Ma, R.; Zhan, J.; Shi, W.; Zhu, Y.; Tian, Y. Superhydrophobic/Superoleophilic Starch-Based Cryogels Coated by Silylated Porous Starch/Fe3O4 Hybrid Micro/Nanoparticles for Removing Discrete Oil Patches from Water. Sep. Purif. Technol. 2022, 291, 120872. [Google Scholar] [CrossRef]
- Wang, F.; Ma, R.; Zhan, J.; Tian, Y. Superhydrophobic Modular Cryogel with Variable Magnetic-Actuated Motion Direction for Discrete Small-Scale Oil Spill Cleanup. J. Hazard. Mater. 2022, 430, 128448. [Google Scholar] [CrossRef]
- Zhou, L.; Chai, K.; Yao, X.; Ji, H. Enhanced Recovery of Acetophenone and 1-Phenylethanol from Petrochemical Effluent by Highly Porous Starch-Based Hypercrosslinked Polymers. Chem. Eng. J. 2021, 418, 129351. [Google Scholar] [CrossRef]
- Zhang, F.; Pei, X.; Zhai, K.; Wang, C.; Bai, Y.; Zhang, B.; Wang, Y.; Tan, Y.; Xu, K.; Wang, P. Starch-Based Nanospheres Modified Filter Paper for o/w Emulsions Separation and Contaminants Removal. Int. J. Biol. Macromol. 2020, 162, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhou, Q.; Liu, F.; Peng, Q.; Bian, Y. Performance and Kinetic of Pesticide Residues Removal by Microporous Starch Immobilized Laccase in a Combined Adsorption and Biotransformation Process. Environ. Technol. Innov. 2021, 21, 101235. [Google Scholar] [CrossRef]
- Suo, F.; You, X.; Ma, Y.; Li, Y. Rapid Removal of Triazine Pesticides by P Doped Biochar and the Adsorption Mechanism. Chemosphere 2019, 235, 918–925. [Google Scholar] [CrossRef]
- Mustofa, D.A.; Gamonchuang, J.; Burakham, R. Magnetic Solid-Phase Extraction Based on Amino-Functionalized Magnetic Starch for Analysis Of Organochlorine Pesticides. Anal. Sci. 2021, 37, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Fallah, Z.; Zare, E.N.; Ghomi, M.; Ahmadijokani, F.; Amini, M.; Tajbakhsh, M.; Arjmand, M.; Sharma, G.; Ali, H.; Ahmad, A.; et al. Toxicity and Remediation of Pharmaceuticals and Pesticides Using Metal Oxides and Carbon Nanomaterials. Chemosphere 2021, 275, 130055. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, N.H.; Shameli, K.; Abdullah, E.C.; Abdullah, L.C. A Facile and Green Synthetic Approach toward Fabrication of Starch-Stabilized Magnetite Nanoparticles. Chin. Chem. Lett. 2017, 28, 1590–1596. [Google Scholar] [CrossRef]
- Yang, C.; Jiang, J.; Wu, Y.; Fu, Y.; Sun, Y.; Chen, F.; Yan, G.; Hu, J. High Removal Rate and Selectivity of Hg(II) Ions Using the Magnetic Composite Adsorbent Based on Starch/Polyethyleneimine. J. Mol. Liq. 2021, 337, 116418. [Google Scholar] [CrossRef]
- Sekhavat Pour, Z.; Ghaemy, M. Removal of Dyes and Heavy Metal Ions from Water by Magnetic Hydrogel Beads Based on Poly(Vinyl Alcohol)/Carboxymethyl Starch-g-Poly(Vinyl Imidazole). RSC Adv. 2015, 5, 64106–64118. [Google Scholar] [CrossRef]
- Sancey, B.; Trunfio, G.; Charles, J.; Minary, J.F.; Gavoille, S.; Badot, P.M.; Crini, G. Heavy Metal Removal from Industrial Effluents by Sorption on Cross-Linked Starch: Chemical Study and Impact on Water Toxicity. J. Environ. Manag. 2011, 92, 765–772. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, C.; Chang, Q. Synthesis of Magnetic Crosslinked Starch-Graft-Poly(Acrylamide)-Co-Sodium Xanthate and Its Application in Removing Heavy Metal Ions. J. Exp. Nanosci. 2017, 12, 270–284. [Google Scholar] [CrossRef]
- Chen, H.; Xie, H.; Zhou, J.; Tao, Y.; Zhang, Y.; Zheng, Q.; Wang, Y. Removal Efficiency of Hexavalent Chromium from Wastewater Using Starch-Stabilized Nanoscale Zero-Valent Iron. Water Sci. Technol. 2019, 80, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Gao, P.; Zhi, H.; Zhao, H.; Yang, Y.; Sun, B. Preparation of Cu(II) Ions Adsorbent from Acrylic Acid-Grafted Corn Starch in Aqueous Solutions. Starch Staerke 2013, 65, 417–424. [Google Scholar] [CrossRef]
- Yu, C.; Tang, X.; Liu, S.; Yang, Y.; Shen, X.; Gao, C. Laponite Crosslinked Starch/Polyvinyl Alcohol Hydrogels by Freezing/Thawing Process and Studying Their Cadmium Ion Absorption. Int. J. Biol. Macromol. 2018, 117, 1–6. [Google Scholar] [CrossRef]
- Darban, Z.; Shahabuddin, S.; Gaur, R.; Ahmad, I.; Sridewi, N. Hydrogel-Based Adsorbent Material for the Effective Removal of Heavy Metals from Wastewater: A Comprehensive Review. Gels 2022, 8, 263. [Google Scholar] [CrossRef]
- Li, P.; Gao, B.; Li, A.; Yang, H. Evaluation of the Selective Adsorption of Silica-Sand/Anionized-Starch Composite for Removal of Dyes and Cupper(II) from Their Aqueous Mixtures. Int. J. Biol. Macromol. 2020, 149, 1285–1293. [Google Scholar] [CrossRef]
- Hou, C.; Ma, H.; Cao, C.; Ding, X.; Duan, J. Adsorption of Cu(Ii) from Solution by Modified Magnetic Starch St/Fe3O4-g-p(AA-r-HEMA). New J. Chem. 2021, 45, 19032–19042. [Google Scholar] [CrossRef]
- Mahdavinia, G.R.; Hasanpour, S.; Behrouzi, L.; Sheykhloie, H. Study on Adsorption of Cu(II) on Magnetic Starch-g-Polyamidoxime/Montmorillonite/Fe3O4 Nanocomposites as Novel Chelating Ligands. Starch Staerke 2016, 68, 188–199. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; El-Sharkawy, R.M.; Ibrahim, G.A.A. Promoted Adsorptive Removal of Chromium(vi) Ions from Water by a Green-Synthesized Hybrid Magnetic Nanocomposite (NFe3O4Starch-Glu-NFe3O4ED). RSC Adv. 2021, 11, 14829–14843. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.N.; Tiwary, D.; Sinha, I. Improved Removal of Cr(VI) by Starch Functionalized Iron Oxide Nanoparticles. J. Environ. Chem. Eng. 2014, 2, 2252–2258. [Google Scholar] [CrossRef]
- Xie, X.; Gao, H.; Luo, X.; Zhang, Y.; Qin, Z.; Ji, H. Polyethyleneimine-Modified Magnetic Starch Microspheres for Cd(II) Adsorption in Aqueous Solutions. Adv. Compos. Hybrid Mater. 2022, 5, 2772–2786. [Google Scholar] [CrossRef]
- Xie, X.; Zhao, X.; Luo, X.; Su, T.; Zhang, Y.; Qin, Z.; Ji, H. Mechanically Activated Starch Magnetic Microspheres for Cd(II) Adsorption from Aqueous Solution. Chin. J. Chem. Eng. 2021, 33, 40–49. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Hamadi, A.; Foroutan, R.; Peighambardoust, S.J.; Ramavandi, B. Decontamination of Cd2+ and Pb2+ from Aqueous Solution Using a Magnetic Nanocomposite of Eggshell/Starch/Fe3O4. J. Water Process Eng. 2022, 48, 102911. [Google Scholar] [CrossRef]
- Konne, J.L.; Opara, K. Remediation of Nickel from Crude Oil Obtained from Bomu Oil Field Using Cassava Waste Water Starch Stabilized Magnetic Nanoparticles. Energy Environ. Res. 2014, 4, 25–31. [Google Scholar] [CrossRef]
- Liu, F.; Peng, G.; Li, T.; Yu, G.; Deng, S. Au(III) Adsorption and Reduction to Gold Particles on Cost-Effective Tannin Acid Immobilized Dialdehyde Corn Starch. Chem. Eng. J. 2019, 370, 228–236. [Google Scholar] [CrossRef]
- Ekebafe, L.O.; Ogbeifun, D.E.; Okieimen, F.E. Equilibrium, Kinetic and Thermodynamic Studies of Lead (II) Sorption on Hydrolyzed Starch Graft Copolymers. J. Polym. Environ. 2018, 26, 807–818. [Google Scholar] [CrossRef]
- Siddiqui, S.I.; Singh, P.N.; Tara, N.; Pal, S.; Chaudhry, S.A.; Sinha, I. Arsenic Removal from Water by Starch Functionalized Maghemite Nano-Adsorbents: Thermodynamics and Kinetics Investigations. Colloids Interface Sci. Commun. 2020, 36, 100263. [Google Scholar] [CrossRef]
- Bisht, G.; Neupane, S. Arsenic Removal through Supercritical Carbon Dioxide-Assisted Modified Magnetic Starch (Starch–Fe3O4) Nanoparticles. Nanotechnol. Environ. Eng. 2018, 3, 8. [Google Scholar] [CrossRef]
- Robinson, M.R.; Coustel, R.; Abdelmoula, M.; Mallet, M. As(V) and As(III) Sequestration by Starch Functionalized Magnetite Nanoparticles: Influence of the Synthesis Route onto the Trapping Efficiency. Sci. Technol. Adv. Mater. 2020, 21, 524–539. [Google Scholar] [CrossRef]
- An, B.; Liang, Q.; Zhao, D. Removal of Arsenic(V) from Spent Ion Exchange Brine Using a New Class of Starch-Bridged Magnetite Nanoparticles. Water Res. 2011, 45, 1961–1972. [Google Scholar] [CrossRef]
- Sami, A.J.; Khalid, M.; Iqbal, S.; Afzal, M.; Shakoori, A.R. Synthesis and Application of ChitosanStarch Based Nanocomposite in Wastewater Treatment for the Removal of Anionic Commercial Dyes. Pak. J. Zool. 2017, 49, 21–26. [Google Scholar] [CrossRef]
- Guibal, E. Interactions of Metal Ions with Chitosan-Based Sorbents: A Review. Sep. Purif. Technol. 2004, 38, 43–74. [Google Scholar] [CrossRef]
- Chassary, P.; Vincent, T.; Guibal, E. Metal Anion Sorption on Chitosan and Derivative Materials: A Strategy for Polymer Modification and Optimum Use. React. Funct. Polym. 2004, 60, 137–149. [Google Scholar] [CrossRef]
- Chiou, M.S.; Li, H.Y. Adsorption Behavior of Reactive Dye in Aqueous Solution on Chemical Cross-Linked Chitosan Beads. Chemosphere 2003, 50, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Pourjavadi, A.; Abedin-Moghanaki, A.; Tavakoli, A. Efficient Removal of Cationic Dyes Using a New Magnetic Nanocomposite Based on Starch-g-Poly(Vinylalcohol) and Functionalized with Sulfate Groups. RSC Adv. 2016, 6, 38042–38051. [Google Scholar] [CrossRef]
- Li, W.; Wei, H.; Liu, Y.; Li, S.; Wang, G.; Han, H. Fabrication of Novel Starch-Based Composite Hydrogel Microspheres Combining Diels-Alder Reaction with Spray Drying for MB Adsorption. J. Environ. Chem. Eng. 2021, 9, 105929. [Google Scholar] [CrossRef]
- Peláez-Cid, A.A.; Herrera-González, A.M.; Salazar-Villanueva, M.; Bautista-Hernández, A. Elimination of Textile Dyes Using Activated Carbons Prepared from Vegetable Residues and Their Characterization. J. Environ. Manag. 2016, 181, 269–278. [Google Scholar] [CrossRef]
- Chen, P.; Cao, Z.F.; Wang, S.; Zhong, H. In Situ Nano-Silicate Functionalized Magnetic Composites by (Poly)Dopamine to Improve MB Removal. Colloids Surf. A Physicochem. Eng. Asp. 2018, 552, 89–97. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, Y.; Cui, Y.; Dai, R.; Shan, Z.; Chen, H. Fabrication of Starch-Based High-Performance Adsorptive Hydrogels Using a Novel Effective Pretreatment and Adsorption for Cationic Methylene Blue Dye: Behavior and Mechanism. Chem. Eng. J. 2021, 405, 126953. [Google Scholar] [CrossRef]
- Xu, E.; Wu, Z.; Ding, T.; Ye, X.; Jin, Z.; Liu, D. Magnetic (Zn-St)10Fe0n (n = 1, 2, 3, 4) Framework of Macro-Mesoporous Biomaterial Prepared via Green Enzymatic Reactive Extrusion for Dye Pollutants Removal. ACS Appl. Mater. Interfaces 2019, 11, 43553–43562. [Google Scholar] [CrossRef]
- Gong, G.; Zhang, F.; Cheng, Z.; Zhou, L. Facile Fabrication of Magnetic Carboxymethyl Starch/Poly(Vinyl Alcohol) Composite Gel for Methylene Blue Removal. Int. J. Biol. Macromol. 2015, 81, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Xu, E.; Ma, S.; Wu, Z.; Wang, W.; Zhang, X.; Tian, J.; Li, D.; Zhou, J.; Liu, D. Bifunctional Fe3O4 Nanoparticles as Magnet and Inducer in Bioextruded Fabrication of Starch-Based Composite with Hierarchical Pore Architecture. Int. J. Biol. Macromol. 2021, 190, 876–886. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, D.; Karak, N. Double Network Hydrophobic Starch Based Amphoteric Hydrogel as an Effective Adsorbent for Both Cationic and Anionic Dyes. Carbohydr. Polym. 2020, 242, 116320. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Fang, K.; Li, J.; Du, N.; Hu, D.; Cao, D.; Tian, R.; Deng, L.; Li, K. Competitive Adsorption Mechanisms of Pigments in Sugarcane Juice on Starch-Based Magnetic Nanocomposites. Int. J. Biol. Macromol. 2023, 231, 123134. [Google Scholar] [CrossRef] [PubMed]
- Grover, A.; Mohiuddin, I.; Malik, A.K.; Aulakh, J.S.; Vikrant, K.; Kim, K.H.; Brown, R.J.C. Magnesium/Aluminum Layered Double Hydroxides Intercalated with Starch for Effective Adsorptive Removal of Anionic Dyes. J. Hazard. Mater. 2022, 424, 127454. [Google Scholar] [CrossRef]
- Foroutan, R.; Peighambardoust, S.J.; Hemmati, S.; Khatooni, H.; Ramavandi, B. Preparation of Clinoptilolite/Starch/CoFe2O4 Magnetic Nanocomposite Powder and Its Elimination Properties for Cationic Dyes from Water and Wastewater. Int. J. Biol. Macromol. 2021, 189, 432–442. [Google Scholar] [CrossRef]
- Esvandi, Z.; Foroutan, R.; Peighambardoust, S.J.; Akbari, A.; Ramavandi, B. Uptake of Anionic and Cationic Dyes from Water Using Natural Clay and Clay/Starch/MnFe2O4 Magnetic Nanocomposite. Surf. Interfaces 2020, 21, 100754. [Google Scholar] [CrossRef]
- Stan, M.; Lung, I.; Soran, M.L.; Opris, O.; Leostean, C.; Popa, A.; Copaciu, F.; Lazar, M.D.; Kacso, I.; Silipas, T.D.; et al. Starch-Coated Green Synthesized Magnetite Nanoparticles for Removal of Textile Dye Optilan Blue from Aqueous Media. J. Taiwan Inst. Chem. Eng. 2019, 100, 65–73. [Google Scholar] [CrossRef]
- Chang, P.R.; Zheng, P.; Liu, B.; Anderson, D.P.; Yu, J.; Ma, X. Characterization of Magnetic Soluble Starch-Functionalized Carbon Nanotubes and Its Application for the Adsorption of the Dyes. J. Hazard. Mater. 2011, 186, 2144–2150. [Google Scholar] [CrossRef]
- Patel, M.; Kumar, R.; Kishor, K.; Mlsna, T.; Pittman, C.U.; Mohan, D. Pharmaceuticals of Emerging Concern in Aquatic Systems: Chemistry, Occurrence, Effects, and Removal Methods. Chem. Rev. 2019, 119, 3510–3673. [Google Scholar] [CrossRef]
- Mohiuddin, I.; Grover, A.; Aulakh, J.S.; Malik, A.K.; Lee, S.S.; Brown, R.J.C.; Kim, K.H. Starch-Mg/Al Layered Double Hydroxide Composites as an Efficient Solid Phase Extraction Sorbent for Non-Steroidal Anti-Inflammatory Drugs as Environmental Pollutants. J. Hazard. Mater. 2021, 401, 123782. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.K.; Mahmoud, M.E. Encapsulation of Starch Hydrogel and Doping Nanomagnetite onto Metal-Organic Frameworks for Efficient Removal of Fluvastatin Antibiotic from Water. Carbohydr. Polym. 2020, 245, 116438. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Xu, M.H.; Wu, T.; Pan, G.X.; Tang, P.S. Adsorption Behavior of Tetracycline on Carboxymethyl Starch Grafted Magnetic Bentonite. Chem. Pap. 2022, 76, 123–135. [Google Scholar] [CrossRef]
- Okoli, C.P.; Ofomaja, A.E. Degree of Time Dependency of Kinetic Coefficient as a Function of Adsorbate Concentration; New Insights from Adsorption of Tetracycline onto Monodispersed Starch-Stabilized Magnetic Nanocomposite. J. Environ. Manag. 2018, 218, 139–147. [Google Scholar] [CrossRef]
- Okoli, C.P.; Ofomaja, A.E. Development of Sustainable Magnetic Polyurethane Polymer Nanocomposite for Abatement of Tetracycline Antibiotics Aqueous Pollution: Response Surface Methodology and Adsorption Dynamics. J. Clean. Prod. 2019, 217, 42–55. [Google Scholar] [CrossRef]
- Okoli, C.P.; Naidoo, E.B.; Ofomaja, A.E. Role of Synthesis Process Variables on Magnetic Functionality, Thermal Stability, and Tetracycline Adsorption by Magnetic Starch Nanocomposite. Environ. Nanotechnol. Monit. Manag. 2018, 9, 141–153. [Google Scholar] [CrossRef]
- He, Q.; Song, P.; Zhang, Z.; You, Z.; Tu, W. Preparation of Magnetic Gelatin-Starch Microspheres and Adsorption Performance for Bovine Serum Album. J. Cent. South Univ. 2015, 22, 1220–1226. [Google Scholar] [CrossRef]
- Chao, Y.; Zhu, W.; Wu, X.; Hou, F.; Xun, S.; Wu, P.; Ji, H.; Xu, H.; Li, H. Application of Graphene-like Layered Molybdenum Disulfide and Its Excellent Adsorption Behavior for Doxycycline Antibiotic. Chem. Eng. J. 2014, 243, 60–67. [Google Scholar] [CrossRef]
- Doǧan, M.; Abak, H.; Alkan, M. Adsorption of Methylene Blue onto Hazelnut Shell: Kinetics, Mechanism and Activation Parameters. J. Hazard. Mater. 2009, 164, 172–181. [Google Scholar] [CrossRef]
- Xu, T.; Qu, R.; Zhang, Y.; Sun, C.; Wang, Y.; Kong, X.; Geng, X.; Ji, C. Preparation of Bifunctional Polysilsesquioxane/Carbon. Nanotube Magnetic Composites and Their Adsorption Properties for Au (III). Chem. Eng. J. 2021, 410, 128225. [Google Scholar] [CrossRef]




| Type of Adsorbent | Type of Pollutant | Pollutant Concentration (mg/L) | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|---|
| Starch-functionalized Iron(III) oxide (Fe2O3) nanoparticles | Lead (II) | 150–450 | 2000 | [64] |
| Activated carbon functionalized magnetic iron oxide nanoparticles | 10–100 | 61.82 | [65] | |
| Activated carbon | 50 | 30 | [66] | |
| Amine functionalized Fe3O4 magnetic nanoparticle dialdehyde starch | Mercury (II) | 150 | 318.87 | [59] |
| Activated carbon | - | 138 | [67] | |
| Starch-based amino-functionalized microspheres | Chromium (VI) | 50 | 734.8 | [60] |
| Activated carbon | 200 | 145 | [68] | |
| Starch-g-polyacrylamide/Fe3O4/graphene oxide nanocomposite | Nickel (II) | 20 | 290 | [69] |
| Activated carbon prepared from coir pith | 20 | 62.5 | [70] | |
| Starch derived zinc carbon foam-like | Malachite green (MG) | 25–100 | 1200 | [71] |
| Carbon prepared from waste jack fruit peel | 20–60 | 166.37 | [72] | |
| Starch-based amino-functionalized microspheres | Indigo carmine (IC) | 50 | 423.69 | [60] |
| Activated carbon | - | 16.3–77.7 | [73] | |
| Starch-g-(acrylic acid-co-acrylamide) functionalized catecholamine | Methylene blue (MB) | 2700 | 2276 | [74] |
| Refused derived fuel | 100 | 83 | [75] |
| Starch-Based Adsorbents | Pollutants | In Situ Remediation | Ref. | |
|---|---|---|---|---|
| Adsorption (%) | Desorption (%) | |||
| Corn starch without pretreatment | Betalain | 36.40 | 35.20 | [93] |
| Corn starch with methanol pretreatment | 21.80 | 58.30 | ||
| Wheat starch without pretreatment | 20.50 | 75.01 | ||
| Wheat starch with methanol pretreatment | 32.72 | 44.40 | ||
| Soluble starch and facultative psychrophilic denitrifier immobilized on macro-porous cellulose | Nitrate | 99.5 | - | [94] |
| Starch-Based Adsorbents | Pollutants | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|
| Sweet potato/Corn/Starch-based adsorbents | Ethanol | 150 | [85] |
| Superhydrophobic starch-based adsorbent | Chloroform | 7560 | [140] |
| n-hexane | 2500 | ||
| Superhydrophobic starch/iron oxide (Fe3O4)/silylated silicon dioxide (SiO2) nanoparticles/cryogel | Chloroform | 7780 | [141] |
| n-hexane | 2720 | ||
| Rice straw-cationic starch aerogel | Oil | 13,000 | [142] |
| Superhydrophobic/oleophilic starch cryogel | Chloroform | 7530 | [143] |
| n-hexane | 2610 | ||
| Superoleophilic starch-based cryogels coated by silylated porous starch/Fe3O4 hybrid micro/nanoparticles | Chloroform | 7570 | [144] |
| n-hexane | 2590 | ||
| Magnetic modular cryogel | Chloroform | 6190 | [145] |
| n-hexane | 2060 | ||
| Starch derived zinc carbon foam-like | Castrol 2T | 2937% | [71] |
| Gear oil | 2375% | ||
| Starch-graft-styrene hypercrosslinked polymers | Acetophenone, | 93.6% | [146] |
| 1-phenylethanol | 74.4% | ||
| Starch-based amino-functionalized microspheres | Oil/water separation | 99.9% | [60] |
| Sweet potato/Corn/Starch-based adsorbents | tert-butyl alcohol (TBA), isopropanol, ethanol | - | [91] |
| Sweet potato/Corn/Starch-based adsorbents | TBA | - | [92] |
| Starch-Based Adsorbents | Pollutants | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|
| Corn/starch-based mesoporous activated carbon (ACS) | pyraclostrobin | 66.2 | [86] |
| Microporous maize starch immobilized laccase | atrazine | 0.2527 | [148] |
| prometryn | 0.1323 | ||
| P-doped biochar from corn straw | triazine | 79.6 | [149] |
| Iron-starch modified with 3,5-diaminobenzidine and (3-aminopropyl) triethoxysilane (Fe-starch@DABA-APTES) | Endosulfan | 0.00025–0.00200 | [150] |
| Heptachlor | 0.00001–0.00075 | ||
| Aldrin | 0.00001–0.00075 | ||
| Isobenzan | 0.00001–0.00075 | ||
| Chlordane | 0.00010–0.00100 | ||
| Dieldrin | 0.00001–0.00075 | ||
| Endrin | 0.00010–0.00100 |
| Starch-Based Adsorbents | Pollutants | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|
| Starch–chitosan-based hydrogel microspheres | Cd(II) | 32.51 | [53] |
| Cu(II) | 47.87 | ||
| Ni(II) | 27.18 | ||
| Starch-functionalized iron(III) oxide (Fe2O3) nanoparticles | Pb(II) | 2000 | [64] |
| Hg(II) | 133.3 | ||
| Cd(II) | 322.58 | ||
| Crosslinked carboxymethyl sago starch/citric acid hydrogel | Pb(II) | 64.48 | [62] |
| Cu(II) | 36.56 | ||
| Ni(II) | 16.21 | ||
| Zn(II) | 18.45 | ||
| Walnut shell ash/starch/iron oxide (Fe3O4) | Cu(II) | 45.4 | [95] |
| Silica-sand/anionized-starch composite | Cu(II) | 383.08 ± 13.50 | [161] |
| Starch/Fe3O4-g-p(AA-r-HEMA) | Cu(II) | 75.5 | [162] |
| Magnetic starch-g-polyamidoxime/montmorillonite/Fe3O4 nanocomposites | Cu(II) | 163 | [163] |
| Starch-based amino-functionalized microspheres | Cr(VI) | 734.8 | [60] |
| Starch-crosslinked magnetic ethylenediamine | Cr(VI) | 210.7 | [164] |
| Starch-functionalized iron oxide nanoparticles | Cr(VI) | 9.02 | [165] |
| Polyethyleneimine-modified magnetic starch microspheres (PEI/MSMs) | Cd(II) | 187.00 | [166] |
| Magnetic starch microspheres (AAM- MSM) | Cd(II) | 39.98 | [167] |
| Cassava-starch-grafted copolymerized AA and AM | Cd(II) | 347.46 | [89] |
| Eggshell/starch/Fe3O4 nanocomposite | Cd(II) | 48.54 | [168] |
| Pb(II) | 57.14 | ||
| Starch-stabilized magnetic nanoparticles | Ni(II) | - | [169] |
| Starch-g-polyacrylamide/Fe3O4/graphene oxide nanocomposite | Ni(II) | 290 | [69] |
| Dialdehyde cornstarch | Gold(III) | 298.5 | [170] |
| Polyethylene-g-poly (acrylic acid)-co-starch/organo-montmorillonite hydrogel | Pb(II) | 430 | [61] |
| Starch graft poly(acrylic) acid | Pb(II) | 118.61 | [171] |
| Starch graft poly(acrylonitrile) | 115.83 | ||
| Amine-functionalized Fe3O4 magnetic nanoparticle dialdehyde starch | Hg(II) | 318.87 | [59] |
| Magnetic starch/polyethyleneimine | Hg(II) | 244.87 | [153] |
| Starch-functionalized maghemite nanoparticles | As(III) | 8.88 | [172] |
| CO2-assisted modified magnetic starch-Fe3O4 nanoparticles | As(III) | 124 | [173] |
| Starch-functionalized magnetite nanoparticles | As(III) | 68.3 | [174] |
| As(V) | 74.8 | ||
| Starch-bridged magnetite nanoparticles | As(V) | 248 | [175] |
| Carboxymethyl starch-g-polyvinyl imidazole | Cu(II) | 83.6 | [154] |
| Cd(II) | 53.2 | ||
| Pb(II) | 65 |
| Starch-Based Adsorbents | Pollutants | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|
| Hydrogel microspheres | Methylene blue (MB) | 106.97 | [53] |
| Eosin yellow (EY) | 143.55 | ||
| Zinc–starch and zerovalent iron extrudates | MB | 61.03 | [185] |
| Magnetism carboxymethyl starch/poly(vinyl alcohol) gel | MB | 23.53 | [186] |
| Zinc–starch–metal–organic coordination polymers-Fe3O4 NPs composite | MB | 37.42 | [187] |
| Double-cross-linked amphoteric hydrogel | MB | 133.65 | [188] |
| Congo red (CR) | 64.73 | ||
| Starch derived zinc carbon foam-like | Crystal violet (CV) | 25,000 | [71] |
| Malachite green (MG) | 1200 | ||
| CR | 1428.57 | ||
| Cationic tapioca starch (CTS)-functionalized magnetic nanoparticles (CTS@Fe3O4) | Caffeic acid (CA) | 185 | [189] |
| Gallic acid (GA) | 160 | ||
| Melanoidin (ME) | 580 | ||
| Rice starch | Methyl orange (MO) | 173.24 | [88] |
| Graham starch | MO | 151.27 | |
| Cassava starch-based hydrogels grafted polyacrylamide | MB | 2000 | [83] |
| Starch-g-(acrylic acid-co-acrylamide)-functionalized catecholamine | MB | 2276 | [74] |
| Starch–magnesium/aluminum-layered double hydroxide | Amaranth | 665 | [190] |
| Tartrazine | 186 | ||
| Sunset yellow (SY) | 71 | ||
| EY | 65 | ||
| Clinoptilolite/Starch/CoFe2O4 | MB | 31.81 | [191] |
| Methylene violet (MV) | 31.15 | ||
| CV | 32.84 | ||
| Corn starch magnetic carbonaceous adsorbent | MV | 344.92 | [78] |
| Carboxymethyl starch-g-polyvinyl imidazole | CR | 83.66 | [154] |
| CV | 91.58 | ||
| Silica-sand/anionized-starch composite | MB | 653.31 ± 27.30 | [161] |
| CV | 1246.40 ± 34.10 | ||
| Clay/starch/MnFe2O4 | SY | 67.82 | [192] |
| Nile blue (NB) | 72.25 | ||
| Magnetic nanoparticles@starch-g-poly(vinyl sulfate) nanocomposite | MB | 621 | [180] |
| MG | 567 | ||
| Starch-coated Fe3O4 magnetic nanoparticles | Option Blue (OB) | 128.83 | [193] |
| Magnetic starch-based composite hydrogel microspheres | MB | 64.05 | [181] |
| Starch-functionalized multiwall carbon nanotube composites | MO | 135.8 | [194] |
| Starch-based amino-functionalized microspheres | Indigo carmine (IC) | 423.69 | [60] |
| Starch-Based Adsorbents | Pollutants | Adsorption Capacity (mg/g) | Ref. |
|---|---|---|---|
| Magnetic metal–organic frameworks (MOFs)-starch hydrogel | Fluvastatin | 782.05 | [197] |
| Carboxymethyl-starch-grafted magnetic bentonite | Tetracycline | 169.7 | [198] |
| Starch-stabilized magnetic nanocomposite | Tetracycline | 24.194 | [199] |
| Magnetic starch polyurethane polymer | Tetracycline | 19.272 | [200] |
| Magnetic starch nanocomposite | Tetracycline | 8.79 | [201] |
| Carboxymethyl cassava starch (CMCS)-functionalized Fe3O4 magnetic nanoparticles | Doxorubicin hydrochloride (Dox) | 235.17 ± 1.75 | [90] |
| Fe3O4 magnetic nanoparticles crosslinked gelatin-starch microspheres | Bovine serum album | 120 | [202] |
| Rape straw/β-CD/Fe3O4 | Ibuprofen | 48.29 | [128] |
| β-Cyclodextrin nanosponge (β−CD−M) | Ibuprofen | 86.21 | [129] |
| Nanocomposite adsorbent based on β-cyclodextrin-PVP-clay | Naproxen | 3.46 | [130] |
| Bio-derived chitosan-EDTA-β-cyclodextrin (CS-ED-CD) trifunctional adsorbent | Bisphenol-S Ciprofoxacin Procaine Imipramine | 43.66 47.11 47.98 41.94 | [126] |
| Calcium(II)-doped chitosan/β-cyclodextrin composite | Acetaminophen | 200.86 | [131] |
| Electronegative silanized β-cyclodextrin adsorbent | Salbutamol Atenolol | 140.24 236.92 | [132] |
| Polyethersulfone nanofibers impregnated with β-cyclodextrin | Estradiol (E2) | 0.000115–0.00029 | [133] |
| β-cyclodextrin/poly (l-glutamic acid) supported magnetic graphene oxide | 17β-estradiol | 85.5 | [134] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoo, P.S.; Ilyas, R.A.; Uda, M.N.A.; Hassan, S.A.; Nordin, A.H.; Norfarhana, A.S.; Ab Hamid, N.H.; Rani, M.S.A.; Abral, H.; Norrrahim, M.N.F.; et al. Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives. Polymers 2023, 15, 3114. https://doi.org/10.3390/polym15143114
Khoo PS, Ilyas RA, Uda MNA, Hassan SA, Nordin AH, Norfarhana AS, Ab Hamid NH, Rani MSA, Abral H, Norrrahim MNF, et al. Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives. Polymers. 2023; 15(14):3114. https://doi.org/10.3390/polym15143114
Chicago/Turabian StyleKhoo, Pui San, R. A. Ilyas, M. N. A. Uda, Shukur Abu Hassan, A. H. Nordin, A. S. Norfarhana, N. H. Ab Hamid, M. S. A. Rani, Hairul Abral, M. N. F. Norrrahim, and et al. 2023. "Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives" Polymers 15, no. 14: 3114. https://doi.org/10.3390/polym15143114
APA StyleKhoo, P. S., Ilyas, R. A., Uda, M. N. A., Hassan, S. A., Nordin, A. H., Norfarhana, A. S., Ab Hamid, N. H., Rani, M. S. A., Abral, H., Norrrahim, M. N. F., Knight, V. F., Lee, C. L., & Rafiqah, S. A. (2023). Starch-Based Polymer Materials as Advanced Adsorbents for Sustainable Water Treatment: Current Status, Challenges, and Future Perspectives. Polymers, 15(14), 3114. https://doi.org/10.3390/polym15143114










