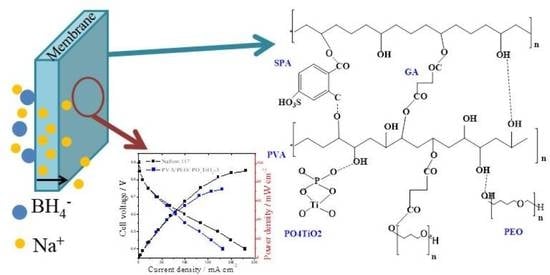
Novel Crosslinked Sulfonated PVA/PEO Doped with Phosphated Titanium Oxide Nanotubes as Effective Green Cation Exchange Membrane for Direct Borohydride Fuel Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis
2.1.1. Synthesis of Phosphated Titanium Oxide Nanotube (PO4TiO2)
2.1.2. Synthesis of SPVA/PEO/PO4TiO2 Membranes
2.2. Characterization
3. Results and Discussion
3.1. Characterization of PO4TiO2 Nanotube and Nanocomposite Membranes
3.2. Mechanical and Thermal Analysis
3.3. Oxidative Stability
3.4. Ionic Conductivity, IEC, and Borohydride Crossover
3.5. Fuel Cell Performance
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanli, A.E.; Gordesel, M.; Yilmaz, E.S.; Ozden, S.K.; Gunlu, G.; Uysal, B.Z. Performance improvement in direct borohydride/peroxide fuel cells. Int. J. Hydrog. Energy 2017, 42, 8119–8129. [Google Scholar] [CrossRef]
- Demirci, U.B.; Akdim, O.; Andrieux, J.; Hannauer, J.; Chamoun, R.; Miele, P. Sodium Borohydride Hydrolysis as Hydrogen Generator: Issues, State of the Art and Applicability Upstream from a Fuel Cell. Fuel Cells 2010, 10, 335–350. [Google Scholar] [CrossRef] [Green Version]
- Akay, R.G.; Ata, K.C.; Kadıoğlu, T.; Çelik, C. Evaluation of SPEEK/PBI blend membranes for possible direct borohydride fuel cell (DBFC) application. Int. J. Hydrog. Energy 2018, 43, 18702–18711. [Google Scholar] [CrossRef]
- Ata, K.C.; Kadıoğlu, T.; Türkmen, A.C.; Çelik, C.; Akay, R.G. Investigation of the effects of SPEEK and its clay composite membranes on the performance of Direct Borohydride Fuel Cell. Int. J. Hydrog. Energy 2020, 45, 5430–5437. [Google Scholar] [CrossRef]
- Ma, J.; Choudhury, N.A.; Sahai, Y. A comprehensive review of direct borohydride fuel cells. Renew. Sustain. Energy Rev. 2010, 14, 183–199. [Google Scholar] [CrossRef]
- Ong, B.; Kamarudin, S.; Basri, S. Direct liquid fuel cells: A review. Int. J. Hydrog. Energy 2017, 42, 10142–10157. [Google Scholar] [CrossRef]
- Jimenez, I.M.; de León, C.P.; Shah, A.; Walsh, F. Developments in direct borohydride fuel cells and remaining challenges. J. Power Sources 2012, 219, 339–357. [Google Scholar] [CrossRef]
- Šljukić, B.; Morais, A.L.; Santos, D.M.F.; Sequeira, C.A.C. Anion- or Cation-Exchange Membranes for NaBH4/H2O2 Fuel Cells? Membranes 2012, 2, 478–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, D.M.F.; Sequeira, C.A.C. Effect of Membrane Separators on the Performance of Direct Borohydride Fuel Cells. J. Electrochem. Soc. 2011, 159, B126–B132. [Google Scholar] [CrossRef]
- Pandey, R.P.; Shukla, G.; Manohar, M.; Shahi, V.K. Graphene oxide based nanohybrid proton exchange membranes for fuel cell applications: An overview. Adv. Colloid Interface Sci. 2017, 240, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.-S.; Rick, J.; Hwang, B.-J. Water Soluble Polymers as Proton Exchange Membranes for Fuel Cells. Polymers 2012, 4, 913–963. [Google Scholar] [CrossRef] [Green Version]
- Gouda, M.H.; Elnouby, M.; Aziz, A.N.; Youssef, M.E.; Santos, D.M.F.; Elessawy, N. Green and Low-Cost Membrane Electrode Assembly for Proton Exchange Membrane Fuel Cells: Effect of Double-Layer Electrodes and Gas Diffusion Layer. Front. Mater. 2020, 6, 337. [Google Scholar] [CrossRef] [Green Version]
- Pourzare, K.; Farhadi, S.; Mansourpanah, Y. Advanced nanocomposite membranes for fuel cell applications: A comprehensive review. Biofuel Res. J. 2016, 3, 496–513. [Google Scholar] [CrossRef] [Green Version]
- Bakangura, E.; Wu, L.; Ge, L.; Yang, Z.; Xu, T. Mixed matrix proton exchange membranes for fuel cells: State of the art and perspectives. Prog. Polym. Sci. 2016, 57, 103–152. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, Y.; Wang, Y.; Chai, W.; Yang, M. Measurement and modeling of the effect of composition ratios on the properties of poly(vinyl alcohol)/poly(vinyl pyrrolidone) membranes. Mater. Des. 2016, 103, 249–258. [Google Scholar] [CrossRef]
- Maarouf, S.; Tazi, B.; Guenoun, F. Preparation and characterization of new composite membranes containing polyvinylpyrrolidone, polyvinyl alcohol, sulfosuccinic acid, silicotungstic acid and silica for direct methanol fuel cell applications. J. Mater. Environ. Sci. 2017, 8, 2870–2876. [Google Scholar]
- Nayak, R.; Ghosh, P.C.; Jana, T. Cross-Linked Polybenzimidazole Membrane for PEM Fuel Cells. ACS Appl. Polym. Mater. 2020, 2, 3161–3170. [Google Scholar] [CrossRef]
- Choudhury, N.A.; Ma, J.; Sahai, Y. High performance and eco-friendly chitosan hydrogel membrane electrolytes for direct borohydride fuel cells. J. Power Sources 2012, 210, 358–365. [Google Scholar] [CrossRef]
- Paseta, L.; Echaide-Górriz, C.; Téllez, C.; Coronas, J. Vapor phase interfacial polymerization: A method to synthesize thin film composite membranes without using organic solvents. Green Chem. 2021, 23, 2449–2456. [Google Scholar] [CrossRef]
- Topuz, F.; Holtzl, T.; Szekely, G. Scavenging organic micropollutants from water with nanofibrous hypercrosslinked cyclodextrin membranes derived from green resources. Chem. Eng. J. 2021, 419, 129443. [Google Scholar] [CrossRef]
- Park, S.-H.; Alammar, A.; Fulop, Z.; Pulido, B.A.; Nunes, S.P.; Szekely, G. Hydrophobic thin film composite nanofiltration membranes derived solely from sustainable sources. Green Chem. 2021, 23, 1175–1184. [Google Scholar] [CrossRef]
- Ong, C.; Falca, G.; Huang, T.; Liu, J.; Chisca, S. Green synthesis of thin film composite membranes for organic solvent nanofilteration. ACS Sustain. Chem. Eng. 2020, 8, 11541–11548. [Google Scholar] [CrossRef]
- Gouda, M.H.; Elessawy, N.A.; Santos, D.M. Synthesis and Characterization of Novel Green Hybrid Nanocomposites for Application as Proton Exchange Membranes in Direct Borohydride Fuel Cells. Energies 2020, 13, 1180. [Google Scholar] [CrossRef] [Green Version]
- Gouda, M.H.; Gouveia, W.; Elessawy, N.A.; Šljukić, B.; Nassr, A.B.A.A.; Santos, D.M.F. Simple design of PVA-based blend doped with SO4(PO4)-functionalised TiO2 as an effective membrane for direct borohydride fuel cells. Int. J. Hydrog. Energy 2020, 45, 15226–15238. [Google Scholar] [CrossRef]
- Gouda, M.; Gouveia, W.; Afonso, M.; Šljukić, B.; El Essawy, N.; Santos, D. Novel Ternary Polymer BlendMembranesDopedwith SO4/PO4-TiO2for Low Temperature Fuel Cells. In Proceedings of the 5th World Congress on Mechanical, Chemical, and Material Engineering, Lisbon, Portugal, 15–17 August 2019. [Google Scholar] [CrossRef]
- Deshmukh, R.R.; Ahamed, M.B.; Sadasivuni, K.K.; Ponnamma, D.; Pasha, S.K.K.; Almaadeed, M.A.-A.; Chidambaram, K. Graphene oxide reinforced polyvinyl alcohol/polyethylene glycol blend composites as high-performance dielectric material. J. Polym. Res. 2016, 23, 1–13. [Google Scholar] [CrossRef]
- Rochliadi, A.; Bundjali, B.; Arcana, I.M.; Dharmi, H. Polymer electrolyte membranes prepared by blending of poly(vinyl alcohol)-poly(ethylene oxide) for lithium battery application. In Proceedings of the Joint International Conference on Electric Vehicular Technology and Industrial, Mechanical, Electrical and Chemical Engineering (ICEVT & IMECE), Surakarta, Indonesia, 4–5 November 2015; pp. 370–373. [Google Scholar]
- Gouda, M.; Gouveia, W.; Afonso, M.; Šljukić, B.; El Essawy, N.; Nassr, A.; Santos, D. Poly(vinyl alcohol)-based crosslinked ternary polymer blend doped with sulfonated graphene oxide as a sustainable composite membrane for direct borohydride fuel cells. J. Power Sources 2019, 432, 92–101. [Google Scholar] [CrossRef]
- Gouda, M.; Konsowa, A.H.; Farag, H.A.; Elessawy, N.A.; Tamer, T.M.; Eldin, M.S.M. Novel nanocomposite membranes based on cross-linked eco-friendly polymers doped with sulfated titania nanotubes for direct methanol fuel cell application. Nanomater. Nanotechnol. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Sedesheva, Y.S.; Ivanov, V.S.; Wozniak, A.I.; Yegorov, A.S. Proton-Exchange Membranes Based on Sulfonated Polymers. Orient. J. Chem. 2016, 32, 2283–2296. [Google Scholar] [CrossRef]
- Awang, N.; Ismail, A.; Jaafar, J.; Matsuura, T.; Junoh, H.; Othman, M.H.D.; Rahman, M. Functionalization of polymeric materials as a high performance membrane for direct methanol fuel cell: A review. React. Funct. Polym. 2015, 86, 248–258. [Google Scholar] [CrossRef]
- Eldin, M.S.M.; Farag, H.A.; Tamer, T.M.; Konsowa, A.H.; Gouda, M. Development of novel iota carrageenan-g-polyvinyl alcohol polyelectrolyte membranes for direct methanol fuel cell application. Polym. Bull. 2019, 77, 4895–4916. [Google Scholar] [CrossRef]
- Benito, H.E.; Del Ángel Sánchez, T.; Alamilla, R.G.; Enríquez, J.M.H.; Robles, G.S.; Delgado, F.P. Synthesis and physicochemical characterization of titanium oxide and sulfated titanium oxide obtained by thermal hydrolysis of titanium tetrachloride. Braz. J. Chem. Eng. 2014, 31, 737–745. [Google Scholar] [CrossRef]
- Lu, M.; Wang, F.; Liao, Q.; Chen, K.; Qin, J.; Pan, S. FTIR spectra and thermal properties of TiO2-doped iron phosphate glasses. J. Mol. Struct. 2015, 1081, 187–192. [Google Scholar] [CrossRef]
- Goswami, P.; Ganguli, J. Synthesis characterization and photocatalytic reactions of phosphatedmesoporoustitania. Bull. Mater. Sci. 2012, 35, 889–896. [Google Scholar] [CrossRef]
- Pucić, I.; Jurkin, T. FTIR assessment of poly(ethylene oxide) irradiated in solid state, melt and aqeuous solution. Radiat. Phys. Chem. 2012, 81, 1426–1429. [Google Scholar] [CrossRef]
- Yu, X.; Qiang, L. Preparation for Graphite Materials and Study on Electrochemical Degradation of Phenol by Graphite Cathodes. Adv. Mater. Phys. Chem. 2012, 2, 63–68. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, P.N.; Dharmalingam, S. Effect of cation transport of SPEEK—Rutile TiO2 electrolyte on microbial fuel cell performance. J. Membr. Sci. 2015, 492, 518–527. [Google Scholar] [CrossRef]
- Ngai, K.S.; Ramesh, S.; Ramesh, K.; Juan, J.C. A review of polymer electrolytes: Fundamental, approaches and applications. Ionics 2016, 22, 1259–1279. [Google Scholar] [CrossRef]
- Kowsari, E.; Zare, A.; Ansari, V. Phosphoric acid-doped ionic liquid-functionalized graphene oxide/sulfonated polyimide composites as proton exchange membrane. Int. J. Hydrog. Energy 2015, 40, 13964–13978. [Google Scholar] [CrossRef]
- Bayer, T.; Cunning, B.V.; Selyanchyn, R.; Daio, T.; Nishihara, M.; Fujikawa, S.; Sasaki, K.; Lyth, S.M. Alkaline anion exchange membranes based on KOH-treated multilayer graphene oxide. J. Membr. Sci. 2016, 508, 51–61. [Google Scholar] [CrossRef] [Green Version]
- Pandey, R.; Shahi, V. Sulphonatedimidized graphene oxide (SIGO) based polymer electrolyte membrane for improved water retention, stability and proton conductivity. J. Power Sources 2015, 299, 104–113. [Google Scholar] [CrossRef]
- Shirdast, A.; Sharif, A.; Abdollahi, M. Effect of the incorporation of sulfonated chitosan/sulfonated graphene oxide on the proton conductivity of chitosan membranes. J. Power Sources 2016, 306, 541–551. [Google Scholar] [CrossRef]
- Beydaghi, H.; Javanbakht, M.; Kowsari, E. Synthesis and Characterization of Poly(vinyl alcohol)/Sulfonated Graphene Oxide Nanocomposite Membranes for Use in Proton Exchange Membrane Fuel Cells (PEMFCs). Ind. Eng. Chem. Res. 2014, 53, 16621–16632. [Google Scholar] [CrossRef]
- Cheng, T.; Feng, M.; Huang, Y.; Liu, X. SGO/SPEN-based highly selective polymer electrolyte membranes for direct methanol fuel cells. Ionics 2017, 23, 2143–2152. [Google Scholar] [CrossRef]
- Luo, T.; Xu, H.; Li, Z.; Gao, S.; Fang, Z.; Zhang, Z.; Wang, F.; Ma, B.; Zhu, C. Novel proton conducting membranes based on copolymers containing hydroxylated poly(ether ether ketone) and sulfonated polystyrenes. J. Appl. Polym. Sci. 2017, 134, 1–8. [Google Scholar] [CrossRef]
- Yuan, C.; Wang, Y. The preparation of novel sulfonated poly(aryl ether ketone sulfone)/TiO2 composite membranes with low methanol permeability for direct methanol fuel cells. High Perform. Polym. 2021, 33, 326–337. [Google Scholar] [CrossRef]
- Yang, C.; Chien, W.; Li, Y.J. Direct methanol fuel cell based on poly(vinyl alcohol)/titanium oxide nanotubes/poly(styrene sulfonic acid) (PVA/nt-TiO2/PSSA) composite polymer membrane. J. Power Sources 2010, 195, 3407–3415. [Google Scholar] [CrossRef]
- Ahmad, H.; Kamarudin, S.; Hasran, U.; Daud, W. A novel hybrid Nafion-PBI-ZP membrane for direct methanol fuel cells. Int. J. Hydrog. Energy 2011, 36, 14668–14677. [Google Scholar] [CrossRef]







| Membrane | PVA: PEO wt% | PO4-TiO2 Nanotubes wt% Respect to PVA |
|---|---|---|
| SPVA/PEO | 85:15 | 0 |
| SPVA/PEO/PO4TiO2-1 | 85:15 | 1 |
| SPVA/PEO/PO4TiO2- 2 | 85:15 | 2 |
| SPVA/PEO/PO4TiO2- 3 | 85:15 | 3 |
| Membrane | Thickness (µm) | WU (%) | SR (%) | Contact Angle (°) | Tensile Strength (MPa) | Oxidative Stability (RW, %) * |
|---|---|---|---|---|---|---|
| SPVA/PEO | 130 | 95 ± 0.5 ** | 90 ± 0.3 | 65.36 ± 1.5° | 15.5 ± 0.5 | 90 ± 2 |
| SPVA/PEO/PO4TiO2-1 | 150 | 40 ± 0.3 | 42 ± 0.3 | 67.23 ± 1.5° | 24.9 ± 0.7 | 94 ± 1.5 |
| SPVA/PEO/PO4TiO2-2 | 175 | 22 ± 0.2 | 13 ± 0.2 | 70.36 ± 1.7° | 32.5 ± 1 | 98 ± 1.5 |
| SPVA/PEO/PO4TiO2-3 | 184 | 16 ± 0.03 | 10 ± 0.1 | 72.30 ± 1.5° | 40.3 ± 1.5 | 99 ± 0.5 |
| Nafion 117 | 183 | 15 | 8 | 102 | 25 | 92 |
| Membrane | IEC (meq g−1) | Ionic Conductivity (mS cm−1) | Borohydride Permeability (10−6 cm2 s−1) | Selectivity (105 S cm−3 s) |
|---|---|---|---|---|
| SPVA/PEO | 0.20 ± 0.01 * | 12 ± 0.05 | 16 | 0.007 |
| SPVA/PEO/PO4TiO2-1 | 0.35 ± 0.01 | 17.7 ± 0.05 | 0.75 | 0.23 |
| SPVA/PEO/PO4TiO2-2 | 0.45 ± 0.01 | 20.5 ± 0.05 | 0.36 | 0.56 |
| SPVA/PEO/PO4TiO2-3 | 0.60 ± 0.01 | 28 ± 0.03 | 0.10 | 2.80 |
| Nafion 117 | 0.89 | 45.0 | 0.40 | 1.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouda, M.H.; Elessawy, N.A.; Toghan, A. Novel Crosslinked Sulfonated PVA/PEO Doped with Phosphated Titanium Oxide Nanotubes as Effective Green Cation Exchange Membrane for Direct Borohydride Fuel Cells. Polymers 2021, 13, 2050. https://doi.org/10.3390/polym13132050
Gouda MH, Elessawy NA, Toghan A. Novel Crosslinked Sulfonated PVA/PEO Doped with Phosphated Titanium Oxide Nanotubes as Effective Green Cation Exchange Membrane for Direct Borohydride Fuel Cells. Polymers. 2021; 13(13):2050. https://doi.org/10.3390/polym13132050
Chicago/Turabian StyleGouda, Marwa H., Noha A. Elessawy, and Arafat Toghan. 2021. "Novel Crosslinked Sulfonated PVA/PEO Doped with Phosphated Titanium Oxide Nanotubes as Effective Green Cation Exchange Membrane for Direct Borohydride Fuel Cells" Polymers 13, no. 13: 2050. https://doi.org/10.3390/polym13132050
APA StyleGouda, M. H., Elessawy, N. A., & Toghan, A. (2021). Novel Crosslinked Sulfonated PVA/PEO Doped with Phosphated Titanium Oxide Nanotubes as Effective Green Cation Exchange Membrane for Direct Borohydride Fuel Cells. Polymers, 13(13), 2050. https://doi.org/10.3390/polym13132050