Amino-Li-Resin—A Fiber Polyacrylamide Resin for Solid-Phase Peptide Synthesis
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Swelling Capacity
2.3. Preliminary Washing
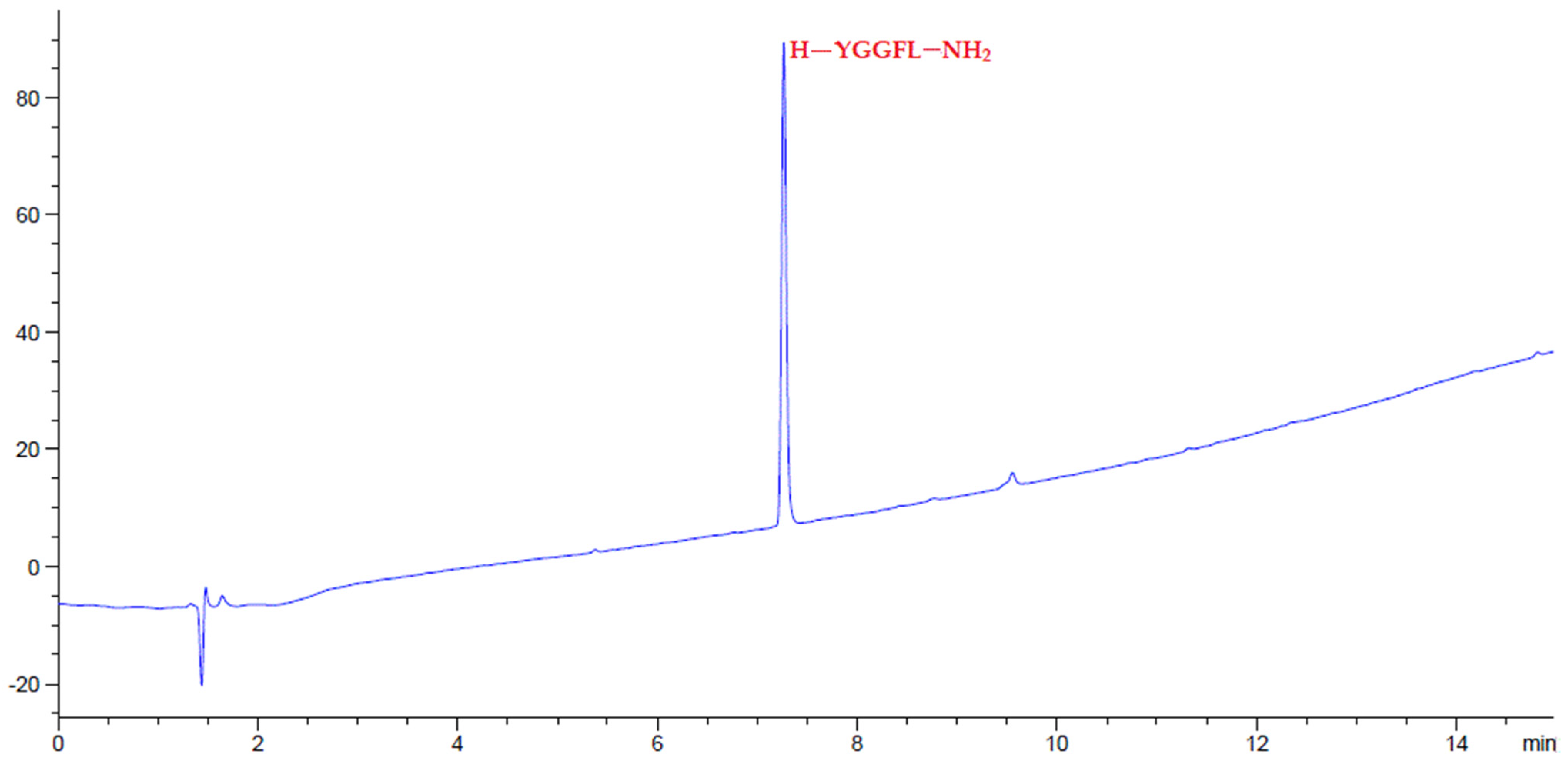
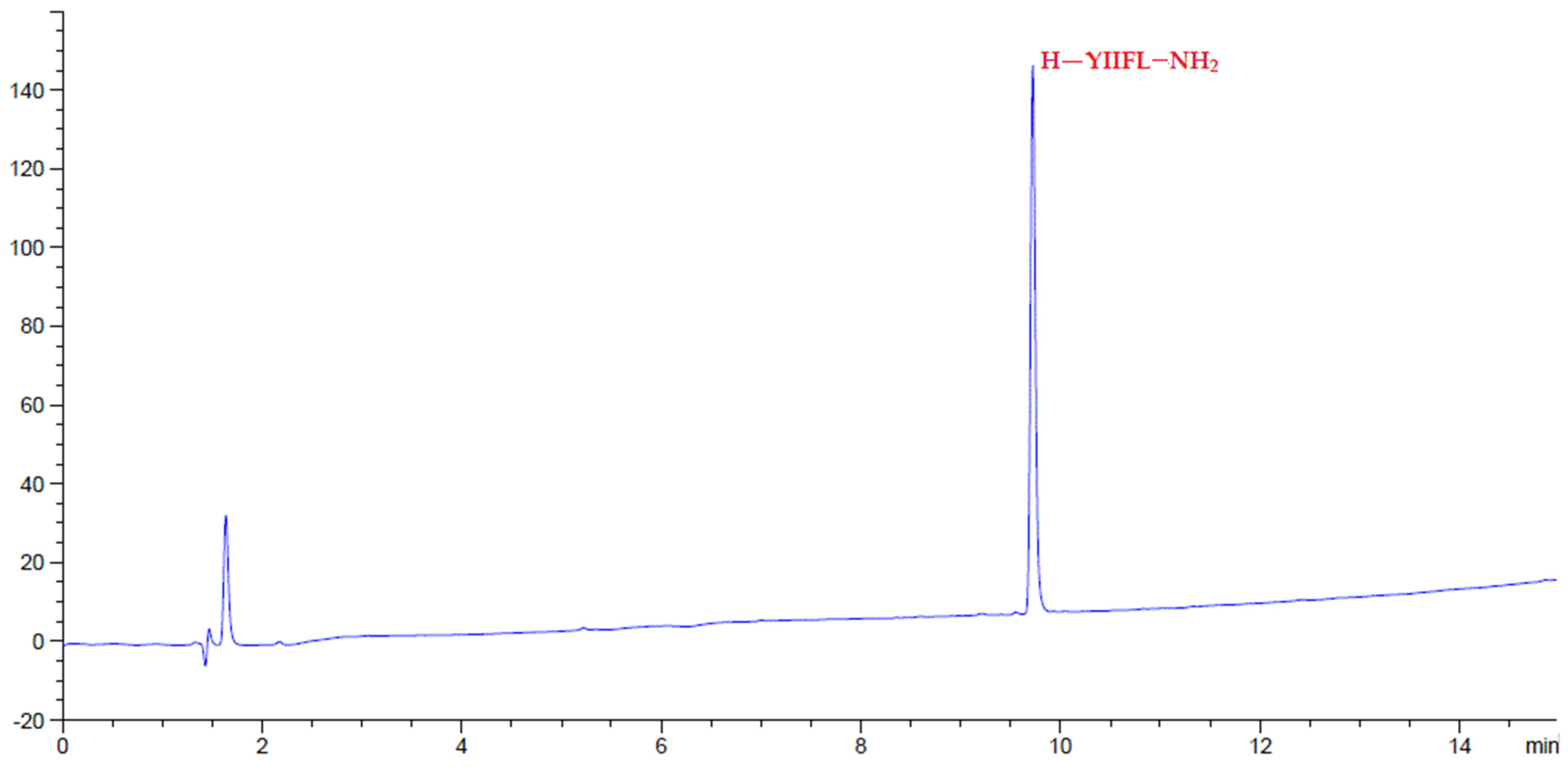
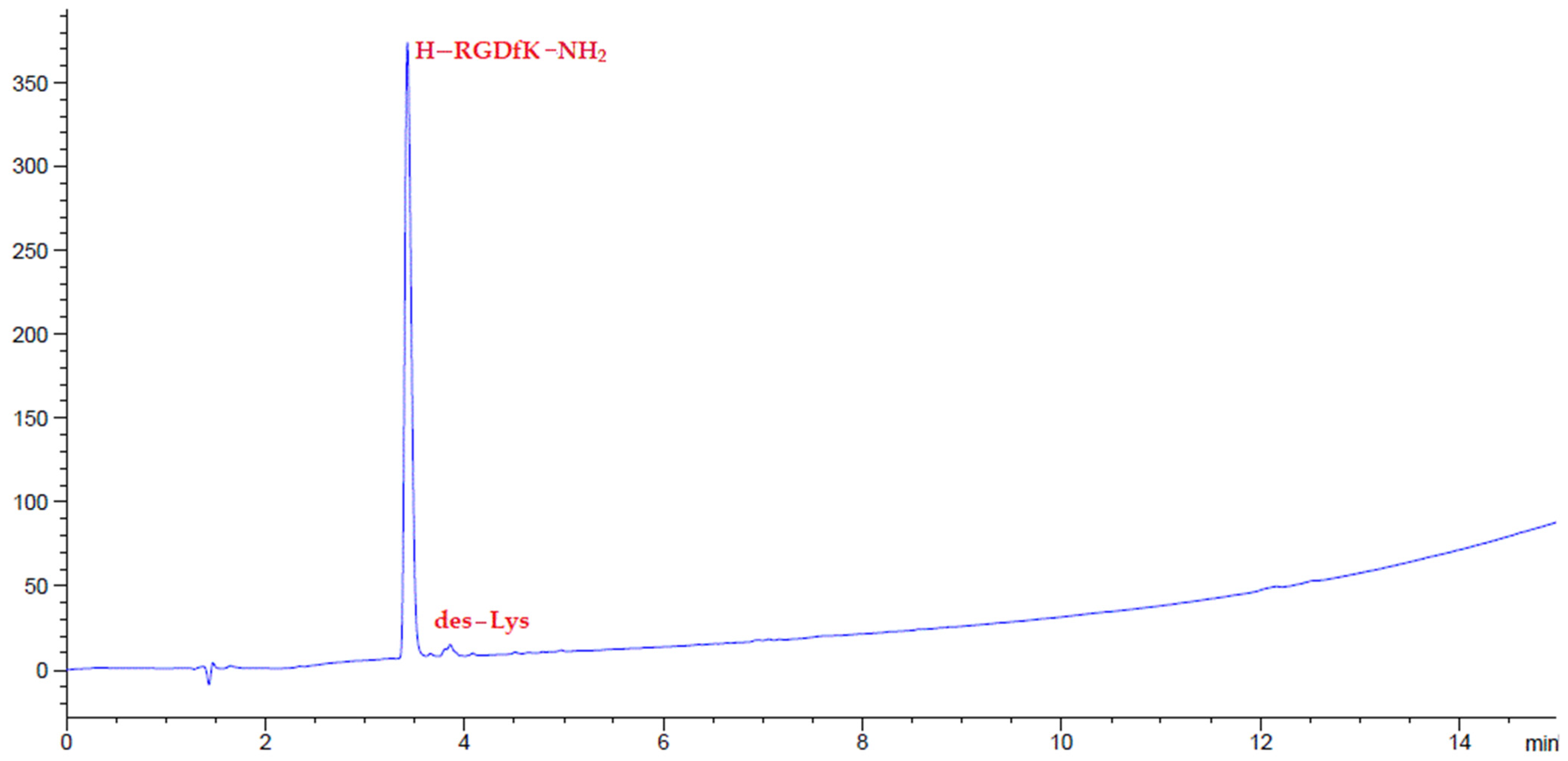
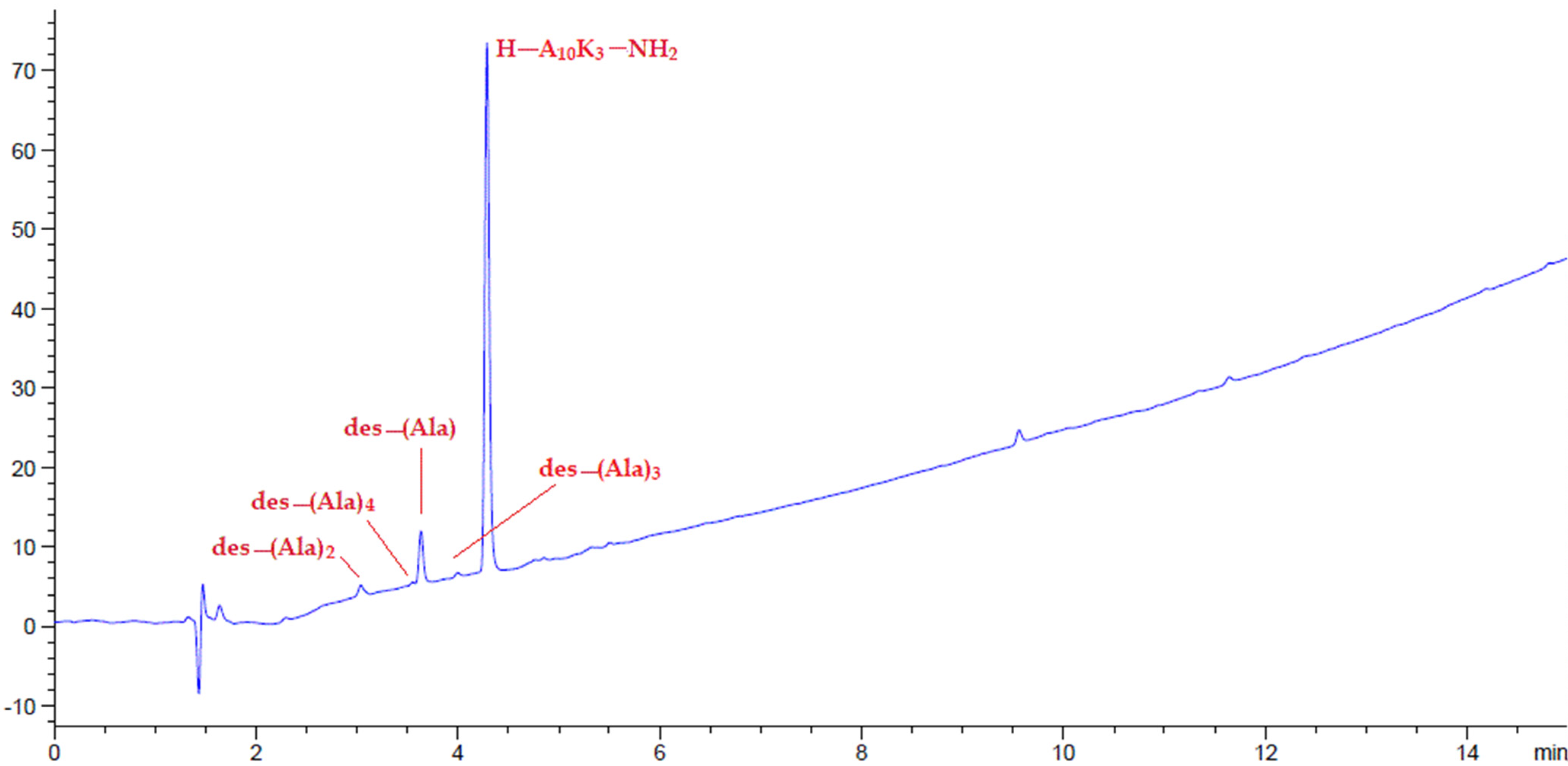
2.4. Peptide Synthesis
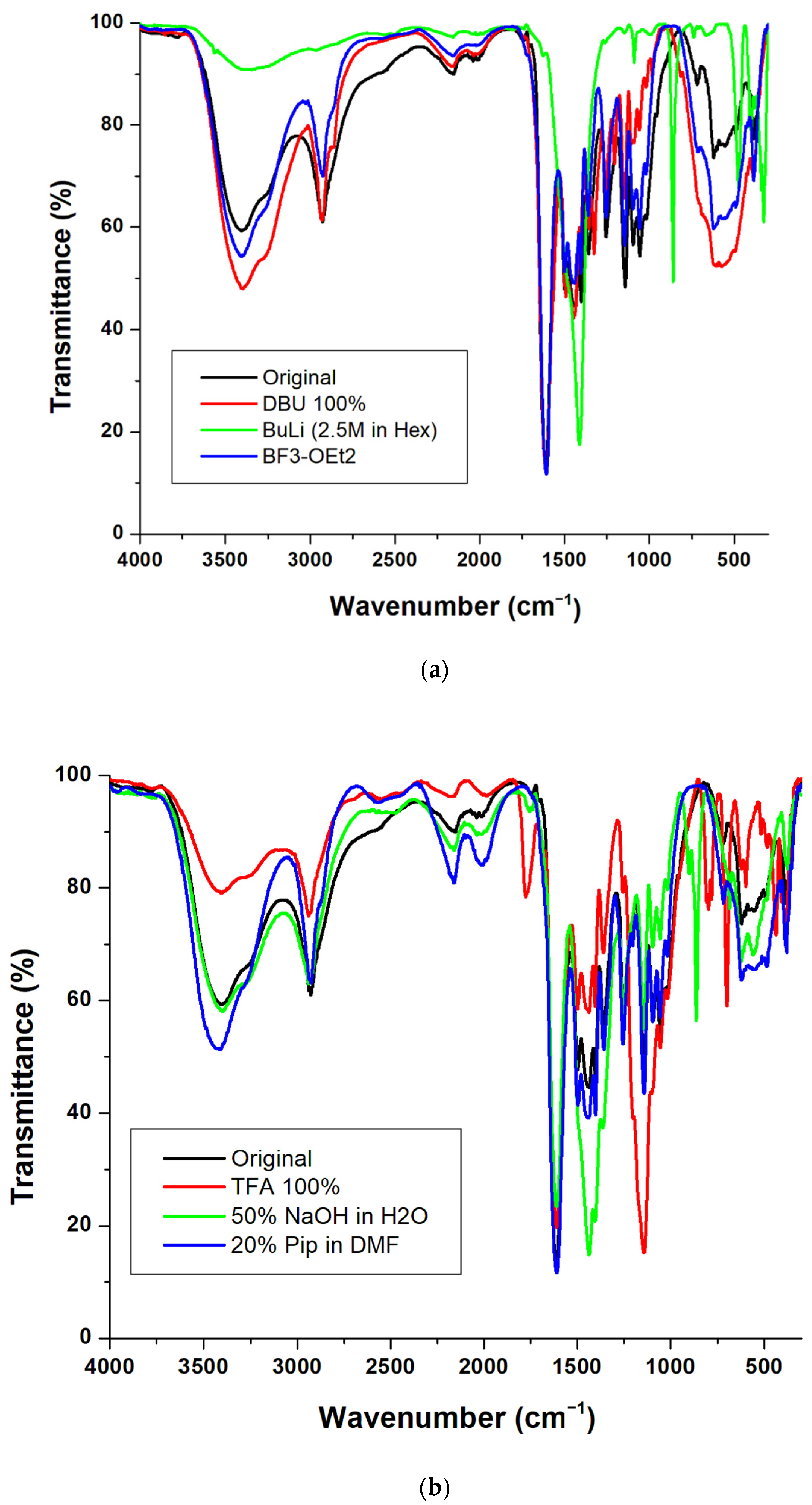
3. Results and Discussion
3.1. Preparation
3.2. Physical Properties
3.3. Swelling
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Merrifield, R.B. Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar] [CrossRef]
- Lloyd-Williams, P.; Albericio, F.; Giralt, E. Chemical Approaches to the Synthesis of Peptides and Proteins, 1st ed.; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar] [CrossRef]
- Zompra, A.A.; Galanis, A.S.; Werbitzky, O.; Albericio, F. Manufacturing peptides as active pharmaceutical ingredients. Future Med. Chem. 2009, 1, 361–377. [Google Scholar] [CrossRef] [PubMed]
- Merrifield, B. Concept and early development of solid-phase peptide synthesis. Methods Enzymol. 1997, 289, 3–13. [Google Scholar] [CrossRef]
- Ramkisson, S.; Jad, Y.E.; Sharma, A.; de la Torre, B.G.; Albericio, F. OctaGel Resin-A New PEG-PS-based Solid Support for Solid-Phase Peptide Synthesis. Lett. Org. Chem. 2019, 16, 935–940. [Google Scholar] [CrossRef]
- García-Martín, F.; Quintanar-Audelo, M.; García-Ramos, Y.; Cruz, L.J.; Gravel, C.; Furic, R.; Côté, S.; Tulla-Puche, J.; Albericio, F. ChemMatrix, a poly (ethylene glycol)-based support for the solid-phase synthesis of complex peptides. J. Comb. Chem. 2006, 8, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Kates, S.A.; McGuinness, B.F.; Blackburn, C.; Griffin, G.W.; Solé, N.A.; Barany, G.; Albericio, F. “High-load” polyethylene glycol–polystyrene (PEG–PS) graft supports for solid-phase synthesis. Pept. Sci. 1998, 47, 365–380. [Google Scholar] [CrossRef]
- Martin, V.; Jadhav, S.; Egelund, P.H.G.; Liffert, R.; Castro, H.J.; Krüger, T.; Haselmann, K.F.; Quement, S.T.L.; Albericio, F.; Dettner, F.; et al. Harnessing polarity and viscosity to identify green binary solvent mixtures as viable alternatives to DMF in solid-phase peptide synthesis. Green Chem. 2021, 23, 3295–3311. [Google Scholar] [CrossRef]
- Acosta, G.A.; del Fresno, M.; Paradis-Bas, M.; Rigau-DeLlobet, M.; Côté, S.; Royo, M.; Albericio, F. Solid-phase peptide synthesis using acetonitrile as a solvent in combination with PEG-based resins. J. Pept. Sci. 2009, 15, 629–633. [Google Scholar] [CrossRef]
- Albericio, F.; Carpino, L.A. [7] Coupling reagents and activation. Methods Enzymol. 1997, 289, 104–126. [Google Scholar] [CrossRef]
- Kamiński, Z.J.; Kolesińska, B.; Kolesińska, J.; Sabatino, G.; Chelli, M.; Rovero, P.; Błaszczyk, M.; Główka, M.L.; Papini, A.M. N-Triazinylammonium tetrafluoroborates. A new generation of efficient coupling reagents useful for peptide synthesis. J. Am. Chem. Soc. 2005, 127, 16912–16920. [Google Scholar] [CrossRef]
- Albericio, F.; Bofill, J.M.; El-Faham, A.; Kates, S.A. Use of Onium Salt-Based Coupling Reagents in Peptide Synthesis1. J. Org. Chem. 1998, 63, 9678–9683. [Google Scholar] [CrossRef]
- Albericio, F. Orthogonal protecting groups for Nα-amino and C-terminal carboxyl functions in solid-phase peptide synthesis. Pept. Sci. 2000, 55, 123–139. [Google Scholar] [CrossRef]
- Zervas, L.; Borovas, D.; Gazis, E. New methods in peptide synthesis. I. Tritylsulfenyl and o-nitrophenylsulfenyl groups as N-protecting groups. J. Am. Chem. Soc. 1963, 85, 3660–3666. [Google Scholar] [CrossRef]
- Ramage, R.; Hopton, D.; Parrott, M.J.; Kenner, G.W.; Moore, G.A. Phosphinamides: A new class of amino protecting groups in peptide synthesis. J. Chem. Soc. Perkin Trans. 1984, 1, 1357–1370. [Google Scholar] [CrossRef]
- Kenner, G.; Moore, G.; Ramage, R. Phosphinamides-a new class of amino protecting groups in peptide chemistry. Tetrahedron Lett. 1976, 17, 3623–3626. [Google Scholar] [CrossRef]
- Neumann, K.; Farnung, J.; Baldauf, S.; Bode, J.W. Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups. Nat. Commun. 2020, 11, 982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paradís-Bas, M.; Tulla-Puche, J.; Albericio, F. 2-Methoxy-4-methylsulfinylbenzyl: A backbone amide safety-catch protecting group for the synthesis and purification of difficult peptide sequences. Chem. Eur. J. 2014, 20, 15031–15039. [Google Scholar] [CrossRef] [PubMed]
- Bedford, J.; Hyde, C.; Johnson, T.; Jun, W.; Owen, D.; Quibell, M.; Sheppard, R.C. Amino acid structure and “difficult sequences” in solid phase peptide synthesis. Int. J. Pept. Protein Res. 1992, 40, 300–307. [Google Scholar] [CrossRef]
- Atherton, E.; Clive, D.L.J.; Sheppard, R.C. Polyamide supports for polypeptide synthesis. In Peptides 1971: Proceedings of the 11th European Peptide Symposium; North Holland Publishing Co.: Amsterdam, The Netherlands, 1973; p. 111. [Google Scholar]
- Atherton, E.; Clive, D.L.; Sheppard, R.C. Polyamide supports for polypeptide synthesis. J. Am. Chem. Soc. 1975, 97, 6584–6585. [Google Scholar] [CrossRef]
- Arshady, R.; Atherton, E.; Gait, M.J.; Lee, K.; Sheppard, R.C. Easily prepared polar support for solid phase peptide and oligonucleotide synthesis. Preparation of substance P and a nonadeoxyribonucleotide. J. Chem. Soc. Chem. Commun. 1979, 9, 423–425. [Google Scholar] [CrossRef]
- Heidmann, W.; Köster, H. Festphasensynthese von oligonucleotiden, 11. Verwendung eines neuartigen hydrophilen perlpolymerisats als träger. Makromol. Chem. 1980, 181, 2495–2506. [Google Scholar] [CrossRef]
- Arshady, R.; Atherton, E.; Clive, D.L.; Sheppard, R.C. Peptide synthesis. Part 1. Preparation and use of polar supports based on poly (dimethylacrylamide). J. Chem. Soc. Perkin Trans. 1981, 529–537. [Google Scholar] [CrossRef]
- Arshady, R. Development of new hydrophilic polymer supports based on dimethylacrylamide. Colloid. Polym. Sci. 1990, 268, 948–958. [Google Scholar] [CrossRef]
- Atherton, E.; Logan, C.J.; Sheppard, R.C. Peptide synthesis. Part 2. Procedures for solid-phase synthesis using N α-fluorenylmethoxycarbonylamino-acids on polyamide supports. Synthesis of substance P and of acyl carrier protein 65–74 decapeptide. J. Chem. Soc. Perkin Trans. 1981, 538–546. [Google Scholar] [CrossRef]
- Kanda, P.; Kennedy, R.C.; Sparrow, J.T. Synthesis of polyamide supports for use in peptide synthesis and as peptide-resin conjugates for antibody production. Int. J. Pept. Protein Res. 1991, 38, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Sparrow, J.T.; Knieb-Cordonier, N.G.; Obeyseskere, N.U.; McMurray, J.S. Large-pore polydimethylacrylamide resin for solid-phase peptide synthesis: Applications in Fmoc chemistry. Pept. Res. 1996, 9, 297–304. [Google Scholar]
- Zalipsky, S.A.; Albericio, F.; Barany, G. Peptides: Structure and Function: Proceedings of the Ninth American Peptide Symposium; Deber, C.M., Hruby, V.J., Kopple, K.D., Eds.; Pierce Chemical Company: Rockford, IL, USA, 1985; pp. 257–260. [Google Scholar]
- Bayer, E.H.; Albert, K.; Rapp, W.; Dengler, M. Peptides, Structure and Function: Proceedings of the Eighth American Peptide Symposium; Hruby, V.J., Rich, D.H., Eds.; Pierce Chemical Company: Rockford, IL, USA, 1983; pp. 87–90. [Google Scholar]
- Rapp, W. Combinatorial Peptide and Nonpeptide Libraries: A Handbook; Jung, G., Ed.; VCH: Weinheim, Germany, 1996; pp. 425–464. [Google Scholar]
- Adams, J.H.; Cook, R.M.; Hudson, D.; Jammalamadaka, V.; Lyttle, M.H.; Songster, M.F. A Reinvestigation of the Preparation, Properties, and Applications of Aminomethyl and 4-Methylbenzhydrylamine Polystyrene Resins1. J. Org. Chem. 1998, 63, 3706–3716. [Google Scholar] [CrossRef]
- Gooding, O.W.; Baudart, S.; Deegan, T.L.; Heisler, K.; Labadie, J.W.; Newcomb, W.S.; Porco, J.A., Jr.; van Eikeren, P. On the development of new poly (styrene-oxyethylene) graft copolymer resin supports for solid-phase organic synthesis. J. Comb. Chem. 1999, 1, 113–122. [Google Scholar] [CrossRef]
- Auzanneau, F.I.; Meldal, M.; Bock, K. Synthesis, characterization and biocompatibility of PEGA resins. J. Pept. Sci. 1995, 1, 31–44. [Google Scholar] [CrossRef]
- Meldal, M. Pega: A flow stable polyethylene glycol dimethyl acrylamide copolymer for solid phase synthesis. Tetrahedron Lett. 1992, 33, 3077–3080. [Google Scholar] [CrossRef]
- Meldal, M. Properties of solid supports. Methods Enzymol. 1997, 289, 83–104. [Google Scholar] [CrossRef] [PubMed]
- Darlak, K.; Wiegandt Long, D.; Czerwinski, A.; Darlak, M.; Valenzuela, F.; Spatola, A.; Barany, G. Facile preparation of disulfide-bridged peptides using the polymer-supported oxidant CLEAR-OXTM. J. Pept. Res. 2004, 63, 303–312. [Google Scholar] [CrossRef]
- Kempe, M.; Barany, G. CLEAR: A novel family of highly cross-linked polymeric supports for solid-phase peptide synthesis1,2. J. Am. Chem. Soc. 1996, 118, 7083–7093. [Google Scholar] [CrossRef]
- Tulla-Puche, J.; Barany, G. On-Resin Native Chemical Ligation for Cyclic Peptide Synthesis1,2. J. Org. Chem. 2004, 69, 4101–4107. [Google Scholar] [CrossRef] [PubMed]
- Rademann, J.; Grøtli, M.; Meldal, M.; Bock, K. SPOCC: A resin for solid-phase organic chemistry and enzymatic reactions on solid phase. J. Am. Chem. Soc. 1999, 121, 5459–5466. [Google Scholar] [CrossRef]
- Renil, M.; Meldal, M. POEPOP and POEPS: Inert polyethylene glycol crosslinked polymeric supports for solid synthesis. Tetrahedron Lett. 1996, 37, 6185–6188. [Google Scholar] [CrossRef]
- Cote, S. New Polyether Based Monomers and Highly Cross-Linked Amphiphile Resins. Patent WO 2005012277, 10 February 2005. [Google Scholar]
- García-Martín, F.; White, P.; Steinauer, R.; Côté, S.; Tulla-Puche, J.; Albericio, F. The synergy of ChemMatrix resin® and pseudoproline building blocks renders Rantes, a complex aggregated chemokine. Pept. Sci. 2006, 84, 566–575. [Google Scholar] [CrossRef]
- García-Ramos, Y.; Paradís-Bas, M.; Tulla-Puche, J.; Albericio, F. ChemMatrix® for complex peptides and combinatorial chemistry. J. Pept. Sci. 2010, 16, 675–678. [Google Scholar] [CrossRef]
- Kijewska, M.; Nuti, F.; Wierzbicka, M.; Waliczek, M.; Ledwoń, P.; Staśkiewicz, A.; Real-Fernandez, F.; Sabatino, G.; Rovero, P.; Stefanowicz, P. An Optimised Di-Boronate-ChemMatrix Affinity Chromatography to Trap Deoxyfructosylated Peptides as Biomarkers of Glycation. Molecules 2020, 25, 755. [Google Scholar] [CrossRef] [Green Version]
- Santini, R.; Griffith, M.C.; Qi, M. A measure of solvent effects on swelling of resins for solid phase organic synthesis. Tetrahedron Lett. 1998, 39, 8951–8954. [Google Scholar] [CrossRef]
- Sabbagh, F.; Muhamad, I.I. Physical and Chemical Characterisation of Acrylamide-Based Hydrogels, Aam, Aam/NaCMC and Aam/NaCMC/MgO. J. Inorg. Organomet. Polym. Mater. 2017, 27, 1439–1449. [Google Scholar] [CrossRef]
- Yraola, F.; Ventura, R.; Vendrell, M.; Colombo, A.; Fernàndez, J.C.; de la Figuera, N.; Fernández-Forner, D.; Royo, M.; Forns, P.; Albericio, F. A Re-evaluation of the Use of Rink, BAL, and PAL Resins and Linkers. QSAR Comb. Sci. 2004, 23, 145–152. [Google Scholar] [CrossRef]
- Martínez-Ceron, M.C.; Marani, M.M.; Taulés, M.; Etcheverrigaray, M.; Albericio, F.; Cascone, O.; Camperi, S.A. Affinity chromatography based on a combinatorial strategy for rerythropoietin purification. ACS Comb. Sci. 2011, 13, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Al Musaimi, O.; Beatriz, G.; Albericio, F. Greening Fmoc/t Bu solid-phase peptide synthesis. Green Chem. 2020, 22, 996–1018. [Google Scholar] [CrossRef]
- Hamdan, F.; Bigdeli, Z.; Balalaie, S.; Sewald, N.; Michalek, C. Efficient synthesis of novel RGD based peptides and the conjugation of the pyrazine moiety to their N-terminus. New J. Chem. 2019, 43, 2702–2709. [Google Scholar] [CrossRef]
- Kates, S.A.; Solé, N.A.; Beyermann, M.; Barany, G.; Albericio, F. Optimized preparation of deca(L-alanyl)-L-valinamide by 9-fluorenylmethyloxycarbonyl (Fmoc) solid-phase synthesis on polyethylene glycol-polystyrene (PEG-PS) graft supports, with 1,8-diazobicyclo [5.4.0]-undec-7-ene (DBU) deprotection. Pept. Res. 1996, 9, 106–113. [Google Scholar]
- Fischer, P.M.; Comis, A.; Howden, M.E.H. Direct immunization with synthetic peptidyl-polyamide resin: Comparison with antibody production from free peptide and conjugates with carrier proteins. J. Immunol. Methods 1989, 118, 119–123. [Google Scholar] [CrossRef]







| Entry (#) | Solvent | Swelling (mL/g) | ||||
|---|---|---|---|---|---|---|
| Li-resin | PS | PEG-PS | CM | CLEAR | ||
| 1 | DMF | 5.75 | 4.75 | 4.25 | 8.25 | 3.25 |
| 2 | DCM | 7.75 | 5.25 | 5.25 | 10.75 | 3.75 |
| 3 | H2O * | 6.75 | 1.75 | 1.75 | 8.25 | 2.25 |
| 4 | MeOH | 10.25 | 2.75 | 2.75 | 9.75 | 3.25 |
| 5 | IPA * | 8.5 | 1.75 | 1.75 | 2.25 | 2.25 |
| 6 | ACN | 4.25 | 1.75 | 3.25 | 8.25 | 3.25 |
| 7 | DMSO * | 6.25 | 6.25 | 3.75 | 8.25 | 3.25 |
| 8 | GVL * | 4.25 | 3.25 | 4.25 | 9.25 | 3.25 |
| 9 | NFM * | 5.25 | 2.75 | 3.75 | 8.25 | 3.25 |
| 10 | NBP * | 2.75 | 6.25 | 4.25 | 2.75 | 3.25 |
| 11 | 2-Me-THF * | 2.25 | 4.25 | 2.75 | 2.25 | 2.75 |
| 12 | AcOEt * | 2.25 | 2.75 | 2.75 | 6.25 | 2.75 |
| 13 | Polar Clean * | 2.25 | 2.25 | 3.75 | 7.25 | 3.25 |
| 14 | CPME * | 2.25 | 3.75 | 2.25 | 3.25 | 2.75 |
| 15 | (CH3O)2CO | 3.25 | 3.25 | 2.75 | 9.25 | 3.75 |
| 16 | Anisole * | 2.75 | 4.25 | 3.75 | 11.25 | 3.25 |
| 17 | 1,3-DMB * | 2.25 | 4.25 | 3.75 | 10.25 | 3.25 |
| 18 | 1,3-DMB-NFM (3:1) * | 3.75 | 4.25 | 4.25 | 10.25 | 4.25 |
| 19 | NBP-AcOEt (1:4) * | 3.25 | 5.75 | 3.75 | 9.25 | 3.75 |
| 20 | TFA | 11.25 | 2.25 | 5.75 | 15.25 | 5.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akintayo, D.C.; de la Torre, B.G.; Li, Y.; Albericio, F. Amino-Li-Resin—A Fiber Polyacrylamide Resin for Solid-Phase Peptide Synthesis. Polymers 2022, 14, 928. https://doi.org/10.3390/polym14050928
Akintayo DC, de la Torre BG, Li Y, Albericio F. Amino-Li-Resin—A Fiber Polyacrylamide Resin for Solid-Phase Peptide Synthesis. Polymers. 2022; 14(5):928. https://doi.org/10.3390/polym14050928
Chicago/Turabian StyleAkintayo, Damilola C., Beatriz G. de la Torre, Yongfu Li, and Fernando Albericio. 2022. "Amino-Li-Resin—A Fiber Polyacrylamide Resin for Solid-Phase Peptide Synthesis" Polymers 14, no. 5: 928. https://doi.org/10.3390/polym14050928
APA StyleAkintayo, D. C., de la Torre, B. G., Li, Y., & Albericio, F. (2022). Amino-Li-Resin—A Fiber Polyacrylamide Resin for Solid-Phase Peptide Synthesis. Polymers, 14(5), 928. https://doi.org/10.3390/polym14050928








