Current State and Perspectives of Simulation and Modeling of Aliphatic Isocyanates and Polyisocyanates
Abstract
1. Introduction
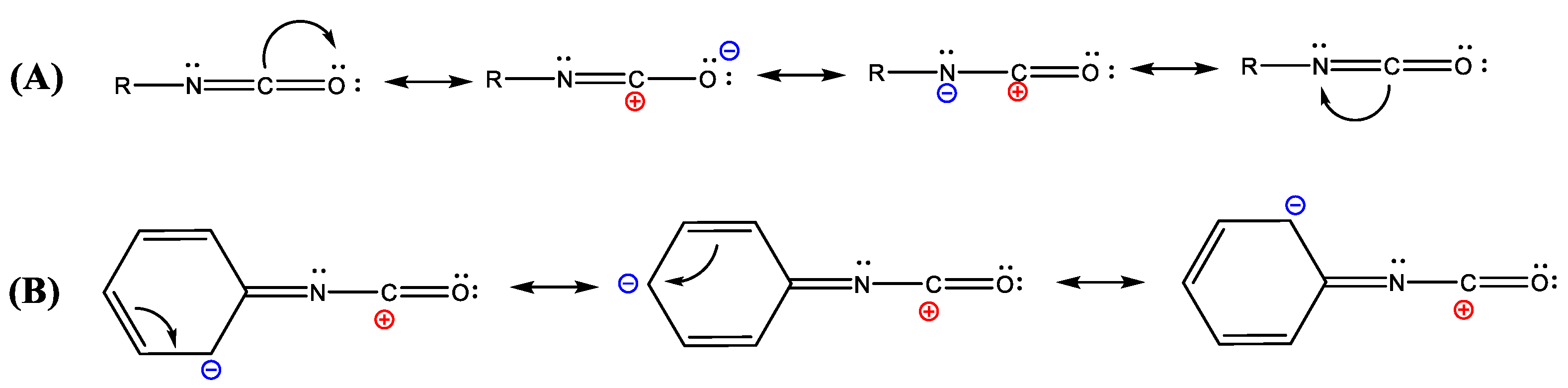
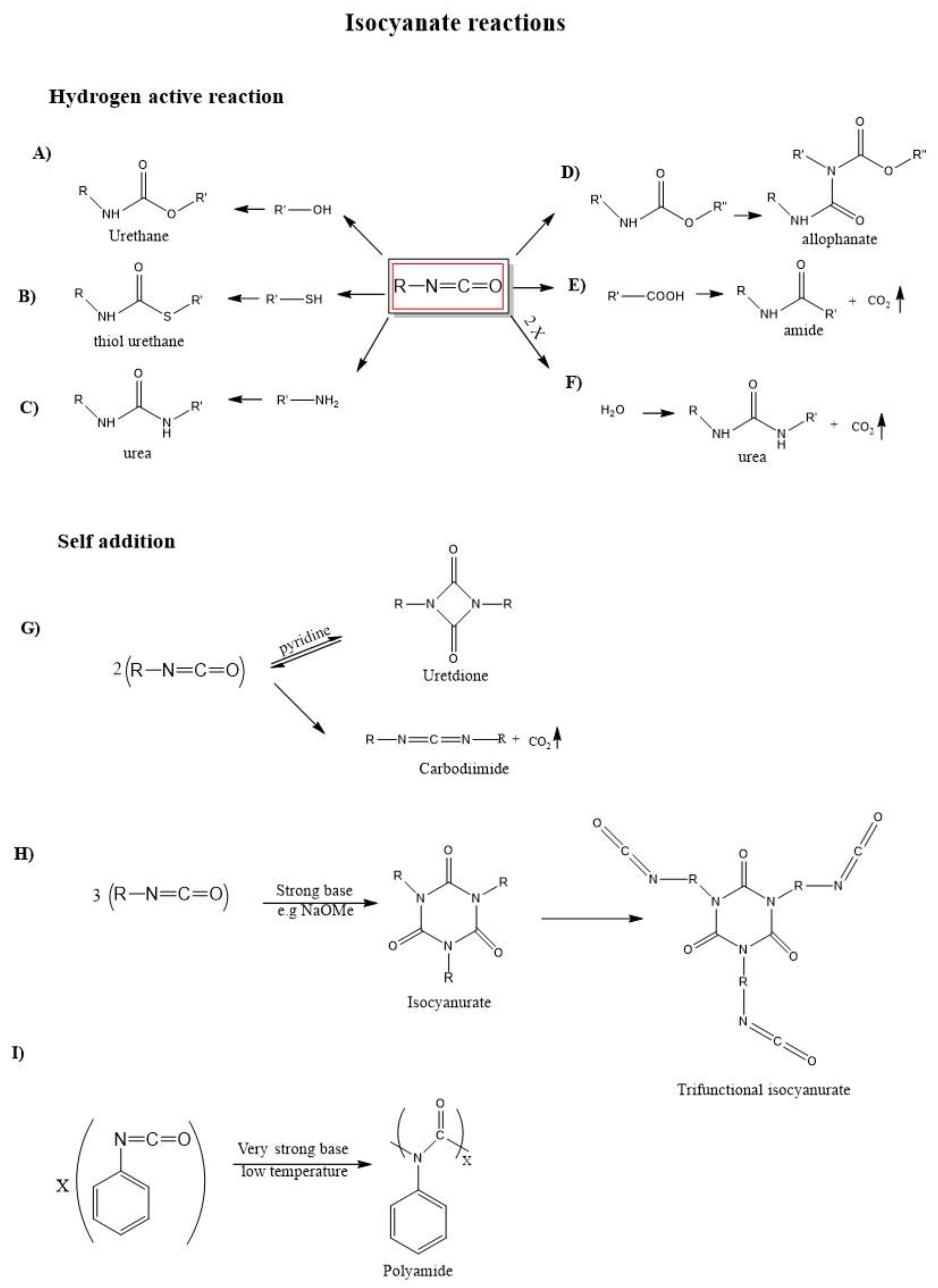
2. Isocyanates and Polyisocyanates: Definitions and Chemistry
3. Applications and Research Trends
4. Modeling and Simulation of Isocyanates
4.1. Ab Initio Studies
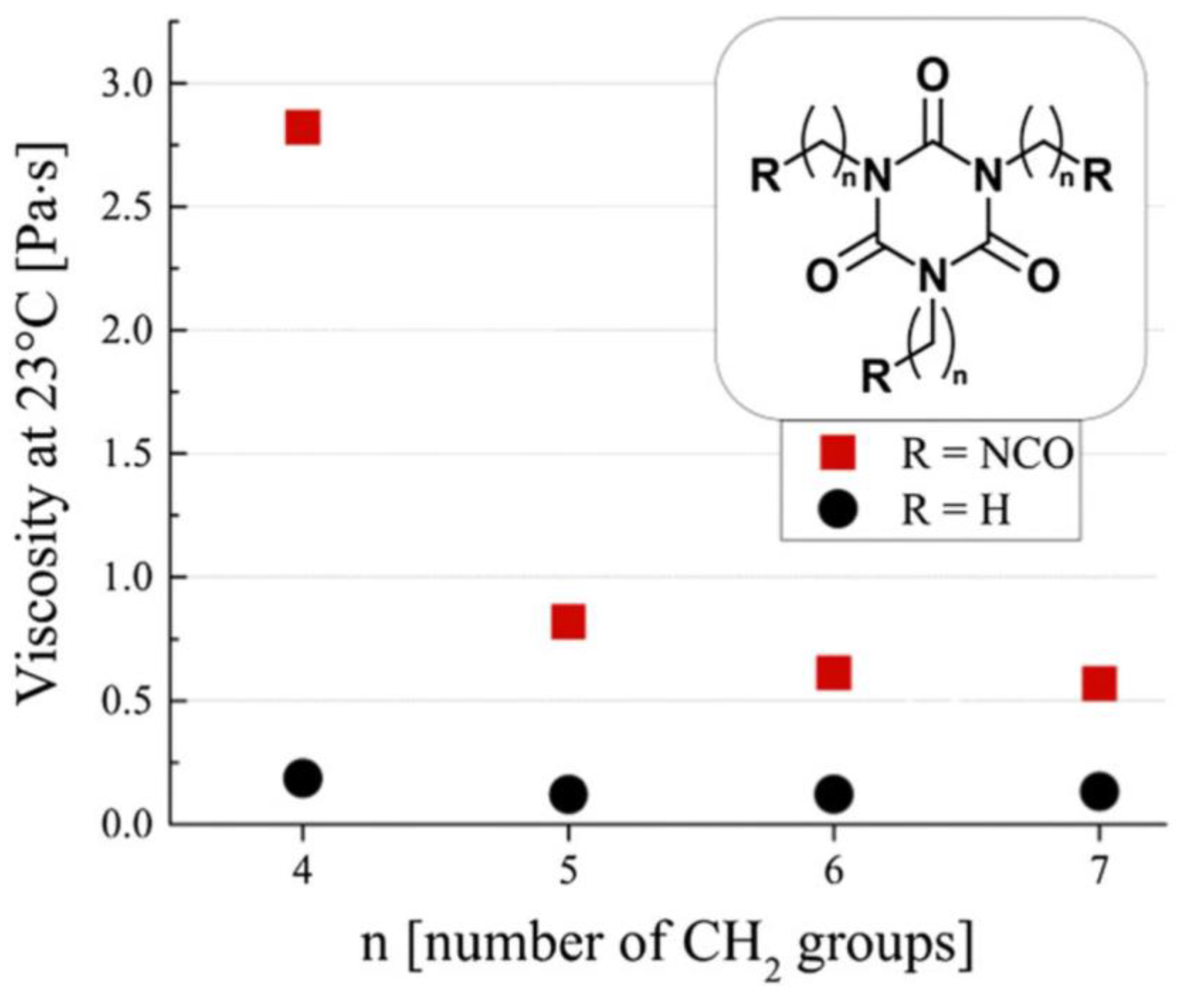
4.2. Molecular Dynamics Force Fields
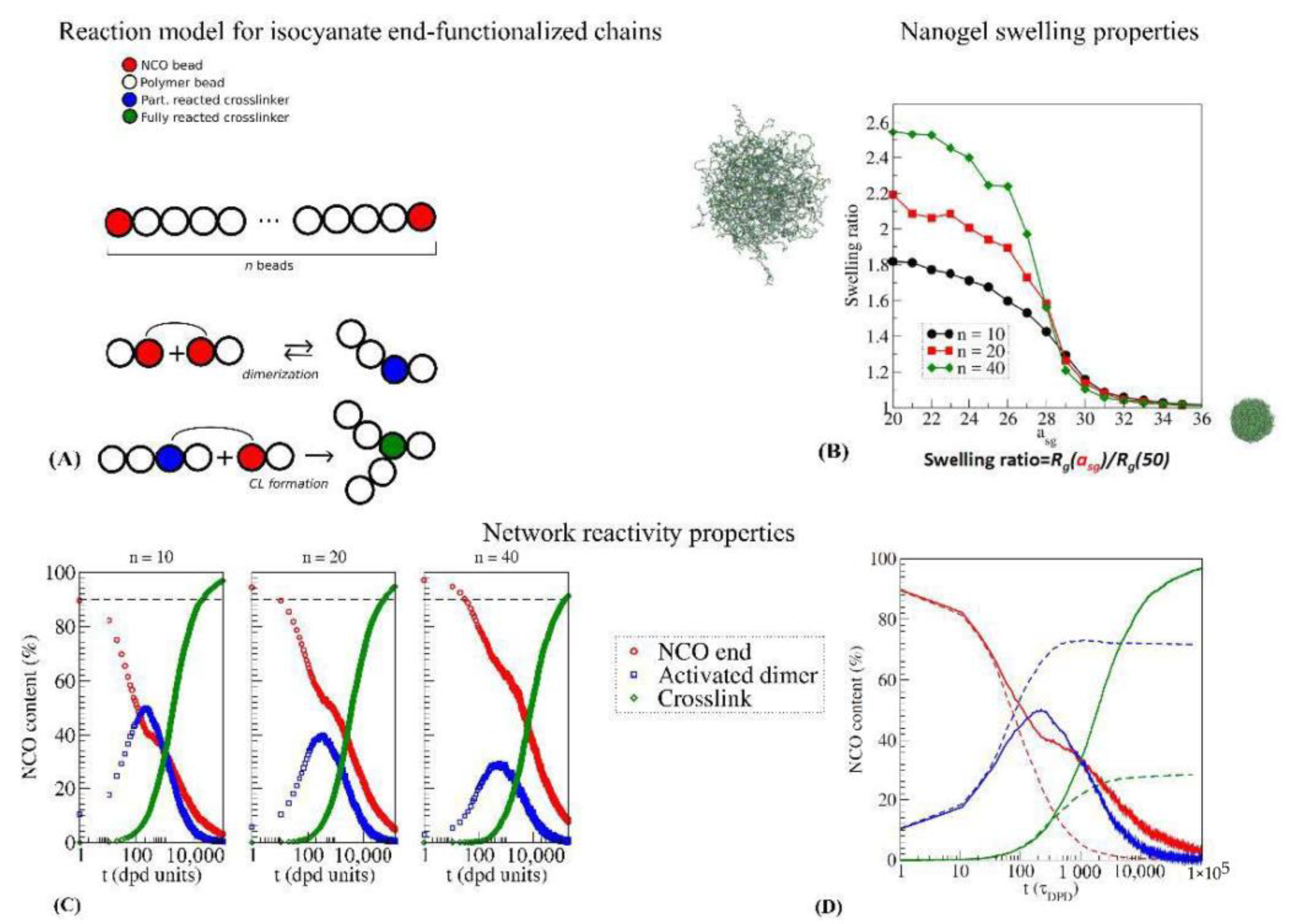
4.3. Coarse-Grained Models
5. Outlook and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Charles, A. Wurtz Recherches Sur Les Éthers Cyaniques et Leurs Dérivés. CR Hebd. Seances. Acad. Sci. 1848, 27, 241–243. [Google Scholar]
- Bayer, O. Das Di-Isocyanat-Polyadditionsverfahren (Polyurethane). Angew. Chem. 1947, 59, 257–272. [Google Scholar] [CrossRef]
- Golling, F.E.; Pires, R.; Hecking, A.; Weikard, J.; Richter, F.; Danielmeier, K.; Dijkstra, D. Polyurethanes for Coatings and Adhesives—Chemistry and Applications. Polym. Int. 2019, 68, 848–855. [Google Scholar] [CrossRef]
- Delebecq, E.; Pascault, J.-P.; Boutevin, B.; Ganachaud, F. On the Versatility of Urethane/Urea Bonds: Reversibility, Blocked Isocyanate, and Non-Isocyanate Polyurethane. Chem. Rev. 2013, 113, 80–118. [Google Scholar] [CrossRef] [PubMed]
- Goesmann, F.; Rosenbauer, H.; Bredehöft, J.H.; Cabane, M.; Ehrenfreund, P.; Gautier, T.; Giri, C.; Krüger, H.; Le Roy, L.; MacDermott, A.J.; et al. Organic Compounds on Comet 67P/Churyumov-Gerasimenko Revealed by COSAC Mass Spectrometry. Science 2015, 349, aab0689. [Google Scholar] [CrossRef]
- Sharmin, E.; Zafar, F. Polyurethane: An Introduction. In Polyurethane; Zafar, F., Ed.; InTech: London, UK, 2012; ISBN 978-953-51-0726-2. [Google Scholar]
- Sonnenschein, M.F. Polyurethanes: Science, Technology, Markets, and Trends, 2nd ed.; Wiley Series on Polymer Engineering and Technology; Wiley: Hoboken, NJ, USA, 2021; ISBN 978-1-119-66941-8. [Google Scholar]
- Polyurethane Market Size, Share & Trends Report, 2021–2028. Available online: https://www.grandviewresearch.com/industry-analysis/polyurethane-pu-market (accessed on 31 March 2022).
- Cooper, S.L.; Guan, J. Advances in Polyurethane Biomaterials; Woodhead Publishing Series in Biomaterials; Woodhead Publishing: Duxford, UK, 2016; ISBN 978-0-08-100614-6. [Google Scholar]
- Konieczny, J.; Loos, K. Polyurethane Coatings Based on Renewable White Dextrins and Isocyanate Trimers. Macromol. Rapid Commun. 2019, 40, 1800874. [Google Scholar] [CrossRef]
- Sahoo, S.; Kalita, H.; Mohanty, S.; Nayak, S.K. Synthesis and Characterization of Vegetable Oil Based Polyurethane Derived from Low Viscous Bio Aliphatic Isocyanate: Adhesion Strength to Wood-Wood Substrate Bonding. Macromol. Res. 2017, 25, 772–778. [Google Scholar] [CrossRef]
- Sharpe, H.R.; Geer, A.M.; Williams, H.E.L.; Blundell, T.J.; Lewis, W.; Blake, A.J.; Kays, D.L. Cyclotrimerisation of Isocyanates Catalysed by Low-Coordinate Mn(ii) and Fe(ii) m-Terphenyl Complexes. Chem. Commun. 2017, 53, 937–940. [Google Scholar] [CrossRef]
- Bahili, M.A.; Stokes, E.C.; Amesbury, R.C.; Ould, D.M.C.; Christo, B.; Horne, R.J.; Kariuki, B.M.; Stewart, J.A.; Taylor, R.L.; Williams, P.A.; et al. Aluminium-Catalysed Isocyanate Trimerization, Enhanced by Exploiting a Dynamic Coordination Sphere. Chem. Commun. 2019, 55, 7679–7682. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, Y.; Yang, J.; Wei, X. Lithium Complex of 2-Amino-Functionalized Benzoylpyrrole: Synthesis, Structure, and Catalytic Activity for the Cyclotrimerization of Isocyanates. Polyhedron 2017, 130, 13–17. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Raju, K.V.S.N. Structural Engineering of Polyurethane Coatings for High Performance Applications. Prog. Polym. Sci. 2007, 32, 352–418. [Google Scholar] [CrossRef]
- Moritsugu, M.; Sudo, A.; Endo, T. Cyclotrimerization of Diisocyanates toward High-Performance Networked Polymers with Rigid Isocyanurate Structure: Combination of Aromatic and Aliphatic Diisocyanates for Tunable Flexibility. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 2631–2637. [Google Scholar] [CrossRef]
- Renz, H.; Bruchmann, B. Pathways Targeting Solvent-Free PUR Coatings. Prog. Org. Coat. 2001, 43, 32–40. [Google Scholar] [CrossRef]
- Preis, E.; Schindler, N.; Adrian, S.; Scherf, U. Microporous Polymer Networks Made by Cyclotrimerization of Commercial, Aromatic Diisocyanates. ACS Macro Lett. 2015, 4, 1268–1272. [Google Scholar] [CrossRef]
- Donthula, S.; Mandal, C.; Leventis, T.; Schisler, J.; Saeed, A.M.; Sotiriou-Leventis, C.; Leventis, N. Shape Memory Superelastic Poly(Isocyanurate-Urethane) Aerogels (PIR-PUR) for Deployable Panels and Biomimetic Applications. Chem. Mater. 2017, 29, 4461–4477. [Google Scholar] [CrossRef]
- Borbone, F.; Carella, A.; Roviello, A.; Casalboni, M.; De Matteis, F.; Stracci, G.; della Rovere, F.; Evangelisti, A.; Dispenza, M. Outstanding Poling Stability of a New Cross-Linked Nonlinear Optical (NLO) Material from a Low Molecular Weight Chromophore. J. Phys. Chem. B 2011, 115, 11993–12000. [Google Scholar] [CrossRef]
- Widemann, M.; Driest, P.J.; Orecchia, P.; Naline, F.; Golling, F.E.; Hecking, A.; Eggert, C.; Pires, R.; Danielmeier, K.; Richter, F.U. Structure–Property Relations in Oligomers of Linear Aliphatic Diisocyanates. ACS Sustain. Chem. Eng. 2018, 6, 9753–9759. [Google Scholar] [CrossRef]
- Driest, P.J.; Lenzi, V.; Marques, L.S.A.; Ramos, M.M.D.; Dijkstra, D.J.; Richter, F.U.; Stamatialis, D.; Grijpma, D.W. Aliphatic Isocyanurates and Polyisocyanurate Networks: Aliphatic Isocyanurates and Polyisocyanurate Networks. Polym. Adv. Technol. 2017, 28, 1299–1304. [Google Scholar] [CrossRef]
- Fernandez-Nieves, A.; Wyss, H.M.; Mattsson, J.; Weitz, D.A. (Eds.) Microgel Suspensions: Fundamentals and Applications, 1st ed.; Wiley: Hoboken, NJ, USA, 2011; ISBN 978-3-527-32158-2. [Google Scholar]
- Grossen, P.; Witzigmann, D.; Sieber, S.; Huwyler, J. PEG-PCL-Based Nanomedicines: A Biodegradable Drug Delivery System and Its Application. J. Control. Release 2017, 260, 46–60. [Google Scholar] [CrossRef]
- Polo Fonseca, L.; Trinca, R.B.; Felisberti, M.I. Amphiphilic Polyurethane Hydrogels as Smart Carriers for Acidic Hydrophobic Drugs. Int. J. Pharm. 2018, 546, 106–114. [Google Scholar] [CrossRef]
- Chen, Y.-P.; Hsu, S. Preparation and Characterization of Novel Water-Based Biodegradable Polyurethane Nanoparticles Encapsulating Superparamagnetic Iron Oxide and Hydrophobic Drugs. J. Mater. Chem. B 2014, 2, 3391–3401. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, Q.; Zhou, Q.; Zhang, P.; Dai, H. Nondegradable Magnetic Poly (Carbonate Urethane) Microspheres with Good Shape Memory as a Proposed Material for Vascular Embolization. J. Mech. Behav. Biomed. Mater. 2018, 82, 9–17. [Google Scholar] [CrossRef]
- Weems, A.C.; Li, W.; Maitland, D.J.; Calle, L.M. Polyurethane Microparticles for Stimuli Response and Reduced Oxidative Degradation in Highly Porous Shape Memory Polymers. ACS Appl. Mater. Interfaces 2018, 10, 32998–33009. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.; Hung, K.-C.; Lin, Y.-Y.; Su, C.-H.; Yeh, H.-Y.; Jeng, U.-S.; Lu, C.-Y.; Dai, S.A.; Fu, W.-E.; Lin, J.-C. Water-Based Synthesis and Processing of Novel Biodegradable Elastomers for Medical Applications. J. Mater. Chem. B 2014, 2, 5083–5092. [Google Scholar] [CrossRef]
- Driest, P.J.; Dijkstra, D.J.; Stamatialis, D.; Grijpma, D.W. The Trimerization of Isocyanate-Functionalized Prepolymers: An Effective Method for Synthesizing Well-Defined Polymer Networks. Macromol. Rapid Commun. 2019, 40, 1800867. [Google Scholar] [CrossRef]
- Driest, P.J.; Dijkstra, D.J.; Stamatialis, D.; Grijpma, D.W. Tough Combinatorial Poly(Urethane-Isocyanurate) Polymer Networks and Hydrogels Synthesized by the Trimerization of Mixtures of NCO-Prepolymers. Acta Biomater. 2020, 105, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Driest, P.J.; Allijn, I.E.; Dijkstra, D.J.; Stamatialis, D.; Grijpma, D.W. Poly(Ethylene Glycol)-based Poly(Urethane Isocyanurate) Hydrogels for Contact Lens Applications. Polym. Int. 2020, 69, 131–139. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Self-Cleaning and Anti-Fog Surfaces via Stimuli-Responsive Polymer Brushes. Adv. Mater. 2007, 19, 3838–3843. [Google Scholar] [CrossRef]
- Kuypers, S.; Pramanik, S.K.; D’Olieslaeger, L.; Reekmans, G.; Peters, M.; D’Haen, J.; Vanderzande, D.; Junkers, T.; Adriaensens, P.; Ethirajan, A. Interfacial Thiol–Isocyanate Reactions for Functional Nanocarriers: A Facile Route towards Tunable Morphologies and Hydrophilic Payload Encapsulation. Chem. Commun. 2015, 51, 15858–15861. [Google Scholar] [CrossRef]
- Abushammala, H.; Mao, J. A Review of the Surface Modification of Cellulose and Nanocellulose Using Aliphatic and Aromatic Mono- and Di-Isocyanates. Molecules 2019, 24, 2782. [Google Scholar] [CrossRef]
- Kang, H.-J.; Cha, E.J.; Park, H.-D. Protein Immobilization onto Various Surfaces Using a Polymer-Bound Isocyanate. Appl. Surf. Sci. 2015, 324, 198–204. [Google Scholar] [CrossRef]
- McDonald, A.R.; Dijkstra, H.P.; Suijkerbuijk, B.M.J.M.; van Klink, G.P.M.; van Koten, G. “Click” Immobilization of Organometallic Pincer Catalysts for C−C Coupling Reactions. Organometallics 2009, 28, 4689–4699. [Google Scholar] [CrossRef]
- Stankovich, S.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Synthesis and Exfoliation of Isocyanate-Treated Graphene Oxide Nanoplatelets. Carbon 2006, 44, 3342–3347. [Google Scholar] [CrossRef]
- Kafy, A.; Sadasivuni, K.K.; Kim, H.-C.; Akther, A.; Kim, J. Designing Flexible Energy and Memory Storage Materials Using Cellulose Modified Graphene Oxide Nanocomposites. Phys. Chem. Chem. Phys. 2015, 17, 5923–5931. [Google Scholar] [CrossRef] [PubMed]
- Dekamin, M.G.; Mehdipoor, F.; Yaghoubi, A. 1,3,5-Tris(2-Hydroxyethyl)Isocyanurate Functionalized Graphene Oxide: A Novel and Efficient Nanocatalyst for the One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-Ones. New J. Chem. 2017, 41, 6893–6901. [Google Scholar] [CrossRef]
- Li, J.; Wang, F.; Liu, C. Tri-Isocyanate Reinforced Graphene Aerogel and Its Use for Crude Oil Adsorption. J. Colloid Interface Sci. 2012, 382, 13–16. [Google Scholar] [CrossRef]
- Wu, X.; Man, J.; Liu, S.; Huang, S.; Lu, J.; Tai, J.; Zhong, Y.; Shen, X.; Cui, S.; Chen, X. Isocyanate-Crosslinked Silica Aerogel Monolith with Low Thermal Conductivity and Much Enhanced Mechanical Properties: Fabrication and Analysis of Forming Mechanisms. Ceram. Int. 2021, 47, 26668–26677. [Google Scholar] [CrossRef]
- Huang, M.; Yang, J. Facile Microencapsulation of HDI for Self-Healing Anticorrosion Coatings. J. Mater. Chem. 2011, 21, 11123. [Google Scholar] [CrossRef]
- Sun, D.; Chong, Y.B.; Chen, K.; Yang, J. Chemically and Thermally Stable Isocyanate Microcapsules Having Good Self-Healing and Self-Lubricating Performances. Chem. Eng. J. 2018, 346, 289–297. [Google Scholar] [CrossRef]
- He, Z.; Jiang, S.; Li, Q.; Wang, J.; Zhao, Y.; Kang, M. Facile and Cost-Effective Synthesis of Isocyanate Microcapsules via Polyvinyl Alcohol-Mediated Interfacial Polymerization and Their Application in Self-Healing Materials. Compos. Sci. Technol. 2017, 138, 15–23. [Google Scholar] [CrossRef]
- He, Z.; Jiang, S.; An, N.; Li, X.; Li, Q.; Wang, J.; Zhao, Y.; Kang, M. Self-Healing Isocyanate Microcapsules for Efficient Restoration of Fracture Damage of Polyurethane and Epoxy Resins. J. Mater. Sci 2019, 54, 8262–8275. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Liu, X.; Gu, J. Synthesis of Isocyanate Microcapsules as Functional Crosslinking Agent for Wood Adhesive. J. Adhes. 2021, 97, 38–52. [Google Scholar] [CrossRef]
- Gody, G.; Rossner, C.; Moraes, J.; Vana, P.; Maschmeyer, T.; Perrier, S. One-Pot RAFT/“Click” Chemistry via Isocyanates: Efficient Synthesis of α-End-Functionalized Polymers. J. Am. Chem. Soc. 2012, 134, 12596–12603. [Google Scholar] [CrossRef] [PubMed]
- Gody, G.; Roberts, D.A.; Maschmeyer, T.; Perrier, S. A New Methodology for Assessing Macromolecular Click Reactions and Its Application to Amine–Tertiary Isocyanate Coupling for Polymer Ligation. J. Am. Chem. Soc. 2016, 138, 4061–4068. [Google Scholar] [CrossRef] [PubMed]
- Sundberg, P.; Karppinen, M. Organic and Inorganic–Organic Thin Film Structures by Molecular Layer Deposition: A Review. Beilstein J. Nanotechnol. 2014, 5, 1104–1136. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Huynh, Q.-U.; Uhlmann, E.; Wagner, M.H. Study of Inkjet Printing as Additive Manufacturing Process for Gradient Polyurethane Material. Prod. Eng. 2014, 8, 25–32. [Google Scholar] [CrossRef]
- Behnken, G.; Hecking, A.; Sánchez, B.V. Sánchez High Performance Enabled by Nature. Eur. Coat. J. 2016, 1, 46–50. [Google Scholar]
- Morales-Cerrada, R.; Tavernier, R.; Caillol, S. Fully Bio-Based Thermosetting Polyurethanes from Bio-Based Polyols and Isocyanates. Polymers 2021, 13, 1255. [Google Scholar] [CrossRef]
- Tai, N.L.; Ghasemlou, M.; Adhikari, R.; Adhikari, B. Starch-Based Isocyanate- and Non-Isocyanate Polyurethane Hybrids: A Review on Synthesis, Performance and Biodegradation. Carbohydr. Polym. 2021, 265, 118029. [Google Scholar] [CrossRef]
- Rolph, M.S.; Inam, M.; O’Reilly, R.K. The Application of Blocked Isocyanate Chemistry in the Development of Tunable Thermoresponsive Crosslinkers. Polym. Chem. 2017, 8, 7229–7239. [Google Scholar] [CrossRef]
- Guo, Z.; Xu, Y.; Wu, X.; Wei, X.; Xi, C. Potassium Complexes Containing Bidentate Pyrrole Ligands: Synthesis, Structures, and Catalytic Activity for the Cyclotrimerization of Isocyanates. Dalton Trans. 2019, 48, 8116–8121. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, W.; He, J.; Zhang, Y. Highly Efficient Cyclotrimerization of Isocyanates Using N-Heterocyclic Olefins under Bulk Conditions. Chem. Commun. 2019, 55, 12563–12566. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Muuronen, M.; Deglmann, P.; Lucas, F.; Sijbesma, R.P.; Tomović, Ž. Role of Acetate Anions in the Catalytic Formation of Isocyanurates from Aromatic Isocyanates. J. Org. Chem. 2021, 86, 5651–5659. [Google Scholar] [CrossRef] [PubMed]
- Methyl Isocyanate (CH3NCO): An Important Missing Organic in Current Astrochemical Networks|Monthly Notices of the Royal Astronomical Society: Letters|Oxford Academic. Available online: https://academic.oup.com/mnrasl/article/473/1/L59/4265278 (accessed on 31 March 2022).
- Rodríguez-Almeida, L.F.; Rivilla, V.M.; Jiménez-Serra, I.; Melosso, M.; Colzi, L.; Zeng, S.; Tercero, B.; de Vicente, P.; Martín, S.; Requena-Torres, M.A.; et al. First Detection of C2H5NCO in the ISM and Search of Other Isocyanates towards the G+0.693-0.027 Molecular Cloud. Astron. Astrophys. 2021, 654, L1. [Google Scholar] [CrossRef]
- Maté, B.; Molpeceres, G.; Tanarro, I.; Peláez, R.J.; Guillemin, J.C.; Cernicharo, J.; Herrero, V.J. Stability of CH$\less$sub$\greater$3$\less$/Sub$\greater$NCO in Astronomical Ices under Energetic Processing: A Laboratory Study. Astrophys. J. 2018, 861, 61. [Google Scholar] [CrossRef]
- Ciaravella, A.; Jiménez-Escobar, A.; Cecchi-Pestellini, C.; Huang, C.H.; Sie, N.E.; Caro, G.M.M.; Chen, Y.J. Synthesis of Complex Organic Molecules in Soft X-Ray Irradiated Ices. Astrophys. J. 2019, 879, 21. [Google Scholar] [CrossRef]
- Schneider, C.; Becker, S.; Okamura, H.; Crisp, A.; Amatov, T.; Stadlmeier, M.; Carell, T. Noncanonical RNA Nucleosides as Molecular Fossils of an Early Earth—Generation by Prebiotic Methylations and Carbamoylations. Angew. Chem. Int. Ed. 2018, 57, 5943–5946. [Google Scholar] [CrossRef]
- Cassone, G.; Saija, F. Interstellar Chemical Reactions toward the Synthesis of the Life’s Building Blocks: Comment on “Insoluble Organic Matter in Chondrites: Archetypal Melanin-like PAH-Based Multifunctionality at the Origin of Life?” By M. d’Ischia et al. Phys. Life Rev. 2021, 38, 140–142. [Google Scholar] [CrossRef]
- Ozensoy, E.; Goodman, D.W. Vibrational Spectroscopic Studies on CO Adsorption, NO Adsorption CO + NO Reaction on Pd Model Catalysts. Phys. Chem. Chem. Phys. 2004, 6, 3765–3778. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864–B871. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Self-Consistent Equations Including Exchange and Correlation Effects. Phys. Rev. 1965, 140, A1133–A1138. [Google Scholar] [CrossRef]
- Zhao, S.; Ren, Y.; Wang, J.; Yin, W. A Density Functional Study of the Interaction of NCO with Small Copper Clusters. J. Phys. Chem. A 2009, 113, 1075–1085. [Google Scholar] [CrossRef] [PubMed]
- Kuang, X.-J.; Wang, X.-Q.; Liu, G.-B. All-Electron Scalar Relativistic Calculation of the Adsorption of NCO Species onto Small Copper Clusters. J. Iran. Chem. Soc. 2011, 8, 750–761. [Google Scholar] [CrossRef]
- Garda, G.R.; Ferullo, R.M.; Castellani, N.J. Chemisorption of NCO on Cu(100): A Density Functional Theory Study. Surf. Sci. 2005, 598, 57–67. [Google Scholar] [CrossRef]
- Zhao, S.; Ren, Y.; Wang, J.; Yin, W. Interaction of NCO with Small Silver Clusters: A Density Functional Study. J. Mol. Struct. THEOCHEM 2009, 897, 100–105. [Google Scholar] [CrossRef]
- Belelli, P.G.; Branda, M.M.; Garda, G.R.; Ferullo, R.M.; Castellani, N.J. Chemisorption of Isocyanate (NCO) on the Pd(100) Surface at Different Coverages. Surf. Sci. 2010, 604, 442–450. [Google Scholar] [CrossRef]
- Deng, H.; Yu, Y.; He, H. The Role of AgOAl Entities in Adsorption of NCO Species and Reduction of NOx. Catal. Today 2015, 258, 35–40. [Google Scholar] [CrossRef]
- Cheuquepán, W.; Orts, J.M.; Rodes, A.; Feliu, J.M. DFT and Spectroelectrochemical Study of Cyanate Adsorption on Gold Single Crystal Electrodes in Neutral Medium. J. Electroanal. Chem. 2017, 793, 147–156. [Google Scholar] [CrossRef]
- Czekaj, I.; Piazzesi, G.; Kröcher, O.; Wokaun, A. DFT Modeling of the Hydrolysis of Isocyanic Acid over the TiO2 Anatase (101) Surface: Adsorption of HNCO Species. Surf. Sci. 2006, 600, 5158–5167. [Google Scholar] [CrossRef]
- Czekaj, I.; Kröcher, O. Decomposition of Urea in the SCR Process: Combination of DFT Calculations and Experimental Results on the Catalytic Hydrolysis of Isocyanic Acid on TiO2 and Al2O3. Top Catal 2009, 52, 1740. [Google Scholar] [CrossRef]
- Evers, J.; Krumm, B.; Axthammer, Q.J.; Martens, J.; Blaha, P.; Steemann, F.X.; Reith, T.; Mayer, P.; Klapötke, T.M. Molecular Structure of Isocyanic Acid, HNCO, the Imide of Carbon Dioxide. J. Phys. Chem. A 2018, 122, 3287–3292. [Google Scholar] [CrossRef] [PubMed]
- Lowenthal, M.S.; Khanna, R.K.; Moore, M.H. Infrared Spectrum of Solid Isocyanic Acid (HNCO): Vibrational Assignments and Integrated Band Intensities. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2002, 58, 73–78. [Google Scholar] [CrossRef]
- Raunier, S.; Chiavassa, T.; Allouche, A.; Marinelli, F.; Aycard, J.-P. Thermal Reactivity of HNCO with Water Ice: An Infrared and Theoretical Study. Chem. Phys. 2003, 288, 197–210. [Google Scholar] [CrossRef]
- Zabardasti, A.; Solimannejad, M. Theoretical Study and AIM Analysis of Hydrogen Bonded Clusters of Water and Isocyanic Acid. J. Mol. Struct. THEOCHEM 2007, 819, 52–59. [Google Scholar] [CrossRef]
- Cassone, G.; Saija, F.; Sponer, J.; Sponer, J.E.; Jiménez-Escobar, A.; Ciaravella, A.; Cecchi-Pestellini, C. Atomistic Simulations of the Free-Energy Landscapes of Interstellar Chemical Reactions: The Case of Methyl Isocyanate. Mon. Not. R. Astron. Soc. 2021, 504, 1565–1570. [Google Scholar] [CrossRef]
- Okumoto, S.; Yamabe, S. A Computational Study of Base-Catalyzed Reactions between Isocyanates and Epoxides Affording 2-Oxazolidones and Isocyanurates. J. Comput. Chem. 2001, 22, 316–326. [Google Scholar] [CrossRef]
- Gibb, J.N.; Goodman, J.M. The Formation of High-Purity Isocyanurate through Proazaphosphatrane-Catalysed Isocyanate Cyclo-Trimerisation: Computational Insights. Org. Biomol. Chem. 2012, 11, 90–97. [Google Scholar] [CrossRef]
- Helberg, J.; Oe, Y.; Zipse, H. Mechanistic Analysis and Characterization of Intermediates in the Phosphane-Catalyzed Oligomerization of Isocyanates. Chem.–A Eur. J. 2018, 24, 14387–14391. [Google Scholar] [CrossRef]
- Han, Y.-S.; Jung, K.-Y.; Kim, Y.-J.; Baeck, K.K.; Lee, G.M.; Lee, S.W. Reactivities of Zero-Valent Group 10 Complexes toward Organic Isocyanates: Synthesis of Metallacycles Containing Dimeric Isocyanate Units, Isocyanate Cyclotrimerization, and Computational Chemistry. New J. Chem. 2019, 43, 15614–15625. [Google Scholar] [CrossRef]
- Cheikh, W.; Rózsa, Z.B.; Camacho López, C.O.; Mizsey, P.; Viskolcz, B.; Szőri, M.; Fejes, Z. Urethane Formation with an Excess of Isocyanate or Alcohol: Experimental and ab initio Study. Polymers 2019, 11, 1543. [Google Scholar] [CrossRef]
- Kim, J.; Cho, J.; Lim, Y.-S. Bonding of Urethane Reactants to Aluminum Surface. J Mater Sci 2005, 40, 2789–2794. [Google Scholar] [CrossRef]
- Tardio, S.; Abel, M.-L.; Carr, R.H.; Watts, J.F. The Interfacial Interaction between Isocyanate and Stainless Steel. Int. J. Adhes. Adhes. 2019, 88, 1–10. [Google Scholar] [CrossRef]
- Lenzi, V.; Driest, P.J.; Dijkstra, D.J.; Ramos, M.M.D.; Marques, L.S.A. Investigation on the Intermolecular Interactions in Aliphatic Isocyanurate Liquids: Revealing the Importance of Dispersion. J. Mol. Liq. 2019, 280, 25–33. [Google Scholar] [CrossRef]
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A.J.; Yang, W. Revealing Noncovalent Interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [Google Scholar] [CrossRef] [PubMed]
- Boros, R.Z.; Rágyanszki, A.; Csizmadia, I.G.; Fiser, B.; Guljas, A.; Farkas, L.; Viskolcz, B. Industrial Application of Molecular Computations on the Dimerization of Methylene Diphenyl Diisocyanate. React. Kinet. Mech. Catal. 2018, 124, 1–14. [Google Scholar] [CrossRef]
- Petelski, A.N.; Peruchena, N.M.; Pamies, S.C.; Sosa, G.L. Insights into the Self-Assembly Steps of Cyanuric Acid toward Rosette Motifs: A DFT Study. J. Mol. Model. 2017, 23, 263. [Google Scholar] [CrossRef] [PubMed]
- Petelski, A.N.; Pamies, S.C.; Sejas, A.G.; Peruchena, N.M.; Sosa, G.L. Impact of Confinement in Multimolecular Inclusion Compounds of Melamine and Cyanuric Acid. Phys. Chem. Chem. Phys. 2019, 21, 8205–8214. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Maxwell, D.S.; Tirado-Rives, J. Development and Testing of the OPLS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 1996, 118, 11225–11236. [Google Scholar] [CrossRef]
- Vanommeslaeghe, K.; Hatcher, E.; Acharya, C.; Kundu, S.; Zhong, S.; Shim, J.; Darian, E.; Guvench, O.; Lopes, P.; Vorobyov, I.; et al. CHARMM General Force Field: A Force Field for Drug-like Molecules Compatible with the CHARMM All-Atom Additive Biological Force Fields. J. Comput. Chem. 2010, 31, 671–690. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and Testing of a General Amber Force Field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- Cornell, W.D.; Cieplak, P.; Bayly, C.I.; Gould, I.R.; Merz, K.M.; Ferguson, D.M.; Spellmeyer, D.C.; Fox, T.; Caldwell, J.W.; Kollman, P.A. A Second Generation Force Field for the Simulation of Proteins, Nucleic Acids, and Organic Molecules. J. Am. Chem. Soc. 1995, 117, 5179–5197. [Google Scholar] [CrossRef]
- Lenzi, V.; Driest, P.J.; Dijkstra, D.J.; Ramos, M.M.D.; Marques, L.S.A. GAFF-IC: Realistic Viscosities for Isocyanate Molecules with a GAFF-Based Force Field. Mol. Simul. 2019, 45, 207–214. [Google Scholar] [CrossRef]
- Lenzi, V. Multiscale Modelling and Simulation of Soft Matter Systems. PhD Thesis, University of Minho, Braga, Portugal, 2020. Completed on 19/05/2020. [Google Scholar]
- Emelianova, A.; Gor, G.Y. Molecular Simulations of Vapor–Liquid Equilibrium of Isocyanates. J. Phys. Chem. B 2021, 125, 12528–12538. [Google Scholar] [CrossRef] [PubMed]
- Voth, G.A. (Ed.) Coarse-Graining of Condensed Phase and Biomolecular Systems; Taylor & Francis: London, UK, 2009; ISBN 978-1-4200-5955-7. [Google Scholar]
- Brini, E.; Algaer, E.A.; Ganguly, P.; Li, C.; Rodríguez-Ropero, F.; van der Vegt, N.F. Systematic Coarse-Graining Methods for Soft Matter Simulations—A Review. Soft Matter 2013, 9, 2108–2119. [Google Scholar] [CrossRef]
- Yang, L.; Tan, C.; Hsieh, M.-J.; Wang, J.; Duan, Y.; Cieplak, P.; Caldwell, J.; Kollman, P.A.; Luo, R. New-Generation Amber United-Atom Force Field. J. Phys. Chem. B 2006, 110, 13166–13176. [Google Scholar] [CrossRef]
- Marrink, S.J.; Risselada, H.J.; Yefimov, S.; Tieleman, D.P.; de Vries, A.H. The MARTINI Force Field: Coarse Grained Model for Biomolecular Simulations. J. Phys. Chem. B 2007, 111, 7812–7824. [Google Scholar] [CrossRef]
- Davtyan, A.; Schafer, N.P.; Zheng, W.; Clementi, C.; Wolynes, P.G.; Papoian, G.A. AWSEM-MD: Protein Structure Prediction Using Coarse-Grained Physical Potentials and Bioinformatically Based Local Structure Biasing. J. Phys. Chem. B 2012, 116, 8494–8503. [Google Scholar] [CrossRef]
- Adema, K.N.; Makki, H.; Peters, E.A.; Laven, J.; van der Ven, L.G.; van Benthem, R.A. Kinetic Monte Carlo Simulation of the Photodegradation Process of Polyester-Urethane Coatings. Phys. Chem. Chem. Phys. 2015, 17, 19962–19976. [Google Scholar] [CrossRef]
- Ghermezcheshme, H.; Makki, H.; Mohseni, M.; Ebrahimi, M. MARTINI-Based Simulation Method for Step-Growth Polymerization and Its Analysis by Size Exclusion Characterization: A Case Study of Cross-Linked Polyurethane. Phys. Chem. Chem. Phys. 2019, 21, 21603–21614. [Google Scholar] [CrossRef]
- Ghermezcheshme, H.; Makki, H.; Mohseni, M.; Ebrahimi, M. Hydrophilic Dangling Chain Interfacial Segregation in Polyurethane Networks at Aqueous Interfaces and Its Underlying Mechanisms: Molecular Dynamics Simulations. Phys. Chem. Chem. Phys. 2020, 22, 26351–26363. [Google Scholar] [CrossRef]
- Vakili, H.; Mohseni, M.; Makki, H.; Yahyaei, H.; Ghanbari, H.; González, A.; Irusta, L. Self-Assembly of a Patterned Hydrophobic-Hydrophilic Surface by Soft Segment Microphase Separation in a Segmented Polyurethane: Combined Experimental Study and Molecular Dynamics Simulation. Polymer 2020, 195, 122424. [Google Scholar] [CrossRef]
- Groot, R.D.; Warren, P.B. Dissipative Particle Dynamics: Bridging the Gap between Atomistic and Mesoscopic Simulation. J. Chem. Phys. 1997, 107, 4423–4435. [Google Scholar] [CrossRef]
- Español, P.; Warren, P.B. Perspective: Dissipative Particle Dynamics. J. Chem. Phys. 2017, 146, 150901. [Google Scholar] [CrossRef]
- Hoogerbrugge, P.J.; Koelman, J.M.V.A. Simulating Microscopic Hydrodynamic Phenomena with Dissipative Particle Dynamics. EPL (Europhys. Lett.) 1992, 19, 155–160. [Google Scholar] [CrossRef]
- Huggins, M.L. The Solubility of Nonelectrolytes. By Joel H. Hildebrand and Robert S. Scott. J. Phys. Chem. 1951, 55, 619–620. [Google Scholar] [CrossRef]
- Mayoral, E.; Goicochea, A.G. Modeling the Temperature Dependent Interfacial Tension between Organic Solvents and Water Using Dissipative Particle Dynamics. J. Chem. Phys. 2013, 138, 094703. [Google Scholar] [CrossRef] [PubMed]
- Esteves, A.C.C.; Lyakhova, K.; van der Ven, L.G.J.; van Benthem, R.A.T.M.; de With, G. Surface Segregation of Low Surface Energy Polymeric Dangling Chains in a Cross-Linked Polymer Network Investigated by a Combined Experimental–Simulation Approach. Macromolecules 2013, 46, 1993–2002. [Google Scholar] [CrossRef]
- Esteves, A.C.C.; Lyakhova, K.; van Riel, J.M.; van der Ven, L.G.J.; van Benthem, R.a.T.M.; de With, G. Self-Replenishing Ability of Cross-Linked Low Surface Energy Polymer Films Investigated by a Complementary Experimental-Simulation Approach. J. Chem. Phys. 2014, 140, 124902. [Google Scholar] [CrossRef]
- Lyakhova, K.; Esteves, A.C.C.; van de Put, M.W.P.; van der Ven, L.G.J.; van Benthem, R.A.T.M.; de With, G. Simulation-Experimental Approach to Investigate the Role of Interfaces in Self-Replenishing Composite Coatings. Adv. Mater. Interfaces 2014, 1, 1400053. [Google Scholar] [CrossRef]
- Iype, E.; Esteves, A.C.C.; With, G. de Mesoscopic Simulations of Hydrophilic Cross-Linked Polycarbonate Polyurethane Networks: Structure and Morphology. Soft Matter 2016, 12, 5029–5040. [Google Scholar] [CrossRef]
- Kacar, G. Dissipative particle dynamics simulation parameters and interactions of a hydrogel. J. Turk. Chem. Soc. Sect. A Chem. 2017, 5, 19–28. [Google Scholar] [CrossRef][Green Version]
- Kacar, G.; Albers, P.T.M.; Esteves, A.C.C.; de With, G. Mesoscopic Structure and Swelling Properties of Crosslinked Polyethylene Glycol in Water. J. Coat. Technol. Res. 2018, 15, 691–701. [Google Scholar] [CrossRef]
- Kacar, G.; Peters, E.A.J.F.; With, G. de Mesoscopic Simulations for the Molecular and Network Structure of a Thermoset Polymer. Soft Matter 2013, 9, 5785–5793. [Google Scholar] [CrossRef]
- Lenzi, V.; Ramos, M.M.D.; Marques, L.S.A. Dissipative Particle Dynamics Simulations of End-Cross-Linked Nanogels. Mol. Simul. 2021, 47, 27–36. [Google Scholar] [CrossRef]
- Masubuchi, Y.; Langeloth, M.; Böhm, M.C.; Inoue, T.; Müller-Plathe, F. A Multichain Slip-Spring Dissipative Particle Dynamics Simulation Method for Entangled Polymer Solutions. Macromolecules 2016, 49, 9186–9191. [Google Scholar] [CrossRef]
- van Duin, A.C.T.; Dasgupta, S.; Lorant, F.; Goddard, W.A. ReaxFF: A Reactive Force Field for Hydrocarbons. J. Phys. Chem. A 2001, 105, 9396–9409. [Google Scholar] [CrossRef]
- Senftle, T.P.; Hong, S.; Islam, M.M.; Kylasa, S.B.; Zheng, Y.; Shin, Y.K.; Junkermeier, C.; Engel-Herbert, R.; Janik, M.J.; Aktulga, H.M.; et al. The ReaxFF Reactive Force-Field: Development, Applications and Future Directions. npj Comput. Mater. 2016, 2, 15011. [Google Scholar] [CrossRef]
- Sengul, M.Y.; Song, Y.; Nayir, N.; Gao, Y.; Hung, Y.; Dasgupta, T.; van Duin, A.C.T. INDEEDopt: A Deep Learning-Based ReaxFF Parameterization Framework. npj Comput. Mater. 2021, 7, 68. [Google Scholar] [CrossRef]
- Unke, O.T.; Chmiela, S.; Sauceda, H.E.; Gastegger, M.; Poltavsky, I.; Schütt, K.T.; Tkatchenko, A.; Müller, K.-R. Machine Learning Force Fields. Chem. Rev. 2021, 121, 10142–10186. [Google Scholar] [CrossRef]
- Vandermause, J.; Torrisi, S.B.; Batzner, S.; Xie, Y.; Sun, L.; Kolpak, A.M.; Kozinsky, B. On-the-Fly Active Learning of Interpretable Bayesian Force Fields for Atomistic Rare Events. npj Comput. Mater. 2020, 6, 20. [Google Scholar] [CrossRef]
- Hajibabaei, A.; Ha, M.; Pourasad, S.; Kim, J.; Kim, K.S. Machine Learning of First-Principles Force-Fields for Alkane and Polyene Hydrocarbons. J. Phys. Chem. A 2021, 125, 9414–9420. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lu, L.; Noid, W.G.; Krishna, V.; Pfaendtner, J.; Voth, G.A. A Systematic Methodology for Defining Coarse-Grained Sites in Large Biomolecules. Biophys. J. 2008, 95, 5073–5083. [Google Scholar] [CrossRef] [PubMed]
- Potter, T.D.; Barrett, E.L.; Miller, M.A. Automated Coarse-Grained Mapping Algorithm for the Martini Force Field and Benchmarks for Membrane–Water Partitioning. J. Chem. Theory Comput. 2021, 17, 5777–5791. [Google Scholar] [CrossRef] [PubMed]
- Fraaije, J.G.E.M.; van Male, J.; Becherer, P.; Serral Gracià, R. Coarse-Grained Models for Automated Fragmentation and Parametrization of Molecular Databases. J. Chem. Inf. Model. 2016, 56, 2361–2377. [Google Scholar] [CrossRef] [PubMed]
- Fraaije, J.G.E.M.; van Male, J.; Becherer, P.; Serral Gracià, R. Calculation of Diffusion Coefficients through Coarse-Grained Simulations Using the Automated-Fragmentation-Parametrization Method and the Recovery of Wilke–Chang Statistical Correlation. J. Chem. Theory Comput. 2018, 14, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.T.; Bai, H.Y.; Li, M.Z.; Wang, W.H. Machine Learning Approach for Prediction and Understanding of Glass-Forming Ability. J. Phys. Chem. Lett. 2017, 8, 3434–3439. [Google Scholar] [CrossRef] [PubMed]
- Pugar, J.A.; Childs, C.M.; Huang, C.; Haider, K.W.; Washburn, N.R. Elucidating the Physicochemical Basis of the Glass Transition Temperature in Linear Polyurethane Elastomers with Machine Learning. J. Phys. Chem. B 2020, 124, 9722–9733. [Google Scholar] [CrossRef]
- Menon, A.; Thompson-Colón, J.A.; Washburn, N.R. Hierarchical Machine Learning Model for Mechanical Property Predictions of Polyurethane Elastomers From Small Datasets. Front. Mater. 2019, 6, 87. [Google Scholar] [CrossRef]
- Pugar, J.A.; Gang, C.; Huang, C.; Haider, K.W.; Washburn, N.R. Predicting Young’s Modulus of Linear Polyurethane and Polyurethane–Polyurea Elastomers: Bridging Length Scales with Physicochemical Modeling and Machine Learning. ACS Appl. Mater. Interfaces 2022, 14, 16568–16581. [Google Scholar] [CrossRef]



| Method | Applications | Advantages | Disadvantages |
|---|---|---|---|
| Ab initio |
|
|
|
| Molecular dynamics |
|
|
|
| Coarse-Grained Molecular Dynamics |
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenzi, V.; Crema, A.; Pyrlin, S.; Marques, L. Current State and Perspectives of Simulation and Modeling of Aliphatic Isocyanates and Polyisocyanates. Polymers 2022, 14, 1642. https://doi.org/10.3390/polym14091642
Lenzi V, Crema A, Pyrlin S, Marques L. Current State and Perspectives of Simulation and Modeling of Aliphatic Isocyanates and Polyisocyanates. Polymers. 2022; 14(9):1642. https://doi.org/10.3390/polym14091642
Chicago/Turabian StyleLenzi, Veniero, Anna Crema, Sergey Pyrlin, and Luís Marques. 2022. "Current State and Perspectives of Simulation and Modeling of Aliphatic Isocyanates and Polyisocyanates" Polymers 14, no. 9: 1642. https://doi.org/10.3390/polym14091642
APA StyleLenzi, V., Crema, A., Pyrlin, S., & Marques, L. (2022). Current State and Perspectives of Simulation and Modeling of Aliphatic Isocyanates and Polyisocyanates. Polymers, 14(9), 1642. https://doi.org/10.3390/polym14091642






