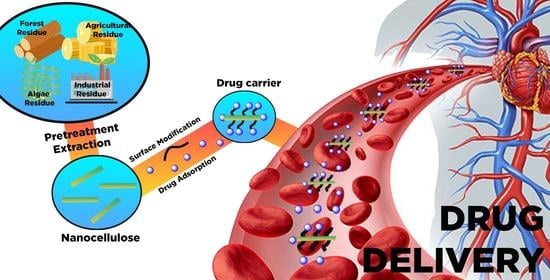
Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers
Abstract
:1. Introduction
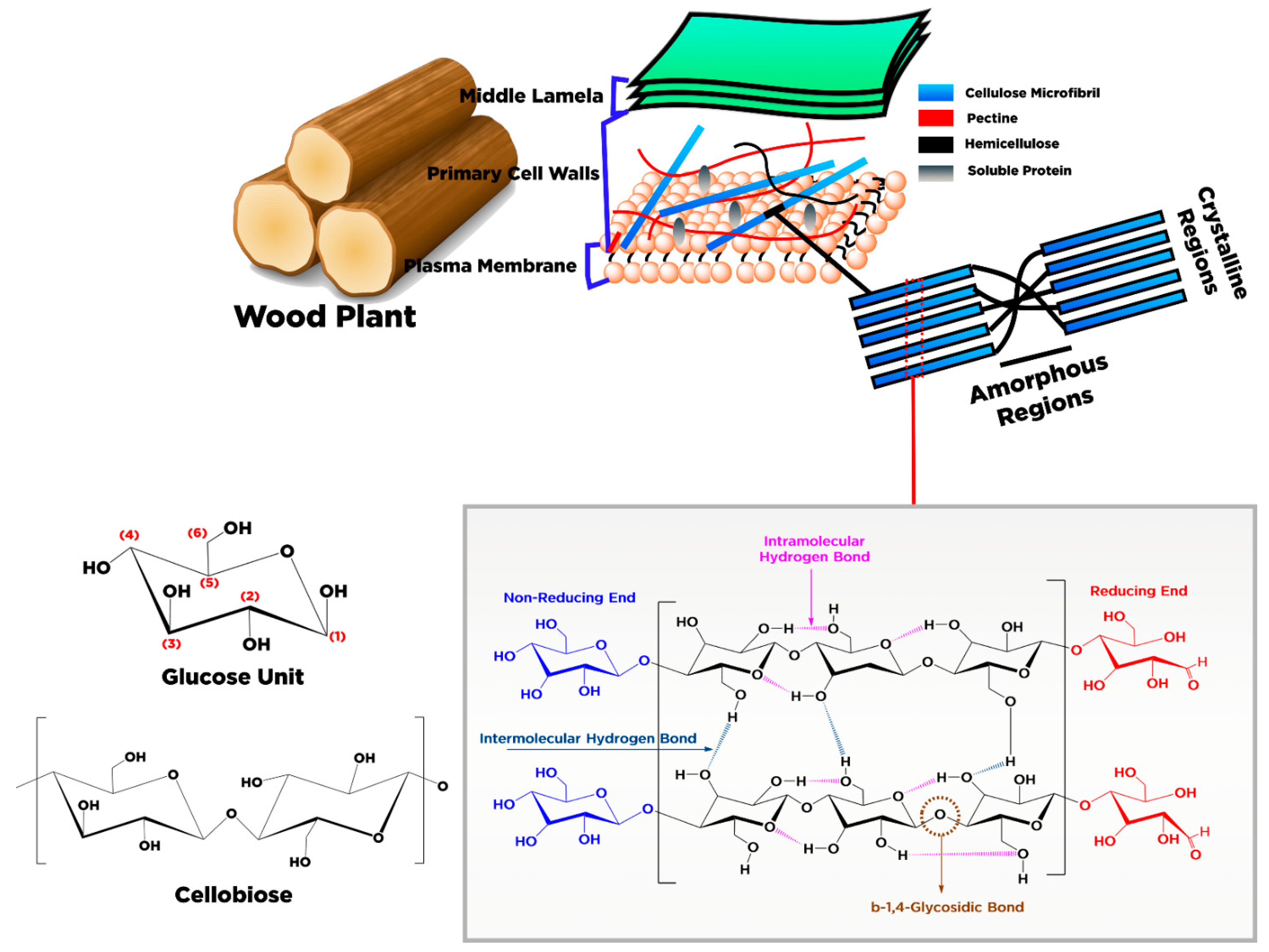
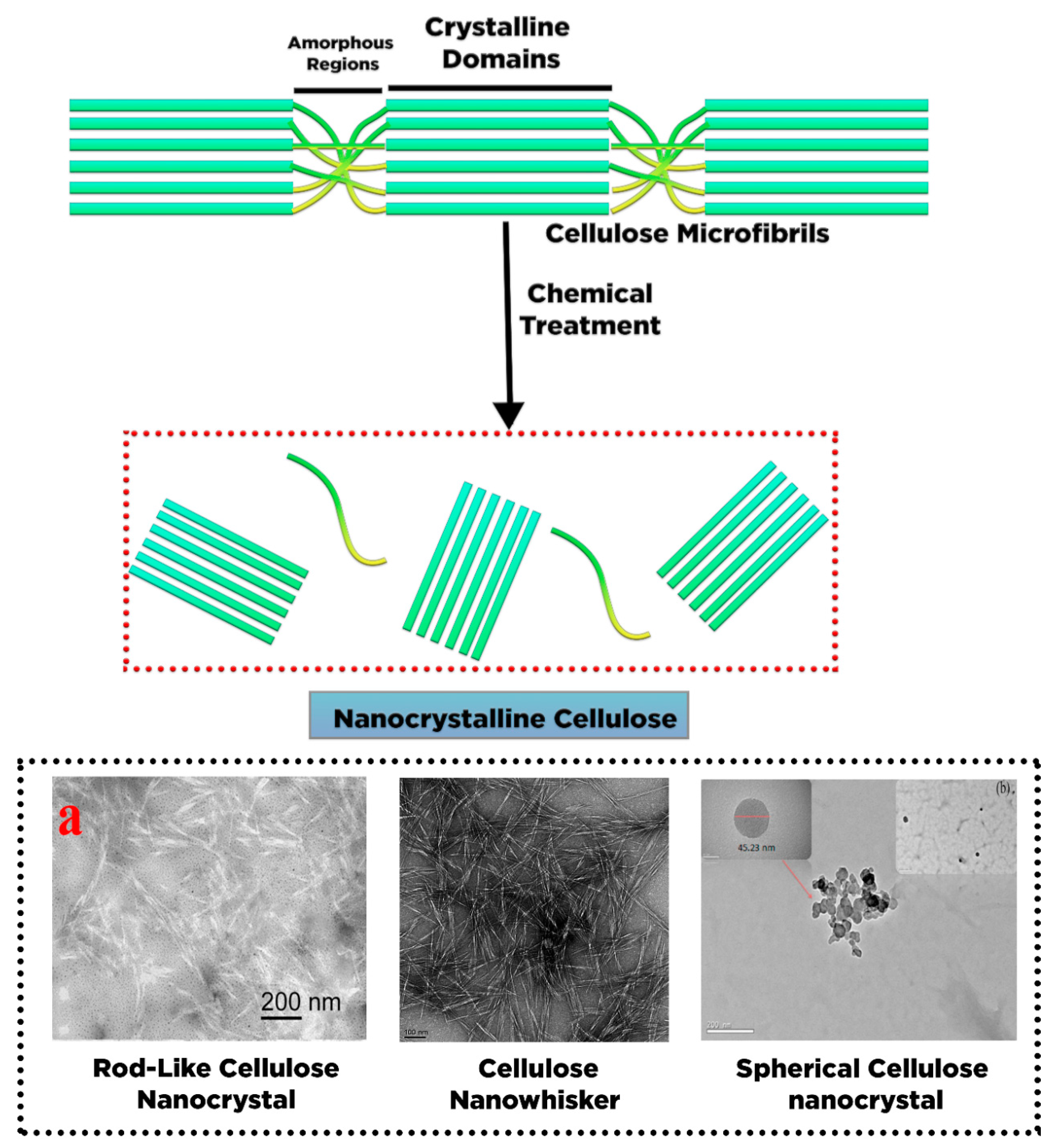
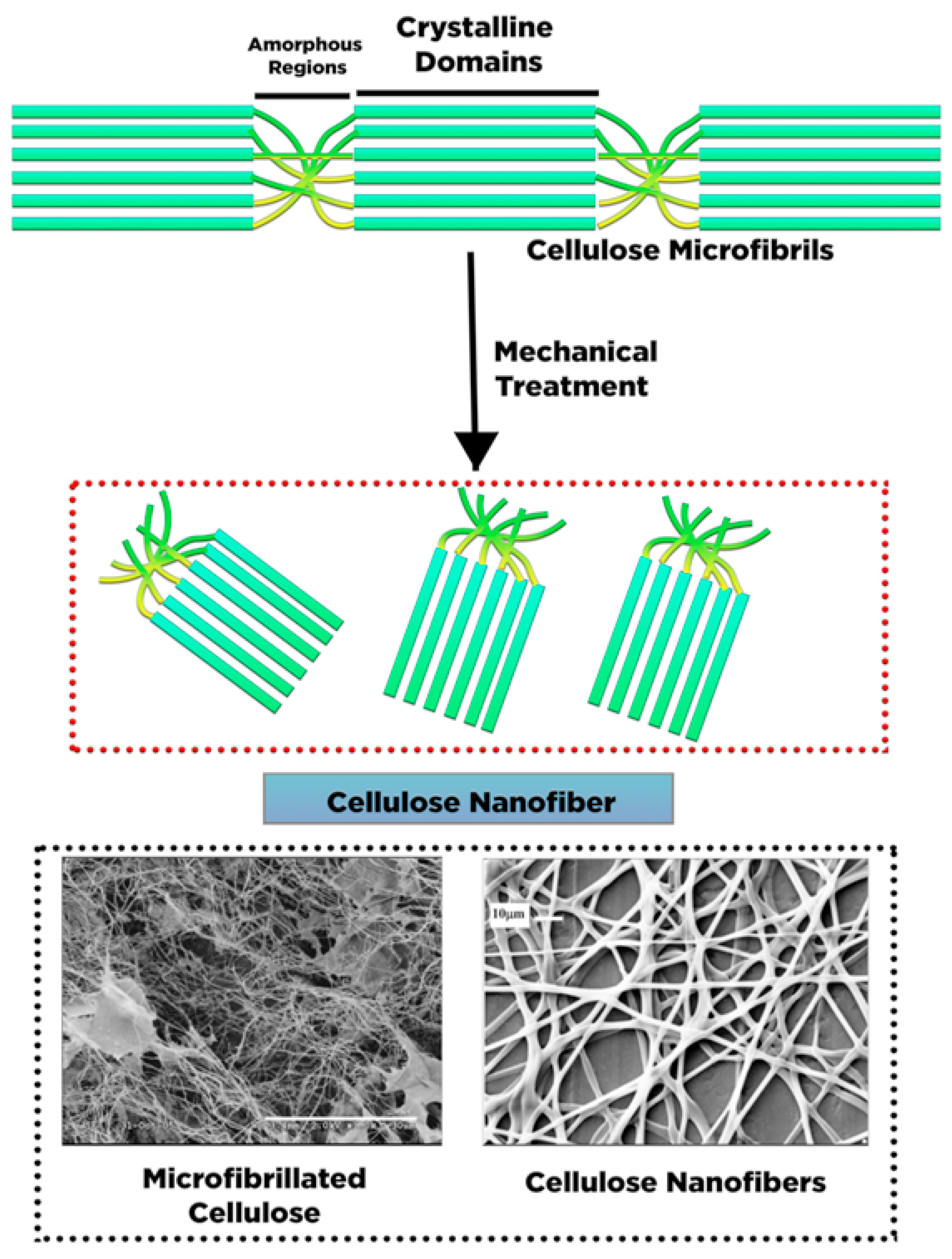
2. Conversion of Cellulose into Nanocellulose and Its Characteristic
3. Sources and Pretreatment of Raw Materials for Nanocellulose Productions
4. Isolation of Nanocellulose
4.1. Isolation of Nano-Fibrillated Cellulose (NFC)
4.2. Isolation of Cellulose Nanocrystal (NCC)
4.3. Isolation of Bacteria Cellulose (BC)
5. Surface Chemistry of Nanocellulose for Drug Delivery
5.1. Functionalization of Nanocellulose through Physical Technique
5.2. Functionalization through Chemical Synthesis of Nanocellulose
5.3. Functionalization through Post Chemical Modification via Covalent and Physical Bonding Strategy
5.4. Polymer Grafting Modified Nanocellulose
5.5. Surfactant Modified Nanocellulose
5.6. Polyelectrolytes-Based Nanocellulose
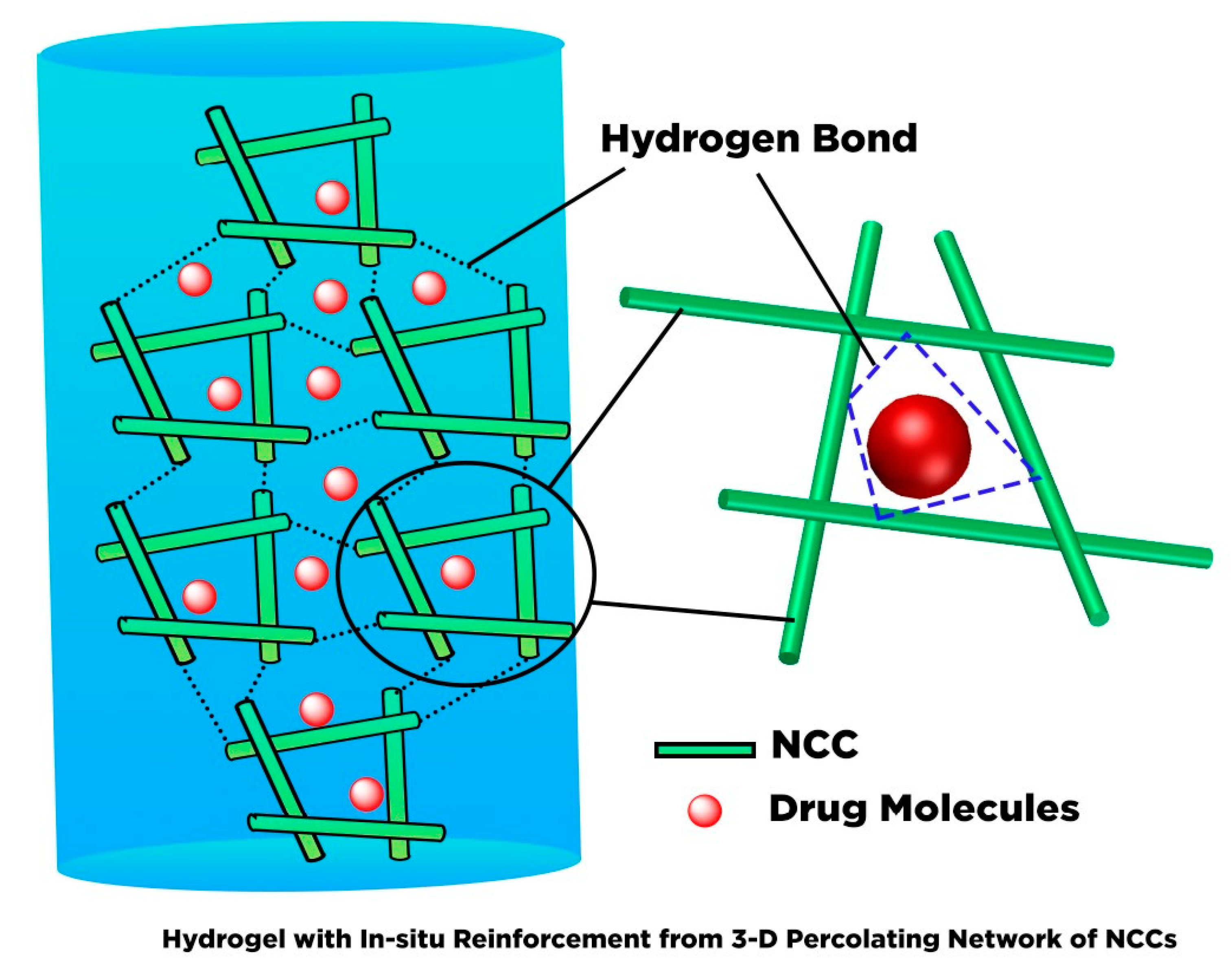
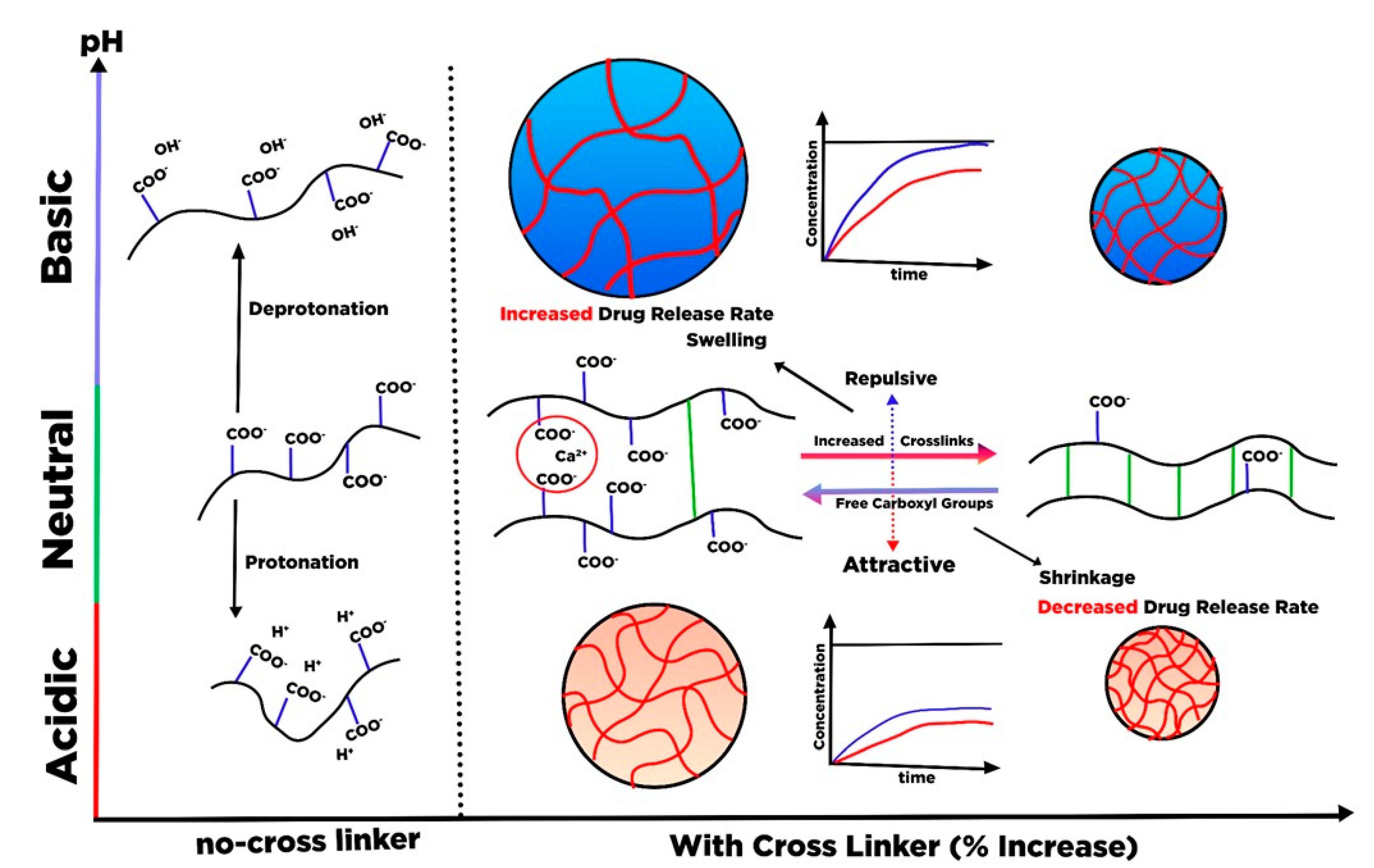
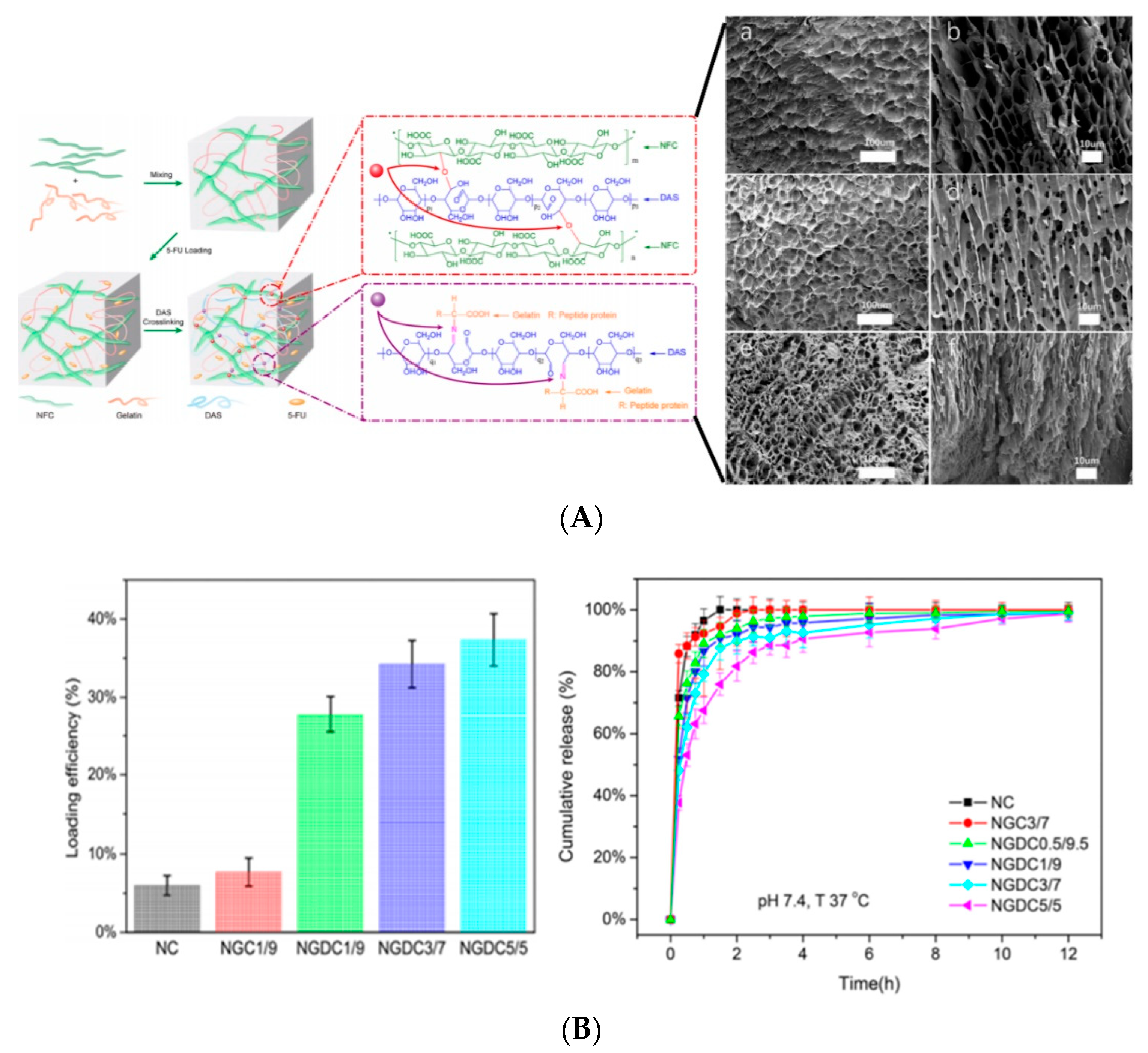
6. Hydrogel Based Nanocellulose for Drug Delivery
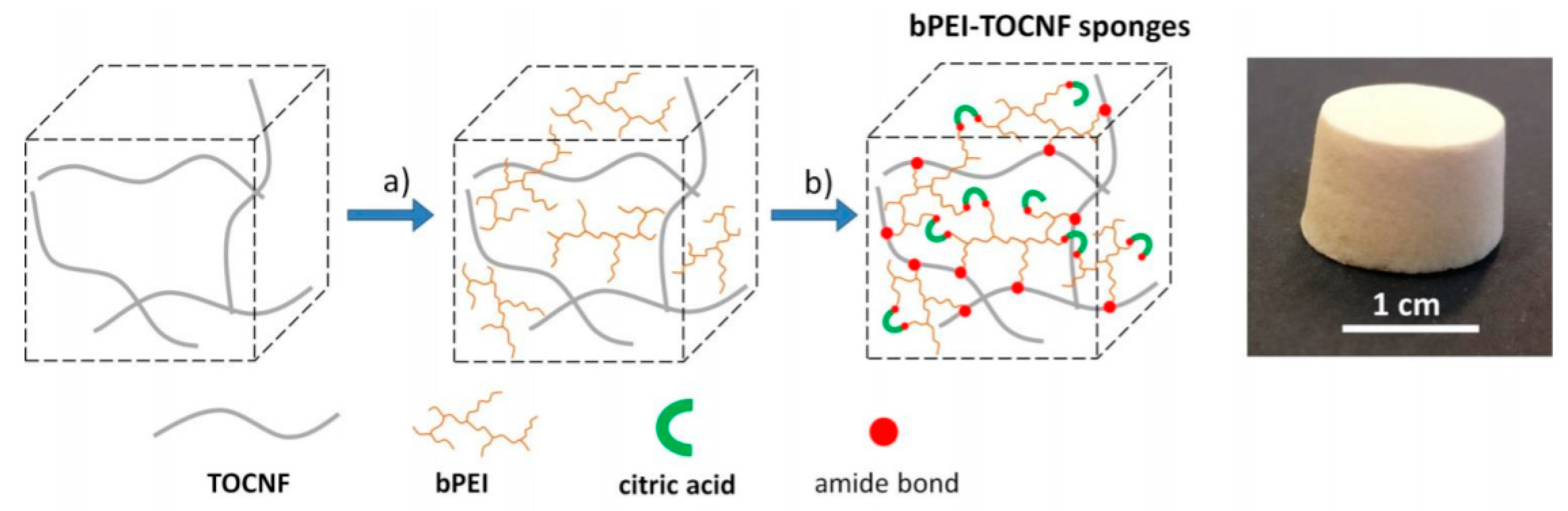
7. Lightweight Porous Based Nanocellulose for Drug Delivery
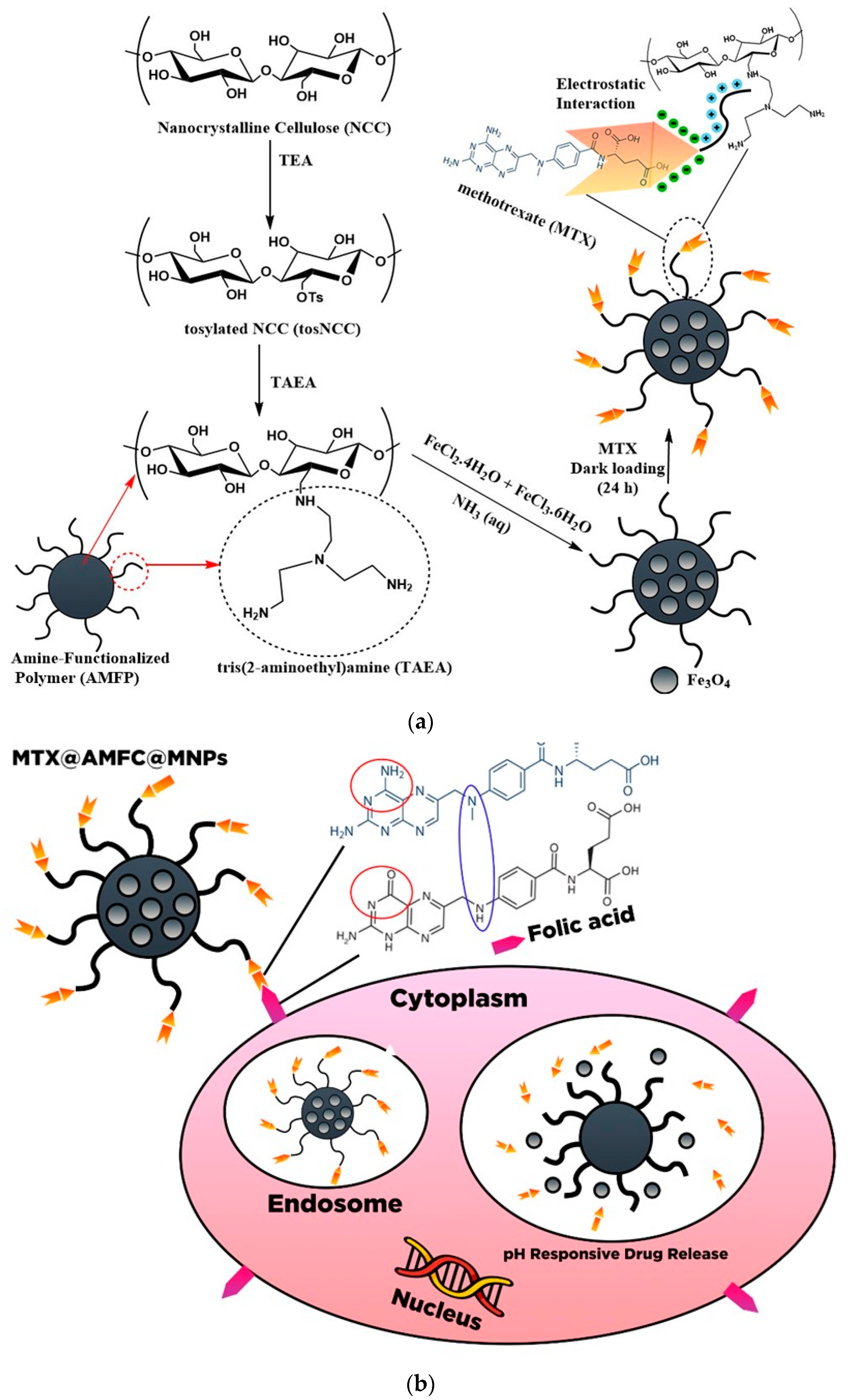
8. Integrated Inorganic/Organic-Based Nanocellulose for Drug Delivery
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Jain, K.K. An overview of drug delivery systems. In Drug Delivery Systems; Springer: New York, NY, USA, 2020. [Google Scholar]
- Sunasee, R.; Hemraz, U.D.; Ckless, K. Cellulose nanocrystals: A versatile nanoplatform for emerging biomedical applications. Expert Opin. Drug Deliv. 2016, 13, 1243–1256. [Google Scholar] [CrossRef] [PubMed]
- Bamrungsap, S.; Zhao, Z.; Chen, T.; Wand, L.; Li, C.; Fu, T.; Tan, W. Nanotechnology in therapeutics: A focus on nanoparticles as a drug delivery system. Nanomedicine 2012, 7, 1253–1271. [Google Scholar] [CrossRef]
- Tong, X.; Pan, W.; Su, T.; Zhang, M.; Dong, W.; Qi, X. Recent advances in natural polymer-based drug delivery systems. React. Funct. Polym. 2020, 148, 104501. [Google Scholar] [CrossRef]
- Cavallaro, G.; Micciulla, S.; Chiappisi, L.; Lazzara, G. Chitosan-based smart hybrid materials: A physico-chemical perspective. J. Mater. Chem. B 2021, 9, 594–611. [Google Scholar] [CrossRef]
- Bertolino, V.; Cavallaro, G.; Milioto, S.; Lazzara, G. Polysaccharides/Halloysite nanotubes for smart bionanocomposite materials. Carbohydr. Polym. 2020, 245, 116502. [Google Scholar] [CrossRef]
- Ahmad, A.; Mubarak, N.; Jannat, F.T.; Ashfaq, T.; Santulli, C.; Rizwan, M.; Najda, A.; Bin-Jumah, M.; Abdel-Daim, M.M.; Hussain, S. A Critical Review on the Synthesis of Natural Sodium Alginate Based Composite Materials: An Innovative Biological Polymer for Biomedical Delivery Applications. Processes 2021, 9, 137. [Google Scholar] [CrossRef]
- Chen, J.; Ouyang, J.; Chen, Q.; Deng, C.; Meng, F.; Zhang, J.; Cheng, R.; Lan, Q.; Zhong, Z. EGFR and CD44 dual-targeted multifunctional hyaluronic acid nanogels boost protein delivery to ovarian and breast cancers in vitro and in vivo. ACS Appl. Mater. Interfaces 2017, 9, 24140–24147. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Wei, W.; Shen, J.; Dong, W. Salecan polysaccharide-based hydrogels and their applications: A review. J. Mater. Chem. B 2019, 7, 2577–2587. [Google Scholar] [CrossRef] [PubMed]
- Dragan, E.S.; Dinu, M.V. Polysaccharides constructed hydrogels as vehicles for proteins and peptides. A review. Carbohydr. Polym. 2019, 225, 115210. [Google Scholar] [CrossRef] [PubMed]
- Jorfi, M.; Foster, E.J. Recent advances in nanocellulose for biomedical applications. J. Appl. Polym. Sci. 2015, 132, 41719. [Google Scholar] [CrossRef]
- Xue, Y.; Mou, Z.; Xiao, H. Nanocellulose as a sustainable biomass material: Structure, properties, present status and future prospects in biomedical applications. Nanoscale 2017, 9, 14758–14781. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef]
- Lin, N.; Dufresne, A. Nanocellulose in biomedicine: Current status and future prospect. Eur. Polym. J. 2014, 59, 302–325. [Google Scholar] [CrossRef] [Green Version]
- Plackett, D.; Letchford, K.; Jackson, J.; Burt, H. A review of nanocellulose as a novel vehicle for drug delivery. Nord. Pulp Pap. Res. J. 2014, 29, 105–118. [Google Scholar] [CrossRef]
- Jawaid, M.; Mohammad, F. Nanocellulose and Nanohydrogel Matrices: Biotechnological and Biomedical Applications; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Hasan, N.; Rahman, L.; Kim, S.-H.; Cao, J.; Arjuna, A.; Lallo, S.; Jhun, B.H.; Yoo, J.-W. Recent advances of nanocellulose in drug delivery systems. J. Pharm. Investig. 2020, 50, 553–572. [Google Scholar] [CrossRef]
- Grishkewich, N.; Mohammed, N.; Tang, J.; Tam, K.C. Recent advances in the application of cellulose nanocrystals. Curr. Opin. Colloid Interface Sci. 2017, 29, 32–45. [Google Scholar] [CrossRef]
- Yan, G.; Chen, B.; Zeng, X.; Sun, Y.; Tan, X.; Lin, L. Recent advances on sustainable cellulosic materials for pharmaceutical carrier applications. Carbohydr. Polym. 2020, 244, 116492. [Google Scholar] [CrossRef] [PubMed]
- Rajinipriya, M.; Nagalakshmaiah, M.; Robert, M.; Elkoun, S. Importance of agricultural and industrial waste in the field of nanocellulose and recent industrial developments of wood based nanocellulose: A review. ACS Sustain. Chem. Eng. 2018, 6, 2807–2828. [Google Scholar] [CrossRef]
- Karimian, A.; Parsian, H.; Majidinia, M.; Rahimi, M.; Mir, S.M.; Kafil, H.S.; Shafiei-Irannejad, V.; Kheyrollah, M.; Ostadi, H.; Yousefi, B. Nanocrystalline cellulose: Preparation, physicochemical properties, and applications in drug delivery systems. Int. J. Biol. Macromol. 2019, 133, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Salimi, S.; Sotudeh-Gharebagh, R.; Zarghami, R.; Chan, S.Y.; Yuen, K.H. Production of nanocellulose and its applications in drug delivery: A critical review. ACS Sustain. Chem. Eng. 2019, 7, 15800–15827. [Google Scholar] [CrossRef]
- Seabra, A.B.; Bernandes, J.S.; Favaro, W.J.; Paula, A.J.; Duran, N. Cellulose nanocrystals as carriers in medicine and their toxicities: A review. Carbohydr. Polym. 2018, 181, 514–527. [Google Scholar] [CrossRef] [PubMed]
- Sheikhi, A.; Hayashi, J.; Eichenbaum, J.; Gutin, M.; Kuntjoro, N.; Khorsandi, D.; Khademhosseini, A. Recent advances in nanoengineering cellulose for cargo delivery. J. Control. Release 2019, 294, 53–76. [Google Scholar] [CrossRef] [PubMed]
- Putro, J.N.; Ismadji, S.; Gunarto, C.; Yuliana, M.; Santoso, S.P.; Soetaredjo, F.E.; Ju, Y.H. The effect of surfactants modification on nanocrystalline cellulose for paclitaxel loading and release study. J. Mol. Liq. 2019, 282, 407–414. [Google Scholar] [CrossRef]
- Bundjaja, V.; Sari, T.M.; Soetaredjo, F.E.; Yuliana, M.; Angkawijaya, A.E.; Ismadji, S.; Cheng, K.-C.; Santoso, S.P. Aqueous sorption of tetracycline using rarasaponin-modified nanocrystalline cellulose. J. Mol. Liq. 2020, 301, 112433. [Google Scholar] [CrossRef]
- Huang, Y.; Zhu, C.; Yang, J.; Nie, Y.; Chen, C.; Sun, D. Recent advances in bacterial cellulose. Cellulose 2014, 21, 1–30. [Google Scholar] [CrossRef]
- Foo, M.L.; Tan, C.R.; Lim, P.D.; Ooi, C.W.; Tan, K.W.; Chew, I.M.L. Surface-modified nanocrystalline cellulose from oil palm empty fruit bunch for effective binding of curcumin. Int. J. Biol. Macromol. 2019, 138, 1064–1071. [Google Scholar] [CrossRef]
- Wijaya, C.J.; Saputra, S.N.; Soetaredjo, F.E.; Putro, J.N.; Lin, C.X.; Kurniawan, A.; Ju, Y.-H.; Ismadji, S. Cellulose nanocrystals from passion fruit peels waste as antibiotic drug carrier. Carbohydr. Polym. 2017, 175, 370–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dufresne, A. Cellulose nanomaterial reinforced polymer nanocomposites. Curr. Opin. Colloid Interface Sci. 2017, 29, 1–8. [Google Scholar] [CrossRef]
- Gopinath, V.; Saravanan, S.; Al-Maleki, A.; Ramesh, M.; Vadivelu, J. A review of natural polysaccharides for drug delivery applications: Special focus on cellulose, starch and glycogen. Biomed. Pharmacother. 2018, 107, 96–108. [Google Scholar] [CrossRef]
- Wertz, J.-L.; Bedue, O.; Mercier, J.P. Cellulose Science and Technology; EPFL Press: Lausanne, Switzerland, 2010. [Google Scholar]
- George, J.; Sabapathi, S. Cellulose nanocrystals: Synthesis, functional properties, and applications. Nanotechnol. Sci. Appl. 2015, 8, 45. [Google Scholar] [CrossRef] [Green Version]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose nanomaterials review: Structure, properties and nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994. [Google Scholar] [CrossRef]
- Medronho, B.; Lindman, B. Brief overview on cellulose dissolution/regeneration interactions and mechanisms. Adv. Colloid Interface Sci. 2015, 222, 502–508. [Google Scholar] [CrossRef]
- Kamel, R.; El-Wakil, N.A.; Dufresne, A.; Elkasabgy, N.A. Nanocellulose: From an agricultural waste to a valuable pharmaceutical ingredient. Int. J. Biol. Macromol. 2020, 163, 1579–1590. [Google Scholar] [CrossRef] [PubMed]
- Miao, C.; Hamad, W.Y. Cellulose reinforced polymer composites and nanocomposites: A critical review. Cellulose 2013, 20, 2221–2262. [Google Scholar] [CrossRef]
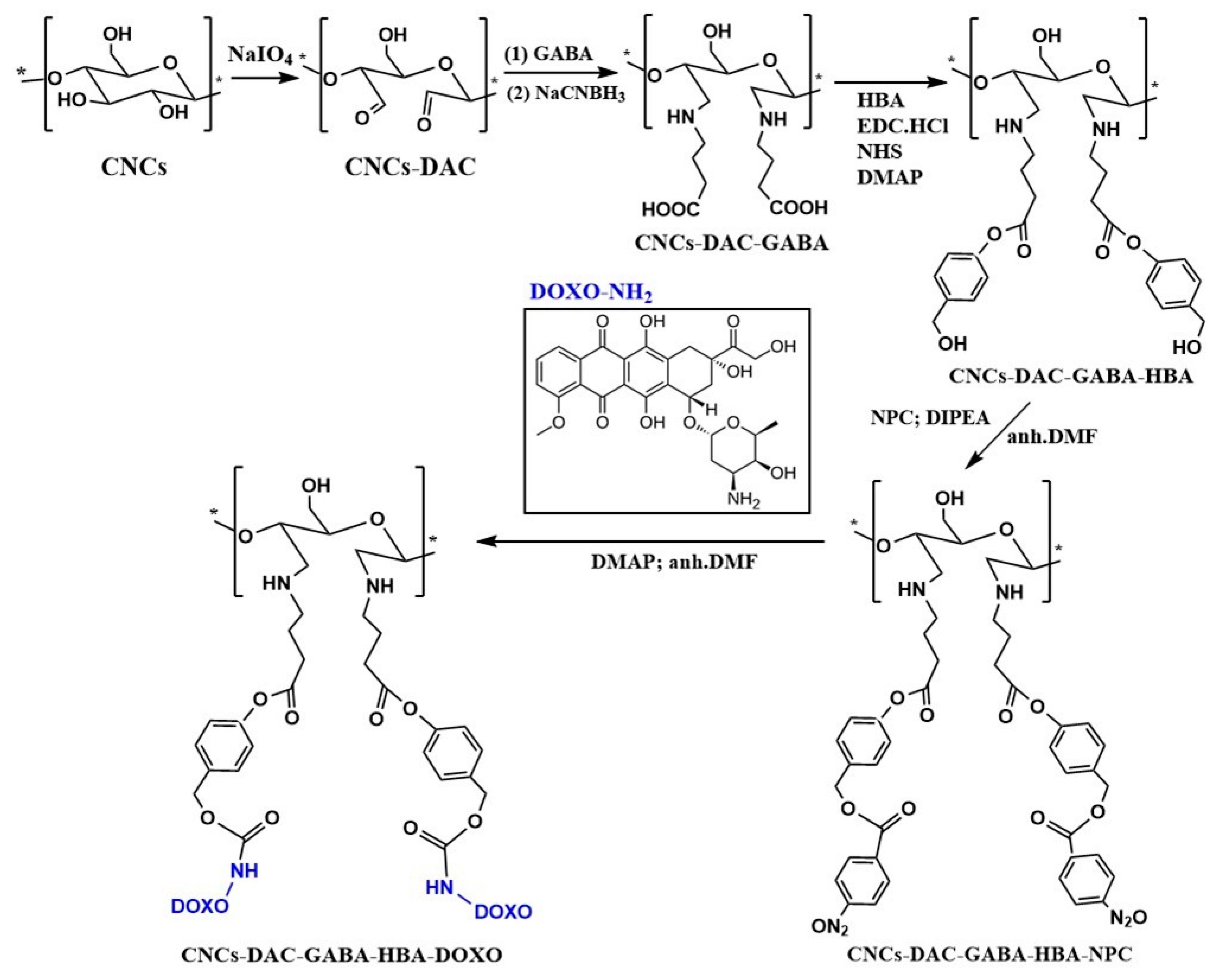
- Li, N.; Lu, W.; Yu, J.; Xiao, Y.; Liu, S.; Gan, L.; Huang, J. Rod-like cellulose nanocrystal/cis-aconityl-doxorubicin prodrug: A fluorescence-visible drug delivery system with enhanced cellular uptake and intracellular drug controlled release. Mater. Sci. Eng. C 2018, 91, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Jia, B.; Li, Y.; Yang, B.; Xiao, D.; Zhang, S.; Rajulu, A.V.; Kondo, T.; Zhang, L.; Zhou, J. Effect of microcrystal cellulose and cellulose whisker on biocompatibility of cellulose-based electrospun scaffolds. Cellulose 2013, 20, 1911–1923. [Google Scholar] [CrossRef]
- Dash, R.; Ragauskas, A.J. Synthesis of a novel cellulose nanowhisker-based drug delivery system. RSC Adv. 2012, 2, 3403–3409. [Google Scholar] [CrossRef]
- Biswas, S.K.; Sano, H.; Yang, X.; Tanphichai, S.; Shams, M.I.; Yano, H. Highly thermal-resilient AgNW transparent electrode and optical device on thermomechanically superstable cellulose nanorod-reinforced nanocomposites. Adv. Opt. Mater. 2019, 7, 1900532. [Google Scholar] [CrossRef]
- Wang, N.; Ding, E.; Cheng, R. Thermal degradation behaviors of spherical cellulose nanocrystals with sulfate groups. Polymer 2007, 48, 3486–3493. [Google Scholar] [CrossRef]
- Ram, B.; Chauhan, G.S.; Mehta, A.; Gupta, R.; Chauhan, K. Spherical nanocellulose-based highly efficient and rapid multifunctional naked-eye Cr (VI) ion chemosensor and adsorbent with mild antimicrobial properties. Chem. Eng. J. 2018, 349, 146–155. [Google Scholar] [CrossRef]
- Mariano, M.; El Kissi, N.; Dufresne, A. Cellulose nanocrystals and related nanocomposites: Review of some properties and challenges. J. Polym. Sci. B Polym. Phys. 2014, 52, 791–806. [Google Scholar] [CrossRef]
- Brinchi, L.; Cotana, F.; Fortunati, E.; Kenny, J. Production of nanocrystalline cellulose from lignocellulosic biomass: Technology and applications. Carbohydr. Polym. 2013, 94, 154–169. [Google Scholar] [CrossRef]
- Phanthong, P.; Reybroycharoen, P.; Hao, X.; Xu, G.; Abudula, A.; Guan, G. Nanocellulose: Extraction and application. Carbon Resour. Convers. 2018, 1, 32–43. [Google Scholar] [CrossRef]
- Lam, E.; Male, K.B.; Chong, J.H.; Leung, A.C.; Luong, J.H. Applications of functionalized and nanoparticle-modified nanocrystalline cellulose. Trends Biotechnol. 2012, 30, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Prathapan, R.; Tabor, R.F.; Garnier, G.; Hu, J. Recent progress in cellulose nanocrystal alignment and its applications. ACS Appl. Bio Mater. 2020, 3, 1828–1844. [Google Scholar] [CrossRef]
- Abitbol, T.; Rivkin, A.; Cao, Y.; Nevo, Y.; Abraham, E.; Ben-Shalom, T.; Lapidot, S.; Shoseyov, O. Nanocellulose, a tiny fiber with huge applications. Curr. Opin. Biotechnol. 2016, 39, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, M.; Henriksson, G.; Berglund, L.; Lindström, T. An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur. Polym. J. 2007, 43, 3434–3441. [Google Scholar] [CrossRef]
- Kim, C.-W.; Kim, D.-S.; Kang, S.-Y.; Marquez, M.; Joo, Y.L. Structural studies of electrospun cellulose nanofibers. Polymer 2006, 47, 5097–5107. [Google Scholar] [CrossRef]
- Dufresne, A. Nanocellulose: A new ageless bionanomaterial. Mater. Today 2013, 16, 220–227. [Google Scholar] [CrossRef]
- Lavoine, N.; Desloges, I.; Dufresne, A.; Bras, J. Microfibrillated cellulose–Its barrier properties and applications in cellulosic materials: A review. Carbohydr. Polym. 2012, 90, 735–764. [Google Scholar] [CrossRef]
- Blanco Parte, F.G.; Santoso, S.P.; Chou, C.-C.; Verma, V.; Wang, H.-T.; Ismadji, S.; Cheng, K.-C. Current progress on the production, modification, and applications of bacterial cellulose. Crit. Rev. Biotechnol. 2020, 40, 397–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siro, I.; Plackett, D. Microfibrillated cellulose and new nanocomposite materials: A review. Cellulose 2010, 17, 459–494. [Google Scholar] [CrossRef]
- Huang, J.; Dufresne, A.; Lin, N. Nanocellulose: From Fundamentals to Advanced Materials; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Jozala, A.F.; De Lencastre-Novaes, L.C.; Lopes, A.M.; De Carvalho Santos-Ebinuma, V.; Mazzola, P.G.; Pessoa-JR, A.; Grotto, D.; Gerenutti, M.; Chaud, M.V. Bacterial nanocellulose production and application: A 10-year overview. Appl. Microbiol. Biotechnol. 2016, 100, 2063–2072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, B.; Yang, G.; Hong, F. Preparation and evaluation of a kind of bacterial cellulose dry films with antibacterial properties. Carbohydr. Polym. 2011, 84, 533–538. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, J.; Gama, M.; Guo, R.; Zhang, Q.; Zhang, P.; Yao, F.; Luo, H. Biofabrication of a novel bacteria/bacterial cellulose composite for improved adsorption capacity. Compos. Part A Appl. Sci. Manuf. 2019, 125, 105560. [Google Scholar] [CrossRef] [Green Version]
- Gorgieva, S.; Trcek, J. Bacterial cellulose: Production, modification and perspectives in biomedical applications. Nanomaterials 2019, 9, 1352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.-H.; Shim, B.S.; Kim, H.S.; Lee, Y.-J.; Min, S.-K.; Jang, D.; Abas, Z.; Kim, J. Review of nanocellulose for sustainable future materials. Int. J. Precis. Eng. Manuf. Green Technol. 2015, 2, 197–213. [Google Scholar] [CrossRef] [Green Version]
- Dufresne, A. Cellulose-based composites and nanocomposites. In Monomers, Polymers, and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Song, F.; Li, X.; Wang, Q.; Liao, L.; Zhang, C. Nanocomposite hydrogels and their applications in drug delivery and tissue engineering. J. Biomed. Nanotechnol. 2015, 11, 40–52. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Fahmy, T.Y.; Salaheldin, E.I.; Mobarak, F.; Youssef, M.A.; Mabrook, M.R. Synthesis of tosylated and trimethylsilylated methyl cellulose as pH-sensitive carrier matrix. Life Sci. J. 2015, 1, 29–37. [Google Scholar]
- Ximenes, F.A.; Gardner, W.D.; Kathuria, A. Proportion of above-ground biomass in commercial logs and residues following the harvest of five commercial forest species in Australia. For. Ecol. Manag. 2008, 256, 335–346. [Google Scholar] [CrossRef]
- Kargarzadeh, H.; Ioelovich, M.; Ahmad, I.; Thomas, S.; Dufresne, A. Methods for extraction of nanocellulose from various sources. Handb. Nanocellulose Cellul. Nanocomposites 2017, 2, 1–49. [Google Scholar]
- Kumar, A.K.; Sharma, S. Recent updates on different methods of pretreatment of lignocellulosic feedstocks: A review. Bioresour. Bioproces. 2017, 4, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Ravindran, R.; Jaiswal, S.; Abu-Ghannam, N.; Jaiswal, A.K. A comparative analysis of pretreatment strategies on the properties and hydrolysis of brewers’ spent grain. Bioresour. Technol. 2018, 248, 272–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pires, J.R.; De Souza, V.G.L.; Fernando, A.L. Production of nanocellulose from lignocellulosic biomass wastes: Prospects and limitations. In Proceedings of the International Conference on Innovation, Engineering and Entrepreneurship, Guimaraes, Portugal, 27–29 June 2018; pp. 719–725. [Google Scholar]
- Shirkavand, E.; Baroutian, S.; Gapes, D.J.; Young, B.R. Combination of fungal and physicochemical processes for lignocellulosic biomass pretreatment–A review. Renew. Sust. Energy Rev. 2016, 54, 217–234. [Google Scholar] [CrossRef]
- Baramee, S.; Siriatcharanon, A.-K.; Ketbot, P.; Teeravivattanakit, T.; Waeonukul, R.; Pason, P.; Tachaapaikoon, C.; Ratanakhanokchai, K.; Phitsuwan, P. Biological pretreatment of rice straw with cellulase-free xylanolytic enzyme-producing Bacillus firmus K-1: Structural modification and biomass digestibility. Renew. Energy 2020, 160, 555–563. [Google Scholar] [CrossRef]
- Khalil, H.A.; Davoudpour, Y.; Islam, M.N.; Mustapha, A.; Sudesh, K.; Dungani, R.; Jawaid, M. Production and modification of nanofibrillated cellulose using various mechanical processes: A review. Carbohydr. Polym. 2014, 99, 649–665. [Google Scholar] [CrossRef]
- Mood, S.H.; Golfeshan, A.H.; Tabatabaei, M.; Jouzani, G.S.; Najafi, G.H.; Gholami, M.; Ardjmand, M. Lignocellulosic biomass to bioethanol, a comprehensive review with a focus on pretreatment. Renew. Sust. Energy Rev. 2013, 27, 77–93. [Google Scholar] [CrossRef]
- Hassan, S.S.; Williams, G.A.; Jaiswal, A.K. Emerging technologies for the pretreatment of lignocellulosic biomass. Bioresour. Technol. 2018, 262, 310–318. [Google Scholar] [CrossRef] [Green Version]
- De Carvalho Benini, K.C.C.; Voorwald, H.J.C.; Cioffi, M.O.H.; Rezende, M.C.; Arantes, V. Preparation of nanocellulose from Imperata brasiliensis grass using Taguchi method. Carbohydr. Polym. 2018, 192, 337–346. [Google Scholar] [CrossRef]
- Maciel, M.M.Á.D.; De Carvalho Benini, K.C.C.; Voorwald, H.J.C.; Cioffi, M.O.H. Obtainment and characterization of nanocellulose from an unwoven industrial textile cotton waste: Effect of acid hydrolysis conditions. Int. J. Biol. Macromol. 2019, 126, 496–506. [Google Scholar] [CrossRef]
- Liu, J.; Korpinen, R.; Mikkonen, K.S.; Willför, S.; Xu, C. Nanofibrillated cellulose originated from birch sawdust after sequential extractions: A promising polymeric material from waste to films. Cellulose 2014, 21, 2587–2598. [Google Scholar] [CrossRef]
- Couret, L.; Irle, M.; Belloncle, C.; Cathala, B. Extraction and characterization of cellulose nanocrystals from post-consumer wood fiberboard waste. Cellulose 2017, 24, 2125–2137. [Google Scholar] [CrossRef]
- Vallejos, M.E.; Felissia, F.E.; Area, M.C.; Ehman, N.V.; Tarrés, Q.; Mutjé, P. Nanofibrillated cellulose (CNF) from eucalyptus sawdust as a dry strength agent of unrefined eucalyptus handsheets. Carbohydr. Polym. 2016, 139, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Rambabu, N.; Panthapulakkal, S.; Sain, M.; Dalai, A. 2016. Production of nanocellulose fibers from pinecone biomass: Evaluation and optimization of chemical and mechanical treatment conditions on mechanical properties of nanocellulose films. Ind. Crops Prod. 2016, 83, 746–754. [Google Scholar] [CrossRef]
- Moriana, R.; Vilaplana, F.; Ek, M. Cellulose nanocrystals from forest residues as reinforcing agents for composites: A study from macro-to nano-dimensions. Carbohydr. Polym. 2016, 139, 139–149. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, L.; Liu, C.; He, Z.; Zhou, X.; Ni, Y. A novel method to prepare lignocellulose nanofibrils directly from bamboo chips. Cellulose 2018, 25, 7043–7051. [Google Scholar] [CrossRef]
- Hua, K.; Strømme, M.; Mihranyan, A.; Ferraz, N. Nanocellulose from green algae modulates the in vitro inflammatory response of monocytes/macrophages. Cellulose 2015, 22, 3673–3688. [Google Scholar] [CrossRef]
- Son, H.N.; Seo, Y.B. Physical and bio-composite properties of nanocrystalline cellulose from wood, cotton liners, cattail, and red algae. Cellulose 2015, 22, 1789–1798. [Google Scholar]
- Rathod, M.; Haldar, S.; Basha, S. Nanocrystalline cellulose for removal of tetracycline hydrochloride from water via biosorption: Equilibrium, kinetic and thermodynamic studies. Ecol. Eng. 2015, 84, 240–249. [Google Scholar] [CrossRef]
- Liu, Z.; Li, X.; Xie, W.; Deng, H. Extraction, isolation and characterization of nanocrystalline cellulose from industrial kelp (Laminaria japonica) waste. Carbohydr. Polym. 2017, 173, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Meng, X.; Zhao, J.; Miao, M.; Shi, L.; Zhang, S.; Fang, J. Extraction and preparation of cellulose nanocrystals from dealginate kelp residue: Structures and morphological characterization. Cellulose 2015, 22, 1763–1772. [Google Scholar] [CrossRef]
- Bhutiya, P.L.; Misra, N.; Rasheed, M.A.; Hasan, S.Z. Nested seaweed cellulose fiber deposited with cuprous oxide nanorods for antimicrobial activity. Int. J. Biol. Macromol. 2018, 117, 435–444. [Google Scholar] [CrossRef]
- De Oliveira, J.P.; Bruni, G.P.; Fabra, M.J.; Da Rosa Zavareze, E.; López-Rubio, A.; Martínez-Sanz, M. Development of food packaging bioactive aerogels through the valorization of Gelidium sesquipedale seaweed. Food Hydrocoll. 2019, 89, 337–350. [Google Scholar] [CrossRef]
- Chen, Y.W.; Lee, H.V.; Juan, J.C.; Phang, S.-M. Production of new cellulose nanomaterial from red algae marine biomass Gelidium elegans. Carbohydr. Polym. 2016, 151, 1210–1219. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Chakrabarty, D. Isolation of nanocellulose from waste sugarcane bagasse (SCB) and its characterization. Carbohydr. Polym. 2011, 86, 1291–1299. [Google Scholar] [CrossRef]
- Thomas, M.G.; Abraham, E.; Jyotishkumar, P.; Maria, H.J.; Pothen, L.A.; Thomas, S. Nanocelluloses from jute fibers and their nanocomposites with natural rubber: Preparation and characterization. Int. J. Biol. Macromol. 2015, 81, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Du, X.; Yin, Z.; Xu, S.; Xu, S.; Zhang, Y. Preparation and characterization of cellulose nanofibrils from coconut coir fibers and their reinforcements in biodegradable composite films. Carbohydr. Polym. 2019, 211, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Mariño, M.; Lopes Da Silva, L.; Durán, N.; Tasic, L. Enhanced materials from nature: Nanocellulose from citrus waste. Molecules 2015, 20, 5908–5923. [Google Scholar] [CrossRef]
- Dilamian, M.; Noroozi, B. A combined homogenization-high intensity ultrasonication process for individualizaion of cellulose micro-nano fibers from rice straw. Cellulose 2019, 26, 5831–5849. [Google Scholar] [CrossRef]
- Kang, X.; Sun, P.; Kuga, S.; Wang, C.; Zhao, Y.; Wu, M.; Huang, Y. Thin cellulose nanofiber from corncob cellulose and its performance in transparent nanopaper. ACS Sust. Chem. Eng. 2017, 5, 2529–2534. [Google Scholar] [CrossRef]
- Karimi, S.; Tahir, P.M.; Karimi, A.; Dufresne, A.; Abdulkhani, A. Kenaf bast cellulosic fibers hierarchy: A comprehensive approach from micro to nano. Carbohydr. Polym. 2014, 101, 878–885. [Google Scholar] [CrossRef]
- Jodeh, S.; Hamed, O.; Melhem, A.; Salghi, R.; Jodeh, D.; Azzaoui, K.; Benmassaoud, Y.; Murtada, K. Magnetic nanocellulose from olive industry solid waste for the effective removal of methylene blue from wastewater. Environ. Sci. Poll. Res. 2018, 25, 22060–22074. [Google Scholar] [CrossRef]
- Jongaroontaprangsee, S.; Chiewchan, N.; Devahastin, S. Production of nanofibrillated cellulose with superior water redispersibility from lime residues via a chemical-free process. Carbohydr. Polym. 2018, 193, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Diop, C.I.K.; Lavoie, J.-M. Isolation of nanocrystalline cellulose: A technological route for valorizing recycled tetra pak aseptic multilayered food packaging wastes. Waste Biomass Valor. 2017, 8, 41–56. [Google Scholar] [CrossRef]
- Putro, J.N.; Santoso, S.P.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.-H. Nanocrystalline cellulose from waste paper: Adsorbent for azo dyes removal. Environ. Nanotech. Monit. Manag. 2019, 12, 100260. [Google Scholar] [CrossRef]
- Ogundare, S.A.; Moodley, V.; Van Zyl, W.E. Nanocrystalline cellulose isolated from discarded cigarette filters. Carbohydr. Polym. 2017, 175, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Peretz, R.; Sterenzon, E.; Gerchman, Y.; Vadivel, V.K.; Luxbacher, T.; Mamane, H. Nanocellulose production from recycled paper mill sludge using ozonation pretreatment followed by recyclable maleic acid hydrolysis. Carbohydr. Polym. 2019, 216, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Cypriano, D.Z.; Da Silva, L.L.; Tasic, L. High value-added products from the orange juice industry waste. Waste Manag. 2018, 79, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.; Singh, J.; Singh, R. Biotransformation of sweet lime pulp waste into high-quality nanocellulose with an excellent productivity using Komagataeibacter europaeus SGP37 under static intermittent fed-batch cultivation. Bioresour. Technol. 2018, 247, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S. Preparation, properties and applications of nanocellulosic materials. Carbohydr. Polym. 2017, 163, 301–316. [Google Scholar] [CrossRef]
- Turbak, A.F.; Snyder, F.W.; Sandberg, K.R. Microfibrillated cellulose, a new cellulose product: Properties, uses, and commercial potential. J. Appl. Polym. Sci. Appl. Polym. Sym. 1983, 37, 815–827. [Google Scholar]
- Herrick, F.W.; Casebier, R.L.; Hamilton, J.K.; Sandberg, K.R. Microfibrillated cellulose: Morphology and accessibility. J. Appl. Polym. Sci. Appl. Polym. Sym. 1983, 37, 797–813. [Google Scholar]
- Kawee, N.; Lam, N.T.; Sukyai, P. Homogenous isolation of individualized bacterial nanofibrillated cellulose by high pressure homogenization. Carbohydr. Polym. 2018, 179, 394–401. [Google Scholar] [CrossRef]
- Yusra, A.I.; Juahir, H.; Firdaus, N.N.A.; Bhat, A.; Endut, A.; Khalil, H.A.; Adiana, G. Controlling of green nanocellulose fiber properties produced by chemo-mechanical treatment process via SEM, TEM, AFM and image analyzer characterization. J. Fund. Appl. Sci. 2018, 10, 1–17. [Google Scholar]
- Kalia, S.; Boufi, S.; Celli, A.; Kango, S. Nanofibrillated cellulose: Surface modification and potential applications. Colloid Polym. Sci. 2014, 292, 5–31. [Google Scholar] [CrossRef]
- Niu, F.; Li, M.; Huang, Q.; Zhang, X.; Pan, W.; Yang, J.; Li, J. The characteristic and dispersion stability of nanocellulose produced by mixed acid hydrolysis and ultrasonic assistance. Carbohydr. Polym. 2017, 165, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Trache, D.; Hussin, M.H.; Haafiz, M.M.; Thakur, V.K. Recent progress in cellulose nanocrystals: Sources and production. Nanoscale 2017, 9, 1763–1786. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Tavakoli, J.; Tang, Y. Bacterial cellulose production, properties and applications with different culture methods–A review. Carbohydr. Polym. 2019, 219, 63–76. [Google Scholar] [CrossRef] [Green Version]
- Czaja, W.; Romanovicz, D.; Malcolm, B.R. Structural investigations of microbial cellulose produced in stationary and agitated culture. Cellulose 2004, 11, 403–411. [Google Scholar] [CrossRef]
- Matsutani, M.; Ito, K.; Azuma, Y.; Ogino, H.; Shirai, M.; Yakushi, T.; Matsushita, K. Adaptive mutation related to cellulose producibility in Komagataeibacter medellinensis (Gluconacetobacter xylinus) NBRC 3288. Appl. Microbiol. Biotechnol. 2015, 99, 7229–7240. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhong, C.; Wu, X.-Y.; Wei, Y.-Q.; Bo, T.; Han, P.-P.; Jia, S.-R. Metabolomic profiling coupled with metabolic network reveals differences in Gluconacetobacter xylinus from static and agitated cultures. Biochem. Eng. J. 2015, 101, 85–98. [Google Scholar] [CrossRef]
- Gullo, M.; La China, S.; Petroni, G.; Di Gregorio, S.; Giudici, P. Exploring K2G30 genome: A high bacterial cellulose producing strain in glucose and mannitol based media. Front. Microbiol. 2019, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Thorat, M.N.; Dastager, S.G. High yield production of cellulose by a Komagataeibacter rhaeticus PG2 strain isolated from pomegranate as a new host. RSC Adv. 2018, 8, 29797–29805. [Google Scholar] [CrossRef] [Green Version]
- Volova, T.G.; Prudnikova, S.V.; Sukovatyi, A.G.; Shishatskaya, E.I. Production and properties of bacterial cellulose by the strain Komagataeibacter xylinus B-12068. App. Microbiol. Biotechnol. 2018, 102, 7417–7428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molina-Ramírez, C.; Castro, M.; Osorio, M.; Torres-Taborda, M.; Gómez, B.; Zuluaga, R.; Gómez, C.; Gañán, P.; Rojas, O.J.; Castro, C. Effect of different carbon sources on bacterial nanocellulose production and structure using the low pH resistant strain Komagataeibacter medellinensis. Materials 2017, 10, 639. [Google Scholar] [CrossRef]
- Mohammadkazemi, F.; Azin, M.; Ashori, A. Production of bacterial cellulose using different carbon sources and culture media. Carbohydr. Polym. 2015, 117, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2020, 20, 101–124. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.I.; Lammers, T.; Ashford, M.B.; Puri, S.; Strom, G.; Barry, S.T. Challenges and strategies in anti-cancer nanomedicine development: An industry perspective. Adv. Drug Deliv. Rev. 2017, 108, 25–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araki, J. Electrostatic or steric?–preparations and characterizations of well-dispersed systems containing rod-like nanowhiskers of crystalline polysaccharides. Soft Mater. 2013, 9, 4125–4141. [Google Scholar] [CrossRef]
- Kargarzadeh, H.; Mariano, M.; Gopakumar, D.; Ahmad, I.; Thomas, S.; Dufresne, A.; Huang, J.; Lin, N. Advances in cellulose nanomaterials. Cellulose 2018, 25, 2151–2189. [Google Scholar] [CrossRef]
- Hebeish, A. , Guthrie, T. The Chemistry and Technology of Cellulosic Copolymers; Springer Science & Business Media: Belin, Germany, 2012. [Google Scholar]
- De La Motte, H.; Hasani, M.; Brelid, H.; Westman, G. Molecular characterization of hydrolyzed cationized nanocrystalline cellulose, cotton cellulose and softwood kraft pulp using high resolution 1D and 2D NMR. Carbohydr. Polym. 2011, 85, 738–746. [Google Scholar] [CrossRef]
- Habibi, Y. Key advances in the chemical modification of nanocelluloses. Chem. Soc. Rev. 2014, 43, 1519–1542. [Google Scholar] [CrossRef]
- Pakharenko, V.; Pervaiz, M.; Pande, H.; Sain, M.; Sain, M. Chemical and physical techniques for surface modification of nanocellulose reinforcements. In Interface/Interphase in Polymer Nanocomposites; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 283–310. [Google Scholar]
- Meneguin, A.B.; da Silva Barud, H.; Sabio, R.M.; de Sousa, P.Z.; Manieri, K.F.; de Freitas, L.A.P.; Pacheco, G.; Alonso, J.D.; Chorilli, M. Spray-dried bacterial cellulose nanofibers: A new generation of pharmaceutical excipient intended for intestinal drug delivery. Carbohydr. Polym. 2020, 249, 116838. [Google Scholar] [CrossRef]
- Khine, Y.Y.; Stenzel, M.H. Surface modified cellulose nanomaterials: A source of non-spherical nanoparticles for drug delivery. Mater. Horiz. 2020, 7, 1727–1758. [Google Scholar] [CrossRef]
- Kusano, Y.; Madsen, B.; Berglund, L.; Oksman, K. Modification of cellulose nanofibre surfaces by He/NH 3 plasma at atmospheric pressure. Cellulose 2019, 26, 7185–7194. [Google Scholar] [CrossRef]
- Taheri, A.; Mohammadi, M. The use of cellulose nanocrystals for potential application in topical delivery of hydroquinone. Chem. Biol. Drug Design 2015, 86, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Camarero Espinosa, S.; Kuhnt, T.; Foster, E.J.; Weder, C. Isolation of thermally stable cellulose nanocrystals by phosphoric acid hydrolysis. Biomacromol. 2013, 14, 1223–1230. [Google Scholar] [CrossRef]
- Wang, N.; Ding, E.; Cheng, R. Preparation and liquid crystalline properties of spherical cellulose nanocrystals. Langmuir 2008, 24, 5–8. [Google Scholar] [CrossRef] [PubMed]
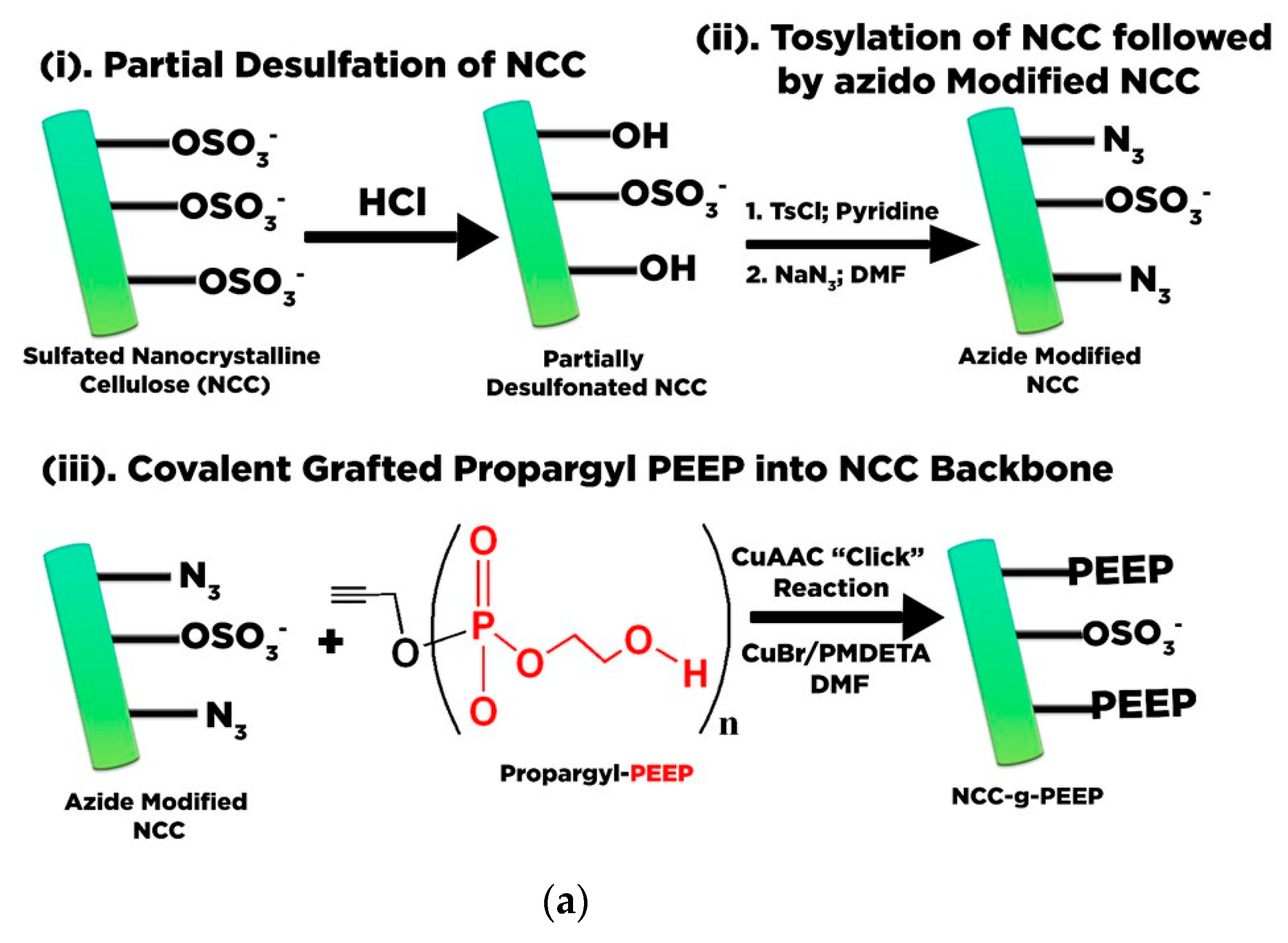
- Lin, N.; Dufresne, A. Surface chemistry, morphological analysis and properties of cellulose nanocrystals with gradiented sulfation degrees. Nanoscale 2014, 6, 5384–5393. [Google Scholar] [CrossRef]
- Kokol, V.; Božič, M.; Vogrinčič, R.; Mathew, A.P. Characterisation and properties of homo- and heterogeneously phosphorylated nanocellulose. Carbohydr. Polym. 2015, 125, 301–313. [Google Scholar] [CrossRef]
- Wang, H.; Xie, H.; Du, H.; Wang, X.; Liu, W.; Duan, Y.; Zhang, X.; Sun, L.; Zhang, X.; Si, C. Highly efficient preparation of functional and thermostable cellulose nanocrystals via H2SO4 intensified acetic acid hydrolysis. Carbohydr. Polym. 2020, 239, 116233. [Google Scholar] [CrossRef]
- Braun, B.; Dorgan, J.R. Single-step method for the isolation and surface functionalization of cellulosic nanowhiskers. Biomacromolecules 2009, 10, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Y.; Yu, H.-Y.; Zhang, C.-H.; Zhou, Y.; Yao, J.-M. A universal route for the simultaneous extraction and functionalization of cellulose nanocrystals from industrial and agricultural celluloses. J. Nanopart. Res. 2016, 18, 48. [Google Scholar] [CrossRef]
- Du, H.; Liu, C.; Mu, X.; Gong, W.; Lv, D.; Hong, Y.; Si, C.; Li, B. Preparation and characterization of thermally stable cellulose nanocrystals via a sustainable approach of FeCl 3-catalyzed formic acid hydrolysis. Cellulose 2016, 23, 2389–2407. [Google Scholar] [CrossRef]
- Spinella, S.; Re, G.L.; Liu, B.; Dorgan, J.; Habibi, Y.; Leclere, P.; Raquez, J.-M.; Dubois, P.; Gross, R.A. Polylactide/cellulose nanocrystal nanocomposites: Efficient routes for nanofiber modification and effects of nanofiber chemistry on PLA reinforcement. Polymer 2015, 65, 9–17. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, J.Y.; Baez, C.; Kitin, P.; Elder, T. Highly thermal-stable and functional cellulose nanocrystals and nanofibrils produced using fully recyclable organic acids. Green Chem. 2016, 18, 3835–3843. [Google Scholar] [CrossRef]
- Bian, H.; Chen, L.; Dai, H.; Zhu, J. Integrated production of lignin containing cellulose nanocrystals (LCNC) and nanofibrils (LCNF) using an easily recyclable di-carboxylic acid. Carbohydr. Polym. 2017, 167, 167–176. [Google Scholar] [CrossRef]
- Jia, W.; Liu, Y. Two characteristic cellulose nanocrystals (CNCs) obtained from oxalic acid and sulfuric acid processing. Cellulose 2019, 26, 8351–8365. [Google Scholar] [CrossRef]
- Luo, J.; Huang, K.; Xu, Y.; Fan, Y. A comparative study of lignocellulosic nanofibrils isolated from celery using oxalic acid hydrolysis followed by sonication and mechanical fibrillation. Cellulose 2019, 26, 5237–5246. [Google Scholar] [CrossRef]
- Spinella, S.; Maiorana, A.; Qian, Q.; Dawson, N.J.; Hepworth, V.; Mccallum, S.A.; Ganesh, M.; Singer, K.D.; Gross, R.A. 2016. Concurrent cellulose hydrolysis and esterification to prepare a surface-modified cellulose nanocrystal decorated with carboxylic acid moieties. ACS Sust. Chem. Eng. 2016, 4, 1538–1550. [Google Scholar] [CrossRef]
- Ji, H.; Xiang, Z.; Qi, H.; Han, T.; Pranovich, A.; Song, T. Strategy towards one-step preparation of carboxylic cellulose nanocrystals and nanofibrils with high yield, carboxylation and highly stable dispersibility using innocuous citric acid. Green Chem. 2019, 21, 1956–1964. [Google Scholar] [CrossRef]
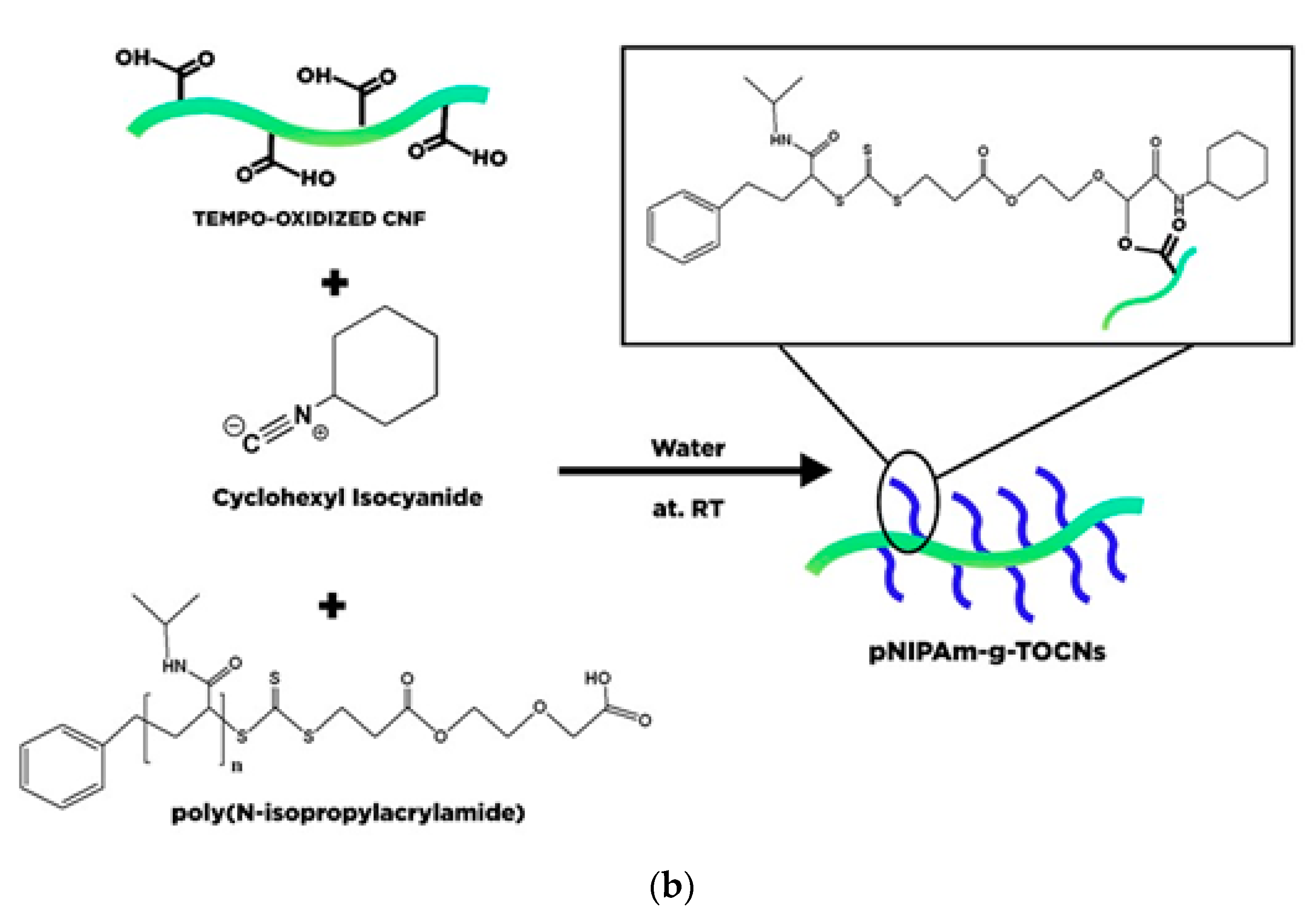
- Habibi, Y.; Chanzy, H.; Vignon, M.R. TEMPO-mediated surface oxidation of cellulose whiskers. Cellulose 2006, 13, 679–687. [Google Scholar] [CrossRef] [Green Version]
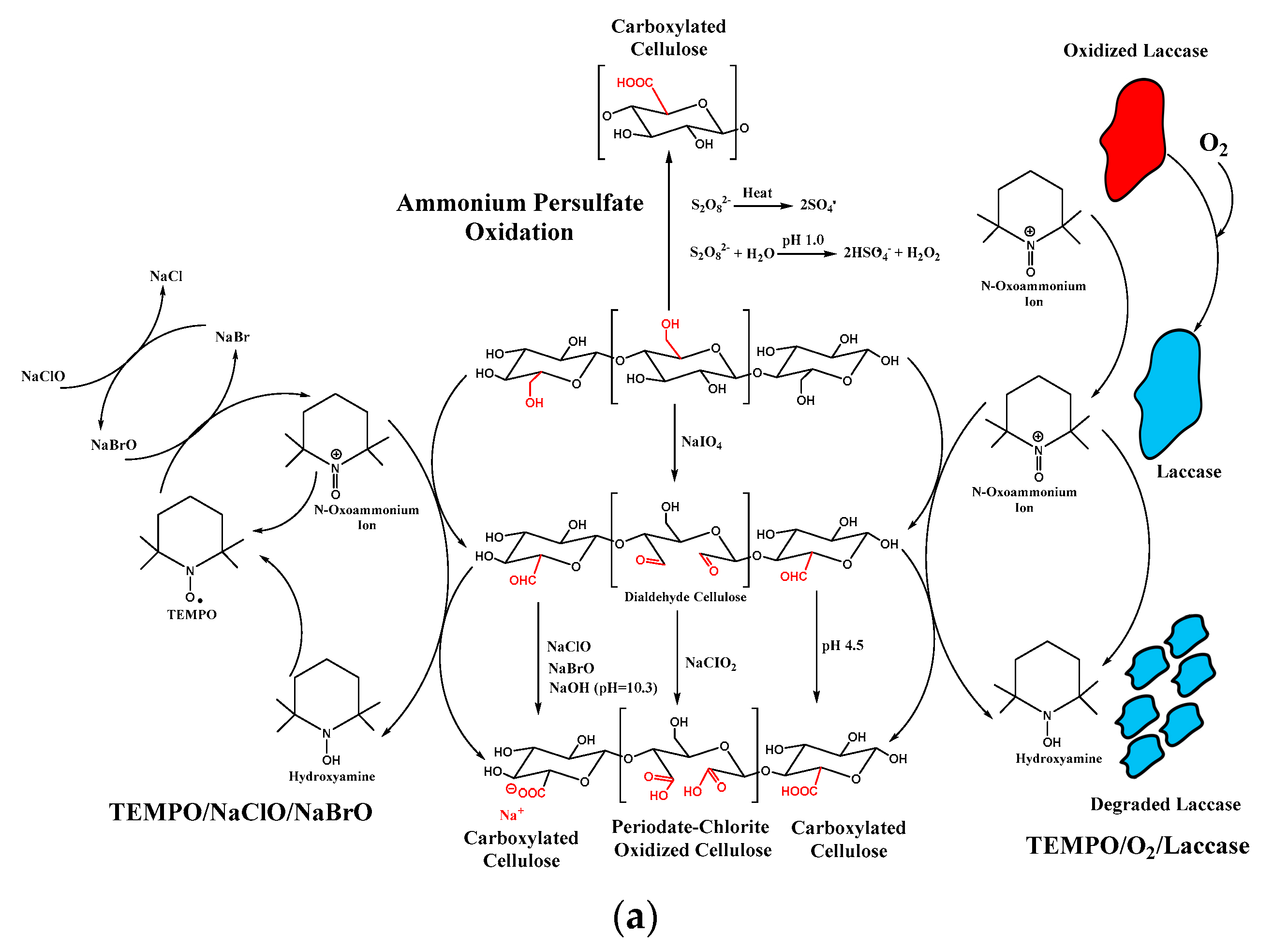
- Jiang, J.; Ye, W.; Liu, L.; Wang, Z.; Fan, Y.; Saito, T.; Isogai, A. Cellulose nanofibers prepared using the TEMPO/laccase/O2 system. Biomacromolecules 2017, 18, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Plappert, S.F.; Liebner, F.W.; Konnerth, J.; Nedelec, J.-M. Anisotropic nanocellulose gel–membranes for drug delivery: Tailoring structure and interface by sequential periodate–chlorite oxidation. Carbohydr. Polym. 2019, 226, 115306. [Google Scholar] [CrossRef] [PubMed]
- Mascheroni, E.; Rampazzo, R.; Ortenzi, M.A.; Piva, G.; Bonetti, S.; Piergiovanni, L. Comparison of cellulose nanocrystals obtained by sulfuric acid hydrolysis and ammonium persulfate, to be used as coating on flexible food-packaging materials. Cellulose 2016, 23, 779–793. [Google Scholar] [CrossRef] [Green Version]
- Montanari, S.; Roumani, M.; Heux, L.; Vignon, M.R. Topochemistry of carboxylated cellulose nanocrystals resulting from TEMPO-mediated oxidation. Macromolecules 2005, 38, 1665–1671. [Google Scholar] [CrossRef]
- Carlsson, D.O.; Hua, K.; Forsgren, J.; Mihranyan, A. Aspirin degradation in surface-charged TEMPO-oxidized mesoporous crystalline nanocellulose. Int. J. Pharm. 2014, 461, 74–81. [Google Scholar] [CrossRef]
- Chen, X.; Xu, X.; Li, W.; Sun, B.; Yan, J.; Chen, C.; Liu, J.; Qian, J.; Sun, D. Effective drug carrier based on polyethylenimine-functionalized bacterial cellulose with controllable release properties. ACS Appl. Bio Mater. 2018, 1, 42–50. [Google Scholar] [CrossRef]
- Singhsa, P.; Narain, R.; Manuspiya, H. Bacterial cellulose nanocrystals (BNCC) preparation and characterization from three bacterial cellulose sources and development of functionalized BNCCs as nucleic acid delivery systems. ACS Appl. Nano Mater. 2017, 1, 209–221. [Google Scholar] [CrossRef]
- Li, L.; Yu, C.; Yu, C.; Chen, Q.; Yu, S. Nanocellulose as template to prepare rough-hydroxy rich hollow silicon mesoporous nanospheres (R-nCHMSNs) for drug delivery. Int. J. Biol. Macromol. 2021, 180, 432–438. [Google Scholar] [CrossRef]
- Akhlaghi, S.P.; Berry, R.C.; Tam, K.C. Surface modification of cellulose nanocrystal with chitosan oligosaccharide for drug delivery applications. Cellulose 2013, 20, 1747–1764. [Google Scholar] [CrossRef]
- Tortorella, S.; Maturi, M.; Dapporto, F.; Spanu, C.; Sambri, L.; Franchini, M.C.; Chiariello, M.; Locatelli, E. Surface modification of nanocellulose through carbamate link for a selective release of chemotherapeutics. Cellulose 2020, 27, 8503–8511. [Google Scholar] [CrossRef]
- Liu, Y.; Sui, Y.; Liu, C.; Liu, C.; Wu, M.; Li, B.; Li, Y. A physically crosslinked polydopamine/nanocellulose hydrogel as potential versatile vehicles for drug delivery and wound healing. Carbohydr. Polym. 2018, 188, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; He, J.; Zhang, M.; Tam, K.C.; Ni, P. A new pathway towards polymer modified cellulose nanocrystals via a “grafting onto” process for drug delivery. Polym. Chem. 2015, 6, 4206–4209. [Google Scholar] [CrossRef]
- Kumar, A.; Durand, H.; Zeno, E.; Balsollier, C.; Watbled, B.; Sillard, C.; Fort, S.; Baussanne, I.; Belgacem, N.; Lee, D. The surface chemistry of a nanocellulose drug carrier unraveled by MAS-DNP. Chem. Sci. 2020, 11, 3868–3877. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khine, Y.Y.; Ganda, S.; Stenzel, M.H. Covalent tethering of temperature responsive pNIPAm onto TEMPO-oxidized cellulose nanofibrils via three-component passerini reaction. ACS Macro Lett. 2018, 7, 412–418. [Google Scholar] [CrossRef]
- Tardy, B.L.; Yokota, S.; Ago, M.; Xiang, W.; Kondo, T.; Bordes, R.; Rojas, O.J. Nanocellulose–surfactant interactions. Curr. Opi. Colloid Interface Sci. 2017, 29, 57–67. [Google Scholar] [CrossRef]
- Zainuddin, N.; Ahmad, I.; Kargarzadeh, H.; Ramli, S. Hydrophobic kenaf nanocrystalline cellulose for the binding of curcumin. Carbohydr. Polym. 2017, 163, 261–269. [Google Scholar] [CrossRef]
- Raghav, N.; Sharma, M.R. Usage of nanocrystalline cellulose phosphate as novel sustained release system for anti-inflammatory drugs. J. Mol. Struct. 2021, 1233, 130108. [Google Scholar] [CrossRef]
- Gupta, R.D.; Raghav, N. Differential effect of surfactants tetra-n-butyl ammonium bromide and N-Cetyl-N,N,N-trimethyl ammonium bromide bound to nano-cellulose on binding and sustained release of some non-steroidal anti-inflammatory drugs. Int. J. Biol. Macromol. 2020, 164, 2745–2752. [Google Scholar] [CrossRef]
- Liu, X.Q.; Picart, C. Layer-by-layer assemblies for cancer treatment and diagnosis. Adv. Mater. 2016, 28, 1295–1301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Koker, S.; Hoogenboom, R.; De Geest, B.G. Polymeric multilayer capsules for drug delivery. Chem. Soc. Rev. 2012, 41, 2867–2884. [Google Scholar] [CrossRef]
- Eivazi, A.; Medronho, B.; Lindman, B.; Norgren, M. On the development of all-cellulose capsules by vesicle-templated layer-by-layer assembly. Polymers 2021, 13, 589. [Google Scholar] [CrossRef]
- Kulkarni, A.D.; Vanjari, Y.H.; Sancheti, K.H.; Patel, H.M.; Belgamwar, V.S.; Surana, S.J.; Pardeshi, C.V. Polyelectrolyte complexes: Mechanisms, critical experimental aspects, and applications. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1615–1625. [Google Scholar] [CrossRef] [Green Version]
- Mohanta, V.; Madras, G.; Patil, S. Layer-by-layer assembled thin films and microcapsules of nanocrystalline cellulose for hydrophobic drug delivery. ACS Appl. Mater. Interfaces 2014, 6, 20093–20101. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Y.; Zhang, L.; Xu, Z.; Dai, H.; Wu, W. Nanocellulose/gelatin composite cryogels for controlled drug release. ACS Sust. Chem. Eng. 2019, 7, 6381–6389. [Google Scholar] [CrossRef]
- Hennink, W.E.; Van Nostrum, C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 223–236. [Google Scholar] [CrossRef]
- Shi, X.; Zheng, Y.; Wang, G.; Lin, Q.; Fan, J. pH-and electro-response characteristics of bacterial cellulose nanofiber/sodium alginate hybrid hydrogels for dual controlled drug delivery. RSC Adv. 2014, 4, 47056–47065. [Google Scholar] [CrossRef]
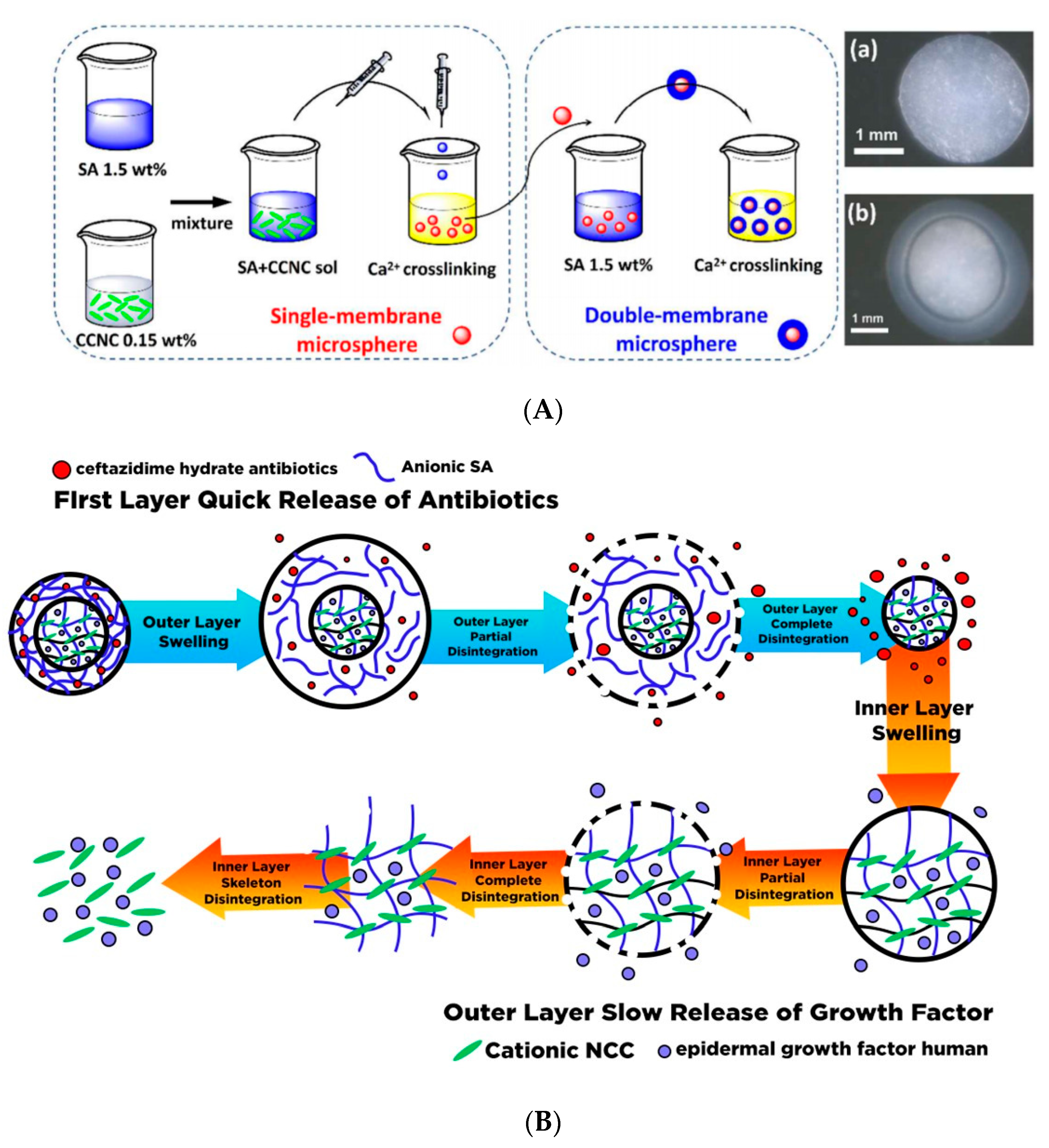
- Lin, N.; Geze, A.; Wouessidjewe, D.; Huang, J.; Dufresne, A. Biocompatible double-membrane hydrogels from cationic cellulose nanocrystals and anionic alginate as complexing drugs codelivery. ACS Appl. Mater. Interfaces 2016, 8, 6880–6889. [Google Scholar] [CrossRef]
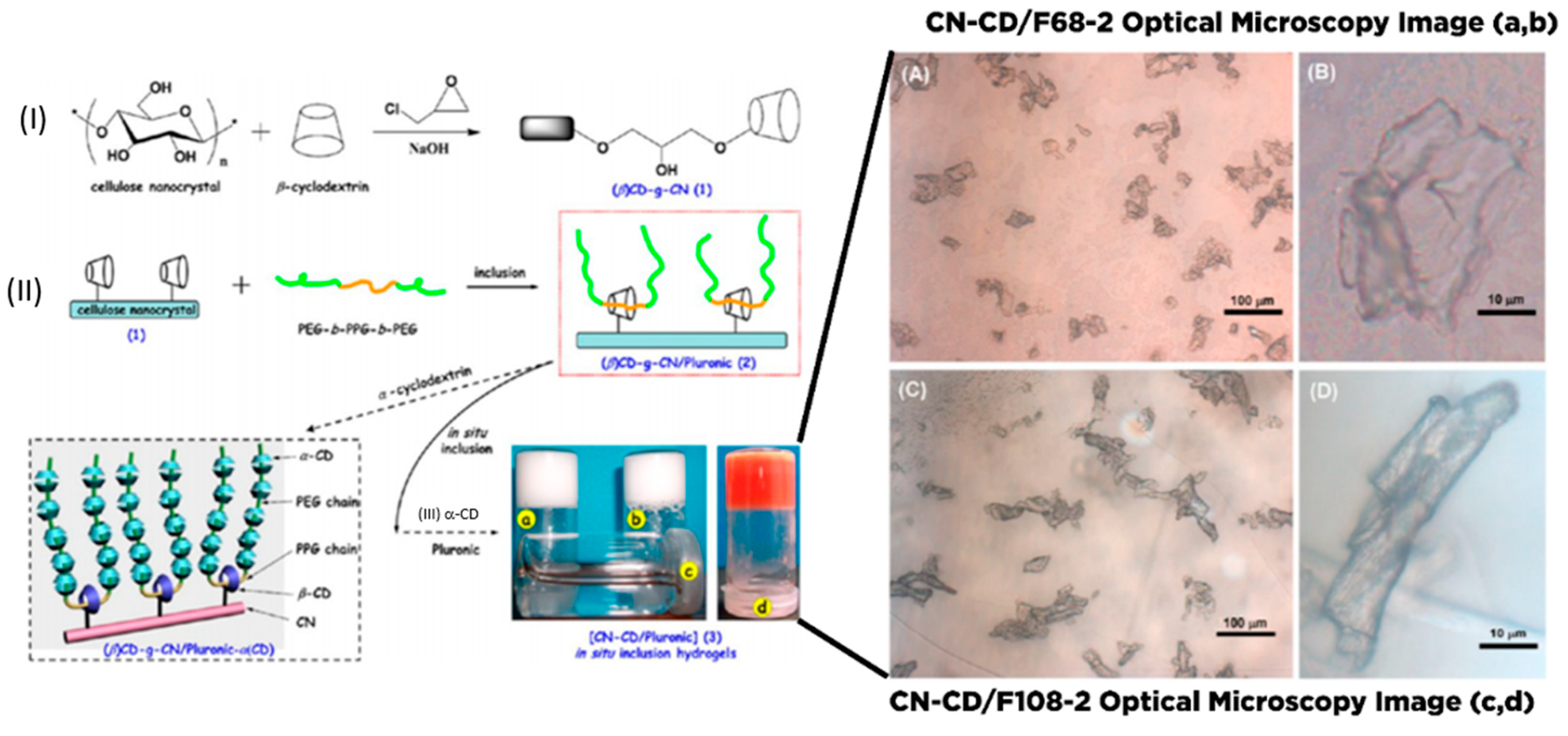
- Lin, N.; Dufresne, A. Supramolecular hydrogels from in situ host–guest inclusion between chemically modified cellulose nanocrystals and cyclodextrin. Biomacromolecules 2013, 14, 871–880. [Google Scholar] [CrossRef]
- Muller, A.; Ni, Z.; Hessler, N.; Wesarg, F.; Muller, F.A.; Kralisch, D.; Fischer, D. The biopolymer bacterial nanocellulose as drug delivery system: Investigation of drug loading and release using the model protein albumin. J. Pharma. Sci. 2013, 102, 579–592. [Google Scholar] [CrossRef]
- Muller, A.; Zink, M.; Hessler, N.; Wesarg, F.; Muller, F.A.; Kralisch, D.; Fischer, D. Bacterial nanocellulose with a shape-memory effect as potential drug delivery system. RSC Adv. 2014, 4, 57173–57184. [Google Scholar] [CrossRef] [Green Version]
- Kopac, T.; Krajnc, M.; Ručigaj, A. A mathematical model for pH-responsive ionically crosslinked TEMPO nanocellulose hydrogel design in drug delivery systems. Int. J. Biol. Macromol. 2021, 168, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Agarwal, S.; Greiner, A. Low-density open cellular sponges as functional materials. Angew. Chem. Int. Ed. 2017, 56, 15520–15538. [Google Scholar] [CrossRef] [PubMed]
- Lavoine, N.; Bergstrom, L. Nanocellulose-based foams and aerogels: Processing, properties, and applications. J. Mater. Chem. A 2017, 5, 16105–16117. [Google Scholar] [CrossRef] [Green Version]
- Pierre, A.C.; Pajonk, G.M. Chemistry of aerogels and their applications. Chem. Rev. 2002, 102, 4243–4266. [Google Scholar] [CrossRef]
- Kargarzadeh, H.; Huang, J.; Lin, N.; Ahmad, I.; Mariano, M.; Dufresne, A.; Thomas, S.; Galeski, A. Recent developments in nanocellulose-based biodegradable polymers, thermoplastic polymers, and porous nanocomposites. Prog. Polym. Sci. 2018, 87, 197–227. [Google Scholar] [CrossRef]
- Sun, Y.; Chu, Y.; Wu, W.; Xiao, H. Nanocellulose-based Lightweight Porous Materials: A Review. Carbohydr. Polym. 2021, 255, 117489. [Google Scholar] [CrossRef]
- Zhao, J.; Lu, C.; He, X.; Zhang, X.; Zhang, W.; Zhang, X. Polyethylenimine-grafted cellulose nanofibril aerogels as versatile vehicles for drug delivery. ACS Appl. Mater. Interfaces 2015, 7, 2607–2615. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Zhu, H.; Wang, L.; He, H.; Wang, S. Biocompatible smart cellulose nanofibres for sustained drug release via pH and temperature dual-responsive mechanism. Carbohydr. Polym. 2020, 249, 116876. [Google Scholar] [CrossRef] [PubMed]
- Svagan, A.J.; Benjamins, J.-W.; Al-Ansari, Z.; Shalom, D.B.; Mullertz, A.; Wagberg, L.; Lobmann, K. Solid cellulose nanofiber based foams–towards facile design of sustained drug delivery systems. J. Controlled Release 2016, 244, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Lobmann, K.; Svagan, A.J. Cellulose nanofibers as excipient for the delivery of poorly soluble drugs. Int. J. Pharm. 2017, 533, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Svagan, A.J.; Mullertz, A.; Lobmann, K. Floating solid cellulose nanofibre nanofoams for sustained release of the poorly soluble model drug furosemide. J. Pharm. Pharmacol. 2017, 69, 1477–1484. [Google Scholar] [CrossRef]
- Bannow, J.; Benjamins, J.-W.; Wohlert, J.; Lobmann, K.; Svagan, A.J. Solid nanofoams based on cellulose nanofibers and indomethacin—The effect of processing parameters and drug content on material structure. Int. J. Pharm. 2017, 526, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Fiorati, A.; Turco, G.; Travan, A.; Caneva, E.; Pastori, N.; Cametti, M.; Punta, C.; Melone, L. Mechanical and drug release properties of sponges from cross-linked cellulose nanofibers. ChemPlusChem 2017, 82, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Rong, L.; Wang, B.; Mao, Z.; Xu, H.; Zhong, Y.; Zhang, L.; Sui, X. A light-weight and high-efficacy antibacterial nanocellulose-based sponge via covalent immobilization of gentamicin. Carbohydr. Polym. 2018, 200, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, M.; Shojaei, S.; Safa, K.D.; Ghasemi, Z.; Salehi, R.; Yousefi, B.; Shafiei-Irannejad, V. Biocompatible magnetic tris (2-aminoethyl) amine functionalized nanocrystalline cellulose as a novel nanocarrier for anticancer drug delivery of methotrexate. New J. Chem. 2017, 41, 2160–2168. [Google Scholar] [CrossRef]
- Supramaniam, J.; Adnan, R.; Kaus, N.H.M.; Bushra, R. Magnetic nanocellulose alginate hydrogel beads as potential drug delivery system. Int. J. Biol. Macromol. 2018, 118, 640–648. [Google Scholar] [CrossRef]
- Jeddi, M.K.; Mahkam, M. Magnetic nano carboxymethyl cellulose-alginate/chitosan hydrogel beads as biodegradable devices for controlled drug delivery. Int. J. Biol. Macromol. 2019, 135, 829–838. [Google Scholar] [CrossRef]
- Cirillo, G.; Hampel, S.; Spizzirri, U.G.; Parisi, O.I.; Picci, N.; Iemma, F. Carbon nanotubes hybrid hydrogels in drug delivery: A perspective review. BioMed Res. Int. 2014, 825017. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, Z.; Zhang, Y. The application of carbon nanotubes in target drug delivery systems for cancer therapies. Nanoscale Res. Lett. 2011, 6, 1–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anirudhan, T.; Shainy, F.; Thomas, J.P. Effect of dual stimuli-responsive dextran/nanocellulose polyelectrolyte complexes for chemo photothermal synergistic cancer therapy. Int. J. Biol. Macromol. 2019, 135, 776–789. [Google Scholar]





| Types | Nanocrystalline Cellulose (NCC) | Cellulose Nanofibers (CNF) | Bacterial Cellulose | |
|---|---|---|---|---|
| Parameter | ||||
| Common names | Cellulose whisker, cellulose nanowhisker, cellulose nanowire, and cellulose nanorod or spherical cellulose nanocrystals | Cellulose nanofibril, micro fibrillated cellulose, Nanofibrillar cellulose, Nanofibrillated cellulose, and cellulose microfibril | Microbial cellulose (MC), bacterial nanocellulose (BC), and bio-cellulose (BC) | |
| Morphological structure | Needles like shape, elongated rod-like shape, and spindle shape | Smooth, extended, and flexible chain | Twisted ribbons like shape | |
| Structure of Nanocellulose | Crystalline domains | amorphous and crystalline domains | Crystalline domains | |
| Chain Length | 500 | 500–15,000 | 4000–10,000 | |
| Crystallinity (%) | 54–88 | - | 84–88 | |
| Other Impurities and contaminant | Possible to contain hemicellulose, lignin, and pectin | Possible to contain hemicellulose, lignin, and pectin | Contain no hemicellulose, lignin, and pectin | |
| Size (Length and Diameter) | Diameter: 5–30 nm and Length: 100–500 nm | Diameter: 1–100 nm and Length: 500–2000 nm | Diameter 20–100 nm and several micrometric lengths | |
| Process System | Top-down system | Top-down system | Bottom-up system | |
| Tensile strength (Gpa) | 7.5–7.7 [34] | 13 | 0.2–0.3 | |
| Modulus Young (Gpa) | 110–220 [45] | Approximately 15 | 18–20 [60] | |
| Density (gr/cm3) | 1.6 [61] | 1.42 | 1.1 | |
| Characteristics | Homogenous nanorod form, exceptional aspect ratio (length to diameter), appreciable specific surface area (SSA), biocompatibility, liquid crystalline attribute, inferior breaking expansion, high young’s modulus, hydrophilicity, outstanding mechanical stiffness, tunable surface characteristic due to the reactive hydroxyl group and low density | Extended length with excellent aspect proportion (length to diameter), superlative surface area, hydrophilicity, biocompatibility and adjustable characteristic through surface modification afforded by high extensive of hydroxyl groups in CNF. | High crystallinity of nanocellulose (84–88%) and polymerization grade, high water uptake capacity (exceeding 100 times of its weight), remarkable surface area (high aspect proportion of fiber), outstanding tensile strength (young modulus 15–18 Gpa), and flexibility, foldability, moldability, mechanical stability, highly biocompatible material, non-cytotoxic, un-genotoxic and high porosity | |
| Waste Residue Sources | Nanocellulose Isolation Technique | Nanocellulose Characteristics | References | |
|---|---|---|---|---|
| Pretreatment | Treatment | |||
| WASTE BASED FOREST RESIDUE | ||||
| Birch and Spruce sawdust | Hot water treatment and subsequent delignification; TEMPO oxidation | Mechanical defibrillation |
CNF σ = 171,6 MPa; E = 6.4 Gpa; | [77] |
| Medium-density fiberboard | Soxhlet extraction (Ethanol and toluene), NaOH, and recurrent bleaching | Acid hydrolysis (H2SO4) | NCC L:164.7 nm; W: 6.7 nm; CrI (%): 71 | [78] |
| Eucalyptus sawdust | Hot water treatment, alkaline delignification, O2 residual delignification, TEMPO-mediated Oxidation | High pressure homogenization | CNF Davg: 41.0 nm; SSA: 60 m2/g; Y (%) = 60 | [79] |
| Pinecone biomass | Alkali treatment followed with acidification (NaClO2:CH3COOH) | Mechanical grinding. | CNF σ: 273 MPa; E: 17 GPa; CrI (%): 70%; D: 5–20 nm. | [80] |
| Logging residues | Alkaline and bleaching pretreatment | Acid hydrolysis (H2SO4) | NCC L/D > 10; CrI (%): 86–93; TS (°C): 208.4–211 | [81] |
| Bamboo log chips | Pretreatment with glycerol; and screw extrusion | Mechanical refining/Milling treatment assisted by H2SO4 (0.15%) as a catalyst | CNF D: 20–80 nm; CrI (%): 52.7%; Y (%): 77.2 | [82] |
| WASTE BASED ALGAE RESIDUE | ||||
| Cladophorales | - | TEMPO Oxidation; | CNF W: 80 nm; SSA: 77 m2/g CrI (%): 93%; D: 80 nm; Excellent mechanical and rheological characteristics | [83] |
| Red algae | - | Acid hydrolysis (H2SO4) | NCC L: 432 nm; W = 28.6 nm; L/D: 15.1; CrI (%): 69.5; Yield: 20.5%; TS (°C): 220 °C | [84] |
| Green Seaweed Ulva lactuca | Methanol pretreatment (Soxhlet extraction) followed by bleaching, alkaline pretreatment, and neutralization | Acid hydrolysis (H2SO4) | NCC CrI (%): 83; TS (°C): 225 °C | [85] |
| Industrial kelp (Laminaria japonica) waste | Two stages of bleaching pretreatment (Chlorine dioxide followed with hydrogen peroxide) | Acid hydrolysis (H2SO4) | NCC L: 100–500 nm; D = 20–50 nm; L/W: 5–20; Yield: 52.3%; TS (°C): 240 °C | [86] |
| Dealginate kelp residue From Giant Kelp (Calrose variety) | Na2CO3 (2% wt) treatment, residual sodium alginate extraction by NaOH (2% wt); Ultrasonic irradiation; NaClO2 (0.7% wt) buffer solution bleaching treatment and delignification | Acid hydrolysis (H2SO4) | NCC L: 100–500 nm; D = 20–50 nm; L/W: 30–70; CrI (%): 74.5; TS (°C): 120–180 °C; l = 120–480 nm | [87] |
| Chaetomorpha antennina | Bleaching method | Acid hydrolysis (HCl) followed with Ultrasonic irradiation | CNF E = 0.9 Gpa; CrI (%): 85.02; Y = 34.09%; TS (°C) = 200–370 °C | [88] |
| Gelidium sesquipedale | Soxhlet Extraction (Ethanol: Toluene) Bleaching treatment, delignification (5% KOH solution) | Acid hydrolysis (H2SO4) followed with neutralization (NaOH) | NCC L: 467–1650 nm; D = 18–29 nm; L/W: ~40; CrI (%): ~70%; | [89] |
| Gelidium elegansred | Alkali and bleaching pretreatment | Acid hydrolysis (H2SO4) | NCC L: 547.3 nm; D = 21.8 nm; L/W: 25; CrI (%): 73%; TS (°C): 334 °C | [90] |
| WASTE BASED AGRICULTURAL RESIDUE | ||||
| Waste sugarcane bagasse | Acidification and alkaline pretreatment | Acid hydrolysis (H2SO4) | NCC L: 170 nm; D = 35 nm; h = 70–90 nm; CrI (%): 93%; TS (°C): 249–345 °C | [91] |
| Jute dried stalks | Alkali treatment followed by steam explosion; sodium chlorite bleaching | Acid hydrolysis (oxalic acid) followed by steam explosion. | CNF L: few micrometers D = 50 nm; CrI (%): 82.2%; E: 138 Gpa; TS (°C): 250–400 °C | [92] |
| Coconut husk | Ultrasonic-aided solvent submersion. Delignification and Bleaching Pretreatment, followed by TEMPO-mediated Oxidation (TEMPO/NaClO/NaClO2; pH = 4.8) | Ultrasonication | CNF L: 150–350; D = 2–10 nm; CrI (%): 56.3%; TS (°C): 190–380 °C | [93] |
| Citrus waste | Alkaline and Bleaching Pretreatment | Enzymatic hydrolysis and ultrasonication | CNF L: 458 nm; W: 10.3 nm; Davg = 10 nm; L/W: 47; CrI (%): 55%; TS (°C): 190–380 °C | [94] |
| Raw rice husk | Size Reduction, Soxhlet extraction (toluene and ethanol); Acidification (NaClO2 and CH3COOH); and delignification (5% KOH) | High pressure homogenization and high-intensity ultrasonication processes (500 W,40 min). | CNF L: 1800 nm; W: 10 nm; CrI (%): 77.5%; L/D > 180; TS (°C): 323 °C | [95] |
| Corn cobs | - | One pot synthesis via mechanochemical esterification |
CNF σ = 110–125 MPa; E = 5.5 Gpa; D: 1.5–2.8 nm | [96] |
| Kenaf bast fiber | Delignification and three stage of bleaching pre-treatments | Mechanical grinder | CNF D: 1.2–34 nm; CrI (%): 82.52%; Y (%) 60.25; TS (°C): 200–400 | [97] |
| Passion Fruit Peels | Alkaline and bleaching pretreatment | Acid hydrolysis (H2SO4) followed with ultrasonication | NCC L: 103–173.5 nm; CrI (%): 77.96%; TS (°C): 303.4; Y (%): 58.1 | [29] |
| WASTE BASED INDUSTRIAL BY PRODUCT | ||||
| Olive industry solid waste | Pretreatment including pulping and bleaching | Acid hydrolysis (H2SO4) | NCC | [98] |
| Lime residues | Autoclaving pretreatment | High shear and high-pressure homogenization | CNF D: 5–28 nm; CrI (%): 44–46 | [99] |
| Recycled Tetra Pak Food Packaging Wastes | Delignification and bleaching pretreatment | Acid hydrolysis (H2SO4) followed with ultrasonication | NCC L: 127–258 nm; D: 11.4–14 nm; L/D: 10; CrI (%): 94.8%; TS (°C): 204 | [100] |
| Waste paper | Deinking method and alkaline pretreatment | Acid hydrolysis (H2SO4) followed with ultrasonication | NCC L: 271 nm | [101] |
| Discarded cigarette filters | Ethanol extraction, alkaline pretreatment, and bleaching pretreatment, | Acid hydrolysis (H2SO4) followed with ultrasonication | NCC L: 143 nm; W: 8 nm; CrI (%): 96.77%; Y (%): 29.4 | [102] |
| Recycled Paper Mill Sludge | Ozonation pretreatment | Acid hydrolysis (Maleic acid) | NCC L: 2431 nm; W: 165 nm; L/D: 16.7 CrI (%): 77%; Y (%): 0.8 | [103] |
| Citrus Pulp of Floater (CPF) | Alkaline and bleaching pretreatment with autoclave | Enzymatic hydrolysis | n.d CrI (%):60 | [104] |
| Sweet lime pulp waste | Blending and acid hydrolysis (H2SO4) | Komagataeibacter europaeus SGP37 incubated in static intermittent fed-batch cultivation | BNC Y(g/L): CrI (%):89.6; TS (°C): 348 | [105] |
| Bacteria Cultivation | Source of Carbon and Its Concentration | Culture Medium | Fermentation Conditions | Yield (g/L) | References |
|---|---|---|---|---|---|
| Komagataeib acter xylinus K2G30 (UMCC 2756) | Glucose | GY Broth | Static; 28 °C; 9 days | 6.17 ± 0.02 | [118] |
| Mannitol | 8.77 ± 0.04 | ||||
| Xylitol | 1.36 ± 0.05 | ||||
| Komagataeibacter rhaeticus PG 2 | Glycerol | Hestrin–Schramm (HS) liquid media | Static; 28 °C; 15 days | ~6.9 | [119] |
| Glucose | ~4.05 | ||||
| Sorbitol and Mannitol | ~1.65–3.41 | ||||
| Komagataeibacter xylinus B12068 | Glucose | Hestrin–Schramm (HS) liquid media | Static; 30 °C; 7 days | ~2.2 | [120] |
| Sucrose | ~1.6 | ||||
| Galactose | ~1.4 | ||||
| Maltose and Mannitol | ~0.1–0.2 | ||||
| Komagataeibacter medellinensis | Glucose | Standard Hestrin–Schramm (HS) Medium | Static; 28 °C; 8 days | 2.80 | [121] |
| Sucrose | 1.68 | ||||
| Fructose | 0.38 | ||||
| Gluconacetobacter xylinus (PTCC 1734) | Date syrup | Yamanaka | 150 rpm; 28 °C; 7 days, | ~1.15 | [122] |
| glucose | ~0.85 | ||||
| mannitol, | ~1.4 | ||||
| sucrose | ~1.45 | ||||
| food-grade sucrose | ~0.7 | ||||
| Date syrup | Hestrin–Schramm | ~0.65 | |||
| glucose | ~0.7 | ||||
| mannitol, | ~1.05 | ||||
| sucrose | ~1.5 | ||||
| food-grade sucrose | ~1.1 | ||||
| Date syrup | Zhou | ~0.9 | |||
| glucose | ~1 | ||||
| mannitol, | ~1.85 | ||||
| sucrose | ~1.65 | ||||
| food-grade sucrose | ~1.15 |
| Methods | Reagents | Aided Reagents | Operation Parameter | Sources of Cellulose | Mechanical Technique | Yield (%) | Morphology (nm) | CI%) | Zeta Potential (mV) | Surface Charge Density (mmol/g) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mineral Acids | H2SO4 | - | 52% H2SO4 50 °C; 60 min | Passion Fruit Peels | Ultrasonication | 58.1 | NCC L: 103–173.5 | 77.96 | −25 | - | [29] |
| - | 63% H2SO4 50 °C; 90 min | Microcrystalline Cellulose | Ultrasonication | 30% | NCC L:250; W: 16 | - | −46.1 | - | [134] | ||
| - | - | Filter Paper | - | - | NCC W: 22 | 85 | - | -SO3H (0.0985) | [135] | ||
| H2SO4/HCl | - | H2SO4:HCl:H2O (3:1:6); Ultrasonic 50 hZ; 10 h | Microcrystalline Cellulose | Ultrasonication | - | S-CNC (D:10–180 nm) | - | - | - | [136] | |
| ClSO3H (Post-sulfonation) | - | ClSO3H in 50 mL DMF; RT; 2 h | Sulfated NCC | Ultrasonication | 79.31 | NCC L:152; W: 22.7; h: 5.0 | 88% | −66.1 | -SO3H 0.409 | [137] | |
| H3PO4 | - | 73.9% H3PO4; 100 °C; 90 min | Filter Paper | Blending (15 min) | 76–80 | NCC L:316; W: 31; | 81 | - | -PO3 (0.0108) | [135] | |
| - | 10.7 M H3PO4; 100 °C; 30 min | Cellulose Biotethanol Residue | Homogenizer (10 times) | NCC | 83 | −27 | -PO3 (0.4352) | [138] | |||
| - | 10.7 M H3PO4; 100 °C; 30 min | CNF L: 2500 nm | 81 | −23 | -PO3 (0.018) | ||||||
| H3PO4 in molten Urea | - | 10.7 M H3PO4; 150 °C; 30 min | NCC L: 610 nm | 83 | −34 | -PO3 (1.038) | |||||
| - | CNF L: 330–480 nm | 86 | −24 | -PO3 (1.173) | |||||||
| HCl | - | 2.5 M HCl; 105 °C; 40 min | Filter Paper | Blending (40 min) | - | NCC W: 20 | 79% | - | - | [135] | |
| Organic Acids | Acetic Acid | H2SO4 | 80 °C; 3 h | Bleached eucalyptus kraft pulp | - | 81 | NCC L: 264; W: 16 | 80 | −33 | -SO3H (0.015) | [139] |
| HCl | 105 °C; 9 h | Cotton | Blending (20 min) | 30 | NCC L: 269; W: 45 | - | - | - | [140] | ||
| Formic Acid | 6M HCl | 80 °C; 4 h | Microcrystalline Cellulose | - | - | NCC L: 236; W: 25 | 88 | −1.7 | Formate (0.4) | [141] | |
| 0.015 M FeCl3 | 90 °C; 6 h | Bleached eucalyptus kraft pulp | - | 75 | NCC L:594 | 75 | −6.53 | Formate | [142] | ||
| Lactic Acid | HCl | 150 °C; 3 h | Cotton | Blending (20 min) | - | NCC L: 200; W = 20 | 80 | - | Lactate | [143] | |
| Butyric Acid | 0.027 M HCl | 105 °C; 9 h | Cotton | Blending (20 min) | 20 | NCC L: 226; W = 34 | - | - | Butyrate | [140] | |
| Maleic Acid (MA) | - | 70% MA; 100 °C; 45 min | Bleached eucalyptus kraft pulp | - | 12% | NCC | - | -33 | -COOH (0.29) | [144] | |
| - | 60% MA; 120 °C; 2 h | Bleached eucalyptus kraft pulp | Microfluidizer (120 mPa; 5 passes) | 3% | L: 329.9; h = 15.9 | - | −46.9 | -COOH (0.368) | [145] | ||
| 84% | CNF h: 13.4 | - | −45.2 | -COOH (0.059) | |||||||
| Oxalic Acid (OA) | - | 8.75% OA; 110 °C; 15 min | Filter paper | Sonication (60 min) | 93.77 | NCC L: 150–200; W: 5–20 | - | −36 | -COOH, 0.29 | [146] | |
| - | 70% OA; 100 °C; 1 h | Bleached eucalyptus kraft pulp | - | 24.7 | NCC | 80 | −42.5 | -COOH | [144] | ||
| - | 30% OA; 100° C; 30 min | Celery | Sonication (18 min) | 76.8 | CNF h: 5.5 | 49 | −32.9 | -COOH | [147] | ||
| Malonic Acid | - | 80% wt of Malonic Acid; 140 °C; 3 h | Ramie Cellulose | Blending (5 min) | 5% | NCC L: ~220; W: ~12 | - | - | -COOH | [148] | |
| 0.025 M HCl | 19.8% | 75 | - | -COOH | |||||||
| Malic acid | - | 80% wt of Malic Acid; 140 °C; 3 h | 3.4% | - | - | -COOH, (1.617) | |||||
| 0.05 M HCl | 20% | 78 | - | -COOH | |||||||
| Citric Acid | - | 80% wt of Citric Acid; 140 °C; 3 h | 5.1 | - | - | -COOH | |||||
| 0.05 M HCl | 20.5 | 78 | - | -COOH, (1.884) | |||||||
| - | 80% wt of Citric Acid; 100 °C; 4 h | Bleached Baggase Pulp | Ultrasonication | 32 | NCC, L: 251; W: 21 | 78 | −122.9 | -COOH, 0.65 | [149] | ||
| - | - | CNF, L: 654; W: 32 | 69 | 190.3 | -COOH, 0.3 | ||||||
| Oxidation Treatment | TEMPO/NaCl /NaBr | - | TEMPO (0.094 mmol)-NaBr (1.57 mmol)- NaClO (1.24 M); 10 °C; 45 min | Nanocrystalline Cellulose | Ultrasonication | - | NCC, L: 100; W: 5–20 | 80% | - | - | [150] |
| - | TEMPO (0.1 mmol mmol)-NaBr (1 mmol)- NaClO (5 mmol/g cellulose); Ambient condition; 1.5 h | HBKP | Ultrasonication | - | CNF | 85% | - | -COOH; -CHO (1.191) | [151] | ||
| TEMPO/O2/Laccase | 50 mM TEMPO, 5 U mL–1 laccase; 96 h | HBKP | Ultrasonication | - | CNF, L: > 100; W: 4–8 | - | - | -COOH; -CHO (0.837) | |||
| Sequential Periodate-Chlorite Oxidation | 1 M Acetic Acid (2) | (1). 46 mmol NaIO4; 50 °C;4.5 h followed by (2). 12 g NaClO2l 50 °C; 40 h | Hardwood Pulp | Homogenizer (5 passes; 80 MPa) | - | CNF, L: 95.8; W: 2.72 | - | −128 | -COOH (2.0) | [152] | |
| APS Oxidation | - | 1 M APS; 75 °C; 16 h | Cotton Linters | - | 34.4 | CNF, L: 95.8; W: 2.72 | 63.8 | - | -COOH (0.16); -SO3 (0.98) | [153] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lunardi, V.B.; Soetaredjo, F.E.; Putro, J.N.; Santoso, S.P.; Yuliana, M.; Sunarso, J.; Ju, Y.-H.; Ismadji, S. Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers. Polymers 2021, 13, 2052. https://doi.org/10.3390/polym13132052
Lunardi VB, Soetaredjo FE, Putro JN, Santoso SP, Yuliana M, Sunarso J, Ju Y-H, Ismadji S. Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers. Polymers. 2021; 13(13):2052. https://doi.org/10.3390/polym13132052
Chicago/Turabian StyleLunardi, Valentino Bervia, Felycia Edi Soetaredjo, Jindrayani Nyoo Putro, Shella Permatasari Santoso, Maria Yuliana, Jaka Sunarso, Yi-Hsu Ju, and Suryadi Ismadji. 2021. "Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers" Polymers 13, no. 13: 2052. https://doi.org/10.3390/polym13132052
APA StyleLunardi, V. B., Soetaredjo, F. E., Putro, J. N., Santoso, S. P., Yuliana, M., Sunarso, J., Ju, Y.-H., & Ismadji, S. (2021). Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers. Polymers, 13(13), 2052. https://doi.org/10.3390/polym13132052