Effect of Tannin Furanic Polymer in Comparison to Its Mimosa Tannin Extract on the Growth of Bacteria and White-Rot Fungi
Abstract
1. Introduction
2. Materials and Methods
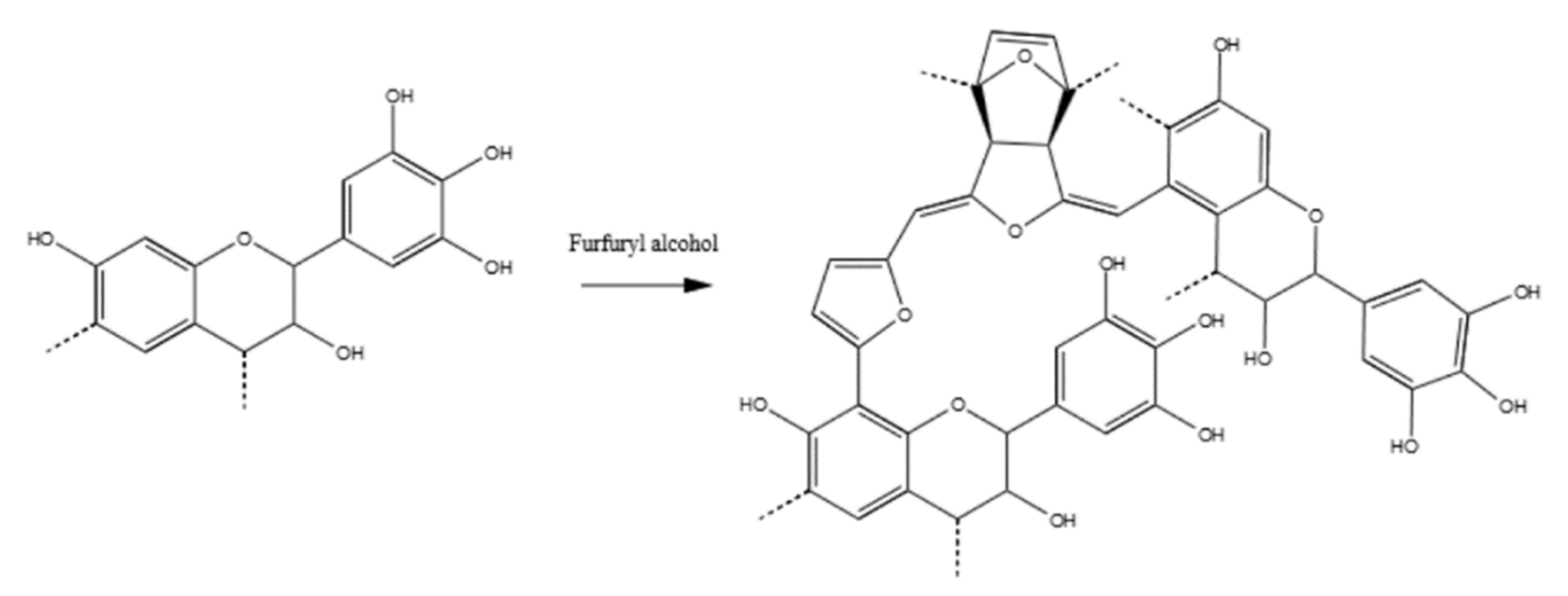
2.1. Materials
2.2. Methods
2.2.1. Bacteria Tests
2.2.2. Fungal Tests
2.3. Statistical Analysis
3. Results
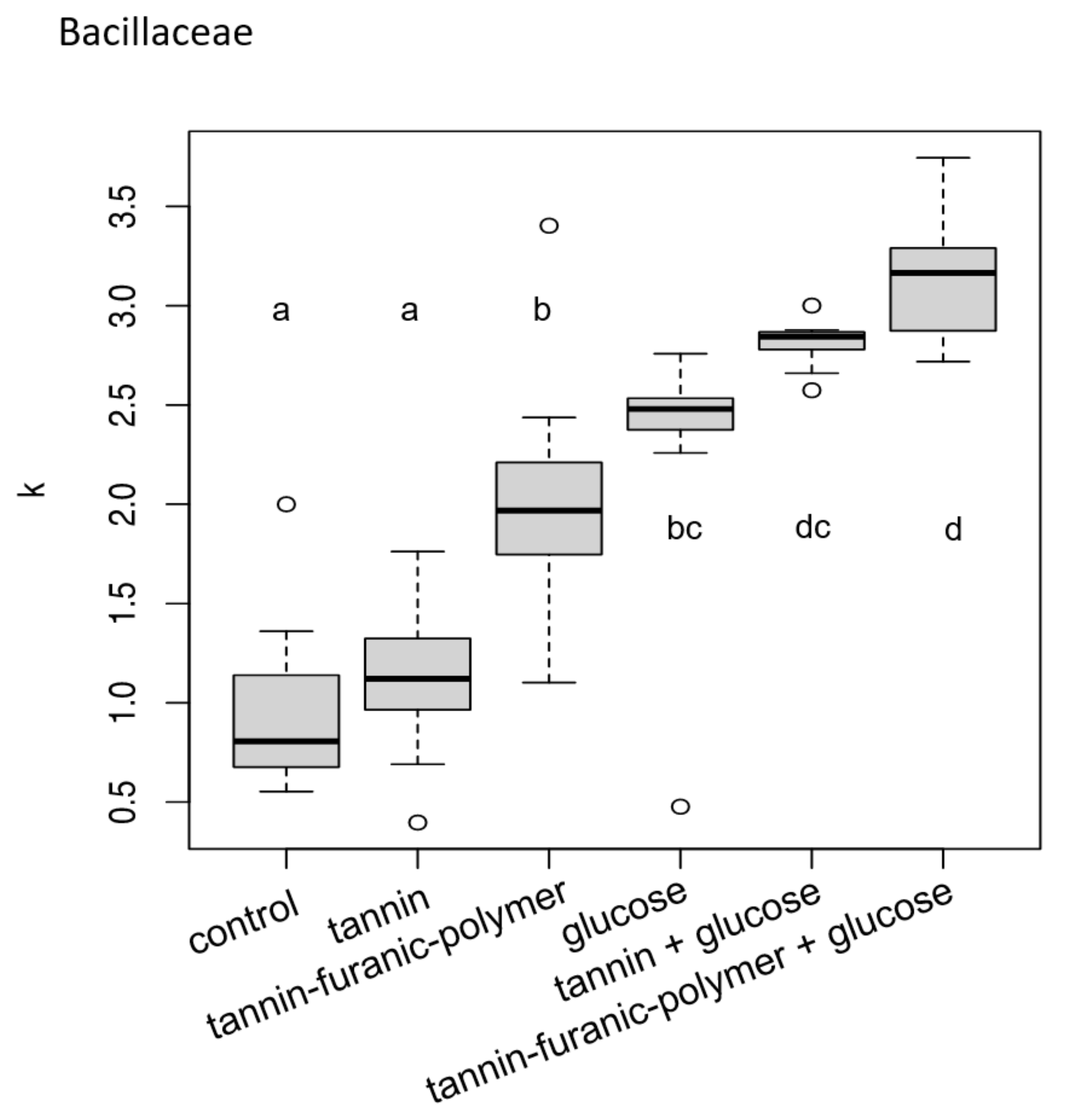
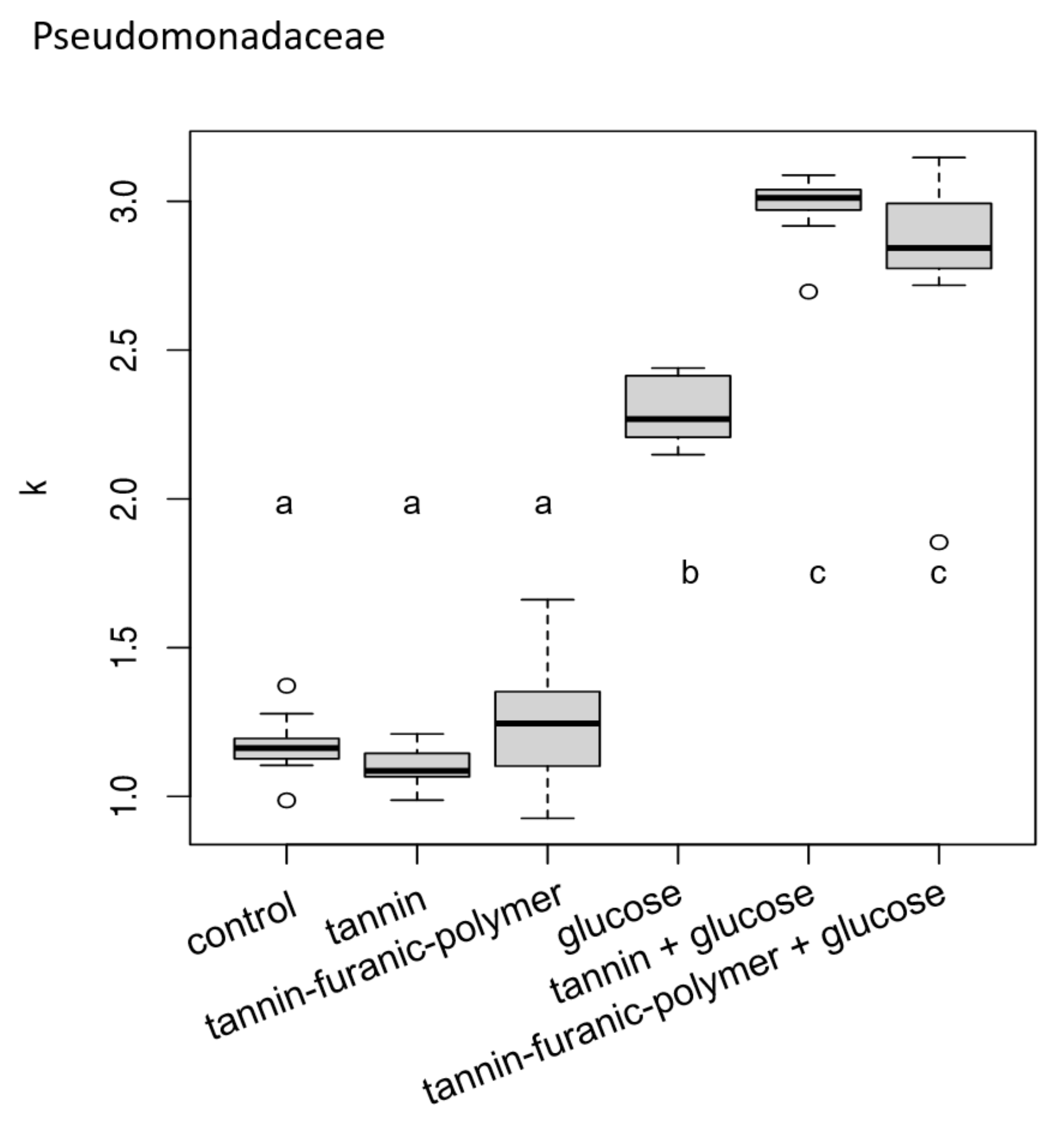
3.1. Bacteria Exposure Tests
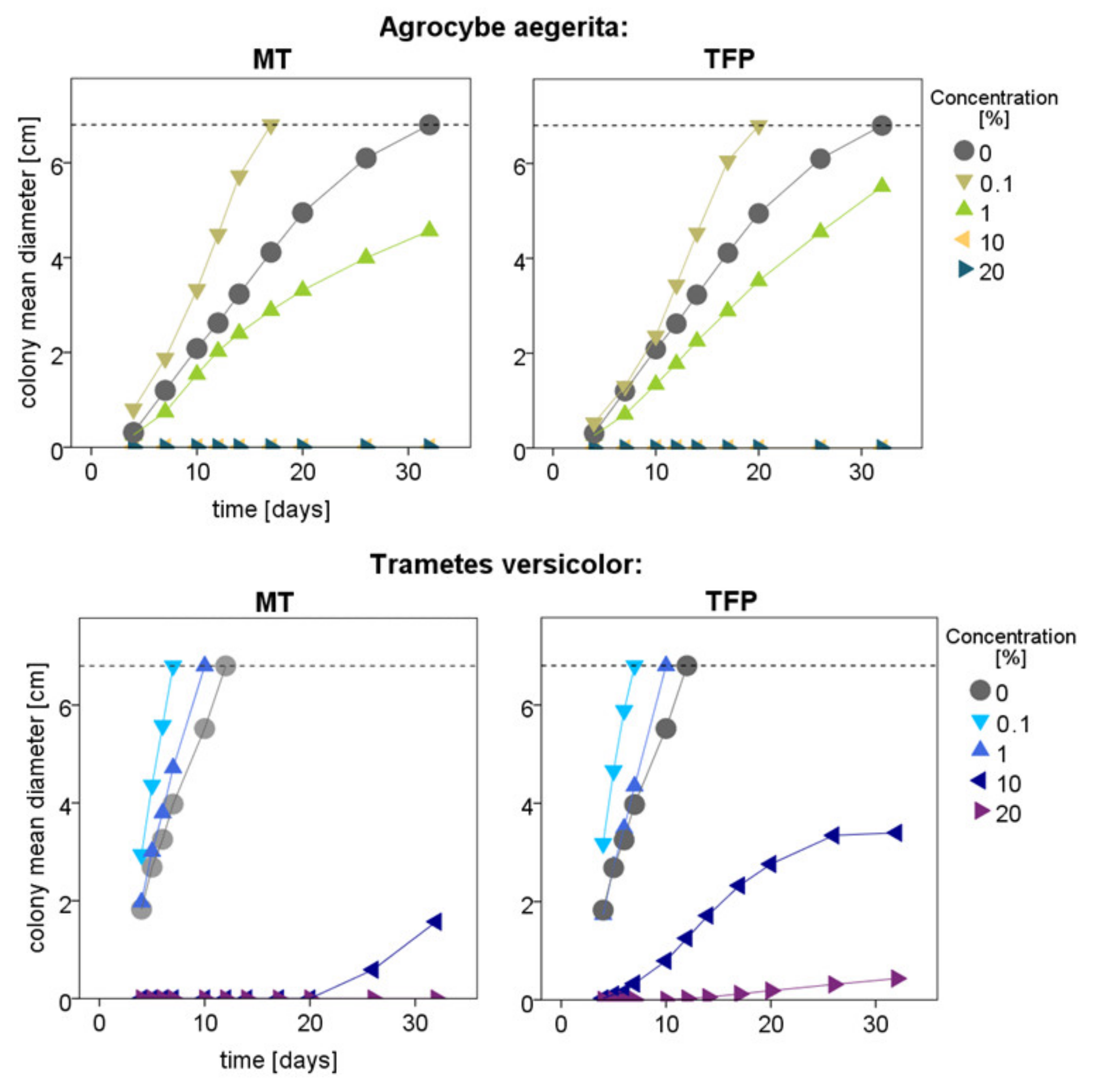
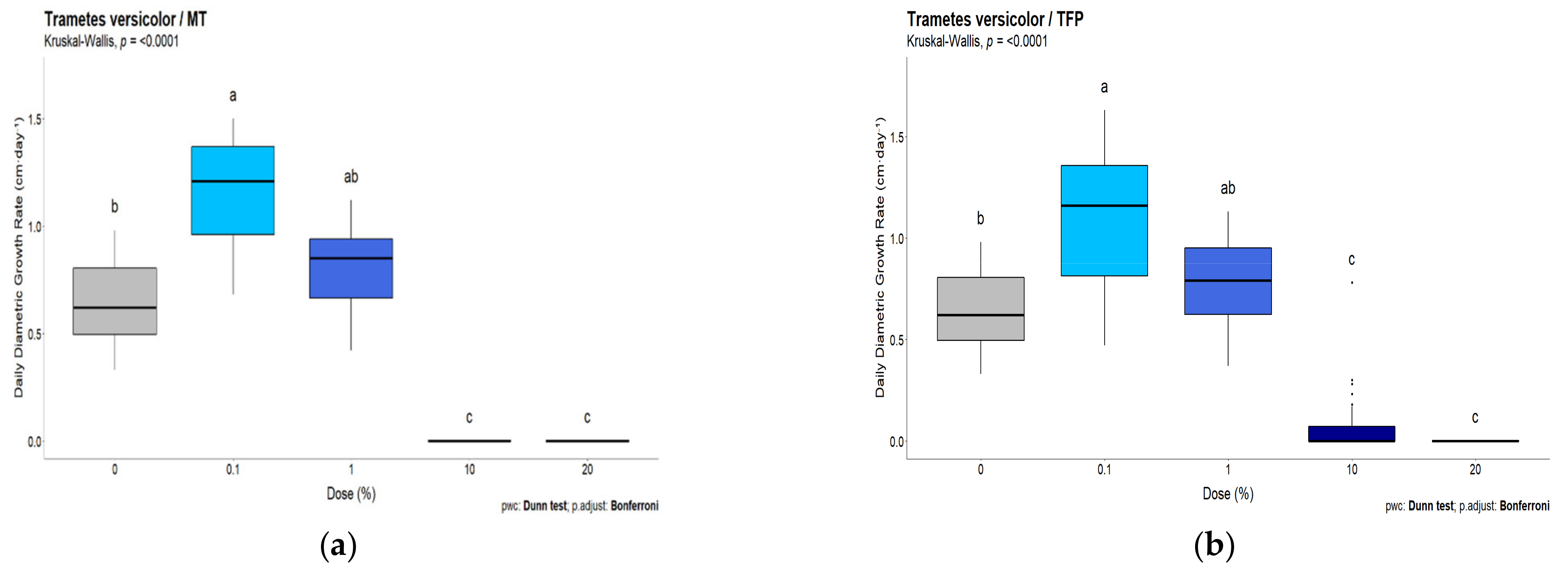
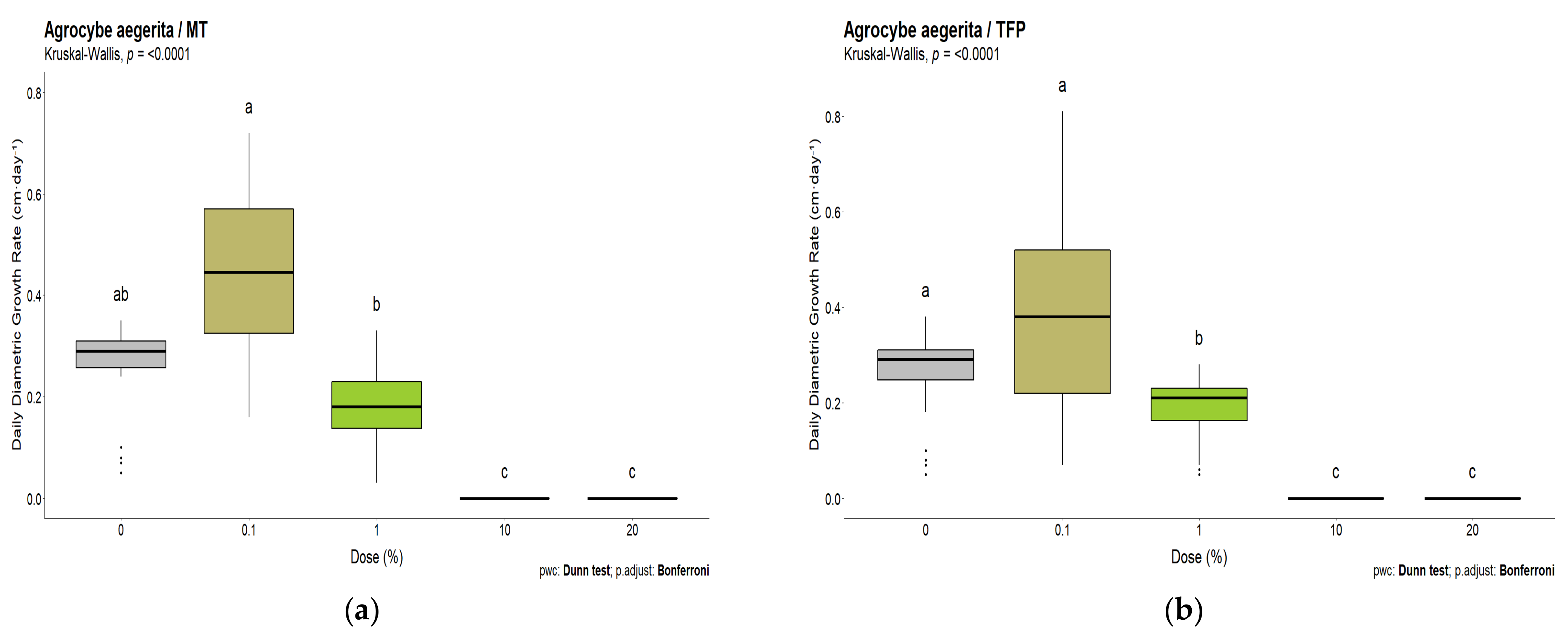
3.2. White-Rot Fungal Exposure Tests
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Valette, N.; Perrot, T.; Sormani, R.; Gelhaye, E.; Morel-Rouhier, M. Antifungal Activities of Wood Extractives. Fungal Biol. Rev. 2017, 31, 113–123. [Google Scholar] [CrossRef]
- Anderson, R.C.; Vodovnik, M.; Min, B.R.; Pinchak, W.E.; Krueger, N.A.; Harvey, R.B.; Nisbet, D.J. Bactericidal Effect of Hydrolysable and Condensed Tannin Extracts on Campylobacter Jejuni in Vitro. Folia Microbiol. 2012, 57, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Schultz, T.P.; Nicholas, D.D. Development of Environmentally-Benign Wood Preservatives Based on the Combination of Organic Biocides with Antioxidants and Metal Chelators. Phytochemistry 2002, 61, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Tomak, E.D.; Gonultas, O. The Wood Preservative Potentials of Valonia, Chestnut, Tara and Sulphited Oak Tannins. J. Wood Chem. Technol. 2018, 38, 183–197. [Google Scholar] [CrossRef]
- Chang, T.-C.; Chang, H.-T.; Wu, C.-L.; Chang, S.-T. Influences of Extractives on the Photodegradation of Wood. Polym. Degrad. Stab. 2010, 95, 516–521. [Google Scholar] [CrossRef]
- Sepperer, T.; Hernandez-Ramos, F.; Labidi, J.; Oostingh, G.J.; Bogner, B.; Petutschnigg, A.; Tondi, G. Purification of Industrial Tannin Extract through Simple Solid-Liquid Extractions. Ind. Crop. Prod. 2019, 139, 111502. [Google Scholar] [CrossRef]
- Tondi, G. Tannin-Based Copolymer Resins: Synthesis and Characterization by Solid State 13C NMR and FT-IR Spectroscopy. Polymers 2017, 9, 223. [Google Scholar] [CrossRef]
- Luckeneder, P.; Gavino, J.; Kuchernig, R.; Petutschnigg, A.; Tondi, G. Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives. Polymers 2016, 8, 396. [Google Scholar] [CrossRef]
- Li, X.; Nicollin, A.; Pizzi, A.; Zhou, X.; Sauget, A.; Delmotte, L. Natural Tannin–Furanic Thermosetting Moulding Plastics. RSC Adv. 2013, 3, 17732. [Google Scholar] [CrossRef]
- Abdullah, U.H.B.; Pizzi, A. Tannin-Furfuryl Alcohol Wood Panel Adhesives without Formaldehyde. Eur. J. Wood Prod. 2013, 71, 131–132. [Google Scholar] [CrossRef]
- Tondi, G.; Pizzi, A.; Olives, R. Natural Tannin-Based Rigid Foams as Insulation for Doors and Wall Panels. Maderas Cienc. Tecnol. 2008, 10, 219–227. [Google Scholar] [CrossRef]
- Tondi, G.; Pizzi, A. Tannin-Based Rigid Foams: Characterization and Modification. Ind. Crop. Prod. 2009, 29, 356–363. [Google Scholar] [CrossRef]
- Celzard, A.; Fierro, V.; Amaral-Labat, G.; Pizzi, A.; Torero, J. Flammability Assessment of Tannin-Based Cellular Materials. Polym. Degrad. Stab. 2011, 96, 477–482. [Google Scholar] [CrossRef]
- Pizzi, A.P.; Khan, A. Furanic Rigid Foams, Furanic-Based Bioplastics and Furanic-Derived Wood Adhesives and Bioadhesives. In Furan Derivatives-Recent Advances and Applications; IntechOpen: London, UK, 2021; pp. 1–19. [Google Scholar]
- Link, M.; Kolbitsch, C.; Tondi, G.; Ebner, M.; Wieland, S.; Petutschnigg, A. Formaldehyde-Free Tannin-Based Foams and Their Use as Lightweight Panels. BioResources 2011, 6, 4218–4228. [Google Scholar] [CrossRef]
- Tondi, G.; Link, M.; Kolbitsch, C.; Lesacher, R.; Petutschnigg, A. Pilot Plant Up-Scaling of Tannin Foams. Ind. Crop. Prod. 2016, 79, 211–218. [Google Scholar] [CrossRef]
- Sepperer, T.; Tondi, G.; Petutschnigg, A.; Young, T.M.; Steiner, K. Mitigation of Ammonia Emissions from Cattle Manure Slurry by Tannins and Tannin-Based Polymers. Biomolecules 2020, 10, 581. [Google Scholar] [CrossRef]
- Tondi, G.; Oo, C.W.; Pizzi, A.; Trosa, A.; Thevenon, M.F. Metal Adsorption of Tannin Based Rigid Foams. Ind. Crop. Prod. 2009, 29, 336–340. [Google Scholar] [CrossRef]
- Sepperer, T.; Neubauer, J.; Eckardt, J.; Schnabel, T.; Petutschnigg, A.; Tondi, G. Pollutant Absorption as a Possible End-Of-Life Solution for Polyphenolic Polymers. Polymers 2019, 11, 911. [Google Scholar] [CrossRef]
- Zanetti, M.; Cesprini, E.; Marangon, M.; Szczurek, A.; Tondi, G. Thermal Valorization and Elemental Composition of Industrial Tannin Extracts. Fuel 2021, 289, 119907. [Google Scholar] [CrossRef]
- Sepperer, T.; Petutschnigg, A.; Steiner, K. Long-Term Study on the Nitrogen Retention Potential of Bark Extracts and a Polymer Based Thereof in Cattle Manure Slurry. Bioresour. Technol. Rep. 2022, 18, 101085. [Google Scholar] [CrossRef]
- Ogawa, S.; Yazaki, Y. Tannins from Acacia mearnsii De Wild. Bark: Tannin determination and biological activities. Molecules 2018, 23, 837. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.; Jin, L.; Niu, Y.D.; Huang, Q.; McAllister, T.A.; Yang, H.E.; Denise, H.; Xu, Z.; Acharya, S.; Wang, S.; et al. Condensed tannins affect bacterial and fungal microbiomes and mycotoxin production during ensiling and upon aerobic exposure. Appl. Environ. Microbiol. 2018, 84, e02274-17. [Google Scholar] [CrossRef] [PubMed]
- Bhat, T.K.; Singh, B.; Sharma, O.P. Microbial Degradation of Tannins—A Current Perspective. Biodegradation 1998, 9, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Prigione, V.; Spina, F.; Tigini, V.; Giovando, S.; Varese, G.C. Biotransformation of industrial tannins by filamentous fungi. Appl. Microbiol. Biotechnol. 2018, 102, 10361–10375. [Google Scholar] [CrossRef] [PubMed]
- Tondi, G.; Petutschnigg, A. Middle Infrared (ATR FT-MIR) Characterization of Industrial Tannin Extracts. Ind. Crop. Prod. 2015, 65, 422–428. [Google Scholar] [CrossRef]
- Eckardt, J.; Neubauer, J.; Sepperer, T.; Donato, S.; Zanetti, M.; Cefarin, N.; Vaccari, L.; Lippert, M.; Wind, M.; Schnabel, T.; et al. Synthesis and Characterization of High-Performing Sulfur-Free Tannin Foams. Polymers 2020, 12, 564. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; Version 3.6.0; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Sprouffske, K.; Wagner, A. Growthcurver: An R Package for Obtaining Interpretable Metrics from Microbial Growth Curves. BMC Bioinform. 2016, 17, 172. [Google Scholar] [CrossRef]
- Kassambara, A. Ggpubr: “ggplot2” Based Publication Ready Plots; Version 0.5.0. CRAN; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Kassambara, A. Rstatix: Pipe-Friendly Framework for Basic Statistical Tests; Version 0.7.0. CRAN; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Wickham, H.; Averick, M.; Bryan, M. Welcome to the Tidyverse. J. Open Source Softw. 2020, 4, 1686. [Google Scholar] [CrossRef]
- Saxena, R.K.; Sharmila, P.; Singh, V.P. Microbial Degradation of Tannins. In Progress in Industrial Microbiology; Elsevier: Amsterdam, The Netherlands, 1995; Volume 32, pp. 259–270. ISBN 978-0-444-81977-2. [Google Scholar]
- Hernández, M.C.; Esquivel, J.C.C.; Lara, F.; Rodríguez, R.; Aguilar, C.N. Isolation and Evaluation of Tannin-Degrading Fungal Strains from the Mexican Desert. Z. Nat. C 2005, 60, 844–848. [Google Scholar] [CrossRef]
- Sambandam, T.; Mahadevan, A. Degradation of Catechin and Purification and Partial Characterization of Catechin Oxygenase FromChaetomium Cupreum. World J. Microbiol. Biotechnol. 1993, 9, 37–44. [Google Scholar] [CrossRef]
- Arunachalam, M.; Mohan, N.; Sugadev, R.; Chellappan, P.; Mahadevan, A. Degradation of (+)-Catechin by Acinetobacter Calcoaceticus MTC 127. Biochim. Biophys. Acta—Gen. Subj. 2003, 1621, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Sinsabaugh, R.L. Phenol Oxidase, Peroxidase and Organic Matter Dynamics of Soil. Soil Biol. Biochem. 2010, 42, 391–404. [Google Scholar] [CrossRef]
- Zengin, G.; Locatelli, M.; Carradori, S.; Mocan, A.M.; Aktumsek, A. Total Phenolics, Flavonoids, Condensed Tannins Content of Eight Centaurea Species and Their Broad Inhibitory Activities against Cholinesterase, Tyrosinase, α-Amylase and α-Glucosidase. Not. Bot. Horti Agrobot. Cluj-Napoca 2016, 44, 195–200. [Google Scholar] [CrossRef]
- Barrett, A.; Ndou, T.; Hughey, C.A.; Straut, C.; Howell, A.; Dai, Z.; Kaletunc, G. Inhibition of α-Amylase and Glucoamylase by Tannins Extracted from Cocoa, Pomegranates, Cranberries, and Grapes. J. Agric. Food Chem. 2013, 61, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.; Mateus, N.; de Freitas, V. Inhibition of α-Amylase Activity by Condensed Tannins. Food Chem. 2011, 125, 665–672. [Google Scholar] [CrossRef]
- Takagi, K.; Mitsunaga, T. Tyrosinase Inhibitory Activity of Proanthocyanidins from Woody Plants. J. Wood Sci. 2003, 49, 461–465. [Google Scholar] [CrossRef]
- Hagerman, A.E. Chemistry of Tannin-Protein Complexation. In Chemistry and Significance of Condensed Tannins; Hemingway, R.W., Karchesy, J.J., Branham, S.J., Eds.; Springer: Boston, MA, USA, 1989; pp. 323–333. ISBN 978-1-4684-7513-5. [Google Scholar]
- Liu, J.; Liu, Y.; He, X.; Teng, B.; McRae, J.M. Valonea Tannin: Tyrosinase Inhibition Activity, Structural Elucidation and Insights into the Inhibition Mechanism. Molecules 2021, 26, 2747. [Google Scholar] [CrossRef]
- Peng, B.; Su, Y.; Li, H.; Han, Y.; Guo, C.; Tian, Y.; Peng, X. Exogenous Alanine and/or Glucose plus Kanamycin Kills Antibiotic-Resistant Bacteria. Cell Metab. 2015, 21, 249–262. [Google Scholar] [CrossRef]
- Cabecas Segura, P.; De Meur, Q.; Tanghe, A.; Onderwater, R.; Dewasme, L.; Wattiez, R.; Leroy, B. Effects of Mixing Volatile Fatty Acids as Carbon Sources on Rhodospirillum Rubrum Carbon Metabolism and Redox Balance Mechanisms. Microorganisms 2021, 9, 1996. [Google Scholar] [CrossRef]
- Anttila, A.-K.; Pirttilä, A.M.; Häggman, H.; Harju, A.; Venäläinen, M.; Haapala, A.; Holmbom, B.; Julkunen-Tiitto, R. Condensed Conifer Tannins as Antifungal Agents in Liquid Culture. Holzforschung 2013, 67, 825–832. [Google Scholar] [CrossRef]
- Goldstein, J.L.; Swain, T. The Inhibition of Enzymes by Tannins. Phytochemistry 1965, 4, 185–192. [Google Scholar] [CrossRef]
- Scalbert, A. Antimicrobial Properties of Tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Türkan, F.; Taslimi, P.; Saltan, F.Z. Tannic Acid as a Natural Antioxidant Compound: Discovery of a Potent Metabolic Enzyme Inhibitor for a New Therapeutic Approach in Diabetes and Alzheimer’s Disease. J. Biochem. Mol. Toxicol. 2019, 33, e22340. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eckardt, J.; Tondi, G.; Fanchin, G.; Lach, A.; Junker, R.R. Effect of Tannin Furanic Polymer in Comparison to Its Mimosa Tannin Extract on the Growth of Bacteria and White-Rot Fungi. Polymers 2023, 15, 175. https://doi.org/10.3390/polym15010175
Eckardt J, Tondi G, Fanchin G, Lach A, Junker RR. Effect of Tannin Furanic Polymer in Comparison to Its Mimosa Tannin Extract on the Growth of Bacteria and White-Rot Fungi. Polymers. 2023; 15(1):175. https://doi.org/10.3390/polym15010175
Chicago/Turabian StyleEckardt, Jonas, Gianluca Tondi, Genny Fanchin, Alexander Lach, and Robert R. Junker. 2023. "Effect of Tannin Furanic Polymer in Comparison to Its Mimosa Tannin Extract on the Growth of Bacteria and White-Rot Fungi" Polymers 15, no. 1: 175. https://doi.org/10.3390/polym15010175
APA StyleEckardt, J., Tondi, G., Fanchin, G., Lach, A., & Junker, R. R. (2023). Effect of Tannin Furanic Polymer in Comparison to Its Mimosa Tannin Extract on the Growth of Bacteria and White-Rot Fungi. Polymers, 15(1), 175. https://doi.org/10.3390/polym15010175







