Direct Synthesis of HKUST-1 onto Cotton Fabrics and Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. In Situ Synthesis of HKUST-1
2.2. Characterization
2.3. Mechanical Properties
2.4. Evaluation of Antibacterial Properties
2.4.1. Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
2.4.2. Bacterial Reduction
2.4.3. Time-Kill Kinetics Test
3. Results
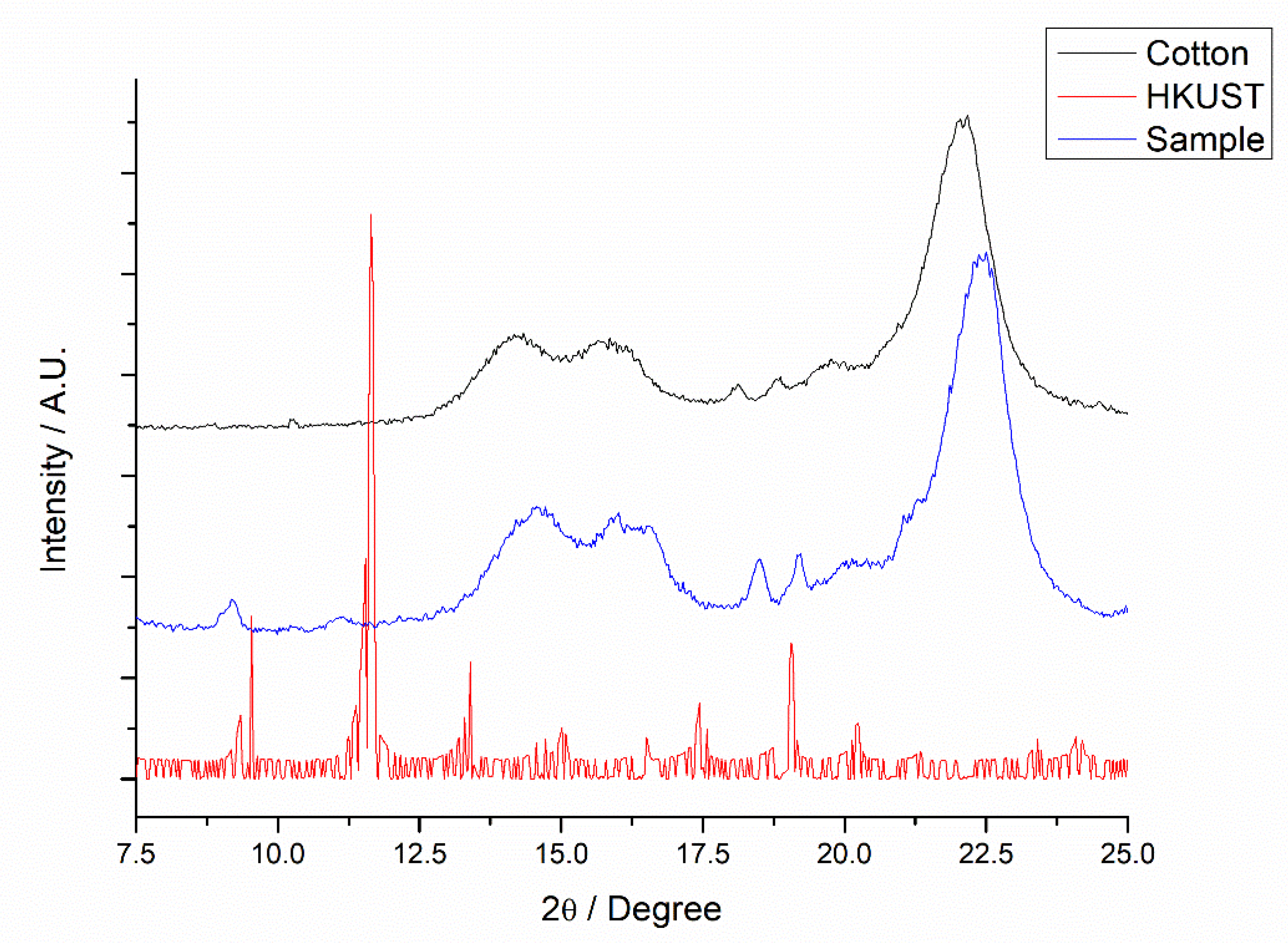
3.1. Characterization
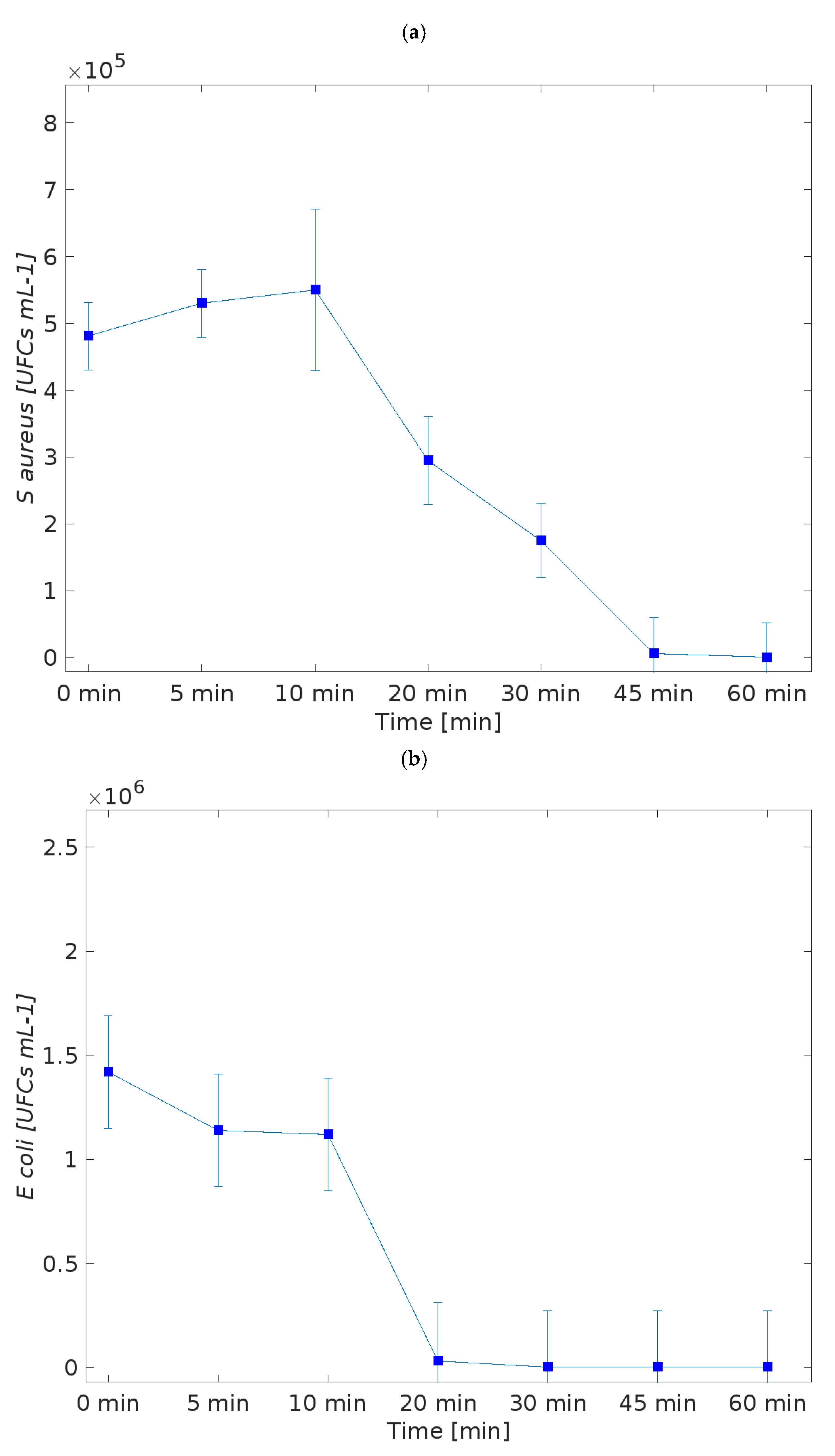
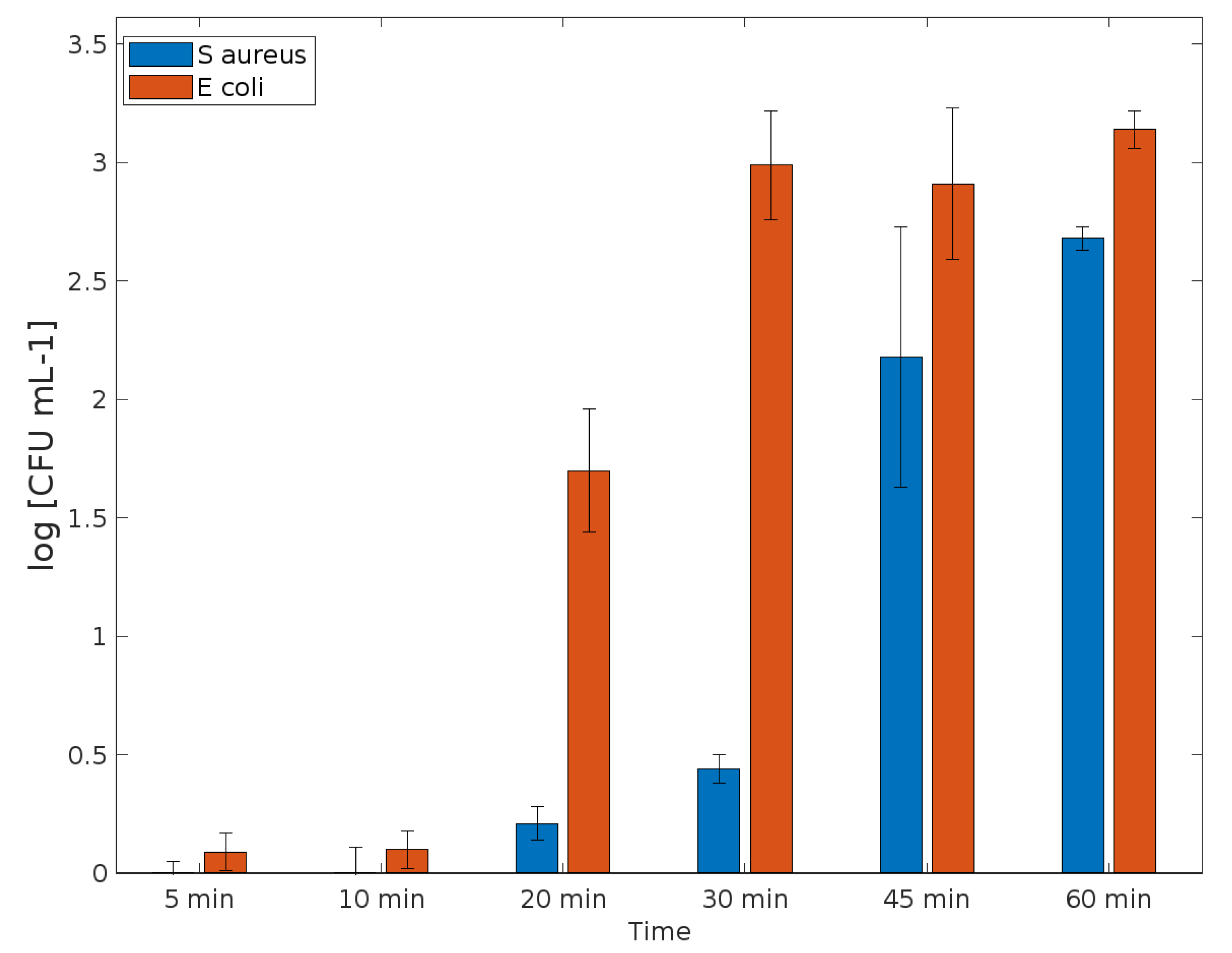
3.2. Evaluation of Antibacterial Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lobanovska, M.; Pilla, G. Focus: Drug development: Penicillin’s discovery and antibiotic resistance: Lessons for the future? Yale J. Biol. Med. 2017, 90, 135. [Google Scholar] [PubMed]
- Iwu, C.D.; Korsten, L.; Okoh, A.I. The incidence of antibiotic resistance within and beyond the agricultural ecosystem: A concern for public health. Microbiologyopen 2020, 9, 1035. [Google Scholar] [CrossRef] [PubMed]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.M.; Wertheim, H.F.L.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic resistance—The need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Al-Khattaf, F.S. Gold and silver nanoparticles: Green synthesis, microbes, mechanism, factors, plant disease management and environmental risks. Saudi J. Biol. Sci. 2021, 28, 3624–3631. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-López, E.; Gomes, D.; Esteruelas, G.; Bonilla, L.; Lopez-Machado, A.L.; Galindo, R.; Cano, A.; Espina, M.; Ettcheto, M.; Camins, A.; et al. Metal-based nanoparticles as antimicrobial agents: An overview. Nanomaterials 2020, 10, 292. [Google Scholar] [CrossRef]
- Nisar, P.; Ali, N.; Rahman, L.; Ali, M.; Shinwari, Z.K. Antimicrobial activities of biologically synthesized metal nanoparticles: An insight into the mechanism of action. J. Biol. Inorg. Chem. 2019, 24, 929–941. [Google Scholar] [CrossRef]
- Calamak, S.; Shahbazi, R.; Eroğlu, I.; Gultekinoglu, M.; Ulubayram, K. An overview of nanofiber-based antibacterial drug design. Expert Opin. Drug Discov. 2017, 12, 391–406. [Google Scholar] [CrossRef]
- Shahidi, S.; Wiener, J. Antibacterial agents in textile industry. In Antimicrobial Agents; Bobbarala, V., Ed.; IntechOpen: London, UK, 2012; pp. 387–406. [Google Scholar]
- Fouda, M.M. Antibacterial Modification of Textiles Using Nanotechnology; IntechOpen: London, UK, 2012. [Google Scholar]
- Hajipour, M.J.; Fromm, K.M.; Ashkarran, A.A.; de Aberasturi, D.J.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef]
- Thorsteinsson, T.; Loftsson, T.; Masson, M. Soft antibacterial agents. Curr. Med. Chem. 2003, 10, 1129–1136. [Google Scholar] [CrossRef]
- Periolatto, M.; Ferrero, F.; Vineis, C.; Varesano, A.; Gozzelino, A.V.A.G. Novel antimicrobial agents and processes for textile applications. In Antibacterial Agents; Kumavath, R., Ed.; IntechOpen: London, UK, 2017; p. 17. [Google Scholar]
- Madhav, S.; Ahamad, A.; Singh, P.; Mishra, P.K. A review of textile industry: Wet processing, environmental impacts, and effluent treatment methods. Environ. Qual. Manag. 2018, 27, 31–41. [Google Scholar] [CrossRef]
- Yetisen, A.K.; Qu, H.; Manbachi, A.; Butt, H.; Dokmeci, M.R.; Hinestroza, J.P.; Skorobogatiy, M.; Khademhosseini, A.; Yun, S.H. Nanotechnology in textiles. ACS Nano 2016, 10, 3042–3068. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; McGuirk, C.M.; d’Aquino, A.; Mason, J.A.; Mirkin, C.A. Metal–organic framework nanoparticles. Adv. Mater. 2018, 30, 1800202. [Google Scholar] [CrossRef] [PubMed]
- Wyszogrodzka, G.; Marszałek, B.; Gil, B.; Dorożyński, P. Metal-organic frameworks: Mechanisms of antibacterial action and potential applications. Drug Discov. Today 2016, 21, 1009–1018. [Google Scholar] [CrossRef]
- Neufeld, M.J.; Harding, J.L.; Reynolds, M.M. Immobilization of metal–organic framework copper (II) benzene-1, 3, 5-tricarboxylate (CuBTC) onto cotton fabric as a nitric oxide release catalyst. ACS Appl. Mater. Interfaces 2015, 7, 26742–26750. [Google Scholar] [CrossRef]
- Textile Exchange. Preferred Fiber & Materials Market Report 2019; Textile Exchange: Lamesa, TX, USA, 2019. [Google Scholar]
- Woolridge, A.C.; Ward, G.D.; Phillips, P.S.; Collins, M.; Gandy, S. Life cycle assessment for reuse/recycling of donated waste textiles compared to use of virgin material: An UK energy saving perspective. Resour. Conserv. Recycl. 2006, 46, 94–103. [Google Scholar] [CrossRef]
- Konwar, A.; Kandimalla, R.; Kalita, S.; Chowdhury, D. Approach to fabricate a compact cotton patch without weaving: A smart bandage material. ACS Sustain. Chem. Eng. 2018, 6, 5806–5817. [Google Scholar] [CrossRef]
- Wang, C.; Qian, X.; An, X. In situ green preparation and antibacterial activity of copper-based metal–organic frameworks/cellulose fibers (HKUST-1/CF) composite. Cellulose 2015, 22, 3789–3797. [Google Scholar] [CrossRef]
- Sadeghi-Kiakhani, M.; Tehrani-Bagha, A.R.; Gharanjig, K.; Hashemi, E. Use of pomegranate peels and walnut green husks as the green antimicrobial agents to reduce the consumption of inorganic nanoparticles on wool yarns. J. Clean. Prod. 2019, 231, 1463–1473. [Google Scholar] [CrossRef]
- Lis, M.J.; Caruzi, B.B.; Gil, G.A.; Samulewski, R.B.; Bail, A.; Scacchetti, F.A.P.; Moisés, M.P.; Bezerra, F.M. In-situ direct synthesis of hkust-1 in wool fabric for the improvement of antibacterial properties. Polymers 2019, 11, 713. [Google Scholar] [CrossRef]
- NBR ISO 105-C06; Têxteis—Ensaios de Solidez da Cor: Parte C06: Solidez da Cor à Lavagem Doméstica e Comercial. Associação Brasileira de Normas Técnicas (ABNT): Rio de Janeiro, Brazil, 2006.
- Yang, F.; Mu, H.; Wang, C.; Xiang, L.; Yao, K.X.; Liu, L.; Pan, Y. Morphological map of ZIF-8 crystals with five distinctive shapes: Feature of filler in mixed-matrix membranes on C3H6/C3H8 separation. Chem. Mater. 2018, 30, 3467–3473. [Google Scholar] [CrossRef]
- Tehrani, A.A.; Safarifard, V.; Morsali, A.; Bruno, G.; Rudbari, H.A. Ultrasound-assisted synthesis of metal–organic framework nanorods of Zn-HKUST-1 and their templating effects for facile fabrication of zinc oxide nanorods via solid-state transformation. Inorg. Chem. Commun. 2015, 59, 41–45. [Google Scholar] [CrossRef]
- Martínez, L.M.T.; Ivanova, S.; Louis, B.; Odriozola, J.A. Synthesis and identification methods for zeolites and MOFs. In Zeolites and Metal-Organic Frameworks; Blay, V., Bobadilla, L.F., Cabrera-García, A., Eds.; Atlantis Press: Dordrecht, The Netherlands, 2010; p. 25. [Google Scholar]
- Matthews, J.M. Laboratory Manual of Dyeing and Textile Chemistry; John Wiley and Sons: Hoboken, NJ, USA, 1909. [Google Scholar]
- Mather, R.R.; Wardman, R.H. The Chemistry of Textiles Fibres; Royal Society of Chemistry: Cambridge, UK, 2015. [Google Scholar]
- Lee, H.V.; Hamid, S.B.A.; Zain, S.K. Conversion of lignocellulosic biomass to nanocellulose: Structure and chemical process. Sci. World J. 2014, 2014, 631013. [Google Scholar] [CrossRef] [PubMed]
- Gascon, J.; Aguado, S.; Kapteijn, F. Manufacture of dense coatings of Cu3(BTC)2 (HKUST-1) on α-alumina. Microporous Mesoporous Mater. 2008, 113, 132–138. [Google Scholar] [CrossRef]
- Zamaro, J.M.; Pérez, N.C.; Miró, E.E.; Casado, C.; Seoane, B.; Téllez, C.; Coronas, J. HKUST-1 MOF: A matrix to synthesize CuO and CuO–CeO2 nanoparticle catalysts for CO oxidation. Chem. Eng. J. 2012, 195, 180–187. [Google Scholar] [CrossRef]
- Liu, Y.; Guo, B.; Xia, Q.; Meng, J.; Chen, W.; Liu, S.; Wang, Q.; Liu, Y.; Li, J.; Yu, H. Efficient cleavage of strong hydrogen bonds in cotton by deep eutectic solvents and facile fabrication of cellulose nanocrystals in high yields. ACS Sustain. Chem. Eng. 2017, 5, 7623–7631. [Google Scholar] [CrossRef]
- Abbasi, A.R.; Akhbari, K.; Morsali, A. Dense coating of surface mounted CuBTC metal–organic framework nanostructures on silk fibers, prepared by layer-by-layer method under ultrasound irradiation with antibacterial activity. Ultrason. Sonochem. 2012, 19, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, H.S.; Hinestroza, J.P.; Ochoa-Puentes, C.; Sierra, C.A.; Soto, C.Y. Antibacterial activity against Escherichia coli of Cu-BTC (MOF-199) metal-organic framework immobilized onto cellulosic fibers. J. Appl. Polym. Sci. 2014, 131, 40815. [Google Scholar] [CrossRef]
- Rickhoff, T.A.; Sullivan, E.; Werth, L.K.; Kissel, D.S.; Keleher, J. A biomimetic cellulose-based composite material that incorporates the antimicrobial metal-organic framework HKUST-1. J. Appl. Polym. Sci. 2019, 136, 46978. [Google Scholar] [CrossRef]

| Sample | Uniaxial Force (N) | Standard Deviation (N) |
|---|---|---|
| Blank | 280.80 | 68.86 |
| 1880 | 74.15 | 17.84 |
| 2480 | 63.78 | 7.73 |
| 4880 | 58.84 | 10.52 |
| 18100 | 45.29 | 3.98 |
| 24100 | 58.50 | 9.90 |
| 48100 | 10.97 | 4.25 |
| Sample | Cie Lab Coordinates | Blank Deviation | Color Difference | ||||
|---|---|---|---|---|---|---|---|
| L | a | b | ΔL | Δa | Δb | ΔE | |
| Blank | 89.19 ± 0.26 | −1.42 ± 0.16 | 0.14 ± 0.45 | − | − | − | − |
| 1880 | 87.87 ± 1.43 | −9.52 ± 1.59 | −0.69 ± 1.20 | −1.32 | −8.09 | −0.82 | 8.24 |
| 2480 | 85.72 ± 2.62 | −11.77 ± 3.81 | −3.68 ± 3.90 | −3.47 | −10.35 | −3.81 | 11.56 |
| 4880 | 87.82 ± 0.83 | −10.75 ± 2.13 | −1.38 ± 1.14 | −1.37 | −9.33 | −1.52 | 9.55 |
| 18,100 | 89.55 ± 1.41 | −4.88 ± 2.09 | 2.18 ± 2.01 | 0.36 | −3.46 | 2.05 | 4.04 |
| 24,100 | 88.18 ± 2.65 | −6.43 ± 1.82 | 1.00 ± 1.39 | −1.01 | −5.01 | 0.87 | 5.18 |
| 48,100 | 89.06 ± 1.22 | −4.72 ± 1.00 | 2.74 ± 1.12 | −0.12 | −3.30 | 2.61 | 4.21 |
| E. coli | S. aureus | |
|---|---|---|
| MIC | 78.125 μg mL−1 | 156.25 μg mL−1 |
| MBC | 5 mg mL−1 | >5 mg mL−1 |
| N. of Bacteria (CFU mL−1) at 0 h (Initial Time) | N. of Bacteria (CFU mL−1) at 1 h (Dynamic Contact) | Log10 Reduction | |
|---|---|---|---|
| E. coli | 1.42 × 106 | 1.0 × 103 | 3.14 ± 0.12 |
| S. aureus | 5.31 × 105 | 1.0 × 103 | 2.68 ± 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Costa, B.L.; Rosa, I.L.A.A.; Silva, V.H.; Wu, Q.; Samulewski, R.B.; Scacchetti, F.A.P.; Moisés, M.P.; Lis, M.J.; Bezerra, F.M. Direct Synthesis of HKUST-1 onto Cotton Fabrics and Properties. Polymers 2022, 14, 4256. https://doi.org/10.3390/polym14204256
da Costa BL, Rosa ILAA, Silva VH, Wu Q, Samulewski RB, Scacchetti FAP, Moisés MP, Lis MJ, Bezerra FM. Direct Synthesis of HKUST-1 onto Cotton Fabrics and Properties. Polymers. 2022; 14(20):4256. https://doi.org/10.3390/polym14204256
Chicago/Turabian Styleda Costa, Braian Lobo, Isadora Letícia Aparecida Ataide Rosa, Vitória Hipolito Silva, Qiuyue Wu, Rafael Block Samulewski, Fabio Alexandre Pereria Scacchetti, Murilo Pereira Moisés, Manuel J. Lis, and Fabricio Maestá Bezerra. 2022. "Direct Synthesis of HKUST-1 onto Cotton Fabrics and Properties" Polymers 14, no. 20: 4256. https://doi.org/10.3390/polym14204256
APA Styleda Costa, B. L., Rosa, I. L. A. A., Silva, V. H., Wu, Q., Samulewski, R. B., Scacchetti, F. A. P., Moisés, M. P., Lis, M. J., & Bezerra, F. M. (2022). Direct Synthesis of HKUST-1 onto Cotton Fabrics and Properties. Polymers, 14(20), 4256. https://doi.org/10.3390/polym14204256










