In Vivo Investigation of Polymer-Ceramic PCL/HA and PCL/β-TCP 3D Composite Scaffolds and Electrical Stimulation for Bone Regeneration
Abstract
:1. Introduction
2. Experimental Methods
2.1. Scaffold Fabrication
2.2. In-Vivo Study
Animals
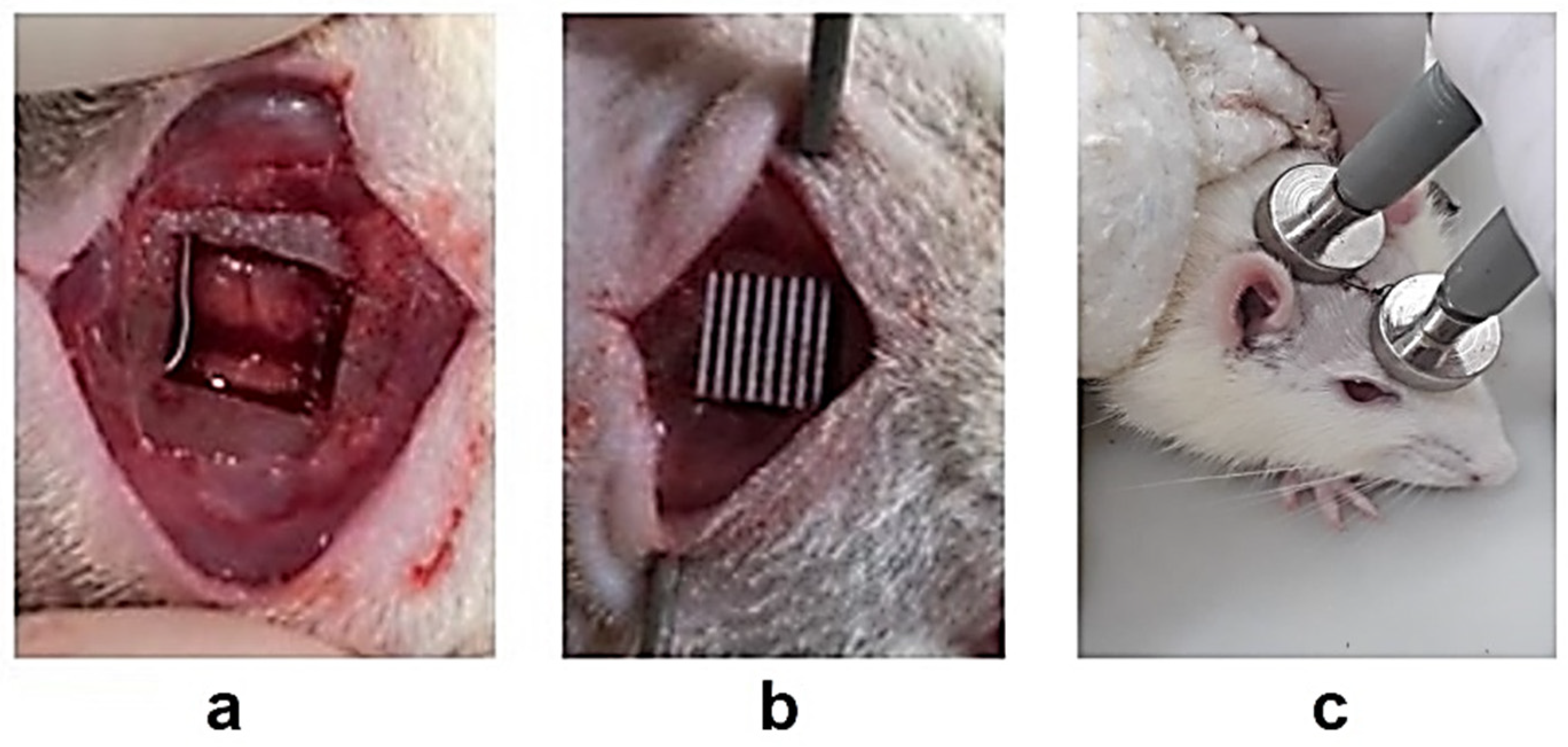
2.3. Surgical Protocol
2.4. Electrical Stimulation and Post Treatment
2.5. Histomorphometry
2.6. Immunohistochemistry
2.7. Quantitative Polymerase Chain Reaction (qPCR)
2.8. Statistical Analysis
3. Results
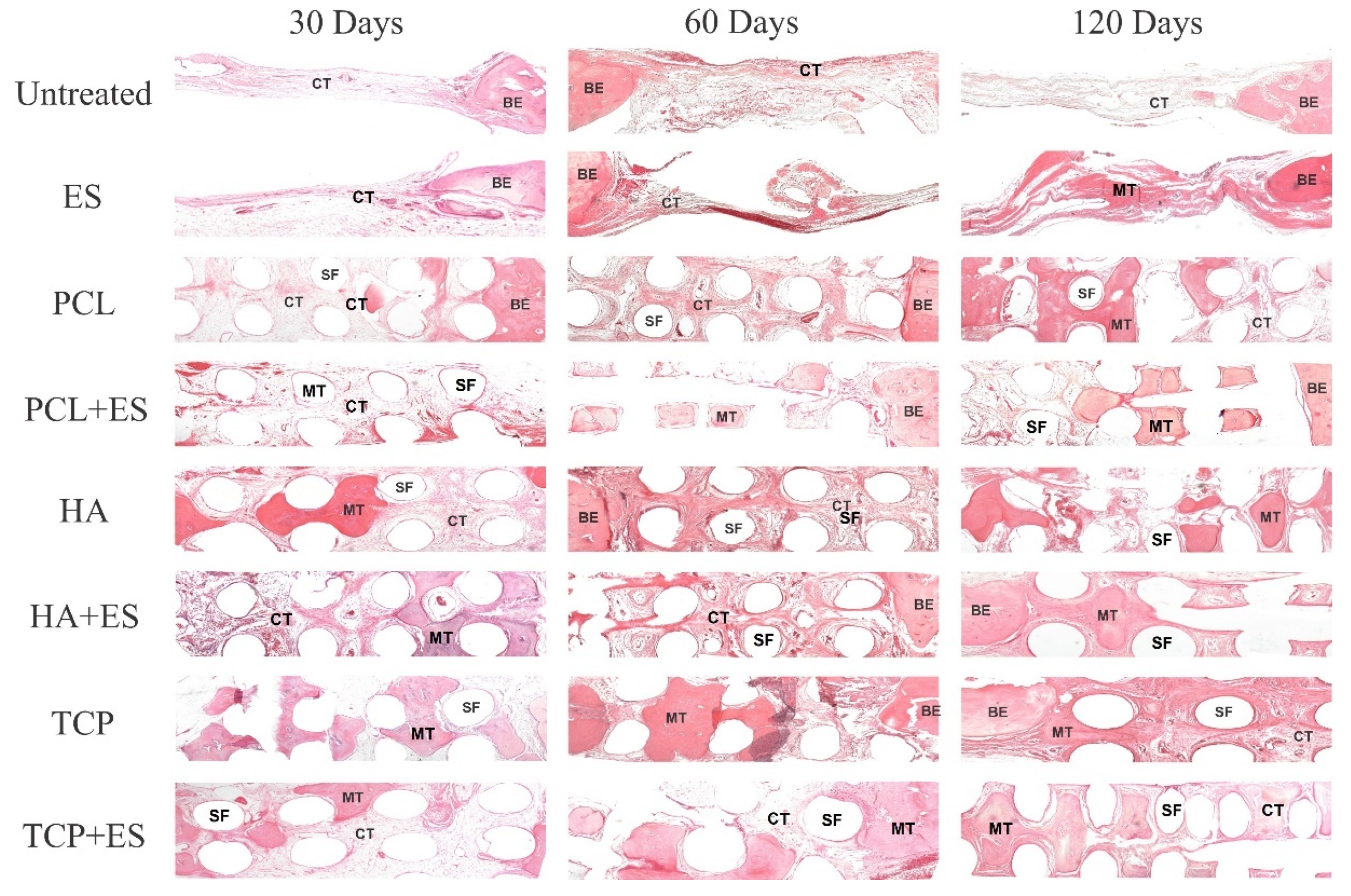
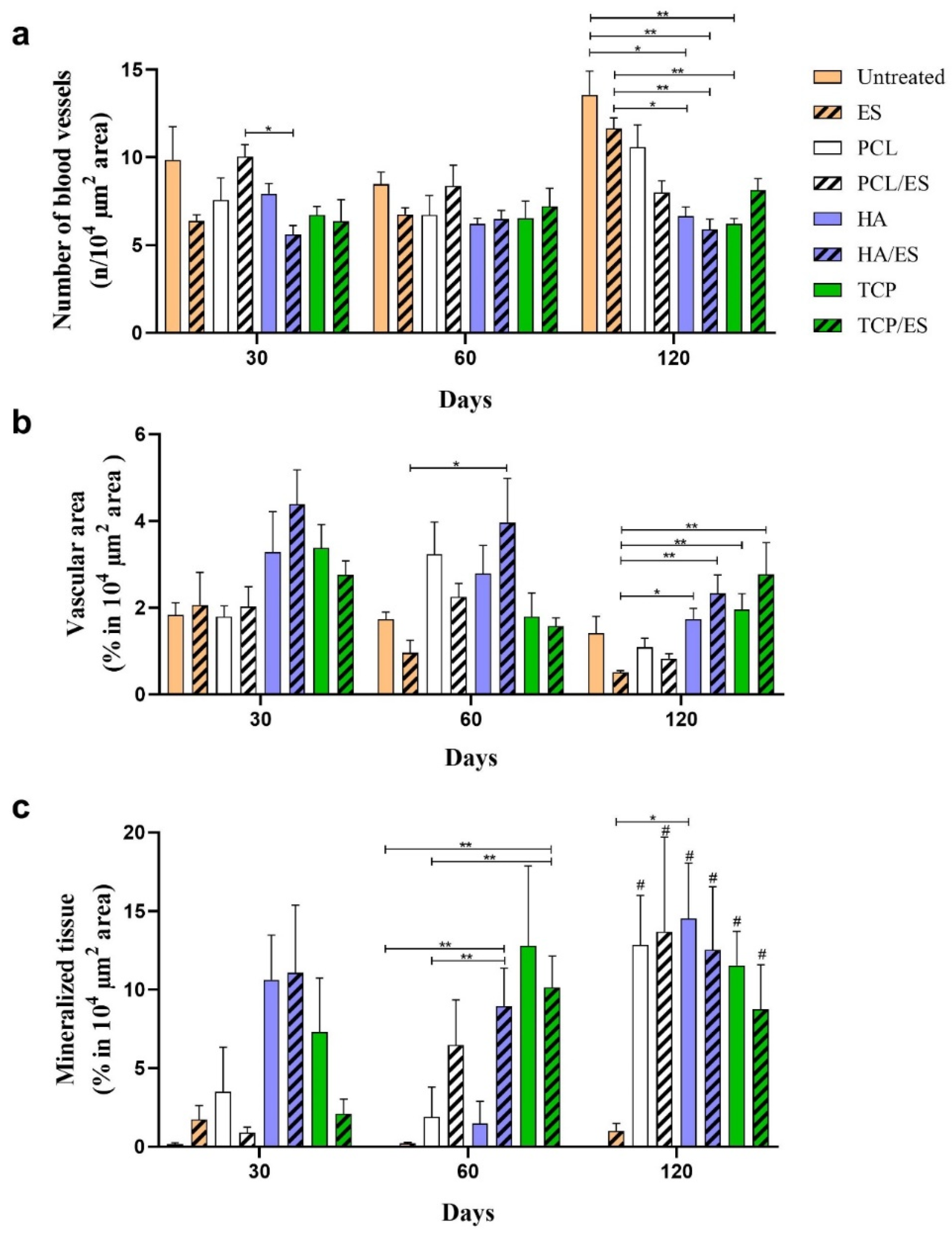
3.1. Histomorphometry
3.2. Osteogenic Gene Expression
3.3. Bone Remodelling
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fernandez-Yague, M.A.; Abbah, S.A.; McNamara, L.; Zeugolis, D.I.; Pandit, A.; Biggs, M.J. Biomimetic Approaches in Bone Tissue Engineering: Integrating Biological and Physicomechanical Strategies. Adv. Drug Deliv. Rev. 2015, 84, 1–29. [Google Scholar] [CrossRef]
- Leppik, L.; Oliveira, K.M.C.; Bhavsar, M.B.; Barker, J.H. Electrical Stimulation in Bone Tissue Engineering Treatments. Eur. J. Trauma Emerg. Surg. 2020, 46, 231–244. [Google Scholar] [CrossRef] [Green Version]
- Dumic-Cule, I.; Peric, M.; Kucko, L.; Grgurevic, L.; Pecina, M.; Vukicevic, S. Bone Morphogenetic Proteins in Fracture Repair. Int. Orthop. 2018, 42, 2619–2626. [Google Scholar] [CrossRef] [Green Version]
- Chronopoulou, L.; Cacciotti, I.; Amalfitano, A.; Di Nitto, A.; D’Arienzo, V.; Nocca, G.; Palocci, C. Biosynthesis of innovative calcium phosphate/hydrogel composites: Physicochemical and biological characterisation. Nanotechnology 2021, 32, 095102. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Jang, T.S.; Pan, H.M.; Jung, H.D.; Sia, M.W.; Xie, S.; Song, J. 3D freeform printing of nanocomposite hydrogels through in situ precipitation in reactive viscous fluid. Int. J. Bioprint. 2020, 6, 29–49. [Google Scholar]
- Kargozar, S.; Mozafari, M.; Hamzehlou, S.; Brouki Milan, P.; Kim, H.; Baino, F. Bone Tissue Engineering Using Human Cells: A Comprehensive Review on Recent Trends, Current Prospects, and Recommendations. Appl. Sci. 2019, 9, 174. [Google Scholar] [CrossRef] [Green Version]
- Huang, B.; Caetano, G.; Vyas, C.; Blaker, J.J.; Diver, C.; Bártolo, P. Polymer-Ceramic Composite Scaffolds: The Effect of Hydroxyapatite and β-tri-Calcium Phosphate. Materials 2018, 11, 129. [Google Scholar] [CrossRef] [Green Version]
- Lohfeld, S.; Cahill, S.; Barron, V.; McHugh, P.; Dürselen, L.; Kreja, L.; Bausewein, C.; Ignatius, A. Fabrication, Mechanical and In Vivo Performance of Polycaprolactone/Tricalcium Phosphate Composite Scaffolds. Acta Biomater. 2012, 8, 3446–3456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denry, I.; Kuhn, L.T. Design and Characterization of Calcium Phosphateceramic Scaffolds for Bone Tissue Engineering. Dent. Mater. 2016, 32, 43–53. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.C.F.M.; Lima, M.G.; de Almeida Lima, L.H.M.; Cordeiro, V.V.; de Souto Viana, K.M.; de Souza, C.V.; de Lucena, H. Hidroxiapatita: Obtenção, caracterização e aplicações. Rev. Eletrôn. Mater. Process. 2009, 4, 29–38. [Google Scholar]
- Cacciotti, I. Multisubstituted hydroxyapatite powders and coatings: The influence of the codoping on the hydroxyapatite performances. J. Appl. Ceram. Technol. 2019, 6, 1864–1884. [Google Scholar] [CrossRef]
- Antoniac, I.V. (Ed.) Cationic and Anionic substitutions in hydroxyapatite. In Handbook of Bioceramics and Biocomposites; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 145–211. [Google Scholar]
- Bianco, A.; Di Federico, E.; Cacciotti, I. Electrospun poly(ε-caprolactone)-based composites using synthesized β-tricalcium phosphate. Polym. Adv. Technol. 2011, 22, 1832–1841. [Google Scholar] [CrossRef]
- Dorozhkin, S.V.; Epple, M. Biological and Medical Significance of Calcium Phosphates. Angew. Chem. Int. Ed. 2002, 41, 3130–3146. [Google Scholar] [CrossRef]
- Samavedi, S.; Whittington, A.R.; Goldstein, A.S. Calcium Phosphate Ceramics in Bone Tissue Engineering: A Review of Properties and Their Influence on Cell Behavior. Acta Biomater. 2013, 9, 8037–8045. [Google Scholar] [CrossRef]
- Gittens, R.A.; Olivares-Navarrete, R.; Tannenbaum, R.; Boyan, B.D.; Schwartz, Z. Electrical Implications of Corrosion for Osseointegration of Titanium Implants. J. Dent. Res. 2011, 90, 1389–1397. [Google Scholar] [CrossRef] [Green Version]
- Mata, D.; Horovistiz, A.L.; Branco, I.; Ferro, M.; Ferreira, N.M.; Belmonte, M.; Lopes, M.A.; Silva, R.F.; Oliveira, F.J. Carbon Nanotube-Based Bioceramic Grafts for Electrotherapy of Bone. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 34, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Leppik, L.; Zhihua, H.; Mobini, S.; Thottakkattumana Parameswaran, V.; Eischen-Loges, M.; Slavici, A.; Helbing, J.; Pindur, L.; Oliveira, K.M.C.; Bhavsar, M.B.; et al. Combining Electrical Stimulation and Tissue Engineering to Treat Large Bone Defects in a Rat Model. Sci. Rep. 2018, 8, 6307. [Google Scholar] [CrossRef]
- Ferrigno, B.; Bordett, R.; Duraisamy, N.; Moskow, J.; Arul, M.R.; Rudraiah, S.; Nukavarapu, S.P.; Vella, A.T.; Kumbar, S.G. Bioactive Polymeric Materials and Electrical Stimulation Strategies for Musculoskeletal Tissue Repair and Regeneration. Bioact. Mater. 2020, 5, 468–485. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.P.; Huang, B.; Helaehil, J.V.; Nalesso, P.R.L.; Bagne, L.; de Oliveira, M.A.; Albiazetti, G.C.C.; Aldalbahi, A.; El-Newehy, M.; Santamaria, M., Jr.; et al. In Vivo Study of Conductive 3D Printed PCL/MWCNTs Scaffolds With Electrical Stimulation for Bone Tissue Engineering. Bio-Des. Manuf. 2021, 4, 190–202. [Google Scholar] [CrossRef]
- Bagne, L.; Oliveira, M.A.; Pereira, A.T.; Caetano, G.F.; Oliveira, C.A.; Aro, A.A.; Chiarotto, G.B.; Santos, G.M.T.; Mendonça, F.A.S.; Santamaria, M., Jr. Electrical Therapies Act on the Ca2+/CaM Signaling Pathway to Enhance Bone Regeneration With Bioactive Glass [S53P4] and Allogeneic Grafts. J. Biomed. Mater. Res. 2021, 109, 2104–2116. [Google Scholar] [CrossRef]
- Nalesso, P.R.L.; Wang, W.; Hou, Y.; Bagne, L.; Pereira, A.T.; Helaehil, J.V.; Andrade, T.A.M.; Chiarotto, G.; Bartolo, P.; Caetano, G.F. In Vivo Investigation of 3D Printed Polycaprolactone/Graphene Electro-Active Bone Scaffolds. Bioprinting 2021, 24, e00164. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Muschler, G.F.; Raut, V.P.; Patterson, T.E.; Wenke, J.C.; Hollinger, J.O. The Design and Use of Animal Models for Translational Research in Bone Tissue Engineering and Regenerative Medicine. Tissue Eng. Part B Rev. 2010, 16, 123–145. [Google Scholar] [CrossRef] [Green Version]
- Kleinhans, C.; Mohan, R.R.; Vacun, G.; Schwarz, T.; Haller, B.; Sun, Y.; Kahlig, A.; Kluger, P.; Finne-Wistrand, A.; Walles, H.; et al. A Perfusion Bioreactor System Efficiently Generates Cell-Loaded Bone Substitute Materials for Addressing Critical Size Bone Defects. Biotechnol. J. 2015, 10, 1727–1738. [Google Scholar] [CrossRef] [Green Version]
- Fonseca, J.H.; Bagne, L.; Meneghetti, D.H.; Dos Santos, G.M.T.; Esquisatto, M.A.M.; de Andrade, T.A.M.; do Amaral, M.E.C.; Felonato, M.; Caetano, G.F.; Santamaria, M., Jr.; et al. Electrical stimulation: Complementary therapy to improve the performance of grafts in bone defects? J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 924–932. [Google Scholar] [CrossRef]
- Sartika, D.; Wang, C.H.; Wang, D.H.; Cherng, J.H.; Chang, S.J.; Fan, G.Y.; Wang, Y.W.; Lee, C.H.; Hong, P.D.; Wang, C.C. Human Adipose-Derived Mesenchymal Stem Cells-Incorporated Silk Fibroin as a Potential Bio-Scaffold in Guiding Bone Regeneration. Polymers 2020, 12, 853. [Google Scholar] [CrossRef] [Green Version]
- Brunello, G.; Panda, S.; Schiavon, L.; Sivolella, S.; Biasetto, L.; Del Fabbro, M. The Impact of Bioceramic Scaffolds on Bone Regeneration in Preclinical In Vivo Studies: A Systematic Review. Materials 2020, 13, 1500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Junior, J.R.P.; Nalesso, P.R.L.; Musson, D.; Cornish, J.; Mendonça, F.; Caetano, G.F.; Bártolo, P. Engineered 3D Printed Poly(ɛ-caprolactone)/Graphene Scaffolds for Bone Tissue Engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 100, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Bins-Ely, L.M.; Cordero, E.B.; Souza, J.C.M.; Teughels, W.; Benfatti, C.A.M.; Magini, R.S. In Vivo Electrical Application on Titanium Implants Stimulating Bone Formation. J. Periodont. Res. 2017, 52, 479–484. [Google Scholar] [CrossRef]
- Zhang, J.; Li, M.; Kang, E.T.; Neoh, K.G. Electrical Stimulation of Adipose-Derived Mesenchymal Stem Cells in Conductive Scaffolds and the Roles of Voltage-Gated Ion Channels. Acta Biomater. 2016, 32, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Kim, G. The Effect of Sinusoidal AC Electric Stimulation of 3D PCL/CNT and PCL/β-TCP Based Bio-Composites on Cellular Activities for Bone Tissue Regeneration. J. Mater. Chem. B 2013, 1, 1439–1452. [Google Scholar] [CrossRef]
- Nakashima, K.; Zhou, X.; Kunkel, G.; Zhang, Z.; Deng, J.M.; Behringer, R.R.; de Crombrugghe, B. The Novel Zinc Finger-Containing Transcription Factor Osterix Is Required for Osteoblast Differentiation and Bone Formation. Cell 2002, 108, 17–29. [Google Scholar] [CrossRef] [Green Version]
- Tang, W.; Yang, F.; Li, Y.; de Crombrugghe, B.; Jiao, H.; Xiao, G.; Zhang, C. Transcriptional Regulation of Vascular Endothelial Growth Factor (VEGF) by Osteoblast-Specific Transcription Factor Osterix (Osx) in Osteoblasts. J. Biol. Chem. 2012, 287, 1671–1678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Johnson, A.W.; Luyten, F.P.; Picart, C. Bone Regeneration Strategies: Engineered Scaffolds, Bioactive Molecules and Stem Cells Current Stage and Future Perspectives. Biomaterials 2018, 180, 143–162. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Jiang, W.; Phillips, F.M.; Haydon, R.C.; Peng, Y.; Zhou, L.; Luu, H.H.; An, N.; Breyer, B.; Vanichakarn, P.; et al. Osteogenic Activity of the Fourteen Types of Human Bone Morphogenetic Proteins (BMPs). J. Bone Jt. Surg. Am. 2003, 85, 1544–1552. [Google Scholar] [CrossRef]
- Fei, L.; Wang, C.; Xue, Y.; Lin, K.; Chang, J.; Sun, J. Osteogenic Differentiation of Osteoblasts Induced by Calcium Silicate and Calcium Silicate/b-Tricalcium Phosphate Composite Bioceramics. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1237–1244. [Google Scholar] [CrossRef]
- Singh, A.; Gill, G.; Kaur, H.; Amhmed, M.; Jakhu, H. Role of Osteopontin in Bone Remodeling and Orthodontic Tooth Movement: A Review. Prog. Orthod. 2018, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Standal, T.; Borset, M.; Sundan, A. Role of Osteopontin in Adhesion, Migration, Cell Survival and Bone Remodeling. Exp. Oncol. 2004, 26, 179–184. [Google Scholar]
- Bartolo, P.J.; Kruth, J.; Silva, J.; Levy, G.; Malshe, A.; Rajurkar, K.; Mitsuishi, M.; Ciurana, J.; Leu, M. Biomedical Production of Implants by Additive Electro-Chemical and Physical Processes. CIRP Ann. Manuf. Technol. 2012, 61, 635–655. [Google Scholar] [CrossRef]
- Lopes, D.; Martins-Cruz, C.; Oliveira, M.B.; Mano, J.F. Bone Physiology as Inspiration for Tissue Regenerative Therapies. Biomaterials 2018, 185, 240–275. [Google Scholar] [CrossRef]
- Lee, D.W.; Kwon, J.Y.; Kim, H.K.; Lee, H.J.; Kim, E.S.; Kim, H.J.; Kim, H.J.; Lee, H.B. Propofol Attenuates Osteoclastogenesis by Lowering RANKL/OPG Ratio in Mouse Osteoblasts. Int. J. Med. Sci. 2018, 15, 723–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaniboni, E.; Bagne, L.; Camargo, T.; do Amaral, M.E.C.; Felonato, M.; de Andrade, T.A.M.; Dos Santos, G.M.T.; Caetano, G.F.; Esquisatto, M.A.M.; Santamaria, M.; et al. Do Electrical Current and Laser Therapies Improve Bone Remodeling during an Orthodontic Treatment with Corticotomy? Clin. Oral Investig. 2019, 23, 4083–4097. [Google Scholar] [CrossRef] [PubMed]



| Group Number | Group Name | Scaffold Implanted | Electrical Stimulation | PCL Polymer Concentration | Ceramic Concentration |
|---|---|---|---|---|---|
| 1 | Untreated | No | No | 0 wt% | 0 wt% |
| 2 | ES | No | Yes | 0 wt% | 0 wt% |
| 3 | PCL | Yes | No | 100 wt% | 0 wt% |
| 4 | PCL/ES | Yes | Yes | 100 wt% | 0 wt% |
| 5 | HA | Yes | No | 80 wt% | HA 20 wt% |
| 6 | HA/ES | Yes | Yes | 80 wt% | HA 20 wt% |
| 7 | TCP | Yes | No | 80 wt% | β-TCP 20 wt% |
| 8 | TCP/ES | Yes | Yes | 80 wt% | β-TCP 20 wt% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helaehil, J.V.; Lourenço, C.B.; Huang, B.; Helaehil, L.V.; de Camargo, I.X.; Chiarotto, G.B.; Santamaria-Jr, M.; Bártolo, P.; Caetano, G.F. In Vivo Investigation of Polymer-Ceramic PCL/HA and PCL/β-TCP 3D Composite Scaffolds and Electrical Stimulation for Bone Regeneration. Polymers 2022, 14, 65. https://doi.org/10.3390/polym14010065
Helaehil JV, Lourenço CB, Huang B, Helaehil LV, de Camargo IX, Chiarotto GB, Santamaria-Jr M, Bártolo P, Caetano GF. In Vivo Investigation of Polymer-Ceramic PCL/HA and PCL/β-TCP 3D Composite Scaffolds and Electrical Stimulation for Bone Regeneration. Polymers. 2022; 14(1):65. https://doi.org/10.3390/polym14010065
Chicago/Turabian StyleHelaehil, Júlia Venturini, Carina Basqueira Lourenço, Boyang Huang, Luiza Venturini Helaehil, Isaque Xavier de Camargo, Gabriela Bortolança Chiarotto, Milton Santamaria-Jr, Paulo Bártolo, and Guilherme Ferreira Caetano. 2022. "In Vivo Investigation of Polymer-Ceramic PCL/HA and PCL/β-TCP 3D Composite Scaffolds and Electrical Stimulation for Bone Regeneration" Polymers 14, no. 1: 65. https://doi.org/10.3390/polym14010065
APA StyleHelaehil, J. V., Lourenço, C. B., Huang, B., Helaehil, L. V., de Camargo, I. X., Chiarotto, G. B., Santamaria-Jr, M., Bártolo, P., & Caetano, G. F. (2022). In Vivo Investigation of Polymer-Ceramic PCL/HA and PCL/β-TCP 3D Composite Scaffolds and Electrical Stimulation for Bone Regeneration. Polymers, 14(1), 65. https://doi.org/10.3390/polym14010065









