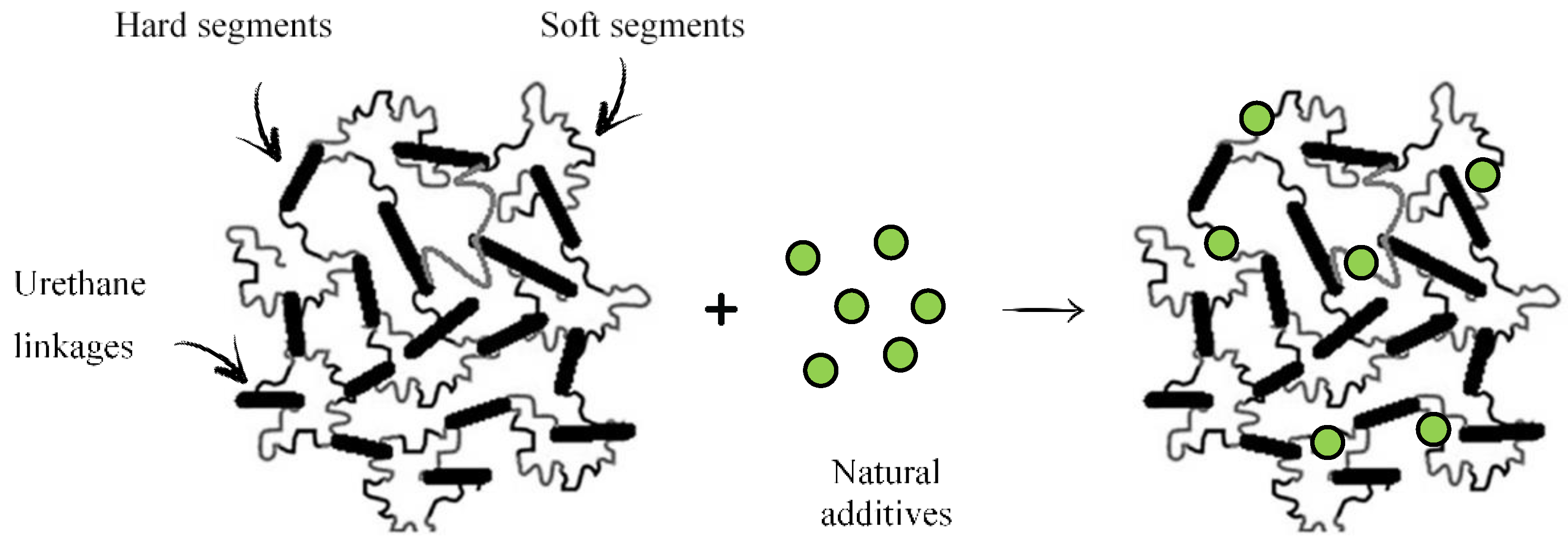
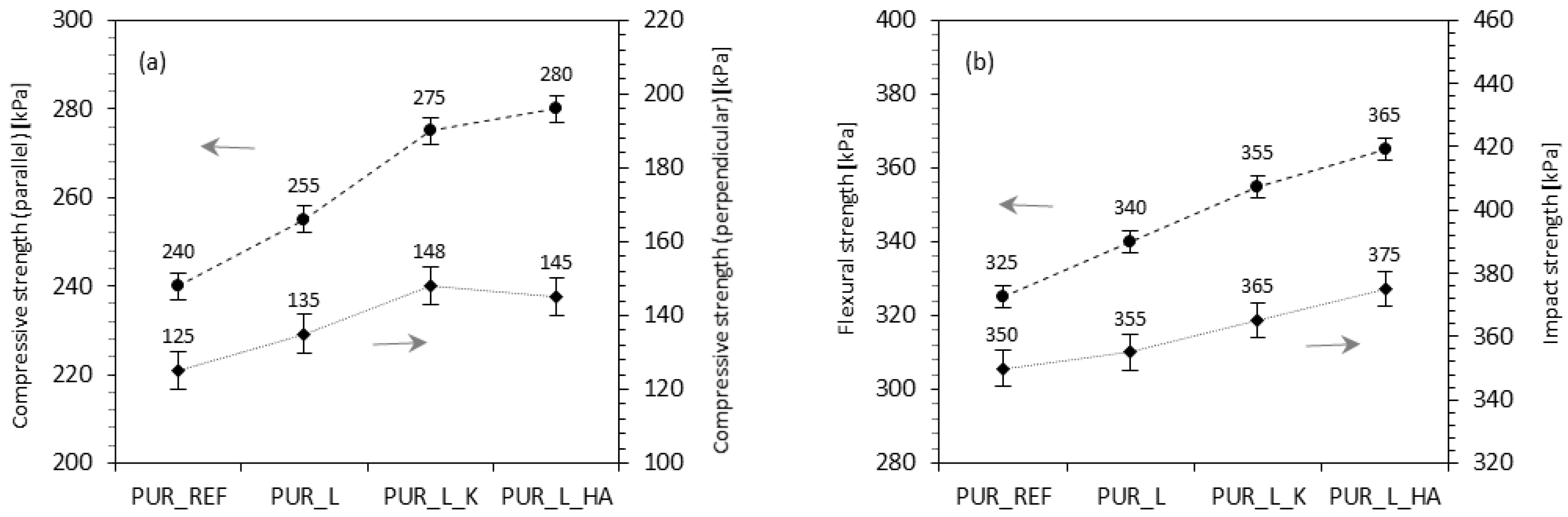
1. Introduction
Nowadays, disinfectants are mostly used for their cost-effectiveness and powerful outcomes, but these chemicals show high toxicity to the environment and human health. Such measures have many disadvantages, e.g., skin irritation on prolonged contact, possible bronchial irritation from inhalation, general caustic effects and corrosion of metals, product deterioration on standing, and others cause those other solutions are currently being sought. Natural antimicrobial additives can be successfully an alternative to chemical antimicrobials. Different kinds of plants, herbs, and spice extracts that have been used in natural medicine for many years have the greatest antimicrobial activity. The bio-resistance potential of clove, oregano, curcumin, cinnamon, birch juice, and thyme essential oils and components, cinnamaldehyde, eugenol, carvacrol, and thymol have been confirmed in the literature [
1,
2]. Many studies show that natural extracts with antibacterial properties can be successfully used as additives to polymers influencing their final properties. The most important feature of materials with antibacterial properties should be to inhibit the appearance, and growth of bacteria and preclude their cumulation. The largest amounts of the most popular bacteria, such as
Escherichia coli,
Klebsiella pneumoniae, and
Staphylococcus aureus, appear on objects which are near to patients, such as bed rails and headboard, armchairs, and cabinets so there are other contact surfaces, from which bacteria can easily transfer to patients and their guardians and other employees [
3,
4]. In the presented works, there are used as additives and modifiers of polyurethanes (PUR), which there are thoroughly researched and used as natural medicines daily. There are no reports in the literature of the use of other toxic natural extracts as polyurethane modifiers. Before starting the study, data on the toxicity of the selected plant should be collected. If the plant is not toxic, continue testing if no toxicity data exist; appropriate determinations should be selected for the toxicity analysis. It is also important to develop and prepare a safety and toxicity protocol. Additionally, it should also be mentioned that the type of polymers used has significant importance in the search for final products that should mainly inhibit the growth of bacteria, fungi, and viruses. It is effectual that the polymers selected to produce specific items should have a bacteriostatic activity to prevent the expansion of bacterial colonies in their proximity or work bactericidal and radiogenic impair nearby bacteria, by changing the course of their cell processes. Important characteristics to keep in mind are the yield and the type of surface to be used. The area with antibacterial features should be prepared with polymers which properties have a long useful time with contemporary high productivity of effect. Moreover, these polymers should show refractory to outside agents so that the process of leaching the antiseptic does not appear because of chemical or mechanical effects and it should also have the odds of constant bonding to different surfaces [
5,
6,
7,
8].
PUR foams are mainly used as insulation materials. Therefore, they should be resistant to viruses, bacteria and fungi. Commercial, synthetic compounds used as antimicrobial and antioxidant agents are mainly derived from phenol, and they are heavily toxic for human health. Nowadays, the tendency is to limit the use of synthetic additives, and replace them with the natural, bio-based antimicrobial and antioxidants additives that do not impact the human health. The application of natural compounds with an antimicrobial and anti-aging activity have been used in previous research. For example, Członka et al. [
9] studied the antioxidant and antimicrobial activity of PUR foams containing clove filler. The obtained materials showed great antibacterial activity against selected bacteria, e.g.,
E. coli and
S. aureus. Similar results were obtained in the case of PUR foams with incorporated nutmeg filler—the inhibitory zone of four bacterial strains (
E. coli,
S. aurous) significantly increased with the incorporation of nutmeg compound [
10]. The antimicrobial activity of cinnamon extract was also confirmed by Liszkowska et al. [
11] in the case of new materials based on cinnamon extract embedded in PUR matrices. The presented results confirmed that the natural, antibacterial compounds can be incorporated into PUR foams and retain their inhibitory effect against microbial growth. Previous studies have shown that strong interfacial interaction, such as hydrogen bonding, can be easily formed between the additive molecules and isocyanate groups, leading to the formation of a cross-linked structure. It has been shown that the functional groups of natural additives (e.g., hydroxyl and amine groups) can react with isocyanates even in the absence of catalyst leading to the formation of urea bonds [
9,
10,
11,
12,
13]. The impact of the addition of natural additives on the cross-linking density of polyurethane composites is presented in
Figure 1.
Due to the constant interest in PUR materials with antibacterial properties, the prepared work contains the latest information on this topic and, in order to be readable for the recipient, has been divided into two main parts, such as antimicrobial PUR and antimicrobial PUR foams. The first chapter describes all forms of polyurethanes, except for foams, which, due to their wide application in many fields, describes the next chapter. The presented review shows that incorporation of natural modifiers with given antimicrobial properties into conventional PUR and PUR foam formulations obtain materials with assumed features.
2. Polyurethanes Modified with Antimicrobial Compounds
Recently, PUR is widely used in the production of equipment that outright comes in contact with a person as medical application and others [
15,
16,
17,
18]. Results in the literature indicate that the possibility and capacity of bacteria, viruses or fungi increasing on the surface of PUR can initiate a human health problem that arises during the use and storage of these materials.
A good example to raise antiseptic properties of PUR is to change them with the application of substances, which expose antimicrobial features like cinnamaldehyde (CA). Kucinska-Lipka and Feldman shown the synthesis of antibacterial porous structure of PUR matrices (MPTLs) in a shape of fine layers by applying solvent-casting/particulate-leaching method jointed with thermally initiated phase separation [
19]. Furthermore, received MPTLs were modified with cinnamaldehyde at various percentage concentrations (0.5–5%) to create the best antimicrobial result of the CA applied into the MPTLs structure. Both unmodified and CA-modified MPTLs samples were defined by mechanical and physicochemical properties as well as by assessment of their antibacterial efficiency. The obtained results shown that the most important antibacterial effect of CA-modified MPTLs was received when the CA amount was 3.5% and it was between all concentrations used. All the described properties arise from the fact that the CA exhibits antibacterial, antifungal, and anti-inflammatory properties. CA in other words, cynamal, is the yellow liquid with an intensive and sweet-spicy flavor, with the group of aromatic aldehydes [
20]. The greatest content of CA is the oil of
Cinnamomum zeylanicum tree [
21]. Because of that CA has in its structure aldehyde groups show that it can be built in the chemical structure of such materials as polyurethane, chitosan, polylactide, cellulose, or alginates. The meaningful advantage of speaking in favor of this substance is the fact that it has been accepted as commonly safe and harmless by the Food and Drug Administration in the USA [
22]. Scientists’ reports confirm and clearly indicates preservative properties of CA towards antimicrobial activity against
Pseudomonas syringae pv.
actinidiae (plant pathogen) [
23],
E. coli, and also
Salmonella [
24]. Importantly, with regard to the quoted studies, CA after inclusion into cellulose-based package film also had antimicrobial activity against pathogenic bacteria such as:
Aeromonas hydrophila,
Bacillus cereus,
E. coli DMST 4212,
E. coli O157: H7,
Listeria monocytogenes,
Micrococcus luteus,
Pseudomonas aeruginosa,
Salmonella enteritidis,
S. aureus,
Enterococcus faecalis, and antifungal working against three yeasts
Candida albicans,
Saccharomyces cerevisiae, and
Zygosaccharomyces rouxii [
25]. The results indicate that the mechanism of antibacterial and antifungal action of CA depends on its concentration in material. Generally, trans-cinnamaldehyde can impede the increase in
E. coli and
S. typhimurium without breaking up the external coating of a bacterial cell or emptying intracellular Adenosine Triphosphate (ATP) Concentration. Research shows at low concentrations, it restrains enzymes committed in cytokine mutuality or different significant cell functions but when the concentration is higher, it restrains ATPase, and at deadly quantities, cinnamaldehyde set whirling the construction of cell membrane [
26]. The mechanical features of samples and morphology advisable that CA uncovers an effect on these properties, but the contact angle of the area has little increased for the reason of CA hydrophobic character. Noticed mass change (over 50% for unmodified and almost over 60% for CA-modified MPTL) shows that these proposed material compositions may find a successful use as biodegradable coating. In accordance with the executed research, it is supposed that received CA-modified MPTLs can find a common use as functional antibacterial matrices for reclamation of injured and damaged skin. Purposeful tests as Tensile strength (TSb), elongation at break (εb), and Young’s modulus (E) were made and the results are as follows. Unmodified MPTLs had TSb of 0.32 ± 0.01 MPa, εb of 78 ± 6%, and E of 0.254 ± 0.15 MPa but which is very interesting modification with CA only slightly affected on the mechanical properties of CA-modified MPTLs. The TSb, εb, and E of CA-modified MPTLs decreased to 0.18 ± 0.02 MPa, 68 ± 4%, 0.244 ± 0.09 MPa, accordingly [
27].
The antiseptic and simultaneously anti-adhesive properties of PUR have been obtained by immobilizing chitosan and heparin on the samples of PUR specially prepared for this purpose via a stepwise method. Antibacterial functions of materials were obtained by plasma and modified with different concentrations of chitosan (0.5 and 2.0%) and heparin immobilization. Prepared samples for analyses were modified to be antibacterial resistant against
S. aureus,
S. epidermidis (both Gram-positive),
E. coli, and
P. aeruginosa (both Gram-negative) bacteria. The obtained bacterial adhesion results were satisfactory and indicated a meaningful decrease in the number of feasible bacteria on alike samples modified chitosan as well as modified heparin where for these samples the results were better and the most efficient (
Figure 2 allows better perceive the presented works and there are not as accurate data and results) [
28]. Kara et al. [
29] showed that the polyurethane surfaces with towering hydrophilicity and surface free energy-adjusted anti-adhesion efficiency versus bacteria. Additionally, stability studies of the obtained systems are presented, which indicate that immobilizing chitosan as well as immobilizing heparin were stable and did not separate from the polyurethane surfaces. Moreover, the results showed that chitosan and heparin were covalently linked to the polyurethane by activating the surface with plasma and glutaraldehyde which ensured reactive aldehyde groups that can simply react with the amine and hydroxyl groups of chitosan and heparin [
29].
New antibacterial wound dressing materials were prepared via coating solution with
Szygium aromatic extract (clove oil) thermoplastic polyurethane nanofibers mats. Used clove oil was obtained by Soxhlet extraction and subjected to further examination by gas chromatography-mass spectrometry analysis, which confirmed that the extract was primarily assembled of eugenol and β-caryophylene with preferable antimicrobial activity. Polyurethane materials were coated with early received clove oil to obtain nanofibers with assumed antibacterial properties. Research proves that even 2 mg cm
−2 clove-oil-coated polyurethane nanofibers show an area of security against
S. aureus and
E. coli, respectively. The inhibition area results are shown in
Table 1 [
30].
The work also noted that air permeability of nanofibrous materials reduced with the growing quantity of clove oil over 5 mg cm
−2, which is due to those clove oil caused modifications in the morphology of nanofibers. It is worth pointing out, that the inherence of clove oil did not affect the morphology of nanofibers coated with 2 mg cm
−2 clove oil and, in this case, the air permeability utility of those bandages was close to that of unmodified nanofiber materials [
30].
Yue et al. [
31] presented the possibility of using thymol loaded ethanol-soluble polyurethane as nanofibrous membranes with antibacterial activity. Thymol is a natural monoterpenoid phenol derivative of p-Cymene, isomeric with carvacrol, found in oil of thyme, and extracted from
Thymus vulgaris (common thyme),
Ajwain (common cumin), and various other kinds of plants as a white crystalline substance of a pleasant aromatic odor and strong antiseptic properties. Scientists obtained a dressing material with complex properties with antibacterial activity, waterproofness, and water vapor permeability used polyurethane-Thymol nanofibrous membranes with various amounts of thymol (2, 4, 6, and 8 wt.%) to determine the effects of thymol on the morphology and execution of fibrous membranes. Finally, the resultant nanofibrous membranes composed of PUR, fluorinated PUR, and thymol had homogeneous structure, good water resistance with the hydrostatic pressure of 17.6 cm H
2O, great breathability of 3.56 kg m
−2 d
−1, the significant tensile stress of 1.83 MPa, and tensile strain of 453%, and very promising antibacterial activity. In the presented research, it was also important to search and define the antibacterial activity of polyurethane-Thymol membranes against
S. aureus and
E. coli based on the method of colony counting. The control plates for nonmodified PUR samples showed crowded colonies of bacteria, whereas the PUR modified with 8 wt.% Thymol plates did not show any colony. It is also worth noticing that all polyurethane-Thymol nanofibrous materials revealed antibacterial activity. Additionally, to investigate the germicidal effects of polyurethane-Thymol materials, the structural changes of
E. coli and
S. aureus were evaluated by SEM. For PUR modified by 14 wt.% Thymol,
E. coli cells were lifeless and shown a specific rod-shaped structure. Nevertheless, for PUR modified by 8 wt.% Thymol, the cells of
E. coli were spoiled, and the cytoplasm issued out what confirmed that the cell membranes were damaged. After the modification with 14 wt.% of Thymol, both
E. coli and
S. aureus had spherical sleek cells. After the modification with 8 wt.% of Thymol, the morphology of the cells became more erratic. To confirm the obtained results, fluorescence-based live/dead bacteria designation was also applied. Calcein, used as a non-fluorescent cell penetrable dye, is enzymatically transformed to potent fluorescent calcein to recognize existing bacteria. Increased red fluorescence was observed in the case of PUR modified with 8 wt% of thymol. This confirmed the antimicrobial properties of the modified PUR fibrous material. Additionally, developed by researchers the in situ electrospinning by the designed mobile equipment showed the potential to develop convenient and preventive fibrous materials for the skin at any time, which will promote the common use of electrospun fibrous injury bandage materials. Scientists used in their research 3 wt%
Salvia against
S. aureus,
E. coli, and
P. aeruginosa [
31].
Another example of the use of natural additives with antibacterial properties in polyurethane materials was presented by Santamaria-Echart et al. [
32]. Their work was focused on the preparation of Salvia-based waterborne polyurethane-urea (WBPUU) dispersions with cellulose nanocrystals (CNC) allowing the preparation of functional green nanocomposite films with enhanced mechanical properties. The work showed that
Salvia officinalis extracts, recognized for their antibacterial and antioxidant properties [
33,
34,
35,
36], were able to transfer the same properties to the nonmodified WBPUU. Furthermore, during the microorganism test, the effect was positive against Gram-positive
S. aureus, and Gram-negative
E. coli, and
P. aeruginosa bacteria. It was noticed that after an incubation period of 1 day, Salvia-based WBPUU films showed a bacteriostatic effect against chosen bacteria, impeding their growth. What is more, after an incubation of 4 days the detention power of the nonmodified WBPUU material was discontinued for all the attempted bacteria, where the modified with natural extract WBPUU films showed individual behaviors dependent on the used incorporation of antibacterial extract. As concerns Gram-positive
S. aureus, it was shown that the arresting effect caused by the adding of
Salvia extract was only efficient in the case where the in situ method was used and applied amounts 3 and 5 wt.% of extract. For samples with
Salvia extract, the bacteria growth was inhibited in most of the modified polyurethane films confirming their yield against this bacterium. The sample containing 1 wt.% natural extract showed slightly different results and the inhibition effect was impeded. A similar tendency was noticed against
P. aeruginosa. Moreover, considering that
P. aeruginosais is more resistant, if compared with
E. coli (being Gram-negative bacteria), the inclusion through the post-method did not score effective, presumably on account of lixiviation effects further time [
32].
Other obtained results showed the low and tight particle size distribution of the Salvia-based WBPUU dispersion, which can be associated with the effective synthesis process with the natural extract supplement. The dispersion was used to obtain novel organic nanocomposites by introducing isolated cellulose nanocrystals with large length-to-diameter ratio. The aspect ratio value is a really important parameter setting the efficiency of the CNC in the nanocomposite. The higher this ratio is, the greater the reinforcing capacity of the system. This worth is in the top scope comparing with congruous materials, take advantage of the reinforcing effect it can transfer to obtained Salvia-based WBPUU samples. What is more, FTIR analyses confirmed that the addition of CNC caused the increase in both, free C=O groups in the soft segment of urethane and hydrogen-bonded C=O groups of urea groups. Differential scanning calorimetry (DSC) results took notice a process of changing how phases induced by CNC incorporation is organized that, in general, resulted in lower the mobility of the soft domain values (indicating the increased mobility of the amorphous fraction of soft domains), while favoring the system of brief range ordered hard spheres, indicated by the increase in ΔH
mHS values. The relations between the parts or elements accepted by the WBPUU, also with the rigid CNC, make it possible to receive composites with stable mechanical properties (higher E), and extension elongation ability [
32,
37].
A new not harmful to living tissue and antimicrobial material with effective wound healing activity can be a specific option for wound dressing use. Eskandarinia et al. [
38] presented polyurethane-hyaluronic acid (PU-HA) nanofibrous wound dressing with three different amounts of ethanolic extract of propolis (EEP). Propolis (bee glue) is a piceous beehive residue collected by honeybees from different plants and next there is applied to the gasket in honeycombs, polish the inside walls and also protect the entry against insects. A lot of pharmaceutical materials including antibacterial, antifungal, antiviral, antiprotozoal, anti-inflammatory, antioxidant, anti-cancer, and other activities have been prepared from propolis [
39,
40,
41,
42]. Ethanol extract of propolis indicates various antimicrobial facilities according to the type of the tested drain off and activity time. This extract of propolis proves severe antimicrobial activities towards Gram-positive cocci strains belonging to
S. aureus types [
35].
The new propolis modified PUR-HA nanofibrous were featured by attenuated total reflectance/Fourier transform infrared spectroscopy, thermal gravimetric analysis, scanning electron microscopy, mechanical investigations, antimicrobial tests, and others. The PUR-HA/1% EEP and PUR-HA/2% EEP ones indicated higher antibacterial activity against
S. aureus (2.36 ± 0.33 and 5.63 ± 0.87 mm) and
E. coli (1.94 ± 0.12 and 3.18 ± 0.63 mm) in comparison with other tested samples. On the other hand, the PUR-HA/1% EEP sample shown notably higher biocompatibility for L929 fibroblast cells in comparison with PUR-HA/2% EEP. Additionally, the PUR-HA/1% EEP sample could crucially speed up the wound healing advancement and wound closure at the animal type. At the histopathological analysis, revised skin growth and collagen deposition at the healed wound place of the PUR-HA/1% EEP sample in comparison with other groups was observed. The obtained results showed that 1 wt.% EEP enriched PUR-HA nanofibrous can be a sanguine material with substantial biocompatibility, wound healing, and antibacterial features for next biomedical uses [
38,
43,
44].
In the study presented by Shababdous et al. [
45] two types of PUR were prepared in a two-step polymerization based on polycaprolactone, hexamethylene diisocyanate, and 1,4-butandiol. Obtained results of FTIR analyses and
1H NMR spectroscopy characterization indicated the achievement of PUR synthesis. Furthermore, thermal features of the PUR were carried by DSC and the obtained effects indicated that soft segments of analyzed PUR are amorphous and solid units of this material are crystalline. Additionally, the hydrophilicity of the presented materials was studied, and the outcomes showed that curcumin-loaded polyurethane is more hydrophilic than nonmodified PUR. Curcumin as a modifier was applied in the polyurethanes by electrospinning process in various amounts. The lowering speed of curcumin from polyurethane samples as well as antimicrobial activity of the matrix was explored and the results have shown that curcumin-modified PUR had satisfying mechanical and anti-bacterial properties, which confirmed that this material are the right candidate for wound dressing uses [
45].
To heal diabetic wounds, PUR/carboxymethylcellulose nanofibers containing
Malva sylvestris extract are used. Malva extract as a modifier was incorporated in the PUR and different amounts of carboxymethyl cellulose blend and has been observed utility, antibacterial, and wound healing properties [
46,
47,
48,
49,
50]. The obtained research results indicated that increasing the diameter of the nanofibers and decreasing the carboxymethyl cellulose concentration raised in progressive the freeing time up to 85 h. The application of natural extract in the polymer material extended the mechanical and thermal properties of nanofibers. FTIR analysis showed the generating of hydrogen bonds between the extract and used PUR. The cell culture investigation was conducted by the protocol reported by Almasian et al. [
51] and applied analysis indicated the non-toxic nature of produced wound dressings. Moreover, the extract containing nanofibers (15% w/w) exhibited 70.66 and 69.83% antibacterial activity against
E. coli and
S. aureus bacteria, accordingly. The wound dressing comprising 15% natural Malva extract presented the wound healing rapidity of 95.05 ± 0.24% by day 14. Additionally, from the histological images, it was noted that macrophage infiltration, neovascularization activity, and fibroblastic proliferation increased on the 7th day and the grade of collagenization and epithelium regeneration was increased on the 14th day [
51,
52].
Table 2 summarizes the methods of polyurethane modification to obtain antibacterial properties of the material.
4. Conclusions
Polyurethanes are one of the common polymeric materials with great control over their physicochemical features based on chemical composition. The properties of polyurethanes enable them to obtain their various forms and to be applied in universally accessible products where are required conveniences, durability, warmth, and sound isolation.
Advisable research of scientists indicated that the chemical inclusion of modifying substances with assured antibacterial and antifungal properties into nonmodified polyurethane formulations enable obtain of antimicrobial products. The scientific description confirmed that the use of an antibacterial modifier can change the rheological behavior, cellular structure, and further mechanical properties of polyurethane materials.
Naturally occurring antimicrobials have gained attention among researchers and material manufacturer due to their safety, easily accessible and nontoxic status. Natural additives as extracts are easy to obtain from various plants. Fruit, vegetables, herbs, and spices have been found to be rich sources of aldehydes, ester terpenoids, phenolics, and sulfur-containing compounds. These natural occurring agents commonly found in roots, flowers, leaves, seeds and bulbs and in other parts of the plants. These substances are produced in defensive mechanism and are helpful for inactivation or inhibition of many microorganisms (bacteria, yeast and molds) and there have not harmful impacts on human health. These naturally occurring antimicrobial agents can be isolated from local sources using various advanced techniques. Due to potential adverse effect of some synthetic fungicides and antibacterial additives on environment and health there is a strong societal emphasis for use less synthetic antimicrobial additives in materials and the use of natural antimicrobial as alternatives. Despite the fact that natural additives have many advantages and can be successfully used as biocides, it must also be remembered that natural extracts may be less active compared with chemical equivalents, and have a shorter period of action. However, the presented research shows that this topic is very interesting because it concerns the use of organic substances in polymer materials [
72,
73,
74,
75,
76,
77,
78,
79,
80,
81,
82]. The antimicrobial properties of the polyurethane were rated toward
E.coli and
S. aureus and others. The methods of polyurethane modification shown in this review may lead to obtaining materials with antibacterial and antifungal properties. Furthermore, this means ways for the formation of antibacterial and antifungal materials in different forms facilitate the future development of new safer, and more effective antibacterial polyurethane materials in the new applications in various fields. So far, there are not many reports on the use of natural antibacterial substances in foams, and the topic is constantly evolving because bacteria and fungi can infect walls, ceilings, and other places in commercial areas, causing unsightly conditions and even breathing problems for the building’s occupants. Frequently, everyday-use products are exposed to fungus and mold which cause ugly black and brown blots or greenish marks which can destroy the condition of materials if they are not protected with an antifungal modifier. Constantly endangering fungus and mold can cause products to fast decrease in product quality, stability, and product shelf life. The presented review confirmed that incorporation of natural modifiers with given antibacterial properties into conventional polyurethane and polyurethane foam formulations allows preparing of antimicrobial materials with effective alternatives to expensive and currently technologies [
83,
84,
85,
86,
87,
88,
89,
90,
91,
92,
93,
94,
95,
96,
97,
98,
99,
100,
101,
102].
Polymer materials have been widely used in various industries. The related need for microbiological protection forces the biostabilization of polymers and their composites. The scope of application of polymeric materials with microbiological protection, additionally enriched with antimicrobial properties, is very extensive and covers many industries. The most important are the medical industry, textile, packaging, water filtration systems, air conditioning. Protection of polymers against adhesion of microorganisms to the surface, as well as the colonization of products by them, are commonly used. The use of biocides for plastics prevents their discoloration, problems with maintaining microbiological cleanliness of the surface, slows down aging.
Summarizing our in-depth analysis of the natural antimicrobial substances used so far as additives to polyurethanes, we consider that the presented results prove that it can be potential strategy or candidate to be further applied extensively. The possibility of using such additives is confirmed not only by the fact of their effectiveness, but also the nature of their origin, the possibility of using, in many cases, bio-waste with antimicrobial properties. There are many natural antibiotics in nature; thus, further research should be carried out in this field because there are few such additives to polyurethanes described so far.