The Interplay between Drug and Sorbitol Contents Determines the Mechanical and Swelling Properties of Potential Rice Starch Films for Buccal Drug Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Rice Starch Films
2.3. Characterisation of Rice Starch Films
2.3.1. Film Thickness
2.3.2. Water Content
2.3.3. Drug Loading Efficiency
2.3.4. Water Absorption Capacity and Kinetics
2.3.5. Mechanical Properties
2.3.6. Attenuated Total Reflectance—Fourier Transform Infrared (ATR–FTIR) Spectroscopy
2.3.7. In Vitro Drug Dissolution Study
3. Results
3.1. Film Thickness, Water Content and Drug Loading Efficiency
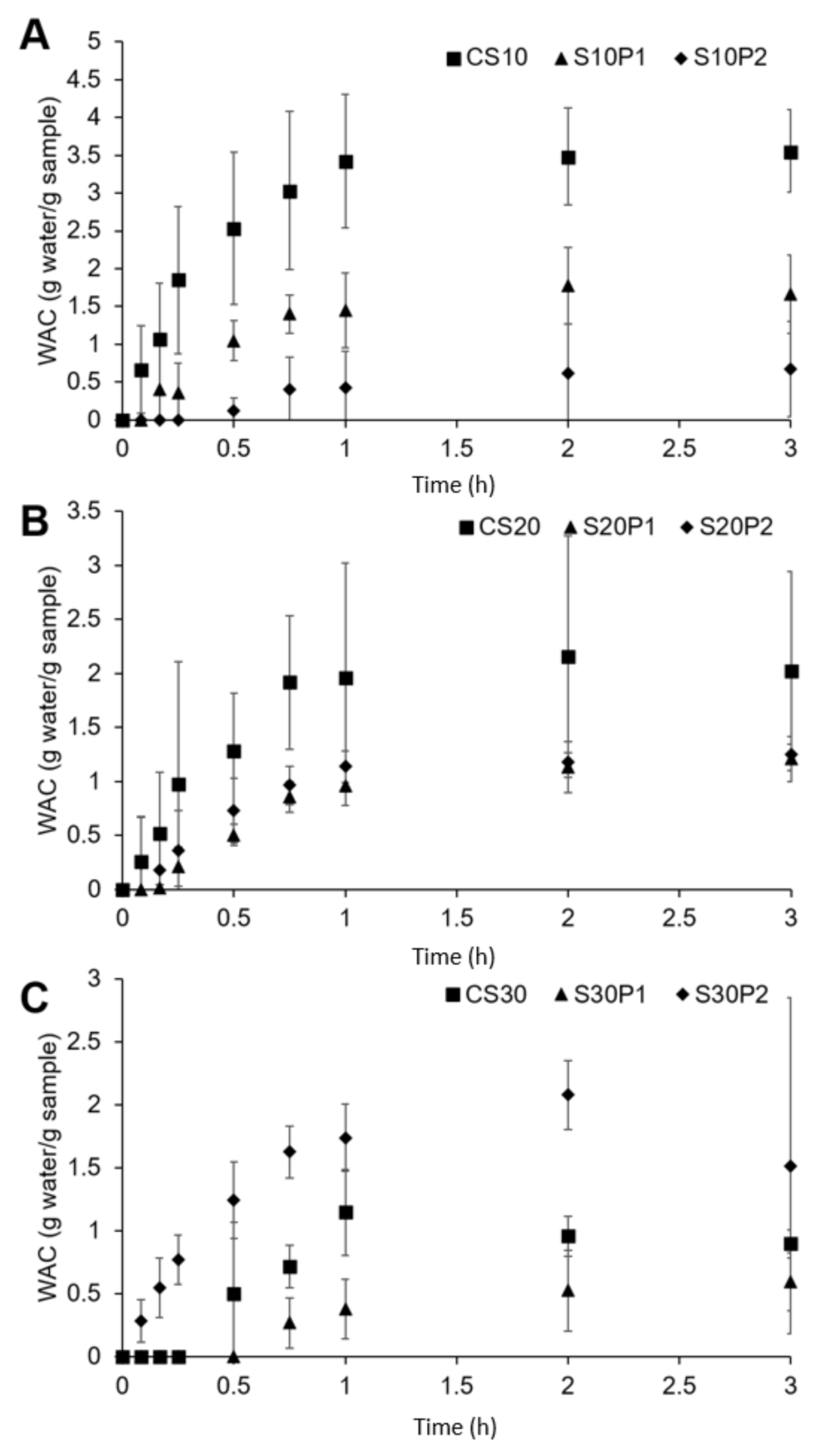
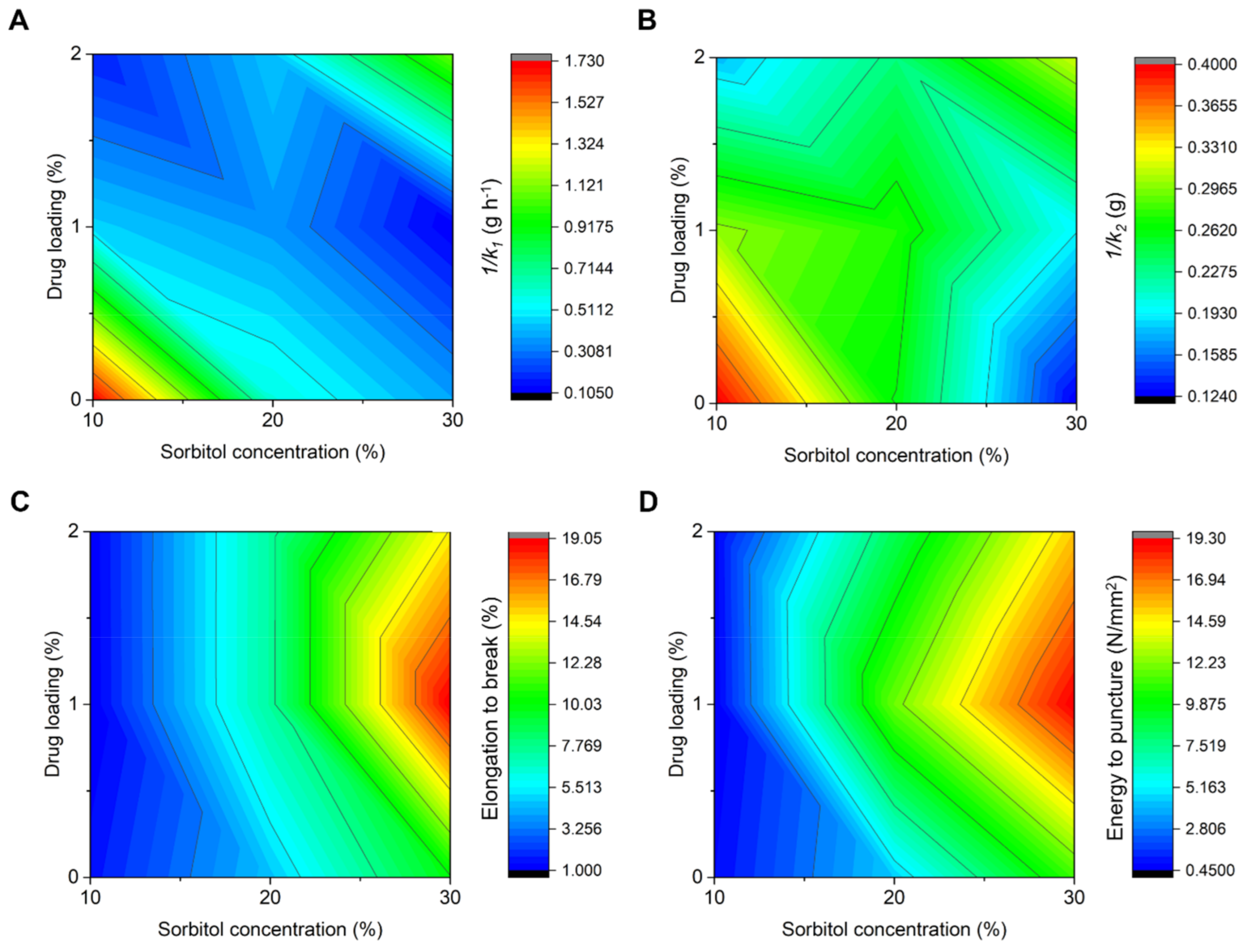
3.2. Water Absorption Capacity and Kinetics
3.3. Mechanical Properties
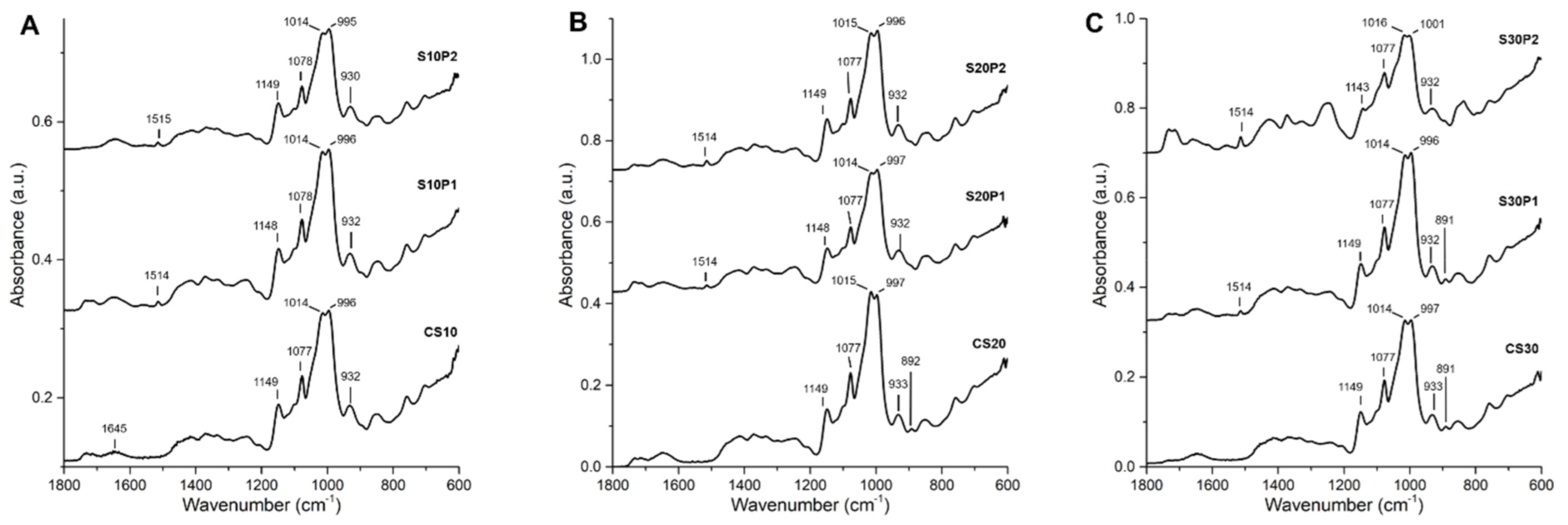
3.4. ATR-FTIR Spectroscopy
3.5. In Vitro Drug Dissolution Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Khan, S.; Boateng, J.S.; Mitchell, J.; Trivedi, V. Formulation, characterisation and stabilisation of buccal films for paediatric drug delivery of omeprazole. AAPS PharmSciTech 2015, 16, 800–810. [Google Scholar]
- Ansari, M.; Sadarani, B.; Majumdar, A. Optimization and evaluation of mucoadhesive buccal films loaded with resveratrol. J. Drug Deliv. Sci. Technol. 2018, 44, 278–288. [Google Scholar] [CrossRef]
- Nair, A.B.; Al-Dhubiab, B.E.; Shah, J.; Vimal, P.; Attimarad, M.; Harsha, S. Development and evaluation of palonosetron loaded mucoadhesive buccal films. J. Drug Deliv. Sci. Technol. 2018, 47, 351–358. [Google Scholar]
- Trastullo, R.; Abruzzo, A.; Saladini, B.; Gallucci, M.C.; Cerchiara, T.; Luppi, B.; Bigucci, F. Design and evaluation of buccal films as paediatric dosage form for transmucosal delivery of ondansetron. Eur. J. Pharm. Biopharm. 2016, 105, 115–121. [Google Scholar] [CrossRef]
- Salamat-Miller, N.; Chittchang, M.; Johnston, T.P. The use of mucoadhesive polymers in buccal drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1666–1691. [Google Scholar] [CrossRef] [PubMed]
- Tejada, G.; Barrera, M.; Piccirilli, G.; Sortino, M.; Frattini, A.; Salomón, C.; Lamas, M.C.; Leonardi, D. Development and evaluation of buccal films based on chitosan for the potential treatment of oral candidiasis. AAPS PharmSciTech 2017, 18, 936–946. [Google Scholar] [CrossRef]
- Preis, M.; Woertz, C.; Kleinebudde, P.; Breitkreutz, J. Oromucosal film preparations: Classification and characterization methods. Expert Opin. Drug Deliv. 2013, 10, 1303–1317. [Google Scholar]
- Aldawsari, H.M.; Badr-Eldin, S.M. Enhanced pharmacokinetic performance of dapoxetine hydrochloride via the formulation of instantly-dissolving buccal films with acidic pH modifier and hydrophilic cyclodextrin: Factorial analysis, in vitro and in vivo assessment. J. Adv. Res. 2020, 24, 281–290. [Google Scholar] [CrossRef]
- Tejada, G.; Lamas, M.; Svetaz, L.; Salomón, C.; Alvarez, V.; Leonardi, D. Effect of drug incorporation technique and polymer combination on the performance of biopolymeric antifungal buccal films. Int. J. Pharm. 2018, 548, 431–442. [Google Scholar] [CrossRef]
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar]
- Bourtoom, T.; Chinnan, M.S. Preparation and properties of rice starch–chitosan blend biodegradable film. LWT—Food Sci. Technol. 2008, 41, 1633–1641. [Google Scholar] [CrossRef]
- Forssell, P.; Lahtinen, R.; Lahelin, M.; Myllärinen, P. Oxygen permeability of amylose and amylopectin films. Carbohydr. Polym. 2002, 47, 125–129. [Google Scholar] [CrossRef]
- Woggum, T.; Sirivongpaisal, P.; Wittaya, T. Characteristics and properties of hydroxypropylated rice starch based biodegradable films. Food Hydrocoll. 2015, 50, 54–64. [Google Scholar] [CrossRef]
- Krochta, J.M.; Baldwin, E.A.; Nisperos-Carriedo, M.O. Edible Coatings and Films to Improve Food Quality; Technomic Publishing Co.: Lancaster, PA, USA, 1994. [Google Scholar]
- Vieira, M.G.A.; da Silva, M.A.; dos Santos, L.O.; Beppu, M.M. Natural-based plasticizers and biopolymer films: A review. Eur. Polym. J. 2011, 47, 254–263. [Google Scholar] [CrossRef]
- Gontard, N.; Guilbert, S.; CUQ, J.L. Water and glycerol as plasticizers affect mechanical and water vapor barrier properties of an edible wheat gluten film. J. Food Sci. 1993, 58, 206–211. [Google Scholar] [CrossRef]
- Sobral, P.d.A.; Menegalli, F.; Hubinger, M.; Roques, M. Mechanical, water vapor barrier and thermal properties of gelatin based edible films. Food Hydrocoll. 2001, 15, 423–432. [Google Scholar] [CrossRef]
- Baldwin, E.; Baker, R. Use of proteins in edible coatings for whole and minimally processed fruits and vegetables. In Protein-Based Films and Coatings; CRC Press: Boca Raton, FL, USA, 2002; pp. 501–515. [Google Scholar]
- Bertuzzi, M.A.; Vidaurre, E.C.; Armada, M.; Gottifredi, J. Water vapor permeability of edible starch based films. J. Food Eng. 2007, 80, 972–978. [Google Scholar] [CrossRef]
- Abdorreza, M.N.; Cheng, L.; Karim, A. Effects of plasticizers on thermal properties and heat sealability of sago starch films. Food Hydrocoll. 2011, 25, 56–60. [Google Scholar] [CrossRef]
- Kusumaningtyas, R.D.; Putri, R.D.; Badriah, N.; Faizah, F.E. Preparation and characterization of edible film from sorghum starch with glycerol and sorbitol as plasticizers. J. Eng. Sci. Technol. 2018, 13, 47–55. [Google Scholar]
- Shi, R.; Li, B. Preparation and characterization of corn starch and lignosulfonate blend film with a high content of lignosulfonate. BioResources 2016, 11, 8860–8874. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, J. Crystallization of high-amylose starch by the addition of plasticizers at low and intermediate concentrations. J. Food Sci. 2010, 75, N8–N16. [Google Scholar] [CrossRef]
- Gaudin, S.; Lourdin, D.; Le Botlan, D.; Ilari, J.; Forssell, P.; Colonna, P. Effect of polymer-plasticizer interactions on the oxygen permeability of starch-sorbitol-water films. In Macromolecular Symposia; WILEY—VCH Verlag: Weinheim, Germany, 1999; Volume 138, pp. 245–248. [Google Scholar]
- Sanyang, M.; Sapuan, S.; Jawaid, M.; Ishak, M.; Sahari, J. Plasticizing and anti-plasticizing effect of different plasticizers on tensile properties of sugar palm starch films. In Proceedings of the Postgraduate Symposium on Biocomposite Technology, Selangor, Malaysia, 3 March 2015. [Google Scholar]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Effect of plasticizer type and concentration on tensile, thermal and barrier properties of biodegradable films based on sugar palm (Arenga pinnata) starch. Polymers 2015, 7, 1106–1124. [Google Scholar] [CrossRef]
- Siepmann, F.; Le Brun, V.; Siepmann, J. Drugs acting as plasticizers in polymeric systems: A quantitative treatment. J. Control Release 2006, 115, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Karim, A.A.; Seow, C. Interactive plasticizing–antiplasticizing effects of water and glycerol on the tensile properties of tapioca starch films. Food Hydrocoll. 2006, 20, 1–8. [Google Scholar] [CrossRef]
- Sanyang, M.; Sapuan, S.; Jawaid, M.; Ishak, M.; Sahari, J. Effect of glycerol and sorbitol plasticizers on physical and thermal properties of sugar palm starch based films. In Proceedings of the 13th International Conference on Environment, Ecosystems and Development (EED ’15), Kuala Lumpur, Malaysia, 23–25 April 2015; pp. 157–162. [Google Scholar]
- Muscat, D.; Adhikari, B.; Adhikari, R.; Chaudhary, D. Comparative study of film forming behaviour of low and high amylose starches using glycerol and xylitol as plasticizers. J. Food Eng. 2012, 109, 189–201. [Google Scholar] [CrossRef]
- Lachman, L.; Drubulis, A. Factors Influencing the Properties of Films Used for Tablet Coating I.: Effects of Plasticizers on the Water Vapor Transmission of Cellulose Acetate Phthalate Films. J. Pharm. Sci. 1964, 53, 639–643. [Google Scholar] [CrossRef]
- Guo, J.-H. Effects of Plasticizers on Water Permeation and Mechanical Properties of Cellulose Acetate: Antiplasticization in Slightly Plasticized Polymer Film. Drug Dev. Ind. Pharm. 1993, 19, 1541–1555. [Google Scholar] [CrossRef]
- Chamarthy, S.P.; Pinal, R. Plasticizer concentration and the performance of a diffusion-controlled polymeric drug delivery system. Colloids Surf. A Physicochem. Eng. Asp. 2008, 331, 25–30. [Google Scholar] [CrossRef]
- Jennings, C.L.; Dziubla, T.D.; Puleo, D.A. Combined Effects of Drugs and Plasticizers on the Properties of Drug Delivery Films. J. Bioact. Compat. Polym. 2016, 31, 323–333. [Google Scholar] [CrossRef]
- Jackson, W.J., Jr.; Caldwell, J.R. Antiplasticization. II. Characteristics of antiplasticizers. J. Appl. Polym. Sci. 1967, 11, 211–226. [Google Scholar] [CrossRef]
- Chan, S.Y.; Goh, C.F.; Lau, J.Y.; Tiew, Y.C.; Balakrishnan, T. Rice starch thin films as a potential buccal delivery system: Effect of plasticiser and drug loading on drug release profile. Int. J. Pharm. 2019, 562, 203–211. [Google Scholar] [CrossRef]
- Fitch-Vargas, P.R.; Aguilar-Palazuelos, E.; Zazueta-Morales, J.d.J.; Vega-García, M.O.; Valdez-Morales, J.E.; Martínez-Bustos, F.; Jacobo-Valenzuela, N. Physicochemical and microstructural characterization of corn starch edible films obtained by a combination of extrusion technology and casting technique. J. Food Sci. 2016, 81, E2224–E2232. [Google Scholar] [CrossRef] [PubMed]
- Talja, R.A.; Helén, H.; Roos, Y.H.; Jouppila, K. Effect of various polyols and polyol contents on physical and mechanical properties of potato starch-based films. Carbohydr. Polym. 2007, 67, 288–295. [Google Scholar] [CrossRef]
- Hassan, L.; Muhammad, A.; Aliyu, R.; Idris, Z.; Izuagie, T.; Umar, K.; Sani, N. Extraction and characterisation of starches from four varieties of Mangifera indica seeds. IOSR J. Appl. Chem. 2013, 3, 16–23. [Google Scholar]
- Peleg, M. An empirical model for the description of moisture sorption curves. J. Food Sci. 1988, 53, 1216–1217. [Google Scholar] [CrossRef]
- Mali, S.; Sakanaka, L.S.; Yamashita, F.; Grossmann, M. Water sorption and mechanical properties of cassava starch films and their relation to plasticizing effect. Carbohydr. Polym. 2005, 60, 283–289. [Google Scholar] [CrossRef]
- Radebaugh, G.W.; Murtha, J.L.; Julian, T.N.; Bondi, J.N. Methods for evaluating the puncture and shear properties of pharmaceutical polymeric films. Int. J. Pharm. 1988, 45, 39–46. [Google Scholar]
- Laohakunjit, N.; Noomhorm, A. Effect of plasticizers on mechanical and barrier properties of rice starch film. Starch-Stärke 2004, 56, 348–356. [Google Scholar] [CrossRef]
- Razavi, S.M.A.; Amini, A.M.; Zahedi, Y. Characterisation of a new biodegradable edible film based on sage seed gum: Influence of plasticiser type and concentration. Food Hydrocoll. 2015, 43, 290–298. [Google Scholar] [CrossRef]
- Sanyang, M.; Sapuan, S.; Jawaid, M.; Ishak, M.; Sahari, J. Effect of plasticizer type and concentration on physical properties of biodegradable films based on sugar palm (Arenga pinnata) starch for food packaging. J. Food Sci. Technol. 2016, 53, 326–336. [Google Scholar] [CrossRef]
- Ghasemlou, M.; Khodaiyan, F.; Oromiehie, A. Physical, mechanical, barrier, and thermal properties of polyol-plasticized biodegradable edible film made from kefiran. Carbohydr. Polym. 2011, 84, 477–483. [Google Scholar] [CrossRef]
- Aguirre, A.; Borneo, R.; León, A.E. Properties of triticale protein films and their relation to plasticizing–antiplasticizing effects of glycerol and sorbitol. Ind. Crop. Prod. 2013, 50, 297–303. [Google Scholar] [CrossRef]
- Etman, M.A.; Naggar, V.F. Thermodynamics of paracetamol solubility in sugar-water cosolvent systems. Int. J. Pharm. 1990, 58, 177–184. [Google Scholar] [CrossRef]
- Turhan, M.; Sayar, S.; Gunasekaran, S. Application of Peleg model to study water absorption in chickpea during soaking. J. Food Eng. 2002, 53, 153–159. [Google Scholar] [CrossRef]
- Preis, M.; Knop, K.; Breitkreutz, J. Mechanical strength test for orodispersible and buccal films. Int. J. Pharm. 2014, 461, 22–29. [Google Scholar] [CrossRef]
- Park, H.-R.; Chough, S.-H.; Yun, Y.-H.; Yoon, S.-D. Properties of Starch/PVA Blend Films Containing Citric Acid as Additive. J. Polym. Environ. 2005, 13, 375–382. [Google Scholar] [CrossRef]
- Suppakul, P.; Chalernsook, B.; Ratisuthawat, B.; Prapasitthi, S.; Munchukangwan, N. Empirical modeling of moisture sorption characteristics and mechanical and barrier properties of cassava flour film and their relation to plasticizing–antiplasticizing effects. LWT—Food Sci. Technol. 2013, 50, 290–297. [Google Scholar] [CrossRef]
- Vicentini, N.M.; Dupuy, N.; Leitzelman, M.; Cereda, M.P.; Sobral, P.J.A. Prediction of Cassava Starch Edible Film Properties by Chemometric Analysis of Infrared Spectra. Spectrosc. Lett. 2005, 38, 749–767. [Google Scholar] [CrossRef]
- De Castro, E.d.S.; Cassella, R.J. Direct determination of sorbitol and sodium glutamate by attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) in the thermostabilizer employed in the production of yellow-fever vaccine. Talanta 2016, 152, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.; Yamashita, F.; Laurindo, J.B. Evaluation of the effects of glycerol and sorbitol concentration and water activity on the water barrier properties of cassava starch films through a solubility approach. Carbohydr. Polym. 2008, 72, 82–87. [Google Scholar]
- Esmaeili, M.; Pircheraghi, G.; Bagheri, R. Optimizing the mechanical and physical properties of thermoplastic starch via tuning the molecular microstructure through co-plasticization by sorbitol and glycerol. Polym. Int. 2017, 66, 809–819. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, H.; Kang, S.; Xia, L.; Jiang, S.; Chen, M.; Jiang, S. An active packaging film based on yam starch with eugenol and its application for pork preservation. Food Hydrocoll. 2019, 96, 546–554. [Google Scholar] [CrossRef]
- Napierała, D.M.; Nowotarska, A. Water vapour transmission properties of wheat starch-sorbitol film. Acta Agrophysica 2006, 7, 151–159. [Google Scholar]
- Gałat, A. Study of the Raman scattering and infrared absorption spectra of branched polysaccharides. Acta Biochim. Pol. 1980, 27, 135–142. [Google Scholar]
- Sibik, J.; Sargent, M.J.; Franklin, M.; Zeitler, J.A. Crystallization and phase changes in paracetamol from the amorphous solid to the liquid phase. Mol. Pharm. 2014, 11, 1326–1334. [Google Scholar] [CrossRef]



| Formulation | Film Thickness (mm) | Water Content (%) | Drug Loding Efficiency (%) |
|---|---|---|---|
| CS10 | 0.090 ± 0.014 | 0.98 ± 0.14 | - |
| CS20 | 0.093 ± 0.019 | 0.64 ± 0.08 | - |
| CS30 | 0.100 ± 0.014 | 1.17 ± 0.33 | - |
| S10P1 | 0.080 ± 0.014 | 0.51 ± 0.36 | 81.9 ± 5.0 |
| S20P1 | 0.086 ± 0.030 | 0.89 ± 0.43 | 93.2 ± 10.6 |
| S30P1 | 0.089 ± 0.017 | 0.42 ± 0.08 | 93.2 ± 15.8 |
| S10P2 | 0.077 ± 0.019 | 0.77 ± 0.15 | 88.4 ± 29.4 |
| S20P2 | 0.088 ± 0.029 | 0.74 ± 0.02 | 71.7 ± 15.1 |
| S30P2 | 0.087 ± 0.020 | 0.68 ± 0.49 | 67.2 ± 21.1 |
| Formulation | k1 (h g−1) | k2 (g−1) | R2 |
|---|---|---|---|
| CS10 | 0.5788 | 2.5004 | 0.9972 |
| CS20 | 1.7085 | 3.8319 | 0.9700 |
| CS30 | 2.6387 | 8.0610 | 0.9134 |
| S10P1 | 2.1822 | 3.3204 | 0.9267 |
| S20P1 | 2.7793 | 3.6524 | 0.9721 |
| S30P1 | 9.1021 | 5.1583 | 0.9673 |
| S10P2 | 5.8184 | 5.6222 | 0.9923 |
| S20P2 | 2.2861 | 4.2975 | 0.9789 |
| S30P2 | 0.9130 | 3.1703 | 0.9475 |
| Formulation | Puncture Strength, PS (N/mm2) | Elongation to Break, EB (%) | Energy to Puncture, EP (N/mm2) |
|---|---|---|---|
| CS10 | 0.43 ± 0.10 | 1.55 ± 0.54 | 0.84 ± 0.26 |
| CS20 | 1.04 ± 0.40 | 4.62 ± 1.13 | 4.43 ± 2.65 |
| CS30 | 1.62 ± 0.40 | 9.93 ± 1.14 | 11.14 ± 3.05 |
| S10P1 | 0.24 ± 0.10 | 1.05 ± 0.14 | 0.49 ± 0.24 |
| S20P1 | 1.01 ± 0.51 | 7.46 ± 1.28 | 11.88 ± 3.98 |
| S30P1 | 1.41 ± 0.04 | 19.04 ± 3.10 | 19.27 ± 2.25 |
| S10P2 | 0.28 ± 0.06 | 1.04 ± 0.22 | 0.61 ± 0.18 |
| S20P2 | 1.40 ± 0.39 | 7.44 ± 0.46 | 8.48 ± 2.67 |
| S30P2 | 1.60 ± 0.67 | 14.63 ± 5.06 | 15.40 ± 3.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alrimawi, B.H.; Chan, M.Y.; Ooi, X.Y.; Chan, S.-Y.; Goh, C.F. The Interplay between Drug and Sorbitol Contents Determines the Mechanical and Swelling Properties of Potential Rice Starch Films for Buccal Drug Delivery. Polymers 2021, 13, 578. https://doi.org/10.3390/polym13040578
Alrimawi BH, Chan MY, Ooi XY, Chan S-Y, Goh CF. The Interplay between Drug and Sorbitol Contents Determines the Mechanical and Swelling Properties of Potential Rice Starch Films for Buccal Drug Delivery. Polymers. 2021; 13(4):578. https://doi.org/10.3390/polym13040578
Chicago/Turabian StyleAlrimawi, Bilal Harieth, May Yee Chan, Xin Yue Ooi, Siok-Yee Chan, and Choon Fu Goh. 2021. "The Interplay between Drug and Sorbitol Contents Determines the Mechanical and Swelling Properties of Potential Rice Starch Films for Buccal Drug Delivery" Polymers 13, no. 4: 578. https://doi.org/10.3390/polym13040578
APA StyleAlrimawi, B. H., Chan, M. Y., Ooi, X. Y., Chan, S.-Y., & Goh, C. F. (2021). The Interplay between Drug and Sorbitol Contents Determines the Mechanical and Swelling Properties of Potential Rice Starch Films for Buccal Drug Delivery. Polymers, 13(4), 578. https://doi.org/10.3390/polym13040578









