Advantages and Disadvantages of Bioplastics Production from Starch and Lignocellulosic Components
Abstract
1. Introduction
2. Starch-Based Bioplastics
2.1. Characteristics and Structure of Starch Grain
2.2. Formation of Filmogenic Starch Solution
2.3. Properties and Characteristics of Starch-Based Bioplastics
2.3.1. Properties and Characteristics of Starch-Based Bioplastics Chemical Modified
2.3.2. Chemical Starch Derivatization Impact on Biodegradation
3. Lignocellulose and Biomass
3.1. Characteristics of Cellulose
3.2. Bioplastics with Cellulose
3.2.1. Properties and Characteristics of Cellulose-Based Bioplastics Chemical Modified
3.2.2. Impact of Chemical Cellulose Derivatization on Biodegradation
3.3. Hemicellulose
Bioplastics with Xylan
4. Extraction of Starch and Lignocellulosic Components (a Challenge)
5. Environmental Impact of Polysaccharide-Based Bioplastics from Plant Biomass
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kaza, S.; Yao, L.C.; Bhada-Tata, P.; Van Woerden, F. What a Waste 2.0: A Global Snapshot of Solid Waste Management to 2050, Urban Development Series. In World Bank: Urban Development Series; World Bank: Washington, DC, USA, 2018. [Google Scholar]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, 25–29. [Google Scholar] [CrossRef]
- Tharanathan, R.N. Biodegradable films and composite coatings: Past, present and future. Trends Food Sci. Technol. 2003, 14, 71–78. [Google Scholar] [CrossRef]
- Hopewell, J.; Dvorak, R.; Kosior, E. Plastics recycling: Challenges and opportunities. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2115–2126. [Google Scholar] [CrossRef]
- Lackner, M. Bioplastics-biobased plastics as renewable and/or biodegradable alternatives to petroplastics. Kirk-Othmer Encycl. Chem. Technol. 2015, 6, 1–41. [Google Scholar] [CrossRef]
- Li, W.C.; Tse, H.F.; Fok, L. Plastic waste in the marine environment: A review of sources, occurrence and effects. Sci. Total Environ. 2016, 566–567, 333–349. [Google Scholar] [CrossRef] [PubMed]
- Paula, F.C.; Paula, C.B.C.; Contiero, J. Prospective Biodegradable Plastics from Biomass Conversion Processes. Biofuels State Dev. 2018. [Google Scholar] [CrossRef]
- Queiroz, A.U.B.; Collares-Queiroz, F.P. Innovation and industrial trends in bioplastics. Polym. Rev. 2009, 49, 65–78. [Google Scholar] [CrossRef]
- Guilbert, S.; Gontard, N.; Cuq, B. Technology and applications of edible protective films. Packag. Technol. Sci. 1995, 8, 339–346. [Google Scholar] [CrossRef]
- Peelman, N.; Ragaert, P.; De Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; Van Impe, F.; Devlieghere, F. Review: Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef]
- Liu, L. Bioplastics in Food Packaging: Innovative Technologies for Biodegradable Packaging. 2006. Available online: http://www.iopp.org/files/public/SanJoseLiuCompetitionFeb06.pdf (accessed on 10 October 2012).
- Gáspár, M.; Benko, Z.; Dogossy, G.; Réczey, K.; Czigány, T. Reducing water absorption in compostable starch-based plastics. Polym. Degrad. Stab. 2005, 90, 563–569. [Google Scholar] [CrossRef]
- Cabedo, L.; Feijoo, J.L.; Villanueva, M.P.; Lagarón, J.M.; Giménez, E. Optimization of biodegradable nanocomposites based on a PLA/PCL blends for food packaging applications. Macromol. Symp. 2006, 233, 191–197. [Google Scholar] [CrossRef]
- Joshi, S. Can nanotechnology improve the sustainability of biobased products? The case of layered silicate biopolymer nanocomposites. J. Ind. Ecol. 2008, 12, 474–489. [Google Scholar] [CrossRef]
- Cyras, V.P.; Soledad, C.M.; Analía, V. Biocomposites based on renewable resource: Acetylated and non acetylated cellulose cardboard coated with polyhydroxybutyrate. Polymer 2009, 50, 6274–6280. [Google Scholar] [CrossRef]
- Shen, L.; Haufe, J.; Patel, M.K. Product Overview and Market Projection of Emerging Bio-Based Plastics PRO-BIP 2009; European Polysaccharide Network of Excellence (EPNOE) and European Bioplastics: Utrecht, The Netherlands, 2009. [Google Scholar]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-Lactic Acid: Production, applications, nanocomposites, and release studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.O.; Laurindo, J.B.; Yamashita, F. Effect of nanoclay incorporation method on mechanical and water vapor barrier properties of starch-based films. Ind. Crop. Prod. 2011, 33, 605–610. [Google Scholar] [CrossRef]
- Yu, J.; Yang, J.; Liu, B.; Ma, X. Preparation and characterization of glycerol plasticized-pea starch/ZnO-carboxymethylcellulose sodium nanocomposites. Bioresour. Technol. 2009, 100, 2832–2841. [Google Scholar] [CrossRef]
- Naik, S.N.; Goud, V.V.; Rout, P.K.; Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renewable and Sustainable. Energy Rev. 2010, 14, 578–597. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report. In Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team]; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Neuling, U.; Kaltschmitt, M. Review of biofuel production—Feedstocks, processess and market. J. Oil Palm Res. 2017, 29, 137–167. [Google Scholar] [CrossRef]
- Sharma, A.; Satyanarayana, T. Microbial acid-stable α-amylases: Characteristics, genetic engineering and applications. Process Biochem. 2013, 48, 201–211. [Google Scholar] [CrossRef]
- Mathlouthi, M. Emballage Et Conservation Des Produits Alimentaire; Polytechnica: Paris, France, 1996. [Google Scholar]
- Coma, V. Polysaccharide-based Biomaterials with Antimicrobial and Antioxidant Properties. Polímeros 2013, 23, 287–297. [Google Scholar] [CrossRef]
- Souza, P.M.; Magalhães, P.O. Application of microbial α-amilase in industry—A review. Brasilian J. Microbiol. 2010, 41, 850–861. [Google Scholar] [CrossRef]
- Peng, L.; Zhongdong, L.; Kennedy, J.F. The study of starch nano-unit chain in the gelatinization process. Carbohydr. Polym. 2007, 68, 360–366. [Google Scholar] [CrossRef]
- Halley, P.J.; Truss, R.W.; Markotsis, M.G.; Chaleat, C.; Russo, M.; Sargent, A.L.; Tan, I.; Sopade, P.A. A review of biodegradable thermoplastic starch polymers. Polym. Durab. Radiat. Eff. 2007, 287–300. [Google Scholar] [CrossRef]
- Copeland, L.; Blazek, J.; Salman, J.; Tang, M.C. Form and functionality of starch. Food Hydrocoll. 2009, 23, 1527–1534. [Google Scholar] [CrossRef]
- Jaillais, B.; Ottenhof, M.A.; Farhat, I.A.; Rutledge, D.N. Outer-product analysis (OPA) using PLS regression to study the retrogradation of starch. Vib. Spectrosc. 2006, 40, 10–19. [Google Scholar] [CrossRef]
- Eliasson, A.C. Starch: Physicochemical and functional aspects. Carbohydr. Food. 2016, 10, 479–578. [Google Scholar]
- Averous, L. Biodegradable multiphase systems based on plasticized starch—Part C: Polymers reviews. J. Macromol. Sci. 2004, 44, 231–274. [Google Scholar] [CrossRef]
- Ma, X.; Yu, J. The effects of plasticizers containing amide groups on the properties of thermoplastic starch. Starch/Starke 2004, 56, 545–551. [Google Scholar] [CrossRef]
- Liu, Z. Edible films and coatings from starches. In Innovations in Food Packaging; Han, J.H., Ed.; Elsevier: Amsterdam, the Netherlands, 2005; pp. 318–336. [Google Scholar]
- Müller, C.M.O.; Laurindo, J.B.; Yamashita, F. Effect of cellulose fibers addition on the mechanical properties and water vapor barrier of starch -based films. Food Hydrocoll. 2009, 23, 1328–1333. [Google Scholar] [CrossRef]
- Manoel, A.F.; Claro, P.I.C.; Mattoso, L.H.C.; Marconcini, J.M.; Mantovani, G.L. Thermoplastic Waxy Starch Films Processed by Extrusion and Pressing: Effect of Glycerol and Water Concentration. Mater. Res. 2017, 20, 353–357. [Google Scholar] [CrossRef]
- Oksman, K.; Aitomäki, Y.; Mathew, A.P.; Siqueira, G.; Zhou, Q.; Butylina, S.; Tanpichai, S.; Zhou, X.; Hooshmand, S. Review of the recent developments in cellulose nanocomposite processing. Compos. Part A Appl. Sci. Manuf. 2016, 83, 2–18. [Google Scholar] [CrossRef]
- Hietala, M.; Rollo, P.; Kekäläinen, K.; Oksman, K. Extrusion processing of green biocomposites: Compounding, fibrillation efficiency, and fiber dispersion. J. Appl. Polym. Sci. 2014, 131, 1–9. [Google Scholar] [CrossRef]
- Taheri, H.; Hietala, M.; Oksman, K. One-step twin-screw extrusion process of cellulose fibers and hydroxyethyl cellulose to produce fibrillated cellulose biocomposite. Cellulose 2020, 27, 8105–8119. [Google Scholar] [CrossRef]
- Herrera, N.; Singh, A.A.; Salaberria, A.M.; Labidi, J.; Mathew, A.P.; Oksman, K. Triethyl citrate (TEC) as a dispersing aid in polylactic acid/chitin nanocomposites prepared via liquid-assisted extrusion. Polymers (Basel) 2017, 9, 406. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, P.; Pontes, A.; Henriques, E. Effect of the injection moulding processing conditions on biopolymers final properties. Mater. Sci. Forum 2012, 730–732, 20–25. [Google Scholar] [CrossRef]
- Perez-Puyana, V.; Felix, M.; Romero, A.; Guerrero, A. Effect of the injection moulding processing conditions on the development of pea protein-based bioplastics. J. Appl. Polym. Sci. 2016, 133, 1–9. [Google Scholar] [CrossRef]
- Sleiman, G.E.H.; Colomines, G.; Deterre, R.; Petit, I.; Leroy, E.; Belhabib, S. PHBV crystallization under injection molding conditions: Influence of packing pressure and mold temperature. J. Renew. Mater. 2018, 6, 270–276. [Google Scholar] [CrossRef]
- Gröndahl, M.; Eriksson, L.; Gatenholm, P. Material properties of plasticized hardwood xylans for potential application as oxygen barrier films. Biomacromolecules 2004, 5, 1528–1535. [Google Scholar] [CrossRef]
- Goksu, E.I.; Karamanlioglu, M.; Bakir, U.; Yilmaz, L.; Yilmazer, U. Production and characterization of films from cotton stalk xylan. J. Agric. Food Chem. 2007, 55, 10685–10691. [Google Scholar] [CrossRef]
- Saxena, A.; Elder, T.J.; Pan, S.; Ragauskas, A.J. Novel nanocellulosic xylan composite film. Compos. Part B Eng. 2009, 40, 727–730. [Google Scholar] [CrossRef]
- Peng, X.W.; Ren, J.L.; Zhong, L.X.; Sun, R.C. Nanocomposite films based on xylan rich hemicelluloses and cellulose nanofibers with enhanced mechanical properties. Biomacromolecules 2011, 12, 3321–3329. [Google Scholar] [CrossRef]
- Gontard, N.; Guilbert, S.; Cuq, J.L. Water and glycerol as plasticizers affect mechanical and water vapor barrier properties of an edible wheat gluten film. J. Food Sci. Chic. 1993, 58, 206–211. [Google Scholar] [CrossRef]
- Wang, S.; Ren, J.; Li, W.; Sun, R.; Liu, S. Properties of polyvinyl alcohol/xylan composite films with citric acid. Carbohydr. Polym. 2014, 103, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Daudt, R.M.; Avena-Bustillos, R.J.; Williams, T.; Wood, D.F.; Külkamp-Guerreiro, I.C.; Marczak, L.D.F.; McHugh, T.H. Comparative study on properties of edible films based on pinhão (Araucaria angustifolia) starch and flour. Food Hydrocoll. 2016, 60, 279–287. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; García, M.A.; Martino, M.N.; Zaritzky, N.E. Barrier, mechanical and optical properties of plasticized yam starch films. Carbohydr. Polym. 2004, 56, 129–135. [Google Scholar] [CrossRef]
- Dias, A.B.; Müller, C.M.; Larotonda, F.D.; Laurindo, J.B. Biodegradable films based on rice starch and rice flour. J. Cereal Sci. 2010, 51, 213–219. [Google Scholar] [CrossRef]
- Cuq, B.; Gontard, N.; Cuq, J.L.; Guilbert, S. Selected functional properties of fish myofibrillar protein-based films as affected by hydrophilic plasticizers. J. Agric. Food Chem. 1997, 45, 622–626. [Google Scholar] [CrossRef]
- Ryu, S.Y.; Rhim, J.W.; Roh, H.J.; Kim, S.S. Preparation and physical properties of zein coated high amylose corn starch film. Lebensm. Wiss. Undtechnologie 2002, 35, 680–686. [Google Scholar] [CrossRef]
- Zahedi, Y.; Ghanbarzadeh, B.; Sedaghat, N. Physical properties of edible emulsified films based on pistachio globulin protein and fatty acids. J. Food Eng. 2010, 100, 102–108. [Google Scholar] [CrossRef]
- Ghanbarzadeh, B.; Almasi, H.; Entezami, A.A. Improving the barrier and mechanical properties of corn starch-based edible films: Effect of citric acid and carboxymethyl cellulose. Ind. Crops Prod. 2011, 33, 229–235. [Google Scholar] [CrossRef]
- Falguera, V.; Quintero, P.J.; Jose, J.A.; Munoz, A.; Ibarz, A. Edible films and coatings: Structures, active function and trends in their use. Trends Food Sci. Technol. 2011, 22, 292–303. [Google Scholar] [CrossRef]
- Souza, A.C.; Benze, R.; Ferrao, E.S.; Ditchfield, C.; Coelho, A.C.V.; Tadin, C.C. Cassava starch biodegradable films: Influence of glycerol and clay nanoparticle content on tensile and barrier properties and glass transition temperature. LWT Food Sci. Technol. 2012, 46, 110–117. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Baraniak, B. Effect of candelilla wax on functional properties of biopolymer emulsion films. A comparative study. Food Hydrocoll. 2014, 41, 195–209. [Google Scholar] [CrossRef]
- Dang, M.K.; Yoksan, R. Development of thermoplastic starch blown film by incorporating plasticized chitosan. Carbohydr. Polym. 2015, 115, 575–581. [Google Scholar] [CrossRef]
- Reis, L.C.B.; Batista, R.; Carolina, O.S.; Alves da Silva, J.B.; Martins, A.C.; Nunes, I.L.; Druzian, J.I. Active biocomposites of cassava starch: The effect of yerba mate extract and mango pulp as antioxidant additives on the properties and the stability of a packaged product. Food Bioproduct. Process. 2015, 94, 382–391. [Google Scholar] [CrossRef]
- Shah, U.; Naqash, F.; Gani, A.; Masoodi, F.A. Art and Science behind Modified Starch Edible Films and Coatings: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 568–580. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xie, F.; Yu, L.; Chen, L.; Lin, L. Thermal processing of starch-based polymers. Prog. Polym. Sci. 2009, 34, 1348–1368. [Google Scholar] [CrossRef]
- Xie, F.; Pollet, E.; Halley, J.P.; Averous, L. Starch-based nano-biocomposites. Prog. Polym. Sci. 2013, 38, 1590–1628. [Google Scholar] [CrossRef]
- Sabetzadeh, M.; Bagheri, R.; Masoomi, M. Study on ternary low density polyethylene/linear low density polyethylene/thermoplastic starch blend films. Carbohydr. Polym. 2015, 119, 126–133. [Google Scholar] [CrossRef]
- Khan, B.; Bilal Khan Niazi, M.; Samin, G.; Jahan, Z. Thermoplastic starch: A possible biodegradable food packaging material—A review. J. Food Process Eng. 2017, 40, 12447. [Google Scholar] [CrossRef]
- Singh, N.; Singh, J.; Kaur, L.; Singh Sodhi, N.; Singh Gill, B. Morphological, thermal and rheological properties of starches from different botanical sources. Food Chem. 2003, 81, 219–231. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; García, M.A.; Martino, M.N.; Zaritzky, N.E. Effects of controlled storage on thermal, mechanical and barrier properties of plasticized films from different starch sources. J. Food Eng. 2006, 75, 453–460. [Google Scholar] [CrossRef]
- Hejna, A.; Lenża, J.; Formela, K.; Korol, J. Studies on the Combined Impact of Starch Source and Multiple Processing on Selected Properties of Thermoplastic Starch/Ethylene-Vinyl Acetate Blends. J. Polym. Environ. 2019, 27, 1112–1126. [Google Scholar] [CrossRef]
- Ehivet, F.E.; Min, B.; Park, M.K.; Oh, J.H. Characterization and antimicrobial activity of sweet potato starch-based edible film containing origanum (Thymus capitatus) oil. J. Food Sci. 2011, 76, 178–184. [Google Scholar] [CrossRef]
- Fakhoury, F.M.; Maria Martelli, S.; Canhadas Bertan, L.; Yamashita, Y.; Innocentini Mei, L.H.; Collares Queiroz, F.P. Edible films made from blends of manioc starch and gelatin—Influence of different types of plasticizer and different levels of macromolecules on their properties. LWT Food Sci. Technol. 2012, 49, 149–154. [Google Scholar] [CrossRef]
- Agustin, M.B.; Ahmmad, B.; Alonzo, S.M.M.; Patriana, F.M. Bioplastic based on Starch and Cellulose Nanocrystals from Rice Straw. J. Reinf. Plast. Compos. 2014, 33, 2205–2213. [Google Scholar] [CrossRef]
- Tulamandi, S.; Rangarajan, V.; Rizvi, S.S.H.; Singhal, R.S.; Chattopadhyay, S.K.; Saha, N.C. A biodegradable and edible packaging film based on papaya puree, gelatin, and defatted soy protein. Food Packag. Shelf Life 2016, 10, 60–71. [Google Scholar] [CrossRef]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Development and characterization of sugar palm starch and poly (lactic acid) bilayer films. Carbohydr. Polym. 2016. [Google Scholar] [CrossRef]
- Oluwasina, O.O.; Olaleye, F.K.; Olusegun, S.J.; Oluwasina, O.O.; Mohallem, N.D.S. Influence of oxidized starch on physico mechanical, thermal properties, and atomic force micrographs of cassava starch bioplastic film. Int. J. Biol. Macromol. 2019, 135, 282–293. [Google Scholar] [CrossRef]
- Makishi, G.; Amorim, E.; Gatti, M.; Ditchfield, C.; Branco, I.; Moraes, I. Physical Properties and Water Vapour Permeability of Blends Produced with Gelatin and Modified Maranta Arundinacea Starch. Chem. Eng. Trans. 2017, 57, 1855–1860. [Google Scholar]
- Sobral, P.J.A.; Menegalli, F.C.; Hubinger, M.D.; Roques, M.A. Mechanical, water vapour barrier and thermal properties of gelatin based edible films. Food Hydrocoll. 2001, 15, 423–432. [Google Scholar] [CrossRef]
- Vanin, F.M.; Hirano, M.H.; Carvalho, R.A.; Moraes, I.C.F.; Bittante, A.M.Q.B.; Sobral, P.J.A. Development of active gelatin-based nanocomposite films produced in an automatic spreader. Food Res. Int. 2014, 63, 16–24. [Google Scholar] [CrossRef]
- Sanyang, M.L.; Sapuan, S.; Jawaid, M.; Ishak, M.; Sahari, J. Effect of Plasticizer Type and Concentration on Tensile, Thermal and Barrier Properties of Biodegradable Films Based on Sugar Palm (Arenga pinnata) Starch. Polymers 2015, 7, 1106–1124. [Google Scholar] [CrossRef]
- Gonçalves, S.M.; Gomes Motta, J.F.; Ribeiro, R.S.; Hidalgo Chávez, D.W.; Ramos de Melo, N. Functional and antimicrobial properties of cellulose acetate films incorporated with sweet fennel essential oil and plasticizers. Curr. Res. Food Sci. 2020. [Google Scholar] [CrossRef]
- López, O.V.; Lecot, C.J.; Zaritzky, N.E.; García, M.A. Biodegradable packages development from starch based heat sealable films. J. Food Eng. 2011, 105, 254–263. [Google Scholar] [CrossRef]
- Kim, H.-Y.; Jane, J.; Lamsal, B. Hydroxypropylation improves film properties of high amylose corn starch. Ind. Crop. Prod. 2017, 95, 175–183. [Google Scholar] [CrossRef]
- Tyagi, V.; Bhattacharya, B. Role of plasticizers in bioplastics. MOJ Food Process Technol. 2019, 7, 128–130. [Google Scholar] [CrossRef]
- Arifa, S.; Al-Zaqri, N.; Tahir, A.; Alsalme, A. Synthesis and characterization of starch based bioplatics using varying plant-based ingredients, plasticizers and natural fillers. Saudi J. Biol. Sci. 2021, 28, 1739–1749. [Google Scholar]
- Bourtoom, T. Plasticizer effect on the properties of biodegradable blend film from rice starch-chitosan. Songklanakarin J. Sci. Technol. 2007, 30, 149. [Google Scholar]
- Bertuzzi, M.A.; Castro Vidaurre, E.F.; Armada, M.; Gottifredi, J.C. Water vapor permeability of edible starch based films. J. Food Eng. 2007, 80, 972–978. [Google Scholar] [CrossRef]
- Ashok, A.; Mathew, M.; Rejeesh, C.R. Innovative value chain development of modified starch for a sustainable environment: A review. Int. J. Polym. Sci. Eng. 2016, 2, 20–32. [Google Scholar]
- Abel, O.M.; Ifeanyi, E.G.; Chinelo, A.S.; Agbajor, G.K.; Imanah, J.E. Surface Hydrophobicity of Starch Acetate for Enhanced Bioplastic Properties. J. Mater. Environ. Sci. 2021, 12, 497–510. [Google Scholar]
- Schmidt, V.C.R.; Blanco-PascuaL, N.; Tribuzi, G.; Laurindo, J.B. Effect of the degree of acetylation, plasticizer concentration and relative humidity on cassava starch films properties. Food Sci. Technol. 2019, 39, 491–499. [Google Scholar] [CrossRef]
- Nevoralová, M.; Koutný, M.; Ujčić, A.; Horák, P.; Kredtusová, J.; Šerá, J.; Růžek, L.; Růžková, M.; Krejčíková, S.; Šlouf, M.; et al. Controlled biodegradability of functionalized thermoplastic starch based materials. Polym. Degrad. Stab. 2019, 170. [Google Scholar] [CrossRef]
- Sondari, D.; Falah, F.; Suryaningrum, R.; Sari, F.P.; Septefani, A.A.; Restu, W.K.; Sampora, Y. Biofilm Based on Modified Sago Starch: Preparation and Characterization. Reaktor 2019, 19, 125–130. [Google Scholar] [CrossRef]
- Oluwasina, O.O.; Akinyele, B.P.; Olusegun, S.J.; Oluwasina, O.; Mohallem, N.D.S. Evaluation of the effects of additives on the properties of starch-based bioplastic film. SN Appl. Sci. 2021, 3, 421. [Google Scholar] [CrossRef]
- Woggum, T.; Sirivongpaisal, P.; Wittaya, T. Characteristics and properties of hydroxypropylated rice starch based biodegradable films. Food Hydrocoll. 2015, 50, 54–64. [Google Scholar] [CrossRef]
- Abe, M.M.; Branciforti, M.C.; Brienzo, M. Biodegradation of Hemicellulose-Cellulose-Starch-Based Bioplastics and Microbial Polyesters. Recycling 2021, 6, 22. [Google Scholar] [CrossRef]
- Rivard, C.; Moens, L.; Roberts, K.; Brigham, J.; Kelley, S. Starch esters as biodegradable plastics: Effects of ester group chain length and degree of substitution on anaerobic biodegradation. Enzym. Microb. Technol. 1995, 17, 848–852. [Google Scholar] [CrossRef]
- Šárka, E.; Kruliš, Z.; Kotek, J.; Růžek, L.; Korbářová, A.; Bubník, Z.; Růžková, M. Application of wheat B-starch in biodegradable plastic materials. Czech J. Food Sci. 2011, 29, 232–242. [Google Scholar] [CrossRef]
- Granza, A.G.; Travalini, A.P.; Farias, F.O.; Colman, T.A.D.; Schnitzler, E.; Demiate, I.M. Effects of acetylation and acetylation–hydroxypropylation (dual-modification) on the properties of starch from Carioca bean (Phaseolus vulgaris L.). J. Therm. Anal. Calorim. 2015, 119, 769–777. [Google Scholar] [CrossRef]
- Ochi, S. Development of high strength biodegradable composites using Manila hemp fiber and starch-based biodegradable resin. Compos. Part A Appl. Sci. Manuf. 2006, 37, 1879–1883. [Google Scholar] [CrossRef]
- Stevens, E.S.; Klamczynski, A.; Glenn, G.M. Starch-lignin foams. Express Polym. Lett. 2010, 4, 311–320. [Google Scholar] [CrossRef]
- Yang, J.; Ching, Y.C.; Chuah, C.H. Applications of Lignocellulosic Fibers and Lignin in Bioplastics: A Review. Polymers 2019, 11, 751. [Google Scholar] [CrossRef]
- Yong, C.K.; Ching, Y.C.; Luqman, C.A.; Liou, N.S. Effect of fiber orientation on mechanical properties of kenaf-reinforced polymer composite. BioResources 2015, 10, 2597–2608. [Google Scholar] [CrossRef]
- Yong, C.K.; Ching, Y.C.; Afzan, M.; Lim, Z.K.; Chong, K.E. Mechanical and thermal properties of chemical treated oil palm empty fruit bunches fiber reinforced polyvinyl alcohol composite. J. Biobased Mater. Bioenergy 2015, 9, 231–235. [Google Scholar]
- Panthapulakkal, S.; Zereshkian, A.; Sain, M. Preparation and characterization of wheat straw fibers for reinforcing application in injection molded thermoplastic composites. Bioresour. Technol. 2006, 97, 265–272. [Google Scholar] [CrossRef]
- Ching, Y.C.; Ershad, A.; Luqman, C.A.; Choo, K.W.; Yong, C.K.; Sabariah, J.J.; Chuah, C.H.; Liou, N.S. Rheological properties of cellulose nanocrystal-embedded polymer composites: A review. Cellulose 2016, 23, 1011–1030. [Google Scholar] [CrossRef]
- Singh, S.; Mohanty, A. Wood fiber reinforced bacterial bioplastic composites: Fabrication and performance evaluation. Compos. Sci. Technol. 2007, 67, 1753–1763. [Google Scholar] [CrossRef]
- Wu, Q.; Rabu, J.; Goulin, K.; Sainlaud, C.; Chen, F.; Johansson, E.; Olsson, R.T.; Hedenqvist, M.S. Flexible strength-improved and crack-resistant biocomposites based on plasticised wheat gluten reinforced with a flax-fibre-weave. Compos. Part A Appl. Sci. Manuf. 2017, 94, 61–69. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in Green Polymer Composites from Lignin for Multifunctional Applications: A Review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Bilo, F.; Pandini, S.; Sartore, L.; Depero, L.E.; Gargiulo, G.; Bonassi, A.; Federici, S.; Bontempi, E. A sustainable bioplastic obtained from rice straw. J. Clen. Prod. 2018, 200, 357–368. [Google Scholar] [CrossRef]
- Vavouraki, A.L.; Angelis, E.M.; Komaros, M. Optimization of thermo-chemical hydrolysis of kitchen waste. Waste Manag. 2013, 33, 740–745. [Google Scholar] [CrossRef]
- Pham, T.P.T.; Kaushik, R.; Parshetti, G.K.; Mahmood, R.; Balasubramaniam, R. Food waste-to-energy conversion technologies: Current status and future directions. Waste Manag. 2015, 38, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Hafid, H.S.; Rahman, N.A.A.; Shah, U.K.M.; Baharuddin, A.S.; Ariff, A.B. Feasibility of using kitchen waste as future substrate for bioethanol production. Renew. Sustain. Energy Rev. 2017, 74, 671–686. [Google Scholar] [CrossRef]
- Hofsetz, K.; Silva, M.A. Brazilian sugarcane bagasse: Energy and non-energy consumption. Biomass Bioenergy 2012, 46, 564–573. [Google Scholar] [CrossRef]
- Souza, O.; Federizzi, M.; Coelho, B.; Wagner, T.M.; Wisbeck, E. Biodegradação de resíduos lignocelulósicos gerados na bananicultura e sua valorização para a produção de biogás. Rev. Bras. Eng. Agrícola Ambient. 2010, 14, 438–443. [Google Scholar] [CrossRef][Green Version]
- Shimizu, F.L.; Monteiro, P.Q.; Ghiraldi, P.H.C.; Melati, R.B.; Pagnocca, F.C.; De Souza, W.; Brienzo, M. Acid, alkali and peroxide pretreatments increase the cellulose accessibility and glucose yield of banana pseudostem. Ind. Crop Prod. 2018, 115, 62–68. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Proagnóstico Agropecuário: Análise da Conjuntura Agropecuária Safra 2010/11: Mandioca. 2011. Available online: http://faostat.fao.org/site/567/DesktopDefault.aspx?PageID=567#ancor (accessed on 23 February 2020).
- ABITRIGO (Associação Brasileira da Indústria do Trigo). Evolução Moagem E Consumo de Farinha—2005–2015. 2016. Available online: http://abitrigo.com.br/ (accessed on 21 September 2020).
- Food and Agriculture Organization (FAO). Wastage Footprint: Impacts on Natural Resources. 2013. Available online: http://www.fao.org/3/i3347e/i3347e.pdf (accessed on 11 September 2020).
- Moon, H.C.; Song, S.; Kim, J.C.; Shirai, Y.; Lee, D.H.; Kim, J.K.; Chung, S.O.; Kim, D.H.; Oh, K.K.; Cho, Y.S. Enzymatic hydrolysis of food waste and ethanol fermentation. Int. J. Energy Res. 2009, 33, 164–172. [Google Scholar] [CrossRef]
- Yan, S.; Li, J.; Chen, X.; Wu, J.; Wang, P.; Ye, J.; Yao, J. Enzymatic hydrolysis of food waste and ethanol production from the hydrolysate. Renew Energy 2011, 36, 1259–1265. [Google Scholar] [CrossRef]
- Jin, Y.; Li, Y.; Li, J. Influence of thermal pretreatment on physical and chemical properties of kitchen waste and the efficiency of anaerobic digestion. J. Environ. Manag. 2016, 180, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Farhat, W.; Venditti, R.; Hubbe, M.; Taha, F.; Becquart, A.; Ayoub, A. Review of Water Resistant Hemicellulose-Based Materials: Processing and Applications. ChemSusChem. 2017, 10, 305–323. [Google Scholar] [CrossRef] [PubMed]
- Festucci-Buselli, R.A.; Otoni, W.C.; Joshi, C.P. Structure, organization, and functions of cellulose synthase complexes in higher plants. Braz. J. Plant Physiol. 2007, 19, 1–13. [Google Scholar] [CrossRef]
- Fengel, D.; Wegener, G. Wood: Chemistry, Ultrastructure, Reactions; W. de Gruyter: Berlin, Germany; New York, NY, USA, 1984. [Google Scholar]
- Melati, R.B.; Shimizu, F.L.; Oliveira, G.; Pagnocca, F.C.; Souza, W.; Sant’Anna c Brienzo, M. Key Factors Affecting the Recalcitrance and Conversion Process of Biomass. Bioenerg. Res. 2019, 12, 1–20. [Google Scholar] [CrossRef]
- Schmatz, A.A.; Tyhoda, L.; Brienzo, M. Sugarcane biomass conversion influenced by lignin. Biofuels Bioprod. Biorefining 2019. [Google Scholar] [CrossRef]
- Zhang, Y.H.P.; Lynd, L.R. Toward an aggregated understanding of enzymatic hydrolysis of cellulose: Noncomplexed cellulase systems. Biotechnol. Bioeng. 2004, 88, 797–824. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink HPBohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. 2005, 44, 3358–3393. [Google Scholar] [CrossRef]
- Mesquita, J.P. Nanocristais de celulose para preparação de bionanocompósitos com quitosana e carbonos nanoestruturados para aplicações tecnológicas e ambientais. Ph.D. Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, 2012. [Google Scholar]
- Liao, Y.; De Beeck, B.O.; Thielemans, K.; Ennaert, T.; Snelders, J.; Dusselier, M.; Courtin, C.M.; Sels, B.F. The role of pretreatment in the catalytic valorization of celulose. Mol. Catal. 2020, 487, 110883. [Google Scholar] [CrossRef]
- Fan, G.Z.; Wang, Y.X.; Song, G.S.; Yan, J.T.; Li, J.F. Preparation of microcrystalline cellulose from rice straw under microwave irradiation. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Sun, B.; Zhang, M.; Ni, Y. Use of sulfated cellulose nanocrystals towards stability enhancement of gelatin-encapsulated tea polyphenols. Cellulose 2018, 25, 5157–5173. [Google Scholar] [CrossRef]
- Kuang, Y.; Chen, C.; Pastel, G.; Li, Y.; Song, J.; Mi, R.; Kong, W.; Liu, B.; Jiang, Y.; Yang, K. Conductive Cellulose Nanofiber Enabled Thick Electrode for Compact and Flexible Energy Storage Devices. Adv. Energy Mater. 2018, 8, 1802398. [Google Scholar] [CrossRef]
- Mathew, A.P.; Oksman, K.; Sain, M. The effect of morphology and chemical characteristics of cellulose reinforcements on the crystallinity of polylactic acid. J. Appl. Polym. Sci. 2006, 101, 300–310. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, C.; Anderson, D.P.; Huneault, M.A.; Chang, P.R. Pea starch based composite films with pea hull fibres and pea hull fibre-derived nanowhiskers. Polym. Eng. Sci. 2009. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, C.; Chang, P.R.; Cao, X.; Anderson, D.P. Bionanocomposites based on pea starch and cellulose nanowhiskers hydrolyzed from pea hull fibre: Effect of hydrolysis time. Carbohydr. Polym. 2009, 76, 607–615. [Google Scholar] [CrossRef]
- Xie, F.; Avérous, L.; Halley, P.J.; Liu, P. Mechanical performance of starch-based biocomposites. In Biocomposites; Woodhead Publishing: Sawston, UK, 2015; pp. 53–92. [Google Scholar]
- Kumar, A.P.; Depan, D.; Singh Tomer, N.; Singh, R.P. Nanoscale particles for polymer degradation and stabilization—Trends and future perspectives. Prog. Polym. Sci. 2009, 34, 479–515. [Google Scholar] [CrossRef]
- Lu, Y.; Weng, L.; Cao, X. Morphological, thermal and mechanical properties of ramie crystallites—Reinforced plasticized starch biocomposites. Carbohydr. Polym. 2006, 63, 198–204. [Google Scholar] [CrossRef]
- Woehl, M.A.; Canestraro, C.D.; Mikowski, A.; Sierakowski, M.R.; Ramos, L.P.; Wypych, F. Bionanocomposites of thermoplastic starch reinforced with bacterial cellulose nanofibres: Effect of enzymatic treatment on mechanical properties. Carbohydr. Polym. 2010, 80, 866–873. [Google Scholar] [CrossRef]
- Kaushik, A.; Singh, M.; Verma, G. Green nanocomposites based on thermoplastic starch and steam exploded cellulose nanofibrils from wheat straw. Carbohyd. Polym. 2010, 82, 337–345. [Google Scholar] [CrossRef]
- Gordobil, O.; Egüés, I.; Urruzola, I.; Labidi, J. Xylan–cellulose films: Improvement of hydrophobicity, thermal and mechanical properties. Carbohydr. Polym. 2014, 112, 56–62. [Google Scholar] [CrossRef]
- Hansen, N.M.L.; Blomfeldt, T.O.J.; Hedenqvist, M.S.; Plackett, D. Properties of plasticized composite films prepared from nanofibrillated cellulose and birch wood xylan. Cellulose 2012, 19, 2015–2031. [Google Scholar] [CrossRef]
- Cao, X.; Chen, Y.; Chang, P.R.; Muir, A.D.; Falk, G. Starch-based nanocomposites reinforced with flax cellulose nanocrystals. Express. Polym. Lett. 2008, 2, 502–510. [Google Scholar] [CrossRef]
- Xie, F.; Pollet, E.; Halley, P.J.; Avérous, L. Advanced Nano-biocomposites Based on Starch. Polysacch. Bioactivity Biotechnol. 2014, 1–75. [Google Scholar] [CrossRef]
- Angles, M.N.; Dufresne, A. Plasticized starch/tunicin whiskers nanocomposites. 1. Structural analysis. Macromolecules 2000, 33, 8344–8353. [Google Scholar] [CrossRef]
- Mathew, A.P.; Dufresne, A. Morphological investigation of nanocomposites from sorbitol plasticized starch and tunicin whiskers. Biomacromolecules 2002, 3, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Sreekala, M.S.; Goda, K.; Devi, P.V. Sorption characteristics of water, oil and diesel in cellulose nanofiber reinforced corn starch resin/ramie fabric composites. Compos Interfaces 2008, 15, 281–299. [Google Scholar] [CrossRef]
- Svagan, A.J.; Hedenqvist, M.; Berglund, L. Reduced water vapour sorption in cellulose nanocomposites with starch matrix. Compos. Sci. Technol. 2009, 69, 500–506. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Luo, H.; He, F.; Liang, H.; Huang, Y.; Li, X.L. Mechanical, moisture absorption, and biodegradation behaviours of bacterial cellulose fibre-reinforced starch biocomposites. Compos. Sci. Technol. 2009, 69, 1212–1217. [Google Scholar] [CrossRef]
- Guimarães, I.C.; dos Reis, K.C.; Menezes, E.G.T.; Rodrigues, A.C.; da Silva, T.F.; de Oliveira, I.R.N.; Vilas Boas, E.V.B. Cellulose microfibrillated suspension of carrots obtained by mechanical defibrillation and their application in edible starch films. Ind. Crop. Prod. 2016, 89, 285–294. [Google Scholar] [CrossRef]
- Do Lago, R.C.; de Oliveira, A.L.M.; Cordasso Dias, M.; de Carvalho, E.E.N.; Denzin Tonoli, G.H.; de Barros Vilas Boas, E.V. Obtaining cellulosic nanofibrils from oat straw for biocomposite reinforcement: Mechanical and barrier properties. Ind. Crop. Prod. 2020, 148, 112264. [Google Scholar] [CrossRef]
- Aravamudhan, A.; Ramos, D.M.; Nada, A.A.; Kumbar, S.G. Natural Polymers. Nat. Synth. Biomed. Polym. 2014, 67–89. [Google Scholar] [CrossRef]
- Suriyatem, R.; Noikang, N.; Kankam, T.; Jantanasakulwong, K.; Leksawasdi, N.; Phimolsiripol, Y.; Insomphun, C.; Seesuriyachan, P.; Chaiyaso, T.; Jantrawut, P. Physical Properties of Carboxymethyl Cellulose from Palm Bunch and Bagasse Agricultural Wastes: Effect of Delignification with Hydrogen Peroxide. Polymers 2020, 12, 1505. [Google Scholar] [CrossRef]
- Ma, X.; Chang, P.R.; Yu, J. Properties of biodegradable thermoplastic pea starch/carboxymethyl cellulose and pea starch/microcrystalline cellulose composites. Carbohydr. Polym. 2008, 72, 369–375. [Google Scholar] [CrossRef]
- Ergun, R.; Guo, J.; Huebner-Keese, B. Cellulose. Encycl. Food Health 2016, 694–702. [Google Scholar] [CrossRef]
- Tamara, T.; Sumari Nazriati Arni, S. Properties of cassava starch-based bioplastics and CMC with sorbitol as A plasticizer. IOP Conf. Ser. Earth Environ. Sci. 2020, 456, 012077. [Google Scholar] [CrossRef]
- Tongdeesoontorn, W.; Mauer, L.; Wongruong, S.; Sriburi, P.; Rachtanapun, P. Effect of carboxymethyl cellulose concentration on physical properties of biodegradable cassava starch-based films. Chem. Cent. J. 2011, 5, 1–8. [Google Scholar] [CrossRef]
- Margaretha, L.; Ratnawulan. The effect of addition sorbitol and carboxymethyl cellulose (CMC) on the quality of biodegradable plastics from avocado seed starch. Pillar Phys. 2020, 13, 103–112. [Google Scholar]
- Mugwagwa Lindleen, R.; Chimphango, A.F.A. Enhancing the functional properties of acetylated hemicellulose films for active food packaging using acetylated nanocellulose reinforcement and polycaprolactone coating. Food Packag. Shelf Life 2020, 24, 100481. [Google Scholar] [CrossRef]
- Fei, Z.; Huang, S.; Yin, J.; Xu, F.; Zhang, Y. Preparation and Characterization of Bio-based Degradable Plastic Films Composed of Cellulose Acetate and Starch Acetate. J. Polym. Environ. 2015, 23, 383–391. [Google Scholar] [CrossRef]
- Akkus, M.; Ozkan, N.; Bakir, U. Efficient acetylation of xylans by exploiting the potassium acetate formed during the alkaline extraction. J. Polym. Environ. 2018, 26, 3397–3403. [Google Scholar] [CrossRef]
- Polman, E.M.N.; Gruter, G.J.M.; Parsons, J.R.; Tietema, A. Comparison of the aerobic biodegradation of biopolymers and the corresponding bioplastics: A review. Sci. Total Environ. 2021, 753, 141953. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Lee, H.S.; Lyoo, W.S. Effect of UV irradiation on cellulase degradation of cellulose acetate containing TiO2. Fibers Polym. 2007, 8, 19–24. [Google Scholar] [CrossRef]
- Ishigaki, T.; Kawagoshi, Y.; Ike, M.; Fujita, M. Abundance of polymer degrading microorganisms in seabased solid waste landfill site. J. Basic Microbiol. 2000, 90, 400–405. [Google Scholar]
- Buchanan, C.M.; Gardner, R.M.; Komarek, R.J. Aerobic biodegradation of cellulose acetate. J. Appl. Polym. Sci. 1993, 47, 1709–1719. [Google Scholar] [CrossRef]
- Nelson, M.; McCarthy, S.P.; Gross, R.A. Isolation of a Pseudomonas paucimobilis capable of using insoluble cellulose acetate as a sole carbon source. Proc. ACS Div. Polym. Mat. Sci. Eng. 1993, 67, 139–140. [Google Scholar]
- Gu, J.D.; Eberiel, D.T.; McCarthy, S.P.; Gross, R.A. Cellulose acetate biodegradability upon exposure to simulated aerobic composting and anaerobic bioreactor environments. J. Environ. Polym. Degrad. 1993, 1, 143–153. [Google Scholar] [CrossRef]
- Mostafa, N.A.; Farag, A.A.; Abo-dief Hala, M.; Tayeb, A.M. Production of biodegradable plastic from agricultural wastes. Arab. J. Chem. 2018, 11, 546–553. [Google Scholar] [CrossRef]
- Yaradoddi, J.S.; Banapurmath, N.R.; Ganachari, S.V.; Soudagar, M.E.; Mubarak, N.M.; Hallad, S.; Hugar, S.; Fayaz, H. Biodegradable carboxymethyl cellulose based material for sustainable packaging application. Sci. Rep. 2020, 10, 21960. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Lavoine, N.; Lucia, L.A.; Dou, C. Formulating bioplastic composites for biodegradability, recycling, and performance: A Review. BioResources 2021, 16, 2021–2083. [Google Scholar] [CrossRef]
- Ekici, S. Intelligent poly(N-isopropylacrylamide)-carboxymethyl cellulose full interpenetrating polymeric networks for protein adsorption studies. J. Mater. Sci. 2011, 46, 2843–2850. [Google Scholar] [CrossRef]
- Anwar, Z.; Gulfraz, M.; Irshad, M. Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: A brief review. J. Rad. Res. App. Sci. 2014, 7, 163–173. [Google Scholar] [CrossRef]
- Shahzadi, T.; Mehmood, S.; Irshad, M.; Anwar, Z.; Afroz, A.; Zeeshan, N.; Rashid, U.; Sughra, K. Advances in lignocellulosic biotechnology: A brief review on lignocellulosic biomass and cellulases. Adv. Biosci. Biotechnol. 2014, 5, 246–251. [Google Scholar] [CrossRef]
- Brienzo, M.; Carvalho, A.F.A.; Figueiredo, F.C.; Neto, P.O. Sugarcane Bagasse Hemicellulose Properties, Extraction Technologies and Xylooligosaccharides Production; Riley, G.L., Ed.; Food Waste: Practices, Management and Challenges, Nova Science Publishers: New York, NY, USA, 2016; pp. 155–188. [Google Scholar]
- Freitas, C.; Carmona, E.; Brienzo, M. Xylooligosaccharides production process from lignocellulosic biomass and bioactive effects. Bioact. Carbohydr. Diet. Fibre 2019, 18, 100184. [Google Scholar] [CrossRef]
- Kusema, B.T.; Tönnov, T.; Mäki-Arvela, P.; Salmi, T.; Willför, S.; Holmbom, B.; Murzin, D.Y. Acid hydrolysis of O-acetyl-galactoglucomannan. Cataly. Sci. Technol. 2013, 3, 116–122. [Google Scholar] [CrossRef]
- Ayoub, A.; Venditti, R.A.; Pawlak, J.J.; Sadeghifar, H.; Salam, A. Development of an acetylation reaction of switch grass hemicellulose in ionic liquid without catalyst. Ind. Crop. Prod. 2013, 44, 306–314. [Google Scholar] [CrossRef]
- Kayserilioglu, B.S.; Bakir, U.; Yilamz, L.; Akkas, N. Use of xylan, an agricultural by-product, in wheat gluten based biodegradable films: Mechanical, solubility and water vapor transfer rate properties. Bioresour. Technol. 2003, 87, 239–246. [Google Scholar] [CrossRef]
- Mikkonen, K.S.; Tenkanen, M. Sustainable food-packaging materials based on future biorefinery products: Xylans and mannans. Trends Food Sci. Technol. 2012, 28, 90–102. [Google Scholar] [CrossRef]
- Melati, R.B.; Sass, D.C.; Paggnoca, F.C.; Brienzo, M. Anatomic influence of sugarcane biomass on xylan solubilization. Ind. Crop. Prod. 2021, 164, 113357. [Google Scholar] [CrossRef]
- Alves, R.C.; Melati, R.B.; Casagrande, G.M.S.; Contiero, J.; Pagnocca, F.C.; Brienzo, M. Sieving process selects sugarcane bagasse with lower recalcitrance to xylan solubilization. J. Chem. Technol. Biotechnol. 2020. [Google Scholar] [CrossRef]
- Escalante, A.; Gonçalves, A.; Bodin, A.; Stepan, A.; Sandström, C.; Toriz, G.; Gatenholm, P. Flexible oxygen barrier films from spruce xylan. Carbohyd. Polym. 2012, 87, 2381–2387. [Google Scholar] [CrossRef]
- Chen, G.G.; Qi, X.M.; Guan, Y.; Peng, F.; Yao, C.L.; Sun, R.C. High Strength Hemicellulose-Based Nanocomposite Film for Food Packaging Applications. ACS Sustain. Chem. Eng. 2016, 4, 1985–1993. [Google Scholar] [CrossRef]
- Oliveira, E.E.; Silva, A.E.; Junior, T.N.; Gomes, M.C.S.; Aguiar, L.M.; Marcelino, H.R.; Araujo, I.B.; Bayer, M.P.; Ricardo, N.M.P.S.; Oliveira, A.G.; et al. Xylan from corn cobs, a promising polymer for drug delivery: Production and characterization. Bioresour. Technol. 2010, 101, 5402–5406. [Google Scholar] [CrossRef]
- Fang, J.M.; Sun, R.C.; Tomkinson, J.; Fowler, P. Acetylation of wheat straw hemicellulose B in a new non-aqueous swelling system. Carbohyd. Polym. 2000, 41, 379–387. [Google Scholar] [CrossRef]
- Salam, A.; Pawlak, J.J.; Venditti, R.A.; El-tahlawy, K. Incorporation of carboxyl groups into xylan for improved absorbency. Cellulose 2011, 18, 1033–1041. [Google Scholar] [CrossRef]
- Stevanic, J.S.; Bergström, E.M.; Gatenholm, P.; Berglund, L.; Salmén, L. Arabinoxylan/nanofibrillated cellulose composite films. J. Mater. Sci. 2012, 47, 6724–6732. [Google Scholar] [CrossRef]
- Mikkonen, K.S.; Pitkänen, L.; Liljeström, V.; Mabasa Bergström, E.; Serimaa, R.; Salmén, L.; Tenkanen, M. Arabinoxylan structure affects the reinforcement of films by microfibrillated cellulose. Cellulose 2012, 19, 467–480. [Google Scholar] [CrossRef]
- Mikkonen, K.S.; Stevanic, J.S.; Joly, C.; Dole, P.; Pirkkalainen, K.; Serimaa, R.; Tenkanen, M. Composite films from spruce galactoglucomannans with microfibrillated spruce wood cellulose. Cellulose 2011, 18, 713–726. [Google Scholar] [CrossRef]
- Long, L.; Shen, F.; Wang, F.; Tian, D.; Hu, J. Synthesis, characterization and enzymatic surface roughing of cellulose/xylan composite films. Carbohydr. Polym. 2019. [Google Scholar] [CrossRef] [PubMed]
- Stevanic, J.S.; Joly, C.; Mikkonen, K.S.; Pirkkalainen, K.; Serimaa, R.; Rémond, C.; Toriz, G.; Gatenholm, P.; Tenkanen, M.; Salmén, L. Bacterial nanocellulose-reinforced arabinoxylan films. J. Appl. Polym. Sci. 2011, 122, 1030–1039. [Google Scholar] [CrossRef]
- Allen, W.R. Structural applications for flexible packaging: Innovations in pouch forms and uses. Polym. Plast. Technol. Eng. 1986, 25, 295–320. [Google Scholar] [CrossRef]
- Briston, J.H. Plasticfilms, 3rd ed.; Wiley: New York, NY, USA, 1988. [Google Scholar]
- Brumer, H.; Zhou, Q.; Baumann, M.J.; Carlsson, K.; Teeri, T.T. Activation of crystalline cellulose surfaces through the chemoenzymatic modification of xyloglucan. J. Am. Chem. Soc. 2004, 126, 5715–5721. [Google Scholar] [CrossRef] [PubMed]
- Jean, B.; Heux, L.; Dubreuil, F.; Chambat, G.; Cousin, F. Nonelectrostatic building of biomimetic cellulose-xyloglucan multilayers. Langmuir 2009, 25, 3920–3923. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Ragauskas, A.J. Water transmission barrier properties of biodegradable films based on cellulosic whiskers and xylan. Carbohydr. Polym. 2009, 78, 357–360. [Google Scholar] [CrossRef]
- Moorthy, S.N. Tropical sources of starch. Starch Food. 2004, 321–359. [Google Scholar] [CrossRef]
- Lacerda, L.G.; Colman, T.A.D.; Bauab, T.; da Silva Carvalho Filho, M.A.; Demiate, I.M.; de Vasconcelos, E.C.; Schnitzler, E. Thermal, structural and rheological properties of starch from avocado seeds (Persea americana, Miller) modified with standard sodium hypochlorite solutions. J. Therm. Anal. Calorim. 2014, 115, 1893–1899. [Google Scholar] [CrossRef]
- Zhu, F. Structures, properties, modifications, and uses of oat starch. Food Chem. 2017, 229, 329–340. [Google Scholar] [CrossRef]
- Khlestkin, V.K.; Peltek, S.E.; Kolchanov, N.A. Review of direct chemical and biochemical transformations of starch. Carbohydr. Polym. 2018, 181, 460–476. [Google Scholar] [CrossRef]
- Saengchan, K.; Nopharatana, M.; Lerdlattaporn, R.; Songkasiri, W. Enhancement of starch-pulp separation in centrifugal-filtration process: Effects of particle size and variety of cassava root on free starch granule separation. Food Bioprod. Process. 2015, 95, 208–217. [Google Scholar] [CrossRef]
- Branco, F.P.; Naka, M.H.; Cereda, M.P. Granulometryand Energy Consumption as Indicators of Disintegration Efficiency in a Hammer Mill Adapted to Extracting Arrowroot Starch (MarantaArundinacea) in Comparison to Starch Extraction from Cassava. Eng. Agrícola. 2019, 39, 341–349. [Google Scholar] [CrossRef]
- Díaz, A.; Dini, C.; Viña, S.Z.; García, M.A. Starch extraction process coupled to protein recovery from leguminous tuberous roots (Pachyrhizus ahipa). Carbohydr. Polym. 2016, 152, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.M.; Park, C.S.; Baik, M.Y.; Kim, H.S.; Choi, Y.S.; Choi, H.W.; Seo, D.H. Enzymatic extraction of starch from broken rice using freeze-thaw infusion with food-grade protease. Starch Stärke. 2017, 70, 1700007. [Google Scholar] [CrossRef]
- Lee, H.; Htoon, A.; Paterson, J. Alkaline extraction of starch from Australian lentil cultivars Matilda and Digger optimised for starch yield and starch and protein quality. Food Chem. 2007, 102, 551–559. [Google Scholar] [CrossRef]
- Versino, F.; García, M.A. Cassava (Manihot esculenta) starch films reinforced with natural fibrous filler. Ind. Crop. Prod. 2014, 58, 305–314. [Google Scholar] [CrossRef]
- Lindeboom, N.; Chang, P.R.; Tyler, R.T. Analytical, Biochemical and Physicochemical Aspects of Starch Granule Size, with Emphasis on Small Granule Starches: A Review. Starch—Stärke 2004, 56, 89–99. [Google Scholar] [CrossRef]
- Galanakis, C.M. Recovery of high added-value components from food wastes: Conventional, emerging technologies and commercialized applications. Trends Food Sci. Technol. 2012, 26, 68–87. [Google Scholar] [CrossRef]
- Lara-Flores, A.A.; Araújo, R.G.; Rodríguez-Jasso, R.M.; Aguedo, M.; Aguilar, C.N.; Trajano, H.L.; Ruiz, H.A. Bioeconomy and Biorefinery: Valorization of Hemicellulose from Lignocellulosic Biomass and Potential Use of Avocado Residues as a Promising Resource of Bioproducts. In Energy, Environment, and Sustainability; Springer: Singapore, 2017; pp. 141–170. [Google Scholar] [CrossRef]
- Araújo, R.G.; Rodríguez-Jasso, R.M.; Ruiz, H.A.; Govea-Salas, M.; Rosas-Flores, W.; Aguilar-González, M.A.; Pintado, M.E.; Lopez-Badillo, C.; Luevanos, C.; Aguilar, C.N. Hydrothermal–Microwave Processing for Starch Extraction from Mexican Avocado Seeds: Operational Conditions and Characterization. Processes 2020, 8, 759. [Google Scholar] [CrossRef]
- Trache, D.; Hussin, M.H.; Haafiz, M.K.M.; Thakur, V.K. Recent progress in cellulose nanocrystals: Sources and production. Nanoscale 2017, 9, 1763–1786. [Google Scholar] [CrossRef] [PubMed]
- Trache, D.; Hussin, M.H.; Hui Chuin, C.T.; Sabar, S.; Fazita, M.R.N.; Taiwo, O.F.A.; Hassan, T.M.; Haafiz, M.K.M. Microcrystalline cellulose: Isolation, characterization and bio-composites application—A review. Int. J. Biol. Macromol. 2016, 93, 789–804. [Google Scholar] [CrossRef]
- Majeed, K.; Jawaid, M.; Hassan, A.; Abu Bakar, A.; Abdul Khalil, H.P.S.; Salema, A.A.; Inuwa, I. Potential materials for food packaging from nanoclay/natural fibres filled hybrid composites. Mater. Des. 2013, 46, 391–410. [Google Scholar] [CrossRef]
- Zulham Efendi Sinaga, M.; Gea, S.; Panindia, N.; Alfinsyah Sihombing, Y. The Preparation of All-Cellulose Nanocomposite Film from Isolated Cellulose of Corncobs as Food Packaging. Orient. J. Chem. 2018, 34, 562–567. [Google Scholar] [CrossRef]
- Wei, X.; Zhang, L.; Wang, J.; Li, J.; Zhou, W. Preparation of cellulose film in ionic liquid by high shearing and application in pineapple preservation. Mater. Res. Express 2020, 7, 025313. [Google Scholar] [CrossRef]
- Ferreira, F.V.; Mariano, M.; Rabelo, S.C.; Gouveia, R.F.; Lona, L.M.F. Isolation and surface modification of cellulose nanocrystals from sugarcane bagasse waste: From a micro- to a nano-scale view. Appl. Surf. Sci. 2018, 436, 1113–1122. [Google Scholar] [CrossRef]
- Robles, E.; Fernández-Rodríguez, J.; Barbosa, A.M.; Gordobil, O.; Carreño, N.L.V.; Labidi, J. Production of cellulose nanoparticles from blue agave waste treated with environmentally friendly processes. Carbohydr. Polym. 2018, 183, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Mokhena, T.C.; John, M.J. Cellulose nanomaterials: New generation materials for solving global issues. Cellulose 2019, 27, 1149–1194. [Google Scholar] [CrossRef]
- Kaur, D.; Bhardwaj, N.K.; Lohchab, R.K. Environmental Aspect of Using Chlorine Dioxide to Improve Effluent and Pulp Quality During Wheat Straw Bleaching. Waste Biomass Valorization 2018, 10, 1231–1239. [Google Scholar] [CrossRef]
- Sartori, J.; Potthast, A.; Ecker, A.; Sixta, H.; Rosenau, T.; Kosma, P. Alkaline degradation kinetics and CE-separation of cello- and xylooligomers. Part I. Carbohydr. Res. 2003, 338, 1209–1216. [Google Scholar] [CrossRef]
- Jackson, M.G. Review article: The alkali treatment of straws. Anim. Feed Sci. Technol. 1977, 2, 105–130. [Google Scholar] [CrossRef]
- Spencer, R.R.; Akin, D.E. Rumen Microbial Degradation of Potassium Hydroxide-treated Coastal Bermudagrass Leaf Blades Examined by Electron Microscopy. J. Anim. Sci. 1980, 51, 1189–1196. [Google Scholar] [CrossRef]
- Mesa, L.; González, E.; Cara, C.; González, M.; Castro, E.; Mussatto, S.I. The effect of organosolv pretreatment variables on enzymatic hydrolysis of sugarcane bagasse. Chem. Eng. J. 2011, 168, 1157–1162. [Google Scholar] [CrossRef]
- Guragain, Y.N.; Bastola, K.P.; Madl, R.L.; Vadlani, P.V. Novel Biomass Pretreatment Using Alkaline Organic Solvents: A Green Approach for Biomass Fractionation and 2,3-Butanediol Production. BioEnergy Res. 2016, 9, 643–655. [Google Scholar] [CrossRef]
- Laure, S.; Leschinsky, M.; Fröhling, M.; Schultmann, F.; Unkelbach, G. Assessment of an organosolv lignocellulose biorefinery concept based on a material flow analysis of a pilot plant. Cell Chem. Technol. 2014, 48, 793–798. [Google Scholar]
- Escarnot, E.; Aguedo, M.; Paquot, M. Enzymatic hydrolysis of arabinoxylans from spelt bran and hull. J. Cereal Sci. 2012, 55, 243–253. [Google Scholar] [CrossRef]
- Souza, R.E.; Gomes, F.J.B.; Brito, E.O.; Costa Lelis, R.C. A review on lignin sources and uses. J. Appl. Biotechnol. Bioeng. 2020, 7, 100–105. [Google Scholar] [CrossRef]
- Jiang, Z.; Hu, C. Selective extraction and conversion of lignin in actual biomass to monophenols: A review. J. Energy Chem. 2016, 25, 947–956. [Google Scholar] [CrossRef]
- Rashid, T.; Kait, C.F.; Regupathi, I.; Murugesan, T. Dissolution of kraft lignin using Protic Ionic Liquids and characterization. Ind. Crop. Prod. 2016, 84, 284–293. [Google Scholar] [CrossRef]
- Wang, G.; Chen, H. Enhanced lignin extraction process from steam exploded corn stalk. Separation and Purification. Technology. 2016, 157, 93–101. [Google Scholar] [CrossRef]
- Ferrer, A.; Vega, A.; Rodríguez, A.; Jiménez, L. Acetosolv pulping for the fractionation of empty fruit bunches from palm oil industry. Bioresour. Technol. 2013, 132, 115–120. [Google Scholar] [CrossRef]
- Gunasekaran, V.; Ramesh, S.; Sathiasivan, K.; Shankar, M.; Rajesh, M.; Tamilarasan, K. Simultaneous organosolv pretreatment and detoxification of agro-biomass for efficient lignin extraction and characterization. Chem. Pap. 2019, 74, 273–283. [Google Scholar] [CrossRef]
- Korol, J.; Hejna, A.; Burchart-Korol, D.; Wachowicz, J. Comparative Analysis of Carbon, Ecological, and Water Footprints of Polypropylene-Based Composites Filled with Cotton, Jute and Kenaf Fibers. Materials 2020, 13, 3541. [Google Scholar] [CrossRef] [PubMed]
- Korol, J.; Hejna, A.; Burchart-Korol, D.; Chmielnicki, B.; Wypiór, K. Water Footprint Assessment of Selected Polymers, Polymer Blends, Composites, and Biocomposites for Industrial Application. Polymers 2019, 11, 1791. [Google Scholar] [CrossRef]
- Broeren, M.L.M.; Kuling, L.; Worrell, E.; Shen, L. Environmental impact assessment of six starch plastics focusing on wastewater-derived starch and additives. Resour. Conserv. Recycl. 2017, 127, 246–255. [Google Scholar] [CrossRef]
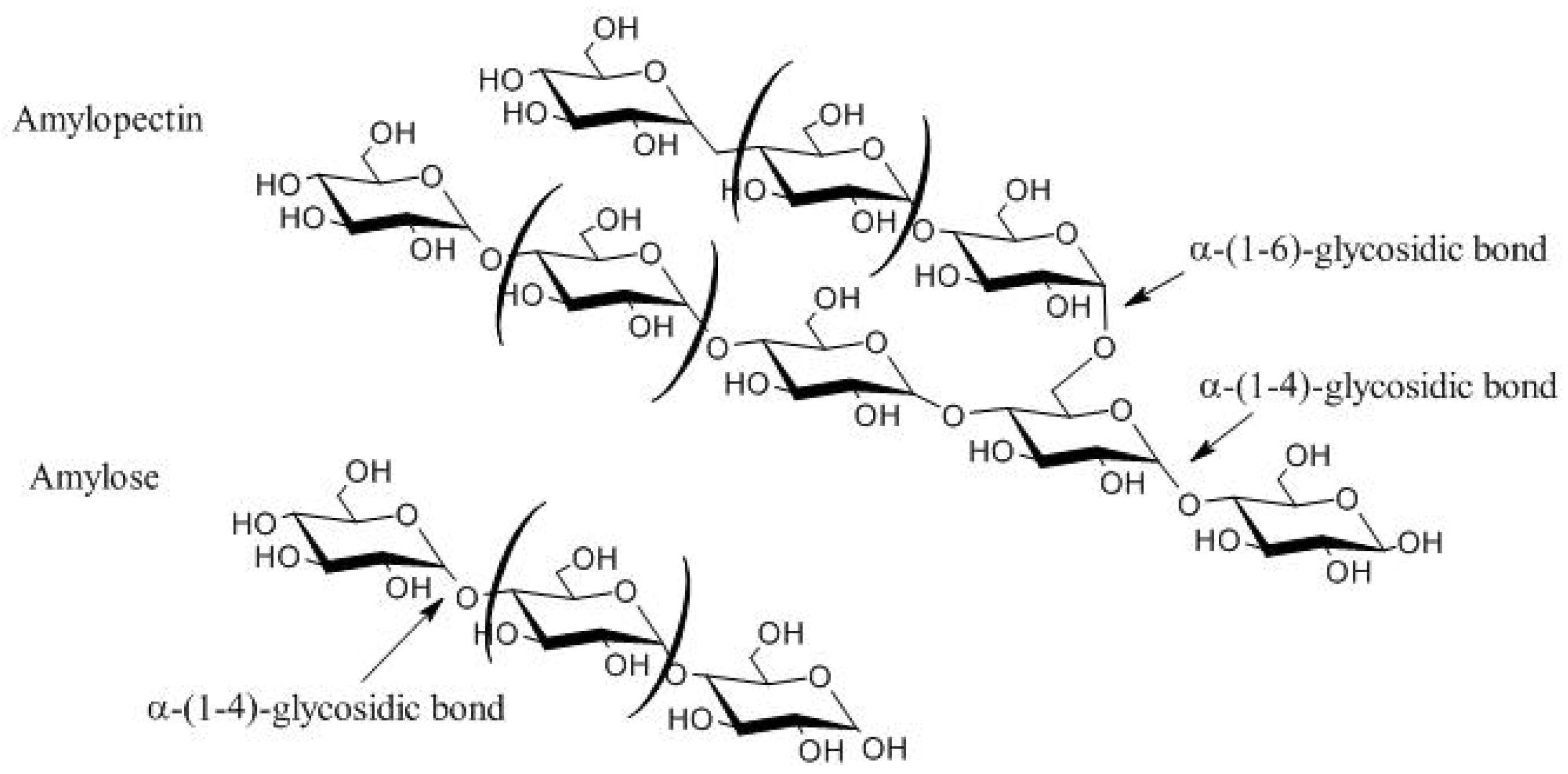

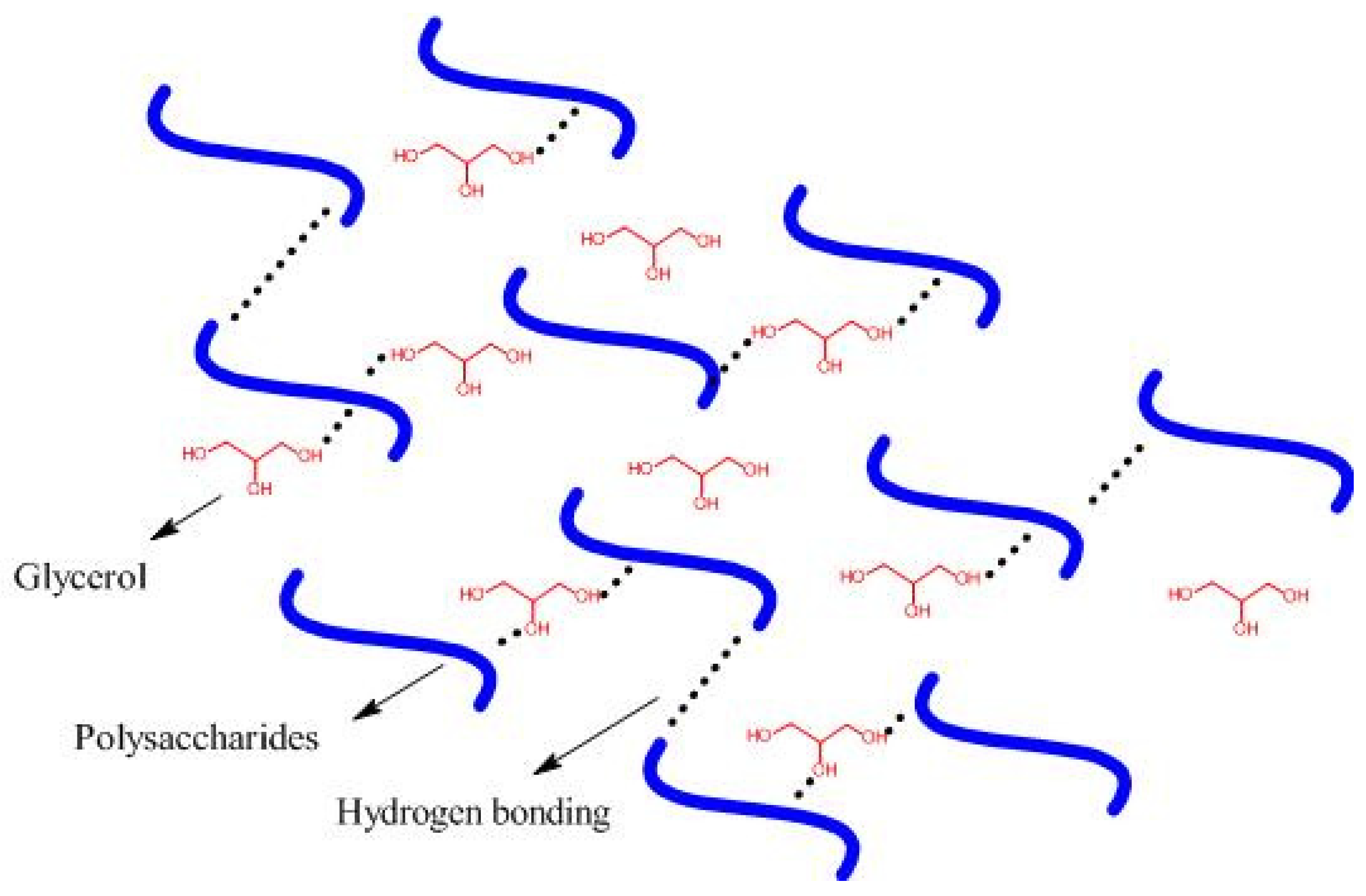

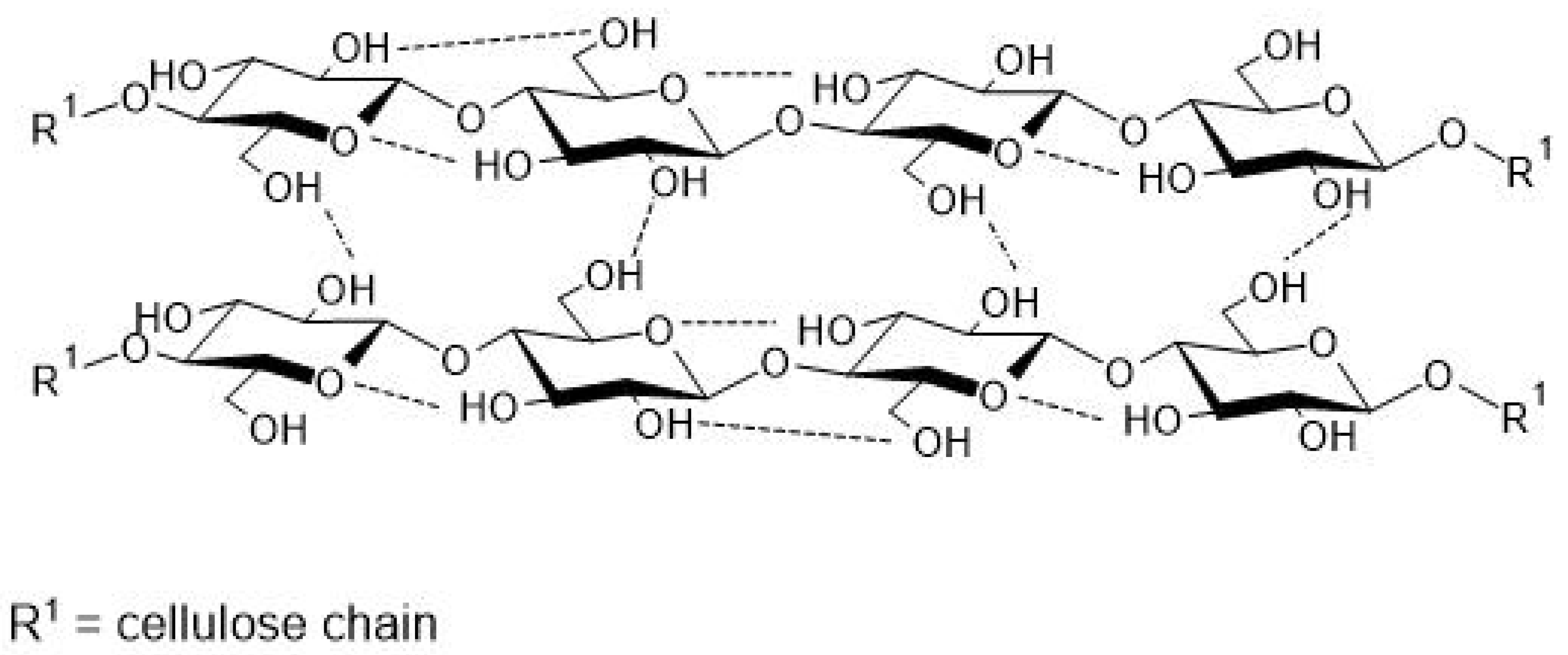

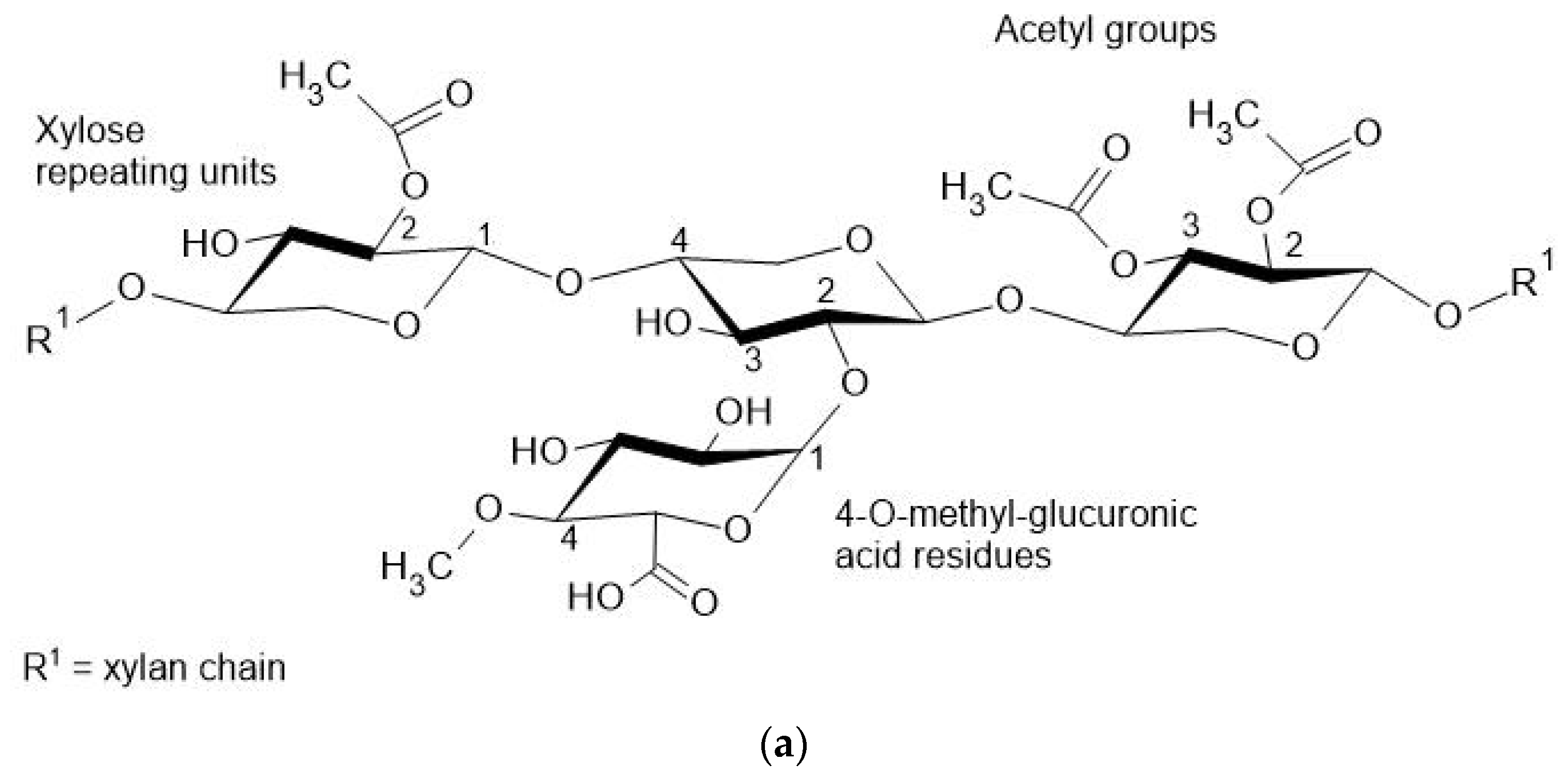

| Starch Source (%, w/v) | Tensile Strength (MPa) | Elongation at Break (%) | Water Solubility (%) | Mixture * (%, w/w) | Plasticizer * (%, w/w) | Bioplastic Processing Method | Reference |
|---|---|---|---|---|---|---|---|
| Sweetpotato (2.5%) | 7.96 | 77.92 | … | None | Sorbitol (40%) | Casting | [70] |
| Manioc (3%) | 64.29 | 3.87 | 20.81 | Gelatin (25%) | Glycerol (10%) | Casting | [71] |
| Manioc (3%) | 108.28 | 6.57 | 28.88 | Gelatin (75%) | Glycerol (10%) | Casting | [71] |
| Corn (5%) | 26 | 3.6 | … | Cellulose nanocrystals (13%) | Glycerol (26%) | Casting | [72] |
| Corn (5%) | 10 | 33.1 | … | None | Glycerol (30%) | Casting | [72] |
| Reag (2%) | 5.21 | 22.25 | 77.54 | Papaya (80%) | Glycerol (30%) | Casting | [73] |
| Rice (5%) | 10.9 | 2.8 | … | None | Glycerol (20%) | Casting | [52] |
| Sugar palm starch (8%) | 7.74 | 46.66 | >31 | None | Glycerol/Sorbitol 1:1 (30%) | Casting | [74] |
| Cassava (5.26%) | 1.14 | 0.22 | 13.48 | None | Glycerol (20%) | Casting | [75] |
| Pinhão (5%) | 18.56 | 2.8 | … | None | Glycerol (20%) | Casting | [50] |
| Arrowroot (2%) | 3.9 | 45.3 | … | None | Glycerol (30%) | Casting | [76] |
| Arrowroot (2%) | 11.5 | 44.4 | … | Gelatin (50%) | Glycerol (30%) | Casting | [76] |
| Hemicellulose (% w/w) * | Mixture (% w/w) * | Tensile Strength (MPa) | Elongationat Break (%) | E (MPa) | Reference |
|---|---|---|---|---|---|
| Xylan (87%) | Nanocellulose (13%) | 20.2 | 2.6 | 1578 | [47] |
| Xylan (50%) | Nanocellulose (50%) | 57 | 1.7 | 5700 | [142] |
| Arabinoxylan (85%) | Microcellulose (15%) | 95 | <15 | 2500 | [188] |
| Galactoglucomannans (85%) | Microcellulose (15%) | 15–20 | 3–4 | 800–1000 | [189] |
| Xylan (20%) | Gluten (80%) | 7–8 | 1–50 | 130–150 | [178] |
| Xylan (10%) | Microcellulose (90.9%) | 160–175 | … | 160–175 | [190] |
| Xylan (95%) | Nanocellulose (5%) | 51 | 2.9 | 3200 | [141] |
| Arabinoxylan (95%) | Bacterial Cellulose (5%) | 68 | 8.1 | 2700 | [191] |
| Arabinoxylan (75%) | Nanocellulose (25%) | 108 | 6 | 4800 | [187] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abe, M.M.; Martins, J.R.; Sanvezzo, P.B.; Macedo, J.V.; Branciforti, M.C.; Halley, P.; Botaro, V.R.; Brienzo, M. Advantages and Disadvantages of Bioplastics Production from Starch and Lignocellulosic Components. Polymers 2021, 13, 2484. https://doi.org/10.3390/polym13152484
Abe MM, Martins JR, Sanvezzo PB, Macedo JV, Branciforti MC, Halley P, Botaro VR, Brienzo M. Advantages and Disadvantages of Bioplastics Production from Starch and Lignocellulosic Components. Polymers. 2021; 13(15):2484. https://doi.org/10.3390/polym13152484
Chicago/Turabian StyleAbe, Mateus Manabu, Júlia Ribeiro Martins, Paula Bertolino Sanvezzo, João Vitor Macedo, Marcia Cristina Branciforti, Peter Halley, Vagner Roberto Botaro, and Michel Brienzo. 2021. "Advantages and Disadvantages of Bioplastics Production from Starch and Lignocellulosic Components" Polymers 13, no. 15: 2484. https://doi.org/10.3390/polym13152484
APA StyleAbe, M. M., Martins, J. R., Sanvezzo, P. B., Macedo, J. V., Branciforti, M. C., Halley, P., Botaro, V. R., & Brienzo, M. (2021). Advantages and Disadvantages of Bioplastics Production from Starch and Lignocellulosic Components. Polymers, 13(15), 2484. https://doi.org/10.3390/polym13152484






