Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Preparation
2.2.1. Master Batch of Gallic Acid with PBAT or PBS
2.2.2. Cast Sheet Extrusion
2.3. Film Characterization
2.3.1. Microstructure
2.3.2. Surface Topography
2.3.3. X-ray Diffraction (XRD)
2.3.4. Dynamic Mechanical Thermal Analysis (DMTA)
2.3.5. Mechanical Properties
2.3.6. Surface Hydrophobicity
2.3.7. Barrier Properties
Water Vapor Permeability
Oxygen Permeability
2.4. Oxygen Absorption Capacity
2.5. Statistical Analysis
3. Results and Discussion
3.1. Microstructures
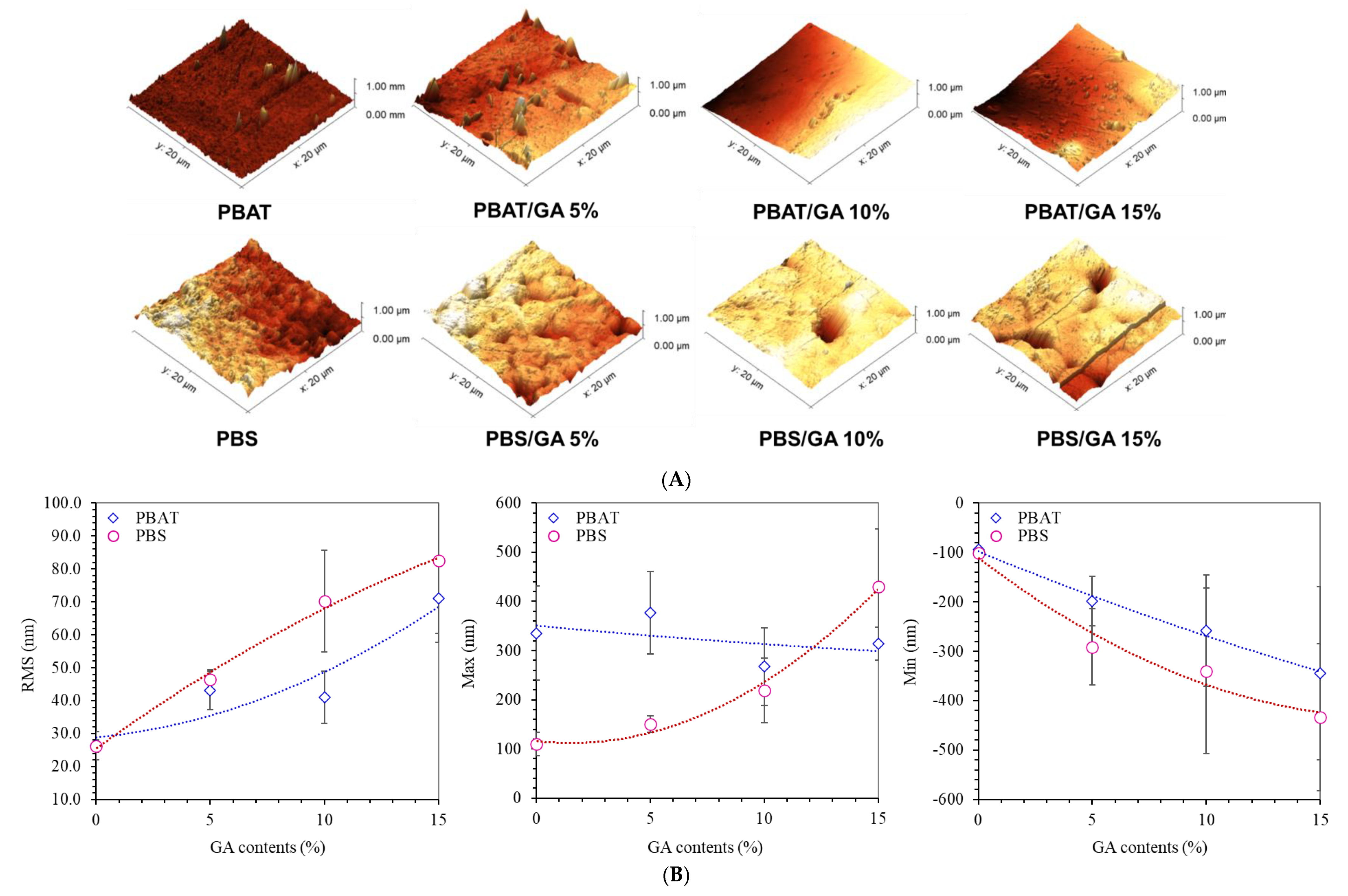
3.2. Surface Topography
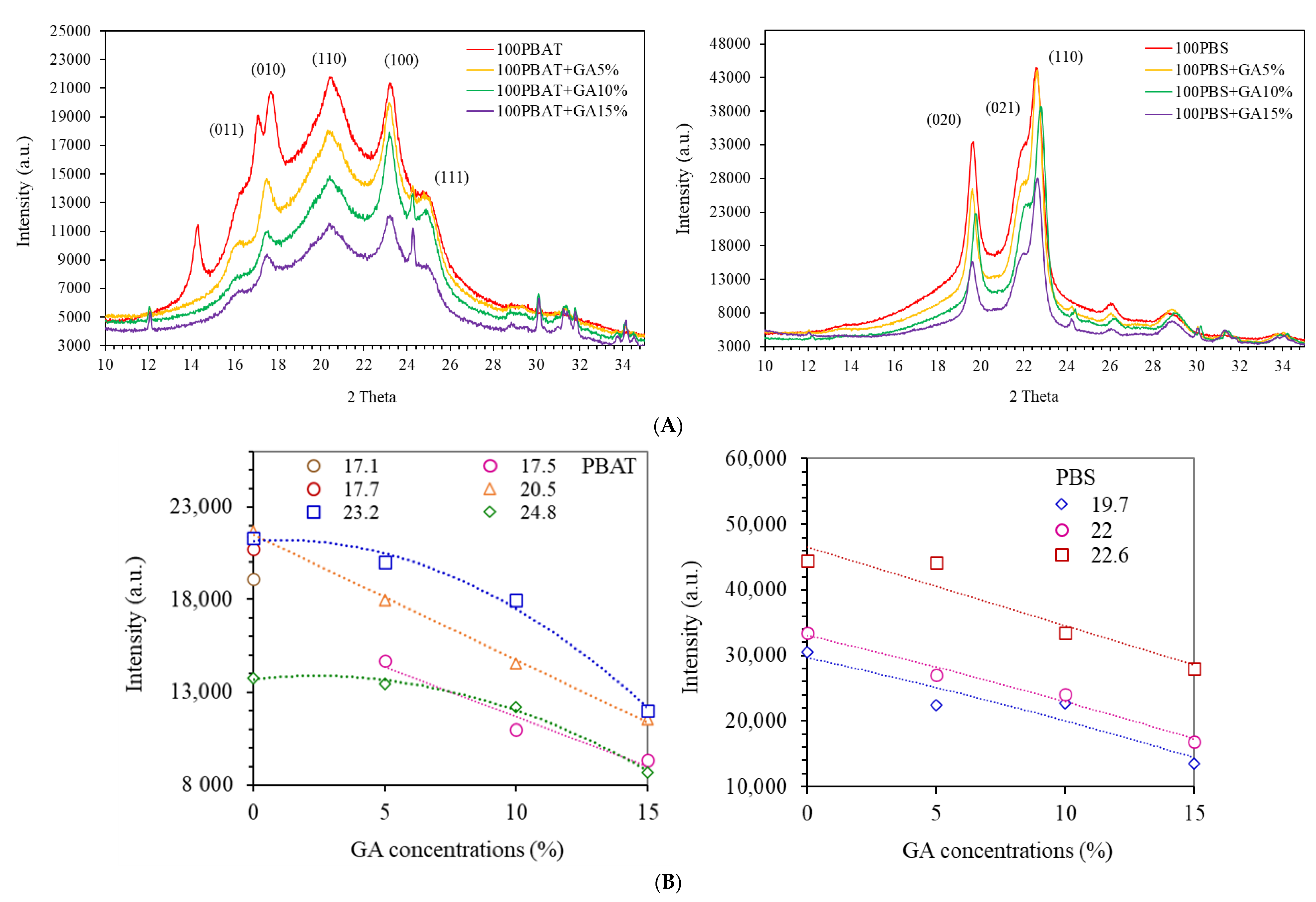
3.3. X-ray Diffraction
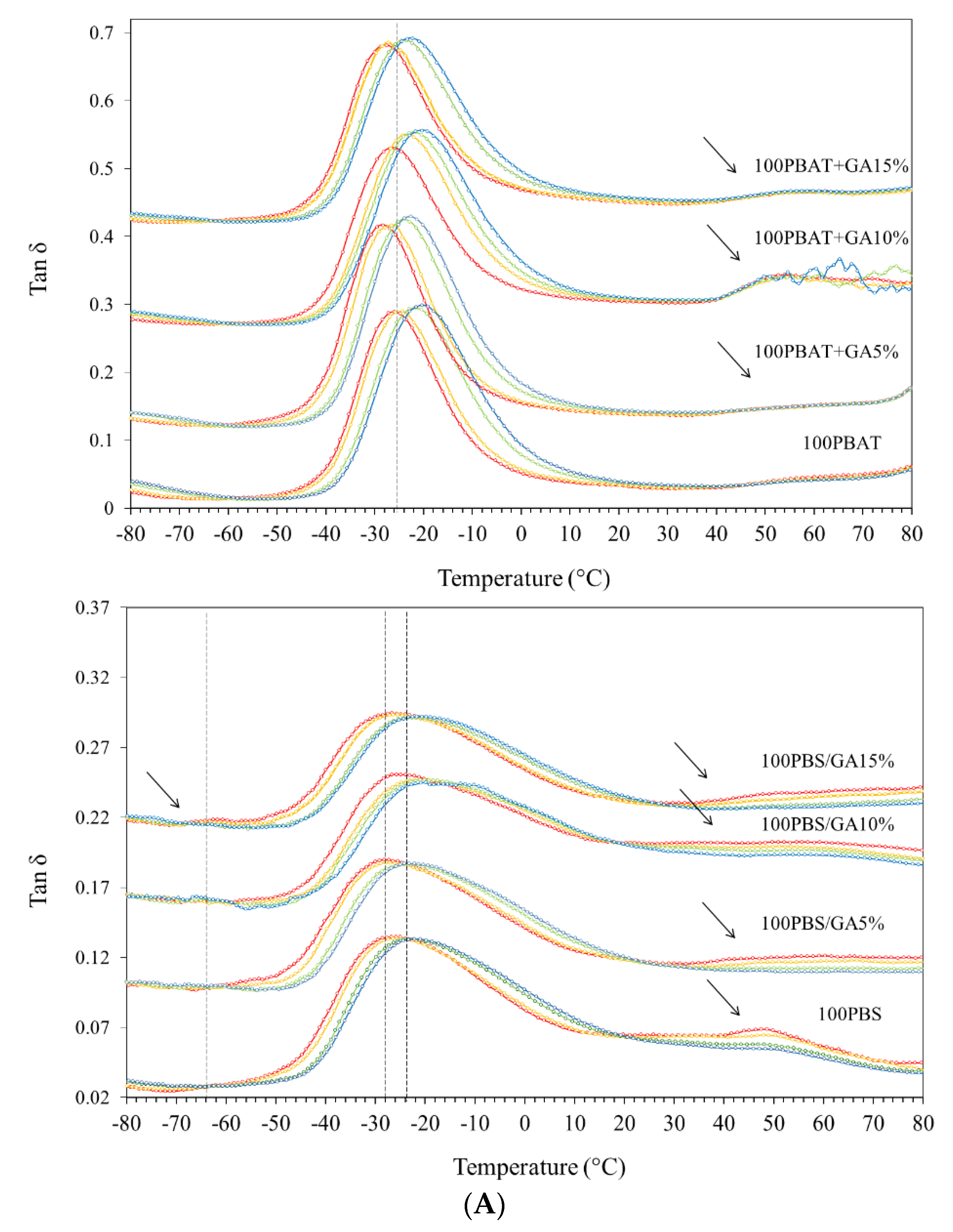
3.4. DMTA
3.5. Mechanical Properties
3.6. Surface Hydrophobicity and Barrier Properties
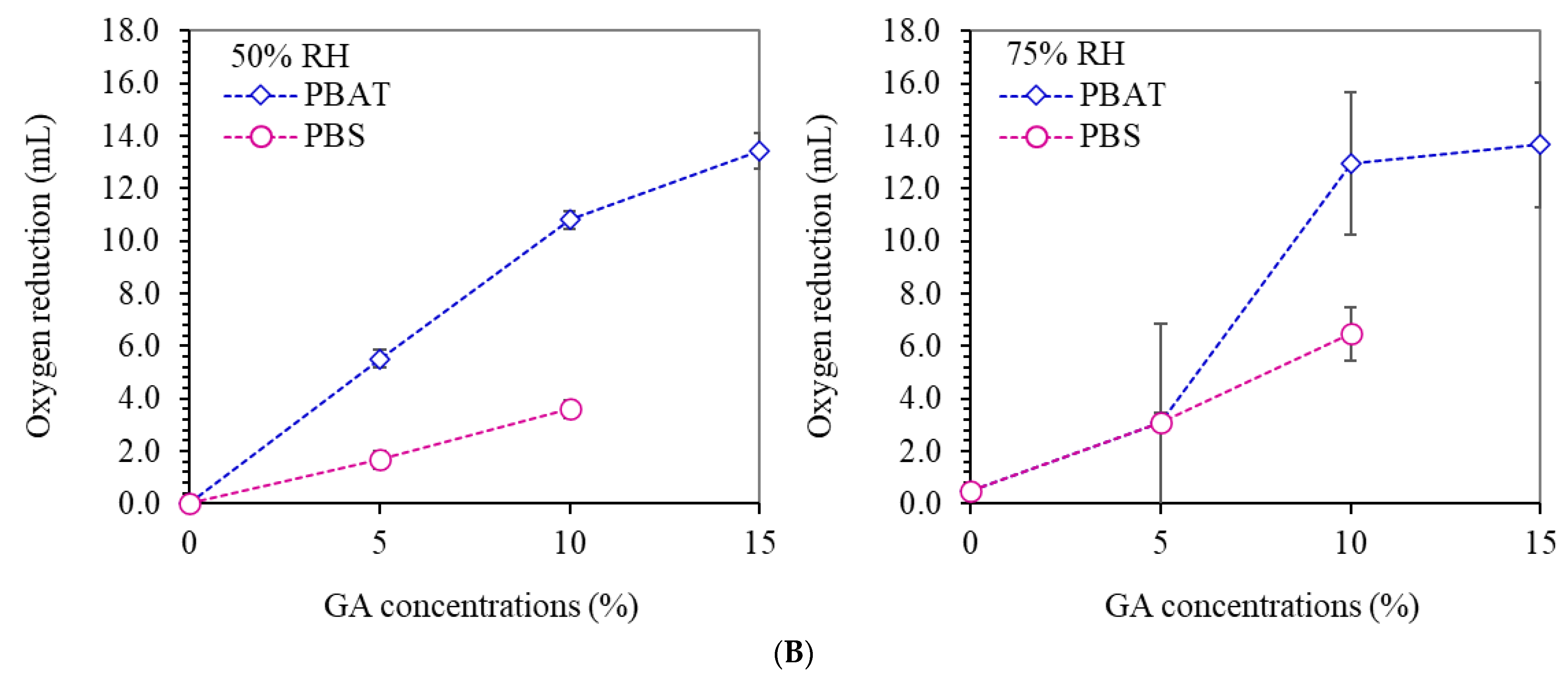
3.7. Oxygen Absorption Capacity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohanty, S.; Nayak, S.K. Starch based biodegradable PBAT nanocomposites: Effect of starch modification on mechanical, thermal, morphological and biodegradability behavior. Int. J. Plast. Technol. 2009, 13, 163–185. [Google Scholar] [CrossRef]
- Garalde, R.A.; Thipmanee, R.; Jariyasakoolroj, P.; Sane, A. The effects of blend ratio and storage time on thermoplastic starch/poly (butylene adipate-co-terephthalate) films. Heliyon 2019, 5, e01251. [Google Scholar]
- Pattanayaiying, R.; Sane, A.; Photjanataree, P.; Cutter, C.N. Thermoplastic starch/polybutylene adipate terephthalate film coated with gelatin containing nisin Z and lauric arginate for control of foodborne pathogens associated with chilled and frozen seafood. Int. J. Food Microbiol. 2019, 290, 59–67. [Google Scholar] [PubMed]
- de Matos Costa, A.R.; Crocitti, A.; Hecker de Carvalho, L.; Carroccio, S.C.; Cerruti, P.; Santagata, G. Properties of biodegradable films based on poly (butylene succinate) (PBS) and poly (butylene adipate-co-terephthalate) (PBAT) blends. Polymers 2020, 12, 2317. [Google Scholar] [CrossRef] [PubMed]
- Yimnak, K.; Thipmanee, R.; Sane, A. Poly (butylene adipate-co-terephthalate)/thermoplastic starch/zeolite 5A films: Effects of compounding sequence and plasticizer content. Int. J. Biol. Macromol. 2020, 164, 1037–1045. [Google Scholar] [PubMed]
- Rafiqah, S.; Khalina, A.; Harmaen, A.S.; Tawakkal, I.A.; Zaman, K.; Asim, M.; Lee, C.H. A review on properties and application of bio-based poly (butylene succinate). Polymers 2021, 13, 1436. [Google Scholar] [CrossRef]
- Di Giuseppe, F.; Coffigniez, F.; Aouf, C.; Guillard, V.; Torrieri, E. Activated gallic acid as radical and oxygen scavenger in biodegradable packaging film. Food Packag. Shelf Life 2022, 31, 100811. [Google Scholar]
- Dey, A.; Neogi, S. Oxygen scavengers for food packaging applications: A review. Trends Food Sci. Technol. 2019, 90, 26–34. [Google Scholar]
- Promsorn, J.; Harnkarnsujarit, N. Pyrogallol loaded thermoplastic cassava starch based films as bio-based oxygen scavengers. Ind. Crops Prod. 2022, 186, 115226. [Google Scholar] [CrossRef]
- Singh, G.; Singh, S.; Kumar, B.; Gaikwad, K.K. Active barrier chitosan films containing gallic acid based oxygen scavenger. J. Food Meas. Charact. 2021, 15, 585–593. [Google Scholar] [CrossRef]
- Pant, A.F.; Dorn, J.; Reinelt, M. Effect of temperature and relative humidity on the reaction kinetics of an oxygen scavenger based on gallic acid. Front. Chem. 2018, 6, 587. [Google Scholar] [PubMed]
- Prichapan, N.; McClements, D.J.; Klinkesorn, U. Iron encapsulation in water-in-oil emulsions: Effect of ferrous sulfate concentration and fat crystal formation on oxidative stability. J. Food Sci. 2018, 83, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Ahn, B.J.; Gaikwad, K.K.; Lee, Y.S. Characterization and properties of LDPE film with gallic-acid-based oxygen scavenging system useful as a functional packaging material. J. Appl. Polym. Sci. 2016, 133, 44138. [Google Scholar]
- Pant, A.F.; Sängerlaub, S.; Müller, K. Gallic acid as an oxygen scavenger in bio-based multilayer packaging films. Materials 2017, 10, 489. [Google Scholar] [PubMed]
- Phothisarattana, D.; Harnkarnsujarit, N. Migration, aggregations and thermal degradation behaviors of TiO2 and ZnO incorporated PBAT/TPS nanocomposite blown films. Food Packag. Shelf Life 2022, 33, 100901. [Google Scholar]
- Wongphan, P.; Panrong, T.; Harnkarnsujarit, N. Effect of different modified starches on physical, morphological, thermomechanical, barrier and biodegradation properties of cassava starch and polybutylene adipate terephthalate blend film. Food Packag. Shelf Life 2022, 32, 100844. [Google Scholar]
- Kadowaki, Y.; Kojio, K. Crystallization behavior of biodegradable poly (L-lactic acid)(PLLA)/poly (butylene succinate)(PBS) blends based on in situ simultaneous wide-angle x-ray diffraction/small-angle x-ray scattering techniques and thermal analyses. J. Polym. Res. 2022, 29, 137. [Google Scholar] [CrossRef]
- Liu, H.; Zhu, H.L.; Wang, Z.; Wu, X.; Huang, Z.; Huqe, M.R.; Choy, W.C. Double-Side Crystallization Tuning to Achieve over 1 µm Thick and Well-Aligned Block-Like Narrow-Bandgap Perovskites for High-Efficiency Near-Infrared Photodetectors. Adv. Funct. Mater. 2021, 31, 2010532. [Google Scholar] [CrossRef]
- Kathyayani, D.; Mahesh, B.; Chamaraja, N.A.; Madhukar, B.S.; Gowda, D.C. Synthesis and Structural Characterization of Elastin-based Polypentapeptide/hydroxypropylmethylcellulose Blend Films: Assessment of Miscibility, Thermal Stability and Surface Characteristics. Colloids Surf. A Physicochem. Eng. Asp. 2022, 649, 129503. [Google Scholar]
- Jariyasakoolroj, P.; Rojanaton, N.; Jarupan, L. Crystallization behavior of plasticized poly (lactide) film by poly (l-lactic acid)-poly (ethylene glycol)-poly (l-lactic acid) triblock copolymer. Polym. Bull. 2020, 77, 2309–2323. [Google Scholar]
- Di Lorenzo, M.L.; Androsch, R.; Righetti, M.C. Low-temperature crystallization of poly (butylene succinate). Eur. Polym. J. 2017, 94, 384–391. [Google Scholar] [CrossRef]
- Ichikawa, Y.; Kondo, H.; Igarashi, Y.; Noguchi, K.; Okuyama, K.; Washiyama, J. Crystal structures of α and β forms of poly (tetramethylene succinate). Polymer 2000, 41, 4719–4727. [Google Scholar]
- Ye, Q.; Wang, X.; Lu, Y. Kinetic behavior of potassium bicarbonate crystallization in a carbonate-based CO2 absorption process. Chem. Eng. Res. Des. 2015, 93, 136–147. [Google Scholar] [CrossRef]
- Ehsani, M.; Khonakdar, H.A.; Ghadami, A. Assessment of morphological, thermal, and viscoelastic properties of epoxy vinyl ester coating composites: Role of glass flake and mixing method. Prog. Org. Coat. 2013, 76, 238–243. [Google Scholar] [CrossRef]
- Ma, Y.; Qin, R.; Xu, M.; Jiang, X.; Sheng, Y.; Wang, M.; Lu, X. Wide temperature range damping polyurethane elastomer based on dynamic disulfide bonds. J. Appl. Polym. Sci. 2022, 139, 51453. [Google Scholar]
- Surendran, A.; Pionteck, J.; Kalarikkal, N.; Thomas, S. Mechanical responses of epoxy/cloisite nanocomposites. Mater. Chem. Phys. 2022, 281, 125755. [Google Scholar] [CrossRef]
- Wadaugsorn, K.; Panrong, T.; Wongphan, P.; Harnkarnsujarit, N. Plasticized hydroxypropyl cassava starch blended PBAT for improved clarity blown films: Morphology and properties. Ind. Crops Prod. 2022, 176, 114311. [Google Scholar]
- Facca, A.G.; Kortschot, M.T.; Yan, N. Predicting the tensile strength of natural fibre reinforced thermoplastics. Compos. Sci. Technol. 2007, 67, 2454–2466. [Google Scholar]
- Kargarzadeh, H.; Galeski, A.; Pawlak, A. PBAT green composites: Effects of kraft lignin particles on the morphological, thermal, crystalline, macro and micromechanical properties. Polymer 2020, 203, 122748. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Wongphan, P.; Promhuad, K.; Promsorn, J.; Harnkarnsujarit, N. Blown film extrusion of PBAT/TPS/ZnO nanocomposites for shelf-life extension of meat packaging. Colloids Surf. B Biointerfaces 2022, 214, 112472. [Google Scholar] [CrossRef]
- Cui, L.; Dong, Y.; Liu, Y.; Fan, S. The Effect of Stretching on the Crystal Structure and Crystal Orientation of PA510/SiO2 Films. Materials 2021, 14, 705. [Google Scholar] [CrossRef] [PubMed]
- Jothi Prakash, C.G.; Prasanth, R. Approaches to design a surface with tunable wettability: A review on surface properties. J. Mater. Sci. 2021, 56, 108–135. [Google Scholar] [CrossRef]
- Pacheco, N.; Naal-Ek, M.G.; Ayora-Talavera, T.; Shirai, K.; Román-Guerrero, A.; Fabela-Morón, M.F.; Cuevas-Bernardino, J.C. Effect of bio-chemical chitosan and gallic acid into rheology and physicochemical properties of ternary edible films. Int. J. Biol. Macromol. 2019, 125, 149–158. [Google Scholar] [PubMed]
- Kishi, M.; Nagatsuka, K.; Toda, T. Effect of membrane hydrophobicity and thickness on energy-efficient dissolved oxygen removal from algal culture. Front. Bioeng. Biotechnol. 2020, 8, 978. [Google Scholar] [CrossRef]
- Kitahara, T.; Nakajima, H.; Mori, K. Hydrophilic and hydrophobic double microporous layer coated gas diffusion layer for enhancing performance of polymer electrolyte fuel cells under no-humidification at the cathode. J. Power Sources 2012, 199, 29–36. [Google Scholar]
- Champion, D.; Le Meste, M.; Simatos, D. Towards an improved understanding of glass transition and relaxations in foods: Molecular mobility in the glass transition range. Trends Food Sci. Technol. 2000, 11, 41–55. [Google Scholar] [CrossRef]




| TS (MPa) | EB (%) | YM (Mpa) | CA | WVP | OP | ||||
|---|---|---|---|---|---|---|---|---|---|
| MD | CD | MD | CD | MD | CD | (°) | (g.mm/m2.Day.atm) | (cm3.mm/m2.Day.atm) | |
| PBAT | 26.9 ± 0.4 e | 21.9 ± 0.6 e | 684.3 ± 100.0 b | 1108.7 ± 0.4 d | 89.3 ± 1.8 a | 99.5 ± 5.8 b | 79.4 ± 2.5 f | 3.164 ± 0.011 c | 50.7 ± 1.6 b |
| PBAT/GA5% | 22.6 ± 0.9 d | 20.3 ± 0.4 d | 1103.5 ± 6.1 d | 1096.3 ± 16.0 d | 83.9 ± 1.9 a | 78.8 ± 3.1 a | 73.9 ± 0.6 e | 3.201 ± 0.021 c | 52.8 ± 4.1 b |
| PBAT/GA10% | 15.9 ± 1.2 b | 8.0 ± 0.3 b | 1064.6 ± 33.1 d | 497.4 ± 53.3 c | 79.9 ± 13.1 a | 75 ± 1.9 a | 71.6 ± 1.1 d | 4.215 ± 0.258 d | 90.4 ± 16.9 c |
| PBAT/GA15% | 10.7 ± 1.3 a | 5.7 ± 0.7 a | 931.5 ± 62.6 c | 271.1 ± 98.0 b | 74.6 ± 4.3 a | 71.4 ± 4.4 a | 67.8 ± 0.8 c | 4.589 ± 0.445 d | 195.4 ± 6.3 e |
| PBS | 35.4 ± 0.9 f | 33.3 ± 0.9 f | 34.9 ± 4.7 a | 33.1 ± 7.1 a | 531.4 ± 9.8 d | 536.6 ± 15.8 e | 83.5 ± 2.9 g | 1.221 ± 0.052 a | 30.1 ± 1.3 a |
| PBS/GA5% | 25.8 + 0.8 e | 20.0 ± 0.6 d | 10.8 ± 0.6 a | 7.5 ± 0.5 a | 453.3 ± 12.0 c | 461.0 ± 6.9 d | 64.4 ± 1.1 b | 1.228 ± 0.142 a | 170.9 ± 18.1 d |
| PBS/GA10% | 18.8 ± 1.7 c | 10.1 ± 0.6 c | 8.8 ± 0.4 a | 5.3 ± 0.3 a | 405.5 ± 24.4 b | 309.1 ± 17.4 c | 60.6 ± 1.8 a | 2.186 ± 0.888 b | 208.2 ± 2.8 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sonchaeng, U.; Promsorn, J.; Bumbudsanpharoke, N.; Chonhenchob, V.; Sablani, S.S.; Harnkarnsujarit, N. Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging. Polymers 2022, 14, 5296. https://doi.org/10.3390/polym14235296
Sonchaeng U, Promsorn J, Bumbudsanpharoke N, Chonhenchob V, Sablani SS, Harnkarnsujarit N. Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging. Polymers. 2022; 14(23):5296. https://doi.org/10.3390/polym14235296
Chicago/Turabian StyleSonchaeng, Uruchaya, Juthathip Promsorn, Nattinee Bumbudsanpharoke, Vanee Chonhenchob, Shyam S. Sablani, and Nathdanai Harnkarnsujarit. 2022. "Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging" Polymers 14, no. 23: 5296. https://doi.org/10.3390/polym14235296
APA StyleSonchaeng, U., Promsorn, J., Bumbudsanpharoke, N., Chonhenchob, V., Sablani, S. S., & Harnkarnsujarit, N. (2022). Polyesters Incorporating Gallic Acid as Oxygen Scavenger in Biodegradable Packaging. Polymers, 14(23), 5296. https://doi.org/10.3390/polym14235296









