Valorization of Honduran Agro-Food Waste to Produce Bioplastics
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Raw Material Obtention
2.3. Flour Characterization
2.3.1. Chemical Composition
2.3.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.3.3. Granulometry
2.3.4. Differential Scanning Calorimetry (DSC)
2.3.5. Thermogravimetric Analysis (TGA)
2.4. Bioplastics Processing
2.5. Bioplastics Characterization
2.5.1. Dynamic-Mechanical Tests
2.5.2. Static Tests
2.5.3. Water-Uptake Capacity (WUC)
2.6. Statistical Analysis
3. Results and Discussion
3.1. Flour Obtention: Process Performance
3.2. Flour Characterization
3.2.1. Chemical Composition
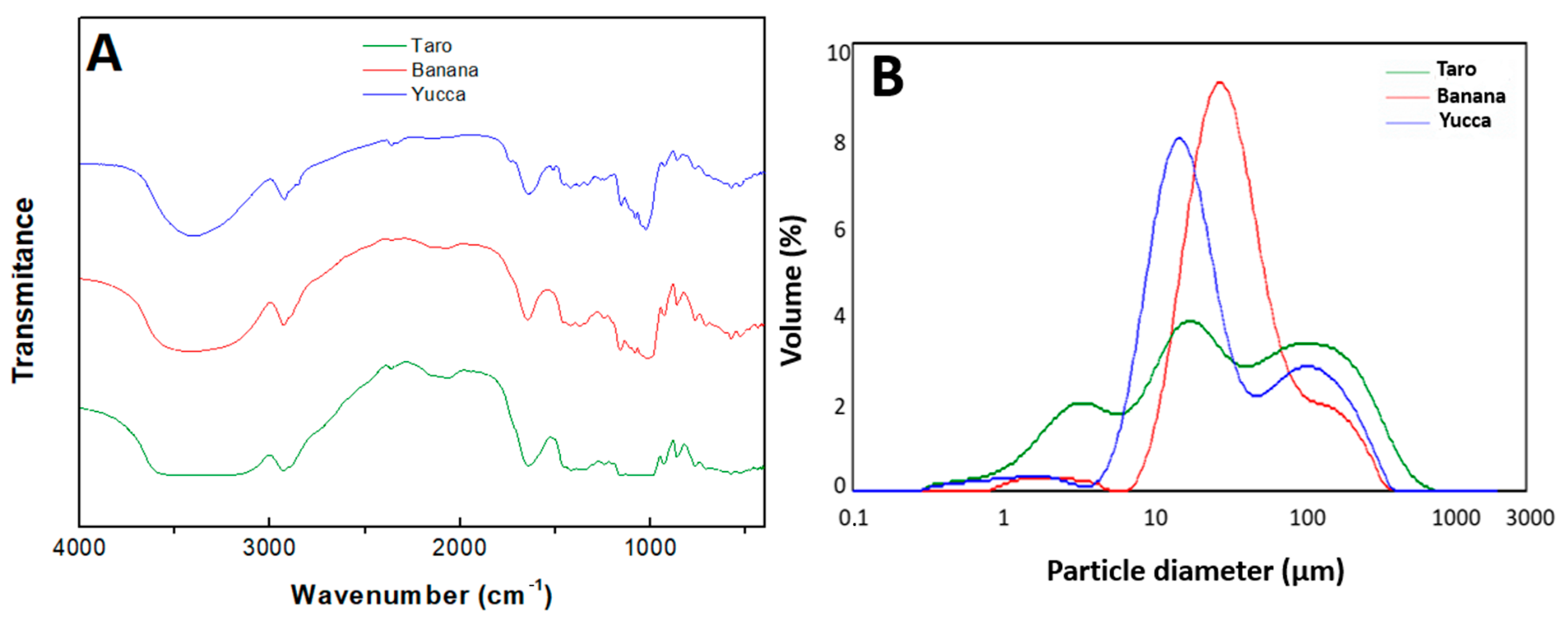
3.2.2. FTIR
3.2.3. Granulometry
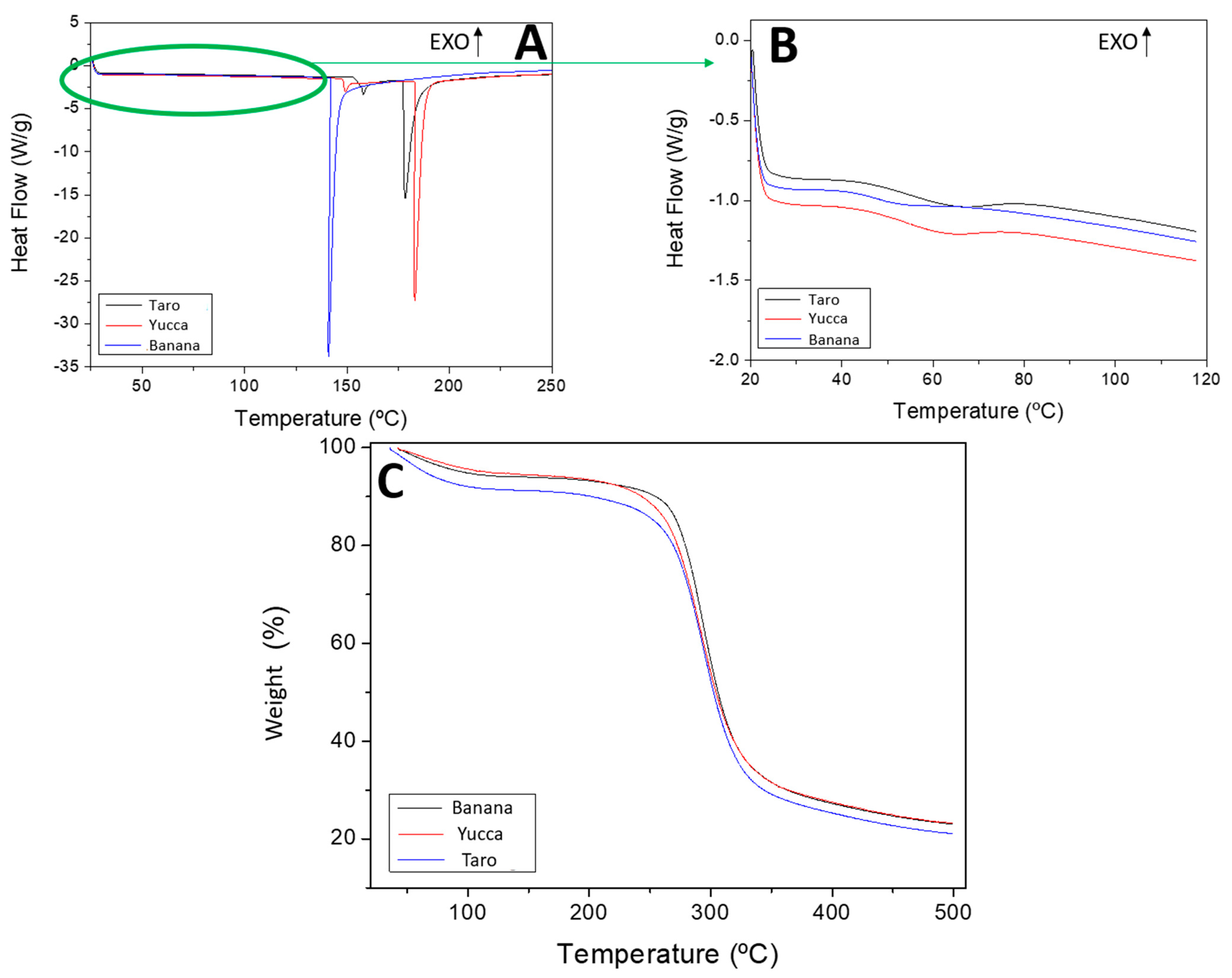
3.2.4. Differential Scanning Calorimetry (DSC)
3.2.5. TGA
3.3. Bioplastics Characterization
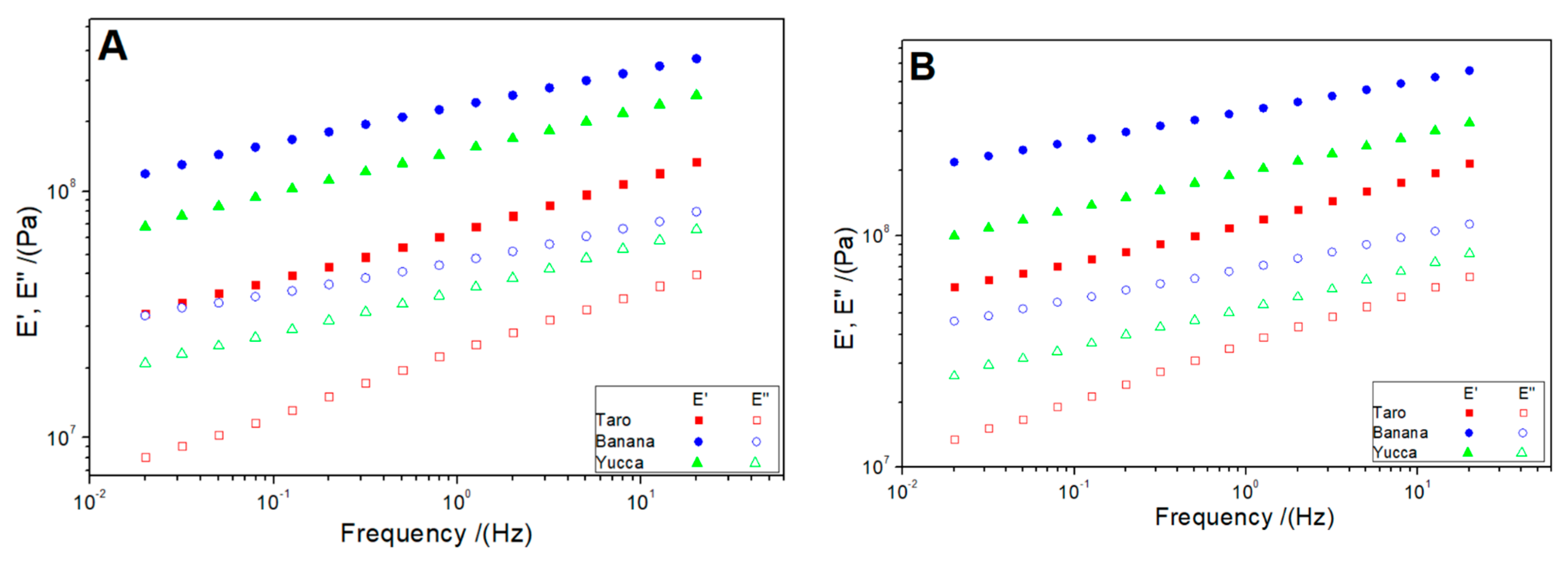
3.3.1. Dynamic-Mechanical Tests
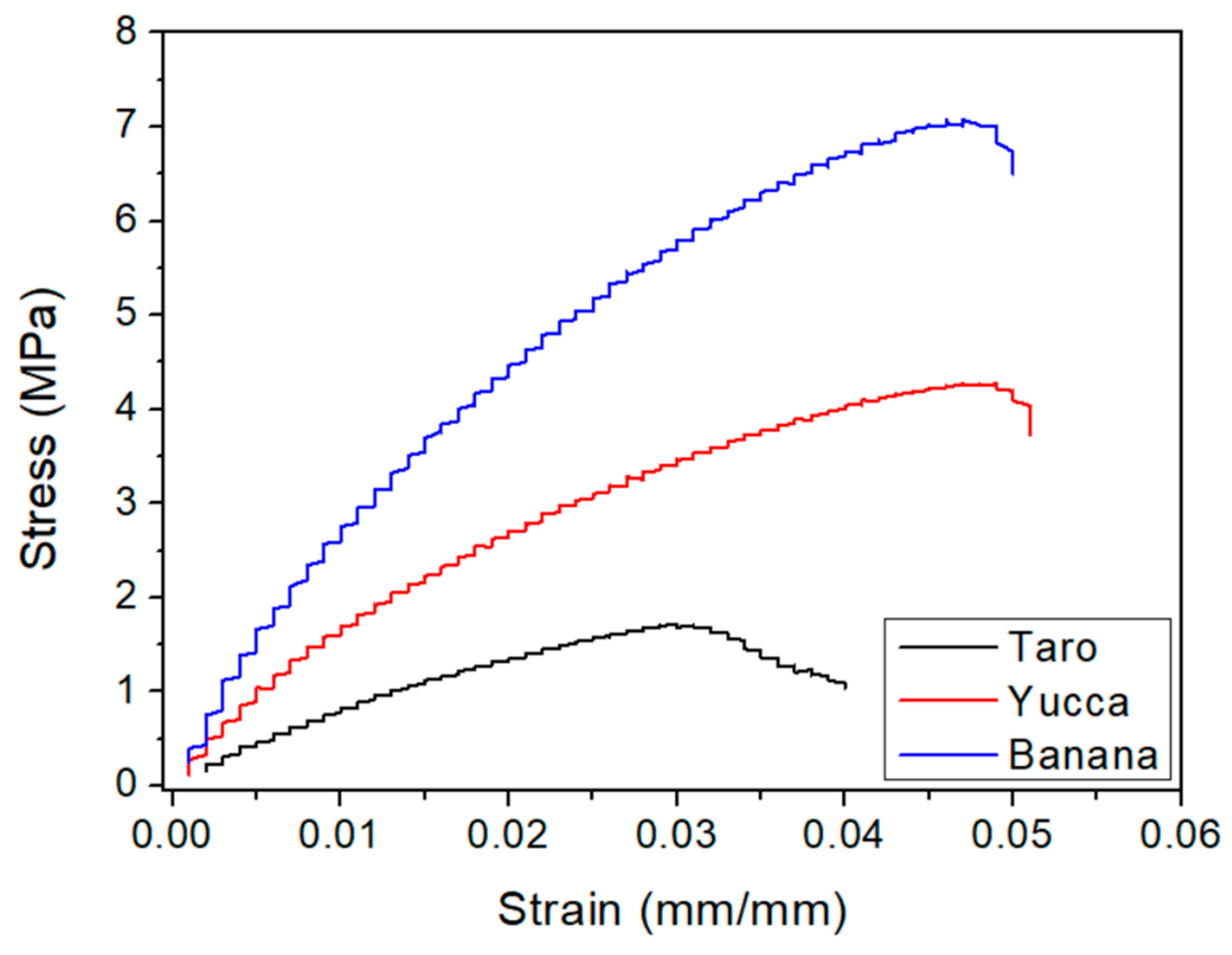
3.3.2. Tensile Static Tests
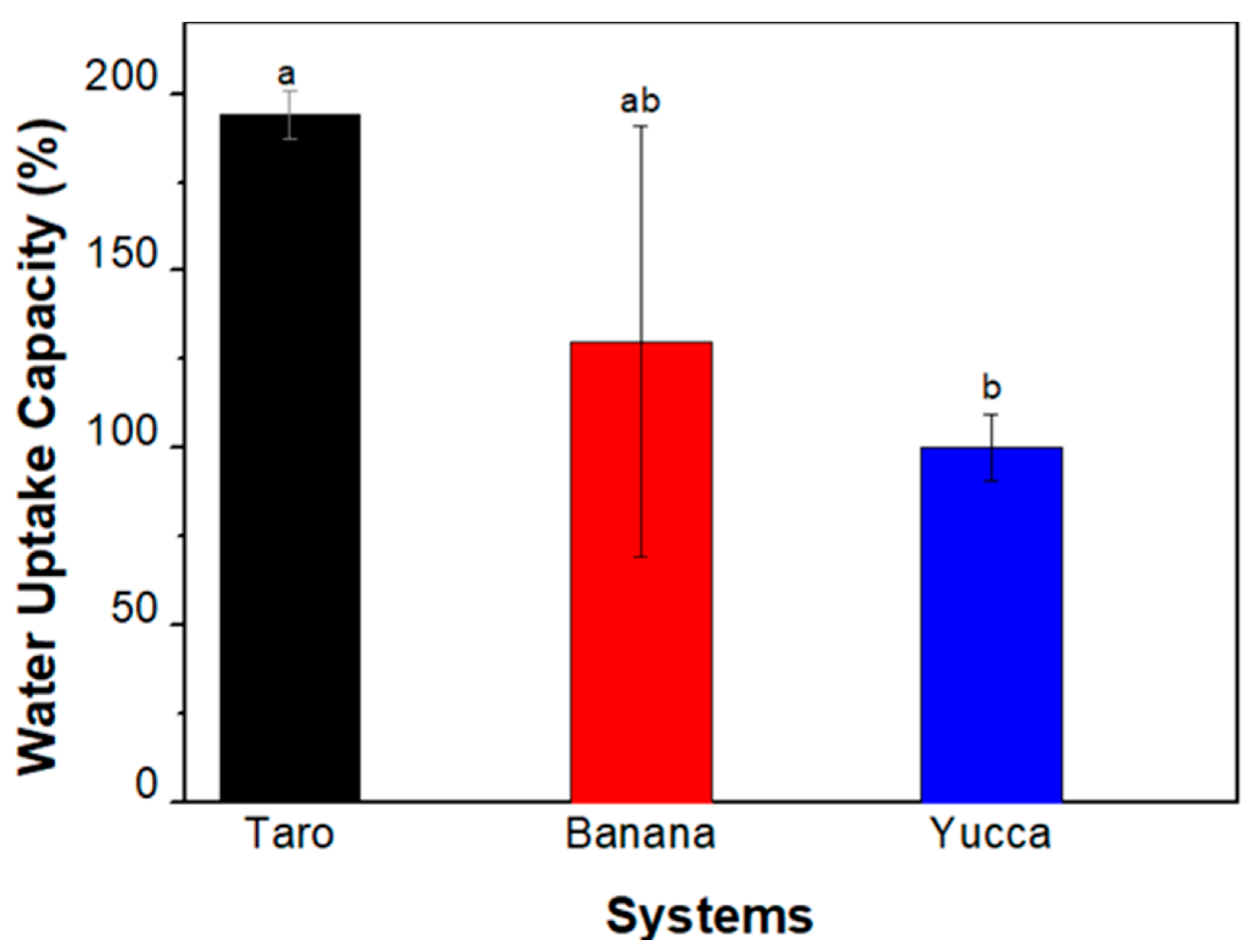
3.3.3. Water-Uptake Capacity (WUC)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.-L.; Zhao, Y.-Y.; Zhang, X.-T.; Shi, X.-P.; Shi, X.-Y.; Li, F.-M. Re-Used Mulching of Plastic Film Is More Profitable and Environmentally Friendly than New Mulching. Soil Tillage Res. 2022, 216, 105256. [Google Scholar] [CrossRef]
- Glaser, T.K.; Plohl, O.; Vesel, A.; Ajdnik, U.; Ulrih, N.P.; Hrnčič, M.K.; Bren, U.; Zemljič, L.F. Functionalization of Polyethylene (PE) and Polypropylene (PP) Material Using Chitosan Nanoparticles with Incorporated Resveratrol as Potential Active Packaging. Materials 2019, 12, 2118. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W.W. Gelatin-Based Film Integrated with Copper Sulfide Nanoparticles for Active Packaging Applications. Appl. Sci. 2021, 11, 6307. [Google Scholar] [CrossRef]
- Kutralam-Muniasamy, G.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Shruti, V.C. Review of Current Trends, Advances and Analytical Challenges for Microplastics Contamination in Latin America. Environ. Pollut. 2020, 267, 115463. [Google Scholar] [CrossRef]
- Raddadi, N.; Fava, F. Biodegradation of Oil-Based Plastics in the Environment: Existing Knowledge and Needs of Research and Innovation. Sci. Total Environ. 2019, 679, 148–158. [Google Scholar] [CrossRef]
- Cazón, P.; Velazquez, G.; Ramírez, J.A.; Vázquez, M. Polysaccharide-Based Films and Coatings for Food Packaging: A Review. Food Hydrocoll. 2017, 68, 136–148. [Google Scholar] [CrossRef]
- Benito-González, I.; López-Rubio, A.; Martínez-Sanz, M. Potential of Lignocellulosic Fractions from Posidonia Oceanica to Improve Barrier and Mechanical Properties of Bio-Based Packaging Materials. Int. J. Biol. Macromol. 2018, 118, 542–551. [Google Scholar] [CrossRef]
- Carvalho, R.A.; Santos, T.A.; de Azevedo, V.M.; Felix, P.H.C.; Dias, M.V.; Borges, S.V. Bio-Nanocomposites for Food Packaging Applications: Effect of Cellulose Nanofibers on Morphological, Mechanical, Optical and Barrier Properties. Polym. Int. 2018, 67, 386–392. [Google Scholar] [CrossRef]
- Zoungranan, Y.; Lynda, E.; Dobi-Brice, K.K.; Tchirioua, E.; Bakary, C.; Yannick, D.D. Influence of Natural Factors on the Biodegradation of Simple and Composite Bioplastics Based on Cassava Starch and Corn Starch. J. Environ. Chem. Eng. 2020, 8, 104396. [Google Scholar] [CrossRef]
- Cruz, R.M.S.; Krauter, V.; Agriopoulou, S.; Weinrich, R.; Herbes, C.; Scholten, P.B.V.; Uysal-unalan, I.; Sogut, E.; Kopacic, S.; Lahti, J.; et al. Bioplastics for Food Packaging: Environmental Impact, Trends and Regulatory Aspects. Foods 2022, 11, 3087. [Google Scholar] [CrossRef]
- Agustin, M.B.; Ahmmad, B.; Alonzo, S.M.M.; Patriana, F.M. Bioplastic Based on Starch and Cellulose Nanocrystals from Rice Straw. J. Reinf. Plast. Compos. 2014, 33, 2205–2213. [Google Scholar] [CrossRef]
- Etxabide, A.; Uranga, J.; Guerrero, P.; de la Caba, K. Development of Active Gelatin Films by Means of Valorisation of Food Processing Waste: A Review. Food Hydrocoll. 2017, 68, 192–198. [Google Scholar] [CrossRef]
- Abdullah, J.A.A.; Yemişken, E.; Guerrero, A.; Romero, A. Marine Collagen-Based Antibacterial Film Reinforced with Graphene and Iron Oxide Nanoparticles. Int. J. Mol. Sci. 2022, 24, 648. [Google Scholar] [CrossRef]
- Abdullah, J.A.A.; Jiménez-Rosado, M.; Benítez, J.J.; Guerrero, A.; Romero, A. Biopolymer-Based Films Reinforced with FexOy-Nanoparticles. Polymers 2022, 14, 4487. [Google Scholar] [CrossRef]
- Abd Elgadir, M.; Mirghani, M.E.S.; Adam, A. Fish Gelatin and Its Applications in Selected Pharmaceutical Aspects as Alternative Source to Pork Gelatin. J. Food Agric. Environ. 2013, 11, 73–79. [Google Scholar]
- Rawdkuen, S.; Thitipramote, N.; Benjakul, S. Preparation and Functional Characterisation of Fish Skin Gelatin and Comparison with Commercial Gelatin. Int. J. Food Sci. Technol. 2013, 48, 1093–1102. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Pérez-Mateos, M.; Gómez-Estaca, J.; López-Caballero, E.; Giménez, B.; Montero, P. Fish Gelatin: A Renewable Material for Developing Active Biodegradable Films. Trends Food Sci. Technol. 2009, 20, 3–16. [Google Scholar] [CrossRef]
- Alfaro, A.D.T.; Balbinot, E.; Weber, C.I.; Tonial, I.B.; Machado-Lunkes, A. Fish Gelatin: Characteristics, Functional Properties, Applications and Future Potentials. Food Eng. Rev. 2015, 7, 33–44. [Google Scholar] [CrossRef]
- Kim, S.-K.; Ngo, D.-H.; Vo, T.-S. Marine Fish-Derived Bioactive Peptides as Potential Antihypertensive Agents. Adv. Food Nutr. Res. 2012, 65, 249–260. [Google Scholar]
- Gudipati, V. Fish Gelatin: A Versatile Ingredient for the Food and Pharmaceutical Industries. In Marine Proteins and Peptides; John Wiley & Sons, Ltd.: Chichester, UK, 2013; pp. 271–295. [Google Scholar]
- Wu Biomedical and Pharmaceutical Application of Fish Collagen and Gelatin: A Review. J. Nutr. Ther. 2013, 2, 218–227. [CrossRef]
- Liu, L.S.; Liu, C.K.; Fishman, M.L.; Hicks, K.B. Composite Films from Pectin and Fish Skin Gelatin or Soybean Flour Protein. J. Agric. Food Chem. 2007, 55, 2349–2355. [Google Scholar] [CrossRef]
- Jiménez-Rosado, M.; Rubio-Valle, J.F.; Perez-Puyana, V.; Guerrero, A.; Romero, A. Use of Heat Treatment for the Development of Protein-Based Bioplastics. Sustain. Chem. Pharm. 2020, 18, 100341. [Google Scholar] [CrossRef]
- Jamilah, B.; Harvinder, K.G. Properties of Gelatins from Skins of Fish—Black Tilapia (Oreochromis Mossambicus) and Red Tilapia (Oreochromis Nilotica). Food Chem. 2002, 77, 81–84. [Google Scholar] [CrossRef]
- Amin, J.; Abdullah, A.; Jiménez-rosado, M.; Guerrero, A.; Romero, A. Biopolymer-Based Films Reinforced with Green Synthesized Zinc Oxide Nanoparticles. Polymers 2022, 14, 5202. [Google Scholar]
- Pratiwi, R.; Rahayu, D.; Barliana, M.I. Characterization of Bioplastic from Rice Straw Cellulose. Res. J. Pharm. Biol. Chem. Sci. 2017, 8, 217–221. [Google Scholar]
- De Corato, U. Bioplastics from Winemaking By-Products in the Buildings Sector: A Feasibility Study on the Main Opportunities, Barriers and Challenges. Circ. Econ. Sustain. 2021, 1, 1313–1333. [Google Scholar] [CrossRef]
- Mohammed, K. Production of Bioplastics from Agricultural Waste, Mainly Banana Peels, “Musa Sapientum”, Using Batch Reactor. Afr. J. Eng. Technol. 2021. [Google Scholar]
- Guliani, E.; Jeyaseelan, C. Bioplastics from microbial and agricultural biomass. In Green. Sustainable Process for Chemical and Environmental Engineering and Science; Elsevier: Amsterdam, The Netherlands, 2023; pp. 395–412. [Google Scholar]
- Shanmathy, M.; Mohanta, M.; Thirugnanam, A. Development of Biodegradable Bioplastic Films from Taro Starch Reinforced with Bentonite. Carbohydr. Polym. Technol. Appl. 2021, 2, 100173. [Google Scholar] [CrossRef]
- Sharmiladevi, S.; Ramesh, N.; Ramesh, S. Production of Bio Degradable Bags Using Cassava Starch. Int. Res. J. Multidiscip. Technovation 2019, 1, 553–559. [Google Scholar] [CrossRef]
- Udo, I.I.; Etokakpan, O.U.; Ukwo, S.P.; Ukpong, E.O. Evaluation of the Proximate Compositions, Dietary Fibre and Resistant Starch Contents of Selected Varieties of Banana and Plantain. J. Adv. Food Sci. Technol. 2021, 8, 1–9. [Google Scholar]
- AOAC International. Official Methods of Analysis of AOAC International; AOAC International: Rockville, MD, USA, 2005; ISBN 9780935584752. [Google Scholar]
- Perera, C.; Brown, R.H. A New Method for Lipid Extraction at Ambient Temperature. Ciencia y Tecnologia Alimentaria 1996, 1, 13–18. [Google Scholar] [CrossRef]
- Perez, V.; Felix, M.; Romero, A.; Guerrero, A. Characterization of Pea Protein-Based Bioplastics Processed by Injection Moulding. Food Bioprod. Process. 2016, 97, 100–108. [Google Scholar] [CrossRef]
- He, J.; Chen, L.; Chu, B.; Zhang, C. Determination of Total Polysaccharides and Total Flavonoids in Chrysanthemum Morifolium Using Near-Infrared Hyperspectral Imaging and Multivariate Analysis. Molecules 2018, 23, 2395. [Google Scholar] [CrossRef]
- ISO 133320:2020; Particle Size Analysis—Laser Diffraction Methods. Aenor: Madrid, Spain, 2020.
- Felix, M.; Romero, A.; Cordobes, F.; Guerrero, A. Development of Crayfish Bio-Based Plastic Materials Processed by Small-Scale Injection Moulding. J. Sci. Food Agric. 2015, 95, 679–687. [Google Scholar] [CrossRef]
- Liu, H.; Xie, F.; Yu, L.; Chen, L.; Li, L. Thermal Processing of Starch-Based Polymers. Prog. Polym. Sci. 2009, 34, 1348–1368. [Google Scholar] [CrossRef]
- Alonso-González, M.; Felix, M.; Guerrero, A.; Romero, A. Effects of Mould Temperature on Rice Bran-Based Bioplastics Obtained by Injection Moulding. Polymers 2021, 13, 398. [Google Scholar] [CrossRef]
- Mekonnen, T.; Mussone, P.; Khalil, H.; Bressler, D. Progress in Bio-Based Plastics and Plasticizing Modifications. J. Mater. Chem. A Mater. 2013, 1, 13379. [Google Scholar] [CrossRef]
- ISO 527-2:2012; Plastics—Determination of Tensile Properties-Part 2: Test Conditions for Moulding and Extrusion Plastics. Aenor: Madrid, Spain, 2012.
- ASTM D570-98; Standard Test Method for Water Absorption of Plastics. ASTM International: West Conshohocken, PA, USA, 2005.
- da Mota, R.V.; Lajolo, F.M.; Cordenunsi, B.R.; Ciacco, C. Composition and Functional Properties of Banana Flour from Different Varieties. Starch Stärke 2000, 52, 63–68. [Google Scholar] [CrossRef]
- Alonso-González, M.; Felix, M.; Romero, A. Rice Bran-Based Bioplastics: Effects of Biopolymer Fractions on Their Mechanical, Functional and Microstructural Properties. Polymers 2021, 14, 100. [Google Scholar] [CrossRef]
- Abdullah, J.A.A.; Rosado, M.J.; Guerrero, A.; Romero, A. Eco-Friendly Synthesis of ZnO-Nanoparticles Using Phoenix Dactylifera L., Polyphenols: Physicochemical, Microstructural, and Functional Assessment. New J. Chem. 2023, 47, 4409–4417. [Google Scholar] [CrossRef]
- Byler, D.M.; Susi, H. Examination of the Secondary Structure of Proteins by Deconvolved FTIR Spectra. Biopolymers 1986, 25, 469–487. [Google Scholar] [CrossRef]
- Muyonga, J.H.; Cole, C.G.B.; Duodu, K.G. Fourier Transform Infrared (FTIR) Spectroscopic Study of Acid Soluble Collagen and Gelatin from Skins and Bones of Young and Adult Nile Perch (Lates niloticus). Food Chem. 2004, 86, 325–332. [Google Scholar] [CrossRef]
- Abdullah, J.A.A.; Jiménez-Rosado, M.; Perez-Puyana, V.; Guerrero, A.; Romero, A. Green Synthesis of FexOy Nanoparticles with Potential Antioxidant Properties. Nanomaterials 2022, 12, 2449. [Google Scholar] [CrossRef]
- Turi, E.A. Thermal Characterisation of Polymeric Materials. Polym. Test. 1997, 16, 523–524. [Google Scholar]
- Jagadeesh, D.; Jeevan Prasad Reddy, D.; Varada Rajulu, A. Preparation and Properties of Biodegradable Films from Wheat Protein Isolate. J. Polym. Environ. 2011, 19, 248–253. [Google Scholar] [CrossRef]
- Ghizdareanu, A.-I.; Banu, A.; Pasarin, D.; Ionita (Afilipoaei), A.; Nicolae, C.-A.; Gabor, A.R.; Pătroi, D. Enhancing the Mechanical Properties of Corn Starch Films for Sustainable Food Packaging by Optimizing Enzymatic Hydrolysis. Polymers 2023, 15, 1899. [Google Scholar] [CrossRef]
- Maraveas, C. Environmental Sustainability of Greenhouse Covering Materials. Sustainability 2019, 11, 6129. [Google Scholar] [CrossRef]
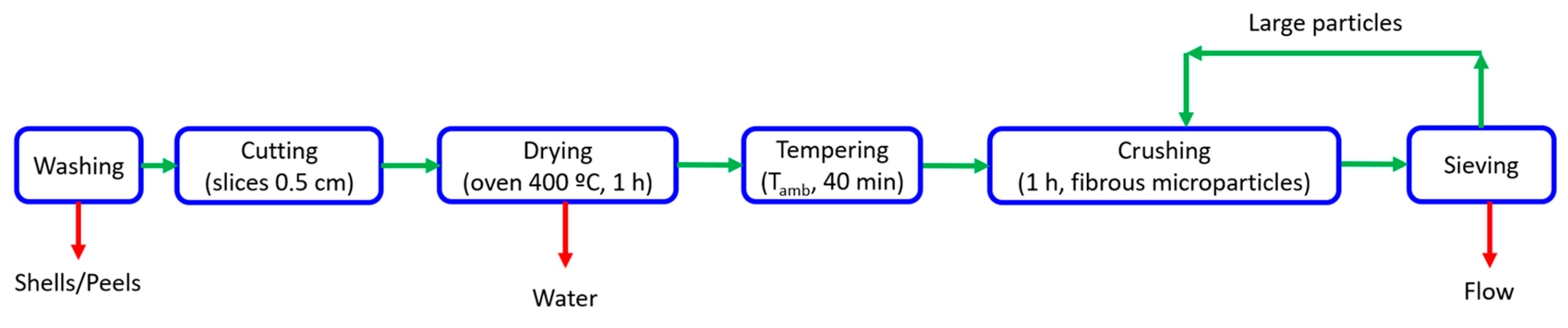
| Sample | Peeling (%) | Cutting (%) | Drying (%) | Crushing + Sieving (%) |
|---|---|---|---|---|
| Taro | 92 | 90 | 33 | 88 |
| Yucca | 80 | 98 | 37 | 92 |
| Banana | 56 | 99 | 45 | 93 |
| Sample | Moisture (wt%) | Ashes (wt%) | Lipids (wt%) | Proteins (wt%) | Polysaccharides (wt%) |
|---|---|---|---|---|---|
| Taro | 0.60 ± 0.04 | 6.90 ± 0.07 | 1.74 ± 0.61 | 4.71 ± 0.10 | 64.4 ± 1.3 |
| Yucca | 1.20 ± 0.03 | 5.45 ± 0.08 | 0.90 ± 0.02 | 2.87 ± 0.06 | 56.4 ± 3.4 |
| Banana | 1.50 ± 0.04 | 7.81 ± 0.05 | 0.22 ± 0.05 | 2.25 ± 0.17 | 59.2 ± 2.2 |
| System | Dynamic-Mechanical Tests | Tensile Static Analysis | |||||
|---|---|---|---|---|---|---|---|
| Flexural Mode | Tensile Mode | ||||||
| E′B (Pa) | tan δ1B (-) | E′1T (Pa) | tan δ1F (-) | E (MPa) | σmax (MPa) | εmax (mm/mm) | |
| Taro | 0.69 ± 0.05 | 0.33 ± 0.02 | 1.13 ± 0.07 | 0.30 ± 0.04 | 99.5 ± 10.0 | 1.8 ± 0.4 | 0.03 ± 0.01 |
| Yucca | 1.46 ± 0.08 | 0.27 ± 0.03 | 1.89 ± 0.10 | 0.26 ± 0.02 | 187.6 ± 14.9 | 4.4 ± 0.1 | 0.05 ± 0.01 |
| Banana | 2.21 ± 0.11 | 0.24 ± 0.02 | 3.46 ± 0.15 | 0.22 ± 0.01 | 308.5 ± 13.9 | 6.3 ± 1.2 | 0.05 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro-Criado, D.; Rivera-Flores, O.; Abdullah, J.A.A.; Castro-Osorto, E.; Alonso-González, M.; Ramos-Casco, L.; Perez-Puyana, V.M.; Sánchez-Barahona, M.; Sánchez-Cid, P.; Jiménez-Rosado, M.; et al. Valorization of Honduran Agro-Food Waste to Produce Bioplastics. Polymers 2023, 15, 2625. https://doi.org/10.3390/polym15122625
Castro-Criado D, Rivera-Flores O, Abdullah JAA, Castro-Osorto E, Alonso-González M, Ramos-Casco L, Perez-Puyana VM, Sánchez-Barahona M, Sánchez-Cid P, Jiménez-Rosado M, et al. Valorization of Honduran Agro-Food Waste to Produce Bioplastics. Polymers. 2023; 15(12):2625. https://doi.org/10.3390/polym15122625
Chicago/Turabian StyleCastro-Criado, Daniel, Octavio Rivera-Flores, Johar Amin Ahmed Abdullah, Elia Castro-Osorto, María Alonso-González, Lucy Ramos-Casco, Víctor M. Perez-Puyana, Marlon Sánchez-Barahona, Pablo Sánchez-Cid, Mercedes Jiménez-Rosado, and et al. 2023. "Valorization of Honduran Agro-Food Waste to Produce Bioplastics" Polymers 15, no. 12: 2625. https://doi.org/10.3390/polym15122625
APA StyleCastro-Criado, D., Rivera-Flores, O., Abdullah, J. A. A., Castro-Osorto, E., Alonso-González, M., Ramos-Casco, L., Perez-Puyana, V. M., Sánchez-Barahona, M., Sánchez-Cid, P., Jiménez-Rosado, M., & Romero, A. (2023). Valorization of Honduran Agro-Food Waste to Produce Bioplastics. Polymers, 15(12), 2625. https://doi.org/10.3390/polym15122625









