Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization
Abstract
1. Introduction
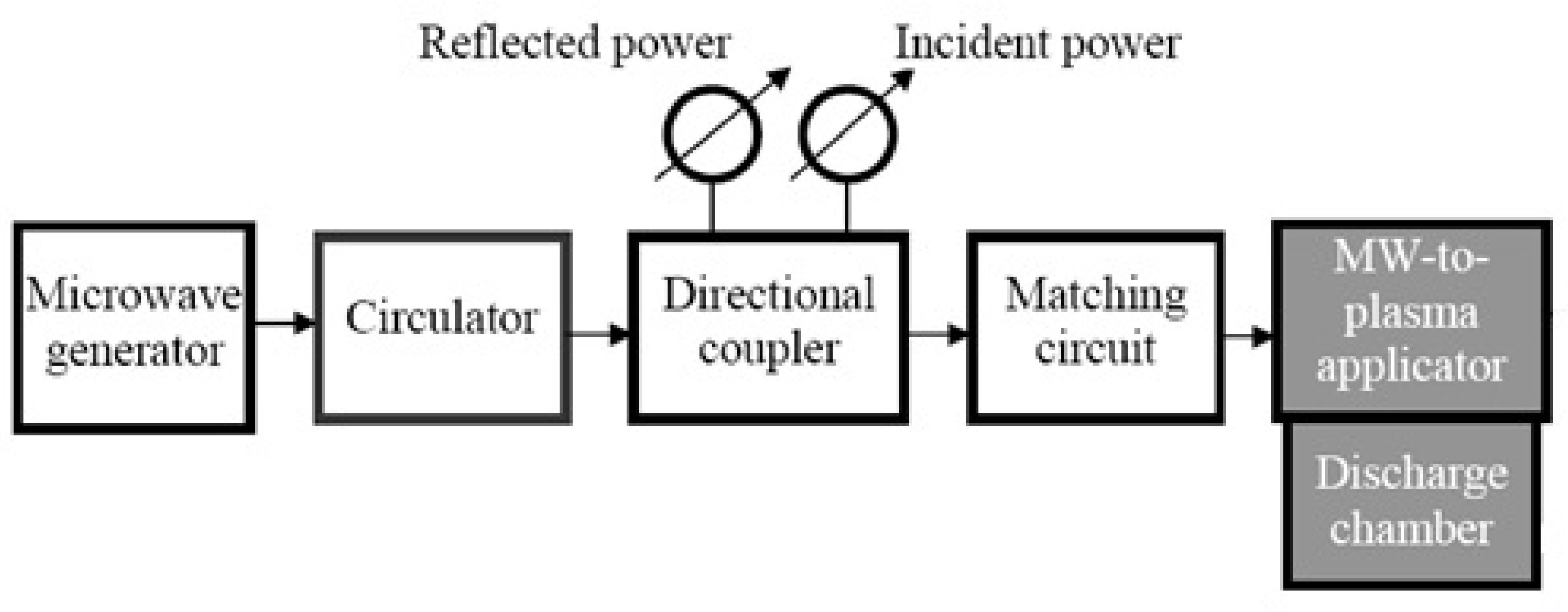
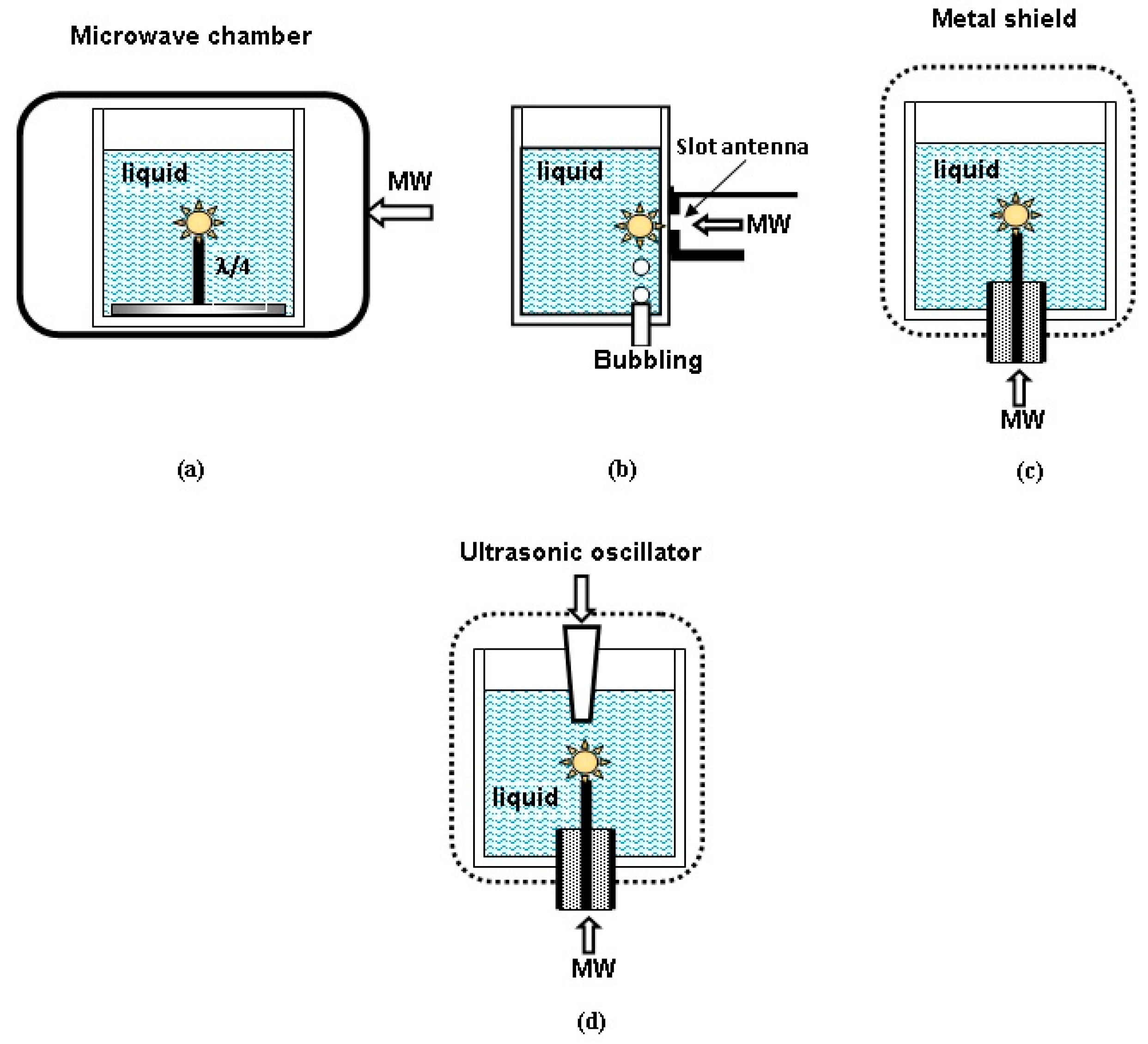
2. Experimental Setups and Methods of Diagnostics
3. Gas Products of Microwave Discharges in Liquid Hydrocarbons
- Increase of the molecular weight of alkanes is accompanied by increase in the yield of acetylene and decrease in the yield of hydrogen (in the series C7–C16, including cycloalkanes).
- Hydrogen and acetylene are predominantly formed in aromatic compounds.
- Gas products of microwave discharge in the investigated liquid cycloalkanes and aromatic compounds without radical groups practically do not contain methane or ethylene. As the number of radical groups increases, the composition approaches the composition of the discharge products in alkanes.
- (i)
- Electron-impact dissociation occurs only in a narrow region adjacent to the electrode because the microwave field is concentrated near the end of central electrode and falls rapidly with increasing distance from it.
- (ii)
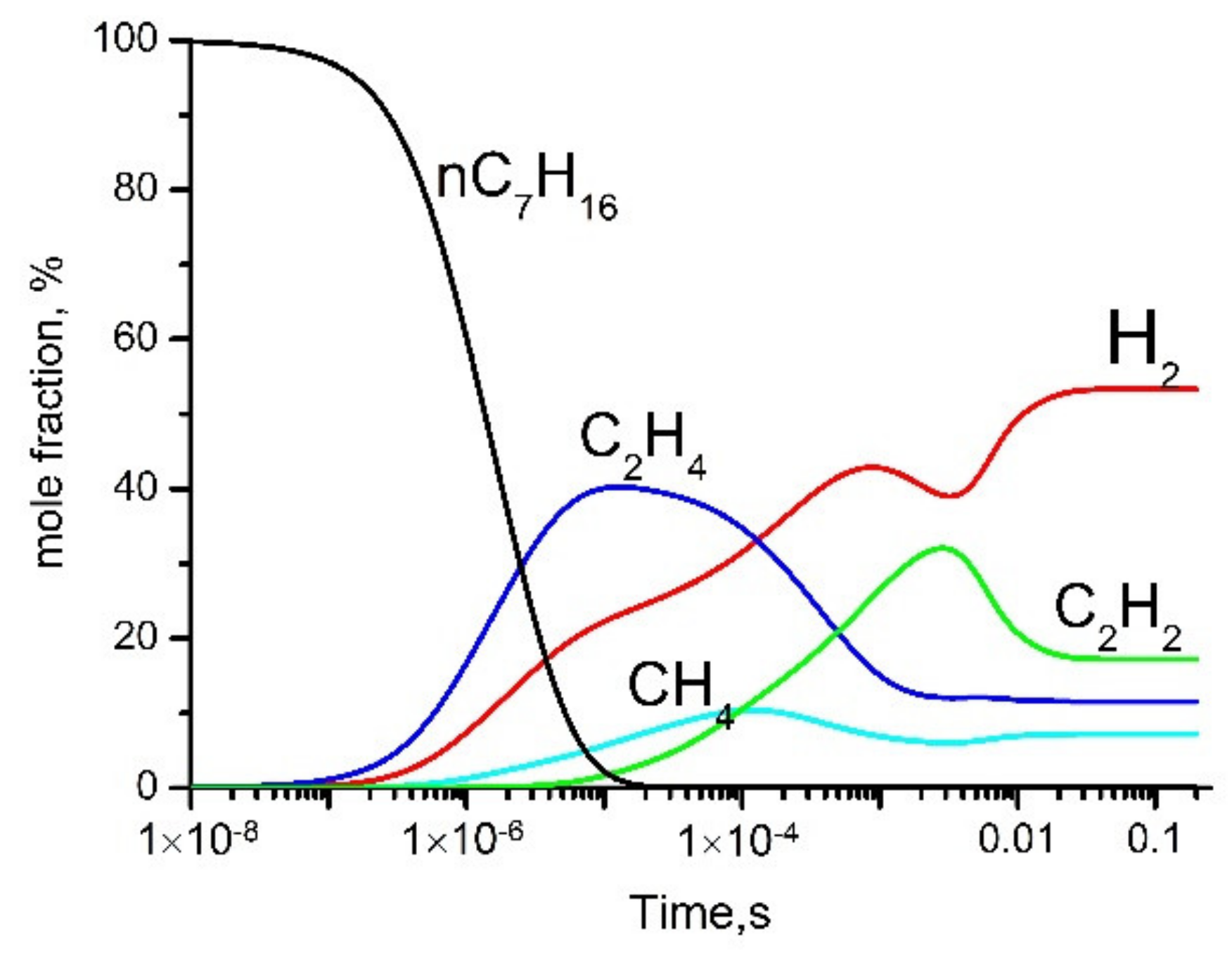
- Electron impact affects the dissociation of n-heptane only during a short time period from discharge ignition (≤10–3 s), when the gas temperature is low (<1300 K).
- (i)
- At times greater than 10–3 s, the dissociation of n-heptane occurs thermally. The role of plasma electrons in the decomposition of n-heptane is reduced to heating the gas inside the plasma bubble (Figure 6).
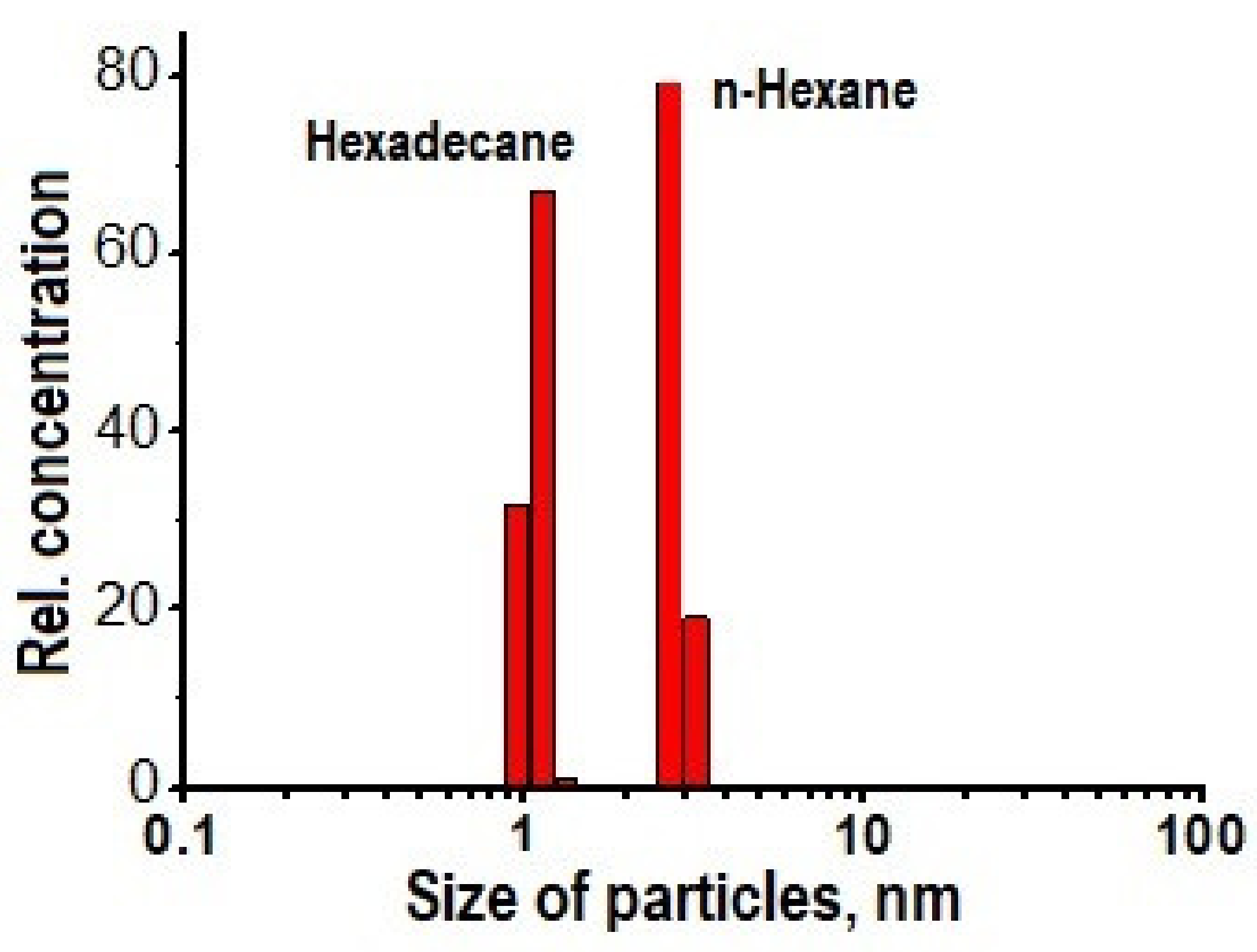
4. Solid Products of Microwave Discharges in Liquid Hydrocarbons
5. Liquid Hydrocarbons after Treatment by Microwave Discharge
6. Basic Information about Plasma Properties (Diagnostics and Modeling)
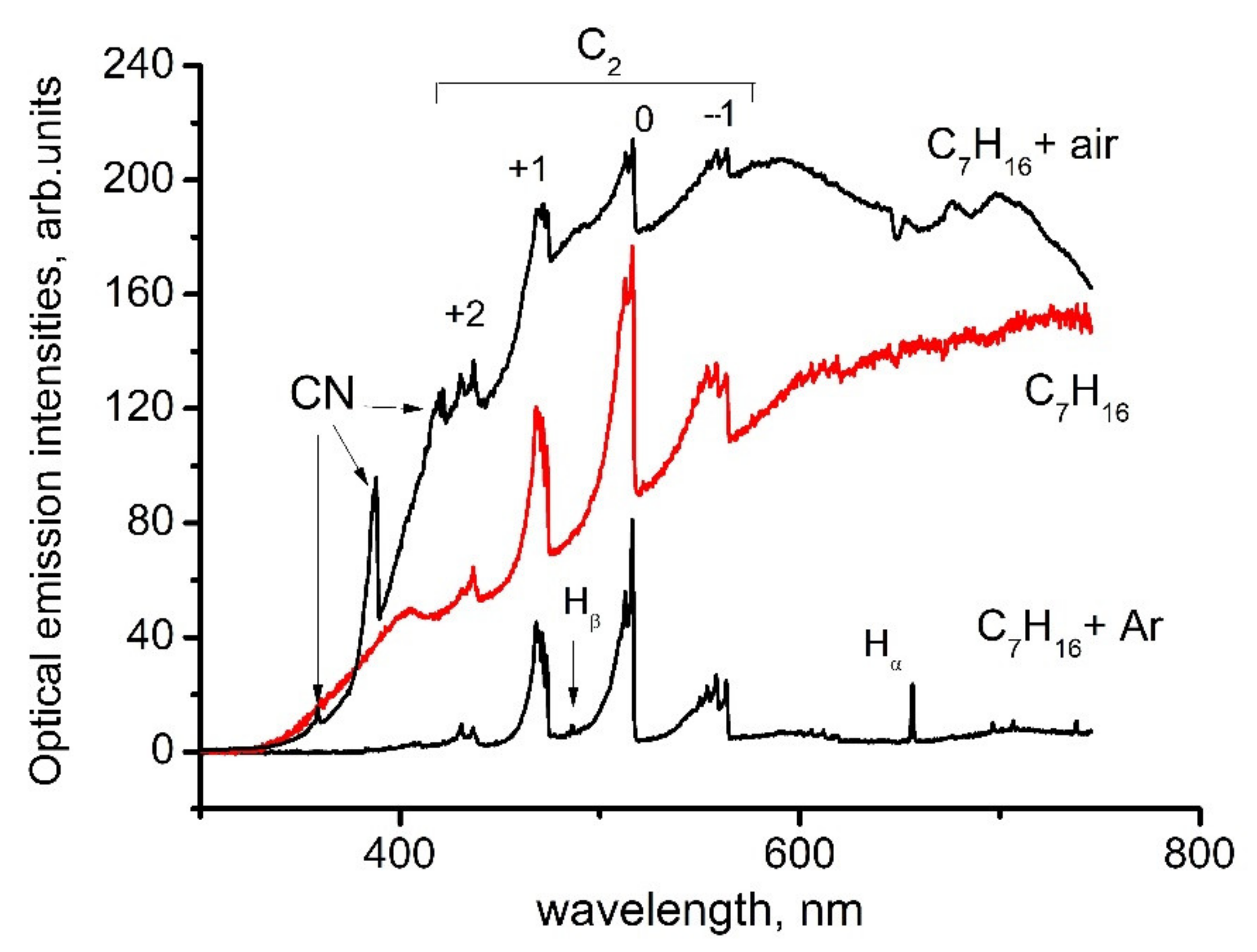
6.1. Diagnostics
6.2. Modeling
6.2.1. 2D Model: Problem Formulation and Main Results
- Gas bubbles are created by evaporation of liquid into the bubble.
- In plasma formed inside the bubble, the main ion is C7H16+.
- Heating of the bubble is due to Joule heating of plasma electrons.
- Cooling of the bubble is due to energy transfer to ambient liquid for evaporation and endothermic pyrolysis of n-heptane.
- The size and shape of the bubbles is determined by the surface tension and the sum of the pressure forces.
- Lifting of the bubble is determined by the Archimedean force and viscosity.
- The initial temperature of the liquid and gas phase is equal to the boiling point.
- A small bubble of evaporated gas of atmospheric pressure is set inside the near-electrode cavity.
- Boiling process depends on the input power. There is a certain range of input power, when there is a regime of periodic formation of bubbles and their further rise.
- The plasma burns only inside the bubble, in close proximity to the central electrode. When a bubble floats up, plasma disappears inside it. This is due to the fact that the microwave field is concentrated near the end of the central electrode and falls very sharply outside it. At a power >500 W, the microwave field at the electrode end reaches 10 kV/cm. The temperature in this area is about 1500–1700 K, which is in good agreement with our experimental data. The maximum electron density is about 1014 cm−3.
- As the bubble rises, it cools very quickly due to energy transfer to ambient boiling liquid needed for its evaporation. Thus, temperature of the rising bubble very soon becomes approximately equal to the boiling point.
- For times less than 10−3, the dissociation of n-heptane occurs under the action of electron impact, and then is dominated by thermal dissociation. The role of the plasma is reduced to the formation of gas bubbles and initiation of thermal reactions.
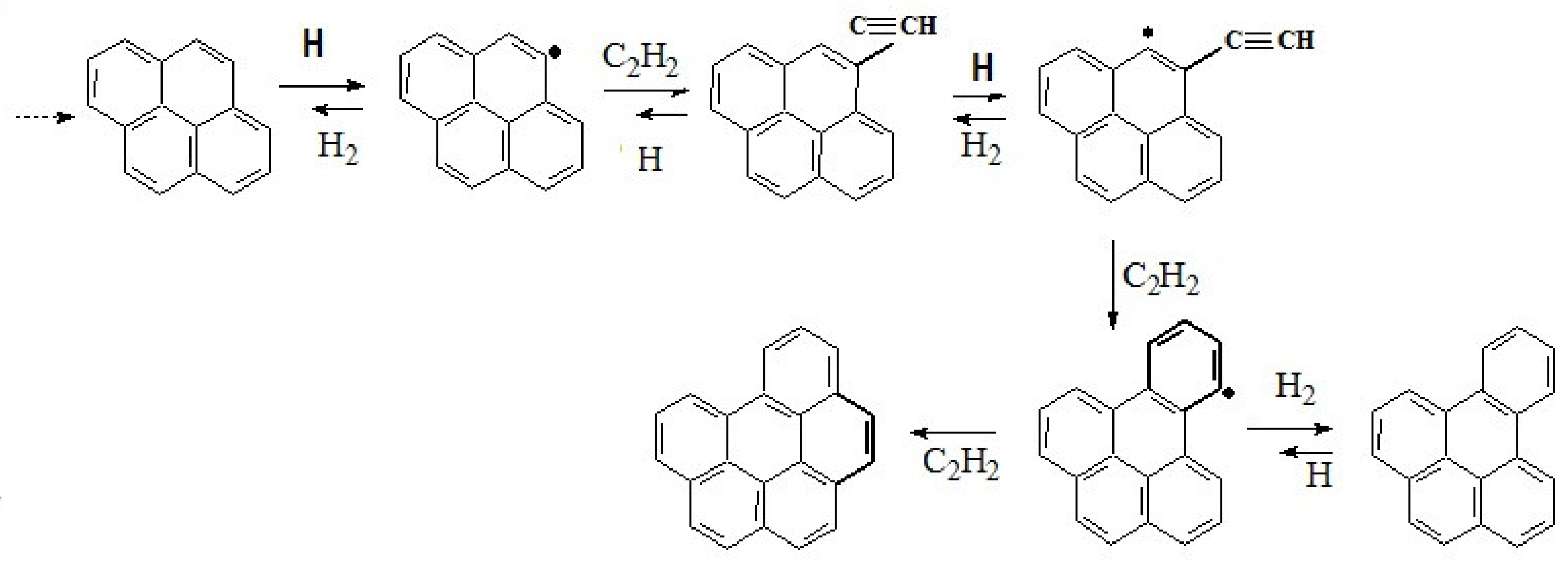
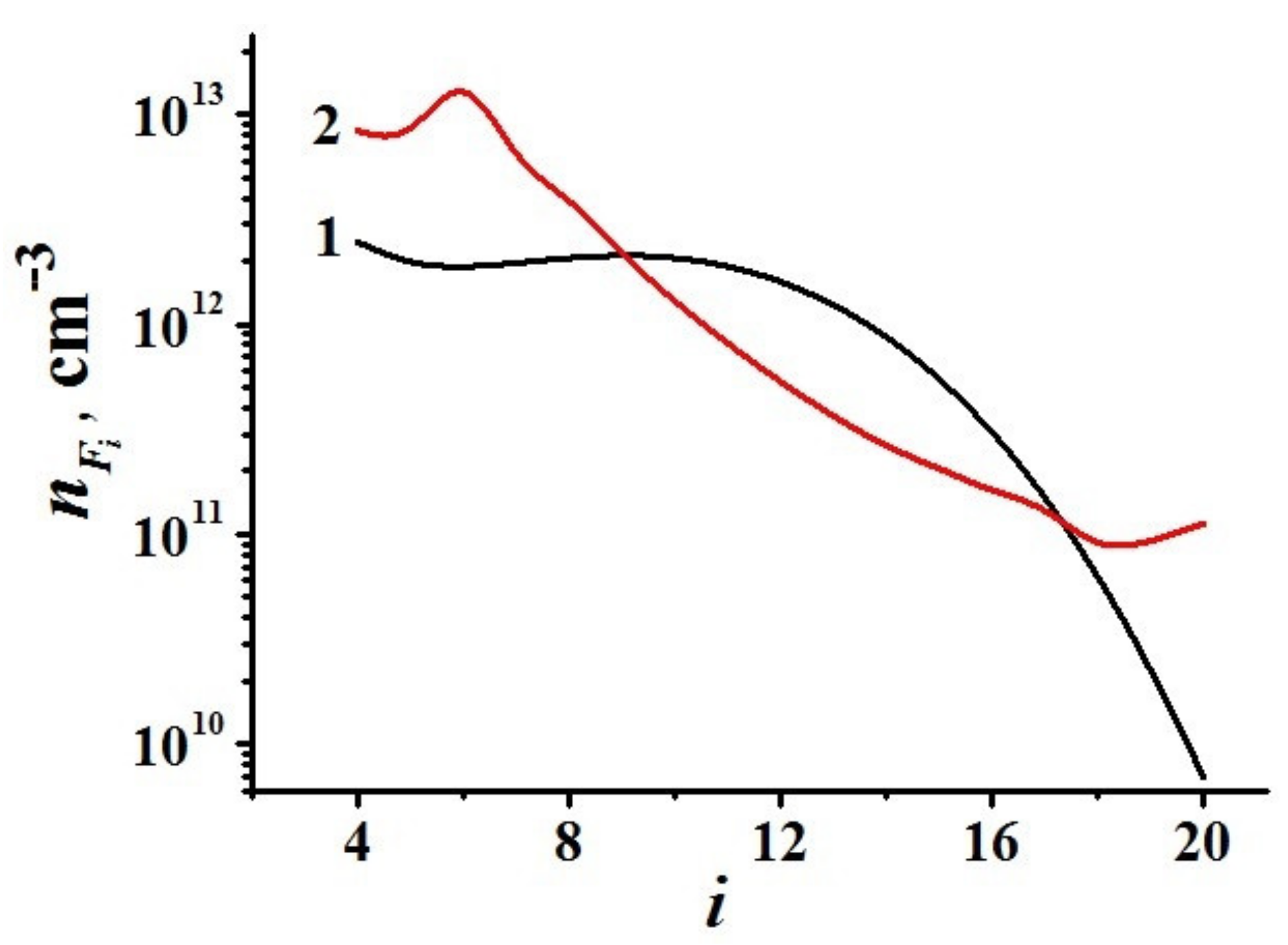
6.2.2. Modeling of Carbonaceous Particle Growth
6.2.3. The Role of Argon Bubbling and Charging of Carbonaceous Particle (0D Models)
7. Conclusions
8. Patents
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bruggeman, P.; Leys, C. Non-thermal plasmas in and in contact with liquids. J. Phys. D Appl. Phys. 2009, 42, 053001. [Google Scholar] [CrossRef]
- Samukawa, S.; Hori, M.; Rauf, S.; Tachibana, K.; Bruggeman, P.; Kroesen, G.; Whitehead, J.C.; Murphy, A.B.; Gutsol, A.F.; Starikovskaia, S.; et al. The 2012 Plasma Roadmap. J. Phys. D Appl. Phys. 2012, 45, 253001. [Google Scholar] [CrossRef]
- Bruggeman, P.J.; Kushner, M.J.; Locke, B.R.; Gardeniers, J.G.; Graham, W.G.; Graves, D.B.; Hofman-Caris, R.C.; Maric, D.; Reid, J.P.; Ceriani, E.; et al. Plasma–liquid interactions: A review and roadmap. Plasma Sources Sci. Technol. 2016, 25, 053002. [Google Scholar] [CrossRef]
- Adamovich, I.; Baalrud, S.D.; Bogaerts, A.; Bruggeman, P.J.; Cappelli, M.; Colombo, V.; Czarnetzki, U.; Ebert, U.; Eden, J.G.; Favia, P.; et al. The 2017 Plasma Roadmap: Low temperature plasma science and technology. J. Phys. D Appl. Phys. 2017, 50, 323001. [Google Scholar] [CrossRef]
- Malik, M.A.; Ghaffar, A.; Malik, S.A. Water purification by electrical discharges. Plasma Sources Sci. Technol. 2001, 10, 82–91. [Google Scholar] [CrossRef]
- Foster, J. Plasma-based water purification: Challenges and prospects for the future. Phys. Plasmas 2017, 24, 055501. [Google Scholar] [CrossRef]
- Rezaei, F.; Vanraes, P.; Nikiforov, A.; Morent, R.; De Geyter, N. Applications of Plasma-Liquid Systems: A Review. Materials 2019, 12, 2751. [Google Scholar] [CrossRef] [PubMed]
- Locke, B.R. Environmental Applications of Electrical Discharge Plasma with Liquid Water—A Mini Review. Int. J. Plasma Environ. Sci. Technol. 2012, 6, 194–203. [Google Scholar]
- Rybkin, V.V.; Shutov, D.A. Atmospheric-Pressure Electric Discharge as an Instrument of Chemical Activation of Water Solutions. Plasma Phys. Rep. 2017, 43, 1089–1113. [Google Scholar] [CrossRef]
- Vanraes, P.; Bogaerts, A. Plasma physics of liquids—A focused review. Appl. Phys. Rev. 2018, 5, 031103. [Google Scholar] [CrossRef]
- Lebedev, Y.A. Microwave Discharges in Liquid Dielectrics. Plasma Phys. Rep. 2017, 43, 685–695. [Google Scholar] [CrossRef]
- Horikoshi, S.; Serpone, N. In-liquid plasma: A novel tool in the fabrication of nanomaterials and in the treatment of wastewaters. RSC Adv. 2017, 7, 47196–47218. [Google Scholar] [CrossRef]
- Lebedev, Y.A. Microwave Discharges in Liquids: Fields of Applications. High Temp. 2018, 56, 811–820. [Google Scholar] [CrossRef]
- Hattori, Y.; Mukasa, S.; Nomura, S.; Toyota, H. Optimization and analysis of shape of coaxial electrode for microwave plasma in water. J. Appl. Phys. 2010, 107, 063305. [Google Scholar] [CrossRef]
- Hattori, Y.; Mukasa, S.; Toyota, H.; Yamashita, H.; Nomura, S. Improvement in preventing metal contamination from an electrode used for generating microwave plasma in liquid. Surf. Coat. Technol. 2012, 206, 2140–2445. [Google Scholar] [CrossRef]
- Mukasa, S.; Nomura, S.; Toyota, H. Observation of Microwave In-Liquid Plasma using High-Speed Camera. Jan. J. Appl. Phys. 2007, 46, 6015–6021. [Google Scholar] [CrossRef]
- Hattori, Y.; Mukasa, S.; Toyota, H.; Nomura, S. Electrical breakdown of microwave plasma in water. Curr. Appl. Phys. 2013, 13, 1050–1054. [Google Scholar] [CrossRef]
- Gidalevich, E.; Boxman, R.L. Microwave excitation of submerged plasma bubbles. J. Phys. D Appl. Phys. 2012, 45, 245204. [Google Scholar] [CrossRef]
- Tatarinov, A.V.; Lebedev, Y.A.; Epstein, I.L. Simulation of Microwave-induced Formation of Gas Bubbles in Liquid n-Heptane. High Energy Chem. 2016, 50, 144–149. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L.; Averin, K.A. The Formation of Gas Bubbles by Processing of Liquid n-Heptane in the Microwave Discharge. Plasma Chem. Plasma Proc. 2016, 36, 535–552. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L.; Bilera, I.V. A 0D kinetic model for the microwave discharge in liquid n-heptane including carbonaceous particles production. J. Phys. D Appl. Phys. 2018, 51, 214007. [Google Scholar]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L. 1D Modeling of the Microwave Discharge in Liquid n-Heptane Including Production of Carbonaceous Particles. Plasma Chem. Plasma Proc. 2019, 39, 787–808. [Google Scholar] [CrossRef]
- Levko, D.; Sharma, A.; Raja, L.L. Plasmas generated in bubbles immersed in liquids: Direct current streamers versus microwave plasma. J. Phys. D Appl. Phys. 2016, 49, 285205. [Google Scholar] [CrossRef]
- Levko, D.; Sharma, A.; Raja, L.L. Microwave plasmas generated in bubbles immersed in liquids for hydrocarbons reforming. J. Phys. D Appl. Phys. 2016, 49, 22LT01. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L. Simulation of Microwave Discharge in Liquid n-Heptane in the Presence of Argon in the Discharge Region. High Energy Chem. 2020, 54, 217–226. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L. Effect of charging solid particles on their growth process and parameters of microwave discharge in liquid n-heptane. Plasma Sources Sci. Technol. 2020, 29, 065013. [Google Scholar] [CrossRef]
- Lebedev, Y.A. Microwave discharges at low pressures and peculiarities of the processes in strongly non-uniform plasma. Plasma Sources Sci. Technol. 2015, 24, 053001. [Google Scholar] [CrossRef]
- Nomura, S.; Toyota, H.; Mukasa, S.; Yamashita, H.; Maehara, T.; Kawashima, A. Production of hydrogen in a conventional microwave oven. J. Appl. Phys. 2009, 106, 073306. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Konstantinov, V.S.; Yablokov, M.Y.; Shchegolikhin, A.N.; Surin, N.M. Microwave Plasma in Liquid n-Heptane: A Study of Plasma Chemical Reaction Products. High Energy Chem. 2014, 48, 385–388. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Epstein, I.L.; Shakhatov, V.A.; Yusupova, E.V.; Konstantinov, V.S. Spectroscopy of Microwave Discharge in Liquid C7–C16 Hydrocarbons. High Temp. 2014, 52, 319–327. [Google Scholar] [CrossRef]
- Averin, K.A.; Lebedev, Y.A.; Shchegolikhin, A.N.; Yablokov, M.Y. Nanosize Carbon Products Formed in Microwave Discharge in Liquid Alkanes. Plasma Proc. Polym. 2017, 14, e201600227. [Google Scholar] [CrossRef]
- Horikoshi, S.; Sawada, S.; Sato, S.; Serpone, N. Microwave-Driven In-liquid Plasma in Chemical and Environmental Applications. III. Examination of Optimum Microwave Pulse Conditions for Prolongation of Electrode Lifetime, and Application to Dye Contaminated Wastewater. Plasma Chem. Plasma Proc. 2019, 39, 51–62. [Google Scholar] [CrossRef]
- Toyota, H.; Nomura, S.; Mukasa, S. A practical electrode for microwave plasma processes. Int. J. Mater. Sci. Appl. 2013, 2, 83–88. [Google Scholar] [CrossRef]
- Ishijima, T.; Hotta, H.; Sugai, H. Multibubble plasma production and solvent decomposition in water by slot-excited microwave discharge. Appl. Phys. Lett. 2007, 91, 121501. [Google Scholar] [CrossRef]
- Ishijima, T.; Sugiura, H.; Satio, R.; Toyada, H.; Sugai, H. Efficient production of microwave bubble plasma in water for plasma processing in liquid. Plasma Sources Sci. Technol. 2010, 19, 015010. [Google Scholar] [CrossRef]
- Ishijima, T.; Nosaka, K.; Tanaka, Y.; Uesugi, Y.; Goto, Y.; Horibe, H. A high-speed photoresist removal process using multibubble microwave plasma under a mixture of multiphase plasma environment. Appl. Phys. Lett. 2013, 103, 142101. [Google Scholar] [CrossRef]
- Wang, B.; Sun, B.; Zhu, X.; Yan, Z.; Liu, Y.; Liu, H. Effect of reactor parameters on matching properties of microwave discharge in liquid. J. Phys. Conf. Ser. 2013, 418, 012099. [Google Scholar] [CrossRef]
- Wang, B.; Sun, B.; Zhu, X.; Yan, Z.; Liu, Y.; Liu, H. Degradation of Methylene Blue by Microwave Discharge Plasma in Liquid. Contrib. Plasma Phys. 2013, 53, 697–702. [Google Scholar] [CrossRef]
- Shu, T.; Sun, B.; Zhu, X.; Wang, L.; Xin, Y.; Liu, J. Mechanism analysis of hydrogen production by microwave discharge in ethanol liquid. J. Anal. And Appl. Pyrolysis 2021, 156, 105111. [Google Scholar]
- Wang, B.; Sun, B.; Zhu, X.; Yan, Z.; Liu, Y.; Liu, H.; Lui, Q. Hydrogen production from alcohol solution by microwave discharge in liquid. Int. J. Hydrogen Energy 2016, 4, 7280–7291. [Google Scholar] [CrossRef]
- Averin, K.A.; Lebedev, Y.A.; Shakhatov, V.A. Some results of study of a microwave discharge in heavy liquid hydrocarbons. Plasma Phys. Rep. 2018, 44, 110–113. [Google Scholar] [CrossRef]
- Hattory, Y.; Mukasa, S.; Toyota, H.; Inoue, T.; Nomura, S. Synthesis of zinc and zinc oxide nanoparticles from zinc electrode using plasma in liquid. Mater. Lett. 2011, 65, 188–190. [Google Scholar] [CrossRef]
- Averin, K.A.; Bilera, I.V.; Lebedev, Y.A.; Shakhatov, V.A.; Epstein, I.L. Microwave discharge in liquid n-heptane with and without bubble flow of argon. Plasma Process Polym. 2019, 16, e1800198. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Averin, K.A.; Tatarinov, A.V. Main Gaseous Products of Microwave Discharge in Various Liquid Hydrocarbons. High Energy Chem. 2019, 54, 331–335. [Google Scholar]
- Lebedev, Y.A.; Shakhatov, V.A. Optical emission spectra of microwave discharge in different liquid hydrocarbons. Plasma Process Polym. 2020, 17, e2000003. [Google Scholar] [CrossRef]
- Nomura, S.; Toyota, H. Sonoplasma generated by a combination of ultrasonic waves and microwave irradiation. Appl. Phys. Lett. 2003, 83, 4503–4505. [Google Scholar] [CrossRef]
- Mukasa, S.; Nomura, S.; Toyota, H. Measurement of Temperature in Sonoplasma. Jpn. J. Appl. Phys. 2004, 43, 2833–2837. [Google Scholar] [CrossRef]
- Nomura, S.; Toyota, H.; Tawara, M.; Yamashota, H. Fuel gas production by microwave plasma in liquid. Appl. Phys. Lett. 2006, 88, 231502. [Google Scholar] [CrossRef]
- Curran, H.J.; Gaffuri, P.; Pitz, W.J.; Westbrook, C.K. A comprehensive modeling study of n-heptane oxidation. Combust. Flame 1998, 114, 149–177. [Google Scholar] [CrossRef]
- Simmie, J.M. Detailed chemical kinetic models for the combustion of hydrocarbon fuels. Prog. Energy Combust. Sci. 2003, 29, 599–634. [Google Scholar] [CrossRef]
- Ahmed, S.S.; Mauss, F.; Moreac, G.; Zeuch, T. A comprehensive and compact n-heptane oxidation model derived using chemical lumping. Phys. Chem. Chem. Phys. 2007, 9, 1107–1126. [Google Scholar] [CrossRef]
- Seidel, L.; Moshammer, K.; Wang, X.; Zeuch, T.; Kohse-Höinghaus, K.; Mauss, F. Comprehensive kinetic modeling and experimental study of a fuel-rich, premixed n-heptane flame. Combust. Flame 2015, 162, 2045–2058. [Google Scholar] [CrossRef]
- Rebick, C. Pyrolysis of Heavy Hydrocarbons in Pyrolysis. In Theory and Industrial Practice; Albright, L.F., Crynes, B.L., Corcoran, W.H., Eds.; Academic Press: Cambridge, MA, USA, 1983; pp. 69–87. [Google Scholar]
- Ranzi, E.; Dente, M.; Pierucci, S.; Biardi, G. Initial product distributions from pyrolysis of normal and branched paraffins. Ind. Eng. Chem. Fundam. 1983, 22, 132–139. [Google Scholar] [CrossRef]
- Safarik, I.; Strausz, O.P. The thermal decomposition of hydro-carbons. Part 1. n-alkanes (C ≥ 5). Res. Chem. Intermed. 1996, 22, 275–314. [Google Scholar] [CrossRef]
- Held, T.J.; Marchese, A.J.; Dryer, F.L. A semi-empirical reaction mechanism for n-heptane oxidation and pyrolysis. Combust. Sci. Technol. 1997, 123, 107–146. [Google Scholar] [CrossRef]
- Murata, M.; Saito, S. Prediction of initial product distribution from n-paraffin pyrolysis at higher temperatures by considering ethyl radical decomposition. J. Chem. Eng. Jpn. 1974, 7, 389–391. [Google Scholar] [CrossRef][Green Version]
- Zamostny, P.; Belohlav, Z.; Starkbaumova, L.; Patera, J. Experimental study of hydrocarbon structure effects on the composition of its pyrolysis products. J. Anal. Appl. Pyrolysis 2010, 87, 207–216. [Google Scholar] [CrossRef]
- Chakraborty, J.P.; Kunzru, D. High pressure pyrolysis of n-heptane. J. Anal. Appl. Pyrolysis 2009, 86, 44–52. [Google Scholar] [CrossRef]
- Yuan, T.; Zhang, L.; Zhou, Z.; Xie, M.; Ye, L.; Qi, F. Pyrolysis of n-Heptane: Experimental and Theoretical Study. J. Phys. Chem. A 2011, 115, 1593–1601. [Google Scholar] [CrossRef]
- Bajus, M.; Veselý, V.; Leclercq, P.A.; Rijks, J.A. Steam Cracking of Hydrocarbons. 1. Pyrolysis of Heptane. Ind. Eng. Chem. Prod. Res. Dev. 1979, 18, 30–37. [Google Scholar] [CrossRef]
- Olahová, N.; Bajus, M.; Hájeková, E.; Šugár, L.; Markoš, J. Kinetics and modelling of heptane steam–cracking. Chem. Pap. 2014, 68, 1678–1689. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, X.; Song, Q.; Zhao, L.; Su, H.; Li, H.; Zeng, X.; Zhao, D.; Xu, J. The effect of temperature and pressure on n-heptane thermal cracking in regenerative cooling channel. Combust. Flame 2018, 194, 233–244. [Google Scholar] [CrossRef]
- Garner, S.; Sivaramakrishnan, R.; Brezinsky, K. The high-pressure pyrolysis of saturated and unsaturated C7 hydrocarbons. Proc. Combust. Inst. 2009, 32, 461–467. [Google Scholar] [CrossRef]
- Yasunaga, K.; Yamada, H.; Oshita, H.; Hattori, K.; Hidaka, Y.; Curran, H. Pyrolysis of n-pentane, n-hexane and n-heptane in a single pulse shock tube. Combust. Flame 2017, 185, 335–345. [Google Scholar] [CrossRef]
- Dey, G.R.; Singh, B.N.; Kumar, S.D.; Das, T.N. Dielectric Barrier Discharge Initiated Gas-Phase Decomposition of CO2 to CO and C6–C9 Alkanes to C1–C3 Hydrocarbons on Glass, Molecular Sieve 10X and TiO2/ZnO Surfaces. Plasma Chem. Plasma Process. 2007, 27, 669–679. [Google Scholar] [CrossRef]
- Taghvaei, H.; Jahanmiri, A.; Rahimpour, M.R.; Shirazi, M.M.; Hooshmand, N. Hydrogen production through plasma cracking of hydrocarbons: Effect of carrier gas and hydrocarbon type. Chem. Eng. J. 2013, 226, 384–392. [Google Scholar] [CrossRef]
- Yao, S.; Weng, S.; Jin, Q.; Wu, Z.; Jiang, B.; Tang, X.; Lu, H.; Han, J.; Cao, Y.; Zhang, X. Comparison of Gasoline-Ranged n-Alkanes Conversions Using Dielectric Barrier Discharge: A Kinetic Study. Plasma Chem. Plasma Process. 2017, 37, 137–148. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Tatarinov, A.V.; Epstein, I.L. On the role of electron impact in an atmospheric pressure microwave discharge in liquid n-heptane. Plasma Phys. Rep. 2017, 43, 510–513. [Google Scholar] [CrossRef]
- Belousov, V.P.; Belousova, I.M.; Ermakov, A.V.; Kiselev, V.M.; Sosnov, E.N. Oxygen adsorption by fullerenes and carbon nanostructures. Russ. J. Phys. Chem. A 2007, 81, 1650–1657. [Google Scholar] [CrossRef]
- Averin, K.A.; Lebedev, Y. A Redistribution of Metal Concentrations in the Products of Microwave Discharge in Liquid Residues of Heavy Petroleum Feedstock. High Energy Chem. 2018, 52, 263–265. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Averin, K.A. Extraction of valuable metals by microwave discharge in crude oil. J. Phys. D Appl. Phys. 2018, 51, 214005. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Khadzhiev, S.N.; Kadiev, K.M.; Averin, K.A.; Visaliev, M.Y.; Mokochunina, T.V. Method of Allocating Concentrate of Valuable Metals Contained in Heavy Oils and Products of Their Processing. RU Patent 2,631,427, 22 September 2017. [Google Scholar]
- Khadzhiev, S.N.; Shpirt, M.Y. Trace Elements in Crude Oils and Their Refining Products; Nauka: Moscow, Russia, 2012. (In Russian) [Google Scholar]
- Khuhawar, M.Y.; Aslam Mirza, M.; Jahangir, T.M. Determination of Metal Ions in Crude Oils. In Crude Oil Emulsions-Composition Stability and Characterization; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Lebedev, Y.A.; Averin, K.A.; Borisov, R.S.; Garifullin, A.R.; Bobkova, E.S.; Kurkin, T.S. Microwave Discharge in Liquid Hydrocarbons: Study of a Liquid Hydrocarbon after Exciting the Discharge. High Energy Chem. 2018, 52, 324–329. [Google Scholar] [CrossRef]
- Averin, K.A.; Borisov, R.S.; Lebedev, Y.A. Microwave Discharge in Liquid Hydrocarbons: Study of a Liquid Hydrocarbon after Its Discharge Treatment Including Air Bubbling. High Energy Chem. 2020, 54, 210–216. [Google Scholar] [CrossRef]
- Skorobogatov, G.A.; Krylov, A.A.; Moskvin, A.L.; Povarov, V.G.; Tret’yachenko, S.A.; Khripun, V.K. Chemical Transformations of Liquid Chloroform, Hexane, and Heptane in Microwave Plasmoid Field. High Energy Chem. 2016, 50, 406–410. [Google Scholar] [CrossRef]
- Scrivens, W.A.; Tour, J.M. Synthesis of gram quantities of C60 by plasma discharge in a modified round-bottomed flask. Key parameters for yield optimization and purification. J. Org. Chem. 1992, 57, 6932–6936. [Google Scholar] [CrossRef]
- Garcia, M.A. Surface plasmons in metallic nanoparticles: Fundamentals and applications. J. Phys. D Appl. Phys. 2011, 44, 283001. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Shakhatov, V.A. Gas temperature in the microwave discharge in liquid n-heptane with argon bubbling. Eur. Phys. J. D 2019, 73, 167–173. [Google Scholar] [CrossRef]
- Ochkin, V.N. Spectroscopy of Low Temperature Plasma; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Mehl, M.; Pitz, W.J.; Westbrook, C.K.; Curran, H. Kinetic modeling of gasoline surrogate components and mixtures under engine conditions. J. Proc. Combust. Inst. 2011, 33, 193–200. [Google Scholar] [CrossRef]
- Wang, H.; Frenklach, M. A detailed kinetic modeling study of aromatics formation in laminar premixed acetylene and ethylene flames. Combust. Flame 1997, 110, 173–221. [Google Scholar] [CrossRef]
- Smirnov, B.M. Cluster plasma. Phys. Uspekhi 2000, 43, 453–491. [Google Scholar] [CrossRef]
- Khrapak, S.A.; Ratynskaia, S.V.; Zobnin, A.V.; Usachev, A.D.; Yaroshenko, V.V.; Thoma, M.H.; Kretschmer, M.; Höfner, H.; Morfill, G.E.; Petrov, O.F.; et al. Particle charge in the bulk of gas discharges. Phys. Rev. E 2005, 72, 016406. [Google Scholar] [CrossRef] [PubMed]
- Bouchoule, A. Dusty Plasmas: Physics, Chemistry and Technological Impacts in Plasma Processing; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1999. [Google Scholar]
- Fortov, V.; Ivlev, S.; Khrapak, S.; Khrapak, A.; Morfill, G. Complex (dusty) plasmas: Current status, open issues, perspectives. Phys. Rep. 2005, 421, 1–103. [Google Scholar] [CrossRef]











| Reference | [61] | [57] | [59] | [58] | [62] | [43] |
| Diluent | Steam | Argon | Nitrogen | Steam | ||
| Temperature and pressure | 760 °C, 101 kPa | 700 °C, 9.2 kPa | 640 °C, 101 kPa | 810 °C, 400 kPa | 760 °C, 101 kPa | ~1700 °C, 101 kPa |
| Conversion, % | 52.51 | 12 | 15 | 87.8 | 64.3 | |
| Component | ||||||
| hydrogen | 2.08 | 0.11 | 0.059 | 1.6 | 0.34 | 7.13 |
| methane | 5.39 | 0.91 | 1.11 | 7.1 | 5.41 | 4.02 |
| ethane | 1.24 | 0.62 | 1.69 | 2.1 | 1.52 | 0.489 |
| ethylene | 23.1 | 4.25 | 4.02 | 47.8 | 32.0 | 12.45 |
| acetylene | 0.6 | 28.39 | ||||
| propane | 0.39 | 0.053 | 0.13 | 0.5 | 0.38 | 0.052 |
| propylene | 7.47 | 2.24 | 2.98 | 17.3 | 13.3 | 0.779 |
| methylacetylene | 0.3 | 0.00 | 0.247 | |||
| allene | 0.1 | 0.00 | 0.067 | |||
| n-butane | 0.070 | 0.12 | tr. | 0.08 | 0.0031 | |
| isobutane | 0.2 | 0.00 | 0.000 | |||
| 1-butene | 4.84 | 1.83 | 2.26 | 4.6 | 6.28 | 0.105 |
| trans-2-butene | 0.11 | 0.5 | 0.26 | 0.0024 | ||
| cis-2-butene | 0.10 | 0.0019 | ||||
| isobutene | 0.1 | 0.00 | 0.0010 | |||
| 1,3-butadiene | 1.17 | 0.13 | 4.3 | 2.02 | 0.106 | |
| vinylacetylene | 0.207 | |||||
| 1-butyne (ethylacetylene) | 0.0068 | |||||
| 2-butyne (dimethylacetylene) | 0.0007 | |||||
| diacetylene | 0.354 | |||||
| n-pentane | 0.0074 | |||||
| isopentane | 0.042 | 0.05 | 0.0038 | |||
| neopentane | 0.000 | |||||
| cyclopentane | 0.0018 | |||||
| 1-pentene | 1.91 | 1.27 | 1.69 | 1.72 | 0.0193 | |
| trans-2-pentene | 0.070 | 0.09 | 0.0006 | |||
| cis-2-pentene | 0.10 | 0.0004 | ||||
| 2-methyl-1-butene | 0.0002 | |||||
| 2-methyl-2-butene | 0.0012 | |||||
| 3-methyl-1-butene | 0.0007 | |||||
| cyclopentene | 0.000 | |||||
| isoprene | 0.000 | |||||
| cyclopentadiene | 0.3 | 0.012 | ||||
| n-hexane | 0.0017 | |||||
| cyclohexane | ||||||
| methylcyclopentane | ||||||
| cyclohexene | 0.0003 | |||||
| 1-methylcyclopentene | 0.00 | |||||
| 1-hexene | 0.82 | 0.71 | 0.81 | 0.55 | 0.00 | |
| trans-2-hexene | 0.0001 | |||||
| cis-2-hexene | ||||||
| benzene | 0.4 | 0.020 | ||||
| other NA C5–C6 | 3.7 | |||||
| n-heptane | 47.5 | 88.0 | 85.0 | 12.2 | 35.7 | 45.1 |
| toluene | 0.1 | 0.0005 | ||||
| ethylbenzene | traces | |||||
| xylenes | traces | |||||
| styrene | traces | |||||
| naphthalene | traces | |||||
| other C7–C12 | traces | 0.390 | ||||
| C12+ | traces | |||||
| CO | 3.57 | 0.24 | ||||
| CO2 | 0.06 | |||||
| SUMMA | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Hydrocarbon, Boiling Temperature and Structure of Molecule | H2 | CH4 | C2H4 | C2H2 | Ref. |
|---|---|---|---|---|---|
n-Heptane C7H16 (Tboil = 98.2 °C)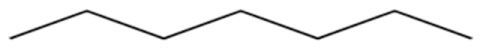 | (71.2) 1 | 0.042 | 0.11 | 0.25 | [44] |
| n-Heptane + Ar | [43] | ||||
| 0 | 1 | 0.059 | 0.18 | 0.27 | |
| 6.8 L/h | 1 | 0.049 | 0.23 | 0.31 | |
| 17.3 | 1 | 0.037 | 0.26 | 0.34 | |
| 36.8 | 1 | 0.040 | 0.18 | 0.28 | |
n-Octane C8H18 (Tboil = 125.52 °C) | (72) 1 | 0.03 | 0.104 | 0.25 | [44] |
Decane C10H22 ((Tboil = 174.1 °C) | (71.7) 1 | 0.024 | 0.088 | 0.28 | [44] |
Dodecane C12H26 (Tboil = 216.2 °C) | (74) 1 | 0.027 | 0.027 | 0.27 | [28] |
Pentadecane C15H32 (Tboil = 270.6 °C) | (67.6) 1 | 0.019 | 0.09 | 0.37 | [44] |
Hexadecane C16H34 (Tboil = 286.79 °C) | (65.6) 1 | 0.015 | 0.083 | 0.43 | [44] |
Isooctane C8H18 (Tboil = 99.3 °C) | (71) 1 | 0.087 | 0.057 | 0.26 | [44] |
Cyclohexane C6H12 (Tboil = 80.74 °C) | (73.4) 1 | 0 | 0.12 | 0.24 | [44] |
Benzene C6H6 (Tboil = 80.1 °C) | (88.80) 1 | 0 | 0 | 0.13 | [44] |
Toluene C6H5-CH3 (Tboil = 110.6 °C)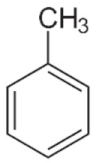 | (86.1) 1 | 0.021 | 0 | 0.14 | [44] |
Ortho-xylole C6H5-(CH3)2 (Tboil = 144 °C) | (74.6) 1 | 0.048 | 0.075 | 0.19 | [44] |
| Petroleum solvent “Nefras S2 80/120” (mixture of light hydrocarbons with boiling temperature range of 33–205 °C) | (66.5) 1 | 0.09 | 0.13 | 0.3 | [44] |
| Metals | Content of Metals (Weight %) | |||
|---|---|---|---|---|
| Source Crude Oil [72] | Tree-Like Structure on Antenna [72] | Source Heavy Petroleum Feedstock [71] | Tree-Like Structure on Antenna [71] | |
| Al | 0.00028 | 0.0028 | 0.0019 | 0.0068 |
| Co | 0.000053 | 0.00012 | 0.000047 | 0.0006 |
| Cu | 0.0002 | 0.0014 | 0.00065 | 0.0025 |
| Fe | 0.00064 | 0.0032 | 0.0019 | 0.034 |
| Mo | 0.00032 | 0.00067 | 0.067 | 0.98 |
| Ni | 0.008 | 0.013 | 0.0049 | 0.018 |
| V | 0.057 | 0.089 | 0.019 | 0.071 |
| Zn | ≤0.000002 | 0.00028 | <0.000002 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lebedev, Y.A. Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization. Polymers 2021, 13, 1678. https://doi.org/10.3390/polym13111678
Lebedev YA. Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization. Polymers. 2021; 13(11):1678. https://doi.org/10.3390/polym13111678
Chicago/Turabian StyleLebedev, Yuri A. 2021. "Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization" Polymers 13, no. 11: 1678. https://doi.org/10.3390/polym13111678
APA StyleLebedev, Y. A. (2021). Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization. Polymers, 13(11), 1678. https://doi.org/10.3390/polym13111678







