Effect of Textile Wastewater Secondary Effluent on UF Membrane Characteristics
Abstract
1. Introduction
2. Materials and Methods
2.1. Membranes
2.2. Textile Wastewater Treatment
2.3. Membrane Characterization
3. Results and Discussion
3.1. TWW Secondary Effluent Characteristics
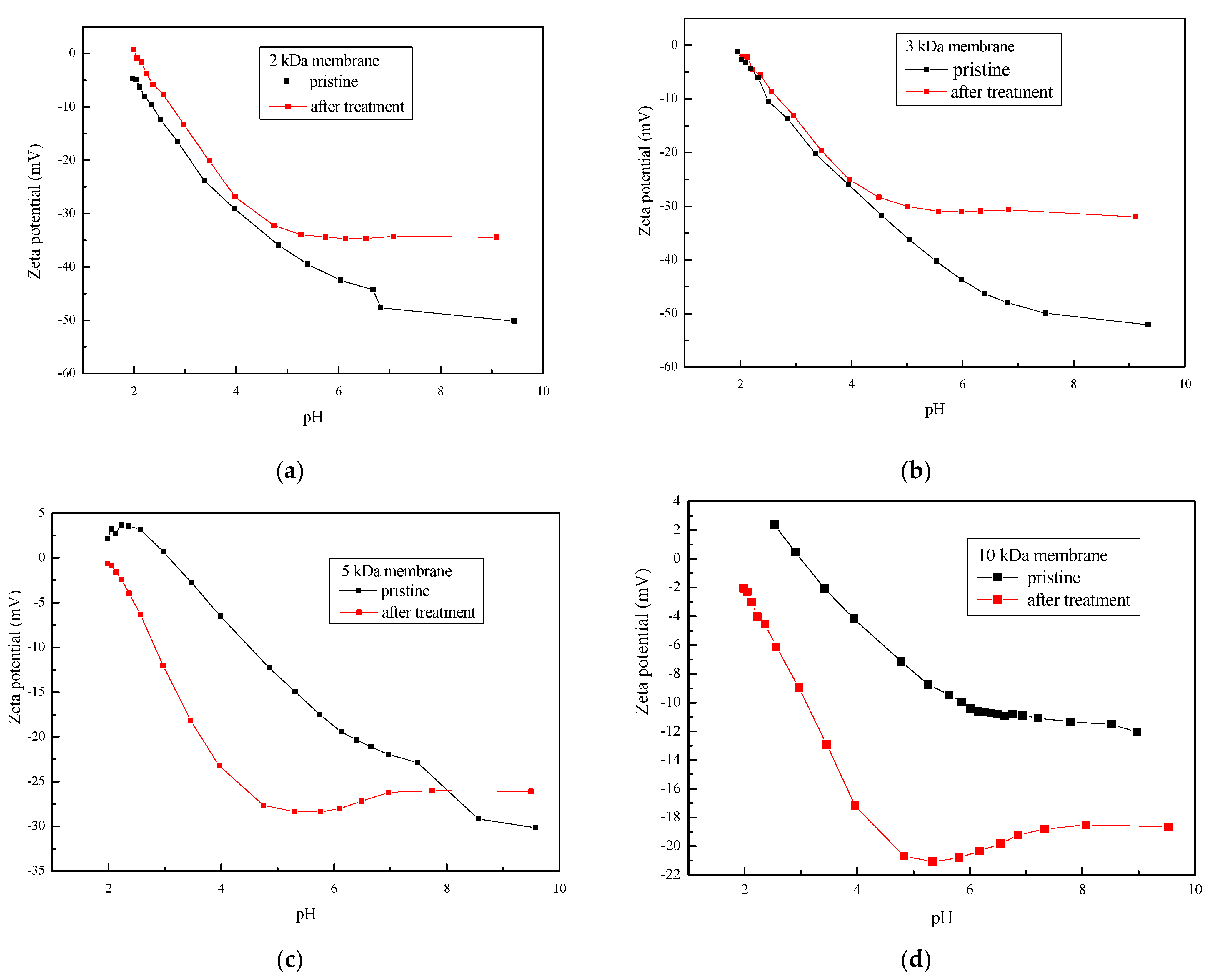
3.2. Membrane Surface Charge
3.3. Hydrophilicity of the Membrane
3.4. Membrane Morphology
3.5. Surface Structure
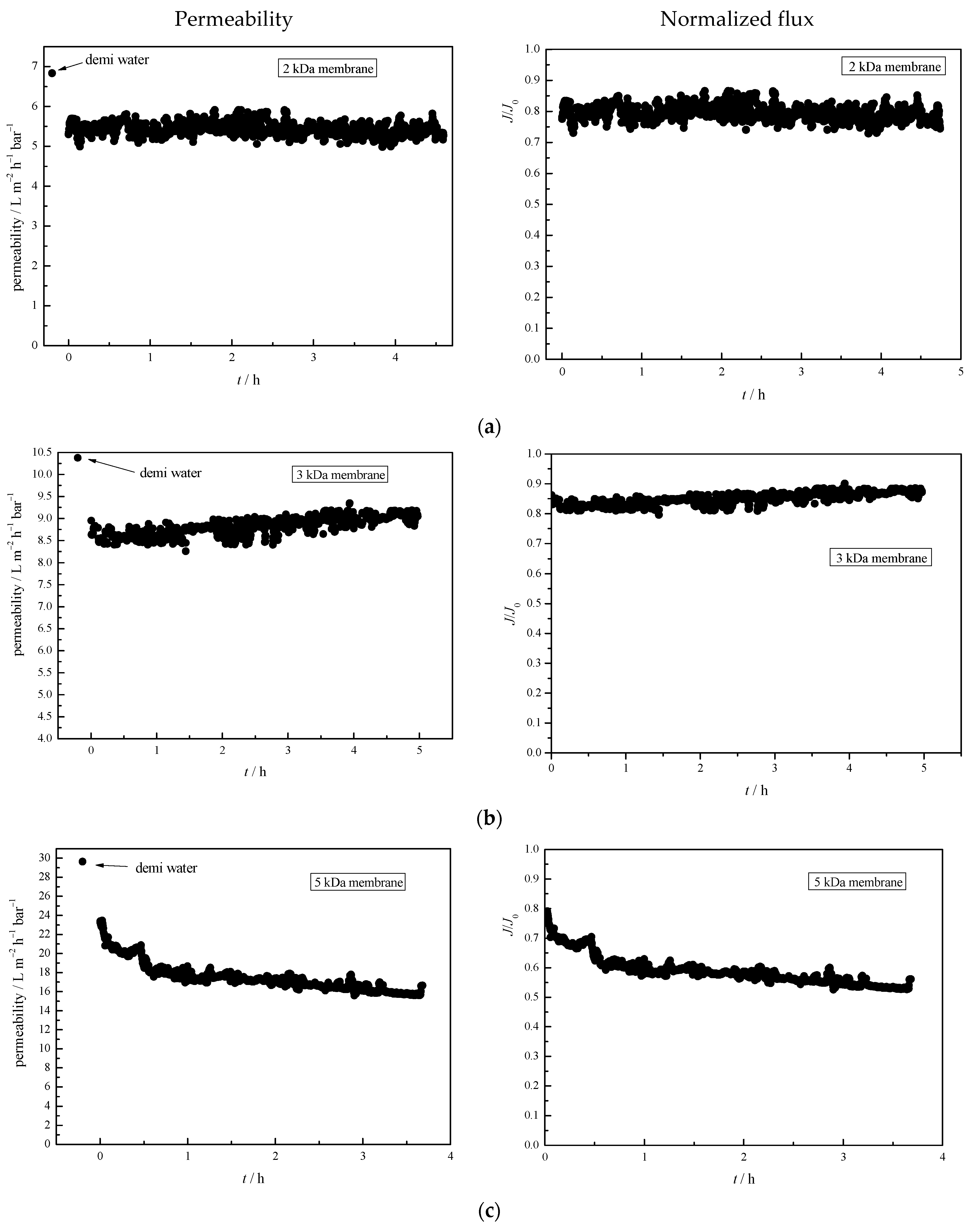
3.6. Membrane Permeability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Keskin, B.; Ersahin, M.E.; Ozgun, H.; Koyuncu, I. Pilot and full-scale applications of membrane processes for textile wastewater treatment: A critical review. J. Water Process Eng. 2021, 42, 102172. [Google Scholar] [CrossRef]
- Ağtaş, M.; Yılmaz, Ö.; Dilaver, M.; Alp, K.; Koyuncu, I. Hot water recovery and reuse in textile sector with pilot scale ceramic ultrafiltration/nanofiltration membrane system. J. Clean. Prod. 2020, 256, 120359. [Google Scholar] [CrossRef]
- Hassanzadeh, E.; Farhadian, M.; Razmjou, A.; Askari, N. An efficient wastewater treatment approach for a real woolen textile industry using a chemical assisted NF membrane process. Environ. Nanotechnol. Monit. Manag. 2017, 8, 92–96. [Google Scholar] [CrossRef]
- Nadeem, K.; Guyer, G.T.; Keskinler, B.; Dizge, N. Investigation of segregated wastewater streams reusability with membraneprocess for textile industry. J. Clean. Prod. 2019, 228, 1437–1445. [Google Scholar] [CrossRef]
- Ağtaş, M.; Yılmaz, Ö.; Dilaver, M.; Alp, K.; Koyuncu, İ. Pilot-scale ceramic ultrafiltration/nanofiltration membrane system application for caustic recovery and reuse in textile sector. Environ. Sci. Pollut. Res. 2021, 28, 41029–41038. [Google Scholar] [CrossRef]
- Koseoglu-Imer, D.Y. The determination of performances of polysulfone (PS) ultrafiltration membranes fabricated at different evaporation temperatures for the pretreatment of textile wastewater. Desalination 2013, 316, 110–119. [Google Scholar] [CrossRef]
- Dasgupta, J.; Sikder, J.; Chakraborty, S.; Curcio, S.; Drioli, E. Remediation of textile effluents by membrane based treatment techniques: A state of the art review. J. Environ. Manag. 2015, 147, 55–72. [Google Scholar] [CrossRef]
- Li, N.N.; Fane, A.G.; Ho, W.W.; Matsuura, T. Advanced Membrane Technology and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Zhang, J.; Chen, G.; Ma, Y.; Xu, M.; Qin, S.; Liu, X.; Feng, H.; Hou, L. Purification of pickling wastewater from the steel industry using membrane filters: Performance and membrane fouling. Environ. Eng. Res. 2020, 27, 200486. [Google Scholar] [CrossRef]
- Thuvander, J.; Zarebska, A.; Hélix-Nielsen, C.; Jönsson, A.-S. Characterization of irreversible fouling after ultrafiltration of thermomechanical pulp mill process water. J. Wood Chem. Technol. 2018, 38, 276–285. [Google Scholar] [CrossRef]
- Shon, H.; Vigneswaran, S.; Kim, I.S.; Cho, J.; Ngo, H. Fouling of ultrafiltration membrane by effluent organic matter: A detailed characterization using different organic fractions in wastewater. J. Membr. Sci. 2006, 278, 232–238. [Google Scholar] [CrossRef]
- Srivastava, H.P.; Arthanareeswaran, G.; Anantharaman, N.; Starov, V.M. Performance of modified poly (vinylidene fluoride) membrane for textile wastewater ultrafiltration. Desalination 2011, 282, 87–94. [Google Scholar] [CrossRef]
- Fersi, C.; Dhahbi, M. Treatment of textile plant effluent by ultrafiltration and/or nanofiltration for water reuse. Desalination 2008, 222, 263–271. [Google Scholar] [CrossRef]
- Ćurić, I.; Dolar, D.; Bošnjak, J. Reuse of textile wastewater for dyeing cotton knitted fabric with hybrid treatment: Coagulation/sand filtration/UF/NF-RO. J. Environ. Manag. 2021, 295, 113133. [Google Scholar] [CrossRef]
- Ćurić, I.; Dolar, D.; Karadakić, K. Textile wastewater reusability in knitted fabric washing process using UF membrane technology. J. Clean. Prod. 2021, 299, 126899. [Google Scholar] [CrossRef]
- Racar, M.; Dolar, D.; Farkaš, M.; Milčić, N.; Špehar, A.; Košutić, K. Wastewater reuse for irrigation by coagulation and ultrafiltration. TC 2017, 66, 85-00. [Google Scholar]
- Hao, O.J.; Kim, H.; Chiang, P.-C. Decolorization of wastewater. Crit. Rev. Environ. Sci. Technol. 2000, 30, 449–505. [Google Scholar] [CrossRef]
- Kumar, R.; Ismail, A. Fouling control on microfiltration/ultrafiltration membranes: Effects of morphology, hydrophilicity, and charge. J. Appl. Polym. Sci. 2015, 132, 42042. [Google Scholar] [CrossRef]
- Lawrence, N.D.; Perera, J.M.; Iyer, M.; Hickey, M.W.; Stevens, G.W. The use of streaming potential measurements to study the fouling and cleaning of ultrafiltration membranes. Sep. Purif. Technol. 2006, 48, 106–112. [Google Scholar] [CrossRef]
- Manawi, Y.; Kochkodan, V.; Mahmoudi, E.; Johnson, D.J.; Mohammad, A.W.; Atieh, M.A. Characterization and separation performance of a novel polyethersulfone membrane blended with acacia gum. Sci. Rep. 2017, 7, 15831. [Google Scholar] [CrossRef]
- Hegoburu, I.; Zedda, K.L.; Velizarov, S. Treatment of electroplating wastewater using nf ph-stable membranes: Characterization and application. Membranes 2020, 10, 399. [Google Scholar] [CrossRef]
- Ru, J.; Qian, X.; Wang, Y. Low-salt or salt-free dyeing of cotton fibers with reactive dyes using liposomes as dyeing/level-dyeing promotors. Sci. Rep. 2018, 8, 13045. [Google Scholar] [CrossRef] [PubMed]
- Kramer, F.; Shang, R.; Rietveld, L.; Heijman, S. Influence of pH, multivalent counter ions, and membrane fouling on phosphate retention during ceramic nanofiltration. Sep. Purif. Technol. 2019, 227, 115675. [Google Scholar] [CrossRef]
- Baek, Y.; Kang, J.; Theato, P.; Yoon, J. Measuring hydrophilicity of RO membranes by contact angles via sessile drop and captive bubble method: A comparative study. Desalination 2012, 303, 23–28. [Google Scholar] [CrossRef]
- Świerczyńska, A.; Bohdziewicz, J.; Kamińska, G.; Wojciechowski, K. Influence of the type of membrane-forming polymer on the membrane fouling. Environ. Prot. Eng. 2016, 42, 197–210. [Google Scholar] [CrossRef]
- Salgin, S.; Salgin, U.; Soyer, N. Streaming potential measurements of polyethersulfone ultrafiltration membranes to determine salt effects on membrane zeta potential. Int. J. Electrochem. Sci 2013, 8, 4073–4084. [Google Scholar]
- Sadeghi, I.; Aroujalian, A.; Raisi, A.; Fathizadeh, M.; Dabir, B. Effect of solvent, hydrophilic additives and corona treatment on performance of polyethersulfone UF membranes for oil/water separation. Procedia Eng. 2012, 44, 1539–1541. [Google Scholar] [CrossRef][Green Version]
- Sagle, A.C.; Van Wagner, E.M.; Ju, H.; McCloskey, B.D.; Freeman, B.D.; Sharma, M.M. PEG-coated reverse osmosis membranes: Desalination properties and fouling resistance. J. Membr. Sci. 2009, 340, 92–108. [Google Scholar] [CrossRef]
- Elcik, H.; Fortunato, L.; Vrouwenvelder, J.S.; Ghaffour, N. Real-time membrane fouling analysis for the assessment of reclamation potential of textile wastewater processed by membrane distillation. J. Water Process Eng. 2021, 43, 102296. [Google Scholar] [CrossRef]







| GH | GK | PT | PU | |
|---|---|---|---|---|
| Membrane | UF | UF | UF | UF |
| MWCO, kDa | 2 | 3 | 5 | 10 |
| Polymer 1 | PA | PA | PES/PSf | PES/PSf |
| Maximum operating pressure, bar | 27 | 27 | 10 | 10 |
| Maximum operating temperature, ℃ | 70 | 70 | 70 | 70 |
| pH range, continuous operation | 1–11 | 1–11 | 1–11 | 1–11 |
| Working pressure 2, bar | 10 | 5 | 4 | 2 |
| Parameter | Average 1 | SD 2 | Parameter | Average | SD |
|---|---|---|---|---|---|
| pH | 8.78 | 0.12 | 525 | 36 | 3 |
| Conductivity, mS cm−1 | 3.33 | 0.10 | 620 | 30 | 3 |
| Turbidity, NTU | 1.37 | 0.77 | Chloride, mg L−1 | 69.5 | 3.2 |
| COD, mg L−1 | 168 | 22 | Sulphate, mg L−1 | 1228.6 | 216.9 |
| TC, mg L−1 | 319.2 | 2.2 | Sodium, mg L−1 | 189.3 | 6.8 |
| IC, mg L−1 | 218.4 | 4.6 | Potassium, mg L−1 | 136.8 | 8.1 |
| TOC, mg L−1 | 100.9 | 4.9 | Magnesium, mg L−1 | 9.9 | 0.3 |
| TNb, mg L−1 | 19.6 | 4.2 | Calcium, mg L−1 | 48.7 | 2.8 |
| SAC, 1 m−1 | Hardness, mg L−1 CaCO3 | 162.3 | 7.5 | ||
| 436 | 62 | 3 | Hardness, D | 9.1 | 0.4 |
| Membrane | Contact Angle/° | |
|---|---|---|
| GH | Pristine | 54.61 ± 1.74 |
| After treatment | 61.86 ± 2.82 | |
| GK | Pristine | 54.10 ± 2.63 |
| After treatment | 56.52 ± 1.80 | |
| PT | Pristine | 58.06 ± 3.58 |
| After treatment | 64.82 ± 2.92 | |
| PU | Pristine | 46.19 ± 1.77 |
| After treatment | 64.09 ± 2.93 | |
| Membrane | Sa/nm | Sq/nm | |
|---|---|---|---|
| 2 kDa | Pristine | 8.59 | 14.21 |
| After treatment | 31.38 | 39.04 | |
| 3 kDa | Pristine | 18.09 | 23.20 |
| After treatment | 30.41 | 39.07 | |
| 5 kDa | Pristine | 13.76 | 20.59 |
| After treatment | 36.14 | 46.84 | |
| 10 kDa | Pristine | 13.91 | 22.09 |
| After treatment | 19.66 | 28.86 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ćurić, I.; Dolar, D.; Horvat, J.; Grgić, K. Effect of Textile Wastewater Secondary Effluent on UF Membrane Characteristics. Polymers 2022, 14, 2035. https://doi.org/10.3390/polym14102035
Ćurić I, Dolar D, Horvat J, Grgić K. Effect of Textile Wastewater Secondary Effluent on UF Membrane Characteristics. Polymers. 2022; 14(10):2035. https://doi.org/10.3390/polym14102035
Chicago/Turabian StyleĆurić, Iva, Davor Dolar, Josip Horvat, and Katia Grgić. 2022. "Effect of Textile Wastewater Secondary Effluent on UF Membrane Characteristics" Polymers 14, no. 10: 2035. https://doi.org/10.3390/polym14102035
APA StyleĆurić, I., Dolar, D., Horvat, J., & Grgić, K. (2022). Effect of Textile Wastewater Secondary Effluent on UF Membrane Characteristics. Polymers, 14(10), 2035. https://doi.org/10.3390/polym14102035







