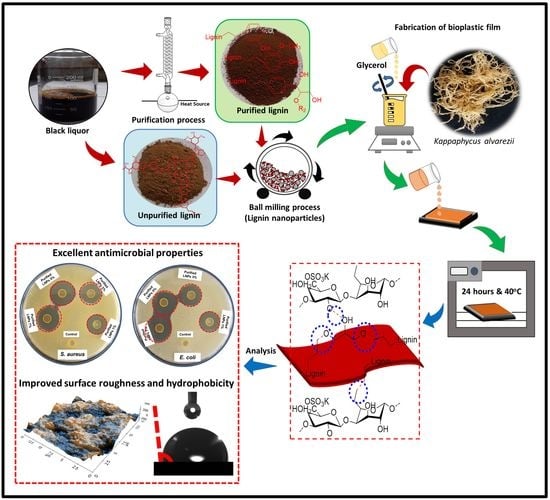
Enhanced Functional Properties of Bioplastic Films Using Lignin Nanoparticles from Oil Palm-Processing Residue
Abstract
1. Introduction
2. Materials and Method
2.1. Materials
2.2. Isolation and Purification of Lignin
2.3. Preparation of LNPs
2.4. Characterization of LNPs
2.5. Fabrication of Lignin Nanoparticles/Kappaphycus Alvarezii Bioplastic Films
2.6. Characterization of LNPs/Kappaphycus Alvarezii Bioplastic Films
2.6.1. Morphological Analysis
2.6.2. Structural Analysis
2.6.3. Hydrophobicity and Physical Properties
2.6.4. Antimicrobial Properties
2.6.5. Soil Burial Test
2.6.6. Statistical Analysis
3. Results and Discussion
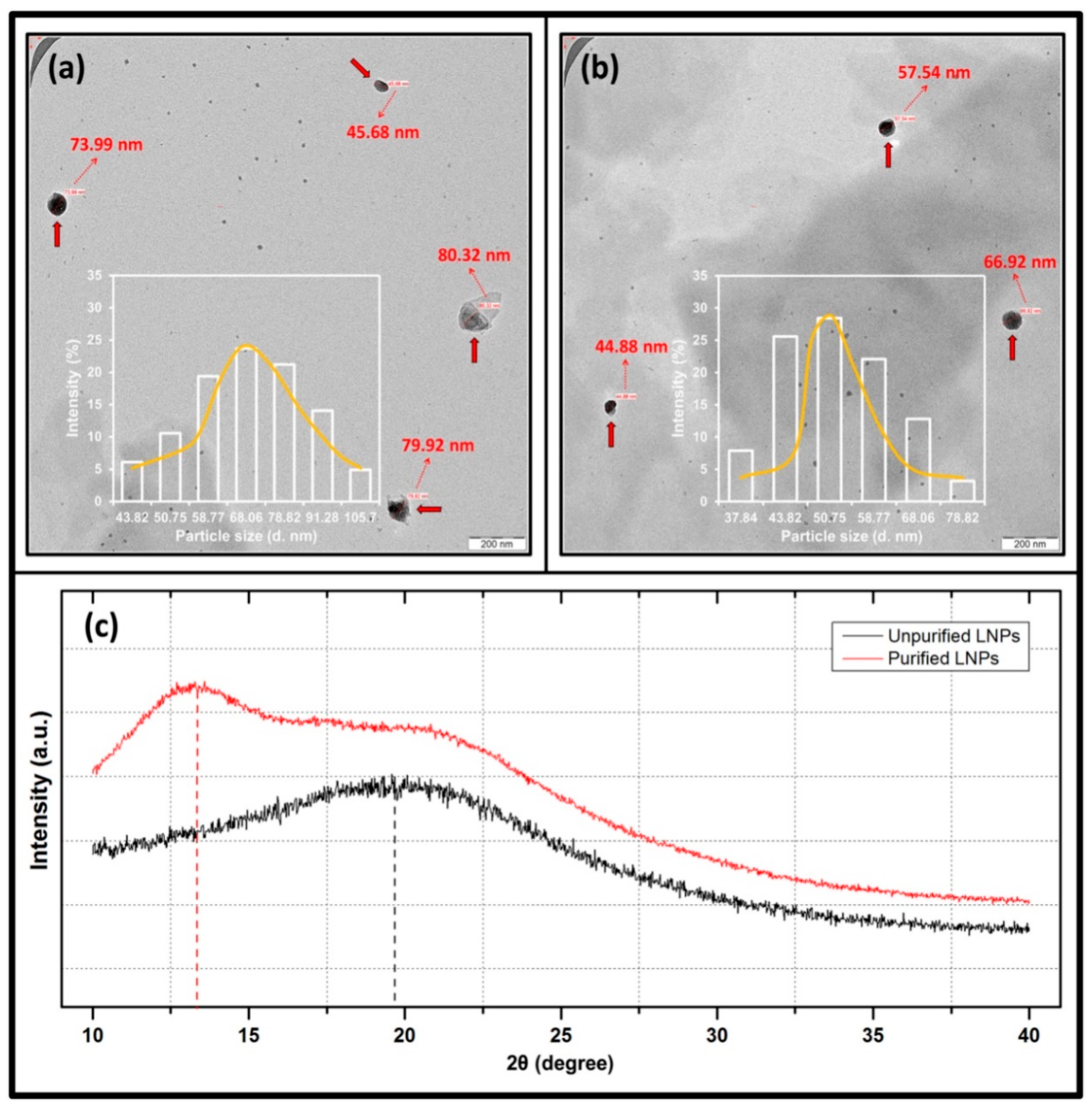
3.1. Characterization of Lignin Nanoparticles
3.2. Characterization of Biopolymer Films Reinforced with Lignin Nanoparticles
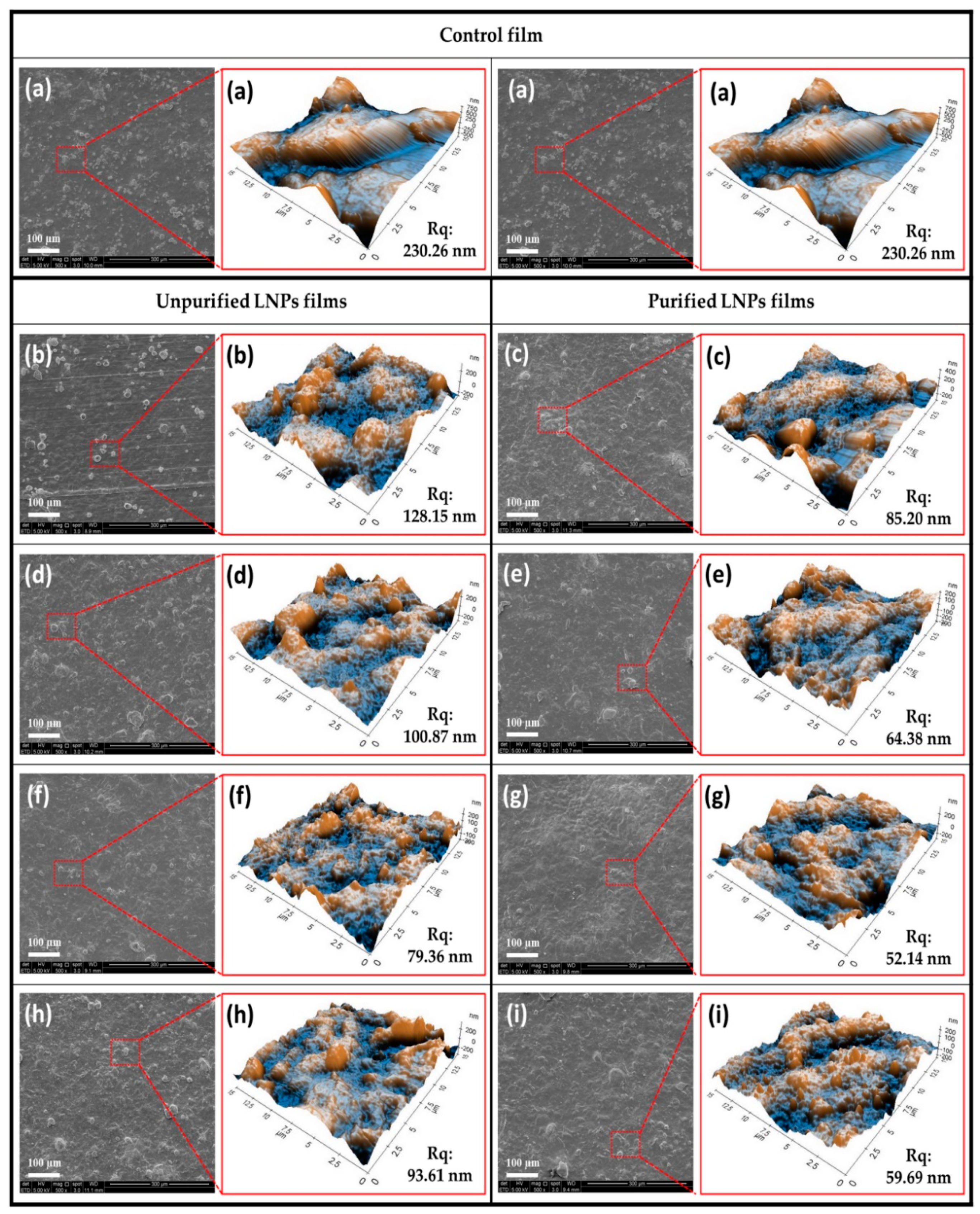
3.2.1. Morphological Analysis
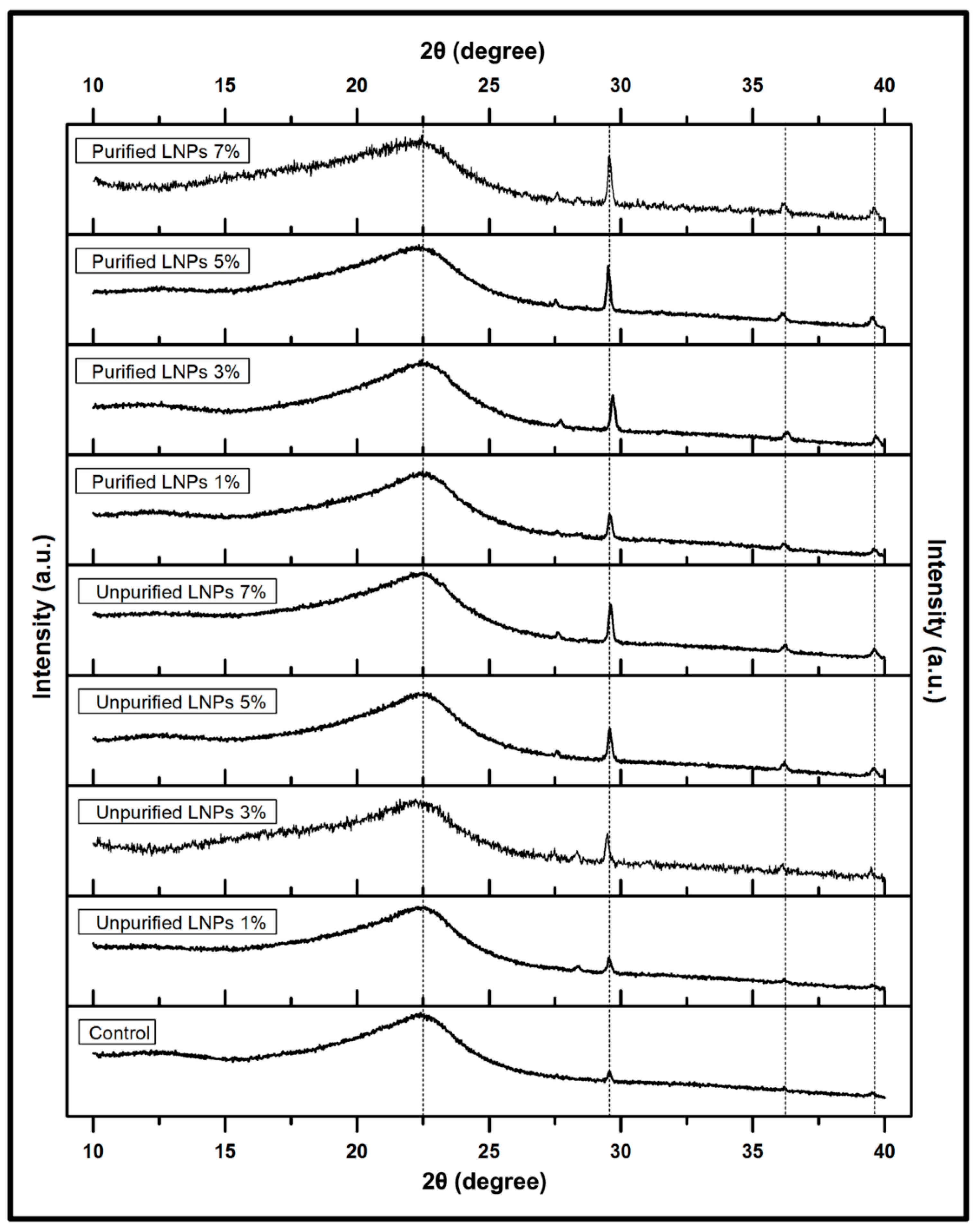
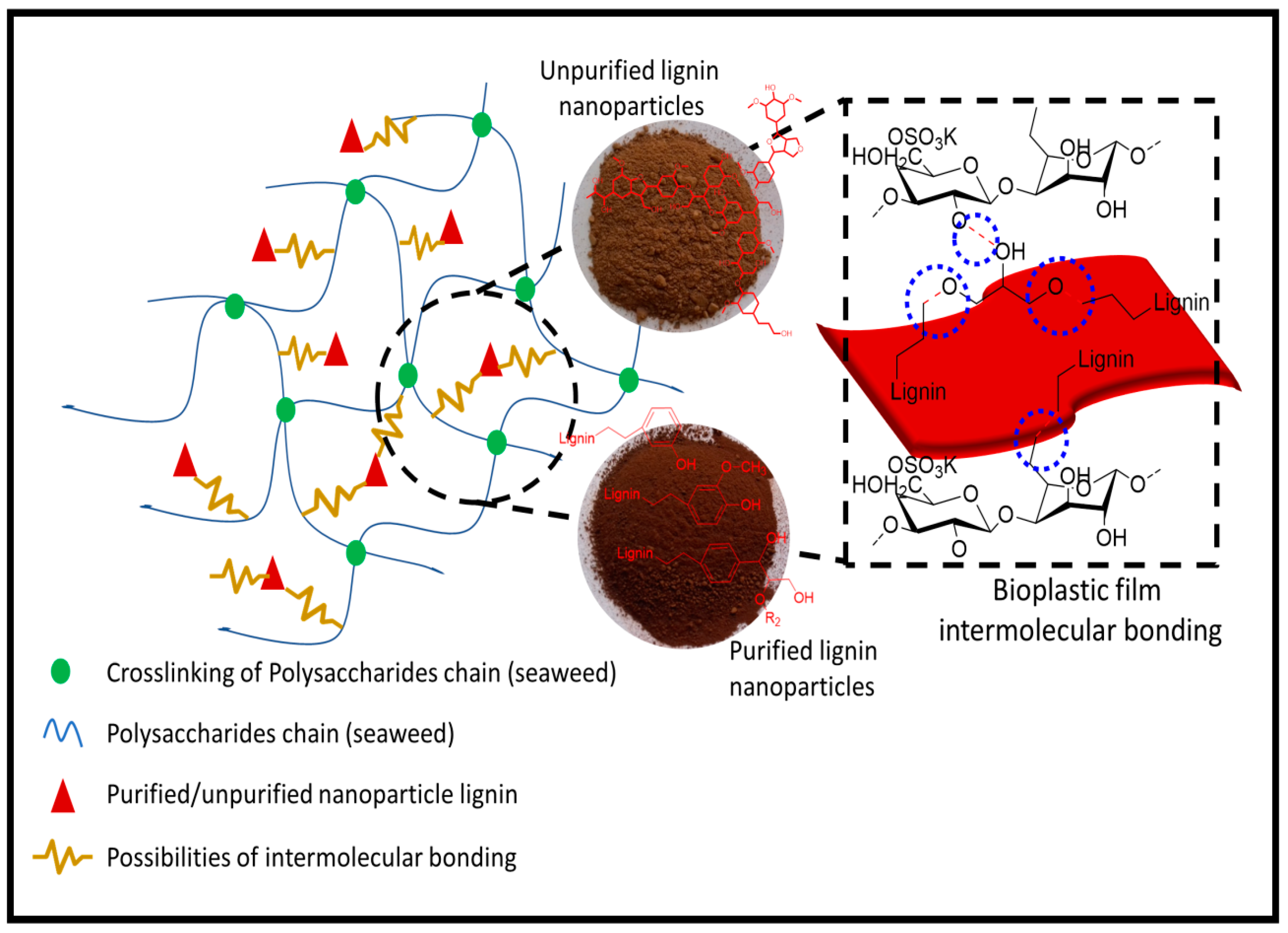
3.2.2. Structural Analysis
3.2.3. Hydrophobicity and Physical Properties
3.2.4. Antimicrobial Properties
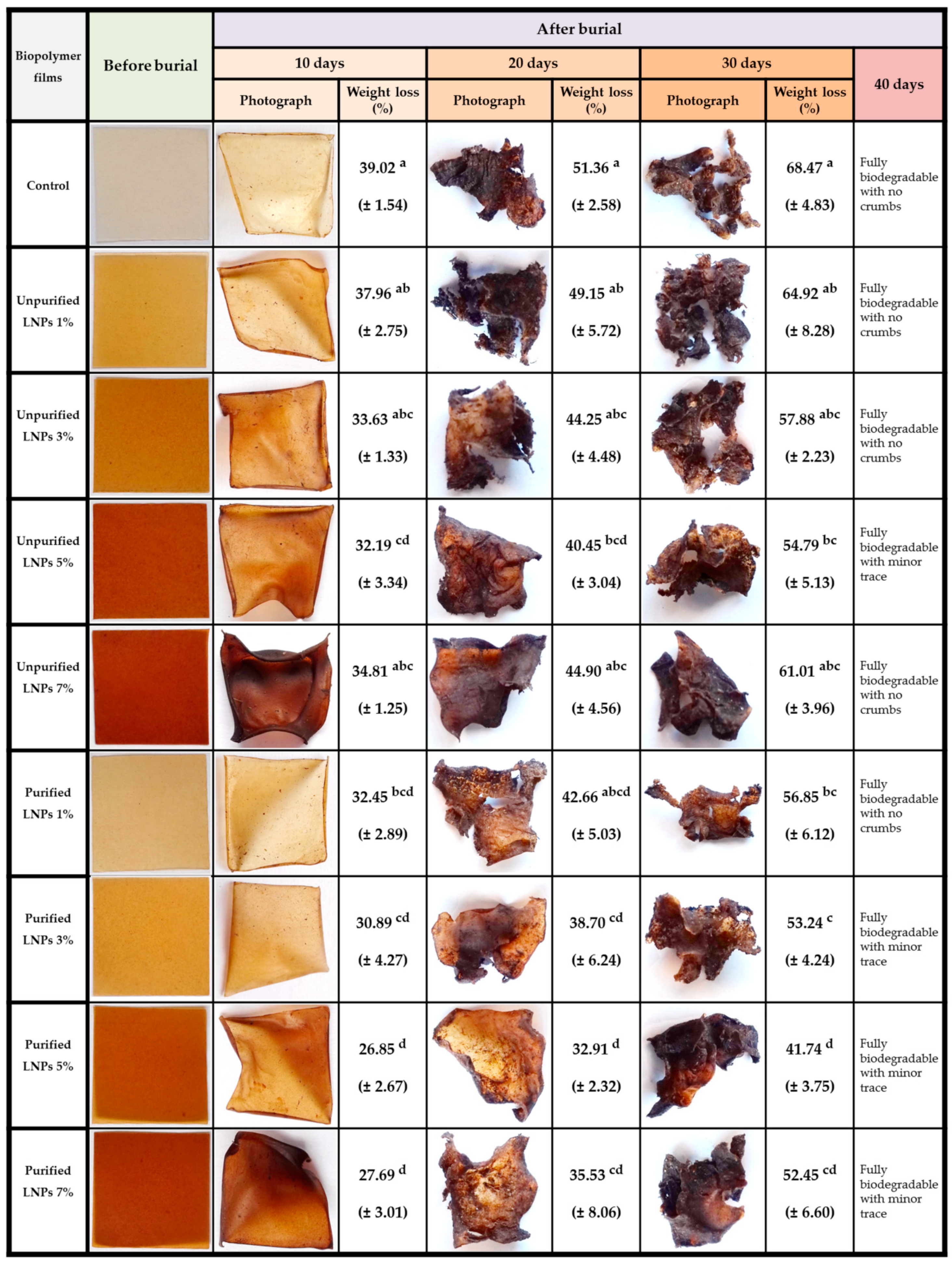
3.2.5. Biodegradability Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McAdam, B.; Fournet, M.B.; McDonald, P.; Mojicevic, M. Production of Polyhydroxybutyrate (PHB) and Factors Impacting Its Chemical and Mechanical Characteristics. Polymers 2020, 12, 2908. [Google Scholar] [CrossRef] [PubMed]
- Vetcher, A.; Hyun Kim, S.; Yeon Cho, J.; Hyun Cho, D.; Ju Jung, H.; Chan Kim, B.; Kant Bhatia, S.; Park, S.-H.; Park, K.; Yang, Y.-H. Acceleration of Polybutylene Succinate Biodegradation by Terribacillus sp. JY49 Isolated from a Marine Environment. Polymers 2022, 14, 3978. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Bashir Yahya, E.; Jummaat, F.; Adnan, A.S.; Olaiya, N.G.; Rizal, S.; Abdullah, C.K.; Pasquini, D.; Thomas, S. Biopolymers based aerogels: A review on revolutionary solutions for smart therapeutics delivery. Prog. Mater. Sci. 2023, 131, 101014. [Google Scholar] [CrossRef]
- Motelica, L.; Ficai, D.; Ficai, A.; Truşcă, R.D.; Ilie, C.I.; Oprea, O.C.; Andronescu, E. Innovative Antimicrobial Chitosan/ZnO/Ag NPs/Citronella Essential Oil Nanocomposite—Potential Coating for Grapes. Foods 2020, 9, 1801. [Google Scholar] [CrossRef] [PubMed]
- Motelica, L.; Ficai, D.; Oprea, O.; Ficai, A.; Trusca, R.D.; Andronescu, E.; Holban, A.M. Biodegradable Alginate Films with ZnO Nanoparticles and Citronella Essential Oil—A Novel Antimicrobial Structure. Pharmaceutics 2021, 13, 1020. [Google Scholar] [CrossRef] [PubMed]
- Motelica, L.; Ficai, D.; Oprea, O.C.; Ficai, A.; Ene, V.L.; Vasile, B.S.; Andronescu, E.; Holban, A.M. Antibacterial Biodegradable Films Based on Alginate with Silver Nanoparticles and Lemongrass Essential Oil–Innovative Packaging for Cheese. Nanomaterials 2021, 11, 2377. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Gopakumar, D.A.; Olaiya, N.G.; Zarlaida, F.; Alfian, A.; Aprinasari, C.; Alfatah, T.; Rizal, S.; Abdul Khalil, H.P.S. Evaluation of the thermomechanical properties and biodegradation of brown rice starch-based chitosan biodegradable composite films. Int. J. Biol. Macromol. 2020, 156, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.; Abdul Khalil, H.P.S.; Sabaruddin, F.A.; Bairwan, R.D.; Oyekanmi, A.A.; Alfatah, T.; Danish, M.; Mistar, E.M.; Abdullah, C.K. Microbial treatment for nanocellulose extraction from marine algae and its applications as sustainable functional material. Bioresour. Technol. Rep. 2021, 16, 100811. [Google Scholar] [CrossRef]
- Baranwal, J.; Barse, B.; Fais, A.; Delogu, G.L.; Kumar, A. Biopolymer: A Sustainable Material for Food and Medical Applications. Polymers 2022, 14, 983. [Google Scholar] [CrossRef]
- Magalhães Júnior, A.I.; Soccol, C.R.; Camara, M.C.; Molina Aulestia, D.T.; Porto de Souza Vandenberghe, L.; Cesar de Carvalho, J. Challenges in the production of second-generation organic acids (potential monomers for application in biopolymers). Biomass Bioenergy 2021, 149, 106092. [Google Scholar] [CrossRef]
- Muneer, F.; Nadeem, H.; Arif, A.; Zaheer, W. Bioplastics from Biopolymers: An Eco-Friendly and Sustainable Solution of Plastic Pollution. Polym. Sci. Ser. C 2021, 63, 47–63. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Chong, E.W.N.; Owolabi, F.A.T.; Asniza, M.; Tye, Y.Y.; Tajarudin, H.A.; Paridah, M.T.; Rizal, S. Microbial-induced CaCO3 filled seaweed-based film for green plasticulture application. J. Clean. Prod. 2018, 199, 150–163. [Google Scholar] [CrossRef]
- Rafiquzzaman, S.M.; Ahmed, R.; Lee, J.M.; Noh, G.; Jo, G.a.; Kong, I.S. Improved methods for isolation of carrageenan from Hypnea musciformis and its antioxidant activity. J. Appl. Phycol. 2015, 28, 1265–1274. [Google Scholar] [CrossRef]
- Uthaya Kumar, U.S.; Abdulmadjid, S.N.; Olaiya, N.G.; Amirul, A.A.; Rizal, S.; Rahman, A.A.; Alfatah, T.; Mistar, E.M.; Abdul Khalil, H.P.S. Extracted Compounds from Neem Leaves as Antimicrobial Agent on the Physico-Chemical Properties of Seaweed-Based Biopolymer Films. Polymers 2020, 12, 1119. [Google Scholar] [CrossRef]
- Lavrič, G.; Oberlintner, A.; Filipova, I.; Novak, U.; Likozar, B.; Vrabič-Brodnjak, U. Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials. Polymers 2021, 13, 2523. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Lai, T.K.; Gopakumar, D.A.; Jawaid, M.; Owolabi, F.A.T.; Mistar, E.M.; Alfatah, T.; Noriman, N.Z.; Haafiz, M.K.M.; Abdul Khalil, H.P.S. Micro Crystalline Bamboo Cellulose Based Seaweed Biodegradable Composite Films for Sustainable Packaging Material. J. Polym. Environ. 2019, 27, 1602–1612. [Google Scholar] [CrossRef]
- Uthaya Kumar, U.S.; Paridah, M.T.; Owolabi, F.A.T.; Gopakumar, D.A.; Rizal, S.; Amirul, A.A.; Rahman, A.A.; Alfatah, T.; Mistar, E.M.; Aprilia, N.A.S.; et al. Neem leaves extract based seaweed bio-degradable composite films with excellent antimicrobial activity for sustainable packaging material. BioResources 2019, 14, 700–713. [Google Scholar] [CrossRef]
- Mariana, M.; Alfatah, T.; Abdul Khalil, H.P.S.; Yahya, E.B.; Olaiya, N.G.; Nuryawan, A.; Mistar, E.M.; Abdullah, C.K.; Abdulmadjid, S.N.; Ismail, H. A current advancement on the role of lignin as sustainable reinforcement material in biopolymeric blends. J. Mater. Res. Technol. 2021, 15, 2287–2316. [Google Scholar] [CrossRef]
- Guo, J.; Chen, X.; Wang, J.; He, Y.; Xie, H.; Zheng, Q. The Influence of Compatibility on the Structure and Properties of PLA/Lignin Biocomposites by Chemical Modification. Polymers 2019, 12, 56. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Meng, X.; Pu, Y.; Ragauskas, A.J. Recent Advances in the Application of Functionalized Lignin in Value-Added Polymeric Materials. Polymers 2020, 12, 2277. [Google Scholar] [CrossRef] [PubMed]
- Gordobil, O.; Diaz, R.H.; Sandak, J.; Sandak, A. One-Step Lignin Refining Process: The Influence of the Solvent Nature on the Properties and Quality of Fractions. Polymers 2022, 14, 2363. [Google Scholar] [CrossRef] [PubMed]
- Ganewatta, M.S.; Lokupitiya, H.N.; Tang, C. Lignin Biopolymers in the Age of Controlled Polymerization. Polymers 2019, 11, 1176. [Google Scholar] [CrossRef] [PubMed]
- Narapakdeesakul, D.; Sridach, W.; Wittaya, T. Recovery, characteristics and potential use as linerboard coatings material of lignin from oil palm empty fruit bunches’ black liquor. Ind. Crops Prod. 2013, 50, 8–14. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Marliana, M.M.; Issam, A.M.; Bakare, I.O. Exploring isolated lignin material from oil palm biomass waste in green composites. Mater. Des. 2011, 32, 2604–2610. [Google Scholar] [CrossRef]
- Jafarzadeh, S.; Alias, A.K.; Ariffin, F.; Mahmud, S. Physico-mechanical and microstructural properties of semolina flour films as influenced by different sorbitol/glycerol concentrations. Int. J. Food Prop. 2018, 21, 983–995. [Google Scholar] [CrossRef]
- Ma, M.; Dai, L.; Xu, J.; Liu, Z.; Ni, Y. A simple and effective approach to fabricate lignin nanoparticles with tunable sizes based on lignin fractionation. Green Chem. 2020, 22, 2011–2017. [Google Scholar] [CrossRef]
- Majdar, R.E.; Ghasemian, A.; Resalati, H.; Saraeian, A.; Crestini, C.; Lange, H. Facile Isolation of LCC-Fraction from Organosolv Lignin by Simple Soxhlet Extraction. Polymers 2019, 11, 225. [Google Scholar] [CrossRef]
- Amin, M.R.; Chowdhury, M.A.; Kowser, M.A. Characterization and performance analysis of composite bioplastics synthesized using titanium dioxide nanoparticles with corn starch. Heliyon 2019, 5, e02009. [Google Scholar] [CrossRef]
- Gomide, R.A.C.; de Oliveira, A.C.S.; Rodrigues, D.A.C.; de Oliveira, C.R.; de Assis, O.B.G.; Dias, M.V.; Borges, S.V. Development and Characterization of Lignin Microparticles for Physical and Antioxidant Enhancement of Biodegradable Polymers. J. Polym. Environ. 2020, 28, 1326–1334. [Google Scholar] [CrossRef]
- Ravishankar, K.; Venkatesan, M.; Desingh, R.P.; Mahalingam, A.; Sadhasivam, B.; Subramaniyam, R.; Dhamodharan, R. Biocompatible hydrogels of chitosan-alkali lignin for potential wound healing applications. Mater. Sci. Eng. C 2019, 102, 447–457. [Google Scholar] [CrossRef]
- Chen, M.; Tang, Z.; Wang, Y.; Shi, J.; Li, C.; Yang, Z.; Wang, J. Catalytic depolymerization of Kraft lignin to liquid fuels and guaiacol over phosphorus modified Mo/Sepiolite catalyst. Chem. Eng. J. 2022, 427, 131761. [Google Scholar] [CrossRef]
- Yan, Z.; Liao, G.; Zou, X.; Zhao, M.; Wu, T.; Chen, Y.; Fang, G. Size-Controlled and Super Long-Term Stable Lignin Nanospheres through a Facile Self-Assembly Strategy from Kraft Lignin. J. Agric. Food Chem. 2020, 68, 8341–8349. [Google Scholar] [CrossRef] [PubMed]
- Oyekanmi, A.A.; Abdul Khalil, H.P.S.; Rahman, A.A.; Mistar, E.M.; Olaiya, N.G.; Alfatah, T.; Yahya, E.B.; Mariana, M.; Hazwan, C.M.; Abdullah, C.K. Extracted supercritical CO2 cinnamon oil functional properties enhancement in cellulose nanofibre reinforced Euchema cottoni biopolymer films. J. Mater. Res. Technol. 2021, 15, 4293–4308. [Google Scholar] [CrossRef]
- Jiang, F.; Yao, Y.; Natarajan, B.; Yang, C.; Gao, T.; Xie, H.; Wang, Y.; Xu, L.; Chen, Y.; Gilman, J.; et al. Ultrahigh-temperature conversion of biomass to highly conductive graphitic carbon. Carbon N. Y. 2019, 144, 241–248. [Google Scholar] [CrossRef]
- Hu, C.; Zhou, Y.; Zhang, T.; Jiang, T.; Zeng, G. Effect of fiber modified by alkali/polyvinyl alcohol coating treatment on properties of sisal fiber plastic composites. J. Reinf. Plast. Compos. 2020, 39, 880–889. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Wongphan, P.; Promhuad, K.; Promsorn, J.; Harnkarnsujarit, N. Blown film extrusion of PBAT/TPS/ZnO nanocomposites for shelf-life extension of meat packaging. Colloids Surf. B Biointerfaces 2022, 214, 112472. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, E.M.; O’Keefe, S.F.; Kim, Y.T. Utilization of Lignin in Biopolymeric Packaging Films. ACS Omega 2018, 3, 7388–7398. [Google Scholar] [CrossRef] [PubMed]
- Marques de Farias, P.; Barros de Vasconcelos, L.; da Silva Ferreira, M.E.; Alves Filho, E.G.; De Freitas, V.A.A.; Tapia-Blácido, D.R. Nopal cladode as a novel reinforcing and antioxidant agent for starch-based films: A comparison with lignin and propolis extract. Int. J. Biol. Macromol. 2021, 183, 614–626. [Google Scholar] [CrossRef] [PubMed]
- Ismail, G.A.; Hessien, M.; Saleh, M.A.; Ismail, M.M. Antioxidant and Antiviral Activity of Sulfated Polysaccharides Derived from Two Sargassum Species of Red Sea Egypt. J. Biol. Act. Prod. Nat. 2022, 12, 324–343. [Google Scholar] [CrossRef]
- Jancikova, S.; Dordevic, D.; Jamroz, E.; Behalova, H.; Tremlova, B. Chemical and Physical Characteristics of Edible Films, Based on κ- and ι-Carrageenans with the Addition of Lapacho Tea Extract. Foods 2020, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Martiny, T.R.; Pacheco, B.S.; Pereira, C.M.P.; Mansilla, A.; Astorga–España, M.S.; Dotto, G.L.; Moraes, C.C.; Rosa, G.S. A novel biodegradable film based on κ-carrageenan activated with olive leaves extract. Food Sci. Nutr. 2020, 8, 3147–3156. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, S.; Mokhtari, H.; Kharaziha, M.; Kermanpur, A.; Talebi, A.; Moshtaghian, J. A multifunctional nanocomposite spray dressing of Kappa-carrageenan-polydopamine modified ZnO/L-glutamic acid for diabetic wounds. Mater. Sci. Eng. C 2020, 111, 110837. [Google Scholar] [CrossRef] [PubMed]
- Gasilova, E.R.; Aleksandrova, G.P.; Volchek, B.Z.; Vlasova, E.N.; Baigildin, V.A. Smart colloids containing ensembles of gold nanoparticles conjugated with κ-carrageenan. Int. J. Biol. Macromol. 2019, 137, 358–365. [Google Scholar] [CrossRef]
- Guo, J.; Shi, F.; Sun, M.; Ma, F.; Li, Y. Antioxidant and aflatoxin B1 adsorption properties of Eucheuma cottonii insoluble dietary fiber. Food Biosci. 2022, 50, 102043. [Google Scholar] [CrossRef]
- Sedayu, B.B.; Cran, M.J.; Bigger, S.W. Reinforcement of Refined and Semi-Refined Carrageenan Film with Nanocellulose. Polymers 2020, 12, 1145. [Google Scholar] [CrossRef] [PubMed]
- Barral-Martínez, M.; Flórez-Fernández, N.; Domínguez, H.; Torres, M.D. Tailoring hybrid carrageenans from Mastocarpus stellatus red seaweed using microwave hydrodiffusion and gravity. Carbohydr. Polym. 2020, 248, 116830. [Google Scholar] [CrossRef]
- Yang, W.; Qi, G.; Kenny, J.M.; Puglia, D.; Ma, P. Effect of Cellulose Nanocrystals and Lignin Nanoparticles on Mechanical, Antioxidant and Water Vapour Barrier Properties of Glutaraldehyde Crosslinked PVA Films. Polymers 2020, 12, 1364. [Google Scholar] [CrossRef]
- Wang, N.; Zhang, C.; Weng, Y. Enhancing gas barrier performance of polylactic acid/lignin composite films through cooperative effect of compatibilization and nucleation. J. Appl. Polym. Sci. 2021, 138, 50199. [Google Scholar] [CrossRef]
- Shao, H.; Zhang, Y.; Pan, H.; Jiang, Y.; Qi, J.; Xiao, H.; Zhang, S.; Lin, T.; Tu, L.; Xie, J. Preparation of flexible and UV-blocking films from lignin-containing cellulose incorporated with tea polyphenol/citric acid. Int. J. Biol. Macromol. 2022, 207, 917–926. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, S.; Wang, W.; Linhardt, R.J.; Ragauskas, A.J. Preparation of Highly Reactive Lignin by Ozone Oxidation: Application as Surfactants with Antioxidant and Anti-UV Properties. ACS Sustain. Chem. Eng. 2020, 8, 22–28. [Google Scholar] [CrossRef]
- Roostazadeh, R.; Behzad, T.; Karimi, K. Isolation and characterization of lignin-rich particles as byproducts of bioethanol production from wheat straw to reinforce starch composite films. Ind. Crops Prod. 2022, 186, 115175. [Google Scholar] [CrossRef]
- Su, W.; Yang, Z.; Wang, H.; Fang, J.; Li, C.; Lyu, G.; Li, H. Synergistic Effect of Sodium Alginate and Lignin on the Properties of Biodegradable Poly(vinyl alcohol) Mulch Films. ACS Sustain. Chem. Eng. 2022, 10, 11800–11814. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, W.; Liu, W.; Qiu, X. High performance PVA/lignin nanocomposite films with excellent water vapor barrier and UV-shielding properties. Int. J. Biol. Macromol. 2020, 142, 551–558. [Google Scholar] [CrossRef]
- Michelin, M.; Marques, A.M.; Pastrana, L.M.; Teixeira, J.A.; Cerqueira, M.A. Carboxymethyl cellulose-based films: Effect of organosolv lignin incorporation on physicochemical and antioxidant properties. J. Food Eng. 2020, 285, 110107. [Google Scholar] [CrossRef]
- Rizal, S.; Alfatah, T.; Abdul Khalil, H.P.S.; Mistar, E.M.; Abdullah, C.K.; Olaiya, F.G.; Sabaruddin, F.A.; Ikramullah; Muksin, U. Properties and Characterization of Lignin Nanoparticles Functionalized in Macroalgae Biopolymer Films. Nanomaterials 2021, 11, 637. [Google Scholar] [CrossRef]
- Zou, Z.; Ismail, B.B.; Zhang, X.; Yang, Z.; Liu, D.; Guo, M. Improving barrier and antibacterial properties of chitosan composite films by incorporating lignin nanoparticles and acylated soy protein isolate nanogel. Food Hydrocoll. 2023, 134, 108091. [Google Scholar] [CrossRef]
- Sedayu, B.B.; Cran, M.J.; Bigger, S.W. Improving the moisture barrier and mechanical properties of semi-refined carrageenan films. J. Appl. Polym. Sci. 2020, 137, 49238. [Google Scholar] [CrossRef]
- Kalinoski, R.M.; Li, W.; Mobley, J.K.; Asare, S.O.; Dorrani, M.; Lynn, B.C.; Chen, X.; Shi, J. Antimicrobial Properties of Corn Stover Lignin Fractions Derived from Catalytic Transfer Hydrogenolysis in Supercritical Ethanol with a Ru/C Catalyst. ACS Sustain. Chem. Eng. 2020, 8, 18455–18467. [Google Scholar] [CrossRef]
- Allegretti, C.; Fontanay, S.; Rischka, K.; Strini, A.; Troquet, J.; Turri, S.; Griffini, G.; D’arrigo, P. Lignin Nanoparticles and Their Nanocomposites. Nanomaterials 2021, 11, 1336. [Google Scholar] [CrossRef]
- Chen, K.; Lei, L.; Lou, H.; Niu, J.; Yang, D.; Qiu, X.; Qian, Y. High internal phase emulsions stabilized with carboxymethylated lignin for encapsulation and protection of environmental sensitive natural extract. Int. J. Biol. Macromol. 2020, 158, 430–442. [Google Scholar] [CrossRef]
- Lourençon, T.V.; de Lima, G.G.; Ribeiro, C.S.P.; Hansel, F.A.; Maciel, G.M.; da Silva, K.; Winnischofer, S.M.B.; de Muniz, G.I.B.; Magalhães, W.L.E. Antioxidant, antibacterial and antitumoural activities of kraft lignin from hardwood fractionated by acid precipitation. Int. J. Biol. Macromol. 2021, 166, 1535–1542. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, Y.; Wu, Q.; Huang, J.; Zhao, Y.; Li, Q.; Wang, S. Improved Hydrophobic, UV Barrier and Antibacterial Properties of Multifunctional PVA Nanocomposite Films Reinforced with Modified Lignin Contained Cellulose Nanofibers. Polymers 2022, 14, 1705. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.E.; Alzagameem, A.; Rumpf, J.; Korte, I.; Kreyenschmidt, J.; Schulze, M. Antimicrobial Activity of Lignin-Derived Polyurethane Coatings Prepared from Unmodified and Demethylated Lignins. Coatings 2019, 9, 494. [Google Scholar] [CrossRef]
- El Bouchtaoui, F.Z.; Ablouh, E.H.; Mhada, M.; Kassem, I.; Salim, M.H.; Mouhib, S.; Kassab, Z.; Sehaqui, H.; El Achaby, M. Methylcellulose/lignin biocomposite as an eco-friendly and multifunctional coating material for slow-release fertilizers: Effect on nutrients management and wheat growth. Int. J. Biol. Macromol. 2022, 221, 398–415. [Google Scholar] [CrossRef]
- Chen, S.; Chen, H.; Yang, S.; Fan, D. Developing an antifungal and high-strength soy protein-based adhesive modified by lignin-based polymer. Ind. Crops Prod. 2021, 170, 113795. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizal, S.; Alfatah, T.; Abdul Khalil, H.P.S.; Yahya, E.B.; Abdullah, C.K.; Mistar, E.M.; Ikramullah, I.; Kurniawan, R.; Bairwan, R.D. Enhanced Functional Properties of Bioplastic Films Using Lignin Nanoparticles from Oil Palm-Processing Residue. Polymers 2022, 14, 5126. https://doi.org/10.3390/polym14235126
Rizal S, Alfatah T, Abdul Khalil HPS, Yahya EB, Abdullah CK, Mistar EM, Ikramullah I, Kurniawan R, Bairwan RD. Enhanced Functional Properties of Bioplastic Films Using Lignin Nanoparticles from Oil Palm-Processing Residue. Polymers. 2022; 14(23):5126. https://doi.org/10.3390/polym14235126
Chicago/Turabian StyleRizal, Samsul, Tata Alfatah, H. P. S. Abdul Khalil, Esam Bashir Yahya, C. K. Abdullah, Eka Marya Mistar, Ikramullah Ikramullah, Rudi Kurniawan, and R. D. Bairwan. 2022. "Enhanced Functional Properties of Bioplastic Films Using Lignin Nanoparticles from Oil Palm-Processing Residue" Polymers 14, no. 23: 5126. https://doi.org/10.3390/polym14235126
APA StyleRizal, S., Alfatah, T., Abdul Khalil, H. P. S., Yahya, E. B., Abdullah, C. K., Mistar, E. M., Ikramullah, I., Kurniawan, R., & Bairwan, R. D. (2022). Enhanced Functional Properties of Bioplastic Films Using Lignin Nanoparticles from Oil Palm-Processing Residue. Polymers, 14(23), 5126. https://doi.org/10.3390/polym14235126