Abstract
In recent years, ecological issues have led to the search for new green materials from biomass as precursors for producing carbon materials (CNFs). Such green materials are more attractive than traditional petroleum-based materials, which are environmentally harmful and non-biodegradable. Biomass could be ideal precursors for nanofibers since they stem from renewable sources and are low-cost. Recently, many authors have focused intensively on nanofibers’ production from biomass using microwave-assisted pyrolysis, hydrothermal treatment, ultrasonication method, but only a few on electrospinning methods. Moreover, still few studies deal with the production of electrospun carbon nanofibers from biomass. This review focuses on the new developments and trends of electrospun carbon nanofibers from biomass and aims to fill this research gap. The review is focusing on recollecting the most recent investigations about the preparation of carbon nanofiber from biomass and biopolymers as precursors using electrospinning as the manufacturing method, and the most important applications, such as energy storage that include fuel cells, electrochemical batteries and supercapacitors, as well as wastewater treatment, CO2 capture, and medicine.
1. Introduction
Increasing environmental awareness and ecological problems have led in recent years to new green materials made from biomass as precursors for the production of carbon materials (CNFs) receiving more research attention, as they seemed to be more attractive than traditional petroleum-based materials, which are polluting, toxic and non-biodegradable [1]. At the same time, there is a need to develop cleaner, more economical, efficient and energy-saving materials and to focus the world’s attention on the new green, renewable energies. Converting biomass waste into carbon materials can help solve the problem of pollution and improve traditional processing methods for producing carbon in the face of the energy crisis and environmental problems [1]. Low-cost renewable biomass materials, such as sawdust, wood residues, rice husks, and corn stover, among many others, are available in large quantities as waste from forestry and agriculture. These renewable biomass materials can be considered promising candidates for carbon precursors [2].
Various techniques for synthesizing carbon nanofibers from biomass, such as electrospinning, pyrolysis, hydrothermal treatment, and ultrasonic treatment, are already known and have already been explained by many research groups [3,4,5,6,7,8,9].
Most of the techniques are complicated and require extensive use of energy resources. For example, the production of carbon nanofibers from hazelnut shell biomass as a carbon resource by hydrothermal technique goes through a series of complex processes, such as hydrothermal carbonization, heat treatment, potassium hydroxide activation, magnesium oxide templating to produce anode materials for lithium-ion batteries at the end of the process [10].
This example shows the complexity of getting carbon nanofibers using traditional techniques, and therefore, the simple and low-cost production of carbon nanofibers by electrospinning offers an excellent solution. However, interestingly, only a few publications have been found on obtaining carbon nanofibers from biomass using the electrospinning method. It seems likely that other methods have been more researched than electrospinning, which has seen a rapid increase in publications in this area in recent years. In addition, the lack of research could be because more well-known polymers are used for the production of nanofiber mats, and the field of biomass is slowly making its entry into electrospinning.
Moreover, the biomass forms a complex composition of different substances that cannot be electrospun alone and should be extracted first, furthermore requiring polymers, such as PAN or PEO, as a carrier material to stimulate nanofiber spinning. The considerations at this point can be diverse, and this review attempts to fill this gap.
Electrospinning is a well-known process for producing nanofibers from various synthetic and bio-based polymers and is very easy to apply [2,11]. Carbon nanofibers can be obtained from nanofibers produced by electrospinning technology and subsequently thermally processed. Nanofiber mats result from producing fibers with diameters of a few tens to a few hundreds of nanometers from a polymer solution or melt in a strong electric field [12,13]. The areas of application for such nanofiber mats are broad due to the numerous positive properties, such as large surface-to-volume ratio and outstanding flexibility, etc. The compositions of electrospun fibers are diverse and much more exciting than any other fibers. Electrospun nanofibers can be made from almost unlimited materials, from synthetic to natural polymers, and by adding particles, new materials and composites can be produced [14,15,16,17]. Biomass-derived carbon nanofibers can be produced from a variety of polymers, such as chitin from seafood [18] or fungi [19], lignocellulosic materials [20], gelatin, proteins and from the combination of polymer blends. In order to manipulate the bonding between the fibers of the nanofiber material and improve the overall electrical conductivity, mesoporosity and surface area, often (polyacrylonitrile) PAN-based carbonized nanofibers are used, blended with biopolymers because producing nanofibers from these biopolymers alone is not possible. Nanofibers can be produced from these bio-based polymer blends at a much lower cost than the conventional processes currently used [21]. These bio-based polymer blends are used for various commercial applications in the industrial field and are specially developed for this purpose. Such precursor electrospun fibers and carbon fibers are of great interest for a range of applications, including drug delivery and regenerative medicine [22], tissue engineering [23], cells growing [24], catalysts [25], filters [26,27,28] or protective clothing [29]. Not only artificial polymers [30] and biopolymers [31,32] can be used for the production of nanofiber mats, but also polymers mixed with non-soluble materials can be used for electrospinning [33,34]. By adding particles, it is also possible to produce magnetic nanofiber mats for various applications [35,36,37]. These magnetic nanofibers are promising for basic research and future applications in spintronics or neuromorphic computing [38,39,40,41,42]. Due to their high surface-to-volume ratio, nanofibers are ideal for filtration applications [43]. As a result of their high porosity and permeability, as well as their small pore size, reliable filtration of the smallest and finest particles can be achieved [44,45]. Other applications of nanofiber mats as filters include optical and chemical sensors [46], energy storage [47], aerospace [48], nanocatalysis [49], protective clothing [50], transportation [51], water and air filters for biotechnological and industrial applications [52,53,54,55,56,57,58], filtering of dye particles from wastewater from the finishing industry [59] or filtering of smoke filtration and flavor retention [60]. Great research interest in recent years shows the use of biomass for the production of nanofibers from the biomass and natural biopolymers used as a precursor for carbon and carbon nanofibers to make the contribution to the ecological point of view and the extraction of raw materials from natural resources. This overview summarizes and presents the current developments. Furthermore, the research gap in the production of carbon nanofibers from biomass by the electrospinning method will be discussed. Carbon nanofibers derived from biomass provide an alternative method for various industrial applications, reducing economic costs and environmental impact.
2. Biomass as Precursor for Carbon Nanofibers
Various natural substances have been used as precursors for the synthesis of carbon materials, such as activated carbon and carbon nanofiber [61]. Biomass is any organic matter that is available on a renewable or recurring basis. It can be classified into forest residues, agricultural residues, food industry waste, municipal solid waste, industrial waste, wastewater, and animal waste [4]. In recent years, the use of biomass as a precursor for carbon and carbon nanofiber has been widely explored due to their contribution to the ecologic system [62] and also due to the low cost of the extraction of the raw materials. Various biomasses used for the production of carbon nanofibers include lignocellulose materials [63,64,65,66,67,68,69] such as cellulose, hemicellulose [70], lignin [71,72], bamboo [73], crab shell [74], natural fungus [75,76], sawdust [9], seafood chitin [77], coconut shell charcoal [78] etc. On other hand, carbon nanofiber also can be prepared from polymers, such as polyacrylonitrile (PAN) [79,80], poly(vinyl alcohol) [81], vanillin polymer [82] and poly-(vinyl pyrrolidone) [83].
3. Preparation of Carbon Nanofibers
3.1. Electrospinning
Electrospinning is a well-known simple, productive process for producing highly functional fibers in the micro- and nanometer range [84]. The advantages of nanofibers include easy and low-cost manufacturing, lightweight, high molecular orientation, and high flexibility in surface functionality, as well as almost unlimited use of biobased and synthetic polymers and blends of various particles [85]. In all electrospinning techniques, such as needle-based, needle-free, electrospraying and others, a high-voltage electric field is basically applied to a polymer solution, causing the fibers to form a Taylor cone at the tip [86].
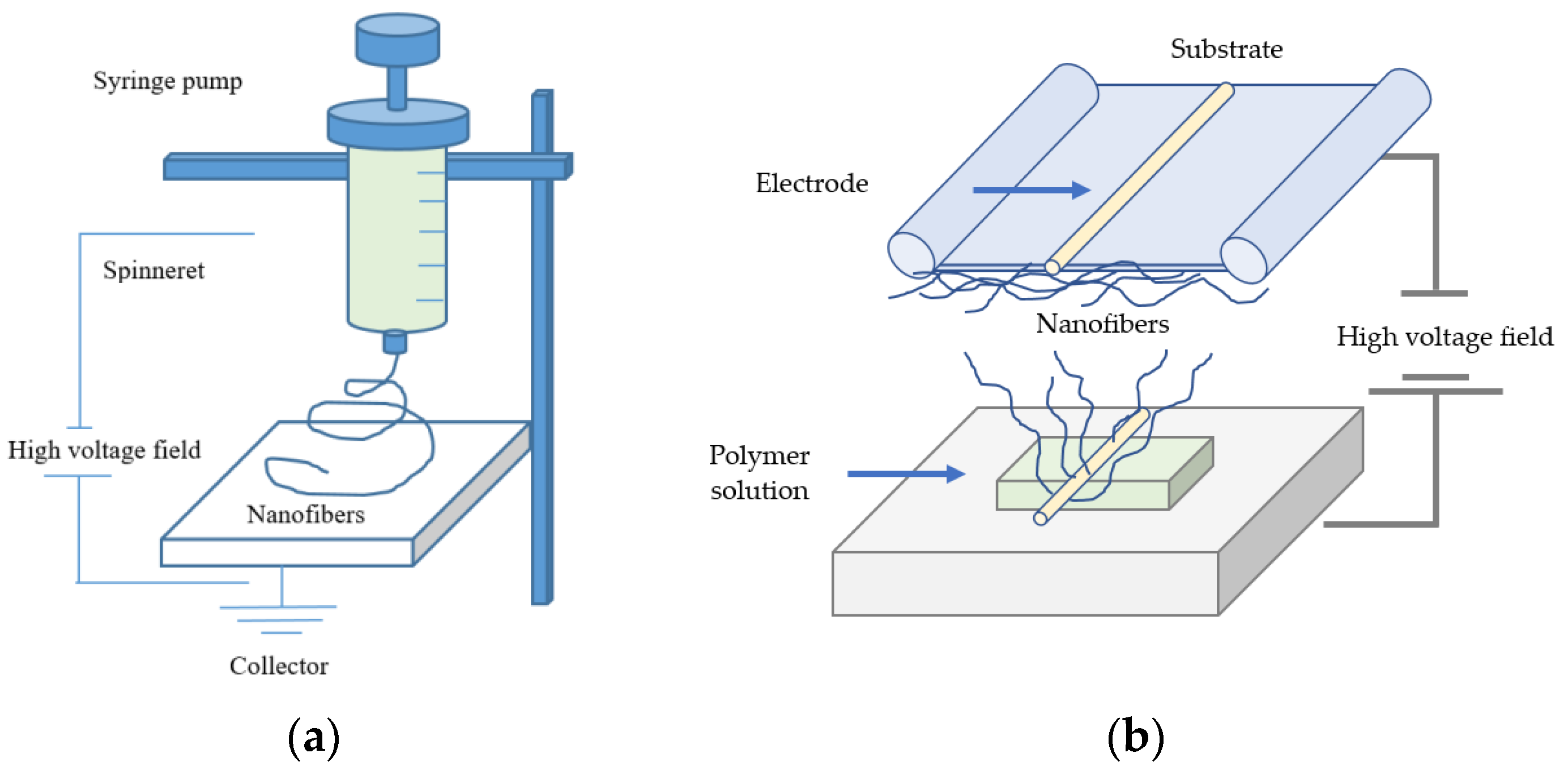
A strong electric field carries electric charges along the polymer surface, and Taylor cones are formed. Subsequently, the droplets of electrospinning solution are attracted to the counter electrode. As a result, the droplets are stretched extensively and form a very thin nanoscale fiber when they reach the substrate [87,88]. In the electrospinning process, a polymer solution or melt is generally deposited through a needle [89,90] or in needleless technique by coating a wire, cylinders, other objects that are used as electrodes or free surfaces with the polymer melt or solution [91,92,93]. The basic setup of electrospinning involves the application of a strong electric field to a droplet of the polymeric solution used (e.g., precursor melting solution). The typical setup of electrospinning essentially consists of a high-voltage field, a syringe pump, a spinneret, and a collector onto, which the nanofibers are deposited [94]. A basic setup of needle-based and needleless electrospinning principles is shown in Figure 1.
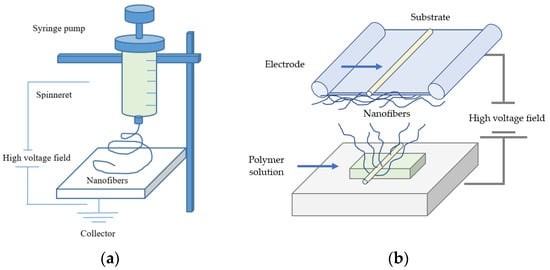
Figure 1.
(a) A basic setup of needle-based and (b) needleless electrospinning methods.
The electrospinning parameters, such as voltage, feed rate, electrode-type, and distance between electrode and collector, significantly affect the properties of the resulting electrospun nanofibers [95]. The electrospinning parameters play a decisive role in the resulting nanofibers. Depending on the change of the parameters, the defined nanofibers with desired properties can be produced [96,97].
3.2. Stabilization and Carbonization of Nanofibers
Various biobased and synthetic polymers are used as precursors for the preparation of CNFs by electrospinning followed by thermal heat treatment process involves oxidative stabilization and subsequent carbonization for the production of carbon fibers [98,99]. Oxidative stabilization is used because it prevents melting or fusing of the fibers and thus increases the final elemental carbon content. In other words, the stabilization process minimizes the volatilization of elemental carbon to maximize the final carbon yield [100]. Both thermal processes exert a great influence on the fiber morphology and the resulting properties [101,102]. Figure 2 depicts the carbonized mycelium/PAN nanofiber composite.

Figure 2.
Confocal laser scanning microscopy (CLSM) images of oyster mushroom mycelium on polyacrylonitrile (PAN) nanofiber mats (the latter not visible) (a) after stabilization of the whole composite for 1 h at 280 °C; (b) after carbonization for 1 h at 500 °C of the whole composite. Scale bars indicate 20 µm. Reproduced from [19], published under a CC BY 4.0 license (MDPI, Basel, Switzerland).
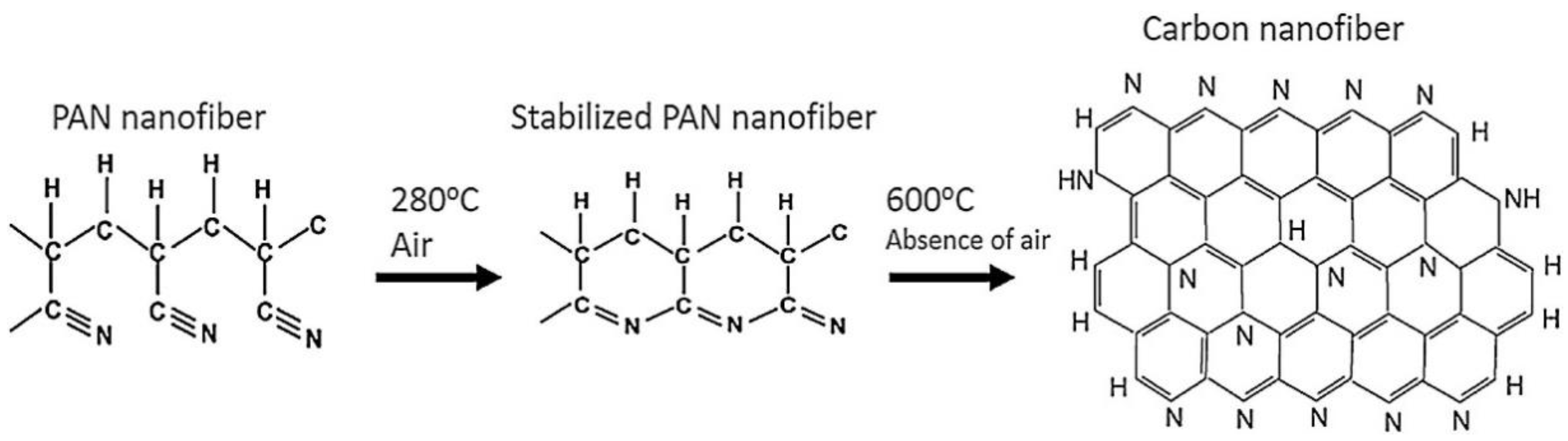
The dimensional change of the nanofiber mats takes place when the temperature is increased during the stabilization process. As the temperature increases, the dimensions of the samples will decrease. These changes happen on the molecular level due to the prevailing processes during the isothermal treatment. During the stabilization process in air, various processes, such as cyclization, dehydrogenation, oxidation, aromatization, and crosslinking reactions, take place, leading to an aromatic ladder structure [103,104]. These processes reduce the size of the sample area and the weight of the sample. The stabilization process plays an important role in the properties of the resulting CNFs and should be carried out in a controlled way. After stabilization, carbonization is performed, which is usually carried out in a nitrogen atmosphere and leads to different chemical and morphological properties of the carbon nanofiber depending on the treatment temperature. Figure 3 shows the changes at the molecular level and the transformation of PAN nanofiber mats into carbon nanofibers.
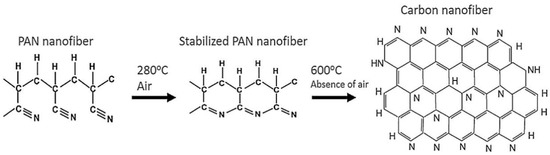
Figure 3.
The effect of thermal treatment in PAN structure change (proposed by Ibupoto et al.) Reprinted with permission from [105]. Copyright (2018) by Elsevier Publishing.
The carbonization process typically occurs under inert gases, such as argon or nitrogen, at temperatures ranging from 500 °C to 1.200 °C (see Table 1). The morphological modification of the resulting CNFs can be achieved by using different techniques. The polymers can be completely removed from the fibers during thermal treatment by pyrolysis [106,107] or by adding ingredients, such as phosphorus [108], nitrogen [109] or particles [110] to modify electrical or mechanical properties. By using blends, carbon nanofibers with adjustable fiber morphology can be produced.

Table 1.
Isothermal treatment conditions of biomass-derived electrospun carbon nanofibers.
During the thermal exposure, not only the dimension of nanofiber mats but also their color changes significantly. After electrospinning, the nanofibers have a matte white color, which changes with the increase of temperature from white to yellow, golden brown and at about 280 °C to dark brown. With the increase of temperature during the carbonization process, the CNFs have a black color. Moreover, after the electrospinning process, the nanofiber mates prove to be very flexible and with the increase of temperature, they become more unstable and brittle, and CNFs break very easily.
Table 1 summarizes some different carbon nanofibers obtained from biomass and biopolymers or a mixture between synthetic and biopolymers prepared by electrospinning.
As shown in Table 1, carbon nanofibers are largely produced from lignin and cellulose-containing biomass. Carbon nanofibers prepared from PAN/fungal mycelium or PAN/gelatine are still a novelty. Lignin and cellulosic materials have been known for a long time and have been intensively researched, and therefore, several studies are available, which deal with these materials.
4. Application of Carbon Nanofibers
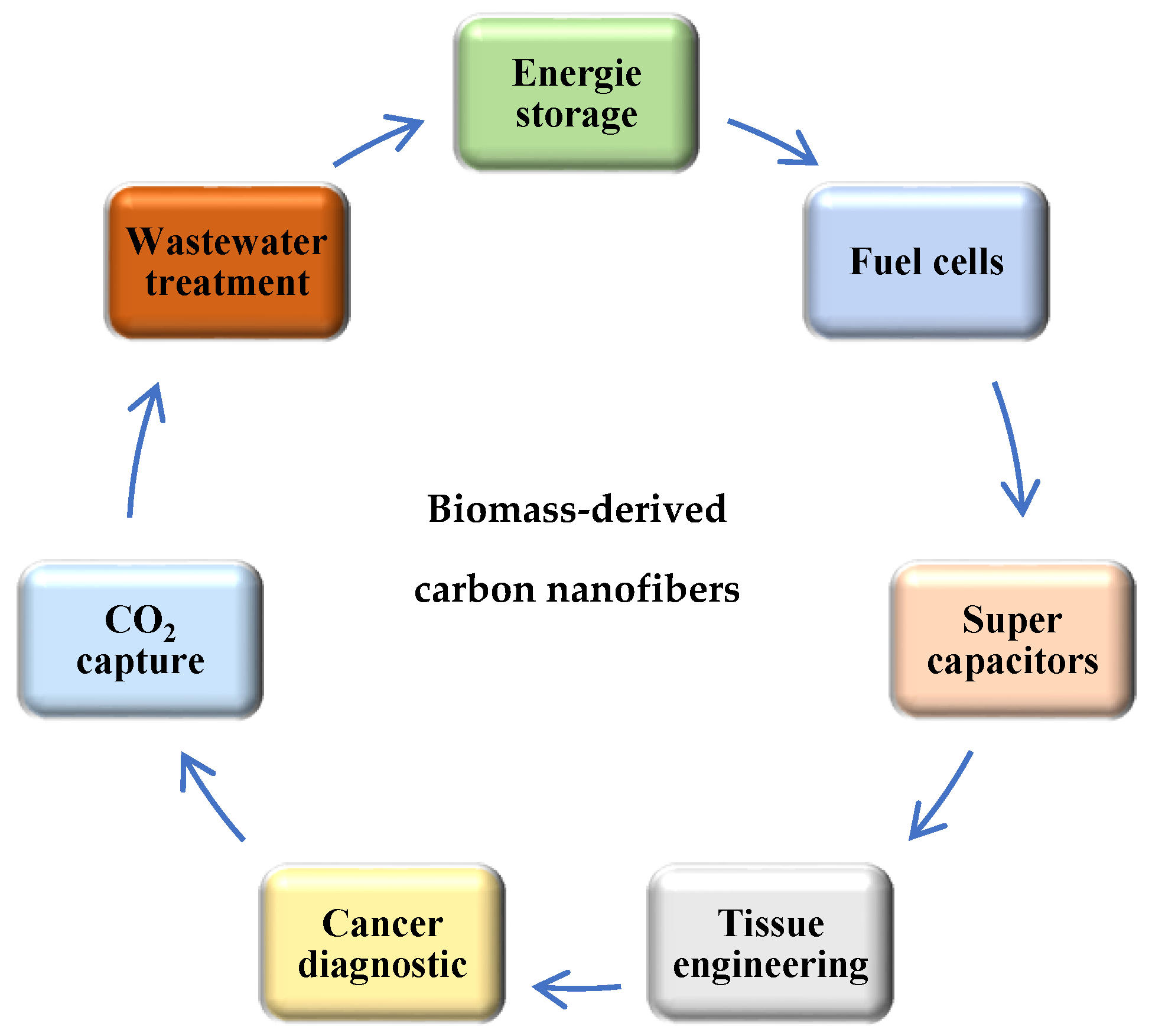
Electrospinning is one of the most productive methods for the preparation of carbon nanofiber using different precursor solutions. Recently, many authors have intensively studied the production of carbon nanofibers by electrospinning from biomass and polymers with or without additives, such as various metals, particles, acids, alkyls, to increase the importance of their application in industry. The importance of carbon nanofibers is based on their porous structure, high specific surface area, high conductivity, and thermal stability. All these properties make the development of the preparation methods a big challenger for recent works. Recently, the manufacturing of carbon nanofibers has significantly improved diverse applications due to their physical and chemical properties, such as high porosity and large specific surface area, numerous active sites, good catalytic properties, high conductivity, good temperature stability, and low-cost, which have been a challenge for many industrial applications (Figure 4), such as energy storage (fuel cells, electrochemical batteries and supercapacitors) [120,121,122], environment science [123,124], tissue engineering [125], optical sensors [126], cancer diagnosis [127].
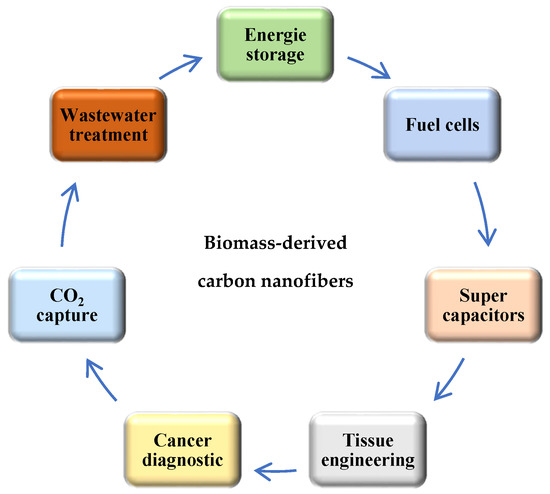
Figure 4.
The different applications of biomass-derived carbon nanofibers.
4.1. For Energy Storage
4.1.1. Fuel Cells
Fuel cells function as batteries, which are electrochemical cells applied to convert chemical energy into electricity through two electrodes (cathode–anode) by redox reactions. The application of rechargeable ion batteries and polymer electrolyte membrane fuel cells has aroused interest in analytical models for calculating the transverse permeability of the gas diffusion layer in proton-exchange membrane fuel cells [128] and effective electrolyte diffusivity considering the electrokinetic effects and microstructure parameters of porous media using the using the fractal theory of porous media [129]. Fuel, such as hydrogen, is supplied to the anode, and the air is supplied to the cathode. Presently, most commercial catalysts used for fuel cells are catalysts doped with metal as Pt due to their high efficacity and stability. However, the high cost of this metal decreases the utilization of this type of fuel cell.
Recently many investigations used carbon nanofiber for fuel cells due to their large surface area, higher conductivity, and stability. Among them, Chung et al. used CNFs prepared by electrospinning of PAN following by heat treatment of stabilization, carbonization and graphitization at different temperatures for Pt/C cathode to improve water management in fuel cells [79]. They concluded that the enhanced air transport in the water-free region in the cathode provided by hydrophobic carbon glass fibers CGFs also suggested this principle for an effective water management method in the electrode. Abdelkarim et al. summarized that Ni-decorated carbon nanofibers from poly(vinyl alcohol) doped with Cd could be an effective co-catalyst for urea oxidation comparable with Ni-carbon nanofibers [81]; they also found that the quality of electrochemical oxidation activity and the number of active sites were improved. In another work, Chan et al. analyzed the performance of carbon nanofiber for polymer electrolyte membranes fuel cell catalyst layers [130]. The results showed that the carbon nanofiber-based catalyst layers significantly outperformed traditional carbon-supporting catalyst layer structures. Also, further optimization can be achieved by improving the control of the morphology and distribution of ionomers, especially in terms of ionomer bridges and connectivity. On the other hand, Ponomarev et al. prepared a complex carbon nanofiber paper by electrospinning PAN with integrated Zr and Ni, followed by further pyrolysis to use it for the high-temperature (HT)- proton exchange membrane fuel cell (HT-PEMFC) [131]. They confirmed that the use of the carbon nanofiber platinum support obtained leads to a higher fuel cell efficiency comparable to the Celtec® P1000, which is one of the best commercial electrodes.
4.1.2. Electrochemical Batteries
Carbon nanofibers can show excellent electrical conductivity, leading to their use as a leading additive in lithium-ion (LIB) and sodium-ion (NIB) electrodes. In the last years, several authors investigated the use of carbon nanofibers in batteries as a novel approach. Amongst them, He et al. reported that TiO2/carbon nanofibers obtained from PAN by electrospinning showed high performance as negative electrodes for vanadium redox flow battery (VRFB) [80]. The authors observed that the cell using CNF–TiO2 as a negative electrode showed an increase of 7.8% in the energy efficiency than the pristine cell. Also, Trung Bui et al. synthesized carbon nanofibers from PAN and PVP (polyvinyl pyrrolidone l with metal (Pt, Co, and Pd) by coaxial electrospinning to improve the performance of Li–O2 battery cathodes [132]. The results indicated that the use of CNF–Pt in Li–O2 cells displays much better electrochemical performance in specific capacity, rate capability, energy efficiency, cycle stability, and O2 efficiency, comparable with the two other metal-embedded carbon nanofibers (CNF–Pd, CNF–Co). They also observed a reduction in overpotential in both charging and discharging with an improvement in cycle life with a limiting capacity of 1000 mAh/gc at a current density of 500 mA/gc. Furthermore, due to the importance of improving the performance of Li–O2 battery cathode, Yoon et al. evaluated the use of carbon nanofibers with metal using cobalt nitride (Co4N) [133]. The authors found that the cathode (Co4N/CNF) had excellent electrochemical performance and exceptional stability over 177 cycles in Li–O2 cells.
The results proved that the favorable formation/decomposition of reaction products and the transmission of side reactions are largely regulated by the appropriate surface chemistry and adapted structure of the cathode materials, which are necessary for lithium batteries. Furthermore, Xia et al. investigated the use of flexible SnSe/C nanofiber membranes by simple electrospinning, followed by calcination, in both LIBs and SIBs batteries [134]. The findings obtained prove the high-performance of the capacity and cycling stability in the batteries. In addition, LIBs battery displayed a higher discharge capacity at higher current density after 500 cycles and the same for SIBs, but after 200 cycles at different current densities.
On the other hand, carbon nanofibers are also used for sodium-ion batteries. Zhao et al. manufactured carbon nanofibers from PAN doped with NiS2 nanoparticles by electrospinning [135]. The authors reported that NiS2 nanoparticles/carbon nanofiber as SIBs anode display a high-performance with a good specific capacity of 500 mAh g−1 at 0.1 A g−1 and also a competitive rate with storage of 200 mAh g−1 at 2 A g−1, in addition to long-lasting consistency within 1000 cycles. Besides, Zhou et al. studied the performance of the ZnSe electrospinning nanofiber form PAN in lithium and sodium batteries as anode materials [136]. They showed that ZnSe@N–CNFs present an excellent performance in LIBs and also in SIBs at higher current density. Furthermore, the sample exhibited a high capacity for both batteries (LIBS and SIBs).
Yin et al. prepared CoSe2 grain attached carbon nanofiber from PAN as a carbon source by electrospinning for flexible sodium-ion batteries [137]. It was demonstrated that the carbon nanofibers form a flexible, binder-free film and thus a three-dimensional, conductive, interconnected network. Due to the carbon nanofiber structure, it was confirmed that electrochemical conductivity and Ni ion diffusion, structural stability and also cycling stability were improved.
4.1.3. Supercapacitors
Supercapacitors are a kind of new energy storage and conversion equipment that should have the potential for high-power density, high circulation characteristics, fast discharge/charge, low self-discharge, safe work, and low cost. Various porous materials, including porous carbon and carbon nanofibers, are used in the manufacture of supercapacitors due to their high electrochemical properties, such as resistance, conductivity, and good temperature stability, also due to their high specific surface area and porosity. Currently, many investigations have been carried out on the use of carbon nanofibers obtained from electrospinning as supercapacitors.
Dai et al. summarized the use of lignin/PAN with graphene doped with nitrogen-sulfur carbon nanofiber obtained by electrospinning for supercapacitor. They mentioned that the high surface area obtained of 2439 m2 g−1 and good surface morphology of the carbon nanofiber exhibited a supercapacitor with high energy and high-power densities, also ultra-high capacity with cycling stability of 97.8% after 5000 cycles of charge/discharge [138] Cao et al. also tested cellulose acetate/lignin-based carbon nanofibers as supercapacitors [72]. The cellulose/lignin-based carbon nanofibers showed a high energy density of 30.2 Wh/kg at a power density of 400 W/kg due to the covalent bond of lignin and cellulose acetate, which give the material good thermal stability, high specific surface area, filamentous morphology, homogenous diameter distribution and high storage capacity. Besides, Ma et al. evaluated carbon nanofibers from lignin and PVP by electrospinning with Mg(NO3)2·6H2O as activate agent at different ratios as an electrode in supercapacitor [112]. The authors suggested that the increase in the ratio of Mg(NO3)2·6H2O increases the surface area and the mesoporosity of the carbon nanofiber, leading to an improvement in capacity when it used as triple- and double-electrode, also they observed that lignocellulose carbon nanofibers (LCNFs) present a good outstanding rate performance and high cycling stability. On other hand, García-Mateos et al. synthesized carbon nanofiber from lignin with phosphoric acid by electrospinning following by two separate heat treatments in air and in an inert atmosphere to be used in supercapacitor electrodes [66]. They reported that the activation under air atmosphere increases the energy storage up to 50% and also ameliorated the power capability of the electrode. In the study by Wang et al., carbon was fabricated from biomass, with a large number of straight holes embedded with nickel particles as the anode framework. The nickel-modified electrode was stably cycled for 1370 h at a current density of 5 mA/cm2 with a capacity of 2 mAh/cm2, and lithium storage with a more substantial capacity (5 mAh/cm2) was used, which kept the cycling stable for 630 h. Furthermore, the nickel-modified electrode was used as the anode. Biomass carbon matrix with pores and sulfur powder was filled and tested as the positive electrode. According to the study, the capacity retention rate is 85% after 100 periods of full cell operation [139].
4.2. Environmental Science
Carbon materials, such as activated carbon and carbon fibers, are widely used in environmental science, more specifically in adsorption and absorption and liquid or gas phases. Adsorption is a phase transfer process mostly used in purification and separation processes and, particularly, in the removal of pollutants from the fluid phase [140]. The process of absorption means that material catches and transforms energy. Therefore, absorption distributes content that it catches through the whole bulk, while adsorption distributes it only along the surface.
4.2.1. Wastewater Treatment
The removal of pollution from wastewater has become a significant challenge for many investigations due to their impact on human health, the environment, and aquatic environments [141]. Nowadays, carbon nanofibers adsorption is one of the most popular technologies for wastewater treatment [142] due to their physical and chemical properties, such as high specific surface area, porosity, surface chemistry and morphology.
Zhao et al. evaluated the use of nanofiber membranes made of biomass vanillin polymer for the adsorption of methylene orange and sodium dodecyl sulfate [82]. They reported that the adsorption capacities reached their maximum at 406.6 and 636.0 mg/g for MO and SDS, respectively. Furthermore, they mentioned that the regeneration of biomass vanillin-derived polymer nanofiber has a high efficiency even after six adsorption-desorption cycles. Recently, Widiyastuti et al. analyzed electrospun activated carbon nanofibers from coconut shell charcoal and PVA with KOH for the adsorption of methylene blue [78]. They concluded that the activated carbon nanofiber obtained at PVA 12 w/v%, 60 wt % C displays the highest adsorption capacity of 166.7 mg/g with the ability to remove 96% at the third cycle.
In addition, some authors carried out the use of the photocatalytic process for wastewater treatment. Among them, Gan et al. analyzed the preparation of biomass cellulose-derived carbon nanofibers by electrospinning with bismuth oxybromide (BiOBr) at different pyrolysis temperatures for the removal of rhodamine B (RhB) and hexavalent chromium (Cr(VI)) [143]. They summarized that the increase in pyrolysis temperatures leads to an increase of the photocatalytic capacity for both RhB and Cr(VI). Additionally, they confirmed that the BiOBr/cellulose-derived carbon nanofibers CCNFs show a high efficacity for the simultaneous removal of the contaminants (RhB and Cr(VI)).
4.2.2. CO2 Capture
A large number of greenhouse gas emissions currently contribute to global warming and pose a worrying threat to the environment and human health [142]. Among all greenhouse gases, CO2 comes from many sources and has a significant impact on global climate change [142]. Actually, many technologies are used for the capture of CO2, the most often applied, of which are absorption, adsorption, membranes, cryogenic, enzymatic and hybrid.
Adsorption is the most commonly used method due to its effectiveness, selectivity, renderability and cost-efficiency. Carbon nanofibers are considered as one of the best alternatives for CO2 capture because they have not only the intrinsic properties of carbon nanomaterials but also an adjustable pore structure [113,142].
In 2016, Calvo-Muñoz et al. successfully manufactured carbon fibers from biomass by electrospinning, where they used alcell lignin with ethanol as precursor fuel cell letter (FCL) for CO2 capture under post-combustion conditions [113]. The results revealed that FCL exhibited capacity values of 1.3 mmol/g, with full regeneration of the carbon material at the same condition without any additional energy. More recently, Zainab et al. analyzed the efficient CO2 capture by polyacrylonitrile–poly(vinyl pyrrolidone) (PAN/PVP) carbon nanofibers obtained via electrospinning with subsequent intensive washing and carbonization treatment [83]. The authors suggested that the PAN/PVP carbon nanofibers showed a high selectivity with a CO2 adsorption capacity of 3.11 mmol/g. They also mentioned the high selectivity of the regeneration up to 50 cycles of adsorption/desorption.
Ali et al. tested polyacrylonitrile–polyvinylidene fluoride (PAN/PVDF) core-shell nanofibers hybridized with tin oxide nanoparticles for adsorption of CO2 [144]. The maximum capacity obtained was 2.6 mmol/g at room temperature, where they confirmed that the carbon nanofiber displays high remarkable cycling stability even after 20 adsorption/desorption cycles and reached a maximum of 95% of the initial CO2 adsorption capacity. Furthermore, Chiang et al. also prepared nanofiber adsorbents from polyacrylonitrile (PAN) via electrospinning, following by different heat treatments (stabilization, carbonization, and activation) for CO2 adsorption application [145]. They reported an adsorption capacity of 1.2 mmol/g (0.15 atm) and 3.2 mmol/g (1 atm), obtained at 298 K, and explained the high efficiency of the carbon nanofibers by the activation temperatures
4.3. Biotechnological and Medical Fields
Today’s medicine progresses greatly and applies more therapeutic solutions based on the field of nanotechnology and nanomaterials. High-performance materials, such as carbon nanotubes, graphene, or carbon nanofibers, have already established their place in developing new implants and medical devices [141]. Due to their properties, high electrical conductivity, unique surface characteristics, and biomimetic shape, these nanomaterials are ideal for constructing implantable electrodes and biosensors. In addition, they can serve as tissue substrates for in vitro and in vivo applications. For this reason, stimulation of an electric field can regulate cell behavior both in vivo and in vitro due to the conductive properties of carbon substrates. Nanofibers resemble the natural structure of cell assembly and can be used in the form of porous mats as membranes for medical reconstruction, substrates for bone and cartilage development in post-traumatic tissues [146,147].
Polymer nanofibers, as well as carbon nanofibers, are promising candidates for diverse medical applications thanks to their physical properties. Due to their conductivity, they can be used as biosensors and electrodes to stimulate the nervous system, as well as for the fabrication of scaffolds for regenerative medicine. As nonwovens, mats, membranes, or other various types of nanocomposites, nanofibers can be used in many biotechnological fields [148,149,150,151,152]. Aoki et al. investigated the application potential of organic nanofibers and electrospun carbon nanofibers for bone regenerative medicine [153].
Previously, the research focus centered on coating nanofiber mates with antibacterial substances. The efficacy of silver nanoparticles and the active healing properties of chitosan polymer hydrogels received numerous publications. With the development of electrospinning processes, the research focus increasingly shifted to electrospun nanofibers, which exhibit antimicrobial properties through the addition of nanoparticles [154,155]. Due to their high mechanical strength and good biocompatibility, carbon-based nanofibers offer further areas of application in biomedicine [156,157,158].
In 2019 Li et al. conducted a study of a superhydrophobic hemostatic material made from a nanocomposite dispersion of a dense network of carbon nanofibers and polytetrafluoroethylene (PTFE) or poly-dimethylsiloxane (PDMS) on support [159]. This nanofiber material has been used for its particular and distinctive way of blood coagulation, which allows rapid blood coagulation due to the presence of microfibers and reduces subsequent blood loss, regardless of the pressure applied, due to its superhydrophobic characteristics.
In tissue engineering for regeneration or organ reconstitution, cells are designed to attach, proliferate, multiply and regenerate multiple organs, such as skin, bone, cartilage, muscle, tendons, heart, nerves, and blood vessels. These strategies depend on appropriate biochemical and physicochemical properties and molecular influences or control of cellular behavior [160]. Carbon nanofibers are potential candidates for tissue engineering applications because they have suitable physical, structural, mechanical, and biological properties [161,162,163]. In addition, carbon nanofibers have exceptional mechanical properties, conductivity, and excellent cytocompatibility properties, as well as osteoblast adhesion, which are suitable for neural and bone tissue engineering applications. In terms of carbon nanofiber adhesion and proliferation, they show the interaction of astrocytes like glial scar tissue-forming cells. These functions of astrocytes make them able to minimize nanoscale fibers and scar tissue formation, reduce the glial scar tissue formation and show positive interaction with neurons, which would be a great support for neural implants [164,165,166].
Recent research indicates that carbon-based nanomaterials are potential candidates for biomedical applications, including drug delivery, repair and regeneration of various tissues, including nerves, muscles, bones, and for imaging [167,168,169]. Stocco et al. have investigated that carbon nanofibers have strong mechanical properties capable of surviving without affecting mesenchymal stem cells for tissue engineering of the knee meniscus [170]. Samadian et al. and Patel et al. have found that carbon nanofibers are promising platforms with a nanoscale surface area that are helpful for tissue healing and bone regeneration process through anti-inflammation, pro-angiogenesis and stem cell stimulation [169,171]. The research group of Serafin et al. has presented that the electrically conductive properties of carbon nanofibers can be used in cardiac or neural tissue engineering applications [172]. In addition, carbon nanofiber composites have special properties, such as large specific area, high porosity, good biodegradability, cytocompatibility and conductivity, etc., making them ideal candidates in the field of tissue engineering and biological medicine [173,174,175,176].
In addition, there is a wide range of further carbon nanostructures such as carbon nanotubes, carbon nanofibers, carbon nano-onions (CNOs), graphene, which have attracted a lot of attention recently due to the promising industrial application areas. Onion-like quasi-spherical CNPs (OCNPs) with hollow cage-like concentric graphene shells have been known since 1992 but are still under-researched compared to other allotropic forms of nanocarbons, such as carbon nanotubes, carbon fibers, fullerenes, graphene, and carbon dots. CNOs are a niche product that has not been explored as much as other carbon nanostructures and offer many advantages, unlike other carbon nanostructures. They exhibit lower toxicity, have one of the exceptional biocompatibilities and are, therefore, of particular interest for medical and biotechnological applications, such as imaging, drug delivery, tissue engineering, sensing and as [177,178,179]. Excellent electrochemical performance is offered by CNOs due to their high surface area, the small size of the carbon-oxygen functional groups and the micro-open 3D graphite structures. These properties provide sufficient space for ion storage, hierarchical porous channels for ion transfer and a carbon matrix with high conductivity for electron transfer [180]. Breczko et al. prepared composites of CNOs and poly(diallyldimethylammonium chloride) (PDDA) or chitosan (chit), and the electrochemical properties were tested and investigated [181]. In another study, CNO–PDDA composite films for dopamine detection were prepared in the presence of ascorbic acid and uric acid in solution [182]. The research group of Giordani et al. prepared a novel near-infrared (NIR)-fluorescent carbon-based nanomaterial, which consists of boron-difluoride azadipyrromethene fluorophores covalently bonded to carbon nano-onions [183]. The cytotoxicity and immunomodulatory properties of the synthesized fluorescein CNO derivative were elucidated and compared with similarly functionalized CNTs. CNOs were found to exhibit efficient cellular uptake, mild inflammatory potential, and low cytotoxicity. These discoveries make CNOs promising materials for biomedical application areas. Moreover, due to a novel concentric graphitic shell structure and a large surface area, CNOs possess many excellent physical properties, such as high electrical conductivity and can be used in the fields of magnetic and gas storage materials, lubricants, for nanoreactors or as substrates for catalyst carriers and electrochemical capacitors [183].
5. Conclusions and Future Outlook
The conversion of biomass into carbon nanofibers offers the possibility to reduce the high carbon cost and provides an alternative to limited petroleum-based resources. Biomass is an available and sustainable material that can be converted into carbon nanofibers for various applications. Currently, there are few studies on the modification of biomass and the basic knowledge of the natural components of biomass and further conversion into carbon nanofibers, although the application potential is promising. This review of carbon nanofibers produced by electrospinning and their potential applications illustrates their usefulness in various technological and biomedical fields. While carbon nanofibers are produced by an electrospinning technique followed by stabilization and carbonization, their physical and chemical properties make them useful in a variety of applications. However, few studies have been conducted on the application of carbon nanofibers from biomass because this topic is still relatively under-researched. In particular, the available information on the application of biomass-derived carbon nanofibers produced by electrospinning is still limited. The production and optimization of the properties of carbon nanofibers from biomass using different types of biomass provide the opportunity for subsequent scaling up to a commercial scale. This study was aimed to clarify the relevant aspects and contribute to this goal.
This work can serve as a basis for further work on carbon nanofibers from biomass and biomass blends and their applications in energy storage and environmental protection, as well as medical technology and biotechnology in the future. First, there are many works evaluating the production of carbon nanofibers and carbon materials by different methods, but the production of carbon nanofibers from biomass by electrospinning is not yet mature and should be analyzed in-depth using natural materials and biopolymers, which can be an alternative for many environmentally friendly applications. The work also proves the improvement of biomass-derived carbon nanofibers by electrospinning and their application due to their physical and chemical properties. Since this topic is lacking in the literature, this review paper can serve as a basis for further research and facilitate the work of other authors.
Moreover, it is possible to use some additives, such as TiO2, Pt, Zr, etc., in the synthesis of CNFs from biomass to increase their surface area and conductivity, which is the key for electrochemical batteries and fuel cells. In conclusion, the use of biomass-derived CNFs from various industrial applications gives a great advantage to some factors, such as cost, performance, stability and the possibility to minimize the environmental damage and to obtain renewable materials as a precursor for the production of CNFs from renewable resources.
Author Contributions
Conceptualization, I.M. and L.S.; visualization, I.M.; M.T., A.M. and L.S.; writing—original draft preparation, I.M., M.T., A.M. and L.S.; writing—review and editing, all authors; supervision, L.S. All authors have read and agreed to the published version of the manuscript.
Funding
Not applicable.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Azwar, E.; Wan Mahari, W.A.; Chuah, J.H.; Vo, D.-V.N.; Ma, N.L.; Lam, W.H.; Lam, S.S. Transformation of biomass into carbon nanofiber for supercapacitor application—A review. Int. J. Hydrogen Energy 2018, 43, 20811–20821. [Google Scholar] [CrossRef]
- Wang, T.; Rony, A.H.; Sun, K.; Gong, W.; He, X.; Lu, W.; Tang, M.; Ye, R.; Yu, J.; Kang, L.; et al. Carbon Nanofibers Prepared from Solar Pyrolysis of Pinewood as Binder-free Electrodes for Flexible Supercapacitors. Cell Rep. Phys. Sci. 2020, 1, 100079. [Google Scholar] [CrossRef]
- Du, B.; Chen, C.; Sun, Y.; Yu, M.; Liu, B.; Wang, X.; Zhou, J. Lignin bio-oil-based electrospun nanofibers with high substitution ratio property for potential carbon nanofibers applications. Polym. Test. 2020, 89, 106591. [Google Scholar] [CrossRef]
- Omoriyekomwan, J.E.; Tahmasebi, A.; Dou, J.; Wang, R.; Yu, J. A review on the recent advances in the production of carbon nanotubes and carbon nanofibers via microwave-assisted pyrolysis of biomass. Fuel Process. Technol. 2020, 214, 106686. [Google Scholar] [CrossRef]
- Du, B.; Chen, C.; Sun, Y.; Yang, M.; Yu, M.; Liu, B.; Wang, X.; Zhou, J. Unlocking the response of lignin structure by depolymerization process improved lignin-based carbon nanofibers preparation and mechanical strength. Int. J. Biol. Macromol. 2020, 156, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Dineshkumar, M.; Meera Sheriffa Begum, K.M.; Shrikar, B.; Ramanathan, A. Synthesis and characterization study of solid carbon biocatalyst produced from novel biomass char in a microwave pyrolysis. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Duran-Jimenez, G.; Monti, T.; Titman, J.J.; Hernandez-Montoya, V.; Kingman, S.W.; Binner, E.R. New insights into microwave pyrolysis of biomass: Preparation of carbon-based products from pecan nutshells and their application in wastewater treatment. J. Anal. Appl. Pyrolysis 2017, 124, 113–121. [Google Scholar] [CrossRef]
- Wan, W.; Zhao, W.; Wu, Y.; Dai, C.; Zhu, X.; Wang, Y.; Qin, J.; Chen, T.; Lü, Z. A highly efficient biomass based electrocatalyst for cathodic performance of lithium–oxygen batteries: Yeast derived hydrothermal carbon. Electrochim. Acta 2020, 349, 136411. [Google Scholar] [CrossRef]
- Zhang, S.; Yin, H.; Wang, J.; Zhu, S.; Xiong, Y. Catalytic cracking of biomass tar using Ni nanoparticles embedded carbon nanofiber/porous carbon catalysts. Energy 2021, 216, 119285. [Google Scholar] [CrossRef]
- Unur, E.; Brutti, S.; Panero, S.; Scrosati, B. Nanoporous carbons from hydrothermally treated biomass as anode materials for lithium ion batteries. Microporous Mesoporous Mater. 2013, 174, 25–33. [Google Scholar] [CrossRef]
- Huang, W.; Tong, Z.; Wang, R.; Liao, Z.; Bi, Y.; Chen, Y.; Ma, M.; Lyu, P.; Ma, Y. A review on electrospinning nanofibers in the field of microwave absorption. Ceram. Int. 2020, 46, 26441–26453. [Google Scholar] [CrossRef]
- Shabafrooz, V.; Mozafari, M.; Vashaee, D.; Tayebi, L. Electrospun nanofibers: From filtration membranes to highly specialized tissue engineering scaffolds. J. Nanosci. Nanotechnol. 2014, 14, 522–534. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhu, J.; Zhang, L.; Qiu, Y. Recent advances in energy materials by electrospinning. Renew. Sustain. Energy Rev. 2018, 81, 1825–1858. [Google Scholar] [CrossRef]
- Sabantina, L.; Wehlage, D.; Klöcker, M.; Mamun, A.; Grothe, T.; García-Mateos, F.J.; Rodríguez-Mirasol, J.; Cordero, T.; Finsterbusch, K.; Ehrmann, A. Stabilization of electrospun PAN/gelatin nanofiber mats for carbonization. J. Nanomater. 2018, 6131085. [Google Scholar] [CrossRef]
- Sabantina, L.; Rodríguez-Cano, M.Á.; Klöcker, M.; García-Mateos, F.J.; Ternero-Hidalgo, J.J.; Mamun, A.; Beermann, F.; Schwakenberg, M.; Voigt, A.L.; Rodríguez-Mirasol, J.; et al. Fixing PAN nanofiber mats during stabilization for carbonization and creating novel metal/carbon composites. Polymers 2018, 10, 735. [Google Scholar] [CrossRef]
- Bier, M.C.; Kohn, S.; Stierand, A.; Grimmelsmann, N.; Homburg, S.V.; Rattenholl, A.; Ehrmann, A. Investigation of eco-friendly casein fibre production methods. IOP Conf. Ser. Mater. Sci. Eng. 2017, 254, 192004. [Google Scholar] [CrossRef]
- Grimmelsmann, N.; Grothe, T.; Homburg, S.V.; Ehrmann, A. Electrospinning and stabilization of chitosan nanofiber mats. IOP Conf. Ser. Mater. Sci. Eng. 2017, 254, 102006. [Google Scholar] [CrossRef]
- Shang, Z.; An, X.; Liu, L.; Yang, J.; Zhang, W.; Dai, H.; Cao, H.; Xu, Q.; Liu, H.; Ni, Y. Chitin nanofibers as versatile bio-templates of zeolitic imidazolate frameworks for N-doped hierarchically porous carbon electrodes for supercapacitor. Carbohydr. Polym. 2021, 251, 117107. [Google Scholar] [CrossRef]
- Sabantina, L.; Kinzel, F.; Hauser, T.; Többer, A.; Klöcker, M.; Döpke, C.; Böttjer, R.; Wehlage, D.; Rattenholl, A.; Ehrmann, A. Comparative Study of Pleurotus ostreatus Mushroom Grown on Modified PAN Nanofiber Mats. Nanomaterirals 2019, 9, 475. [Google Scholar] [CrossRef]
- Cao, M.; Cheng, W.; Ni, X.; Hu, Y.; Han, G. Lignin-based multi-channels carbon nanofibers @ SnO2 nanocomposites for high-performance supercapacitors. Electrochim. Acta 2020, 345, 136172. [Google Scholar] [CrossRef]
- Sharma, A.; Mandal, T.; Goswami, S. Fabrication of cellulose acetate nanocomposite films with lignocelluosic nanofiber filler for superior effect on thermal, mechanical and optical properties. Nano Struct. Nano Objects 2021, 25, 100642. [Google Scholar] [CrossRef]
- Chen, S.; Li, R.; Li, X.; Xie, J. Electrospinning: An enabling nanotechnology platform for drug delivery and regenerative medicine. Adv. Drug Deliv. Rev. 2018, 132, 188–213. [Google Scholar] [CrossRef]
- Mamun, A. Review of possible applications of nanofibrous mats for wound dressings. Tekstilec 2019, 62, 89–100. [Google Scholar] [CrossRef]
- Ranjan, V.D.; Zeng, P.; Li, B.; Zhang, Y. In vitro cell culture in hollow microfibers with porous structures. Biomater. Sci. 2020, 8, 2175–2188. [Google Scholar] [CrossRef]
- Zarei, M.; Samimi, A.; Khorram, M.; Abdi, M.M.; Golestaneh, S.I. Fabrication and characterization of conductive polypyrrole/chitosan/collagen electrospun nanofiber scaffold for tissue engineering application. Int. J. Biol. Macromol. 2021, 168, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Yalcinkaya, B.; Yalcinkaya, F.; Chaloupek, J. Thin Film Nanofibrous Composite Membrane for Dead-End Seawater Desalination. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
- Roche, R.; Yalcinkaya, F. Electrospun Polyacrylonitrile Nanofibrous Membranes for Point-of-Use Water and Air Cleaning. ChemistryOpen 2019, 8, 97–103. [Google Scholar] [CrossRef]
- Torres-Mendieta, R.; Yalcinkaya, F.; Boyraz, E.; Havelka, O.; Wacławek, S.; Maryška, J.; Černík, M.; Bryjak, M. PVDF nanofibrous membranes modified via laser-synthesized Ag nanoparticles for a cleaner oily water separation. Appl. Surf. Sci. 2020, 526, 146575. [Google Scholar] [CrossRef]
- Choi, J.; Moon, D.S.; Ryu, S.G.; Lee, B.; Ying, W.B.; Lee, K. J. N-chloro hydantoin functionalized polyurethane fibers toward protective cloth against chemical warfare agents. Polymer 2018, 138, 146–155. [Google Scholar] [CrossRef]
- Grothe, T.; Wehlage, D.; Böhm, T.; Remche, A.; Ehrmann, A. Needleless electrospinning of PAN nanofiber mats. Tekstilec 2017, 60, 290–295. [Google Scholar] [CrossRef]
- Maver, T.; Kurečič, M.; Smrke, D.M.; Kleinschek, K.S.; Maver, U. Electrospun nanofibrous CMC/PEO as a part of an effective pain-relieving wound dressing. J. Sol-Gel Sci. Technol. 2016, 79, 475–486. [Google Scholar] [CrossRef]
- Ebrahimi-Hosseinzadeh, B.; Pedram, M.; Hatamian-Zarmi, A.; Salahshour-Kordestani, S.; Rasti, M.; Mokhtari-Hosseini, Z.B.; Mir-Derikvand, M. In vivo evaluation of gelatin/hyaluronic acid nanofiber as Burn-wound healing and its comparison with ChitoHeal gel. Fibers Polym. 2016, 17, 820–826. [Google Scholar] [CrossRef]
- Na, K.H.; Kim, W.T.; Park, D.C.; Shin, H.G.; Lee, S.H.; Park, J.; Song, T.H.; Choi, W.Y. Fabrication and characterization of the magnetic ferrite nanofibers by electrospinning process. Thin Solid Films 2018, 660, 358–364. [Google Scholar] [CrossRef]
- Matos, R.J.R.; Chaparro, C.I.P.; Silva, J.C.; Valente, M.A.; Borges, J.P.; Soares, P.I.P. Electrospun composite cellulose acetate/iron oxide nanoparticles non-woven membranes for magnetic hyperthermia applications. Carbohydr. Polym. 2018, 198, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, Y.; Yuan, M.; Sun, G.; Liao, Q.; Zhang, Y. Solid and macroporous Fe3C/N-C nanofibers with enhanced electromagnetic wave absorbability. Sci. Rep. 2018, 8, 16832. [Google Scholar] [CrossRef]
- Lekota, M.W.; Mpupa, A.; Dimpe, K.M.; Nomngongo, P.N. Preparation of ferric oxide-aluminium oxide carbon nanofiber nanocomposites for ultrasound-assisted dispersive magnetic solid phase extraction of 17-beta estradiol in wastewater. Emerg. Contam. 2020, 6, 162–171. [Google Scholar] [CrossRef]
- Ponnamma, D.; Aljarod, O.; Parangusan, H.; Ali Al-Maadeed, M. Al Electrospun nanofibers of PVDF-HFP composites containing magnetic nickel ferrite for energy harvesting application. Mater. Chem. Phys. 2020, 239, 122257. [Google Scholar] [CrossRef]
- Amini, F.; Blachowicz, T.; Ehrmann, A. Systematic study of magnetization reversal in beaded fibers from different magnetic materials. J. Magn. Magn. Mater. 2021, 529, 167855. [Google Scholar] [CrossRef]
- Kerker, E.; Steinhäußer, D.; Mamun, A.; Trabelsi, M.; Fiedler, J.; Sabantina, L.; Juhász Junger, I.; Schiek, M.; Ehrmann, A.; Kaschuba, R. Spectroscopic investigation of highly-scattering nanofiber mats during drying and film formation. Optik 2020, 208, 164081. [Google Scholar] [CrossRef]
- Garg, C.; Yang, S.H.; Phung, T.; Pushp, A.; Parkin, S.S.P. Dramatic influence of curvature of nanowire on chiral domain wall velocity. Sci. Adv. 2017, 3, e1602804. [Google Scholar] [CrossRef]
- Blachowicz, T.; Ehrmann, A. Magnetization reversal in bent nanofibers of different cross sections. J. Appl. Phys. 2018, 124, 152112. [Google Scholar] [CrossRef]
- Döpke, C.; Grothe, T.; Steblinski, P.; Klöcker, M.; Sabantina, L.; Kosmalska, D.; Blachowicz, T.; Ehrmann, A. Magnetic nanofiber mats for data storage and transfer. Nanomaterials 2019, 9, 92. [Google Scholar] [CrossRef] [PubMed]
- Asmatulu, R.; Khan, W.S. Chapter 7—Electrospun nanofibers for filtration applications. In Synthesis and Applications of Electrospun Nanofibers; Asmatulu, R., Khan, W.S., Eds.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2019; pp. 135–152. ISBN 978-0-12-813914-1. [Google Scholar]
- Xiong, J.; Zhou, M.; Zhang, H.; Quan, Z.; Wang, R.; Qin, X. Sandwich-structured fibrous membranes with low filtration resistance for effective PM 2.5 capture via one-step needleless electrospinning. Mater. Res. Express 2018, 6, 035027. [Google Scholar] [CrossRef]
- Yalcinkaya, F. A review on advanced nanofiber technology for membrane distillation. J. Eng. Fiber. Fabr. 2019, 14, 1558925018824901. [Google Scholar] [CrossRef]
- Mohammed, H.A.; Yaacob, M.H. Chapter 20—Polyaniline-graphite nanocomposite based modified cladding optical fiber gas sensors. In Handbook of Polymer Nanocomposites for Industrial Applications; Hussain, C.M., Ed.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2021; pp. 545–570. ISBN 978-0-12-821497-8. [Google Scholar]
- Chi, Q.; Zhou, Y.; Feng, Y.; Cui, Y.; Zhang, Y.; Zhang, T.; Chen, Q. Excellent energy storage performance of polyetherimide filled by oriented nanofibers with optimized diameters. Mater. Today Energy 2020, 18, 100516. [Google Scholar] [CrossRef]
- Behera, A.; Mallick, P. Chapter 20—Application of nanofibers in aerospace industry. In Fiber-Reinforced Nanocomposites: Fundamentals and Applications; Han, B., Sharma, S., Nguyen, T.A., Longbiao, L., Bhat, K.S., Eds.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2020; pp. 449–457. ISBN 978-0-12-819904-6. [Google Scholar]
- Wang, Y.; Wei, Y.; Wang, B.; Jing, P.; Zhang, Y.; Zhang, Y.; Wang, Q.; Wu, H. Bio-assisted engineering of hierarchical porous carbon nanofiber host in-situ embedded with iron carbide nanocatalysts toward high-performance Li–S batteries. Carbon N. Y. 2021, 177, 60–70. [Google Scholar] [CrossRef]
- Serbezeanu, D.; Popa, A.M.; Stelzig, T.; Sava, I.; Rossi, R.M.; Fortunato, G. Preparation and characterization of thermally stable polyimide membranes by electrospinning for protective clothing applications. Text. Res. J. 2015, 85, 1763–1775. [Google Scholar] [CrossRef]
- Mathew, J.; Joy, J.; George, S.C. Potential applications of nanotechnology in transportation: A review. J. King Saud Univ. Sci. 2019, 31, 586–594. [Google Scholar] [CrossRef]
- Kalalinia, F.; Taherzadeh, Z.; Jirofti, N.; Amiri, N.; Foroghinia, N.; Beheshti, M.; Bazzaz, B.S.F.; Hashemi, M.; Shahroodi, A.; Pishavar, E.; et al. Evaluation of wound healing efficiency of vancomycin-loaded electrospun chitosan/poly ethylene oxide nanofibers in full thickness wound model of rat. Int. J. Biol. Macromol. 2021, 177, 100–110. [Google Scholar] [CrossRef]
- Luu, Y.K.; Kim, K.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA–PEG block copolymers. J. Control. Release 2003, 89, 341–353. [Google Scholar] [CrossRef]
- Subbiah, T.; Bhat, G.S.; Tock, R.W.; Parameswaran, S.; Ramkumar, S.S. Electrospinning of nanofibers. J. Appl. Polym. Sci. 2005, 96, 557–569. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Fujihara, K.; Teo, W.-E.; Yong, T.; Ma, Z.; Ramaseshan, R. Electrospun nanofibers: Solving global issues. Mater. Today 2006, 9, 40–50. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.-J. Drug Delivery Applications of Core-Sheath Nanofibers Prepared by Coaxial Electrospinning: A Review. Pharmaceutics 2019, 11, 305. [Google Scholar] [CrossRef]
- Barnes, C.P.; Sell, S.A.; Knapp, D.C.; Walpoth, B.H.; Brand, D.D.; Bowlin, G.L. Preliminary Investigation of Electrospun Collagen and Polydioxanone for Vascular Tissue Engineering Applications. Int. J. Electrospun Nanofibers Appl. 2007, 1, 73–87. [Google Scholar]
- Welle, A.; Kröger, M.; Döring, M.; Niederer, K.; Pindel, E.; Chronakis, I.S. Electrospun aliphatic polycarbonates as tailored tissue scaffold materials. Biomaterials 2007, 28, 2211–2219. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ali, J.; Zhang, C.; Mailhot, G.; Pan, G. Simultaneously enhanced photocatalytic and antibacterial activities of TiO2/Ag composite nanofibers for wastewater purification. J. Environ. Chem. Eng. 2020, 8, 102104. [Google Scholar] [CrossRef]
- Wu, X.; Li, P.; Cong, L.; Yu, H.; Zhang, D.; Yue, Y.; Xu, H.; Xu, K.; Zheng, X.; Wang, X. Electrospun poly(vinyl alcohol) nanofiber films containing menthol/β-cyclodextrin inclusion complexes for smoke filtration and flavor retention. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 605, 125378. [Google Scholar] [CrossRef]
- Kurian, M.; Paul, A. Recent Trends in the Use of Green Sources for Carbon Dot Synthesis—A Short Review. Carbon Trends 2021, 3, 100032. [Google Scholar] [CrossRef]
- Scott, E.; Peter, F.; Sanders, J. Biomass in the manufacture of industrial products-the use of proteins and amino acids. Appl. Microbiol. Biotechnol. 2007, 75, 751–762. [Google Scholar] [CrossRef]
- García-Mateos, F.J.; Cordero-Lanzac, T.; Berenguer, R.; Morallón, E.; Cazorla-Amorós, D.; Rodríguez-Mirasol, J.; Cordero, T. Lignin-derived Pt supported carbon (submicron)fiber electrocatalysts for alcohol electro-oxidation. Appl. Catal. B Environ. 2017, 211, 18–30. [Google Scholar] [CrossRef]
- García-Mateos, F.J.; Berenguer, R.; Valero-Romero, M.J.; Rodríguez-Mirasol, J.; Cordero, T. Phosphorus functionalization for the rapid preparation of highly nanoporous submicron-diameter carbon fibers by electrospinning of lignin solutions. J. Mater. Chem. A 2018, 6, 1219–1233. [Google Scholar] [CrossRef]
- Berenguer, R.; García-Mateos, F.J.; Ruiz-Rosas, R.; Cazorla-Amorós, D.; Morallón, E.; Rodríguez-Mirasol, J.; Cordero, T. Biomass-derived binderless fibrous carbon electrodes for ultrafast energy storage. Green Chem. 2016, 18, 1506–1515. [Google Scholar] [CrossRef]
- García-Mateos, F.J.; Ruiz-Rosas, R.; María Rosas, J.; Morallón, E.; Cazorla-Amorós, D.; Rodríguez-Mirasol, J.; Cordero, T. Activation of electrospun lignin-based carbon fibers and their performance as self-standing supercapacitor electrodes. Sep. Purif. Technol. 2020, 241, 116724. [Google Scholar] [CrossRef]
- Li, Z.; Liu, J.; Jiang, K.; Thundat, T. Carbonized nanocellulose sustainably boosts the performance of activated carbon in ionic liquid supercapacitors. Nano Energy 2016, 25, 161–169. [Google Scholar] [CrossRef]
- Deng, L.; Young, R.J.; Kinloch, I.A.; Abdelkader, A.M.; Holmes, S.M.; De Haro-Del Rio, D.A.; Eichhorn, S.J. Supercapacitance from cellulose and carbon nanotube nanocomposite fibers. ACS Appl. Mater. Interfaces 2013, 5, 9983–9990. [Google Scholar] [CrossRef]
- Lai, C.; Zhou, Z.; Zhang, L.; Wang, X.; Zhou, Q.; Zhao, Y.; Wang, Y.; Wu, X.F.; Zhu, Z.; Fong, H. Free-standing and mechanically flexible mats consisting of electrospun carbon nanofibers made from a natural product of alkali lignin as binder-free electrodes for high-performance supercapacitors. J. Power Sources 2014, 247, 134–141. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, Y.; Woo, M.W.; Zhang, L.; Xu, K.Z. Preparation of steam activated carbon from black liquor by flue gas precipitation and its performance in hydrogen sulfide removal: Experimental and simulation works. Rev. Mex. Urol. 2016, 76, 395–404. [Google Scholar] [CrossRef]
- Kumar, M.; Hietala, M.; Oksman, K. Lignin-based electrospun carbon nanofibers. Front. Mater. 2019, 6, 1–6. [Google Scholar] [CrossRef]
- Cao, Q.; Zhang, Y.; Chen, J.; Zhu, M.; Yang, C.; Guo, H.; Song, Y.; Li, Y.; Zhou, J. Electrospun biomass based carbon nanofibers as high-performance supercapacitors. Ind. Crops Prod. 2020, 148, 112181. [Google Scholar] [CrossRef]
- Yang, C.; Chen, C.; Pan, Y.; Li, S.; Wang, F.; Li, J.; Li, N.; Li, X.; Zhang, Y.; Li, D. Flexible highly specific capacitance aerogel electrodes based on cellulose nanofibers, carbon nanotubes and polyaniline. Electrochim. Acta 2015, 182, 264–271. [Google Scholar] [CrossRef]
- Ifuku, S.; Nogi, M.; Abe, K.; Yoshioka, M.; Morimoto, M.; Saimoto, H.; Yano, H. Preparation of chitin nanofibers with a uniform width as α-chitin from crab shells. Biomacromolecules 2009, 10, 1584–1588. [Google Scholar] [CrossRef]
- Luo, M.; Ming, Y.; Wang, L.; Li, Y.; Li, B.; Chen, J.; Shi, S. Local delivery of deep marine fungus-derived equisetin from polyvinylpyrrolidone (PVP) nanofibers for anti-MRSA activity. Chem. Eng. J. 2018, 350, 157–163. [Google Scholar] [CrossRef]
- Deng, L.; Zhong, W.; Wang, J.; Zhang, P.; Fang, H.; Yao, L.; Liu, X.; Ren, X.; Li, Y. The enhancement of electrochemical capacitance of biomass-carbon by pyrolysis of extracted nanofibers. Electrochim. Acta 2017, 228, 398–406. [Google Scholar] [CrossRef]
- Duan, B.; Gao, X.; Yao, X.; Fang, Y.; Huang, L.; Zhou, J.; Zhang, L. Unique elastic N-doped carbon nanofibrous microspheres with hierarchical porosity derived from renewable chitin for high rate supercapacitors. Nano Energy 2016, 27, 482–491. [Google Scholar] [CrossRef]
- Widiyastuti, W.; Fahrudin Rois, M.; Suari, N.M.I.P.; Setyawan, H. Activated carbon nanofibers derived from coconut shell charcoal for dye removal application. Adv. Powder Technol. 2020, 31, 3267–3273. [Google Scholar] [CrossRef]
- Chung, S.; Shin, D.; Choun, M.; Kim, J.; Yang, S.; Choi, M.; Kim, J.W.; Lee, J. Improved water management of Pt/C cathode modified by graphitized carbon nanofiber in proton exchange membrane fuel cell. J. Power Sources 2018, 399, 350–356. [Google Scholar] [CrossRef]
- He, Z.; Li, M.; Li, Y.; Zhu, J.; Jiang, Y.; Meng, W.; Zhou, H.; Wang, L.; Dai, L. Flexible electrospun carbon nanofiber embedded with TiO2 as excellent negative electrode for vanadium redox flow battery. Electrochim. Acta 2018, 281, 601–610. [Google Scholar] [CrossRef]
- Abdelkareem, M.A.; Al Haj, Y.; Alajami, M.; Alawadhi, H.; Barakat, N.A.M. Ni-Cd carbon nanofibers as an effective catalyst for urea fuel cell. J. Environ. Chem. Eng. 2018, 6, 332–337. [Google Scholar] [CrossRef]
- Zhao, R.; Yong, X.; Pan, M.; Deng, J.; Pan, K. Aldehyde-containing nanofibers electrospun from biomass vanillin-derived polymer and their application as adsorbent. Sep. Purif. Technol. 2020, 246, 116916. [Google Scholar] [CrossRef]
- Zainab, G.; Babar, A.A.; Ali, N.; Aboalhassan, A.A.; Wang, X.; Yu, J.; Ding, B. Electrospun carbon nanofibers with multi-aperture/opening porous hierarchical structure for efficient CO2 adsorption. J. Colloid Interface Sci. 2020, 561, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Hellert, C.; Wortmann, M.; Frese, N.; Grötsch, G.; Cornelißen, C.; Ehrmann, A. Adhesion of Electrospun Poly(acrylonitrile) Nanofibers on Conductive and Isolating Foil Substrates. Coatings 2021, 11, 249. [Google Scholar] [CrossRef]
- Banner, J.; Dautzenberg, M.; Feldhans, T.; Hofmann, J.; Plümer, P.; Ehrmann, A. Water resistance and morphology of electrospun gelatine blended with citric acid and coconut oil. Tekstilec 2018, 61, 129–135. [Google Scholar] [CrossRef]
- Liu, Y.; Li, K.; Mohideen, M.; Ramakrishna, S. (Eds.) Chapter 1—Development of melt electrospinning: The past, present, and future. In Melt Electrospinning; Academic Press: Cambridge, MA, USA, 2019; pp. 1–5. ISBN 978-0-12-816220-0. [Google Scholar]
- Kijeńska, E.; Swieszkowski, W. 2—General requirements of electrospun materials for tissue engineering: Setups and strategy for successful electrospinning in laboratory and industry. In Electrospun Materials for Tissue Engineering and Biomedical Applications; Uyar, T., Kny, E., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 43–56. ISBN 978-0-08-101022-8. [Google Scholar]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Khalili Amand, F.; Esmaeili, A. Investigating the properties of electrospun nanofibers made of hybride polymer containing anticoagulant drugs. Carbohydr. Polym. 2020, 228, 115397. [Google Scholar] [CrossRef] [PubMed]
- Jahan, I.; Jadhav, A.; Wang, L.; Wang, X. Electrospinning from a convex needle with multiple jet toward better controlling and enhanced production rate. J. Appl. Polym. Sci. 2019, 136, 1–7. [Google Scholar] [CrossRef]
- Hwang, M.; Karenson, M.O.; Elabd, Y.A. High Production Rate of High Purity, High Fidelity Nafion Nanofibers via Needleless Electrospinning. ACS Appl. Polym. Mater. 2019, 1, 2731–2740. [Google Scholar] [CrossRef]
- Rosenthal, T.; Weller, J.M.; Chan, C.K. Needleless Electrospinning for High Throughput Production of Li7La3Zr2O12 Solid Electrolyte Nanofibers. Ind. Eng. Chem. Res. 2019, 58, 17399–17405. [Google Scholar] [CrossRef]
- Yalcinkaya, F.; Komarek, M. Polyvinyl Butyral (PVB) Nanofiber/Nanoparticle-Covered Yarns for Antibacterial Textile Surfaces. Int. J. Mol. Sci. 2019, 20, 4317. [Google Scholar] [CrossRef]
- Bavatharani, C.; Muthusankar, E.; Wabaidur, S.M.; Alothman, Z.A.; Alsheetan, K.M.; mana AL-Anazy, M.; Ragupathy, D. Electrospinning technique for production of polyaniline nanocomposites/nanofibres for multi-functional applications: A review. Synth. Met. 2021, 271, 116609. [Google Scholar] [CrossRef]
- Sundera Murthe, S.; Mohamed Saheed, M.S.; Perumal, V.; Mohamed Saheed, M.S.; Mohamed, N.M. 11—Electrospun Nanofibers for Biosensing Applications. In Nanobiosensors for Biomolecular Targeting; Gopinath, S.C.B., Lakshmipriya, T., Eds.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2019; pp. 253–267. ISBN 978-0-12-813900-4. [Google Scholar]
- Trabelsi, M.; Mamun, A.; Klöcker, M.; Sabantina, L.; Großerhode, C.; Blachowicz, T.; Ehrmann, A. Increased mechanical properties of carbon nanofiber mats for possible medical applications. Fibers 2019, 7, 98. [Google Scholar] [CrossRef]
- Sabantina, L.; Hes, L.; Mirasol, J.R.; Cordero, T.; Ehrmann, A. Water vapor permeability through PAN nanofiber mat with varying membrane-like areas. Fibres Text. East. Eur. 2019, 27, 12–15. [Google Scholar] [CrossRef]
- Molnár, K.; Szolnoki, B.; Toldy, A.; Vas, L.M. Thermochemical stabilization and analysis of continuously electrospun nanofibers: Carbon nanotube-loaded polyacrylonitrile nanofibers for high performance carbon nanofiber mass production. J. Therm. Anal. Calorim. 2014, 117, 1123–1135. [Google Scholar] [CrossRef]
- Liu, C.; Lafdi, K. Fabrication and characterization of carbon nanofibers from polyacrylonitrile/pitch blends. J. Appl. Polym. Sci. 2017, 134, 45388. [Google Scholar] [CrossRef]
- Gergin, I.; Ismar, E.; Sarac, A.S. Oxidative stabilization of polyacrylonitrile nanofibers and carbon nanofibers containing graphene oxide (GO): A spectroscopic and electrochemical study. Beilstein J. Nanotechnol. 2017, 8, 1616–1628. [Google Scholar] [CrossRef]
- Sabantina, L.; Klöcker, M.; Wortmann, M.; Mirasol, J.R.; Cordero, T.; Moritzer, E.; Finsterbusch, K.; Ehrmann, A. Stabilization of polyacrylonitrile nanofiber mats obtained by needleless electrospinning using dimethyl sulfoxide as solvent. J. Ind. Text. 2020, 50, 224–239. [Google Scholar] [CrossRef]
- Sabantina, L.; Mirasol, J.R.; Cordero, T.; Finsterbusch, K.; Ehrmann, A. Investigation of needleless electrospun PAN nanofiber mats. AIP Conf. Proc. 2018, 1952, 020085. [Google Scholar] [CrossRef]
- Bashir, Z. A critical review of the stabilisation of polyacrylonitrile. Carbon N. Y. 1991, 29, 1081–1090. [Google Scholar] [CrossRef]
- Dalton, S.; Heatley, F.; Budd, P.M. Thermal stabilization of polyacrylonitrile fibres. Polymer 1999, 40, 5531–5543. [Google Scholar] [CrossRef]
- Ibupoto, A.S.; Qureshi, U.A.; Ahmed, F.; Khatri, Z.; Khatri, M.; Maqsood, M.; Brohi, R.Z.; Kim, I.S. Reusable carbon nanofibers for efficient removal of methylene blue from aqueous solution. Chem. Eng. Res. Des. 2018, 136, 744–752. [Google Scholar] [CrossRef]
- Jo, E.; Yeo, J.G.; Kim, D.K.; Oh, J.S.; Hong, C.K. Preparation of well-controlled porous carbon nanofiber materials by varying the compatibility of polymer blends. Polym. Int. 2014, 63, 1471–1477. [Google Scholar] [CrossRef]
- Ji, L.; Yao, Y.; Toprakci, O.; Lin, Z.; Liang, Y.; Shi, Q.; Medford, A.J.; Millns, C.R.; Zhang, X. Fabrication of carbon nanofiber-driven electrodes from electrospun polyacrylonitrile/polypyrrole bicomponents for high-performance rechargeable lithium-ion batteries. J. Power Sources 2010, 195, 2050–2056. [Google Scholar] [CrossRef]
- García-Mateos, F.J.; Ruiz-Rosas, R.; Rosas, J.M.; Rodríguez-Mirasol, J.; Cordero, T. Controlling the Composition, Morphology, Porosity, and Surface Chemistry of Lignin-Based Electrospun Carbon Materials. Front. Mater. 2019, 6, 114. [Google Scholar] [CrossRef]
- Miao, F.; Shao, C.; Li, X.; Wang, K.; Liu, Y. Flexible solid-state supercapacitors based on freestanding nitrogen-doped porous carbon nanofibers derived from electrospun polyacrylonitrile@polyaniline nanofibers. J. Mater. Chem. A 2016, 4, 4180–4187. [Google Scholar] [CrossRef]
- Li, F.; Li, J.; Chen, L.; Dong, Y.; Xie, P.; Li, Q. Hydrogen production through hydrolysis of sodium borohydride: Highly dispersed CoB particles immobilized in carbon nanofibers as a novel catalyst. Int. J. Hydrogen Energy 2020, 45, 32145–32156. [Google Scholar] [CrossRef]
- Mamun, A.; Trabelsi, M.; Klöcker, M.; Lukas Storck, J.; Böttjer, R.; Sabantina, L. Needleless electrospun polyacrylonitrile/konjac glucomannan nanofiber mats. J. Eng. Fiber. Fabr. 2020, 15, 1558925020964806. [Google Scholar] [CrossRef]
- Ma, C.; Li, Z.; Li, J.; Fan, Q.; Wu, L.; Shi, J.; Song, Y. Lignin-based hierarchical porous carbon nanofiber films with superior performance in supercapacitors. Appl. Surf. Sci. 2018, 456, 568–576. [Google Scholar] [CrossRef]
- Calvo-Muñoz, E.M.; García-Mateos, F.J.; Rosas, J.M.; Rodríguez-Mirasol, J.; Cordero, T. Biomass waste carbon materials as adsorbents for CO 2 capture under post-combustion conditions. Front. Mater. 2016, 3, 1–14. [Google Scholar] [CrossRef]
- Wang, Z.; Kang, K.; Wu, J.; Hu, Q.; Harper, D.P.; Du, G.; Wang, S.; Xu, K. Comparative effects of electrospinning ways for fabricating green, sustainable, flexible, porous, nanofibrous cellulose/chitosan carbon mats as anode materials for lithium-ion batteries. J. Mater. Res. Technol. 2021, 11, 50–61. [Google Scholar] [CrossRef]
- Tao, L.; Huang, Y.; Zheng, Y.; Yang, X.; Liu, C.; Di, M.; Larpkiattaworn, S.; Nimlos, M.R.; Zheng, Z. Porous carbon nanofiber derived from a waste biomass as anode material in lithium-ion batteries. J. Taiwan Inst. Chem. Eng. 2019, 95, 217–226. [Google Scholar] [CrossRef]
- Song, K.; Wu, Q.; Zhang, Z.; Ren, S.; Lei, T.; Negulescu, I.I.; Zhang, Q. Porous Carbon Nanofibers from Electrospun Biomass Tar/Polyacrylonitrile/Silver Hybrids as Antimicrobial Materials. ACS Appl. Mater. Interfaces 2015, 7, 15108–15116. [Google Scholar] [CrossRef] [PubMed]
- Golmohammadi, F.; Amiri, M. Fabrication of MEA from Biomass-Based Carbon Nanofibers Composited with Nickel-Cobalt Oxides as a New Electrocatalyst for Oxygen Reduction Reaction in Passive Direct Methanol Fuel Cells. Electrocatalysis 2021, 11, 485–496. [Google Scholar] [CrossRef]
- Zhang, Y.-Q.; Shi, G.-F.; Chen, B.; Wang, G.-Y.; Guo, T.-C. Biomass-Based Carbon Nanofibers Prepared by Electrospinning for Supercapacitor. J. Nanosci. Nanotechnol. 2018, 18, 5731–5737. [Google Scholar] [CrossRef]
- Zhu, M.; Liu, H.; Cao, Q.; Zheng, H.; Xu, D.; Guo, H.; Wang, S.; Li, Y.; Zhou, J. Electrospun Lignin-Based Carbon Nanofibers as Supercapacitor Electrodes. ACS Sustain. Chem. Eng. 2020, 8, 12831–12841. [Google Scholar] [CrossRef]
- Jung, K.H.; Deng, W.; Smith, D.W.; Ferraris, J.P. Carbon nanofiber electrodes for supercapacitors derived from new precursor polymer: Poly(acrylonitrile-co-vinylimidazole). Electrochem. Commun. 2012, 23, 149–152. [Google Scholar] [CrossRef]
- Guo, J.; Sun, G.; Wang, Q.; Wang, G.; Zhou, Z.; Tang, S.; Jiang, L.; Zhou, B.; Xin, Q. Carbon nanofibers supported Pt-Ru electrocatalysts for direct methanol fuel cells. Carbon N. Y. 2006, 44, 152–157. [Google Scholar] [CrossRef]
- Zou, G.; Zhang, D.; Dong, C.; Li, H.; Xiong, K.; Fei, L.; Qian, Y. Carbon nanofibers: Synthesis, characterization, and electrochemical properties. Carbon N. Y. 2006, 44, 828–832. [Google Scholar] [CrossRef]
- García-Díaz, I.; López, F.A.; Alguacil, F.J. Carbon nanofibers: A new adsorbent for copper removal from wastewater. Metals 2018, 8, 914. [Google Scholar] [CrossRef]
- Chakraborty, A.; Deva, D.; Sharma, A.; Verma, N. Adsorbents based on carbon microfibers and carbon nanofibers for the removal of phenol and lead from water. J. Colloid Interface Sci. 2011, 359, 228–239. [Google Scholar] [CrossRef]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15–30. [Google Scholar] [CrossRef]
- Kruss, S.; Hilmer, A.J.; Zhang, J.; Reuel, N.F.; Mu, B.; Strano, M.S. Carbon nanotubes as optical biomedical sensors. Adv. Drug Deliv. Rev. 2013, 65, 1933–1950. [Google Scholar] [CrossRef] [PubMed]
- Sheikhpour, M.; Golbabaie, A.; Kasaeian, A. Carbon nanotubes: A review of novel strategies for cancer diagnosis and treatment. Mater. Sci. Eng. C 2017, 76, 1289–1304. [Google Scholar] [CrossRef]
- Liang, M.; Liu, Y.; Xiao, B.; Yang, S.; Wang, Z.; Han, H. An analytical model for the transverse permeability of gas diffusion layer with electrical double layer effects in proton exchange membrane fuel cells. Int. J. Hydrogen Energy 2018, 43, 17880–17888. [Google Scholar] [CrossRef]
- Liang, M.; Fu, C.; Xiao, B.; Luo, L.; Wang, Z. A fractal study for the effective electrolyte diffusion through charged porous media. Int. J. Heat Mass Transf. 2019, 137, 365–371. [Google Scholar] [CrossRef]
- Chan, S.; Jankovic, J.; Susac, D.; Saha, M.S.; Tam, M.; Yang, H.; Ko, F. Electrospun carbon nanofiber catalyst layers for polymer electrolyte membrane fuel cells: Structure and performance. J. Power Sources 2018, 392, 239–250. [Google Scholar] [CrossRef]
- Ponomarev, I.I.; Skupov, K.M.; Naumkin, A.V.; Basu, V.G.; Zhigalina, O.M.; Razorenov, D.Y.; Ponomarev, I.I.; Volkova, Y.A. Probing of complex carbon nanofiber paper as gas-diffusion electrode for high temperature polymer electrolyte membrane fuel cell. RSC Adv. 2019, 9, 257–267. [Google Scholar] [CrossRef]
- Bui, H.T.; Kim, D.Y.; Kim, D.W.; Suk, J.; Kang, Y. Carbon nanofiber@platinum by a coaxial electrospinning and their improved electrochemical performance as a Li−O2 battery cathode. Carbon N. Y. 2018, 130, 94–104. [Google Scholar] [CrossRef]
- Yoon, K.R.; Shin, K.; Park, J.; Cho, S.H.; Kim, C.; Jung, J.W.; Cheong, J.Y.; Byon, H.R.; Lee, H.M.; Kim, I.D. Brush-Like Cobalt Nitride Anchored Carbon Nanofiber Membrane: Current Collector-Catalyst Integrated Cathode for Long Cycle Li-O2 Batteries. ACS Nano 2018, 12, 128–139. [Google Scholar] [CrossRef]
- Xia, J.; Yuan, Y.; Yan, H.; Liu, J.; Zhang, Y.; Liu, L.; Zhang, S.; Li, W.; Yang, X.; Shu, H.; et al. Electrospun SnSe/C nanofibers as binder-free anode for lithium–ion and sodium-ion batteries. J. Power Sources 2020, 449, 227559. [Google Scholar] [CrossRef]
- Zhao, W.; Ci, S.; Hu, X.; Chen, J.; Wen, Z. Highly dispersed ultrasmall NiS2 nanoparticles in porous carbon nanofiber anodes for sodium ion batteries. Nanoscale 2019, 11, 4688–4695. [Google Scholar] [CrossRef]
- Zhou, P.; Zhang, M.; Wang, L.; Huang, Q.; Su, Z.; Li, L.; Wang, X.; Li, Y.; Zeng, C.; Guo, Z. Synthesis and Electrochemical Performance of ZnSe Electrospinning Nanofibers as an Anode Material for Lithium Ion and Sodium Ion Batteries. Front. Chem. 2019, 7, 1–10. [Google Scholar] [CrossRef]
- Yin, H.; Qu, H.Q.; Liu, Z.; Jiang, R.Z.; Li, C.; Zhu, M.Q. Long cycle life and high rate capability of three dimensional CoSe 2 grain-attached carbon nanofibers for flexible sodium-ion batteries. Nano Energy 2019, 58, 715–723. [Google Scholar] [CrossRef]
- Dai, Z.; Ren, P.-G.; Jin, Y.-L.; Zhang, H.; Ren, F.; Zhang, Q. Nitrogen-sulphur Co-doped graphenes modified electrospun lignin/polyacrylonitrile-based carbon nanofiber as high performance supercapacitor. J. Power Sources 2019, 437, 226937. [Google Scholar] [CrossRef]
- Wang, P.; Gong, Z.; Ye, K.; Gao, Y.; Zhu, K.; Yan, J.; Wang, G.; Cao, D. The stable lithium metal cell with two-electrode biomass carbon. Electrochim. Acta 2020, 356, 136824. [Google Scholar] [CrossRef]
- Worch, E. Adsorption Technology in Water Treatment; Walter de Gruyter GmbH: Berlin, Germany; Co. KG: Boston, MA, USA, 2012; ISBN 9783110240221. [Google Scholar]
- Dey, S.; Bano, F.; Malik, A. Pharmaceuticals and personal care product (PPCP) contamination—A global discharge inventory. In Pharmaceuticals and Personal Care Products: Waste Management and Treatment Technology; Butterworth-Heinemann, Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 1–26. ISBN 9780128161890. [Google Scholar]
- Zhou, X.; Liu, B.; Chen, Y.; Guo, L.; Wei, G. Carbon nanofiber-based three-dimensional nanomaterials for energy and environmental applications. Mater. Adv. 2020, 1, 2163–2181. [Google Scholar] [CrossRef]
- Gan, L.; Geng, A.; Song, C.; Xu, L.; Wang, L.; Fang, X.; Han, S.; Cui, J.; Mei, C. Simultaneous removal of rhodamine B and Cr(VI) from water using cellulose carbon nanofiber incorporated with bismuth oxybromide: The effect of cellulose pyrolysis temperature on photocatalytic performance. Environ. Res. 2020, 185, 109414. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Babar, A.A.; Zhang, Y.; Iqbal, N.; Wang, X.; Yu, J.; Ding, B. Porous, flexible, and core-shell structured carbon nanofibers hybridized by tin oxide nanoparticles for efficient carbon dioxide capture. J. Colloid Interface Sci. 2020, 560, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.C.; Wu, C.Y.; Chen, Y.J. Effects of activation on the properties of electrospun carbon nanofibers and their adsorption performance for carbon dioxide. Sep. Purif. Technol. 2020, 233, 116040. [Google Scholar] [CrossRef]
- Chaniotakis, N.; Vamvakaki, V.; Tsagaraki, K.; Chaniotakis, N. Carbon Nanofiber-Based Glucose Biosensor Carbon Nanofiber-Based Glucose Biosensor. Anal. Chem. 2006, 78, 5538–5542. [Google Scholar]
- Tran, P.A.; Zhang, L.; Webster, T.J. Carbon nanofibers and carbon nanotubes in regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 1097–1114. [Google Scholar] [CrossRef] [PubMed]
- Swisher, L.Z.; Prior, A.M.; Gunaratna, M.J.; Shishido, S.; Madiyar, F.; Nguyen, T.A.; Hua, D.H.; Li, J. Quantitative electrochemical detection of cathepsin B activity in breast cancer cell lysates using carbon nanofiber nanoelectrode arrays toward identification of cancer formation. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1695–1704. [Google Scholar] [CrossRef]
- Olenic, L.; Mihailescu, G.; Pruneanu, S.; Lupu, D.; Biris, A.R.; Margineanu, P.; Garabagiu, S.; Biris, A.S. Investigation of carbon nanofibers as support for bioactive substances. J. Mater. Sci. Mater. Med. 2009, 20, 177–183. [Google Scholar] [CrossRef]
- Nguyen-Vu, T.D.B.; Chen, H.; Cassell, A.M.; Andrews, R.J.; Meyyappan, M.; Li, J. Vertically aligned carbon nanofiber architecture as a multifunctional 3-D neural electrical interface. IEEE Trans. Biomed. Eng. 2007, 54, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Webster, T.J.; Waid, M.C.; McKenzie, J.L.; Price, R.L.; Ejiofor, J.U. Nano-biotechnology: Carbon nanofibres as improved neural and orthopaedic implants. Nanotechnology 2004, 15, 48–54. [Google Scholar] [CrossRef]
- Khang, D.; Sato, M.; Price, R.L.; Ribbe, A.E.; Webster, T.J. Selective adhesion and mineral deposition by osteoblasts on carbon nanofiber patterns. Int. J. Nanomed. 2006, 1, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.; Haniu, H.; Kim, Y.A.; Saito, N. The use of electrospun organic and carbon nanofibers in bone regeneration. Nanomaterials 2020, 10, 562. [Google Scholar] [CrossRef]
- Pilehvar-Soltanahmadi, Y.; Dadashpour, M.; Mohajeri, A.; Fattahi, A.; Sheervalilou, R.; Zarghami, N. An Overview on Application of Natural Substances Incorporated with Electrospun Nanofibrous Scaffolds to Development of Innovative Wound Dressings. Mini Rev. Med. Chem. 2017, 18, 414–427. [Google Scholar] [CrossRef]
- Sylvester, M.A.; Amini, F.; Keat, T.C. Electrospun nanofibers in wound healing. Mater. Today Proc. 2019, 29, 1–6. [Google Scholar] [CrossRef]
- Saito, N.; Aoki, K.; Usui, Y.; Shimizu, M.; Hara, K.; Narita, N.; Ogihara, N.; Nakamura, K.; Ishigaki, N.; Kato, H.; et al. Application of carbon fibers to biomaterials: A new era of nano-level control of carbon fibers after 30-years of development. Chem. Soc. Rev. 2011, 40, 3824–3834. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Aboagye, A.; Kelkar, A.; Lai, C.; Fong, H. A review: Carbon nanofibers from electrospun polyacrylonitrile and their applications. J. Mater. Sci. 2014, 49, 463–480. [Google Scholar] [CrossRef]
- Smolka, W.; Panek, A.; Gubernat, M.; Szczypta-Fraczek, A.; Jelen, P.; Paluszkiewicz, C.; Markowski, J.; Blazewicz, M. Structure and Biological Properties of Surface-Engineered Carbon Nanofibers. J. Nanomater. 2019, 2019, 4146190. [Google Scholar] [CrossRef]
- Li, Z.; Milionis, A.; Zheng, Y.; Yee, M.; Codispoti, L.; Tan, F.; Poulikakos, D.; Yap, C.H. Superhydrophobic hemostatic nanofiber composites for fast clotting and minimal adhesion. In Proceedings of the Nature Communications; Springer: New York, NY, USA, 2019; Volume 10. [Google Scholar]
- Ding, Y.; Li, W.; Zhang, F.; Liu, Z.; Zanjanizadeh Ezazi, N.; Liu, D.; Santos, H.A. Electrospun Fibrous Architectures for Drug Delivery, Tissue Engineering and Cancer Therapy. Adv. Funct. Mater. 2019, 29, 1802852. [Google Scholar] [CrossRef]
- Scaffaro, R.; Maio, A.; Lopresti, F.; Botta, L. Nanocarbons in electrospun polymeric nanomats for tissue engineering: A review. Polymers 2017, 9, 76. [Google Scholar] [CrossRef] [PubMed]
- Ngadiman, N.H.A.; Noordin, M.Y.; Idris, A.; Kurniawan, D. A review of evolution of electrospun tissue engineering scaffold: From two dimensions to three dimensions. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2017, 231, 597–616. [Google Scholar] [CrossRef]
- Ma, G.; Fang, D.; Liu, Y.; Zhu, X.; Nie, J. Electrospun sodium alginate/poly(ethylene oxide) core-shell nanofibers scaffolds potential for tissue engineering applications. Carbohydr. Polym. 2012, 87, 737–743. [Google Scholar] [CrossRef]
- Xu, C.; Inai, R.; Kotaki, M.; Ramakrishna, S. Electrospun nanofiber fabrication as synthetic extracellular matrix and its potential for vascular tissue engineering. Tissue Eng. 2004, 10, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Ndreu, A.; Nikkola, L.; Ylikauppila, H.; Ashammakhi, N.; Hasirci, V. Electrospun biodegradable nanofibrous mats for tissue engineering. Nanomedicine 2008, 3, 45–60. [Google Scholar] [CrossRef]
- Heidari, M.; Bahrami, S.H.; Ranjbar-Mohammadi, M.; Milan, P.B. Smart electrospun nanofibers containing PCL/gelatin/graphene oxide for application in nerve tissue engineering. Mater. Sci. Eng. C 2019, 103, 109768. [Google Scholar] [CrossRef] [PubMed]
- Yadav, D.; Amini, F.; Ehrmann, A. Recent advances in carbon nanofibers and their applications—A review. Eur. Polym. J. 2020, 138, 109963. [Google Scholar] [CrossRef]
- Nekounam, H.; Allahyari, Z.; Gholizadeh, S.; Mirzaei, E.; Shokrgozar, M.A.; Faridi-Majidi, R. Simple and robust fabrication and characterization of conductive carbonized nanofibers loaded with gold nanoparticles for bone tissue engineering applications. Mater. Sci. Eng. C 2020, 117, 111226. [Google Scholar] [CrossRef]
- Samadian, H.; Mobasheri, H.; Hasanpour, S.; Ai, J.; Azamie, M.; Faridi-Majidi, R. Electro-conductive carbon nanofibers as the promising interfacial biomaterials for bone tissue engineering. J. Mol. Liq. 2020, 298, 112021. [Google Scholar] [CrossRef]
- Stocco, T.D.; Antonioli, E.; Romagnolli, M.L.; Sousa, G.F.; Ferretti, M.; Lobo, A.O. Aligned biomimetic scaffolds based on carbon nanotubes-reinforced polymeric nanofibers for knee meniscus tissue engineering. Mater. Lett. 2020, 264, 127351. [Google Scholar] [CrossRef]
- Patel, K.D.; Kim, T.H.; Mandakhbayar, N.; Singh, R.K.; Jang, J.H.; Lee, J.H.; Kim, H.W. Coating biopolymer nanofibers with carbon nanotubes accelerates tissue healing and bone regeneration through orchestrated cell- and tissue-regulatory responses. Acta Biomater. 2020, 108, 97–110. [Google Scholar] [CrossRef]
- Serafin, A.; Murphy, C.; Rubio, M.C.; Collins, M.N. Printable alginate/gelatin hydrogel reinforced with carbon nanofibers as electrically conductive scaffolds for tissue engineering. Mater. Sci. Eng. C 2021, 122, 111927. [Google Scholar] [CrossRef] [PubMed]
- Zhijiang, C.; Cong, Z.; Jie, G.; Qing, Z.; Kongyin, Z. Electrospun carboxyl multi-walled carbon nanotubes grafted polyhydroxybutyrate composite nanofibers membrane scaffolds: Preparation, characterization and cytocompatibility. Mater. Sci. Eng. C 2018, 82, 29–40. [Google Scholar] [CrossRef]
- Dai, J.; Luo, Y.; Nie, D.; Jin, J.; Yang, S.; Li, G.; Yang, Y.; Zhang, W. pH/photothermal dual-responsive drug delivery and synergistic chemo-photothermal therapy by novel porous carbon nanofibers. Chem. Eng. J. 2020, 397, 125402. [Google Scholar] [CrossRef]
- Abdal-hay, A.; Taha, M.; Mousa, H.M.; Bartnikowski, M.; Hassan, M.L.; Dewidar, M.; Ivanovski, S. Engineering of electrically-conductive poly(ε-caprolactone)/ multi-walled carbon nanotubes composite nanofibers for tissue engineering applications. Ceram. Int. 2019, 45, 15736–15740. [Google Scholar] [CrossRef]
- Shrestha, B.K.; Shrestha, S.; Tiwari, A.P.; Kim, J.I.; Ko, S.W.; Kim, H.J.; Park, C.H.; Kim, C.S. Bio-inspired hybrid scaffold of zinc oxide-functionalized multi-wall carbon nanotubes reinforced polyurethane nanofibers for bone tissue engineering. Mater. Des. 2017, 133, 69–81. [Google Scholar] [CrossRef]
- Mamidi, N.; Delgadillo, R.M.V.; Ortiz, A.G.; Barrera, E.V. Carbon nano-onions reinforced multilayered thin film system for stimuli-responsive drug release. Pharmaceutics 2020, 12, 1208. [Google Scholar] [CrossRef] [PubMed]
- Mykhailiv, O.; Zubyk, H.; Plonska-Brzezinska, M.E. Carbon nano-onions: Unique carbon nanostructures with fascinating properties and their potential applications. Inorg. Chim. Acta 2017, 468, 49–66. [Google Scholar] [CrossRef]
- Ahlawat, J.; Masoudi Asil, S.; Guillama Barroso, G.; Nurunnabi, M.; Narayan, M. Application of carbon nano onions in the biomedical field: Recent advances and challenges. Biomater. Sci. 2021, 9, 626–644. [Google Scholar] [CrossRef]
- Jin, H.; Wu, S.; Li, T.; Bai, Y.; Wang, X.; Zhang, H.; Xu, H.; Kong, C.; Wang, H. Synthesis of porous carbon nano-onions derived from rice husk for high-performance supercapacitors. Appl. Surf. Sci. 2019, 488, 593–599. [Google Scholar] [CrossRef]
- Breczko, J.; Winkler, K.; Plonska-Brzezinska, M.E.; Villalta-Cerdas, A.; Echegoyen, L. Electrochemical properties of composites containing small carbon nano-onions and solid polyelectrolytes. J. Mater. Chem. 2010, 20, 7761–7768. [Google Scholar] [CrossRef]
- Breczko, J.; Plonska-Brzezinska, M.E.; Echegoyen, L. Electrochemical oxidation and determination of dopamine in the presence of uric and ascorbic acids using a carbon nano-onion and poly(diallyldimethylammonium chloride) composite. Electrochim. Acta 2012, 72, 61–67. [Google Scholar] [CrossRef]
- Giordani, S.; Bartelmess, J.; Frasconi, M.; Biondi, I.; Cheung, S.; Grossi, M.; Wu, D.; Echegoyen, L.; O’Shea, D.F. NIR fluorescence labelled carbon nano-onions: Synthesis, analysis and cellular imaging. J. Mater. Chem. B 2014, 2, 7459–7463. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).