Optimization of the Rheological Properties of Self-Assembled Tripeptide/Alginate/Cellulose Hydrogels for 3D Printing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
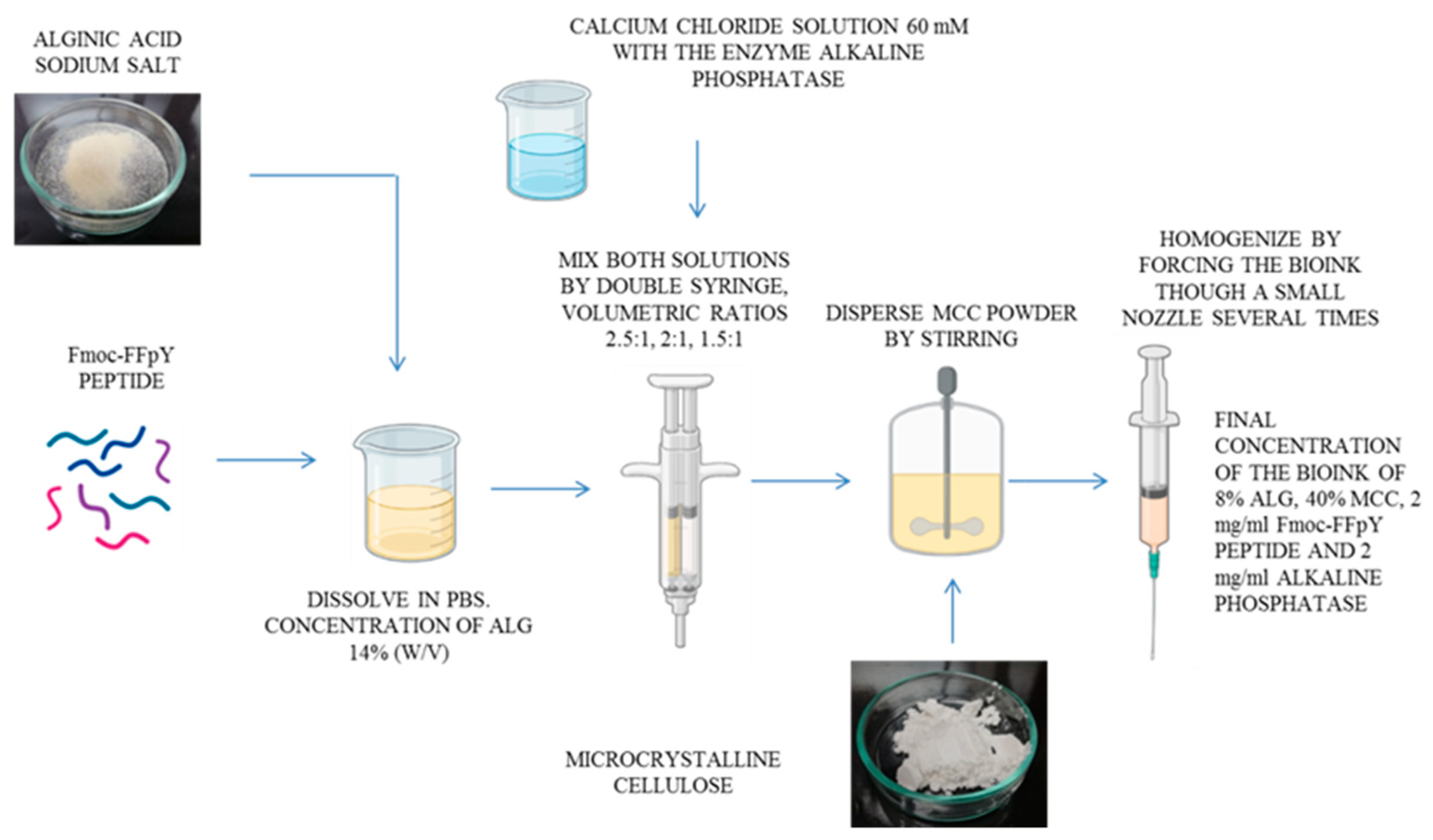
2.2. Ink Preparation
2.2.1. Preparation of the Pre-Crosslinked Alginate Inks with CaCl2
2.2.2. Modulation of the Pre-Crosslinked Inks Viscosity by Incorporation of Microcrystalline Cellulose and Printability Tests
2.2.3. Incorporation of the Fmoc-FFpY Peptide into the Ink Formulation
2.3. Rheological Characterization of the Inks
2.4. 3D Printing of Alginate Inks
2.4.1. Transformation of a Filament Extrusion 3D Printer into a Bioprinter
2.4.2. Optimization of Printing Parameters
2.4.3. 3D Printing of Alginate Scaffolds
2.5. Characterization of the Printed Scaffolds
2.5.1. Morphological Characterization by Scanning Electron Microscopy
2.5.2. Water Content
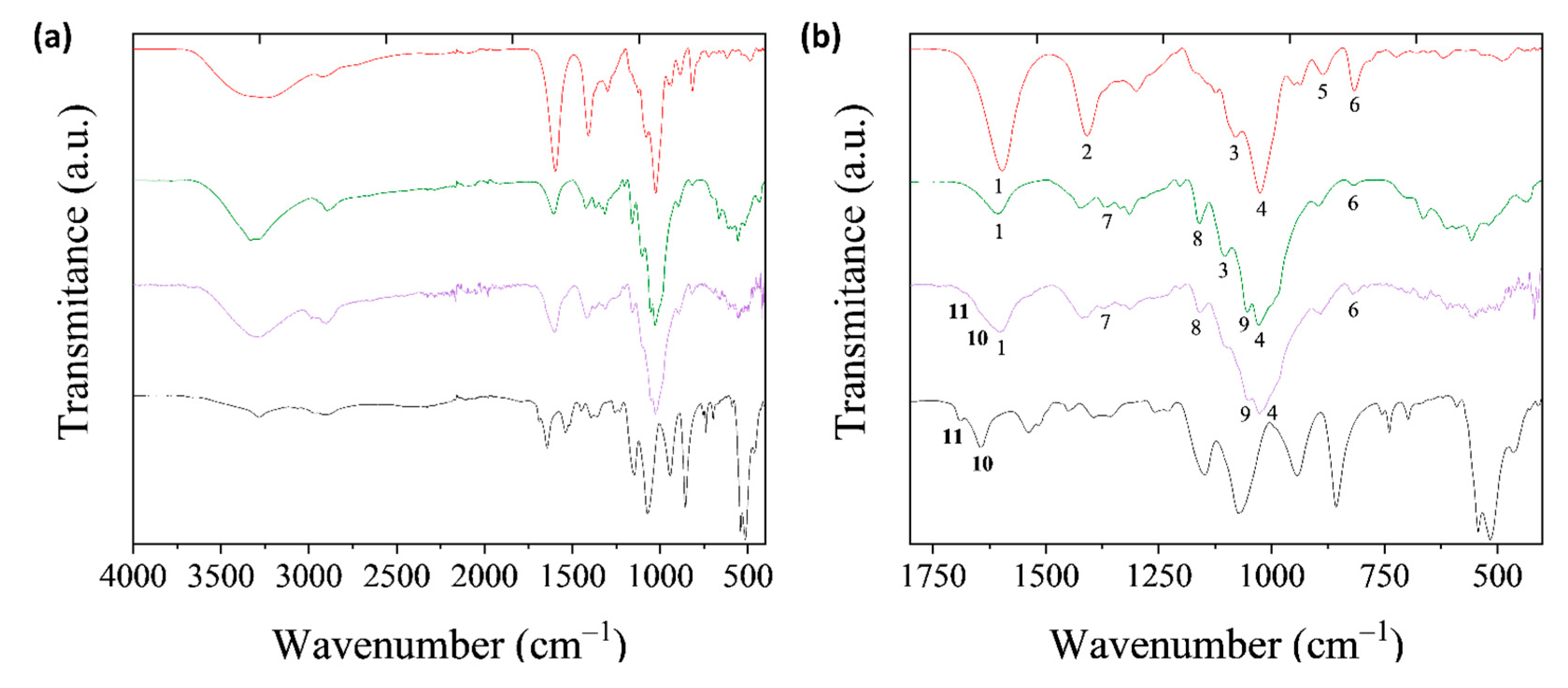
2.5.3. Fourier Transform Infrared Spectroscopy (FTIR)
2.6. In Vitro Cytotoxicity Tests
2.6.1. Cell Culture
2.6.2. Cytotoxicity Assay
2.6.3. Cell Adhesion and Proliferation
3. Results and Discussion
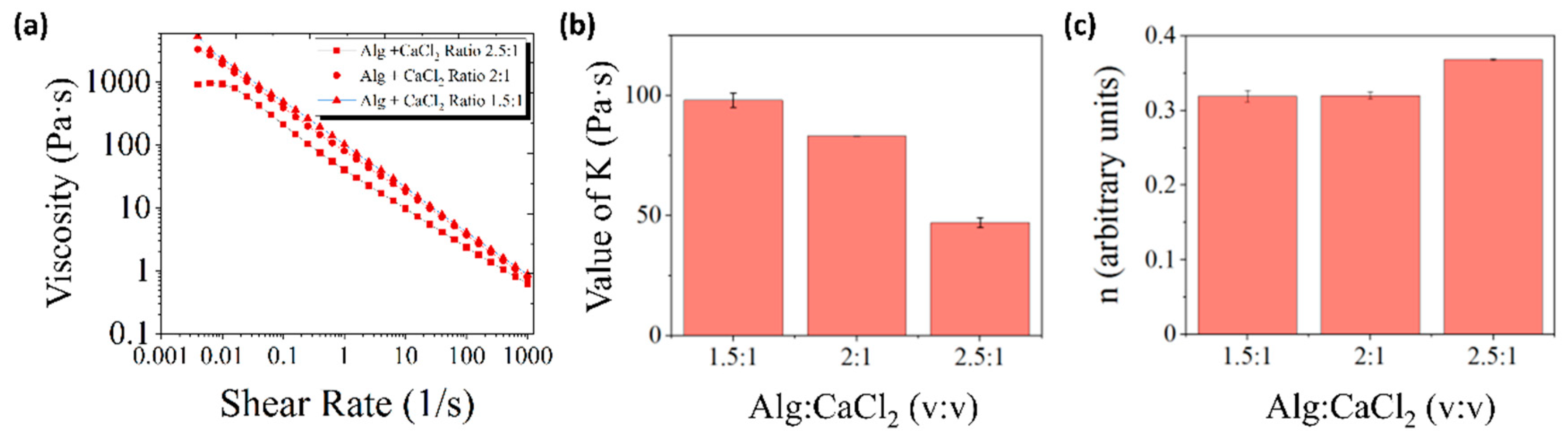
3.1. Rheological Characterization of Alginate Inks Pre-Crosslinked with CaCl2
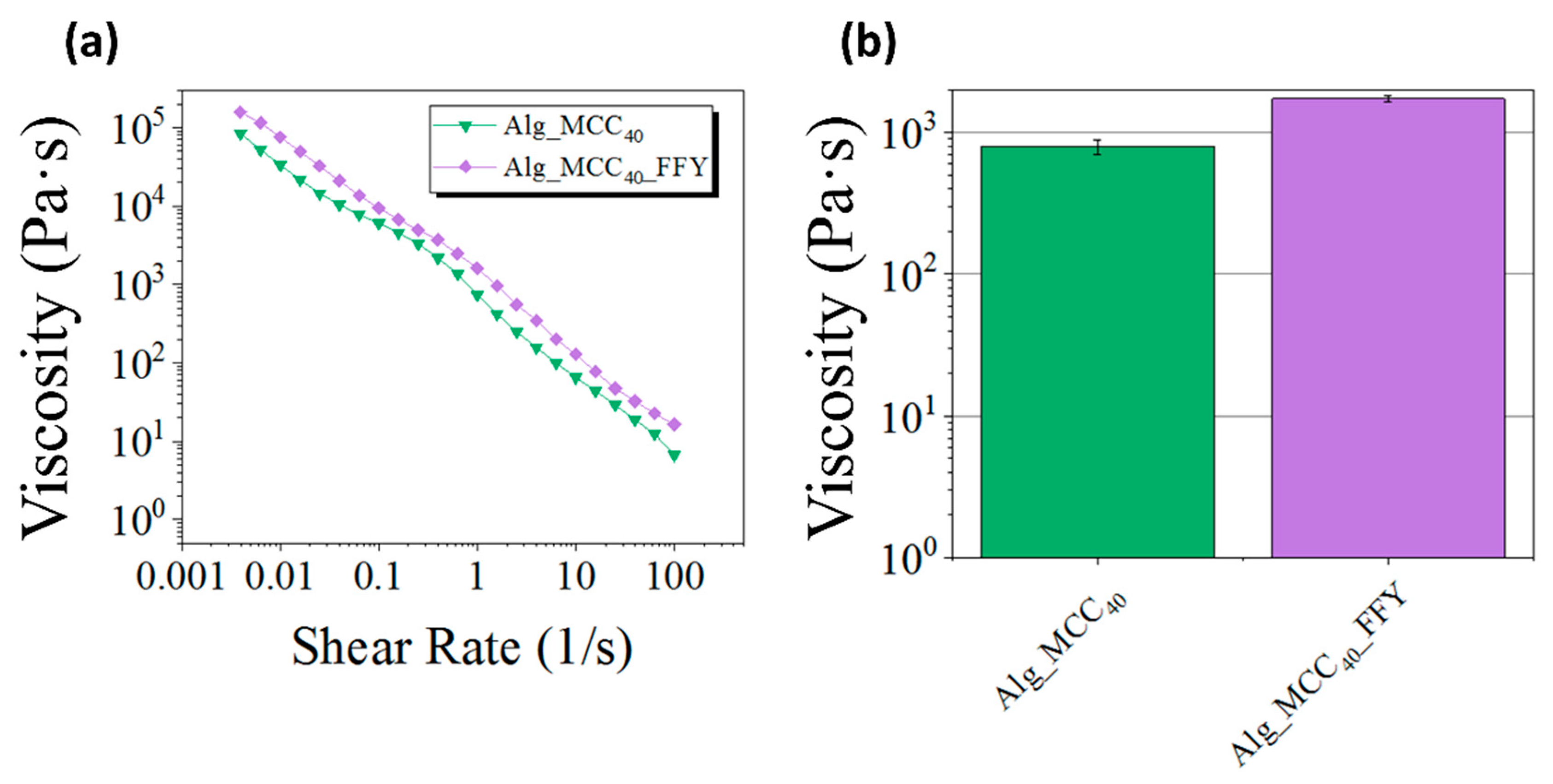
3.2. Modulation of the Viscosity by Incorporation of Microcrystalline Cellulose within the Pre-Crosslinked Alginate Ink
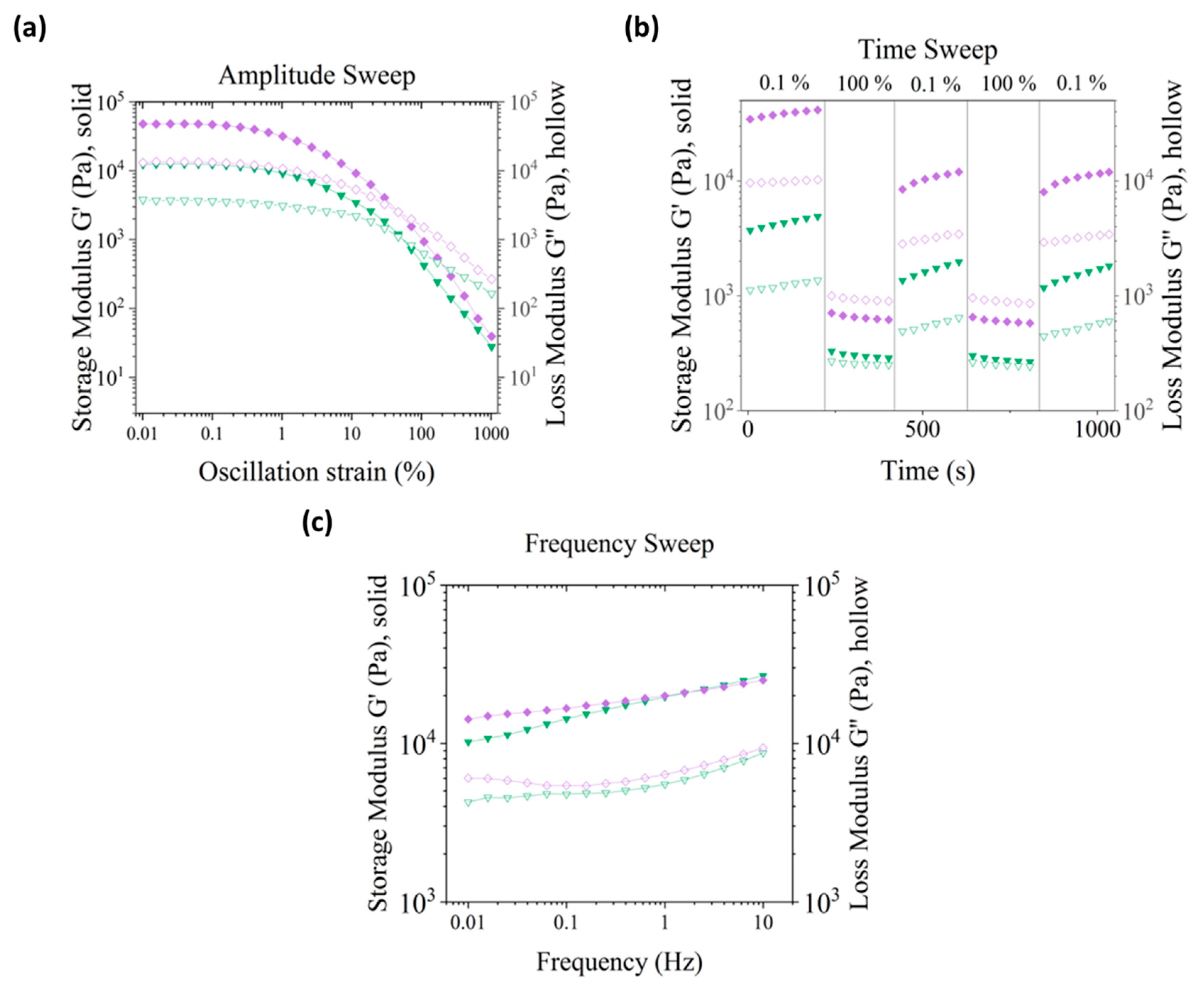
3.3. Rheological Characterization of the Optimized Alginate-Cellulose Ink
3.4. Optimization of Printing Parameters
3.5. Morphological Characterization of 3D Printed Scaffolds
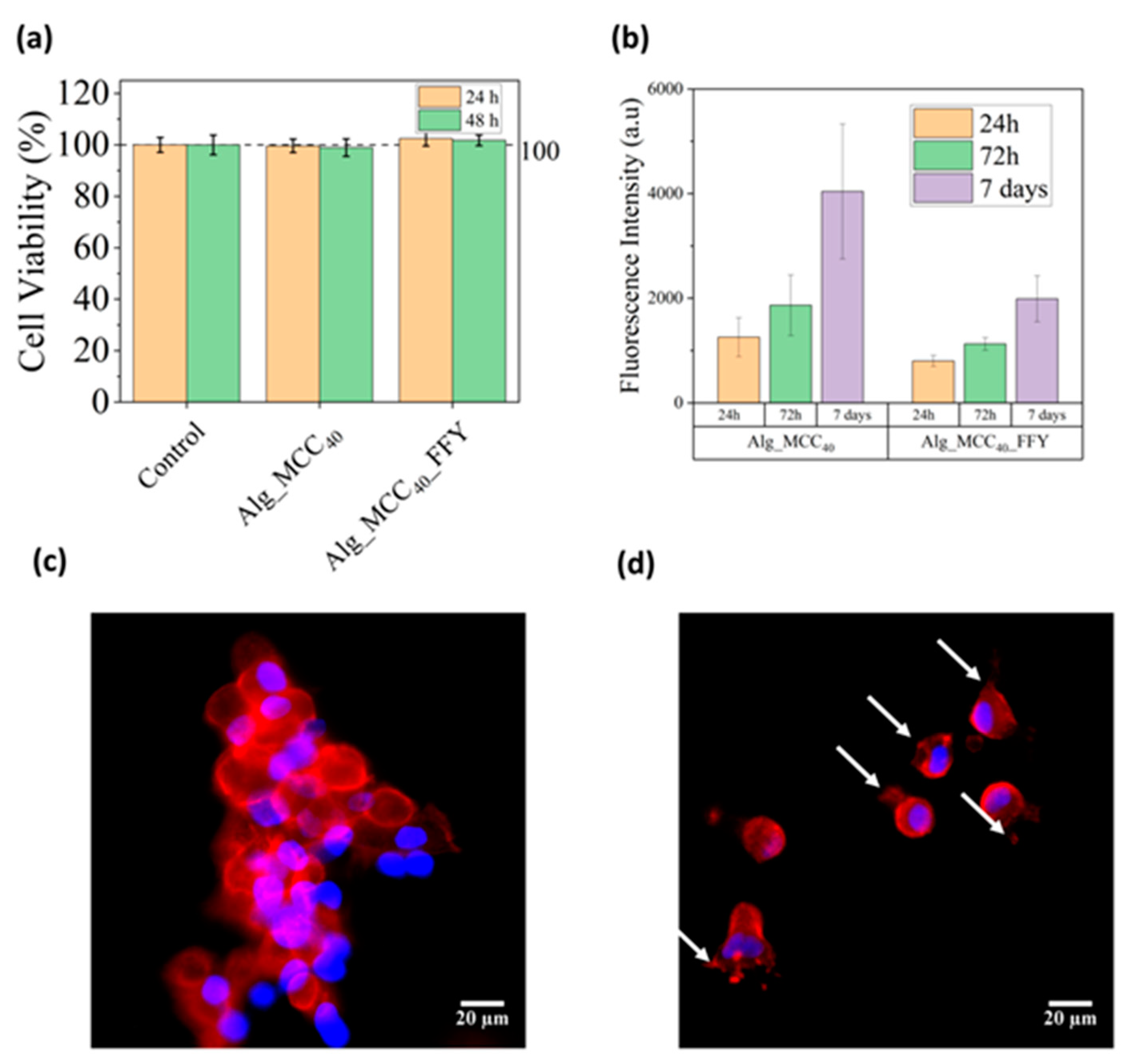
3.6. In Vitro Cytotoxicity Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mancha Sánchez, E.; Gómez-Blanco, J.C.; López Nieto, E.; Casado, J.G.; Macías-García, A.; Díaz Díez, M.A.; Carrasco-Amador, J.P.; Torrejón Martín, D.; Sánchez-Margallo, F.M.; Pagador, J.B. Hydrogels for Bioprinting: A Systematic Review of Hydrogels Synthesis, Bioprinting Parameters, and Bioprinted Structures Behavior. Front. Bioeng. Biotechnol. 2020, 8, 776. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.S.; Mijangos, C.; Hernandez, R. Polysaccharide Coating of Gelatin Gels for Controlled BSA Release. Polymers 2019, 11, 702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernández, R.; Mijangos, C. Determining the Rheological Properties of Polymer Hydrogels for the Development of Advanced Applications. In Rheology: Theory, Properties and Practical Applications; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2013; pp. 383–407. ISBN 9781626189997. [Google Scholar]
- Criado-Gonzalez, M.; Corbella, L.; Senger, B.; Boulmedais, F.; Hernández, R. Photoresponsive Nanometer-Scale Iron Alginate Hydrogels: A Study of Gel-Sol Transition Using a Quartz Crystal Microbalance. Langmuir 2019, 35, 11397–11405. [Google Scholar] [CrossRef] [Green Version]
- Boffito, M.; Sartori, S.; Ciardelli, G. Polymeric Scaffolds for Cardiac Tissue Engineering: Requirements and Fabrication Technologies. Polym. Int. 2014, 63, 2–11. [Google Scholar] [CrossRef]
- Qian, H.; Wang, J.; Yan, L. Synthesis of Lignin-Poly(N-Methylaniline)-Reduced Graphene Oxide Hydrogel for Organic Dye and Lead Ions Removal. J. Bioresour. Bioprod. 2020, 5, 204–210. [Google Scholar] [CrossRef]
- Zhang, J.; Jin, J.; Wan, J.; Jiang, S.; Wu, Y.; Wang, W.; Gong, X.; Wang, H. Quantum Dots-Based Hydrogels for Sensing Applications. Chem. Eng. J. 2021, 408, 127351. [Google Scholar] [CrossRef]
- Han, X.; Xiao, G.; Wang, Y.; Chen, X.; Duan, G.; Wu, Y.; Gong, X.; Wang, H. Design and Fabrication of Conductive Polymer Hydrogels and Their Applications in Flexible Supercapacitors. J. Mater. Chem. A 2020, 8, 23059–23095. [Google Scholar] [CrossRef]
- Criado-Gonzalez, M.; Dominguez-Alfaro, A.; Lopez-Larrea, N.; Alegret, N.; Mecerreyes, D. Additive Manufacturing of Conducting Polymers: Recent Advances, Challenges, and Opportunities. ACS Appl. Polym. Mater. 2021, 3, 2865–2883. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, F.; Zhai, W.; Cheng, S.; Li, J.; Wang, Y. Unraveling of Advances in 3D-Printed Polymer-Based Bone Scaffolds. Polymers 2022, 14, 566. [Google Scholar] [CrossRef]
- Zhang, A.; Wang, F.; Chen, L.; Wei, X.; Xue, M.; Yang, F.; Jiang, S. 3D Printing Hydrogels for Actuators: A Review. Chin. Chem. Lett. 2021, 32, 2923–2932. [Google Scholar] [CrossRef]
- Li, J.; Wu, C.; Chu, P.K.; Gelinsky, M. 3D Printing of Hydrogels: Rational Design Strategies and Emerging Biomedical Applications. Mater. Sci. Eng. R Rep. 2020, 140, 100543. [Google Scholar] [CrossRef]
- Markstedt, K.; Mantas, A.; Tournier, I.; Martínez Ávila, H.; Hägg, D.; Gatenholm, P. 3D Bioprinting Human Chondrocytes with Nanocellulose-Alginate Bioink for Cartilage Tissue Engineering Applications. Biomacromolecules 2015, 16, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Gorroñogoitia, I.; Urtaza, U.; Zubiarrain-Laserna, A.; Alonso-Varona, A.; Zaldua, A.M. A Study of the Printability of Alginate-Based Bioinks by 3D Bioprinting for Articular Cartilage Tissue Engineering. Polymers 2022, 14, 354. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, S.; Li, L. Rheological Study on 3D Printability of Alginate Hydrogel and Effect of Graphene Oxide. Int. J. Bioprinting 2016, 2, 54–66. [Google Scholar] [CrossRef]
- Gao, T.; Gillispie, G.J.; Copus, J.S.; Kumar, A.; Rajan, P.; Seol, Y.; Atala, A.; Yoo, J.J.; Lee, S.J. Optimization of Gelatin-Alginate Composite Bioink Printability Using Rheological Parameters: A Systematic Approach. Biofabrication 2019, 10, 034106. [Google Scholar] [CrossRef]
- Cooke, M.E.; Rosenzweig, D.H. The Rheology of Direct and Suspended Extrusion Bioprinting. APL Bioeng. 2021, 5, 11502. [Google Scholar] [CrossRef]
- Axpe, E.; Oyen, M.L. Applications of Alginate-Based Bioinks in 3D Bioprinting. Int. J. Mol. Sci. 2016, 17, 1976. [Google Scholar] [CrossRef] [Green Version]
- Tabriz, A.G.; Hermida, M.A.; Leslie, N.R.; Shu, W. Three-Dimensional Bioprinting of Complex Cell Laden Alginate Hydrogel Structures. Biofabrication 2015, 7, 45012. [Google Scholar] [CrossRef]
- Mirdamadi, E.; Tashman, J.W.; Shiwarski, D.J.; Palchesko, R.N.; Feinberg, A.W. FRESH 3D Bioprinting a Full-Size Model of the Human Heart. ACS Biomater. Sci. Eng. 2020, 6, 6453–6459. [Google Scholar] [CrossRef]
- Place, E.S.; Rojo, L.; Gentleman, E.; Sardinha, J.P.; Stevens, M.M. Strontium-and Zinc-Alginate Hydrogels for Bone Tissue Engineering. Tissue Eng. Part A 2011, 17, 2713–2722. [Google Scholar] [CrossRef]
- Iskandar, L.; Rojo, L.; Di Silvio, L.; Deb, S. The Effect of Chelation of Sodium Alginate with Osteogenic Ions, Calcium, Zinc, and Strontium. J. Biomater. Appl. 2019, 34, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Hernández, R.; Sacrista, J.; Mijangos, C. Sol/Gel Transition of Aqueous Alginate Solutions Induced by Fe 2 R Cations A. Macromol. Chem. Phys. 2010, 211, 1254–1260. [Google Scholar] [CrossRef]
- Wu, Y.; Yuan, Z.; Lin, W.; Wenger, A.C.; Tam, K.C.; Shirley, X. 3D Bioprinting of Liver-Mimetic Construct with Alginate / Cellulose Nanocrystal Hybrid Bioink. Bioprinting 2018, 9, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Yang, G.; Song, Y.; Xu, Y.; Zhao, S.; Zhang, W. 3D Bioprinted Integrated Osteochondral Scaffold-Mediated Repair of Articular Cartilage Defects in the Rabbit Knee. J. Med. Biol. Eng. 2020, 40, 71–81. [Google Scholar] [CrossRef]
- Thakur, A.; Kaur, H. Synthetic Chemistry of Cellulose Hydrogels—A Review. Mater. Today Proc. 2022, 48, 1431–1438. [Google Scholar] [CrossRef]
- Zainal, S.H.; Mohd, N.H.; Suhaili, N.; Anuar, F.H.; Lazim, A.M.; Othaman, R. Preparation of Cellulose-Based Hydrogel: A Review. J. Mater. Res. Technol. 2021, 10, 935–952. [Google Scholar] [CrossRef]
- Wang, C.; Bai, J.; Tian, P.; Xie, R.; Duan, Z.; Lv, Q.; Tao, Y. The Application Status of Nanoscale Cellulose-Based Hydrogels in Tissue Engineering and Regenerative Biomedicine. Front. Bioeng. Biotechnol. 2021, 9, 939. [Google Scholar] [CrossRef]
- Miao, X.; Lin, J.; Bian, F. Utilization of Discarded Crop Straw to Produce Cellulose Nanofibrils and Their Assemblies. J. Bioresour. Bioprod. 2020, 5, 26–36. [Google Scholar] [CrossRef]
- Yadav, C.; Saini, A.; Zhang, W.; You, X.; Chauhan, I.; Mohanty, P.; Li, X. Plant-Based Nanocellulose: A Review of Routine and Recent Preparation Methods with Current Progress in Its Applications as Rheology Modi Fi Er and 3D Bioprinting. Int. J. Biol. Macromol. 2021, 166, 1586–1616. [Google Scholar] [CrossRef]
- Du, L.; Zhong, T.; Wolcott, M.P.; Zhang, Y.; Qi, C.; Zhao, B.; Wang, J.; Yu, Z. Dispersing and Stabilizing Cellulose Nanoparticles in Acrylic Resin Dispersions with Unreduced Transparency and Changed Rheological Property. Cellulose 2018, 25, 2435–2450. [Google Scholar] [CrossRef]
- Zhong, F.; Nsor-Atindana, J. Chapter 16—Microcrystalline Cellulose and Nanocrystalline Cellulose. In Handbook of Hydrocolloids, 3rd ed.; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Sawston, UK, 2021; pp. 509–536. ISBN 978-0-12-820104-6. [Google Scholar]
- Jia, J.; Richards, D.J.; Pollard, S.; Tan, Y.; Rodriguez, J.; Visconti, R.P.; Trusk, T.C.; Yost, M.J.; Yao, H.; Markwald, R.R.; et al. Engineering Alginate as Bioink for Bioprinting. Acta Biomater. 2014, 10, 4323–4331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golunova, A.; Velychkivska, N.; Mikšovská, Z.; Chochola, V.; Jaroš, J.; Hampl, A.; Pop-Georgievski, O.; Proks, V. Direct and Indirect Biomimetic Peptide Modification of Alginate: Efficiency, Side Reactions, and Cell Response. Int. J. Mol. Sci. 2021, 22, 5731. [Google Scholar] [CrossRef] [PubMed]
- Dalheim, M.Ø.; Vanacker, J.; Najmi, M.A.; Aachmann, F.L.; Strand, B.L.; Christensen, B.E. Efficient Functionalization of Alginate Biomaterials. Biomaterials 2016, 80, 146–156. [Google Scholar] [CrossRef]
- Criado-Gonzalez, M.; Loftin, B.; Rodon Fores, J.; Vautier, D.; Kocgozlu, L.; Jierry, L.; Schaaf, P.; Boulmedais, F.; Harth, E. Enzyme Assisted Peptide Self-Assemblies Trigger Cell Adhesion in High Density Oxime Based Host Gels. J. Mater. Chem. B 2020, 8, 4419–4427. [Google Scholar] [CrossRef] [PubMed]
- Criado-Gonzalez, M.; Rodon Fores, J.; Wagner, D.; Schröder, A.P.; Carvalho, A.; Schmutz, M.; Harth, E.; Schaaf, P.; Jierry, L.; Boulmedais, F. Enzyme-Assisted Self-Assembly within a Hydrogel Induced by Peptide Diffusion. Chem. Commun. 2019, 55, 1156–1159. [Google Scholar] [CrossRef] [Green Version]
- Fores, J.R.; Bigo-Simon, A.; Wagner, D.; Payrastre, M.; Damestoy, C.; Blandin, L.; Boulmedais, F.; Kelber, J.; Schmutz, M.; Rabineau, M.; et al. Localized Enzyme-Assisted Self-Assembly in the Presence of Hyaluronic Acid for Hybrid Supramolecular Hydrogel Coating. Polymers 2021, 13, 1793. [Google Scholar] [CrossRef] [PubMed]
- Criado-Gonzalez, M.; Iqbal, M.H.; Carvalho, A.; Schmutz, M.; Jierry, L.; Schaaf, P.; Boulmedais, F. Surface Triggered Self-Assembly of Fmoc-Tripeptide as an Antibacterial Coating. Front. Bioeng. Biotechnol. 2020, 8, 938. [Google Scholar] [CrossRef] [PubMed]
- Criado-Gonzalez, M.; Espinosa-Cano, E.; Rojo, L.; Boulmedais, F.; Aguilar, M.R.; Hernández, R. Injectable Tripeptide/Polymer Nanoparticles Supramolecular Hydrogel: A Candidate for the Treatment of Inflammatory Pathologies. ACS Appl. Mater. Interfaces 2022, 14, 10068–10080. [Google Scholar] [CrossRef]
- Connell, C.O.; Ren, J.; Pope, L.; Li, Y.; Mohandas, A.; Blanchard, R.; Duchi, S.; Onofrillo, C. Characterizing Bioinks for Extrusion Bioprinting: Printability and Rheology. In 3D Bioprinting: Principles and Protocols, Methods in Molecular Biology; Humana: New York, NY, USA, 2020; Volume 2140, pp. 111–133. ISBN 9781071605202. [Google Scholar]
- Osswald, T.; Rudolph, N. Polymer Rheology Fundamentals and Applications; Strohm, C., Ed.; Hanser Publications: Munich, Germany, 2015; pp. 64–100. ISBN 978-1-56990-517-3. [Google Scholar]
- Freeman, F.E.; Kelly, D.J. Tuning Alginate Bioink Stiffness and Composition for Controlled Growth Factor Delivery and to Spatially Direct MSC Fate within Bioprinted Tissues. Sci. Rep. 2017, 7, 17042. [Google Scholar] [CrossRef] [Green Version]
- Cui, X.; Li, J.; Hartanto, Y.; Durham, M.; Tang, J.; Zhang, H.; Hooper, G.; Lim, K.; Woodfield, T. Advances in Extrusion 3D Bioprinting: A Focus on Multicomponent Hydrogel-Based Bioinks. Adv. Healthc. Mater. 2020, 9, 1901648. [Google Scholar] [CrossRef]
- Kyle, S.; Jessop, Z.M.; Al-Sabah, A.; Whitaker, I.S. ‘Printability’ of Candidate Biomaterials for Extrusion Based 3D Printing: State-of-the-Art. Adv. Healthc. Mater. 2017, 6, 1700264. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ramos, A.; Li, M.; Li, Z.; Bhatta, S.; Jeyaseelan, A. Improvement of Cell Deposition by Self-Absorbent Capability of Freeze- Dried 3D-Bioprinted Scaffolds Derived from Cellulose Material-Alginate Hydrogels. Biomed. Phys. Eng. Express 2020, 6, 045009. [Google Scholar] [CrossRef] [PubMed]
- Fermani, M.; Platania, V.; Kavasi, R.; Karavasili, C.; Zgouro, P.; Fatouros, D.; Chatzinikolaidou, M.; Bouropoulos, N. 3D-Printed Scaffolds from Alginate / Methyl Cellulose / Trimethyl Chitosan / Silicate Glasses for Bone Tissue Engineering. Appl. Sci. 2021, 11, 8677. [Google Scholar] [CrossRef]
- Koch, M.; Włodarczyk-Biegun, M.K. Faithful Scanning Electron Microscopic (SEM) Visualization of 3D Printed Alginate-Based Scaffolds. Bioprinting 2020, 20, e00098. [Google Scholar] [CrossRef]
- Liu, Q.; Li, Q.; Xu, S.; Zheng, Q.; Cao, X. Preparation and Properties of 3D Printed Alginate–Chitosan Polyion Complex Hydrogels for Tissue Engineering. Polymers 2018, 10, 664. [Google Scholar] [CrossRef] [Green Version]
- Mahmoud, Y.; Safidine, Z.; Belhaneche-Bensemra, N. Characterization of Microcrystalline Cellulose Extracted from Walnut and Apricots Shells by Alkaline Treatment. J. Serb. Chem. Soc. 2021, 86, 521–532. [Google Scholar] [CrossRef]
- Gómez-Ordóñez, E.; Rupérez, P. FTIR-ATR Spectroscopy as a Tool for Polysaccharide Identification in Edible Brown and Red Seaweeds. Food Hydrocoll. 2011, 25, 1514–1520. [Google Scholar] [CrossRef]
- Müller, G.; Schöpper, C.; Vos, H.; Kharazipour, A.; Polle, A. FTIR-ATR Spectroscopic Analyses of Changes in Wood Properties during Particleand Fibreboard Production of Hardand Softwood Trees. BioResources 2009, 4, 49–71. [Google Scholar] [CrossRef]
- Abidi, N.; Cabrales, L.; Haigler, C.H. Changes in the Cell Wall and Cellulose Content of Developing Cotton Fibers Investigated by FTIR Spectroscopy. Carbohydr. Polym. 2014, 100, 9–16. [Google Scholar] [CrossRef]
- Oyeoka, H.C.; Ewulonu, C.M.; Nwuzor, I.C.; Obele, C.M.; Nwabanne, J.T. Packaging and Degradability Properties of Polyvinyl Alcohol/Gelatin Nanocomposite Films Filled Water Hyacinth Cellulose Nanocrystals. J. Bioresour. Bioprod. 2021, 6, 168–185. [Google Scholar] [CrossRef]
- Criado-Gonzalez, M.; Fores, J.R.; Carvalho, A.; Blanck, C.; Schmutz, M.; Kocgozlu, L.; Schaaf, P.; Jierry, L.; Boulmedais, F. Phase Separation in Supramolecular Hydrogels Based on Peptide Self-Assembly from Enzyme-Coated Nanoparticles. Langmuir 2019, 35, 10838–10845. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wei, D.; Zhu, Y.; Zhong, M.; Zuo, Y.; Fan, H.; Zhang, X. A Spatial Patternable Macroporous Hydrogel with Cell-Affinity Domains to Enhance Cell Spreading and Differentiation. Biomaterials 2014, 35, 4759–4768. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Sosa, A.; Ramírez-Jiménez, R.A.; Rojo, L.; Boulmedais, F.; Aguilar, M.R.; Criado-Gonzalez, M.; Hernández, R. Optimization of the Rheological Properties of Self-Assembled Tripeptide/Alginate/Cellulose Hydrogels for 3D Printing. Polymers 2022, 14, 2229. https://doi.org/10.3390/polym14112229
Hernández-Sosa A, Ramírez-Jiménez RA, Rojo L, Boulmedais F, Aguilar MR, Criado-Gonzalez M, Hernández R. Optimization of the Rheological Properties of Self-Assembled Tripeptide/Alginate/Cellulose Hydrogels for 3D Printing. Polymers. 2022; 14(11):2229. https://doi.org/10.3390/polym14112229
Chicago/Turabian StyleHernández-Sosa, Alejandro, Rosa Ana Ramírez-Jiménez, Luis Rojo, Fouzia Boulmedais, María Rosa Aguilar, Miryam Criado-Gonzalez, and Rebeca Hernández. 2022. "Optimization of the Rheological Properties of Self-Assembled Tripeptide/Alginate/Cellulose Hydrogels for 3D Printing" Polymers 14, no. 11: 2229. https://doi.org/10.3390/polym14112229
APA StyleHernández-Sosa, A., Ramírez-Jiménez, R. A., Rojo, L., Boulmedais, F., Aguilar, M. R., Criado-Gonzalez, M., & Hernández, R. (2022). Optimization of the Rheological Properties of Self-Assembled Tripeptide/Alginate/Cellulose Hydrogels for 3D Printing. Polymers, 14(11), 2229. https://doi.org/10.3390/polym14112229








