Highlighting Thermo-Elastic Effects in Confined Fluids
Abstract
1. Introduction
2. Experimental
- -
- The ordinary polymer sample was an amorphous polymer: a polybutyl acrylate (PBuA) of Mn = 40,000 molecular weight and 1.19 polydispersity (Polymer Source Inc. manufacturer, Dorval (Montreal), Quebec). This molecular weight corresponds to the onset of the entanglement state (Mw ≅ 2 Me where Me = 22,000) [28]. The melt was studied at room temperature (i.e., at about 100 °C above the glass transition temperature (Tg = −64 °C)) with a 0.285 mm gap thickness. Its room temperature terminal viscoelastic time as deduced from conventional viscoelastic measurements was τrelax = 0.03 s [9]. At the low frequency probed for the thermal study (1 rad/s) and macroscopic scale, the PBuA exhibits a viscous response with η = 380 Pa s [6].
- -
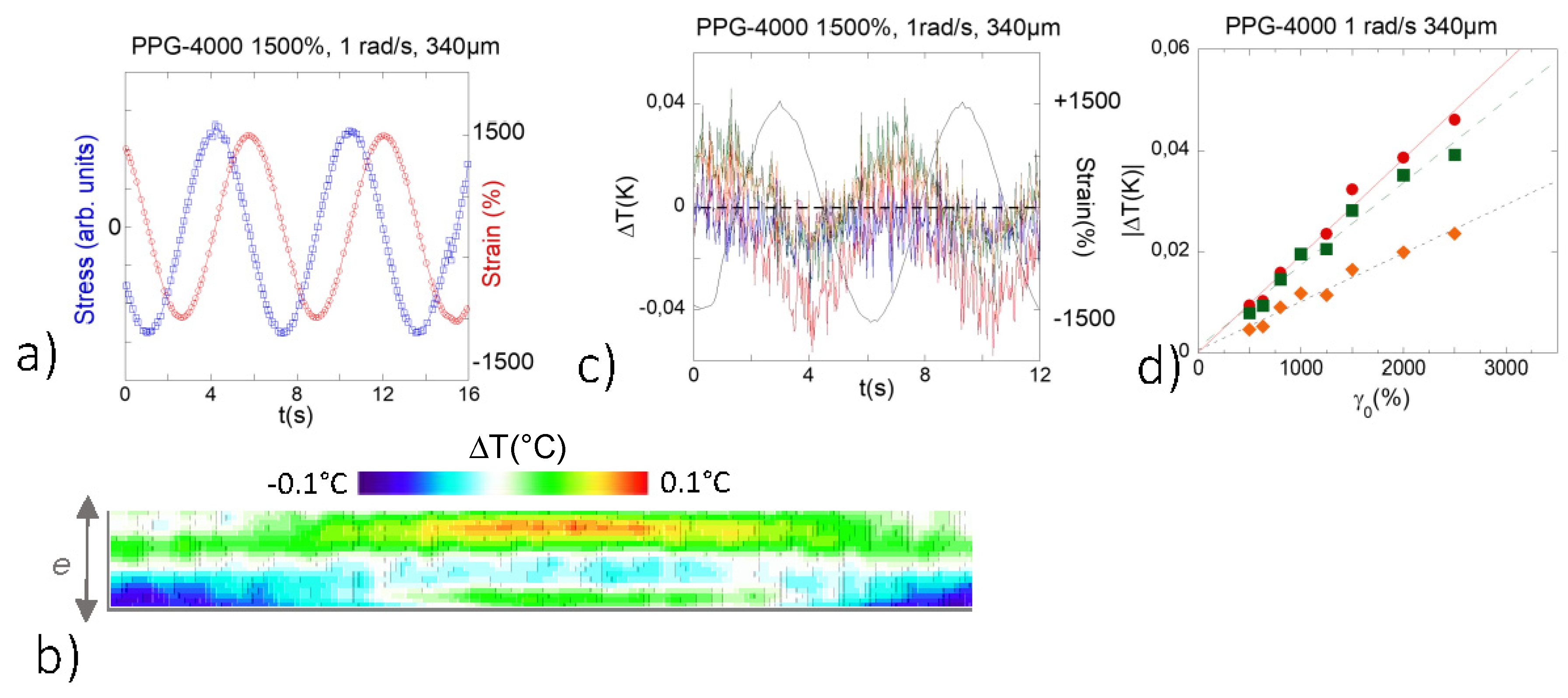
- The polymer glass former studied was polypropylene glycol-4000 (Sigma-Aldrich manufacturer, St. Louis, MO, USA). The molecular weight is given as 3500–4500 g/mol, which corresponds to about 55 repetition units. It is a viscous liquid at room temperature (η = 100 mPa s) and its relaxation time is out of the dynamic range of conventional mechanical tools [29].
- -
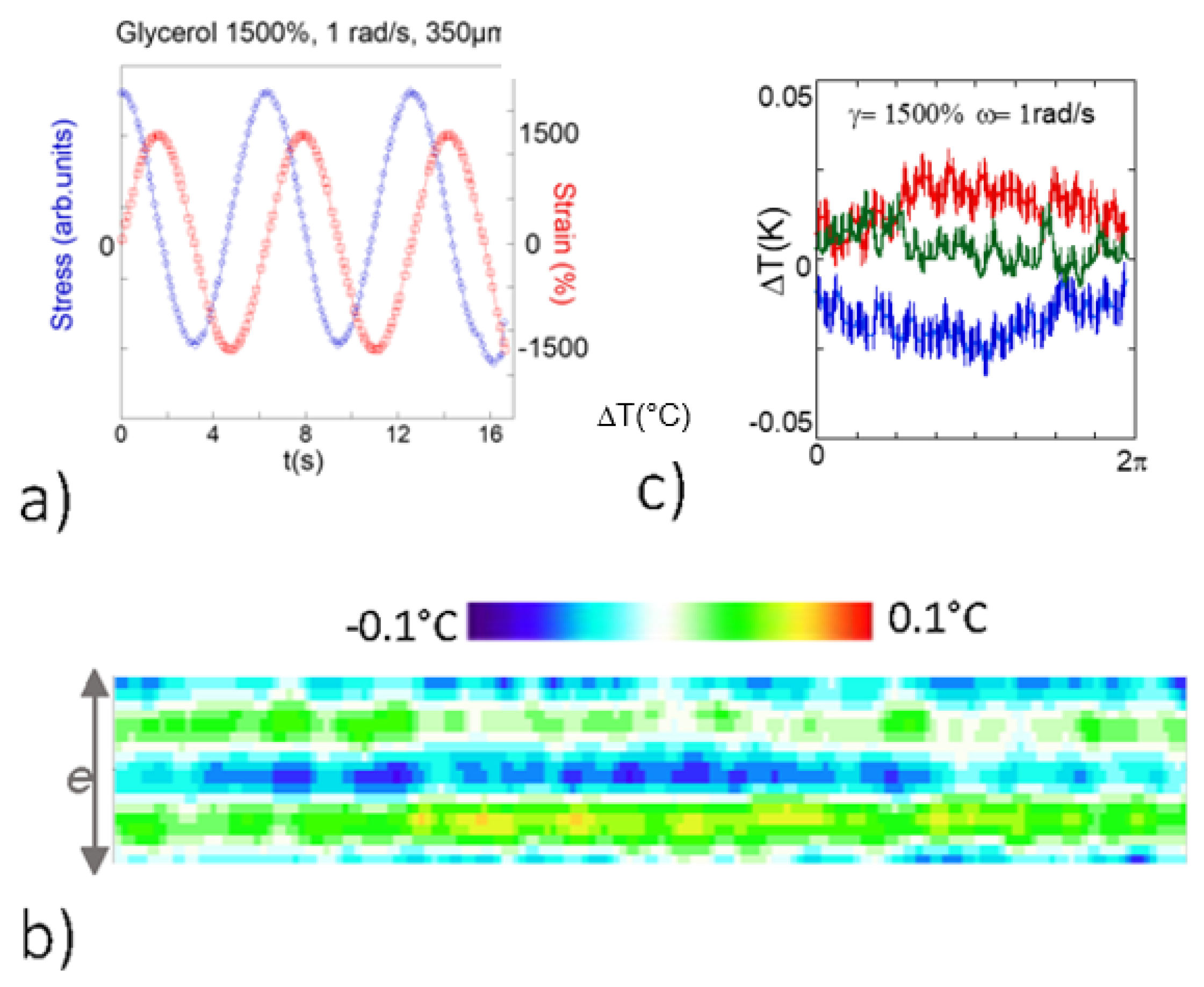
- Glycerol is a very well-studied liquid due its extremely wide range of uses and its biocompatibility. It is a glass former that exhibits at room temperature of viscosity (η = 1.41 Pa s). The molecular relaxation time is far away from the dynamic range of mechanical tools, being accessible by Brillouin scattering at 7 GHz [30].
3. Results
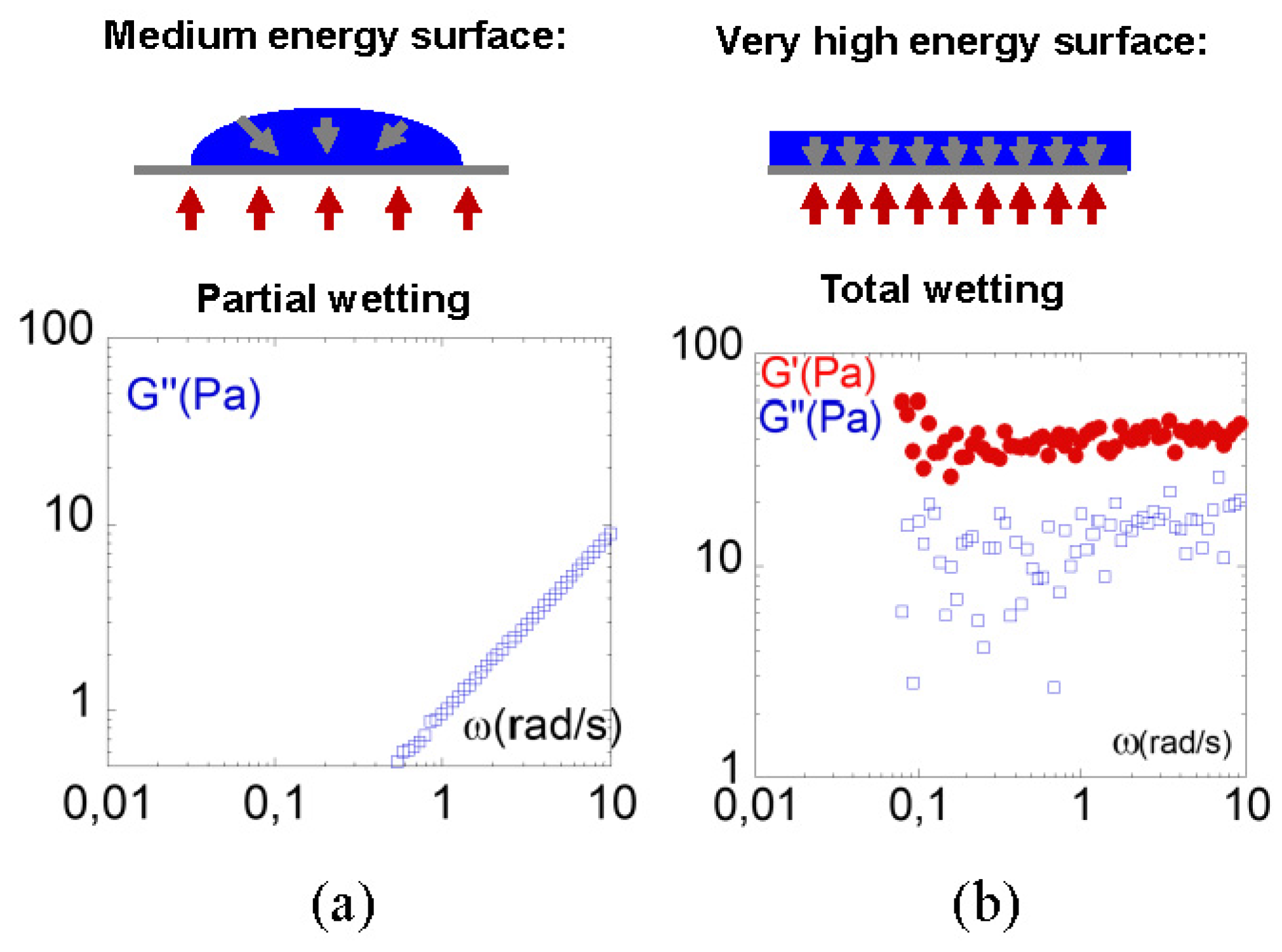
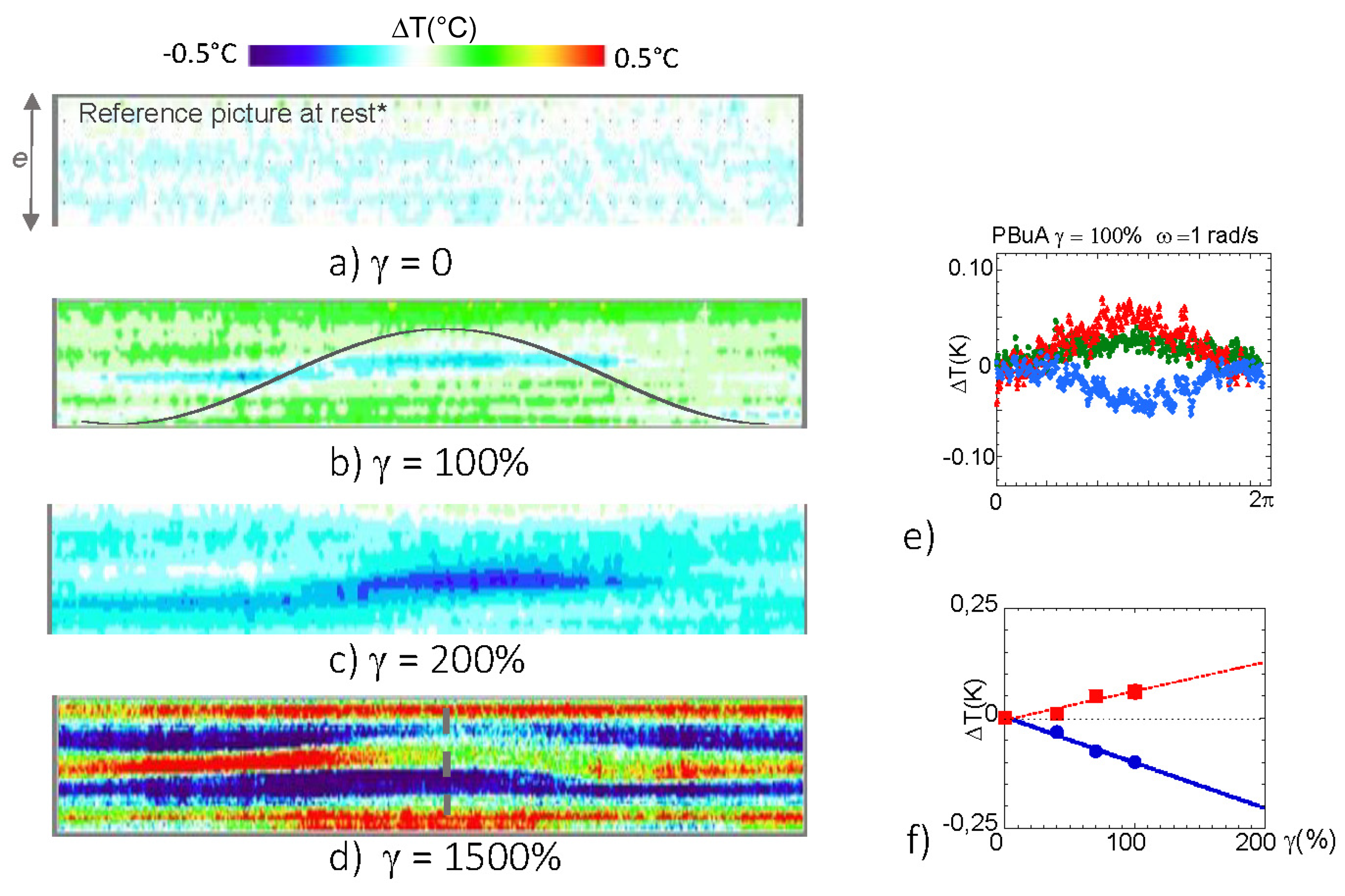
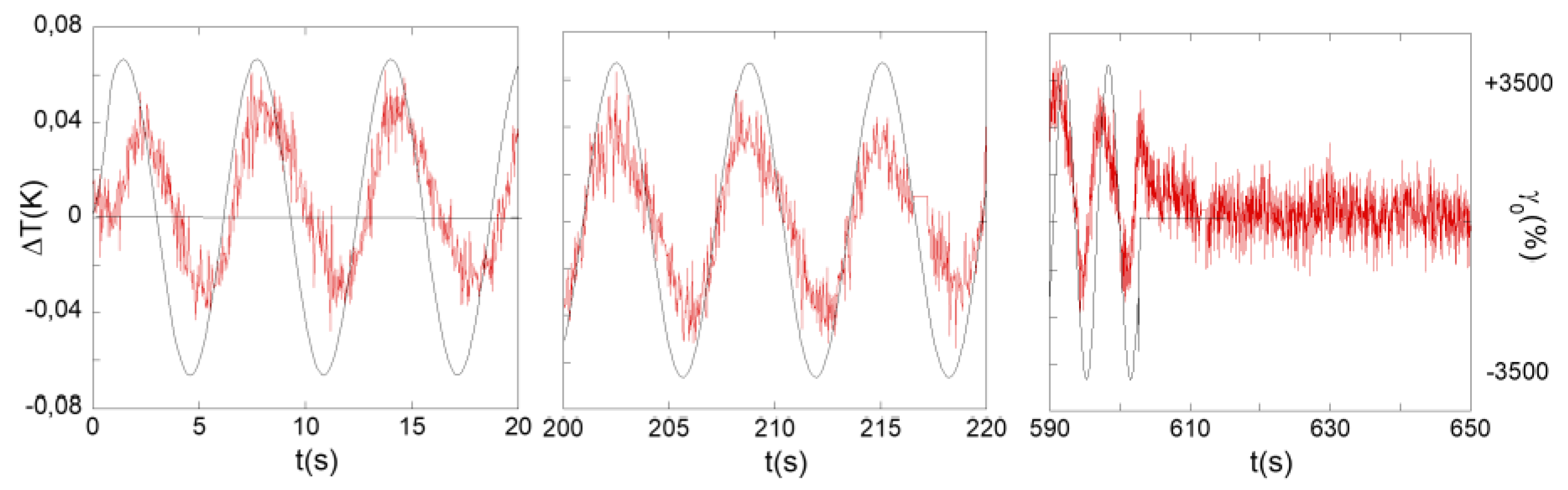
3.1. Identification of a Thermal Response of a Polymer Melt (PBuA) to an Oscillatory Shear Deformation
3.2. Identification of a Thermal Response in H-Bond Glass Formers upon Low Frequency Shear Deformation
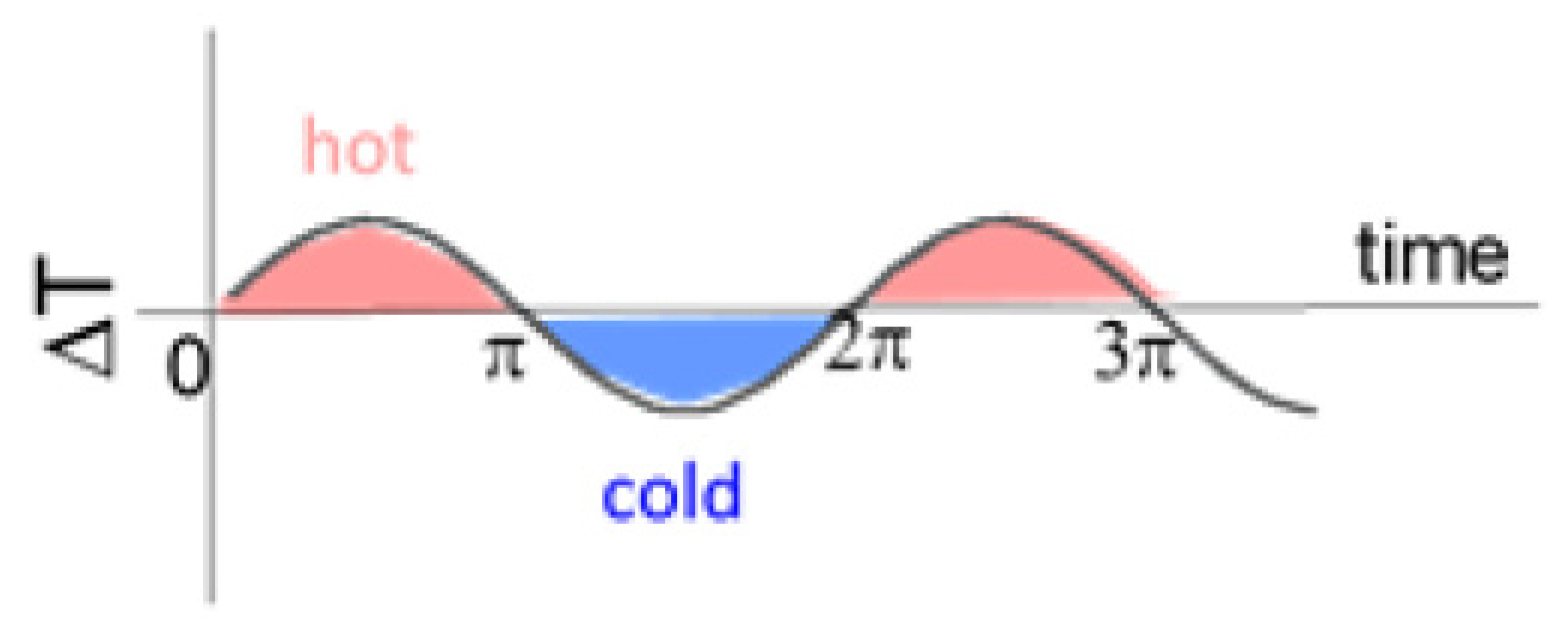
4. Discussion: Remarkable Net Temperature Invariance
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, T.-D.; Riedo, E. Nonlinear viscoelastic dynamics of nanoconfined wetting liquids. Phys. Rev. Lett. 2008, 100, 106102. [Google Scholar] [CrossRef] [PubMed]
- Xua, R.-G.; Lenga, Y. Squeezing and stick–slip friction behaviors of lubricants in boundary lubrication. Proc. Natl. Acad. Sci. USA 2018, 115, 6560–6565. [Google Scholar] [CrossRef]
- Reiter, G.; Demirel, A.L.; Peanasky, J.; Cai, L.L.; Granick, S. Stick to slip transition and adhesion of lubricated surfaces in moving contact. J. Chem. Phys. 1994, 101, 2606–2615. [Google Scholar] [CrossRef][Green Version]
- Lv, P.; Yang, Z.; Hua, Z.; Li, M.; Lin, M.; Dong, Z. Viscosity of water and hydrocarbon changes with micro-crevice thickness. Colloids Surf. A 2016, 504, 287–297. [Google Scholar] [CrossRef]
- Derjaguin, B.; Bazaron, U.; Zandanova, K.; Budaev, O. The complex shear modulus of polymeric and small-molecule liquids. Polymer 1989, 30, 97–103. [Google Scholar] [CrossRef]
- Derjaguin, B.V.; Bazaron, U.B.; Lamazhapova, K.D.; Tsidypov, B.D. Shear elasticity of low-viscosity liquids at low frequencies. Phys. Rev. A 1990, 42, 2255–2258. [Google Scholar] [CrossRef]
- Collin, D.; Martonity, P. Dynamic macroscopic heterogeneities in a fexible linear polymer melt. Physica A 2003, 320, 235–248. [Google Scholar] [CrossRef]
- Mendil, H.; Baroni, P.; Noirez, L. Solid-like rheological response of non-entangled polymers in the molten state. Eur. Phys. J. E 2006, 19, 77–85. [Google Scholar] [CrossRef]
- Noirez, L.; Baroni, P. Revealing the solid-like nature of glycerol at ambient temperature. J. Mol. Struct. 2010, 972, 16–21. [Google Scholar] [CrossRef]
- Windberger, U.; Noirez, L. Human Blood Plasma in Capillary-size Flow: Revealing Hidden Elasticity and Scale Dependence hal-02513618. Available online: https://hal.archives-ouvertes.fr/hal-02513618 (accessed on 21 June 2021).
- Zaccone, A.; Trachenko, K. Explaining the low-frequency shear elasticity of confined liquids. Proc. Natl. Acad. Sci. USA 2020, 117, 19653–19655. [Google Scholar] [CrossRef]
- Baggioli, M.; Vasin, M.; Brazhkin, V.V.; Trachenko, K. Field theory of dissipative systems with gapped momentum states. Phys. Rev. D 2020, 102, 025012. [Google Scholar] [CrossRef]
- Phillips, A.E.; Baggioli, M.; Sirk, T.W.; Trachenko, K.; Zaccone, A. Universal L−3 finite-size effects in the viscoelasticity of amorphous systems. Phys. Rev. Mater. 2021, 5, 035602. [Google Scholar] [CrossRef]
- Zaccone, A.; Noirez, L. Universal G′∼L–3 law for the low-frequency shear modulus of confined liquids. J. Phys. Chem. Lett. 2021, 12, 650–657. [Google Scholar] [CrossRef]
- Frenkel, J. Kinetic Theory of Liquids; Clarendon Press: Oxford, UK, 1946. [Google Scholar]
- Trachenko, K. Quantum dissipation in a scalar field theory with gapped momentum states. Sci. Rep. 2019, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Trachenko, K. Lagrangian formulation and symmetrical description of liquid dynamics. Phys. Rev. E 2017, 96, 062134. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Dove, M.; Brazhkin, V.V.; Trachenko, K. Emergence and evolution of the k gap in spectra of liquid and supercritical states. Phys. Rev. Lett. 2017, 118, 215502. [Google Scholar] [CrossRef] [PubMed]
- Trachenko, K.; Brazhkin, V.V. Collective modes and thermodynamics of the liquid state. Rep. Prog. Phys. 2016, 79, 016502. [Google Scholar] [CrossRef]
- Baggioli, M.; Vasin, M.; Brazhkin, V.; Trachenko, K. Gapped momentum states. Phys. Rep. 2020, 865, 1–44. [Google Scholar] [CrossRef]
- Baggioli, M.; Landry, M.; Zaccone, A. Deformations, relaxation and broken symmetries in liquids, solids and glasses: A unified topological field theory. arXiv 2021, arXiv:2101.05015. [Google Scholar]
- Volino, F. Théorie visco-élastique non-extensive. Ann. Phys. 1997, 22, 7–41. [Google Scholar] [CrossRef]
- Stillinger, F.H.; Weber, T.A. Hidden structu, re in liquids. Phys. Rev. A 1982, 25, 978–989. [Google Scholar] [CrossRef]
- Guenza, M. Intermolecular effects in the center-of-mass dynamics of unentangled polymer fluids. Macromolecules 2002, 35, 2714. [Google Scholar] [CrossRef]
- Zaccone, A.; Scossa-Romano, E. Approximate analytical description of the nonaffine response of amorphous solids. Phys. Rev. B 2011, 83, 184205. [Google Scholar] [CrossRef]
- Barnes, H.A. Handbook of Elementary Rheology; University of Wales: Aberystwyth, UK, 2000. [Google Scholar]
- Baroni, P.; Mendil, H.; Noirez, L. Method and Device for Determining at Least One Dynamic Property of a Deformable Solid or Fluid Material. Patent No. 0510988, 27 November 2005. [Google Scholar]
- Lakrout, H.; Creton, C.; Ahn, D.; Shull, K.R. Influence of molecular features on the tackiness of acrylic polymer melts. Macromolecules 2001, 34, 7448–7458. [Google Scholar] [CrossRef]
- Yahsi, U.; Coskun, B.; Yumak, A.; Boubaker, K.; Tav, C. Relaxation time of polypropylene glycol and polypropylene glycol dimethylether-like polymers in terms of fluid-phase temperature and pressure dependent hole fraction. Eur. Polym. J. 2015, 68, 226–232. [Google Scholar] [CrossRef]
- Scarponi, F.; Comez, L.; Fioretto, D.; Palmieri, L. Brillouin light scattering from transverse and longitudinal acoustic waves in glycerol. Phys. Rev. B 2004, 70, 054203. [Google Scholar] [CrossRef]
- Pineda-Pimentel, M.G.; Flores-Ramirez, N.; Farías Sanchez, J.C.; Domratcheva-Lvova, L.; Vasquez-Garcia, S.R.; García-Gonzalez, L. Theoretical analysis and FTIR of cellulose nanowhiskers/Poly(ButylAcrylate). Superf. Vacío 2016, 29, 83–86. [Google Scholar]
- De Zárate, M.O.; Sengers, J.V. Chapter 3: Fluctuations in fluids in thermodynamic equilibrium. In Hydrodynamic Fluctuations in Fluids and Fluid Mixtures; Elsevier Science: Amsterdam, The Netherlands, 2006; pp. 39–62. [Google Scholar]
- Morini, G.L. Viscous heating in liquid flows in micro-channels. Int. J. Heat Mass Transf. 2005, 48, 3637–3647. [Google Scholar] [CrossRef]
- Giacomin, J.; Bird, R.B.; Aumnate, C.; Mertz, A.M.; Schmalzer, A.; Mix, A.W. Viscous heating in large-amplitude oscillatory shear flow. Phys. Fluids 2012, 24, 103101. [Google Scholar] [CrossRef]
- Kume, E.; Baroni, P.; Noirez, L. Strain-induced violation of temperature uniformity in mesoscale liquids. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Kume, E.; Zaccone, A.; Noirez, L. Unexpected thermo-elastic effects in liquid glycerol by mechanical deformation. Phys. Fluids 2021, 33, 072007. [Google Scholar] [CrossRef]
- Cugliandolo, L.F.; Kurchan, J.; Peliti, L. Energy flow, partial equilibration, and effective temperatures in systems with slow dynamics. Phys. Rev. E 1997, 55, 3898–3914. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kume, E.; Baroni, P.; Noirez, L. Highlighting Thermo-Elastic Effects in Confined Fluids. Polymers 2021, 13, 2378. https://doi.org/10.3390/polym13142378
Kume E, Baroni P, Noirez L. Highlighting Thermo-Elastic Effects in Confined Fluids. Polymers. 2021; 13(14):2378. https://doi.org/10.3390/polym13142378
Chicago/Turabian StyleKume, Eni, Patrick Baroni, and Laurence Noirez. 2021. "Highlighting Thermo-Elastic Effects in Confined Fluids" Polymers 13, no. 14: 2378. https://doi.org/10.3390/polym13142378
APA StyleKume, E., Baroni, P., & Noirez, L. (2021). Highlighting Thermo-Elastic Effects in Confined Fluids. Polymers, 13(14), 2378. https://doi.org/10.3390/polym13142378






