Influence of Ulluco Starch Modified by Annealing on the Physicochemical Properties of Biodegradable Films
Abstract
1. Introduction
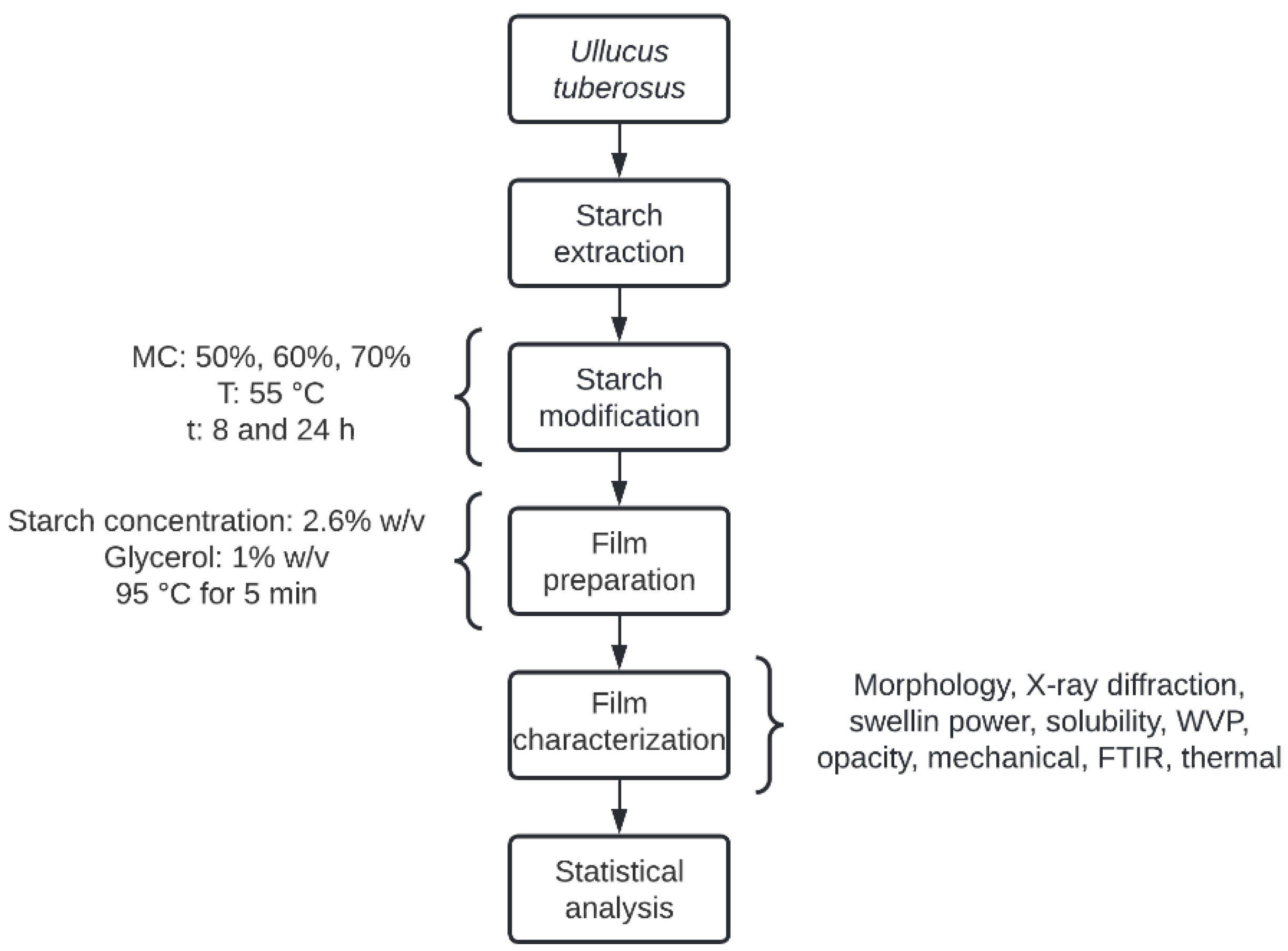
2. Materials and Methods
2.1. Raw Material and Starch Extraction
2.2. Film Preparation
2.3. Morphology of Edible Films
2.4. X-ray Diffraction (XRD) of Edible Films
2.5. Swelling Power and Solubility
2.6. Water Vapor Permeability
2.7. Opacity
2.8. Mechanical Properties
2.9. Attenuated Total Reflectance-Fourier Transform Infrared Spectroscopy (ATR-FTIR)
2.10. Thermal Analysis of Starch and Edible Films
2.11. Statistical Analysis
3. Results
3.1. Morphology of Biodegradable Films
3.2. X-ray Diffraction (XRD) of Edible Films
3.3. Solubility and Swelling Power of Biodegradable Films
3.4. Water Vapor Permeability of the Films
3.5. Opacity
3.6. Mechanical Properties
3.7. Attenuated Total Reflectance-Fourier Transform Infrared Spectroscopy (ATR-FTIR)
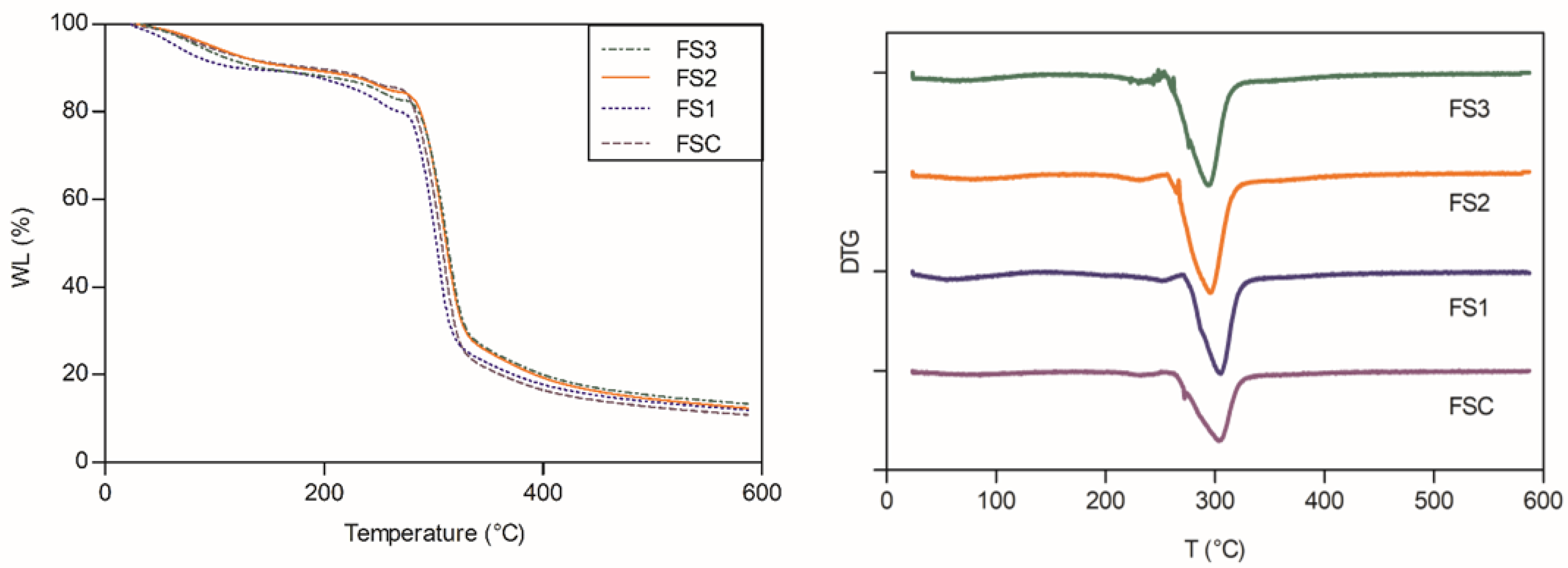
3.8. Thermal Analysis of Starch and Edible Films
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Plastics Europe Plastics—The Facts 2021, An Analysis of European Plastics Production, Demand and Waste Data. 2020. Available online: https://issuu.com/plasticseuropeebook/docs/plastics_the_facts-web-dec2020 (accessed on 1 September 2022).
- Rafey, A.; Siddiqui, F.Z. A Review of Plastic Waste Management in India–Challenges and Opportunities. Int. J. Environ. Anal. Chem. 2021. [Google Scholar] [CrossRef]
- Cheng, H.; Chen, L.; McClements, D.J.; Yang, T.; Zhang, Z.; Ren, F.; Miao, M.; Tian, Y.; Jin, Z. Starch-Based Biodegradable Packaging Materials: A Review of Their Preparation, Characterization and Diverse Applications in the Food Industry. Trends Food Sci. Technol. 2021, 114, 70–82. [Google Scholar] [CrossRef]
- Jiang, T.; Duan, Q.; Zhu, J.; Liu, H.; Yu, L. Starch-Based Biodegradable Materials: Challenges and Opportunities. Adv. Ind. Eng. Polym. Res. 2020, 3, 8–18. [Google Scholar] [CrossRef]
- Bhargava, N.; Sharanagat, V.S.; Mor, R.S.; Kumar, K. Active and Intelligent Biodegradable Packaging Films Using Food and Food Waste-Derived Bioactive Compounds: A Review. Trends Food Sci. Technol. 2020, 105, 385–401. [Google Scholar] [CrossRef]
- Henning, F.G.; Ito, V.C.; Demiate, I.M.; Lacerda, L.G. Non-conventional starches for biodegradable films: A review focussing on characterisation and recent applications in food. Carbohydr. Polym. Technol. Appl. 2021, 100157. [Google Scholar] [CrossRef]
- Parra, D.O.; Daza Ramírez, L.D.; Sandoval-Aldana, A.; Eim, V.S.; Váquiro, H.A. Annealing Treatment of Ulluco Starch: Effect of Moisture Content and Time on the Physicochemical Properties. J. Food Process. Preserv. 2022, 1–11. [Google Scholar] [CrossRef]
- Fonseca, L.M.; El Halal, S.L.M.; Dias, A.R.G.; Zavareze, E.d.R. Physical Modification of Starch by Heat-Moisture Treatment and Annealing and Their Applications: A Review. Carbohydr. Polym. 2021, 274. [Google Scholar] [CrossRef]
- Galindez, A.; Daza, L.D.; Homez-Jara, A.; Eim, V.S.; Váquiro, H.A. Characterization of Ulluco Starch and Its Potential for Use in Edible Films Prepared at Low Drying Temperature. Carbohydr. Polym. 2019, 215, 143–150. [Google Scholar] [CrossRef]
- Daza, L.D.; Homez-Jara, A.; Solanilla, J.F.; Váquiro, H.A. Effects of Temperature, Starch Concentration, and Plasticizer Concentration on the Physical Properties of Ulluco (Ullucus Tuberosus Caldas)-Based Edible Films. Int. J. Biol. Macromol. 2018, 120, 1834–1845. [Google Scholar] [CrossRef]
- Daza, L.D.; Eim, V.S.; Váquiro, H.A. Influence of Ulluco Starch Concentration on the Physicochemical Properties of Starch–Chitosan Biocomposite Films. Polymers 2021, 13, 4232. [Google Scholar] [CrossRef]
- Valcárcel-Yamani, B.; Rondán-Sanabria, G.G.; Finardi-Filho, F. The Physical, Chemical and Functional Characterization of Starches from Andean Tubers: Oca (Oxalis Tuberosa Molina), Olluco (Ullucus Tuberosus Caldas) and Mashua (Tropaeolum Tuberosum Ruiz & Pavón). Braz. J. Pharm. Sci. 2013, 49, 453–464. [Google Scholar] [CrossRef]
- Feijoo, P.; Samaniego-Aguilar, K.; Sánchez-Safont, E.; Torres-Giner, S.; Lagaron, J.M.; Gamez-Perez, J.; Cabedo, L. Development and Characterization of Fully Renewable and Biodegradable Polyhydroxyalkanoate Blends with Improved Thermoformability. Polymers 2022, 14, 2527. [Google Scholar] [CrossRef] [PubMed]
- Baron, R.D.; Pérez, L.L.; Salcedo, J.M.; Córdoba, L.P.; Sobral, P.J.d.A. Production and Characterization of Films Based on Blends of Chitosan from Blue Crab (Callinectes Sapidus) Waste and Pectin from Orange (Citrus Sinensis Osbeck) Peel. Int. J. Biol. Macromol. 2017, 98, 676–683. [Google Scholar] [CrossRef] [PubMed]
- E96/E96M-16; Standard Test Methods for Water Vapor Transmission of Materials. Annual Book of ASTM Standards; American Society for Testing and Materials: West Conshohocken, PA, USA, 2016; pp. 719–725.
- D882-18; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM International: West Conshohocken, PA, USA, 2018; Volume 8, pp. 182–190.
- Tavares, K.M.; de Campos, A.; Luchesi, B.R.; Resende, A.A.; de Oliveira, J.E.; Marconcini, J.M. Effect of Carboxymethyl Cellulose Concentration on Mechanical and Water Vapor Barrier Properties of Corn Starch Films. Carbohydr. Polym. 2020, 246, 116521. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liao, L.; Liu, H.; Wang, Y.; Zhang, L.; Chen, L.; Yu, L. Effect of Annealing on Morphologies and Performances of Hydroxypropyl Methylcellulose/Hydroxypropyl Starch Blends. J. Appl. Polym. Sci. 2020, 137, 1–8. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; García, M.A.; Martino, M.N.; Zaritzky, N.E. Effects of Controlled Storage on Thermal, Mechanical and Barrier Properties of Plasticized Films from Different Starch Sources. J. Food Eng. 2006, 75, 453–460. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Herniou-Julien, C.; Álvarez, K.; Alvarez, V.A. Structural Properties and in Vitro Digestibility of Edible and pH-Sensitive Films Made from Guinea Arrowroot Starch and Wastes from Wine Manufacture. Carbohydr. Polym. 2018, 184, 135–143. [Google Scholar] [CrossRef]
- Majzoobi, M.; Pesaran, Y.; Mesbahi, G.; Golmakani, M.T.; Farahnaky, A. Physical Properties of Biodegradable Films from Heat-Moisture-Treated Rice Flour and Rice Starch. Starch/Staerke 2015, 67, 1053–1060. [Google Scholar] [CrossRef]
- Zavareze, E.D.R.; Pinto, V.Z.; Klein, B.; El Halal, S.L.M.; Elias, M.C.; Prentice-Hernández, C.; Dias, A.R.G. Development of Oxidised and Heat-Moisture Treated Potato Starch Film. Food Chem. 2012, 132, 344–350. [Google Scholar] [CrossRef]
- Punia Bangar, S.; Nehra, M.; Siroha, A.K.; Petrů, M.; Ilyas, R.A.; Devi, U.; Devi, P. Development and Characterization of Physical Modified Pearl Millet Starch-Based Films. Foods 2021, 10, 1609. [Google Scholar] [CrossRef]
- Shahbazi, Y. The Properties of Chitosan and Gelatin Films Incorporated with Ethanolic Red Grape Seed Extract and Ziziphora Clinopodioides Essential Oil as Biodegradable Materials for Active Food Packaging. Int. J. Biol. Macromol. 2017, 99, 746–753. [Google Scholar] [CrossRef]
- Alimi, B.A.; Workneh, T.S.; Femi, F.A. Fabrication and Characterization of Edible Films from Acha (Digitalia Exilis) and Iburu (Digitalia Iburua) Starches. CYTA J. Food 2021, 19, 493–500. [Google Scholar] [CrossRef]
- Colivet, J.; Carvalho, R.A. Hydrophilicity and Physicochemical Properties of Chemically Modified Cassava Starch Films. Ind. Crops Prod. 2017, 95, 599–607. [Google Scholar] [CrossRef]
- Lee, H.; Yamaguchi, K.; Nagaishi, T.; Murai, M.; Kim, M.; Wei, K.; Zhang, K.Q.; Kim, I.S. Enhancement of Mechanical Properties of Polymeric Nanofibers by Controlling Crystallization Behavior Using a Simple Freezing/Thawing Process. RSC Adv. 2017, 7, 43994–44000. [Google Scholar] [CrossRef]
- Ibrahim, M.I.J.; Sapuan, S.M.; Zainudin, E.S.; Zuhri, M.Y.M. Physical, Thermal, Morphological, and Tensile Properties of Cornstarch-Based Films as Affected by Different Plasticizers. Int. J. Food Prop. 2019, 22, 925–941. [Google Scholar] [CrossRef]
- Osman, A.F.; Siah, L.; Alrashdi, A.A.; Ul-Hamid, A.; Ibrahim, I. Improving the Tensile and Tear Properties of Thermoplastic Starch/Dolomite Biocomposite Film through Sonication Process. Polymers 2021, 13, 274. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Kapin, É.; Fekete, E. Effects of Preparation Methods on the Structure and Mechanical Properties of Wet Conditioned Starch/Montmorillonite Nanocomposite Films. Carbohydr. Polym. 2014, 113, 569–576. [Google Scholar] [CrossRef]
- Mantovan, J.; Bersaneti, G.T.; Faria-Tischer, P.C.S.; Celligoi, M.A.P.C.; Mali, S. Use of Microbial Levan in Edible Films Based on Cassava Starch. Food Packag. Shelf Life 2018, 18, 31–36. [Google Scholar] [CrossRef]
- Homez-Jara, A.; Daza, L.D.; Aguirre, D.M.; Muñoz, J.A.; Solanilla, J.F.; Váquiro, H.A. Characterization of Chitosan Edible Films Obtained with Various Polymer Concentrations and Drying Temperatures. Int. J. Biol. Macromol. 2018, 113, 1233–1240. [Google Scholar] [CrossRef]
- Sessini, V.; Arrieta, M.P.; Kenny, J.M.; Peponi, L. Processing of Edible Films Based on Nanoreinforced Gelatinized Starch. Polym. Degrad. Stab. 2016, 132, 157–168. [Google Scholar] [CrossRef]
- Basiak, E.; Lenart, A.; Debeaufort, F. Effect of Starch Type on the Physico-Chemical Properties of Edible Films. Int. J. Biol. Macromol. 2017, 98, 348–356. [Google Scholar] [CrossRef] [PubMed]



| Sample | S (%) | SP (%) | WVP × 10−9 (g m−1 s−1 Pa−1) | Thickness | Opacity | EB (%) | YM (MPa) | TS (%) |
|---|---|---|---|---|---|---|---|---|
| FSC | 21.8 ± 0.8 a | 299 ± 39 a | 2.18 ± 0.10 c | 0.13 ± 0.01 | 0.96 ± 0.07 b | 25 ± 2 b | 288 ± 42 c | 5.1 ± 0.3 c |
| FS1 | 20.7 ± 0.9 ab | 191 ± 16 b | 3.45 ± 0.21 a | 0.11 ± 0.01 | 0.95 ± 0.09 b | 22 ± 4 b | 892 ± 89 a | 10.5 ± 0.8 a |
| FS2 | 20.6 ± 0.8 b | 207 ± 22 b | 2.46 ± 0.18 c | 0.13 ± 0.01 | 1.35 ± 0.12 a | 30 ± 3 a | 237 ± 61 c | 5.5 ± 0.7 c |
| FS3 | 19.6 ± 0.8 b | 153 ± 11 c | 2.88 ± 0.10 b | 0.11 ± 0.01 | 0.83 ± 0.04 c | 22 ± 3 b | 597 ± 77 b | 9.4 ± 0.5 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daza, L.D.; Parra, D.O.; Rosselló, C.; Arango, W.M.; Eim, V.S.; Váquiro, H.A. Influence of Ulluco Starch Modified by Annealing on the Physicochemical Properties of Biodegradable Films. Polymers 2022, 14, 4251. https://doi.org/10.3390/polym14204251
Daza LD, Parra DO, Rosselló C, Arango WM, Eim VS, Váquiro HA. Influence of Ulluco Starch Modified by Annealing on the Physicochemical Properties of Biodegradable Films. Polymers. 2022; 14(20):4251. https://doi.org/10.3390/polym14204251
Chicago/Turabian StyleDaza, Luis Daniel, Daniela O. Parra, Carmen Rosselló, Walter Murillo Arango, Valeria Soledad Eim, and Henry Alexander Váquiro. 2022. "Influence of Ulluco Starch Modified by Annealing on the Physicochemical Properties of Biodegradable Films" Polymers 14, no. 20: 4251. https://doi.org/10.3390/polym14204251
APA StyleDaza, L. D., Parra, D. O., Rosselló, C., Arango, W. M., Eim, V. S., & Váquiro, H. A. (2022). Influence of Ulluco Starch Modified by Annealing on the Physicochemical Properties of Biodegradable Films. Polymers, 14(20), 4251. https://doi.org/10.3390/polym14204251







