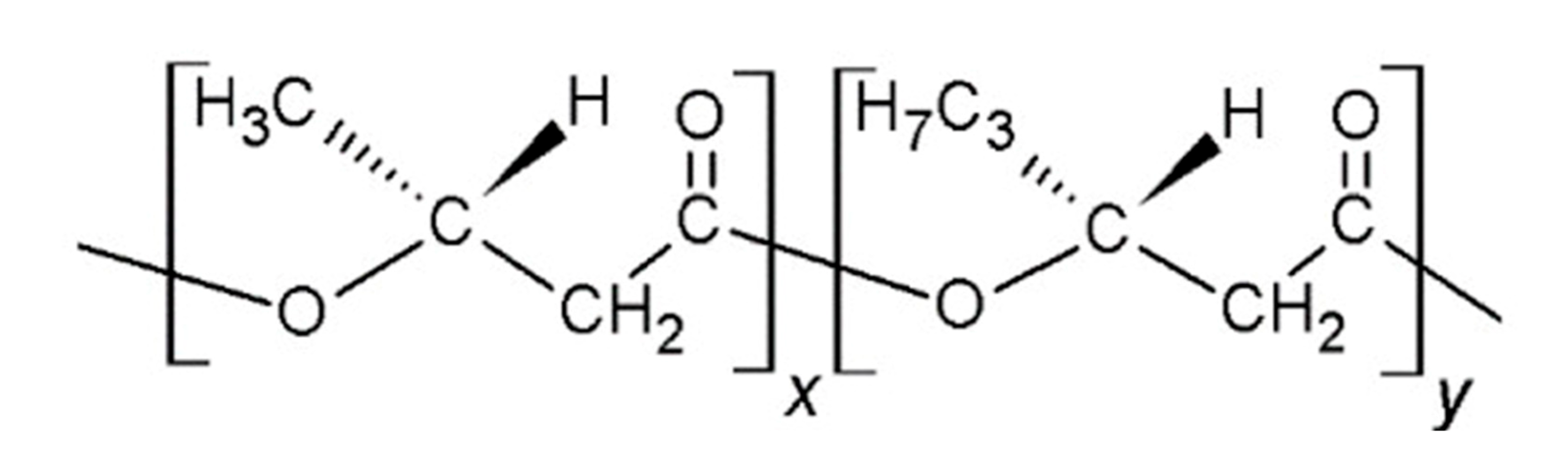
Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis of GO
2.3. Preparation of PHBHHx/GO Nanocomposites
2.4. Characterization Techniques
3. Results and Discussion
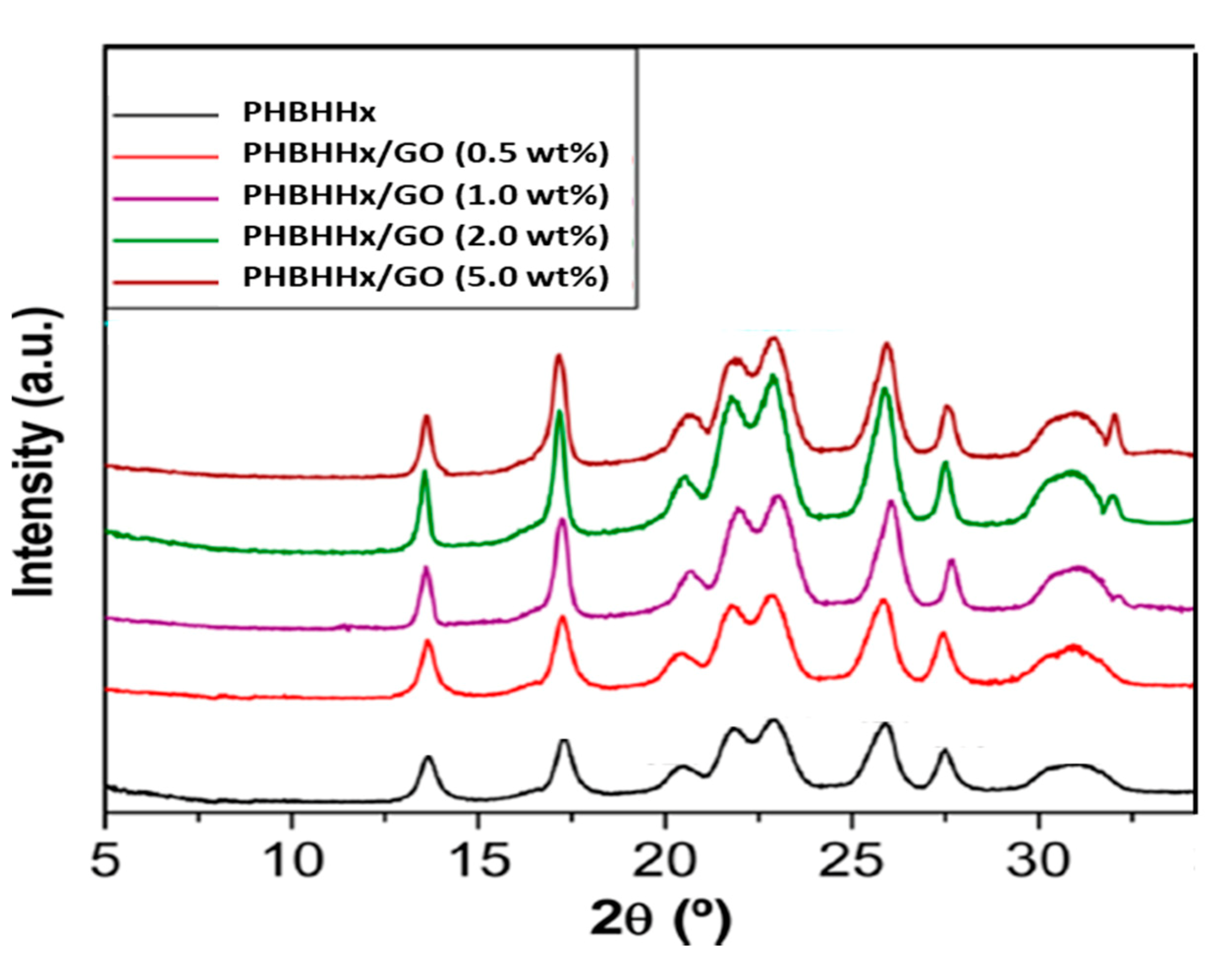
3.1. Surface Morphology and Crystalline Structure
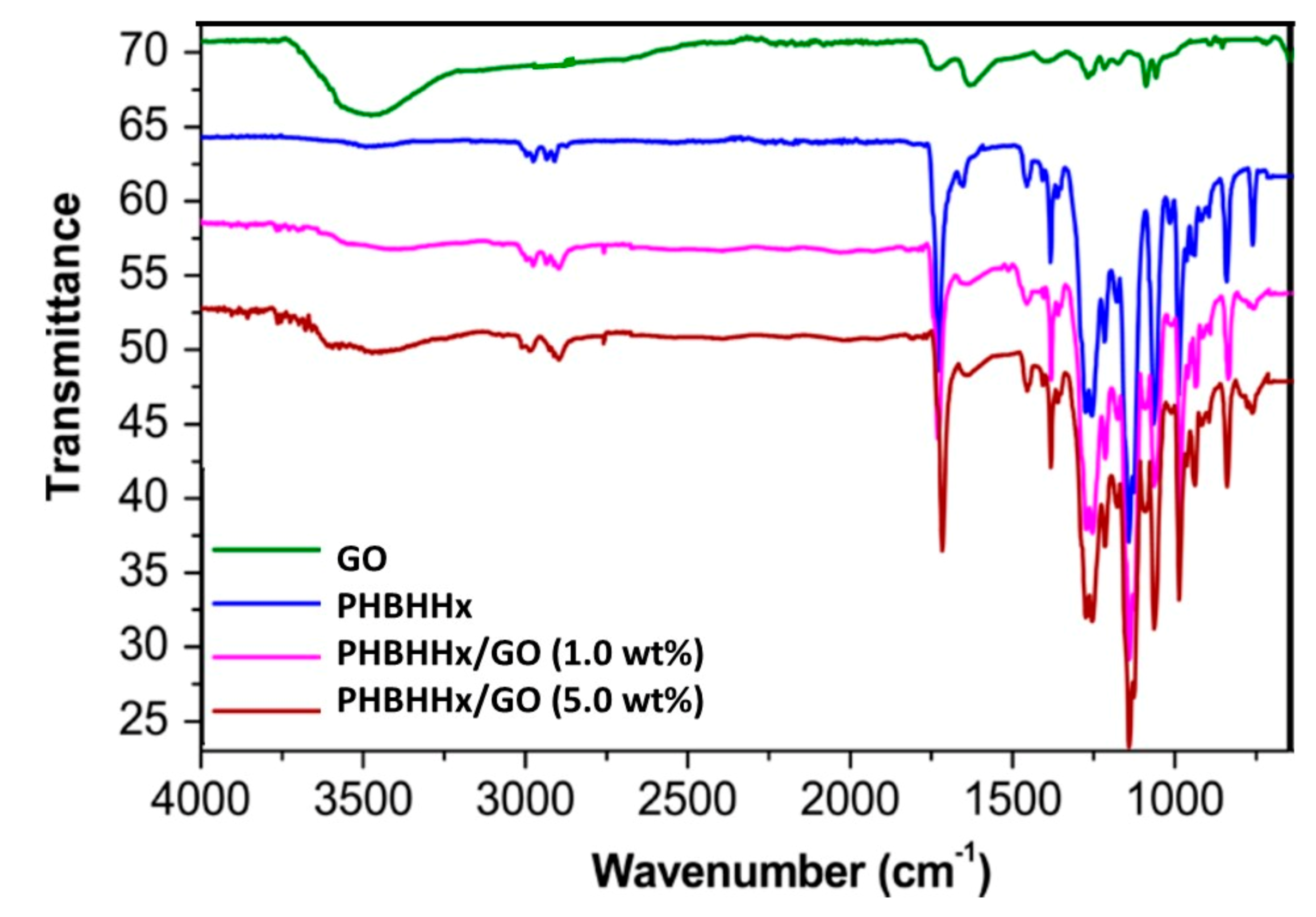
3.2. FT-IR Study
3.3. Thermal Stability
3.4. Mechanical Performance
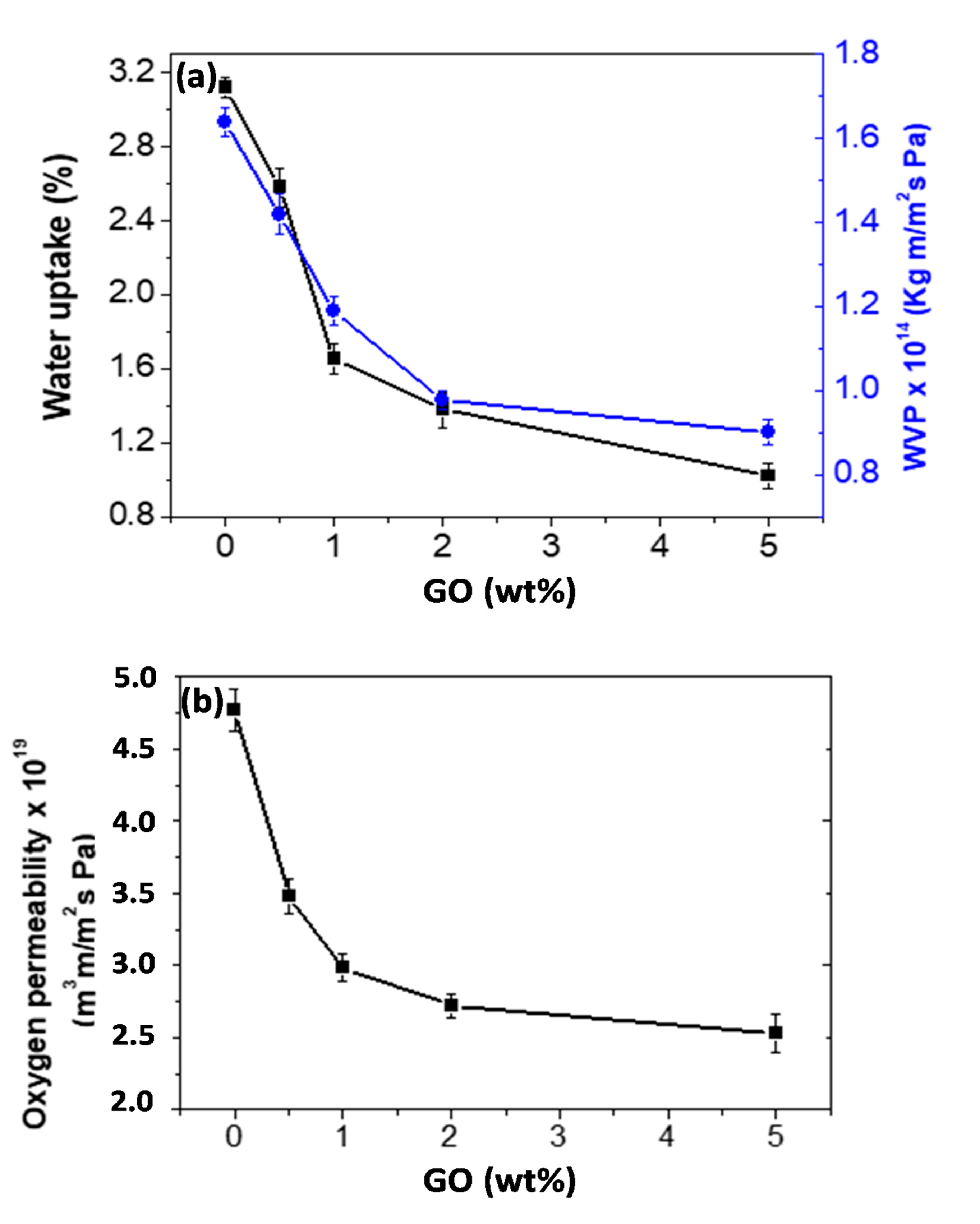
3.5. Barrier Properties
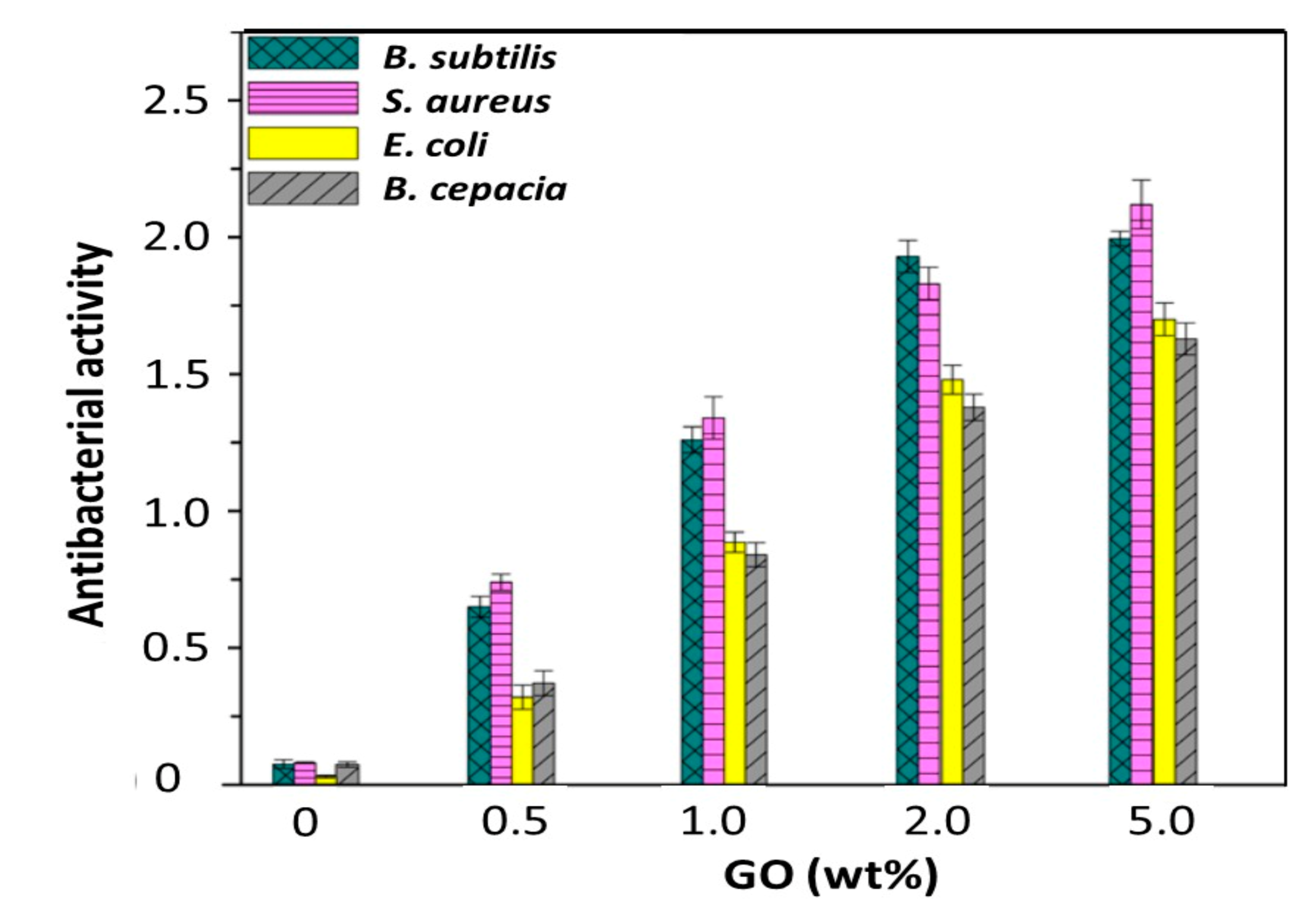
3.6. Antibacterial Properties
4. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Koller, M. A Review on Established and Emerging Fermentation Schemes for Microbial Production of Polyhydroxyalkanoate (PHA) Biopolyesters. Ferment. 2018, 4, 30. [Google Scholar] [CrossRef] [Green Version]
- Markl, E.; Grünbichler, H.; Lackner, M. PHB - Bio Based and Biodegradable Replacement for PP: A Review. Nov. Tech. Nutr. Food Sci. 2018, 2, 1–4. [Google Scholar] [CrossRef]
- Vroman, I.; Tighzert, L. Biodegradable Polymers. Materials 2009, 2, 307–344. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Fortunati, E.; Dominici, F.; Rayón, E.; Lopez, J.; Kenny, J.M. Multifunctional PLA–PHB/Cellulose Nanocrystal Films: Processing, Structural and Thermal Properties. Carbohydr. Polym. 2014, 107, 16–24. [Google Scholar] [CrossRef]
- Vogel, C.; Wessel, E.; Siesler, H.W. FT-IR Imaging Spectroscopy of Phase Separation in Blends of Poly(3-Hydroxybutyrate) With Poly (l-Lactic Acid) and Poly(ϵ-Caprolactone). Biomacromolecules 2008, 9, 523–527. [Google Scholar] [CrossRef]
- Prakalathan, K.; Mohanty, S.; Nayak, S.K. Reinforcing Effect and Isothermal Crystallization Kinetics of poly(3-Hydroxybutyrate) Nanocomposites Blended with Organically Modified Montmorillonite. Polym. Compos. 2014, 35, 999–1012. [Google Scholar] [CrossRef]
- Xu, C.; Qiu, Z. Crystallization Behavior and Thermal Property of Biodegradable poly(3-hydroxybutyrate)/Multi-Walled Carbon Nanotubes Nanocomposite. Polym. Adv. Technol. 2011, 22, 538–544. [Google Scholar] [CrossRef]
- Sadat-Shojai, M.; Khorasani, M.-T.; Jamshidi, A.; Irani, S. Nano-Hydroxyapatite Reinforced Polyhydroxybutyrate Composites: A Comprehensive Study on the Structural and in Vitro Biological Properties. Mater. Sci. Eng. C 2013, 33, 2776–2787. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Poly(3-hydroxybutyrate)/ZnO Bionanocomposites with Improved Mechanical, Barrier and Antibacterial Properties. Int. J. Mol. Sci. 2014, 15, 10950–10973. [Google Scholar] [CrossRef] [Green Version]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. ZnO-Reinforced Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerate) Bionanocomposites with Antimicrobial Function for Food Packaging. ACS Appl. Mater. Interfaces 2014, 6, 9822–9834. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Liu, Q.; Mark, J.E.; Noda, I. A Novel Biodegradable Nanocomposite Based on Poly (3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) and Silylated kaolinite/Silica core–shell Nanoparticles. Appl. Clay Sci. 2009, 46, 51–56. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Electrospun Fibers of Chitosan-Grafted polycaprolactone/Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) Blends. J. Mater. Chem. B 2015, 4, 600–612. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Wang, S.; Kong, M.; Geng, W.; Li, R.K.; Song, C.; Kong, D. Phase Morphology, Physical Properties, and Biodegradation Behavior of Novel PLA/PHBHHx Blends. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 100, 23–31. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Sánchez, J.A.L.; Capilla, R.P.; Díaz, P.G. Recent Developments in Graphene/Polymer Nanocomposites for Application in Polymer Solar Cells. Polymer 2018, 10, 217. [Google Scholar] [CrossRef] [Green Version]
- Díez-Pascual, A.M. Antibacterial Activity of Nanomaterials. Nanomaterial 2018, 8, 359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luceño-Sánchez, J.A.; Maties, G.; Gonzalez-Arellano, C.; Diez-Pascual, A.M. Synthesis and Characterization of Graphene Oxide Derivatives via Functionalization Reaction with Hexamethylene Diisocyanate. Nanomaterial 2018, 8, 870. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Ruan, J.; Song, H.; Zhang, J.; Wo, Y.; Guo, S.; Cui, D. Biocompatibility of Graphene Oxide. Nanoscale Res. Lett. 2010, 6, 8. [Google Scholar] [CrossRef] [Green Version]
- Lukowiak, A.; Kędziora, A.; Strek, W. Antimicrobial Graphene Family Materials: Progress, Advances, Hopes and Fears. Adv. Colloid Interface Sci. 2016, 236, 101–112. [Google Scholar] [CrossRef]
- Han, W.; Wu, Z.; Li, Y.; Wang, Y. Graphene Family Nanomaterials (GFNs)—promising Materials for Antimicrobial Coating and Film: A Review. Chem. Eng. J. 2019, 358, 1022–1037. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E. Toxicity of Graphene and Graphene Oxide Nanowalls Against Bacteria. ACS Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef]
- Chen, J.; Peng, H.; Wang, X.; Shao, F.; Yuan, Z.; Han, H.-Y. Graphene Oxide Exhibits Broad-Spectrum Antimicrobial Activity Against Bacterial Phytopathogens and Fungal Conidia by Intertwining and Membrane Perturbation. Nanoscale 2014, 6, 1879–1889. [Google Scholar] [CrossRef]
- Sánchez, J.A.L.; Capilla, R.P.; Díez-Pascual, A.M. High-Performance PEDOT: PSS/Hexamethylene Diisocyanate-Functionalized Graphene Oxide Nanocomposites: Preparation and Properties. Polymer 2018, 10, 1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ASTM E96–95. Standard Test Methods for Water Vapor Transmission of Materials. In ASTM, Annual Book of ASTM; ASTM International: Philadelphia, PA, USA, 1995; pp. 406–413. [Google Scholar]
- ASTM D3985–05. Standard Test Method for Oxygen Gas Transmission Rate through Plastic Film and Sheeting Using a Coulometric Sensor. In ASTM, Annual Book of ASTM; ASTM International: Philadelphia, PA, USA, 2010. [Google Scholar]
- Pan, P.; Liang, Z.; Nakamura, N.; Miyagawa, T.; Inoue, Y. Uracil as Nucleating Agent for Bacterial Poly[(3-Hydroxybutyrate)-Co-(3-hydroxyhexanoate)] Copolymers. Macromol. Biosci. 2009, 9, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.-T.; Ke, Y.-L. Superior Crystallization Kinetics Caused by the Remarkable Nucleation Effect of Graphene Oxide in Novel Ternary Biodegradable Polymer Composites. ACS Omega 2020, 5, 30643–30656. [Google Scholar] [CrossRef] [PubMed]
- Alexander, L.E. X-Ray Diffraction Methods in Polymer Science, 1st ed.; Wiley-Interscience: New York, NY, USA, 1969. [Google Scholar]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Poly (propylene fumarate)/Polyethylene Glycol-Modified Graphene Oxide Nanocomposites for Tissue Engineering. ACS Appl. Mater. Interfaces 2016, 8, 17902–17914. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Dabbs, D.M.; Liu, L.-M.; Aksay, I.A.; Car, R.; Selloni, A. Combined Effects of Functional Groups, Lattice Defects, and Edges in the Infrared Spectra of Graphene Oxide. J. Phys. Chem. C 2015, 119, 18167–18176. [Google Scholar] [CrossRef]
- Lim, J.S.; Noda, I.; Im, S.S. Effect of Hydrogen Bonding on the Crystallization Behavior of poly(3-Hydroxybutyrate-Co-3-hydroxyhexanoate)/Silica Hybrid Composites. Polymer 2007, 48, 2745–2754. [Google Scholar] [CrossRef]
- Liu, C.; Jia, M.; Qu, J.; Noda, I.; Chase, D.B.; Street, R.; Rabolt, J.F. Intermolecular Hydrogen Bonding Between Poly[(R)-3-Hydroxybutyrate] (PHB) and Pseudoboehmite and Its Effect on Crystallization of PHB. ACS Appl. Polym. Mater. 2020, 2, 4762–4769. [Google Scholar] [CrossRef]
- Suttiwijitpukdee, N.; Sato, H.; Unger, M.; Ozaki, Y. Effects of Hydrogen Bond Intermolecular Interactions on the Crystal Spherulite of Poly(3-Hydroxybutyrate) and Cellulose Acetate Butyrate Blends: Studied by FT-IR and FT-NIR Imaging Spectroscopy. Macromology 2012, 45, 2738–2748. [Google Scholar] [CrossRef]
- Grassie, N.; Murray, E.; Holmes, P. The Thermal Degradation of Poly (-(d)-β-Hydroxybutyric acid): Part 3—The Reaction Mechanism. Polym. Degrad. Stab. 1984, 6, 127–134. [Google Scholar] [CrossRef]
- Chancelier, L.; Boyron, O.; Gutel, T.; Santini, C. Thermal Stability of Imidazolium-Based Ionic Liquids. French-Ukrainian J. Chem. 2016, 4, 51–64. [Google Scholar] [CrossRef] [Green Version]
- He, J.-D.; Cheung, M.K.; Yu, P.H.; Chen, G.-Q. Thermal Analyses of poly(3-hydroxybutyrate), poly(3-Hydroxybutyrate-Co-3-hydroxyvalerate), and poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate). J. Appl. Polym. Sci. 2001, 82, 90–98. [Google Scholar] [CrossRef]
- Sang, B.; Li, Z.-W.; Li, X.-H.; Yu, L.-G.; Zhang, Z.-J. Graphene-Based Flame Retardants: A Review. J. Mater. Sci. 2016, 51, 8271–8295. [Google Scholar] [CrossRef]
- Yin, Y.; Jiang, B.; Zhu, X.; Meng, L.; Huang, Y. Investigation of Thermostability of Modified Graphene Oxide/Methylsilicone Resin Nanocomposites. Eng. Sci. 2018, 5, 73–78. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Tian, H.; Yao, Y.; Xiang, A.; Qi, H.; Wu, Q.; Rajulu, A.V. Polyimide Foams with Outstanding Flame Resistance and Mechanical Properties by the Incorporation of Noncovalent Bond Modified Graphene Oxide. New J. Chem. 2020, 44, 12068–12078. [Google Scholar] [CrossRef]
- Yang, H.-X.; Sun, M.; Zhang, Y.; Zhou, P. Degradable PHBHHx Modified by the Silk Fibroin for the Applications of Cardiovascular Tissue Engineering. ISRN Mater. Sci. 2011, 2011, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Khoei, A.R.; Khorrami, M.S. Mechanical Properties of Graphene Oxide: A Molecular Dynamics Study. Full- Nanotub. Carbon Nanostructures 2016, 24, 594–603. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Naffakh, M. Towards the Development of (poly (phenylene Sulphide) Based Nanocomposites with Enhanced Mechanical, Electrical and Tribological Properties. Mater. Chem. Phys. 2012, 135, 348–357. [Google Scholar] [CrossRef] [Green Version]
- Díez-Pascual, A.M.; Naffakh, M. Enhancing the Thermomechanical Behaviour of poly(phenylene Sulphide) Based Composites via Incorporation of Covalently Grafted Carbon Nanotubes. Compos. Part A: Appl. Sci. Manuf. 2013, 54, 10–19. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Rosa, M.D. Biodegradable Polymers for Food Packaging: A Review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Bunch, J.S.; Verbridge, S.S.; Alden, J.S.; van der Zande, A.M.; Parpia, J.M.; Craighead, H.G.; McEuen, P.L. Impermeable Atomic Membranes from Graphene Sheets. Nano Lett. 2008, 8, 2458–2462. [Google Scholar] [CrossRef] [Green Version]
- Cui, Y.; Kundalwal, S.; Kumar, S. Gas Barrier Performance of graphene/Polymer Nanocomposites. Carbon 2016, 98, 313–333. [Google Scholar] [CrossRef] [Green Version]
- Ke, Y.; Liu, C.; Zhang, X.; Xiao, M.; Wu, G. Surface Modification of Polyhydroxyalkanoates Toward Enhancing Cell Compatibility and Antibacterial Activity. Macromol. Mater. Eng. 2017, 302, 1700258. [Google Scholar] [CrossRef]
- Hancock, J.T.; Desikan, R.; Neill, S. Role of Reactive Oxygen Species in Cell Signalling Pathways. Biochem. Soc. Trans. 2001, 29, 345–349. [Google Scholar] [CrossRef] [PubMed]

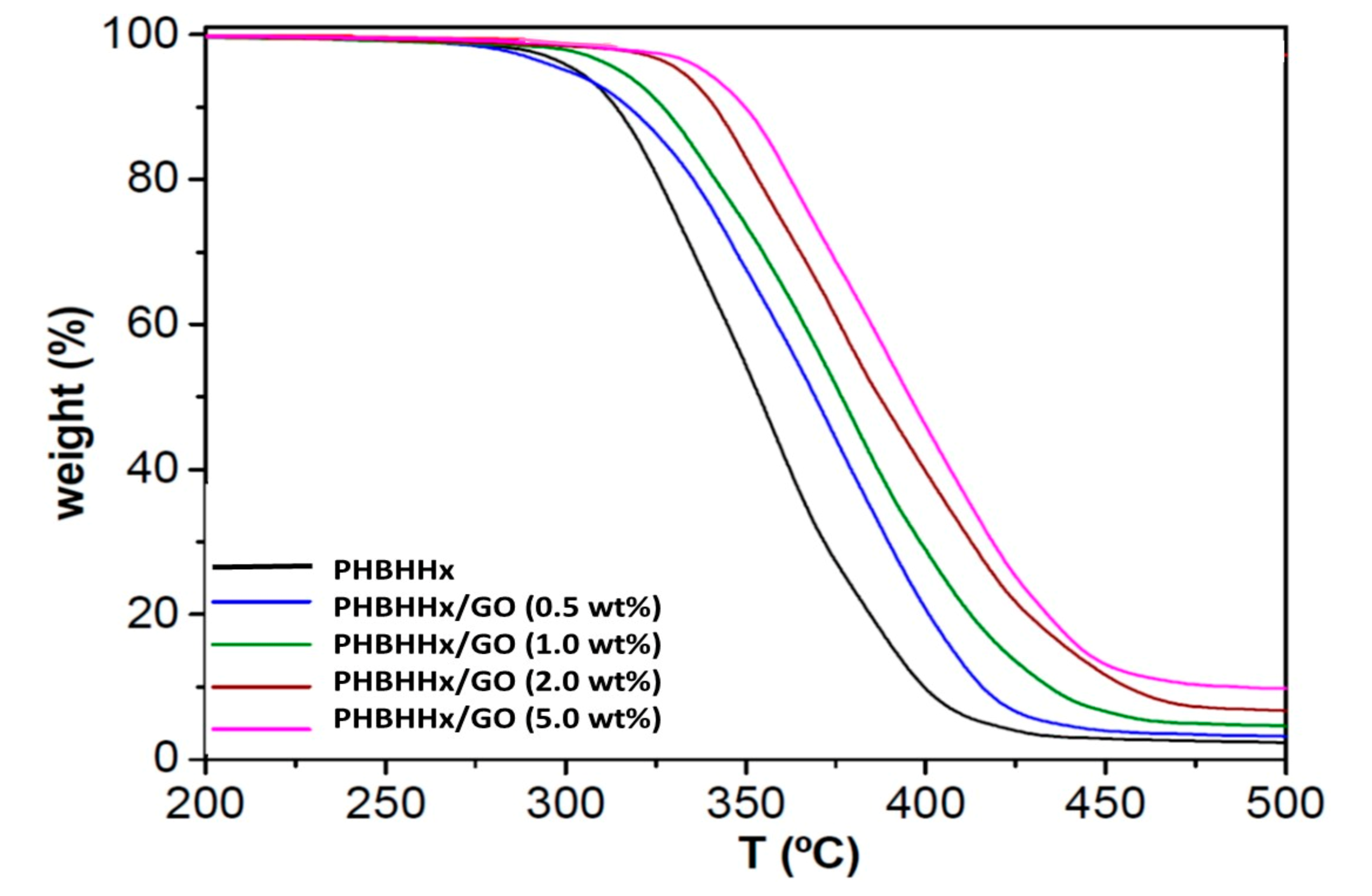

| Sample | Ra (µm) | Tstart (°C) | T10 (°C) | Tpeak (°C) | R (wt%) |
|---|---|---|---|---|---|
| PHBHHx | 0.22 | 280 | 314 | 325 | 2.2 |
| PHBHHx/GO (0.5 wt%) | 0.25 | 277 | 316 | 339 | 2.9 |
| PHBHHx/GO (1.0 wt%) | 0.36 | 289 | 325 | 343 | 4.0 |
| PHBHHx/GO (2.0 wt%) | 0.59 | 302 | 343 | 361 | 6.1 |
| PHBHHx/GO (5.0 wt%) | 0.78 | 304 | 348 | 365 | 10.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díez-Pascual, A.M. Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate). Polymers 2021, 13, 2233. https://doi.org/10.3390/polym13142233
Díez-Pascual AM. Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate). Polymers. 2021; 13(14):2233. https://doi.org/10.3390/polym13142233
Chicago/Turabian StyleDíez-Pascual, Ana M. 2021. "Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate)" Polymers 13, no. 14: 2233. https://doi.org/10.3390/polym13142233
APA StyleDíez-Pascual, A. M. (2021). Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate). Polymers, 13(14), 2233. https://doi.org/10.3390/polym13142233






